The Effect of Short-Term Upwelling Events on Fish Assemblages at the South-Eastern Coast of the Baltic Sea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Upwelling Statistics
2.3. Coastal Fish Communities Monitoring Data
2.4. Fish Community-Based Indices for Marine Environment Status Assessment
2.5. Data Analysis
3. Results
3.1. Fish Catches
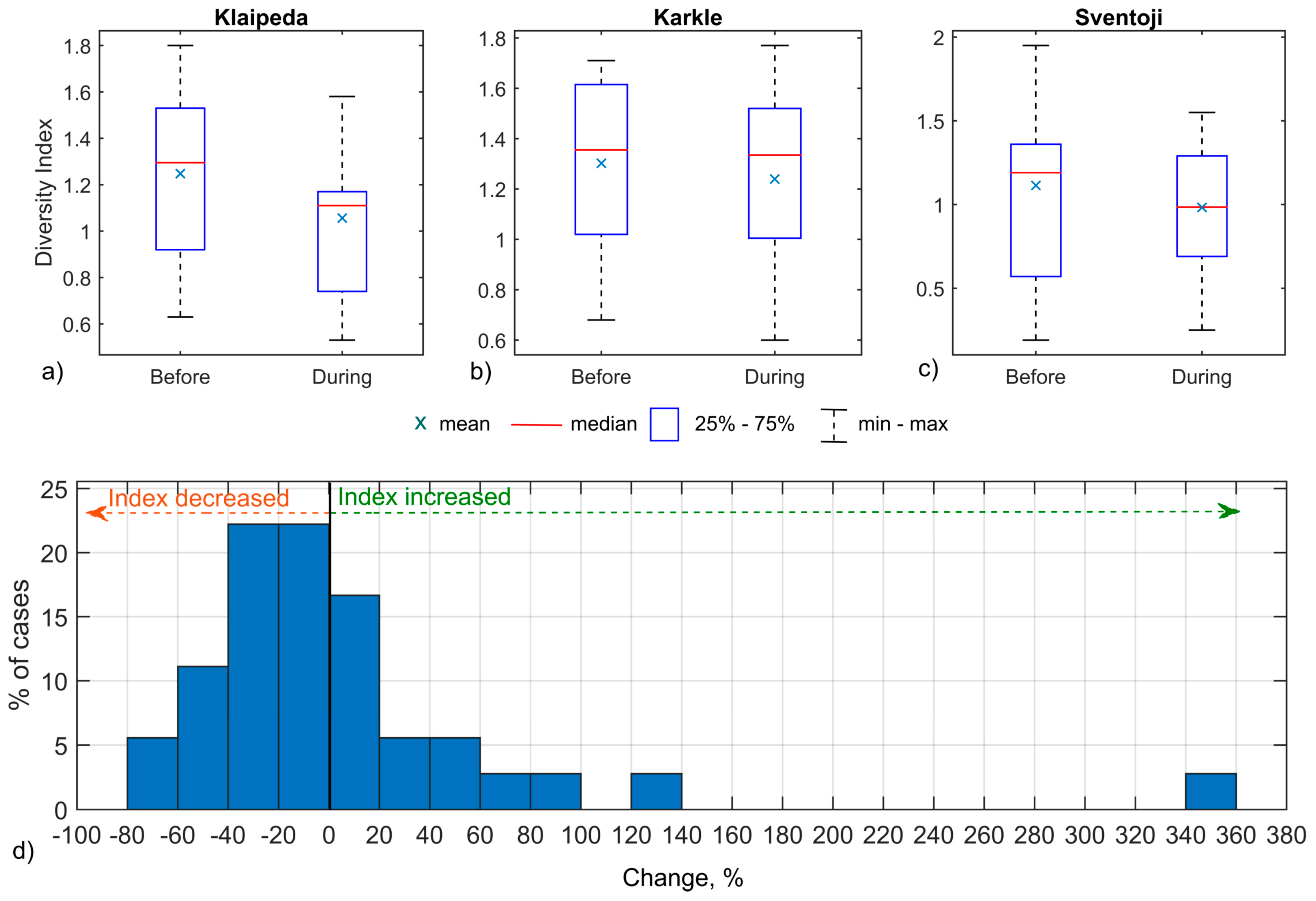
3.2. Upwelling Influence on Fish Community-Based Ecological Indices
4. Discussion
4.1. Fish Community Response to Short-Term Upwellings
4.2. Upwelling Influence on Fish Community-Based Ecological Indices
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Capotondi, A.; Jacox, M.; Bowler, C.; Kavanaugh, M.; Lehodey, P.; Barrie, D.; Brodie, S.; Chaffron, S.; Cheng, W.; Dias, D.F.; et al. Observational Needs Supporting Marine Ecosystems Modeling and Forecasting: From the Global Ocean to Regional and Coastal Systems. Front. Mar. Sci. 2019, 6, 623. [Google Scholar] [CrossRef] [Green Version]
- UNEP. Marine and Coastal Ecosystems and Human Wellbeing: A Synthesis Report Based on the Findings of the Millennium Ecosystem Assessment; UNEP: Nairobi, Kenya, 2006; 76p. [Google Scholar]
- Kumaran, M. Our Fisheries Resources and the Role of Upwelling in Their Fluctuations Part III The Role of Upwelling on Fluctuations. Seaf. Export J. 1978, 10, 21–35. [Google Scholar]
- Neokye, E.O.; Dossou, S.; Iniga, M.; Alabi-Doku, B.N. The Role of Oceanic Environmental Conditions on Catch of Sardinella spp. in Ghana. Reg. Stud. Mar. Sci. 2021, 44, 101768. [Google Scholar] [CrossRef]
- Zuloaga, R.; Varas, O.; Ahrendt, C.; Pulgar, V.M.; Valdés, J.A.; Molina, A.; Duarte, C.; Urzúa, Á.; Guzmán-Rivas, F.; Aldana, M.; et al. Revealing Coastal Upwelling Impact on the Muscle Growth of an Intertidal Fish. Sci. Total. Environ. 2023, 858, 159810. [Google Scholar] [CrossRef] [PubMed]
- Kampf, J.; Chapman, P. Upwelling Systems of the World; Springer International Publishing: Cham, Switzerland, 2016; 433p. [Google Scholar] [CrossRef]
- Aguirre, C.; Rojas, M.; Garreaud, R.D.; Rahn, D.A. Role of Synoptic Activity on Projected Changes in Upwelling-Favourable Winds at the Ocean’s Eastern Boundaries. NPJ Clim. Atmos. Sci. 2019, 2, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Xiu, P.; Chai, F.; Curchitser, E.N.; Castruccio, F.S. Future Changes in Coastal Upwelling Ecosystems with Global Warming: The Case of the California Current System. Sci. Rep. 2018, 8, 2866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sreeush, M.G.; Valsala, V.; Pentakota, S.; Prasad, K.V.S.R.; Murtugudde, R. Biological Production in the Indian Ocean Upwelling Zones –Part 1: Refined Estimation via the Use of a Variable Compensation Depth in Ocean Carbon Models. Biogeosciences 2018, 15, 1895–1918. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Wang, X.H. Progress on Upwelling Studies in the China Seas. Rev. Geophys. 2016, 54, 653–673. [Google Scholar] [CrossRef]
- Johns, B.; Marsaleix, P.; Estournel, C.; Véhil, R. On the Wind-Driven Coastal Upwelling in the Gulf of Lions. J. Mar. Syst. 1992, 3, 309–320. [Google Scholar] [CrossRef]
- Mamoutos, I.; Zervakis, V.; Tragou, E.; Karydis, M.; Frangoulis, C.; Kolovoyiannis, V.; Georgopoulos, D.; Psarra, S. The Role of Wind-Forced Coastal Upwelling on the Thermohaline Functioning of the North Aegean Sea. Cont. Shelf Res. 2017, 149, 52–68. [Google Scholar] [CrossRef]
- Mikhailova, E.N.; Ivanov, V.A.; Kosnyrev, V.R. Upwelling in the North-Western Black Sea during the Period of Summer-Time Warming. Phys. Oceanogr. 1997, 8, 243–251. [Google Scholar] [CrossRef]
- Silvestrova, K.P.; Zatsepin, A.G.; Myslenkov, S.A. Coastal Upwelling in the Gelendzhik Area of the Black Sea: Effect of Wind and Dynamics. Oceanology 2017, 57, 469–477. [Google Scholar] [CrossRef]
- Gidhagen, L. Coastal Upwelling in the Baltic Sea—Satellite and in Situ Measurements of Sea-Surface Temperatures Indicating Coastal Upwelling. Estuar. Coast. Shelf Sci. 1987, 24, 449–462. [Google Scholar] [CrossRef]
- Lehmann, A.; Myrberg, K. Upwelling in the Baltic Sea—A Review. J. Mar. Syst. 2008, 74, S3–S12. [Google Scholar] [CrossRef]
- Dabuleviciene, T.; Kozlov, I.E.; Vaiciute, D.; Dailidiene, I. Remote Sensing of Coastal Upwelling in the South-Eastern Baltic Sea: Statistical Properties and Implications for the Coastal Environment. Remote Sens. 2018, 10, 1752. [Google Scholar] [CrossRef] [Green Version]
- Dabuleviciene, T.; Vaiciute, D.; Kozlov, I.E. Chlorophyll-a Variability during Upwelling Events in the South-Eastern Baltic Sea and in the Curonian Lagoon from Satellite Observations. Remote Sens. 2020, 12, 3661. [Google Scholar] [CrossRef]
- Lehmann, A.; Myrberg, K.; Hoflich, K. A Statistical Approach to Coastal Upwelling in the Baltic Sea Based on the Analysis of Satellite Data for 1990–2009. Oceanologia 2012, 54, 369–393. [Google Scholar] [CrossRef] [Green Version]
- Sproson, D.; Sahlée, E. Modelling the Impact of Baltic Sea Upwelling on the Atmospheric Boundary Layer. Tellus A Dyn. Meteorol. Oceanogr. 2014, 66, 24041. [Google Scholar] [CrossRef]
- Laanemets, J.; Kononen, K.; Pavelson, J.; Poutanen, E.-L. Vertical Location of Seasonal Nutriclines in the Western Gulf of Finland. J. Mar. Syst. 2004, 52, 1–13. [Google Scholar] [CrossRef]
- Lips, I.; Lips, U. Phytoplankton Dynamics Affected by the Coastal Upwelling Events in the Gulf of Finland in July–August 2006. J. Plankton Res 2010, 32, 1269–1282. [Google Scholar] [CrossRef]
- Vahtera, E.; Laanemets, J.; Pavelson, J.; Huttunen, M.; Kononen, K. Effect of Upwelling on the Pelagic Environment and Bloom-Forming Cyanobacteria in the Western Gulf of Finland, Baltic Sea. J. Mar. Syst. 2005, 58, 67–82. [Google Scholar] [CrossRef]
- Kuvaldina, N.; Lips, I.; Lips, U.; Liblik, T. The Influence of a Coastal Upwelling Event on Chlorophyll a and Nutrient Dynamics in the Surface Layer of the Gulf of Finland, Baltic Sea. Hydrobiologia 2010, 639, 221–230. [Google Scholar] [CrossRef]
- Oey, L.-Y.; Wang, J.; Lee, M.-A. Fish Catch Is Related to the Fluctuations of a Western Boundary Current. J. Phys. Oceanogr. 2018, 48, 705–721. [Google Scholar] [CrossRef]
- Steinberg, C.E.W. Stress Ecology: Environmental Stress as Ecological Driving Force and Key Player in Evolution; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012; ISBN 978-94-007-2072-5. [Google Scholar]
- Donaldson, M.R.; Cooke, S.J.; Patterson, D.A.; Macdonald, J.S. Cold Shock and Fish. J. Fish Biol. 2008, 73, 1491–1530. [Google Scholar] [CrossRef]
- Volkoff, H.; Rønnestad, I. Effects of Temperature on Feeding and Digestive Processes in Fish. Temperature 2020, 7, 307–320. [Google Scholar] [CrossRef]
- Tiedemann, M.; Fock, H.O.; Brehmer, P.; Doering, J.; Moellmann, C. Does Upwelling Intensity Determine Larval Fish Habitats in Upwelling Ecosystems? The Case of Senegal and Mauritania. Fish. Oceanogr. 2017, 26, 655–667. [Google Scholar] [CrossRef] [Green Version]
- ICES. Baltic Sea Ecoregion—Fisheries Overview. In Report of the ICES Advisory Committee, 2018. ICES Adv. 2018, 13. [Google Scholar] [CrossRef]
- HELCOM. HELCOM Thematic Assessment of Biodiversity 2011–2016. 2018. Available online: http://www.helcom.fi/baltic-seatrends/holistic-assessments/state-of-the-baltic-sea-2018/reports-and-materials/ (accessed on 11 October 2019).
- Olsson, J. Past and Current Trends of Coastal Predatory Fish in the Baltic Sea with a Focus on Perch, Pike, and Pikeperch. Fishes 2019, 4, 7. [Google Scholar] [CrossRef] [Green Version]
- EU-COMDirective 2008/56/EC of the European Parliament and of the Council of 17 June 2008 Establishing a Framework for Community Action in the Field of Marine Environmental Policy (Marine Strategy Framework Directive). 2008. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2008:164:0019:0040:EN:PDF (accessed on 5 June 2019).
- Kuliński, K.; Pempkowiak, J. The Carbon Budget of the Baltic Sea. Biogeosciences 2011, 8, 3219–3230. [Google Scholar] [CrossRef] [Green Version]
- Liblik, T.; Lips, U. Stratification Has Strengthened in the Baltic Sea—An Analysis of 35 Years of Observational Data. Front. Earth Sci. 2019, 7, 174. [Google Scholar] [CrossRef]
- Kozlov, I.; Dailidienė, I.; Korosov, A.; Klemas, V.; Mingėlaitė, T. MODIS-Based Sea Surface Temperature of the Baltic Sea Curonian Lagoon. J. Mar. Syst. 2014, 129, 157–165. [Google Scholar] [CrossRef]
- Olenin, S.; Daunys, D. Coastal Typology Based on Benthic Biotope and Community Data: The Lithuanian Case Study. Coastline Rep. 2004, 4, 65–84. [Google Scholar]
- Vaiciute, D. Distribution Patterns of Optically Active Components and Phytoplankton in the Estuarine Plume in the South Eastern Baltic Sea. Ph.D. Thesis, Klaipeda University, Klaipeda, Lithuania, 2012; 128p. [Google Scholar]
- Myrberg, K.; Andrejev, O. Main upwelling regions in the Baltic Sea—A statistical analysis based on three-dimensional modelling. Boreal Environ. Res. 2003, 8, 97–112. [Google Scholar]
- HELCOM. Guidelines for Coastal Fish Monitoring Sampling Methods of HELCOM. 2015. Available online: http://www.helcom.fi/Documents/Action%20areas/Monitoring%20and%20assessment/Manuals%20and%20Guidelines/Guidelines%20for%20Coastal%20fish%20Monitoring%20of%20HELCOM.pdf (accessed on 16 December 2018).
- Maunder, M.N.; Punt, A.E. Standardizing Catch and Effort Data: A Review of Recent Approaches. Fish. Res. 2004, 70, 141–159. [Google Scholar] [CrossRef]
- HELCOM. Development of a Set of Core Indicators: Interim Report of the HELCOM CORESET Project. PART A. Description of the Selection Process. Baltic Sea Environment Proceedings No. 129A. 2012. Available online: https://helcom.fi/wp-content/uploads/2019/08/BSEP129A.pdf (accessed on 16 December 2018).
- Magurran, A.E. Measuring Biological Diversity; Blackwell Scientific: Oxford, UK, 2004. [Google Scholar]
- Skabeikis, A. Role of the Round Goby (Neogobius Melanostomus) in the Food Web of the South-Eastern Baltic Sea Coastal Waters. Ph.D. Thesis, Klaipeda University, Klaipeda, Lithuania, 2019; 165p. [Google Scholar]
- HELCOM. Indicator Based Assessment of Coastal Fish Community Status in the Baltic Sea 2005–2009. Baltic Sea Environment Proceedings No. 131. 2012. Available online: https://helcom.fi/wp-content/uploads/2019/08/BSEP131.pdf (accessed on 16 December 2018).
- HELCOM. Changing Communities of Baltic Coastal Fish. Executive summary: Assessment of coastal fish in the Baltic Sea. Balt. Sea Environ. Proc. 2006, 103B, 3–11. [Google Scholar]
- HELCOM. FISH-PRO II 1-2014, GES Values and Baselines for the Coastal Fish Core Indicators. Document 1-1 Agenda Item 3. 2014. Available online: https://portal.helcom.fi/meetings/HELCOM%20FISH-PRO%20II%201-2014-88/MeetingDocuments/3-2%20GES%20values%20and%20baselines%20for%20the%20coastal%20fish%20core%20indicators.pdf (accessed on 5 June 2018).
- Baba, K.; Renwick, J. Aspects of intraseasonal variability of Antarctic sea ice in austral winter related to ENSO and SAM events. J. Glaciol. 2017, 63, 838–846. [Google Scholar] [CrossRef] [Green Version]
- Zuur, A.F.; Ieno, E.N.; Smith, G.M. Analysing Ecological Data; Springer: New York, NY, USA, 2007; p. 672. [Google Scholar] [CrossRef] [Green Version]
- Oksanen, J. Vegan: Community Ecology Package. R Package Version 2. 2020. Available online: https://cran.r-project.org/package=vegan (accessed on 10 October 2022).
- Nowacki, J.; Matciak, M.; Szymelfenig, M.; Kowalewski, M. Upwelling Characteristics in the Puck Bay (the Baltic Sea). Oceanol. Hydrobiol. Stud. 2009, 38, 3–16. [Google Scholar] [CrossRef]
- Laanemets, J.; Vali, G.; Zhurbas, V.; Elken, J.; Lips, I.; Lips, U. Simulation of mesoscale structures and nutrient transport during summer upwelling events in the Gulf of Finland in 2006. Boreal Environ. Res. 2011, 16 (Suppl. SA), 15–26. [Google Scholar]
- Iles, A.C.; Gouhier, T.C.; Menge, B.A.; Stewart, J.S.; Haupt, A.J.; Lynch, M.C. Climate-driven Trends and Ecological Implications of Event-scale Upwelling in the California Current System. Glob. Chang. Biol. 2012, 18, 783–796. [Google Scholar] [CrossRef]
- Krężel, A.; Szymanek, L.; Kozłowski, Ł.; Szymelfenig, M. Influence of Coastal Upwelling on Chlorophyll a Concentration in the Surface Water along the Polish Coast of the Baltic Sea. Oceanologia 2005, 47, 433–452. [Google Scholar]
- Olsson, J.; Bergström, L.; Gårdmark, A. Abiotic Drivers of Coastal Fish Community Change during Four Decades in the Baltic Sea. ICES J. Mar. Sci. 2012, 69, 961–970. [Google Scholar] [CrossRef]
- Pihl, L.; Wennhage, H. Structure and Diversity of Fish Assemblages on Rocky and Soft Bottom Shores on the Swedish West Coast. J. Fish Biol. 2002, 61, 148–166. [Google Scholar] [CrossRef]
- Ådjers, K.; Appelberg, M.; Eschbaum, R.; Lappalainen, A.; Minde, A.; Repečka, R.; Thoresson, G. Trends in coastal fish stocks of the Baltic Sea. Boreal Environ. Res. 2006, 11, 13–25. [Google Scholar]
- Dainys, J.; Jakubavičiūtė, E.; Gorfine, H.; Pūtys, Ž.; Virbickas, T.; Jakimavičius, D.; Šarauskienė, D.; Meilutytė-Lukauskienė, D.; Povilaitis, A.; Bukantis, A.; et al. Predicted Climate Change Effects on European Perch (Perca Fluviatilis L.)—A Case Study from the Curonian Lagoon, South-Eastern Baltic. Estuar. Coast. Shelf Sci. 2019, 221, 83–89. [Google Scholar] [CrossRef]
- Bergström, L.; Bergström, U.; Olsson, J.; Carstensen, J. Coastal fish indicators response to natural and anthropogenic driverse variability at temporal and different spatial scales. Estuar. Coast. Shelf Sci. 2016, 183, 62–72. [Google Scholar] [CrossRef]
- Ojaveer, E.; Kalejs, M. The Impact of Climate Change on the Adaptation of Marine Fish in the Baltic Sea. ICES J. Mar. Sci. 2005, 62, 1492–1500. [Google Scholar] [CrossRef]
- Raid, T. The influence of hydrodynamic conditions on the spatial distribution of young fish and their prey organisms. Rapp. Pv Réun. Cons. Int. Explor. Mer. 1989, 190, 166–172. [Google Scholar]
- Krause, G.; Budeus, G.; Gerdes, D.; Schaumann, K.; Hesse, K. Frontal Systems in the German Bight and Their Physical and Biological Effects. In Marine Interfaces Ecohydrodynamics; Nihoul, J.C.J., Ed.; Elsevier Oceanography Series; Elsevier: Amsterdam, The Netherlands, 1986; Volume 42, pp. 119–140. [Google Scholar]
- Issifu, I.; Alava, J.J.; Lam, V.W.Y.; Sumaila, U.R. Impact of Ocean Warming, Overfishing and Mercury on European Fisheries: A Risk Assessment and Policy Solution Framework. Front. Mar. Sci. 2022, 8, 770805. [Google Scholar] [CrossRef]
- Nissling, A.; Johansson, U.; Jacobsson, M. Effects of Salinity and Temperature Conditions on the Reproductive Success of Turbot (Scophthalmus Maximus) in the Baltic Sea. Fish. Res. 2006, 80, 230–238. [Google Scholar] [CrossRef]
- von Westernhagen, H. Incubation of garpike eggs (Belone belone Linné) under controlled temperature and salinity conditions. J. Mar. Biol Ass. UK 1974, 54, 625–634. [Google Scholar] [CrossRef]
- Lappalainen, J.; Malinen, T. Hydroacoustics and Concurrent Experimental Trawling Reveal Extreme Annual Variation in the Density of 0+ Pikeperch in Late Summer. Fish. Res. 2022, 251, 106316. [Google Scholar] [CrossRef]
- Lappalainen, J.; Lehtonen, H.; Böhling, P.; Erm, V. Covariation in Year-Class Strength of Perch, Perca Fluviatilis L. and Pikeperch, Stizostedion Lucioperca (L.). Ann. Zool. Fenn. 1996, 33, 421–426. [Google Scholar]
- Heikinheimo, O.; Pekcan-Hekim, Z.; Raitaniemi, J. Spawning Stock–Recruitment Relationship in Pikeperch Sander Lucioperca (L.) in the Baltic Sea, with Temperature as an Environmental Effect. Fish. Res. 2014, 155, 1–9. [Google Scholar] [CrossRef]
- Kokkonen, E.; Heikinheimo, O.; Pekcan-Hekim, Z.; Vainikka, A. Effects of Water Temperature and Pikeperch (Sander Lucioperca) Abundance on the Stock–Recruitment Relationship of Eurasian Perch (Perca Fluviatilis) in the Northern Baltic Sea. Hydrobiologia 2019, 841, 79–94. [Google Scholar] [CrossRef] [Green Version]
- Hallett, C.S.; Trayler, K.M.; Valesini, F.J. The Fish Community Index: A Practical Management Tool for Monitoring and Reporting Estuarine Ecological Condition. Integr. Environ. Assess. Manag. 2019, 15, 726–738. [Google Scholar] [CrossRef] [PubMed]
- Myrberg, K.; Korpinen, S.; Uusitalo, L. Physical Oceanography Sets the Scene for the Marine Strategy Framework Directive Implementation in the Baltic Sea. Mar. Policy 2019, 107, 103591. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dabuleviciene, T.; Jucevicius, D.; Zolubas, T.; Vaiciute, D.; Nika, N. The Effect of Short-Term Upwelling Events on Fish Assemblages at the South-Eastern Coast of the Baltic Sea. Water 2023, 15, 452. https://doi.org/10.3390/w15030452
Dabuleviciene T, Jucevicius D, Zolubas T, Vaiciute D, Nika N. The Effect of Short-Term Upwelling Events on Fish Assemblages at the South-Eastern Coast of the Baltic Sea. Water. 2023; 15(3):452. https://doi.org/10.3390/w15030452
Chicago/Turabian StyleDabuleviciene, Toma, Deividas Jucevicius, Tomas Zolubas, Diana Vaiciute, and Nerijus Nika. 2023. "The Effect of Short-Term Upwelling Events on Fish Assemblages at the South-Eastern Coast of the Baltic Sea" Water 15, no. 3: 452. https://doi.org/10.3390/w15030452







