Application of Different Coagulants and Cost Evaluation for the Treatment of Oil and Gas Produced Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Coagulation Experiments and Analytical Methods
3. Results and Discussions
3.1. Characterization of PW
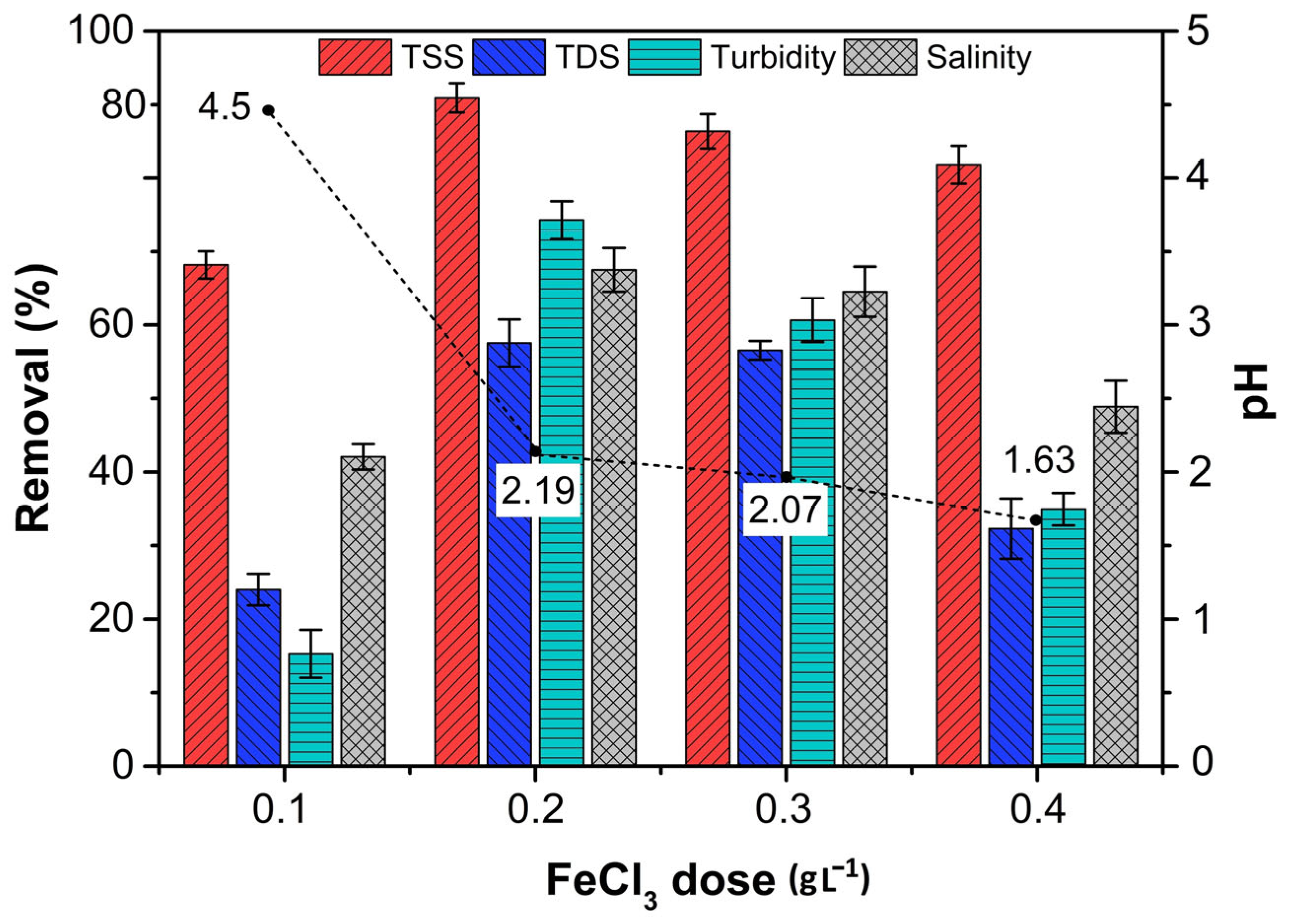
3.2. Treatment of PW Using Ferric Chloride
3.3. Treatment of PW Using Sodium Sulfide
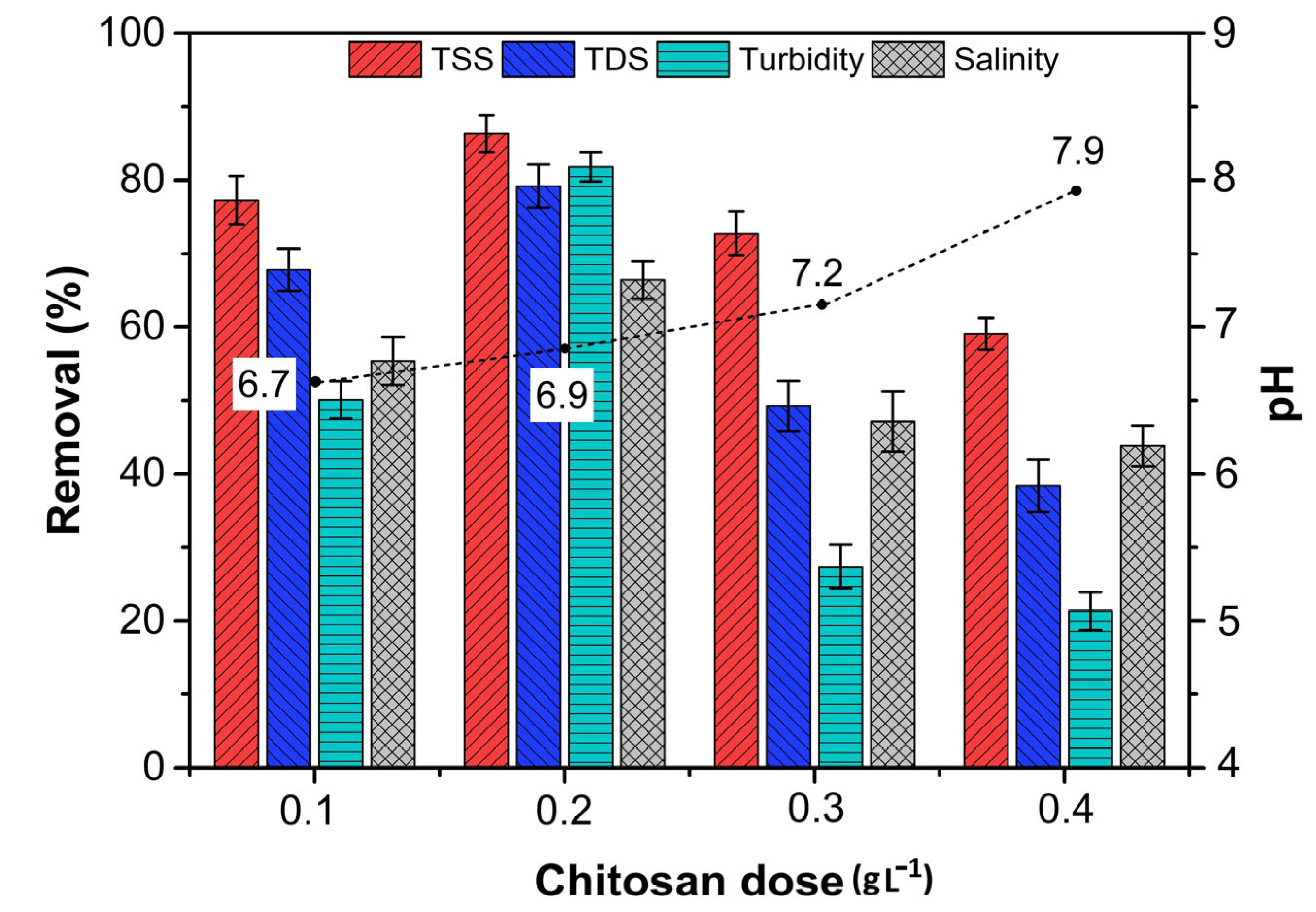
3.4. Treatment of PW Using Chitosan
3.5. Treatment of PW Using Aluminum Potassium Sulphate
3.6. Treatment of PW Using Magnesium Oxide
3.7. Cost Comparison of Different Coagulants
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, S.; Huang, G.; Cheng, G.; Wang, Y.; Fu, H. Hardness, COD and turbidity removals from produced water by electrocoagulation pretreatment prior to Reverse Osmosis membranes. Desalination 2014, 344, 454–462. [Google Scholar] [CrossRef]
- Bakke, T.; Klungsøyr, J.; Sanni, S. Environmental impacts of produced water and drilling waste discharges from the Norwegian offshore petroleum industry. Mar. Environ. Res. 2013, 92, 154–169. [Google Scholar] [CrossRef] [Green Version]
- Dickhout, J.; Moreno, J.; Biesheuvel, P.; Boels, L.; Lammertink, R.; de Vos, W. Produced water treatment by membranes: A review from a colloidal perspective. J. Colloid Interface Sci. 2017, 487, 523–534. [Google Scholar] [CrossRef]
- Nadersha, S.; Hassan, A.A. Biodesalination and treatment of raw hypersaline produced water samples using indigenous wastewater algal consortia. Desalination 2022, 528, 115638. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Yue, P.L. Separation of pollutants from restaurant wastewater by electrocoagulation. Sep. Purif. Technol. 2000, 19, 65–76. [Google Scholar] [CrossRef]
- Neff, J.M. Bioaccumulation in Marine Organisms: Effect of Contaminants from Oil Well Produced Water; Elsevier: Amsterdam, The Netherlands, 2002. [Google Scholar]
- Ahmad, F.; Morris, K.; Law, G.T.; Taylor, K.G.; Shaw, S. Fate of radium on the discharge of oil and gas produced water to the marine environment. Chemosphere 2021, 273, 129550. [Google Scholar] [CrossRef]
- Isehunwa, S.O.; Onovae, S. Evaluation of Produced Water Discharge in the Niger-Delta. J. Eng. Appl. Sci. 2011, 6, 66–72. [Google Scholar]
- D’Unger, C.; Chapman, D.; Carr, R.S. Discharge of oilfield-produced water in nueces bay, texas: A case study. Environ. Manag. 1996, 20, 143–150. [Google Scholar] [CrossRef]
- Mathieu, A.; Hanlon, J.; Myers, M.; Melvin, W.; French, B.; DeBlois, E.M.; King, T.; Lee, K.; Williams, U.P.; Wight, F.M.; et al. Studies on Fish Health Around the Terra Nova Oil Development Site on the Grand Banks Before and After Discharge of Produced Water. In Produced Water; Springer: New York, NY, USA, 2011; pp. 375–399. [Google Scholar] [CrossRef]
- Schifter, I.; González-Macías, C.; Salazar-Coria, L.; Sánchez-Reyna, G.; González-Lozano, C. Ecological and human risk assessment of long-term produced water discharge to the ocean at the Sonda de Campeche, Gulf of México. Environ. Earth Sci. 2015, 74, 5813–5826. [Google Scholar] [CrossRef]
- Manfra, L.; Moltedo, G.; Lamberti, C.V.; Maggi, C.; Finoia, M.G.; Giuliani, S.; Onorati, F.; Gabellini, M.; Di Mento, R.; Cicero, A.M. Metal Content and Toxicity of Produced Formation Water (PFW): Study of the Possible Effects of the Discharge on Marine Environment. Arch. Environ. Contam. Toxicol. 2007, 53, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, S.; Andreozzi, M.; Micó, M.M.; Alvarez, M.G.; Contreras, S. Produced water treatment by advanced oxidation processes. Sci. Total. Environ. 2019, 666, 12–21. [Google Scholar] [CrossRef]
- Xu, P.; Drewes, J.E.; Heil, D. Beneficial use of co-produced water through membrane treatment: Technical-economic assessment. Desalination 2008, 225, 139–155. [Google Scholar] [CrossRef]
- Kumar, M.; Adham, S.S.; Pearce, W.R. Investigation of Seawater Reverse Osmosis Fouling and Its Relationship To Pretreatment Type. Environ. Sci. Technol. 2006, 40, 2037–2044. [Google Scholar] [CrossRef]
- Tzotzi, C.; Pahiadaki, T.; Yiantsios, S.; Karabelas, A.; Andritsos, N. A study of CaCO3 scale formation and inhibition in RO and NF membrane processes. J. Membr. Sci. 2007, 296, 171–184. [Google Scholar] [CrossRef]
- Bellona, C.; Drewes, J.E. The role of membrane surface charge and solute physico-chemical properties in the rejection of organic acids by NF membranes. J. Membr. Sci. 2005, 249, 227–234. [Google Scholar] [CrossRef]
- Zhu, B.; Clifford, D.A.; Chellam, S. Comparison of electrocoagulation and chemical coagulation pretreatment for enhanced virus removal using microfiltration membranes. Water Res. 2005, 39, 3098–3108. [Google Scholar] [CrossRef]
- Montaño-Medina, C.U.; Lopéz-Martínez, L.M.; Ochoa-Terán, A.; López-Maldonado, E.A.; Salazar-Gastelum, M.I.; Trujillo-Navarrete, B.; Pérez-Sicairos, S.; Cornejo-Bravo, J.M. New pyridyl and aniline-functionalized carbamoylcarboxylic acids for removal of metal ions from water by coagulation-flocculation process. Chem. Eng. J. 2023, 451, 138396. [Google Scholar] [CrossRef]
- Kumar, V.; Othman, N.; Asharuddin, S. Applications of Natural Coagulants to Treat Wastewater—A Review. MATEC Web Conf. 2017, 103, 06016. [Google Scholar] [CrossRef] [Green Version]
- Garbutt, C.F. Water Treatment Process for Reducing the Hardness of an Oilfield Produced Water. U.S. Patent 5879562, 9 March 1999. [Google Scholar]
- Zhou, F.S.; Zhao, M.F.; Ni, W.X.; Dang, Y.S.; Pu, C.S.; Lu, F.J. Inorganic polymeric flocculent FMA for purifying oilfield produced water: Preparation and uses. Oilfield Chem. 2000, 17, 256–259. [Google Scholar]
- Houcine, M. Solution for heavy metals decontamination in produced water/case study in southern Tunisia. In Proceedings of the SPE International Conference on Health, Safety and Environment in Oil and Gas Exploration and Production, Kuala Lumpur, Malaysia, 20–22 March 2002. [Google Scholar]
- Frankiewicz, T.C.; Gerlach, J. Union Oil Co of California. Removal of Hydrocarbons, Mercury, and Arsenic from Oil-Field Produced Water. U.S. Patent 6117333, 2000. [Google Scholar]
- Gao, J.-M.; Wang, B.; Li, W.; Cui, L.; Guo, Y.; Cheng, F. High-efficiency leaching of Al and Fe from fly ash for preparation of polymeric aluminum ferric chloride sulfate coagulant for wastewater treatment. Sep. Purif. Technol. 2023, 306, 122545. [Google Scholar] [CrossRef]
- DePaolis, M.; De Respino, S.; Samineni, L.; Brighton, S.; Kumar, M. Cottonseed extract as a coagulant for water treatment. Environ. Sci. Adv. 2022. [Google Scholar] [CrossRef]
- Altaher, H.; ElQada, E.; Omar, W. Pretreatment of Wastewater Streams from Petroleum/Petrochemical Industries Using Coagulation. Adv. Chem. Eng. Sci. 2011, 01, 245–251. [Google Scholar] [CrossRef] [Green Version]
- Otálora, M.C.; Wilches-Torres, A.; Lara, C.R.; Castaño, J.A.G.; Cifuentes, G.R. Evaluation of Turbidity and Color Removal in Water Treatment: A Comparative Study between Opuntia ficus-indica Fruit Peel Mucilage and FeCl3. Polymers 2022, 15, 217. [Google Scholar] [CrossRef] [PubMed]
- Bottero, J.Y.; Manceau, A.; Villieras, F.; Tchoubar, D. Structure and mechanisms of formation of iron oxide hy-droxide (chloride) polymers. Langmuir 1994, 10, 316–319. [Google Scholar] [CrossRef]
- Siéliéchi, J.-M.; Lartiges, B.; Kayem, G.; Hupont, S.; Frochot, C.; Thieme, J.; Ghanbaja, J.; de la Caillerie, J.D.; Barrès, O.; Kamga, R.; et al. Changes in humic acid conformation during coagulation with ferric chloride: Implications for drinking water treatment. Water Res. 2008, 42, 2111–2123. [Google Scholar] [CrossRef]
- Duan, J.; Gregory, J. Coagulation by hydrolysing metal salts. Adv. Colloid Interface Sci. 2003, 100, 475–502. [Google Scholar] [CrossRef]
- Liang, Z.; Wang, Y.; Zhou, Y.; Liu, H. Coagulation removal of melanoidins from biologically treated molasses wastewater using ferric chloride. Chem. Eng. J. 2009, 152, 88–94. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, W.; Wei, D.; Wang, W.; Cui, B.; Liu, W. Effect of copper ions on the flotation separation of chalco-pyrite and molybdenite using sodium sulfide as a depressant. Miner. Eng. 2018, 115, 44–52. [Google Scholar] [CrossRef]
- Trahar, W. A laboratory study of the influence of sodium sulphide and oxygen on the collectorless flotation of chalcopyrite. Int. J. Miner. Process. 1983, 11, 57–74. [Google Scholar] [CrossRef]
- Luttrell, G.H.; Yoon, R.-H. The collectorless flotation of chalcopyrite ores using sodium sulfide. Int. J. Miner. Process. 1984, 13, 271–283. [Google Scholar] [CrossRef]
- Pavez, O.; Peres, A.E.C. Effect of sodium metasilicate and sodium sulphide on the floatability of mona-zite-zircon-rutile with oleate and hydroxamates. Miner. Eng. 1993, 6, 69–78. [Google Scholar] [CrossRef]
- Herrera-Urbina, R.; Sotillo, F.; Fuerstenau, D. Effect of sodium sulfide additions on the pulp potential and amyl xanthate flotation of cerussite and galena. Int. J. Miner. Process. 1999, 55, 157–170. [Google Scholar] [CrossRef]
- Wang, K.-J.; Wang, P.-S.; Nguyen, H.-P. A data-driven optimization model for coagulant dosage decision in industrial wastewater treatment. Comput. Chem. Eng. 2021, 152, 107383. [Google Scholar] [CrossRef]
- Bobadilla, M.C.; Lorza, R.L.; García, R.E.; Gómez, F.S.; González, E.P.V. Coagulation: Determination of Key Operating Parameters by Multi-Response Surface Methodology Using Desirability Functions. Water 2019, 11, 398. [Google Scholar] [CrossRef] [Green Version]
- Zainuddin, N.A.; Mamat, T.A.R.; Maarof, H.I.; Puasa, S.W.; Yatim, S.R.M. Removal of nickel, zinc and copper from plating process industrial raw effluent via hydroxide precipitation versus sulfide precipitation. In Proceedings of the IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; Volume 551, p. 012122. [Google Scholar]
- Lochyński, P.; Wiercik, P.; Charazińska, S.; Ostrowski, M. Research on neutralization of wastewater from pickling and electropolishing processes. Arch. Environ. Prot. 2021, 47, 18–29. [Google Scholar]
- Blais, J.F.; Djedidi, Z.; Ben Cheikh, R.; Tyagi, R.D.; Mercier, G. Metals Precipitation from Effluents: Review. Pr. Period. Hazardous, Toxic, Radioact. Waste Manag. 2008, 12, 135–149. [Google Scholar] [CrossRef]
- Renault, F.; Sancey, B.; Badot, P.-M.; Crini, G. Chitosan for coagulation/flocculation processes—An eco-friendly approach. Eur. Polym. J. 2009, 45, 1337–1348. [Google Scholar] [CrossRef]
- Roberts, G.A.; Roberts, G.A. Chitin Chemistry; Macmillan: London, UK, 1992; pp. 167–172. [Google Scholar]
- Castro-Riquelme, C.L.; López-Maldonado, E.A.; Ochoa-Terán, A.; Alcántar-Zavala, E.; Trujillo-Navarrete, B.; Pérez-Sicairos, S.; Miranda-Soto, V.; Zizumbo-López, A. Chitosan-carbamoylcarboxylic acid grafted polymers for removal of metal ions in wastewater. Chem. Eng. J. 2023, 456, 141034. [Google Scholar] [CrossRef]
- Bolto, B.; Gregory, J. Organic polyelectrolytes in water treatment. Water Res. 2007, 41, 2301–2324. [Google Scholar] [CrossRef]
- Huang, C.; Chen, Y. Coagulation of colloidal particles in water by chitosan. J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol. 1996, 66, 227–232. [Google Scholar] [CrossRef]
- Ahmad, A.; Sumathi, S.; Hameed, B. Coagulation of residue oil and suspended solid in palm oil mill effluent by chitosan, alum and PAC. Chem. Eng. J. 2006, 118, 99–105. [Google Scholar] [CrossRef]
- Yukselen, M.A.; Gregory, J. The effect of rapid mixing on the break-up and re-formation of flocs. J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol. 2004, 79, 782–788. [Google Scholar] [CrossRef]
- Jiang, J.-Q. The role of coagulation in water treatment. Curr. Opin. Chem. Eng. 2015, 8, 36–44. [Google Scholar] [CrossRef]
- Roques, H. Chemical Water Treatment: Principles and Practice; VCH Publishers, Inc.: New York, NY, USA, 1996; p. 620. [Google Scholar]
- Zeng, Y.; Yang, C.; Pu, W.; Zhang, X. Removal of silica from heavy oil wastewater to be reused in a boiler by combining magnesium and zinc compounds with coagulation. Desalination 2007, 216, 147–159. [Google Scholar] [CrossRef]
- Bolto, B.; Dixon, D.; Eldridge, R.; King, S. Cationic polymer and clay or metal oxide combinations for natural organic matter removal. Water Res. 2001, 35, 2669–2676. [Google Scholar] [CrossRef]



| Location | Impact of PW Discharge | Reference |
|---|---|---|
| United Kingdom | Elevated level of radium (226Ra) up to 97% was detected in marine sediment samples at sites downstream of the point of PW discharge. | [7] |
| Nigeria | Even after treatment, the PW discharged in the Niger Delta exceeded the discharge limits for the oil and grease content (60 ± 20), TDS (6200 ± 700), and chloride ions (4100 ± 500). | [8] |
| Texas | PW discharged in Nueces Bay made the sediments in the vicinity of the discharge toxic, with EC50 values ranging between 7.3 and 24.6%. | [9] |
| Canada | After the release of PW in the Grand Banks, selected health bioindicators of fish (cell count, visible skin and organ lesions, cellular alteration, etc.) were absent or similar to the reference site. | [10] |
| Mexico | Long-term PW discharge into the ocean at the Gulf of Mexico could cause an increase in ecological and human risks over time and space due to high concentrations of PAHs (up to 2324 ng/g). | [11] |
| Norway | PW discharge may have toxic effects on organisms close to the outlet and severe cumulative effects. | [2] |
| Italy | PW discharged in Adriatic Sea led to the toxicity of water (EC50 value of 20.73% for P. lividus); however, no significant effect was observed on marine sediment. | [12] |
| S. No. | Parameter | PW Composition |
|---|---|---|
| 1 | pH | 7.8 |
| 2 | Electrical conductivity (mS cm−1) | 151.2 |
| 3 | Salinity (parts per thousand) | 90.8 |
| 4 | TDS (g L−1) | 80.8 |
| 5 | Turbidity (NTU) | 6.6 |
| 6 | TSS (mg L−1) | 22 |
| Coagulants | |||||
|---|---|---|---|---|---|
| FeCl3 | Na2S | Chitosan | AlKSO4 | MgO | |
| Coagulant dose used (mg L−1) | 200 | 400 | 200 | 100 | 200 |
| Coagulant dose used (kg 10−3 m−3) | 200 | 400 | 200 | 100 | 200 |
| Commercial cost of coagulants (USD kg−1) | 0.02 | 0.09 | 0.2 | 0.19 | 0.6 |
| Cost of treatment (USD m−3) | 4 | 36 | 40 | 19 | 120 |
| Removal efficiency (%) * | 74.25 | 69.5 | 82.5 | 73.5 | 84.5 |
| Treatment cost efficiency (USD m−3 %−1) ** | 5.38(1) | 51.7(4) | 48.4(3) | 25.8(2) | 142.0 (5) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, N.; Rajput, H.; Aly Hassan, A.; Kumar, S. Application of Different Coagulants and Cost Evaluation for the Treatment of Oil and Gas Produced Water. Water 2023, 15, 464. https://doi.org/10.3390/w15030464
Das N, Rajput H, Aly Hassan A, Kumar S. Application of Different Coagulants and Cost Evaluation for the Treatment of Oil and Gas Produced Water. Water. 2023; 15(3):464. https://doi.org/10.3390/w15030464
Chicago/Turabian StyleDas, Namita, Himadri Rajput, Ashraf Aly Hassan, and Sandeep Kumar. 2023. "Application of Different Coagulants and Cost Evaluation for the Treatment of Oil and Gas Produced Water" Water 15, no. 3: 464. https://doi.org/10.3390/w15030464