Porous Biochar Materials for Sustainable Water Treatment: Synthesis, Modification, and Application
Abstract
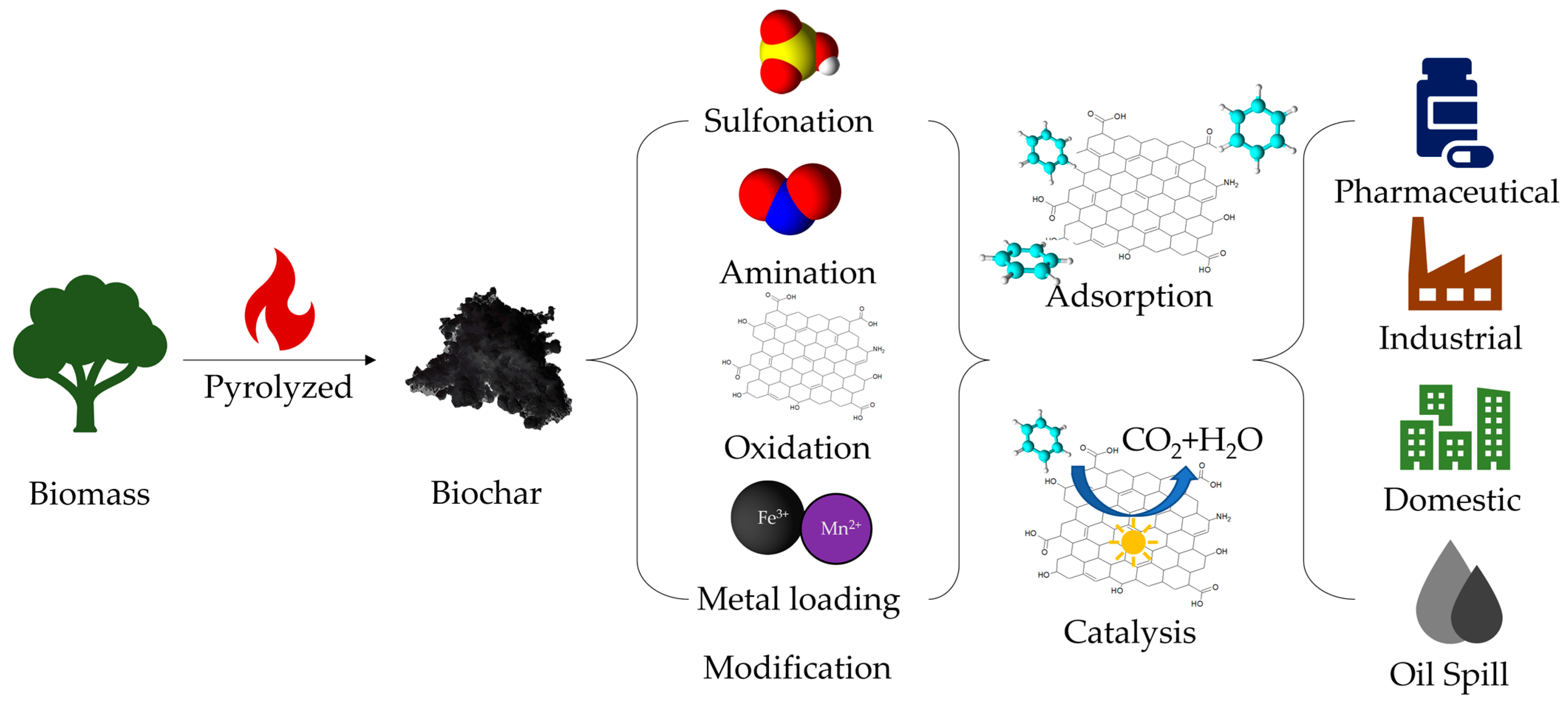
:1. Introduction
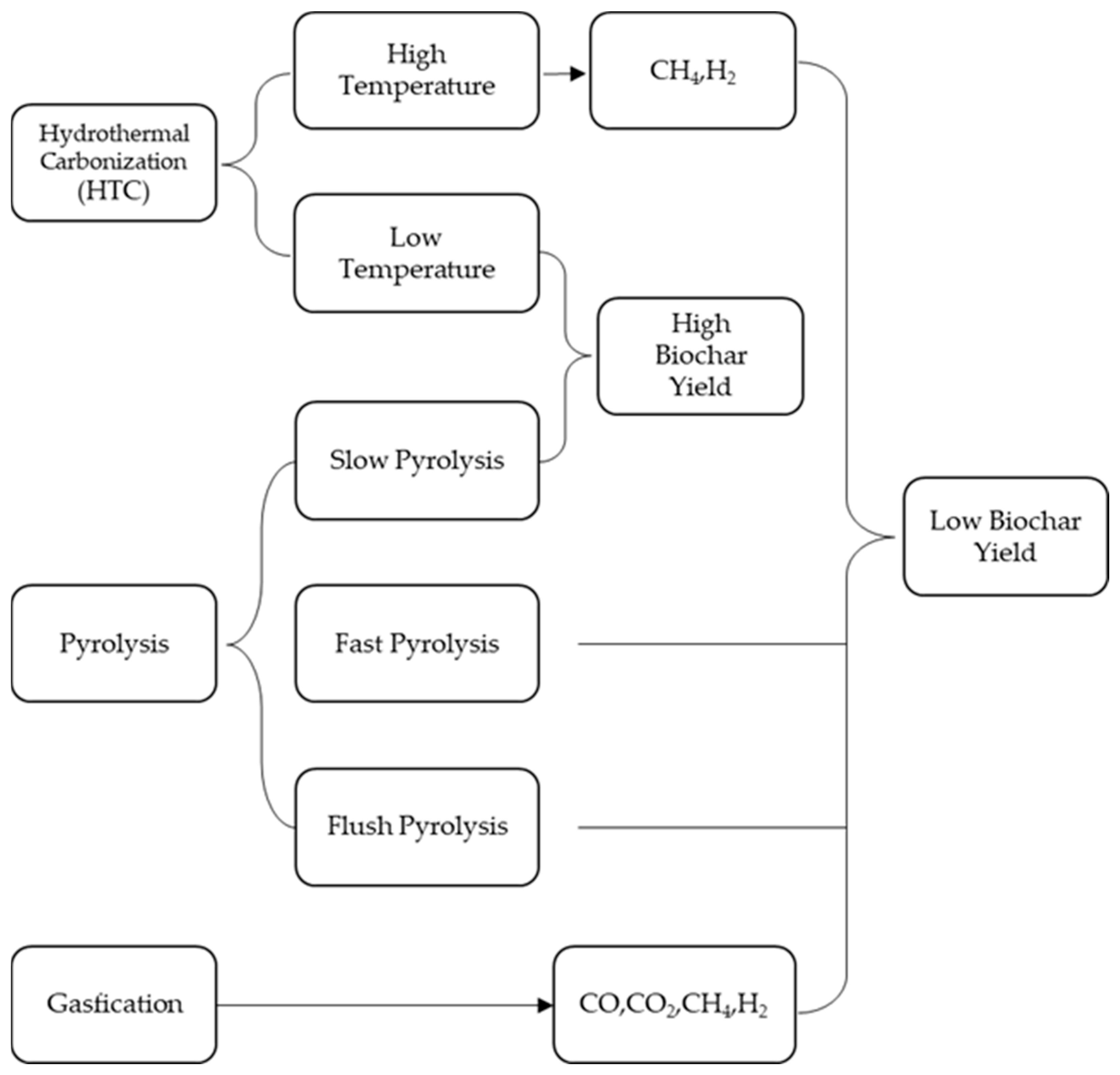
2. Preparation of Biochar Materials
2.1. Pyrolysis
2.2. Gasification
2.3. Hydrothermal Carbonization
3. Modification of Biochar Materials
3.1. Surface Oxidation
3.2. Surface Amination
3.3. Surface Sulfonation
3.4. Metal Nanoparticle Loading
3.5. Surface Binding of Nanostructures
3.6. Other Modification Methods
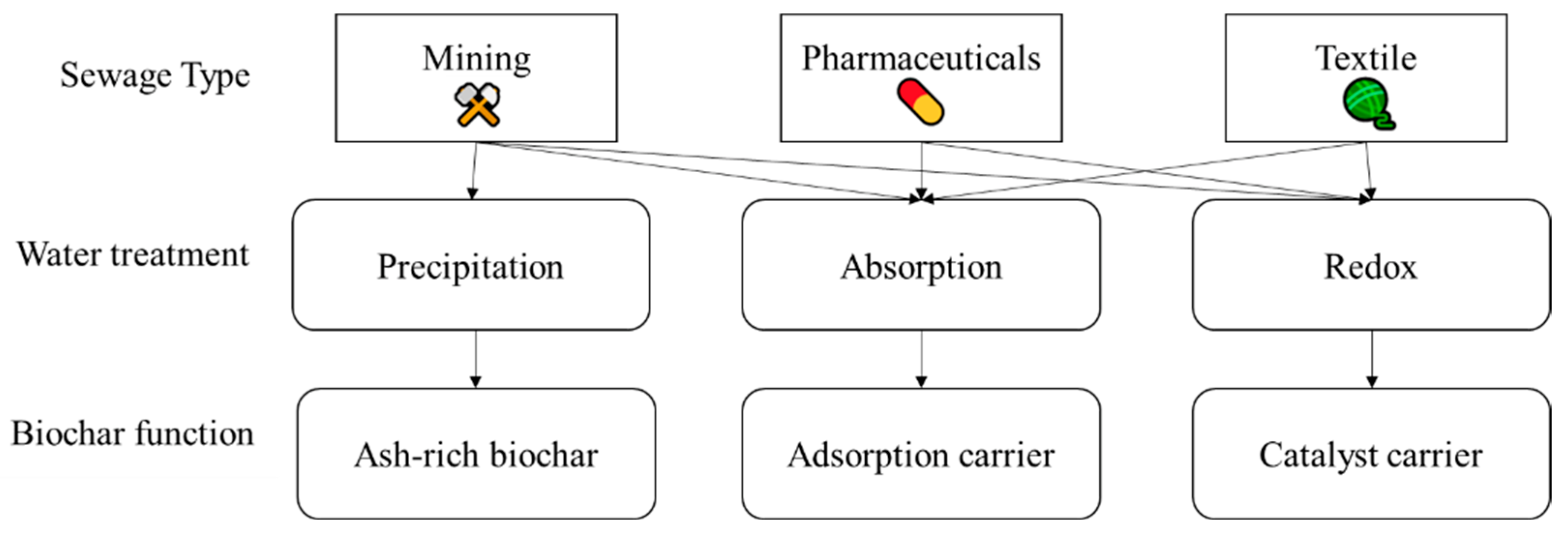
4. Application of Biochar Material for Water Treatment
4.1. Treatment of Domestic Sewage
4.2. Treatment of Industrial Wastewater
4.3. Biochar in Oil Spills
4.4. Different Biochar in Water Treatment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Selvarajoo, A.; Lee, C.W.; Oochit, D.; Almashjary, K.H.O. Bio-pellets from empty fruit bunch and durian rinds with cornstarch adhesive for potential renewable energy. Mater. Sci. Energy Technol. 2021, 4, 242–248. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933. [Google Scholar] [CrossRef]
- Mortensen, P.M.; Grunwaldt, J.D.; Jensen, P.A.; Knudsen, K.G.; Jensen, A.D. A review of catalytic upgrading of bio-oil to engine fuels. Appl. Catal. A Gen. 2011, 407, 1–19. [Google Scholar] [CrossRef]
- Swain, P.K.; Das, L.M.; Naik, S.N. Biomass to liquid: A prospective challenge to research and development in 21st century. Renew. Sustain. Energy Rev. 2011, 15, 4917–4933. [Google Scholar] [CrossRef]
- Kumar, A.; Jones, D.; Hanna, M. Thermochemical Biomass Gasification: A Review of the Current Status of the Technology. Energies 2009, 2, 556–581. [Google Scholar] [CrossRef] [Green Version]
- Petrus, L.; Noordermeer, M.A. Biomass to biofuels, a chemical perspective. Green Chemistry 2006, 8, 861. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Neugschwandtner, R.W.; Kopecky, M.; Amirahmadi, E.; Moudry, J., Jr.; Mensik, L. Preliminary Findings on Cadmium Bioaccumulation and Photosynthesis in Rice (Oryza sativa L.) and Maize (Zea mays L.) Using Biochar Made from C3- and C4-Originated Straw. Plants 2022, 11, 1424. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Neugschwandtner, R.W.; Kopecký, M.; Amirahmadi, E.; Bucur, D.; Walkiewicz, A. Interaction of Biochar with Chemical, Green and Biological Nitrogen Fertilizers on Nitrogen Use Efficiency Indices. Agronomy 2022, 12, 2106. [Google Scholar] [CrossRef]
- Thompson, K.A.; Shimabuku, K.K.; Kearns, J.P.; Knappe, D.R.U.; Summers, R.S.; Cook, S.M. Environmental Comparison of Biochar and Activated Carbon for Tertiary Wastewater Treatment. Environ. Sci. Technol. 2016, 50, 11253–11262. [Google Scholar] [CrossRef]
- Qian, K.; Kumar, A.; Zhang, H.; Bellmer, D.; Huhnke, R. Recent advances in utilization of biochar. Renew. Sustain. Energy Rev. 2015, 42, 1055–1064. [Google Scholar] [CrossRef]
- Huggins, T.; Wang, H.; Kearns, J.; Jenkins, P.; Ren, Z.J. Biochar as a sustainable electrode material for electricity production in microbial fuel cells. Bioresour. Technol. 2014, 157, 114–119. [Google Scholar] [CrossRef]
- Titirici, M.-M.; White, R.J.; Falco, C.; Sevilla, M. Black perspectives for a green future: Hydrothermal carbons for environment protection and energy storage. Energy Environ. Sci. 2012, 5, 6796. [Google Scholar] [CrossRef]
- Manyà, J.J. Pyrolysis for Biochar Purposes: A Review to Establish Current Knowledge Gaps and Research Needs. Environ. Sci. Technol. 2012, 46, 7939–7954. [Google Scholar] [CrossRef]
- Ghorbani, M.; Neugschwandtner, R.W.; Konvalina, P.; Asadi, H.; Kopecký, M.; Amirahmadi, E. Comparative effects of biochar and compost applications on water holding capacity and crop yield of rice under evaporation stress: A two-years field study. Paddy Water Environ. 2023, 21, 47–58. [Google Scholar] [CrossRef]
- Ghorbani, M.; Amirahmadi, E.; Konvalina, P.; Moudrý, J.; Bárta, J.; Kopecký, M.; Teodorescu, R.I.; Bucur, R.D. Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.). Water 2022, 14, 3506. [Google Scholar] [CrossRef]
- IBI. Standardized Product Definition and Product Testing Guidelines for Biochar that Is Used in Soil (Version 1.1), International Biochar Initiative. 2012. Available online: http://www.biochar-international.org/characterizationstandard (accessed on 12 January 2023).
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Ghorbani, M.; Amirahmadi, E.; Neugschwandtner, R.W.; Konvalina, P.; Kopecký, M.; Moudrý, J.; Perná, K.; Murindangabo, Y.T. The Impact of Pyrolysis Temperature on Biochar Properties and Its Effects on Soil Hydrological Properties. Sustainability 2022, 14, 14722. [Google Scholar] [CrossRef]
- Liu, W.-J.; Jiang, H.; Yu, H.-Q. Development of Biochar-Based Functional Materials: Toward a Sustainable Platform Carbon Material. Chem. Rev. 2015, 115, 12251–12285. [Google Scholar] [CrossRef]
- Meyer, S.; Glaser, B.; Quicker, P. Technical, Economical, and Climate-Related Aspects of Biochar Production Technologies: A Literature Review. Environ. Sci. Technol. 2011, 45, 9473–9483. [Google Scholar] [CrossRef]
- Qian, K.; Kumar, A.; Patil, K.; Bellmer, D.; Wang, D.; Yuan, W.; Huhnke, R. Effects of Biomass Feedstocks and Gasification Conditions on the Physiochemical Properties of Char. Energies 2013, 6, 3972–3986. [Google Scholar] [CrossRef]
- Hu, B.; Wang, K.; Wu, L.; Yu, S.-H.; Antonietti, M.; Titirici, M.-M. Engineering Carbon Materials from the Hydrothermal Carbonization Process of Biomass. Adv. Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Kruse, A.; Funke, A.; Titirici, M.-M. Hydrothermal conversion of biomass to fuels and energetic materials. Curr. Opin. Chem. Biol. 2013, 17, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Becidan, M.; Skreiberg, Ø.; Hustad, J.E. NOx and N2O precursors (NH3 and HCN) in pyrolysis of biomass residues. Energy Fuels 2007, 21, 1173–1180. [Google Scholar] [CrossRef]
- Antal, M.J.; Grønli, M. The Art, Science, and Technology of Charcoal Production. Ind. Eng. Chem. Res. 2003, 42, 1619–1640. [Google Scholar] [CrossRef]
- Yaman, S. Pyrolysis of biomass to produce fuels and chemical feedstocks. Energy Convers. Manag. 2004, 45, 651–671. [Google Scholar] [CrossRef]
- Leng, L.; Xiong, Q.; Yang, L.; Li, H.; Zhou, Y.; Zhang, W.; Jiang, S.; Li, H.; Huang, H. An overview on engineering the surface area and porosity of biochar. Sci. Total Environ. 2021, 763, 144204. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Kopecký, M.; Kolář, L. A meta-analysis on the impacts of different oxidation methods on the surface area properties of biochar. Land Degrad. Dev. 2022, 1–14. [Google Scholar] [CrossRef]
- Nguyen, V.H.; Nguyen, D.T.; Nguyen, T.T.; Nguyen, H.P.T.; Khuat, H.B.; Nguyen, T.H.; Tran, V.K.; Woong Chang, S.; Nguyen-Tri, P.; Nguyen, D.D.; et al. Activated carbon with ultrahigh surface area derived from sawdust biowaste for the removal of rhodamine B in water. Environ. Technol. Innov. 2021, 24, 101811. [Google Scholar] [CrossRef]
- Li, M.; Liu, Q.; Guo, L.; Zhang, Y.; Lou, Z.; Wang, Y.; Qian, G. Cu(II) removal from aqueous solution by Spartina alterniflora derived biochar. Bioresour. Technol. 2013, 141, 83–88. [Google Scholar] [CrossRef]
- Xu, D.; Zhao, Y.; Sun, K.; Gao, B.; Wang, Z.; Jin, J.; Zhang, Z.; Wang, S.; Yan, Y.; Liu, X.; et al. Cadmium adsorption on plant- and manure-derived biochar and biochar-amended sandy soils: Impact of bulk and surface properties. Chemosphere 2014, 111, 320–326. [Google Scholar] [CrossRef]
- He, H.; Qian, T.-T.; Liu, W.-J.; Jiang, H.; Yu, H.-Q. Biological and chemical phosphorus solubilization from pyrolytical biochar in aqueous solution. Chemosphere 2014, 113, 175–181. [Google Scholar] [CrossRef]
- Anfruns, A.; García-Suárez, E.J.; Montes-Morán, M.A.; Gonzalez-Olmos, R.; Martin, M.J. New insights into the influence of activated carbon surface oxygen groups on H2O2 decomposition and oxidation of pre-adsorbed volatile organic compounds. Carbon 2014, 77, 89–98. [Google Scholar] [CrossRef]
- Wu, L.; Sitamraju, S.; Xiao, J.; Liu, B.; Li, Z.; Janik, M.J.; Song, C. Effect of liquid-phase O3 oxidation of activated carbon on the adsorption of thiophene. Chem. Eng. J. 2014, 242, 211–219. [Google Scholar] [CrossRef]
- Sun, C.; Snape, C.E.; Liu, H. Development of Low-Cost Functional Adsorbents for Control of Mercury (Hg) Emissions from Coal Combustion. Energy Fuels 2013, 27, 3875–3882. [Google Scholar] [CrossRef]
- Gokce, Y.; Aktas, Z. Nitric acid modification of activated carbon produced from waste tea and adsorption of methylene blue and phenol. Appl. Surf. Sci. 2014, 313, 352–359. [Google Scholar] [CrossRef]
- Yang, G.; Chen, H.; Qin, H.; Feng, Y. Amination of activated carbon for enhancing phenol adsorption: Effect of nitrogen-containing functional groups. Appl. Surf. Sci. 2014, 293, 299–305. [Google Scholar] [CrossRef]
- Adelodun, A.A.; Lim, Y.H.; Jo, Y.M. Stabilization of potassium-doped activated carbon by amination for improved CO2 selective capture. J. Anal. Appl. Pyrolysis 2014, 108, 151–159. [Google Scholar] [CrossRef]
- Shafeeyan, M.S.; Wan Daud, W.M.A.; Houshmand, A.; Arami-Niya, A. The application of response surface methodology to optimize the amination of activated carbon for the preparation of carbon dioxide adsorbents. Fuel 2012, 94, 465–472. [Google Scholar] [CrossRef]
- Jansen, R.J.J.; Van Bekkum, H. Amination and ammoxidation of activated carbons. Carbon 1994, 32, 1507–1516. [Google Scholar] [CrossRef]
- Stöhr, B.; Boehm, H.P.; Schlögl, R. Enhancement of the catalytic activity of activated carbons in oxidation reactions by thermal treatment with ammonia or hydrogen cyanide and observation of a superoxide species as a possible intermediate. Carbon 1991, 29, 707–720. [Google Scholar] [CrossRef]
- Titirici, M.-M.; Thomas, A.; Antonietti, M. Aminated hydrophilic ordered mesoporous carbons. J. Mater. Chem. 2007, 17, 3412. [Google Scholar] [CrossRef]
- Zhao, L.; Bacsik, Z.; Hedin, N.; Wei, W.; Sun, Y.; Antonietti, M.; Titirici, M.-M. Carbon Dioxide Capture on Amine-Rich Carbonaceous Materials Derived from Glucose. ChemSusChem 2010, 3, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, W.-J.; Zhang, N.; Li, Y.-S.; Jiang, H.; Sheng, G.-P. Polyethylenimine modified biochar adsorbent for hexavalent chromium removal from the aqueous solution. Bioresour. Technol. 2014, 169, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; He, Y.; He, Y.; Liu, X.; Xu, B.; Yu, J.; Dai, C.; Huang, A.; Pang, Y.; Luo, L. Analyses of tetracycline adsorption on alkali-acid modified magnetic biochar: Site energy distribution consideration. Sci. Total Environ. 2019, 650, 2260–2266. [Google Scholar] [CrossRef] [PubMed]
- Takagaki, A.; Tagusagawa, C.; Hayashi, S.; Hara, M.; Domen, K. Nanosheets as highly active solid acid catalysts for green chemical syntheses. Energy Environ. Sci. 2010, 3, 82–93. [Google Scholar] [CrossRef]
- Nakajima, K.; Hara, M. Amorphous Carbon with SO3H groups as a Solid Brønsted Acid Catalyst. ACS Catal. 2012, 2, 1296–1304. [Google Scholar] [CrossRef]
- Dehkhoda, A.M.; Ellis, N. Biochar-based catalyst for simultaneous reactions of esterification and transesterification. Catal. Today 2013, 207, 86–92. [Google Scholar] [CrossRef]
- Dehkhoda, A.M.; West, A.H.; Ellis, N. Biochar based solid acid catalyst for biodiesel production. Appl. Catal. A Gen. 2010, 382, 197–204. [Google Scholar] [CrossRef]
- Kastner, J.R.; Miller, J.; Geller, D.P.; Locklin, J.; Keith, L.H.; Johnson, T. Catalytic esterification of fatty acids using solid acid catalysts generated from biochar and activated carbon. Catal. Today 2012, 190, 122–132. [Google Scholar] [CrossRef]
- Ormsby, R.; Kastner, J.R.; Miller, J. Hemicellulose hydrolysis using solid acid catalysts generated from biochar. Catal. Today 2012, 190, 89–97. [Google Scholar] [CrossRef]
- Li, S.; Gu, Z.; Bjornson, B.E.; Muthukumarappan, A. Biochar based solid acid catalyst hydrolyze biomass. J. Environ. Chem. Eng. 2013, 1, 1174–1181. [Google Scholar] [CrossRef]
- Wu, Y.; Fu, Z.; Yin, D.; Xu, Q.; Liu, F.; Lu, C.; Mao, L. Microwave-assisted hydrolysis of crystalline cellulose catalyzed by biomass char sulfonic acids. Green Chem. 2010, 12, 696. [Google Scholar] [CrossRef]
- Hara, M. Biomass conversion by a solid acid catalyst. Energy Environ. Sci. 2010, 3, 601. [Google Scholar] [CrossRef]
- Zhang, C.; Fu, Z.; Liu, Y.C.; Dai, B.; Zou, Y.; Gong, X.; Wang, Y.; Deng, X.; Wu, H.; Xu, Q.; et al. Ionic liquid-functionalized biochar sulfonic acid as a biomimetic catalyst for hydrolysis of cellulose and bamboo under microwave irradiation. Green Chem. 2012, 14, 1928. [Google Scholar] [CrossRef]
- Liu, W.-J.; Tian, K.; Jiang, H.; Yu, H.-Q. Facile synthesis of highly efficient and recyclable magnetic solid acid from biomass waste. Sci. Rep. 2013, 3, 2419. [Google Scholar] [CrossRef] [Green Version]
- Richardson, Y.; Blin, J.; Volle, G.; Motuzas, J.; Julbe, A. In situ generation of Ni metal nanoparticles as catalyst for H2-rich syngas production from biomass gasification. Appl. Catal. A Gen. 2010, 382, 220–230. [Google Scholar] [CrossRef]
- Richardson, Y.; Motuzas, J.; Julbe, A.; Volle, G.; Blin, J. Catalytic Investigation of in Situ Generated Ni Metal Nanoparticles for Tar Conversion during Biomass Pyrolysis. J. Phys. Chem. C 2013, 117, 23812–23831. [Google Scholar] [CrossRef]
- Lai, C.; Huang, F.; Zeng, G.; Huang, D.; Qin, L.; Cheng, M.; Zhang, C.; Li, B.; Yi, H.; Liu, S.; et al. Fabrication of novel magnetic MnFe2O4/bio-char composite and heterogeneous photo-Fenton degradation of tetracycline in near neutral pH. Chemosphere 2019, 224, 910–921. [Google Scholar] [CrossRef]
- Inyang, M.; Gao, B.; Zimmerman, A.; Zhang, M.; Chen, H. Synthesis, characterization, and dye sorption ability of carbon nanotube–biochar nanocomposites. Chem. Eng. J. 2014, 236, 39–46. [Google Scholar] [CrossRef]
- Su, D.S.; Chen, X.-W. Natural Lavas as Catalysts for Efficient Production of Carbon Nanotubes and Nanofibers. Angew. Chem. 2007, 119, 1855–1856. [Google Scholar] [CrossRef]
- Shen, Y.; Zhao, P.; Shao, Q. Porous silica and carbon derived materials from rice husk pyrolysis char. Microporous Mesoporous Mater. 2014, 188, 46–76. [Google Scholar] [CrossRef]
- Ao, W.; Fu, J.; Mao, X.; Kang, Q.; Ran, C.; Liu, Y.; Zhang, H.; Gao, Z.; Li, J.; Liu, G.; et al. Microwave assisted preparation of activated carbon from biomass: A review. Renew. Sustain. Energy Rev. 2018, 92, 958–979. [Google Scholar] [CrossRef]
- Zhang, Y.; Fan, S.; Liu, T.; Fu, W.; Li, B. A review of biochar prepared by microwave-assisted pyrolysis of organic wastes. Sustain. Energy Technol. Assess. 2022, 50, 101873. [Google Scholar] [CrossRef]
- Sun, Y.; Yu, I.K.M.; Tsang, D.C.W.; Fan, J.; Clark, J.H.; Luo, G.; Zhang, S.; Khan, E.; Graham, N.J.D. Tailored design of graphitic biochar for high-efficiency and chemical-free microwave-assisted removal of refractory organic contaminants. Chem. Eng. J. 2020, 398, 125505. [Google Scholar] [CrossRef]
- Qu, J.; Wang, S.; Jin, L.; Liu, Y.; Yin, R.; Jiang, Z.; Tao, Y.; Huang, J.; Zhang, Y. Magnetic porous biochar with high specific surface area derived from microwave-assisted hydrothermal and pyrolysis treatments of water hyacinth for Cr(Ⅵ) and tetracycline adsorption from water. Bioresour. Technol. 2021, 340, 125692. [Google Scholar] [CrossRef]
- Wang, H.; Teng, H.; Wang, X.; Xu, J.; Sheng, L. Physicochemical modification of corn straw biochar to improve performance and its application of constructed wetland substrate to treat city tail water. J. Environ. Manag. 2022, 310, 114758. [Google Scholar] [CrossRef]
- Gholami, P.; Dinpazhoh, L.; Khataee, A.; Hassani, A.; Bhatnagar, A. Facile hydrothermal synthesis of novel Fe-Cu layered double hydroxide/biochar nanocomposite with enhanced sonocatalytic activity for degradation of cefazolin sodium. J. Hazard. Mater. 2020, 381, 120742. [Google Scholar] [CrossRef]
- Wang, Y.; Miao, J.; Saleem, M.; Yang, Y.; Zhang, Q. Enhanced adsorptive removal of carbendazim from water by FeCl3-modified corn straw biochar as compared with pristine, HCl and NaOH modification. J. Environ. Chem. Eng. 2022, 10, 107024. [Google Scholar] [CrossRef]
- Mao, W.; Zhang, Y.; Luo, J.; Chen, L.; Guan, Y. Novel co-polymerization of polypyrrole/polyaniline on ferrate modified biochar composites for the efficient adsorption of hexavalent chromium in water. Chemosphere 2022, 303, 135254. [Google Scholar] [CrossRef]
- Du, H.; Xi, C.; Tang, B.; Chen, W.; Deng, W.; Cao, S.; Jiang, G. Performance and mechanisms of NaOH and ball-milling co-modified biochar for enhanced the removal of Cd2+ in synthetic water: A combined experimental and DFT study. Arab. J. Chem. 2022, 15, 103817. [Google Scholar] [CrossRef]
- Wang, Q.; Duan, C.-J.; Xu, C.-Y.; Geng, Z.-C. Efficient removal of Cd(II) by phosphate-modified biochars derived from apple tree branches: Processes, mechanisms, and application. Sci. Total Environ. 2022, 819, 152876. [Google Scholar] [CrossRef]
- Khan, B.A.; Ahmad, M.; Iqbal, S.; Bolan, N.; Zubair, S.; Shafique, M.A.; Shah, A. Effectiveness of the engineered pinecone-derived biochar for the removal of fluoride from water. Environ. Res. 2022, 212, 113540. [Google Scholar] [CrossRef]
- Liu, Z.; Zhen, F.; Zhang, Q.; Qian, X.; Li, W.; Sun, Y.; Zhang, L.; Qu, B. Nanoporous biochar with high specific surface area based on rice straw digestion residue for efficient adsorption of mercury ion from water. Bioresour. Technol. 2022, 359, 127471. [Google Scholar] [CrossRef]
- Yang, T.; Xu, Y.; Huang, Q.; Sun, Y.; Liang, X.; Wang, L. Removal mechanisms of Cd from water and soil using Fe–Mn oxides modified biochar. Environ. Res. 2022, 212, 113406. [Google Scholar] [CrossRef]
- Iqbal, J.; Mohamed Al Hajeri, B.; Shah, N.S.; Wilson, K.; Xavier, C.; Shaalan, J.; Al-Taani, A.A.; Howari, F.; Nazzal, Y. Preparation of H3PO4 modified Sidr biochar for the enhanced removal of ciprofloxacin from water. Int. J. Phytoremediat. 2022, 24, 1231–1242. [Google Scholar] [CrossRef]
- Li, X.; Shi, J.; Luo, X. Enhanced adsorption of rhodamine B from water by Fe-N co-modified biochar: Preparation, performance, mechanism and reusability. Bioresour. Technol. 2022, 343, 126103. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, Y.; Song, T.; Su, R.; Luo, J. Activated peroxymonosulfate with ferric chloride-modified biochar to degrade bisphenol A: Characteristics, influencing factors, reaction mechanism and reuse performance. Sep. Purif. Technol. 2022, 300, 121857. [Google Scholar] [CrossRef]
- Wei, Y.; Wei, S.; Liu, C.; Chen, T.; Tang, Y.; Ma, J.; Yin, K.; Luo, S. Efficient removal of arsenic from groundwater using iron oxide nanoneedle array-decorated biochar fibers with high Fe utilization and fast adsorption kinetics. Water Res. 2019, 167, 115107. [Google Scholar] [CrossRef]
- Wei, Y.; Yang, W.; Wang, Z.; Yin, K.; Chen, T.; Liu, C. Enhanced removal of As(III) by heterogeneous catalytic oxidation of As(III) on Fe-biochar fibers with H2O2 and hydroxylamine. Chem. Eng. J. 2022, 428, 131200. [Google Scholar] [CrossRef]
- Zhu, Y.; Fan, W.; Feng, W.; Wang, Y.; Liu, S.; Dong, Z.; Li, X. Removal of EDTA-Cu(II) from Water Using Synergistic Fenton Reaction-Assisted Adsorption by Nanomanganese Oxide-Modified Biochar: Performance and Mechanistic Analysis. ACS EST Water 2021, 1, 1302–1312. [Google Scholar] [CrossRef]
- Sun, R.; Zhang, X.; Wang, C.; Cao, Y. Co-carbonization of red mud and waste sawdust for functional application as Fenton catalyst: Evaluation of catalytic activity and mechanism. J. Environ. Chem. Eng. 2021, 9, 105368. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Xie, X.; Wang, Z. Bifunctional MnFe2O4/chitosan modified biochar composite for enhanced methyl orange removal based on adsorption and photo-Fenton process. Colloids Surf. A Physicochem. Eng. Asp. 2021, 613, 126104. [Google Scholar] [CrossRef]
- Sang, F.; Yin, Z.; Wang, W.; Almatrafi, E.; Wang, Y.; Zhao, B.; Gong, J.; Zhou, C.; Zhang, C.; Zeng, G.; et al. Degradation of ciprofloxacin using heterogeneous Fenton catalysts derived from natural pyrite and rice straw biochar. J. Clean. Prod. 2022, 378, 134459. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, Y. Preparation of hydrogen sulfide adsorbent derived from spent Fenton-like reagent modified biochar and its removal characteristics for hydrogen sulfide. Fuel Process. Technol. 2022, 238, 107495. [Google Scholar] [CrossRef]
- Xin, S.; Liu, G.; Ma, X.; Gong, J.; Ma, B.; Yan, Q.; Chen, Q.; Ma, D.; Zhang, G.; Gao, M.; et al. High efficiency heterogeneous Fenton-like catalyst biochar modified CuFeO2 for the degradation of tetracycline: Economical synthesis, catalytic performance and mechanism. Appl. Catal. B Environ. 2021, 280, 119386. [Google Scholar] [CrossRef]
- Yi, K.; Lei, M.; Peng, L.; Chen, A.; Luo, S. Sunlight-driven degradation of diethyl phthalate via magnetically modified biochar catalysts in water: Internal electron transfer mechanism. Chemosphere 2021, 269, 129366. [Google Scholar] [CrossRef]
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. MWH's Water Treatment: Principles and Design; John Wiley & Sons: Hoboken, NJ, USA, 2012; pp. 34–35. [Google Scholar]
- Jin, L.; Zhang, G.; Tian, H. Current state of sewage treatment in China. Water Res. 2014, 66, 85–98. [Google Scholar] [CrossRef]
- Yang, X.; Zeng, L.; Huang, J.; Mo, Z.; Guan, Z.; Sun, S.; Liang, J.; Huang, S. Enhanced sludge dewaterability by a novel MnFe2O4-Biochar activated peroxymonosulfate process combined with Tannic acid. Chem. Eng. J. 2022, 429, 132280. [Google Scholar] [CrossRef]
- He, M.; Xu, Z.; Hou, D.; Gao, B.; Cao, X.; Ok, Y.S.; Rinklebe, J.; Bolan, N.S.; Tsang, D.C.W. Waste-derived biochar for water pollution control and sustainable development. Nat. Rev. Earth Environ. 2022, 3, 444–460. [Google Scholar] [CrossRef]
- Wang, C.; Liu, Y.; Gao, X.; Chen, H.; Xu, X.; Zhu, L. Role of biochar in the granulation of anaerobic sludge and improvement of electron transfer characteristics. Bioresour. Technol. 2018, 268, 28–35. [Google Scholar] [CrossRef]
- Barbusiński, K.; Parzentna-Gabor, A.; Kasperczyk, D. Removal of Odors (Mainly H2S and NH3) Using Biological Treatment Methods. Clean Technol. 2021, 3, 138–155. [Google Scholar] [CrossRef]
- He, M.; Xiong, X.; Wang, L.; Hou, D.; Bolan, N.S.; Ok, Y.S.; Rinklebe, J.; Tsang, D.C.W. A critical review on performance indicators for evaluating soil biota and soil health of biochar-amended soils. J. Hazard. Mater. 2021, 414, 125378. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Walkiewicz, A.; Neugschwandtner, R.W.; Kopecky, M.; Zamanian, K.; Chen, W.H.; Bucur, D. Feasibility of Biochar Derived from Sewage Sludge to Promote Sustainable Agriculture and Mitigate GHG Emissions-A Review. Int. J. Environ. Res. Public Health 2022, 19, 12983. [Google Scholar] [CrossRef]
- Wu, J.; Wang, T.; Wang, J.; Zhang, Y.; Pan, W.-P. A novel modified method for the efficient removal of Pb and Cd from wastewater by biochar: Enhanced the ion exchange and precipitation capacity. Sci. Total Environ. 2021, 754, 142150. [Google Scholar] [CrossRef]
- Xu, X.; Cao, X.; Zhao, L.; Wang, H.; Yu, H.; Gao, B. Removal of Cu, Zn, and Cd from aqueous solutions by the dairy manure-derived biochar. Environ. Sci. Pollut. Res. 2012, 20, 358–368. [Google Scholar] [CrossRef]
- Xu, Z.; Xu, X.; Tsang, D.C.W.; Cao, X. Contrasting impacts of pre- and post-application aging of biochar on the immobilization of Cd in contaminated soils. Environ. Pollut. 2018, 242, 1362–1370. [Google Scholar] [CrossRef]
- Xu, X.; Cao, X.; Zhao, L. Comparison of rice husk- and dairy manure-derived biochars for simultaneously removing heavy metals from aqueous solutions: Role of mineral components in biochars. Chemosphere 2013, 92, 955–961. [Google Scholar] [CrossRef]
- Sun, T.; Levin, B.D.A.; Guzman, J.J.L.; Enders, A.; Muller, D.A.; Angenent, L.T.; Lehmann, J. Rapid electron transfer by the carbon matrix in natural pyrogenic carbon. Nat. Commun. 2017, 8, 14873. [Google Scholar] [CrossRef] [Green Version]
- Fan, Z.; Fang, J.; Zhang, G.; Qin, L.; Fang, Z.; Jin, L. Improved Adsorption of Tetracycline in Water by a Modified Caulis spatholobi Residue Biochar. ACS Omega 2022, 7, 30543–30553. [Google Scholar] [CrossRef]
- Mao, Q.; Zhou, Y.; Yang, Y.; Zhang, J.; Liang, L.; Wang, H.; Luo, S.; Luo, L.; Jeyakumar, P.; Ok, Y.S.; et al. Experimental and theoretical aspects of biochar-supported nanoscale zero-valent iron activating H2O2 for ciprofloxacin removal from aqueous solution. J. Hazard. Mater. 2019, 380, 120848. [Google Scholar] [CrossRef]
- Rubeena, K.K.; Hari Prasad Reddy, P.; Laiju, A.R.; Nidheesh, P.V. Iron impregnated biochars as heterogeneous Fenton catalyst for the degradation of acid red 1 dye. J. Environ. Manag. 2018, 226, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Rong, X.; Xie, M.; Kong, L.; Natarajan, V.; Ma, L.; Zhan, J. The magnetic biochar derived from banana peels as a persulfate activator for organic contaminants degradation. Chem. Eng. J. 2019, 372, 294–303. [Google Scholar] [CrossRef]
- Li, S.; Wang, Z.; Zhao, X.; Yang, X.; Liang, G.; Xie, X. Insight into enhanced carbamazepine photodegradation over biochar-based magnetic photocatalyst Fe3O4/BiOBr/BC under visible LED light irradiation. Chem. Eng. J. 2019, 360, 600–611. [Google Scholar] [CrossRef]
- Zeng, S.; Kan, E. FeCl3-activated biochar catalyst for heterogeneous Fenton oxidation of antibiotic sulfamethoxazole in water. Chemosphere 2022, 306, 135554. [Google Scholar] [CrossRef] [PubMed]
- Lingamdinne, L.P.; Choi, J.-S.; Angaru, G.K.R.; Karri, R.R.; Yang, J.-K.; Chang, Y.-Y.; Koduru, J.R. Magnetic-watermelon rinds biochar for uranium-contaminated water treatment using an electromagnetic semi-batch column with removal mechanistic investigations. Chemosphere 2022, 286, 131776. [Google Scholar] [CrossRef]
- Wang, Y.; Liang, J.; Wang, J.; Gao, S. Combining stable carbon isotope analysis and petroleum-fingerprinting to evaluate petroleum contamination in the Yanchang oilfield located on loess plateau in China. Environ. Sci. Pollut. Res. Int. 2018, 25, 2830–2841. [Google Scholar] [CrossRef]
- Chen, M.; Xu, P.; Zeng, G.; Yang, C.; Huang, D.; Zhang, J. Bioremediation of soils contaminated with polycyclic aromatic hydrocarbons, petroleum, pesticides, chlorophenols and heavy metals by composting: Applications, microbes and future research needs. Biotechnol. Adv. 2015, 33, 745–755. [Google Scholar] [CrossRef]
- Harshvardhan, K.; Jha, B. Biodegradation of low-density polyethylene by marine bacteria from pelagic waters, Arabian Sea, India. Mar. Pollut. Bull. 2013, 77, 100–106. [Google Scholar] [CrossRef]
- Saeed, M.; Ilyas, N.; Jayachandran, K.; Shabir, S.; Akhtar, N.; Shahzad, A.; Sayyed, R.Z.; Bano, A. Advances in Biochar and PGPR engineering system for hydrocarbon degradation: A promising strategy for environmental remediation. Environ. Pollut. 2022, 305, 119282. [Google Scholar] [CrossRef]
- Zeng, G.; Huang, X.; Yue, J.; Fan, B.; Liu, Y.; Tang, X.-Z. Solar-assisted efficient cleanup of high-viscosity oil spills using magnetic porous biochar. J. Alloy. Compd. 2022, 924, 166474. [Google Scholar] [CrossRef]
- Bazargan, A.; Tan, J.; Hui, C.W.; McKay, G. Utilization of rice husks for the production of oil sorbent materials. Cellulose 2014, 21, 1679–1688. [Google Scholar] [CrossRef]
- Jain, R.; Mathur, M.; Sikarwar, S.; Mittal, A. Removal of the hazardous dye rhodamine B through photocatalytic and adsorption treatments. J. Environ. Manag. 2007, 85, 956–964. [Google Scholar] [CrossRef]
- Da Silva, W.L.; Muraro, P.C.L.; Pavoski, G.; Espinosa, D.C.R.; Dos Santos, J.H.Z. Preparation and characterization of biochar from cement waste for removal of rhodamine B dye. J. Mater. Cycles Waste Manag. 2022, 24, 1333–1342. [Google Scholar] [CrossRef]
- Adekola, F.; Ayodele, S.; Inyinbor, A. Efficient Rhodamine B Removal Using Acidand Alkaline-Activated Musa paradisiaca Biochar. Pol. J. Environ. Stud. 2019, 28, 3063–3070. [Google Scholar] [CrossRef]
- Albanio, I.I.; Muraro, P.C.L.; Da Silva, W.L. Rhodamine B Dye Adsorption onto Biochar from Olive Biomass Waste. Water, Air, Soil Pollut. 2021, 232, 1–11. [Google Scholar] [CrossRef]
- Adekola, F.A.; Ayodele, S.B.; Inyinbor, A.A. Activated biochar prepared from plaintain peels: Characterization and Rhodamine B adsorption data set. Chem. Data Collect. 2019, 19, 100170. [Google Scholar] [CrossRef]
- Peng, Z.; Fan, Z.; Chen, X.; Zhou, X.; Gao, Z.F.; Deng, S.; Wan, S.; Lv, X.; Shi, Y.; Han, W. Fabrication of Nano Iron Oxide–Modified Biochar from Co-Hydrothermal Carbonization of Microalgae and Fe(II) Salt for Efficient Removal of Rhodamine B. Nanomaterials 2022, 12, 2271. [Google Scholar] [CrossRef]
- Hou, Y.; Huang, G.; Li, J.; Yang, Q.; Huang, S.; Cai, J. Hydrothermal conversion of bamboo shoot shell to biochar: Preliminary studies of adsorption equilibrium and kinetics for rhodamine B removal. J. Anal. Appl. Pyrolysis 2019, 143, 104694. [Google Scholar] [CrossRef]
- Zhu, B.; Yu, Y.; Ding, Y.; Ge, S. Iron-modified granular sludge biochar-based catalysts for improved Rhodamine B degradation by activating peroxymonosulfate. Biomass Convers. Biorefinery 2022, 1–11. [Google Scholar] [CrossRef]
- Fatimah, I.; Purwiandono, G.; Sahroni, I.; Wijayana, A.; Faraswati, M.; Dwi Putri, A.; Oh, W.-C.; Doong, R.-A. Magnetically-separable photocatalyst of magnetic biochar from snake fruit peel for rhodamine B photooxidation. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100669. [Google Scholar] [CrossRef]
- Cheng, Z.; Li, S.; Nguyen, T.T.; Gao, X.; Luo, S.; Guo, M. Biochar loaded on MnFe2O4 as Fenton catalyst for Rhodamine B removal: Characterizations, catalytic performance, process optimization and mechanism. Colloids Surf. A Physicochem. Eng. Asp. 2021, 631, 127651. [Google Scholar] [CrossRef]
- Kang, F.; Shi, C.; Li, W.; Eqi, M.; Liu, Z.; Zheng, X.; Huang, Z. Honeycomb like CdS/sulphur-modified biochar composites with enhanced adsorption-photocatalytic capacity for effective removal of rhodamine B. J. Environ. Chem. Eng. 2022, 10, 106942. [Google Scholar] [CrossRef]

| Slow Pyrolysis | Fast Pyrolysis | Flush Pyrolysis | Pyrolytic Gasification | |
|---|---|---|---|---|
| Heating rate (°C·min−1) | 5–7 | 300–800 | ~1000 | - |
| Temperature (°C) | 300–800 | 400–600 | 400–1000 | 750–1000 |
| Vapor residence time | >1 h | 0.5–10 s | <2 s | 10–20 s |
| Typical reactor | Fixed Bed | Fluidized Bed | Fluidized Bed | Fluidized Bed |
| Main Product | Biochar | Bio-oil | Biogas | Biogas |
| Biochar yield (wt%) | 35–50 | 15–35 | 10–20 | 10–20 |
| Type | Surface Oxidation | Surface Amination | Surface Sulfonation | Metal Nanoparticle Loading | Surface Binding of Nanostructures |
|---|---|---|---|---|---|
| Target functional groups | Carbonyl, hydroxyl, carboxyl | Nitro | Sulfonic acid group | - | - |
| Reagents used | H2O2, O3, KMnO4, HNO3 | NH3 | Fuming sulfuric acid or Chlorosulfonic acid | Metal solutions | CNT |
| Features | Adsorption capacity | Hydrophilic or Hydrophobic | Efficiency and stability | Catalytically active | Rich surface functional groups |
| Feedstock | Modifier | Modification Method | Pyrolysis temperature (°C) | Specific Surface Area (m2·g−1) | Contaminant | Treatment | References |
|---|---|---|---|---|---|---|---|
| Corn straw | FeCl3 | Pyrolyzed, stirred, pyrolyzed | 300–700 | 4.35–173.9 | Carbendazim | Adsorption | [69] |
| Forestry waste | K2FeO4, Pyrrole, Aniline | Soaked, pyrolyzed, vigorously stirred | 300 | 56.97 | Cr(VI) | Absorption and Photocatalytic | [70] |
| Citrus peel | NaOH, Fe3O4 | Soaked, pyrolyzed | 500–800 | 64.45–288.91 | Cd(II) | Adsorption | [71] |
| Apple tree branches | Phosphate | Stirred, pyrolyzed | 500 | 37.8 | Cd(II) | Adsorption | [72] |
| Pinecone | FeCl3, AlCl3 | Pyrolyzed, stirred | 600 | - | Fluoride | Adsorption | [73] |
| Corn straw | NaOH, KMnO4, H2SO4 | Pyrolyzed, Freeze-thaw cycles, Soaked | 450 | 412.058 | City tail water | Adsorption | [67] |
| Rice straw | KOH | Hydrothermal, pyrolyzed | 750 | 2372.51 | Hg | Adsorption | [74] |
| Durian shell, branches of Robinia pseudoacacia | Fe (NO)3⋅9H2O MnSO4⋅H2O | Pyrolyzed, shaken, pyrolyzed | 500 | 96.35 | Cd(II) | Adsorption | [75] |
| Sidr plant leaves | H3PO4 | Pyrolyzed, soaked | 450 | 4.2948–6.0873 | Ciprofloxacin | Adsorption | [76] |
| Coconut shell | Urea, FeSO4 | Soaked, Pyrolyzed | 500 | 637.0493–972.8714 | Rhodamine B | Adsorption | [77] |
| Rice husks | FeCl3 | Pyrolyzed, stirred, pyrolyzed | 600–800 | 79.3736–264.5860 | Bisphenol A | Adsorption and Catalytic | [78] |
| Cotton | FeCl3, NaSO4, HNO3 | Pyrolyzed, soaked, hydrothermal | 800 | 2.45–8.68 | As(III) | Adsorption | [79,80] |
| Coconut shell | KMnO4 | Soaked, pyrolyzed | 300–600 | 396.41–465.84 | EDTA-Cu(II) | Fenton-like catalytic (Redox) | [81] |
| Waste sawdust | Red mud | Mixed, pyrolyzed | 800 | 23.51 | Rhodamine B | Fenton-like catalytic (Redox) | [82] |
| Potato straw | MnFe2O4 | Pyrolyzed, sonicated | 500 | 99.43 | Methyl orange | Fenton-like catalytic (Redox) | [83] |
| Rice straw | FeS2 | Mixed, pyrolyzed (calcined) | 500 | 15.07 | Ciprofloxacin | Fenton-like catalytic (Redox) | [84] |
| Rice straw | Cu(NO3)2 | Microwaved, stirred, pyrolyzed (calcined) | 600 | 5.57–280.07 36.059 after modification | Hydrogen sulfide | Adsorption and Fenton-like catalytic (Redox) | [85] |
| Pepper stalks | CuFeO2 | Pyrolyzed, hydrothermal | 450 | 25.4–37.3 | Tetracycline | Fenton-like catalytic (Redox) | [86] |
| Rice straw | FeCl3 | Pyrolyzed, sonicated, pyrolyzed | 800 | - | Diethyl phthalate | Sunlight-driven degradation (Redox) | [87] |
| Feedstock | Modifier | Treatment | Pyrolysis Temperature (°C) | Specific Surface Area (m2·g−1) | pH in Reaction | Adsorption Capacity or Catalysis Efficiency | References |
|---|---|---|---|---|---|---|---|
| Cement waste | ZnCl2 | Adsorption | 600 | 23.30 | 4.3 | 531.836 mg·g−1 | [115] |
| Musa paradisiaca | KOH and H3PO4 | Adsorption | 500 | 985 | 4 | 7.003 mg·g−1 and 6.878 mg·g−1 | [116] |
| Olive Biomass Waste | ZnCl2 | Adsorption | 600 | - | 4 or 3 | 263.71 mg·g−1 | [117] |
| Plaintain peels | Fe(NO3)3 | Adsorption | 500 | - | 7 | 84.41 mg·g−1 | [118] |
| Chlorella | FeCl3 and FeSO4 | Adsorption | 900 | 338.6, 350.2, 527.6 | 7 | 179.7 mg·g−1 (CBC) | [119] |
| 185.1 mg·g−1 CBC-Fe(II) | |||||||
| 289.6 mg·g−1 CBC-Fe(III) | |||||||
| Bamboo shoot shell | hydrothermal carbonization | Adsorption | 800 | 513 | 7 | 85.8 mg·g−1 | [120] |
| Anaerobic granular sludge | Fe(NO3)3 | Catalysis | 800 | 249.145 | 3 | 90% at 0.4 g·L−1 | [121] |
| Snake fruit peel | FeCl3 and FeSO4 | Catalysis | 400 | 126.8 | 7 | 99.90% | [122] |
| Poplar wood flour | FeCl3 and MnSO4 | Catalysis | 600 | 115.11 | 5 | 93.1% at 1.0 g·L−1 | [123] |
| Bagasse | Cd(NO3)2 | Catalysis | 600 | 609.35 | 4.14 | Nearly 100% at 0.5 g·L−1 | [124] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, R.; Wu, Y.; Lou, X.; Li, H.; Cheng, J.; Shen, B.; Qin, L. Porous Biochar Materials for Sustainable Water Treatment: Synthesis, Modification, and Application. Water 2023, 15, 395. https://doi.org/10.3390/w15030395
Li R, Wu Y, Lou X, Li H, Cheng J, Shen B, Qin L. Porous Biochar Materials for Sustainable Water Treatment: Synthesis, Modification, and Application. Water. 2023; 15(3):395. https://doi.org/10.3390/w15030395
Chicago/Turabian StyleLi, Ruichenzhi, Yujiao Wu, Xujun Lou, Haorui Li, Jing Cheng, Bin Shen, and Lei Qin. 2023. "Porous Biochar Materials for Sustainable Water Treatment: Synthesis, Modification, and Application" Water 15, no. 3: 395. https://doi.org/10.3390/w15030395





