Comparative Screening Study on the Adsorption of Aqueous Pb(II) Using Different Metabolically Inhibited Bacterial Cultures from Industry
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microbial Culture
2.2. Metabolic Activity Measurement
2.3. Lead Removal Experiments
2.4. Adsorption Kinetics
2.5. Diffusion Model of Crank
2.6. Adsorption Equilibrium Experiments
2.7. Characterization of Adsorbents
2.8. Regeneration
3. Results and Discussion
3.1. Lead Removal Experiments
3.2. Adsorption Kinetics
3.3. Crank Mass Transfer Model
3.4. Adsorption Isotherm
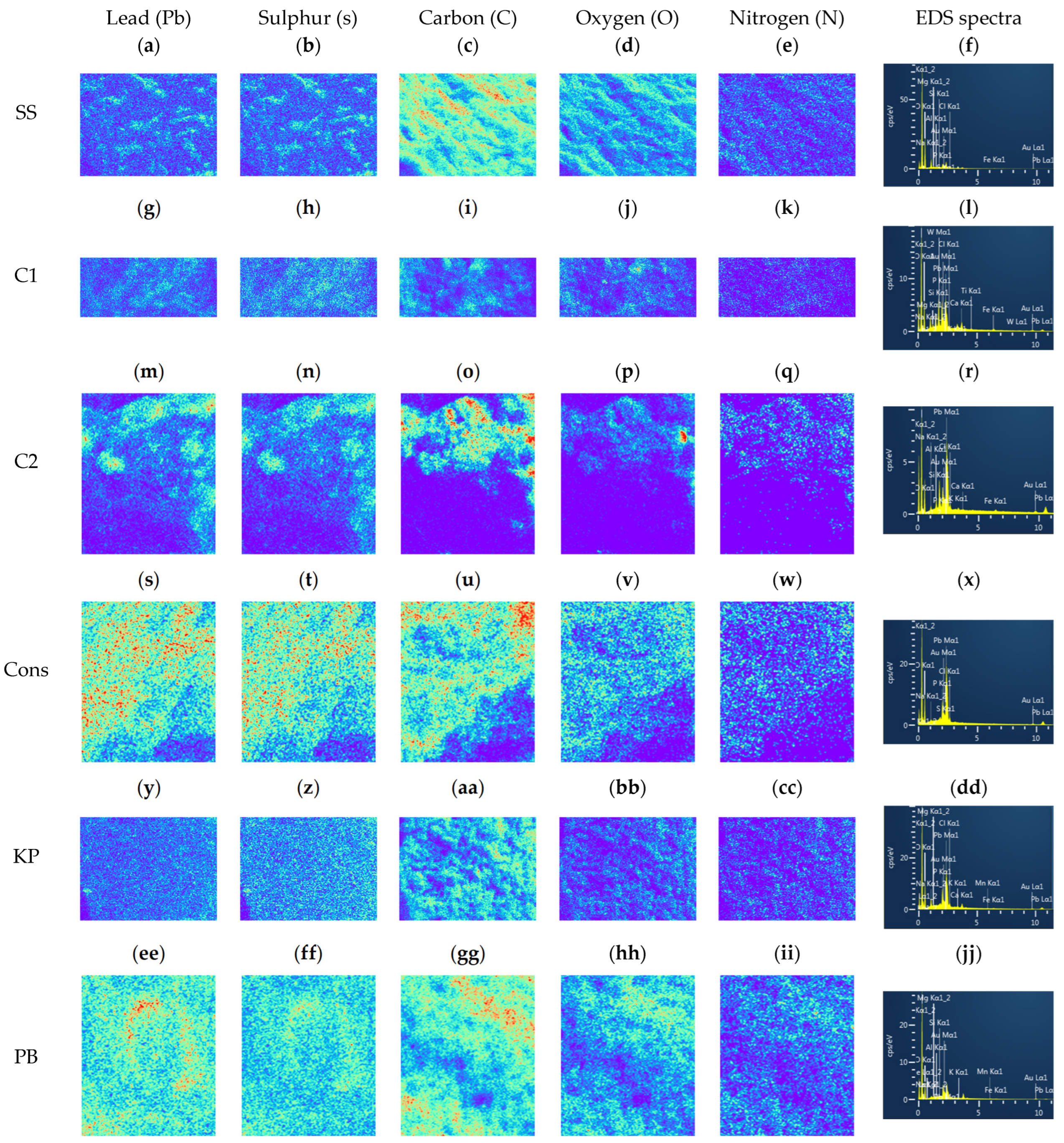
3.5. Characterization of Adsorbents
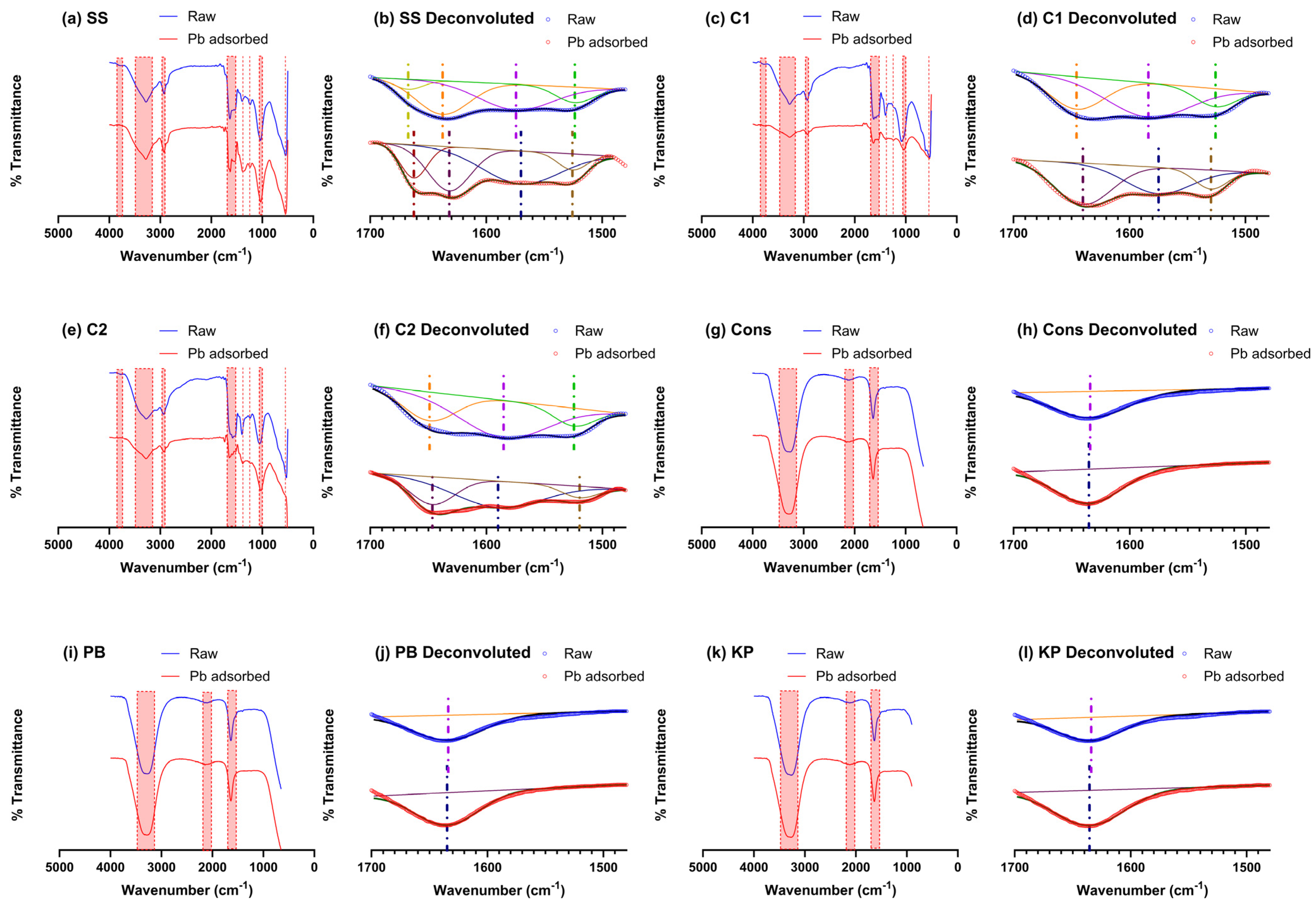
3.6. FTIR Analysis
3.7. Regeneration and Reusability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tiwari, S.; Tripathi, I.P.; Tiwari, H.I. Effects of lead on Environment. Int. J. Emerg. Res. Manag. Technol. 2013, 2, 23–45. [Google Scholar]
- Van Veenhuyzen, B.; Chirwa, E.M.N.; Brink, H.G. Microbial Pb(II) Precipitation: The Role of Biosorption as a Pb(II) Removal Mechanism. Chem. Eng. Trans. 2021, 86, 181–185. [Google Scholar] [CrossRef]
- Mason, L.H.; Harp, J.P.; Han, D.Y. Pb Neurotoxicity: Neuropsychological Effects of Lead Toxicity. Biomed Res. Int. 2014, 2014, 840547. [Google Scholar] [CrossRef] [PubMed]
- Jakubowski, M. Low-level environmental lead exposure and intellectual impairment in children—The current concepts of risk assessment. Int. J. Occup. Med. Environ. Health 2011, 24, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Klingberg, T. Training and plasticity of working memory. Trends Cogn. Sci. 2010, 14, 317–324. [Google Scholar] [CrossRef]
- van Veenhuyzen, B.; Tichapondwa, S.; Hörstmann, C.; Chirwa, E.; Brink, H.G. High capacity Pb(II) adsorption characteristics onto raw- and chemically activated waste activated sludge. J. Hazard. Mater. 2021, 416, 125943. [Google Scholar] [CrossRef]
- Yang, X.; Wan, Y.; Zheng, Y.; He, F.; Yu, Z.; Huang, J.; Wang, H.; Ok, Y.S.; Jiang, Y.; Gao, B. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review. Chem. Eng. J. 2019, 366, 608–621. [Google Scholar] [CrossRef]
- Gupta, A.; Yunus, M.; Sankararamakrishnan, N. Chitosan-and iron–chitosan-coated sand filters: A cost-effective approach for enhanced arsenic removal. Ind. Eng. Chem. Res. 2013, 52, 2066–2072. [Google Scholar] [CrossRef]
- Heidari, P.; Panico, A. Sorption Mechanism and Optimization Study for the Bioremediation of Pb(II) and Cd(II) Contamination by Two Novel Isolated Strains Q3 and Q5 of Bacillus sp. Int. J. Environ. Res. Public Health 2020, 17, 4059. [Google Scholar] [CrossRef]
- Cui, Z.; Zhang, X.; Yang, H.; Sun, L. Bioremediation of heavy metal pollution utilizing composite microbial agent of Mucor circinelloides, Actinomucor sp. and Mortierella sp. J. Environ. Chem. Eng. 2017, 5, 3616–3621. [Google Scholar] [CrossRef]
- Choińska-Pulit, A.; Sobolczyk-Bednarek, J.; Łaba, W. Optimization of copper, lead and cadmium biosorption onto newly isolated bacterium using a Box-Behnken design. Ecotoxicol. Environ. Saf. 2018, 149, 275–283. [Google Scholar] [CrossRef]
- Hörstmann, C.; Brink, H.G.; Chirwa, E.M.N. Pb(II) bio-removal, viability, and population distribution of an industrial microbial consortium: The ediect of Pb(II) and nutrient concentrations. Sustainability 2020, 12, 2511. [Google Scholar] [CrossRef]
- Neveling, O.; Ncube, T.M.C.; Ngxongo, Z.P.; Chirwa, E.M.N.; Brink, H.G. Microbial Precipitation of Pb(II) with Wild Strains of Paraclostridium bifermentans and Klebsiella pneumoniae Isolated from an Industrially Obtained Microbial Consortium. Int. J. Mol. Sci. 2022, 23, 12255. [Google Scholar] [CrossRef]
- Singh, M.; Raorane, C.J.; Alka; Shastri, D.; Raj, V.; Kim, S.-C.; Tuteja, M. Recent Progress on Modified Gum Katira Polysaccharides and Their Various Potential Applications. Polymers 2022, 14, 3648. [Google Scholar] [CrossRef]
- Peens, J.; Wu, Y.W.; Brink, H.G. Microbial Pb(II) precipitation: The influence of elevated Pb(II) Concentrations. Chem. Eng. Trans. 2018, 64, 583–588. [Google Scholar] [CrossRef]
- Tan, K.L.; Hameed, B.H. Insight into the adsorption kinetics models for the removal of contaminants from aqueous solutions. J. Taiwan Inst. Chem. Eng. 2017, 74, 25–48. [Google Scholar] [CrossRef]
- Wang, X.; Liang, X.; Wang, Y.; Wang, X.; Liu, M.; Yin, D.; Xia, S.; Zhao, J.; Zhang, Y. Adsorption of Copper (II) onto activated carbons from sewage sludge by microwave-induced phosphoric acid and zinc chloride activation. Desalination 2011, 278, 231–237. [Google Scholar] [CrossRef]
- Largitte, L.; Pasquier, R. A review of the kinetics adsorption models and their application to the adsorption of lead by an activated carbon. Chem. Eng. Res. Des. 2016, 109, 495–504. [Google Scholar] [CrossRef]
- Boyd, G.E.; Adamson, A.W.; Myers Jr, L.S. The exchange adsorption of ions from aqueous solutions by organic zeolites. II. Kinetics1. J. Am. Chem. Soc. 1947, 69, 2836–2848. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Yang, R.T. Gas Separation by Adsorption Processes; World Scientific: Singapore, 1997; Volume 1, ISBN 1911298100. [Google Scholar]
- Do, D.D. Adsorption Analysis: Equilibria and Kinetics (with Cd Containing Computer MATLAB Programs); World Scientific: Singapore, 1998; Volume 2, ISBN 1783262249. [Google Scholar]
- Weber, T.W.; Chakravorti, R.K. Pore and solid diffusion models for fixed-bed adsorbers. AIChE J. 1974, 20, 228–238. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Bolster, C.H.; Hornberger, G.M. On the Use of Linearized Langmuir Equations. Soil Sci. Soc. Am. J. 2007, 71, 1796–1806. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. chem 1906, 57, 1100–1107. [Google Scholar]
- Berkessa, Y.W.; Mereta, S.T.; Feyisa, F.F. Simultaneous removal of nitrate and phosphate from wastewater using solid waste from factory. Appl. Water Sci. 2019, 9, 28. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.J.; Hosseini-Bandegharaei, A.; Chao, H.P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef]
- Duma, Z.S.; Sihvonen, T.; Havukainen, J.; Reinikainen, V.; Reinikainen, S.P. Optimizing energy dispersive X-Ray Spectroscopy (EDS) image fusion to Scanning Electron Microscopy (SEM) images. Micron 2022, 163, 103361. [Google Scholar] [CrossRef]
- Pearson, K. VII. Note on regression and inheritance in the case of two parents. Proc. R. Soc. London 1895, 58, 240–242. [Google Scholar] [CrossRef]
- Han, J.; Kamber, M.; Pei, J. Getting to Know Your Data. In Data Mining; Elsevier: Amsterdam, The Netherlands, 2012; pp. 39–82. [Google Scholar]
- Foreman, J. Cosine Distance, Cosine Similarity, Angular Cosine Distance, Angular Cosine Similarity. Available online: https://www.itl.nist.gov/div898/software/dataplot/refman2/auxillar/cosdist.htm (accessed on 27 October 2023).
- Goyal, P.; Sharma, P.; Srivastava, S.; Srivastava, M.M. Saraca indica leaf powder for decontamination of Pb: Removal, recovery, adsorbent characterization and equilibrium modeling. Int. J. Environ. Sci. Technol. 2008, 5, 27–34. [Google Scholar] [CrossRef]
- Katsou, E.; Malamis, S.; Tzanoudaki, M.; Haralambous, K.J.; Loizidou, M. Regeneration of natural zeolite polluted by lead and zinc in wastewater treatment systems. J. Hazard. Mater. 2011, 189, 773–786. [Google Scholar] [CrossRef] [PubMed]
- Lazim, Z.M.; Hadibarata, T.; Puteh, M.H.; Yusop, Z. Adsorption characteristics of bisphenol A onto low-cost modified phyto-waste material in aqueous solution. Water Air Soil Pollut. 2015, 226, 34. [Google Scholar] [CrossRef]
- Gupta, A.; Sharma, V.; Sharma, K.; Kumar, V.; Choudhary, S.; Mankotia, P.; Kumar, B.; Mishra, H.; Moulick, A.; Ekielski, A.; et al. A review of adsorbents for heavy metal decontamination: Growing approach to wastewater treatment. Materials 2021, 14, 4702. [Google Scholar] [CrossRef]
- Muedi, K.L.L.; Brink, H.G.G.; Masindi, V.; Maree, J.P.P. Effective removal of arsenate from wastewater using aluminium enriched ferric oxide-hydroxide recovered from authentic acid mine drainage. J. Hazard. Mater. 2021, 414, 125491. [Google Scholar] [CrossRef]
- Wang, J.; Guo, X. Adsorption kinetic models: Physical meanings, applications, and solving methods. J. Hazard. Mater. 2020, 390, 122156. [Google Scholar] [CrossRef]
- Guo, X.; Wang, J. A general kinetic model for adsorption: Theoretical analysis and modeling. J. Mol. Liq. 2019, 288, 111100. [Google Scholar] [CrossRef]
- Vishan, I.; Saha, B.; Sivaprakasam, S.; Kalamdhad, A. Evaluation of Cd (II) biosorption in aqueous solution by using lyophilized biomass of novel bacterial strain Bacillus badius AK: Biosorption kinetics, thermodynamics and mechanism. Environ. Technol. Innov. 2019, 14, 100323. [Google Scholar] [CrossRef]
- Sato, H.; Yui, M.; Yoshikawa, H. Ionic Diffusion Coefficients of Cs−, Pb2+, Sm3+, Ni2+, SeO2-4 and TcO−4 in Free Water Determined from Conductivity Measurements. J. Nucl. Sci. Technol. 1996, 33, 950–955. [Google Scholar] [CrossRef]
- Sick, T. Synthesis and Characterization of Nanoporous Covalent Organic Frameworks for Optoelectronic Applications. Ph.D. Thesis, LMU Munich, Munich, Germany, 2018. [Google Scholar]
- Ruthven, D.M. Principles of Adsorption and Adsorption Processes; John Wiley & Sons: Hoboken, NJ, USA, 1984; ISBN 0471866067. [Google Scholar]
- Kowanga, K.D.; Gatebe, E.; Mauti, G.O.; Mauti, E.M. Kinetic, sorption isotherms, pseudo-first-order model and pseudo-second-order model studies of Cu (II) and Pb (II) using defatted Moringa oleifera seed powder. J. Phytopharm. 2016, 5, 71–78. [Google Scholar] [CrossRef]
- Igwe, J.C.; Abia, A.A. Equilibrium sorption isotherm studies of Cd (II), Pb (II) and Zn (II) ions detoxification from waste water using unmodified and EDTA-modified maize husk. Electron. J. Biotechnol. 2007, 10, 536–548. [Google Scholar] [CrossRef]
- Ge, X.; Wu, Z.; Wu, Z.; Yan, Y.; Cravotto, G.; Ye, B.-C. Microwave-assisted modification of activated carbon with ammonia for efficient pyrene adsorption. J. Ind. Eng. Chem. 2016, 39, 27–36. [Google Scholar] [CrossRef]
- Asuquo, E.; Martin, A.; Nzerem, P.; Siperstein, F.; Fan, X. Adsorption of Cd (II) and Pb (II) ions from aqueous solutions using mesoporous activated carbon adsorbent: Equilibrium, kinetics and characterisation studies. J. Environ. Chem. Eng. 2017, 5, 679–698. [Google Scholar] [CrossRef]
- Hamilton, T.; Huai, Y.; Peng, Y. Lead adsorption on copper sulphides and the relevance to its contamination in copper concentrates. Miner. Eng. 2020, 154, 106381. [Google Scholar] [CrossRef]
- Macías-García, A.; Valenzuela-Calahorro, C.; Espinosa-Mansilla, A.; Bernalte-García, A.; Gómez-Serrano, V. Adsorption of Pb2+ in aqueous solution by SO2-treated activated carbon. Carbon N. Y. 2004, 42, 1755–1764. [Google Scholar] [CrossRef]
- Li, F.; Wang, X.; Yuan, T.; Sun, R. A lignosulfonate-modified graphene hydrogel with ultrahigh adsorption capacity for Pb(II) removal. J. Mater. Chem. A 2016, 4, 11888–11896. [Google Scholar] [CrossRef]
- Mohapatra, R.K.; Parhi, P.K.; Pandey, S.; Bindhani, B.K.; Thatoi, H.; Panda, C.R. Active and passive biosorption of Pb(II)using live and dead biomass of marine bacterium Bacillus xiamenensis PbRPSD202: Kinetics and isotherm studies. J. Environ. Manag. 2019, 247, 121–134. [Google Scholar] [CrossRef]
- Muñoz, A.J.; Espínola, F.; Moya, M.; Ruiz, E. Biosorption of Pb(II) Ions by Klebsiella sp. 3S1 Isolated from a Wastewater Treatment Plant: Kinetics and Mechanisms Studies. Biomed Res. Int. 2015, 2015, 719060. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Kowalczuk, P.B.; Drzymala, J. Physical meaning of the Sauter mean diameter of spherical particulate matter. Part. Sci. Technol. 2016, 34, 645–647. [Google Scholar] [CrossRef]
- Levin, P.A.; Angert, E.R. Small but mighty: Cell size and bacteria. Cold Spring Harb. Perspect. Biol. 2015, 7, a019216. [Google Scholar] [CrossRef] [PubMed]
- Pareek, N.; Gillgren, T.; Jönsson, L.J. Adsorption of proteins involved in hydrolysis of lignocellulose on lignins and hemicelluloses. Bioresour. Technol. 2013, 148, 70–77. [Google Scholar] [CrossRef]
- Gale, M.; Nguyen, T.; Moreno, M.; Gilliard-Abdulaziz, K.L. Physiochemical Properties of Biochar and Activated Carbon from Biomass Residue: Influence of Process Conditions to Adsorbent Properties. ACS Omega 2021, 6, 10224–10233. [Google Scholar] [CrossRef]
- Nekvapil, F.; Mihet, M.; Lazar, G.; Pinzaru, S.C.; Gavrilović, A.; Ciorîță, A.; Levei, E.; Tamaș, T.; Soran, M.L. Comparative Analysis of Composition and Porosity of the Biogenic Powder Obtained from Wasted Crustacean Exoskeletonsafter Carotenoids Extraction for the Blue Bioeconomy. Water 2023, 15, 2591. [Google Scholar] [CrossRef]
- Venkatesan, S.; Pugazhendy, K.; Sangeetha, D.; Vasantharaja, C.; Prabakaran, S.; Meenambal, M. Fourier transform infrared (FT-IR) spectoroscopic analysis of Spirulina. Int. J. Pharm. Biol. Arch 2012, 3, 969–972. [Google Scholar]
- Muruganantham, S.; Anbalagan, G.; Ramamurthy, N. FT-IR and SEM-EDS comparative analysis of medicinal plants, Eclipta alba Hassk and Eclipta prostrata Linn. Rom. J. Biophys. 2009, 19, 285–294. [Google Scholar]
- Tipson, R.S. Infrared Spectroscopy of Carbohydrates: A Review of the Literature; National Institute of Standards and Technology: Gaithersburg, MD, USA, 1968.
- Lingegowda, D.C.; Kumar, J.K.; Prasad, A.D.; Zarei, M.; Gopal, S. FTIR spectroscopic studies on cleome gynandra-Comparative analysis of functional group before and after extraction. Rom. J. Biophys 2012, 22, 137–143. [Google Scholar]
- Sitko, R.; Turek, E.; Zawisza, B.; Malicka, E.; Talik, E.; Heimann, J.; Gagor, A.; Feist, B.; Wrzalik, R. Adsorption of divalent metal ions from aqueous solutions using graphene oxide. Dalt. Trans. 2013, 42, 5682–5689. [Google Scholar] [CrossRef]
- Alizadeh, B.; Ghorbani, M.; Salehi, M.A. Application of polyrhodanine modified multi-walled carbon nanotubes for high efficiency removal of Pb(II) from aqueous solution. J. Mol. Liq. 2016, 220, 142–149. [Google Scholar] [CrossRef]
- Li, K.; Cao, J.; Li, H.; Liu, J.; Lu, M.; Tang, D. Nitrogen functionalized hierarchical microporous/mesoporous carbon with a high surface area and controllable nitrogen content for enhanced lead(II) adsorption. RSC Adv. 2016, 6, 92186–92196. [Google Scholar] [CrossRef]
- Salimon, J.; Abdullah, B.M.; Salih, N. Hydrolysis optimization and characterization study of preparing fatty acids from Jatropha curcas seed oil. Chem. Cent. J. 2011, 5, 67. [Google Scholar] [CrossRef] [PubMed]
- Francioso, O.; Rodriguez-Estrada, M.T.; Montecchio, D.; Salomoni, C.; Caputo, A.; Palenzona, D. Chemical characterization of municipal wastewater sludges produced by two-phase anaerobic digestion for biogas production. J. Hazard. Mater. 2010, 175, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Alessi, D.S.; Owttrim, G.W.; Kenney, J.P.L.; Zhou, Q.; Lalonde, S.V.; Konhauser, K.O. Cell surface acid-base properties of the cyanobacterium Synechococcus: Influences of nitrogen source, growth phase and N:P ratios. Geochim. Cosmochim. Acta 2016, 187, 179–194. [Google Scholar] [CrossRef]
- Yang, X.; Cui, X. Adsorption characteristics of Pb (II) on alkali treated tea residue. Water Resour. Ind. 2013, 3, 1–10. [Google Scholar] [CrossRef]
- Atieh, M.A.; Bakather, O.Y.; Al-Tawbini, B.; Bukhari, A.A.; Abuilaiwi, F.A.; Fettouhi, M.B. Effect of carboxylic functional group functionalized on carbon nanotubes surface on the removal of lead from water. Bioinorg. Chem. Appl. 2010, 2010, 603978. [Google Scholar] [CrossRef]
- Sheng, P.X.; Ting, Y.P.; Chen, J.P.; Hong, L. Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: Characterization of biosorptive capacity and investigation of mechanisms. J. Colloid Interface Sci. 2004, 275, 131–141. [Google Scholar] [CrossRef]
- Muhaisen, L.F. Nickel Ions Removal From Aqueous Solutions Using Sawdust As Adsorbent: Equilibrium, Kinetic and Thermodynamic Studies. J. Eng. Sustain. Dev. 2017, 21, 60–71. [Google Scholar]
- Ryu, S.R.; Noda, I.; Jung, Y.M. What is the origin of positional fluctuation of spectral features: True frequency shift or relative intensity changes of two overlapped bands? Appl. Spectrosc. 2010, 64, 1017–1021. [Google Scholar] [CrossRef]
- Tao, Y.; Zhang, C.; Lü, T.; Zhao, H. Removal of Pb (II) ions from wastewater by using polyethyleneimine-functionalized Fe3O4 magnetic nanoparticles. Appl. Sci. 2020, 10, 948. [Google Scholar] [CrossRef]
- Hu, X.; Cao, J.; Yang, H.; Li, D.; Qiao, Y.; Zhao, J.; Zhang, Z.; Huang, L. Pb2+ biosorption from aqueous solutions by live and dead biosorbents of the hydrocarbon-degrading strain Rhodococcus sp. HX-2. PLoS ONE 2020, 15, e0226557. [Google Scholar] [CrossRef]
- Selatnia, A.; Boukazoula, A.; Kechid, N.; Bakhti, M.Z.; Chergui, A.; Kerchich, Y. Biosorption of lead (II) from aqueous solution by a bacterial dead Streptomyces rimosus biomass. Biochem. Eng. J. 2004, 19, 127–135. [Google Scholar] [CrossRef]
- He, Y.; Wu, P.; Xiao, W.; Li, G.; Yi, J.; He, Y.; Chen, C.; Ding, P.; Duan, Y. Efficient removal of Pb (II) from aqueous solution by a novel ion imprinted magnetic biosorbent: Adsorption kinetics and mechanisms. PLoS ONE 2019, 14, e0213377. [Google Scholar] [CrossRef] [PubMed]
- Ifthikar, J.; Wang, T.; Khan, A.; Jawad, A.; Sun, T.; Jiao, X.; Chen, Z.; Wang, J.; Wang, Q.; Wang, H.; et al. Highly Efficient Lead Distribution by Magnetic Sewage Sludge Biochar: Sorption Mechanisms and Bench Applications. Bioresour. Technol. 2017, 238, 399–406. [Google Scholar] [CrossRef]
- Wang, S.; Guo, W.; Gao, F.; Yang, R. Characterization and Pb(II) removal potential of corn straw- and municipal sludge-derived biochars. R. Soc. Open Sci. 2017, 4, 170402. [Google Scholar] [CrossRef] [PubMed]
- Rozada, F.; Otero, M.; Morán, A.; García, A.I. Adsorption of heavy metals onto sewage sludge-derived materials. Bioresour. Technol. 2008, 99, 6332–6338. [Google Scholar] [CrossRef] [PubMed]
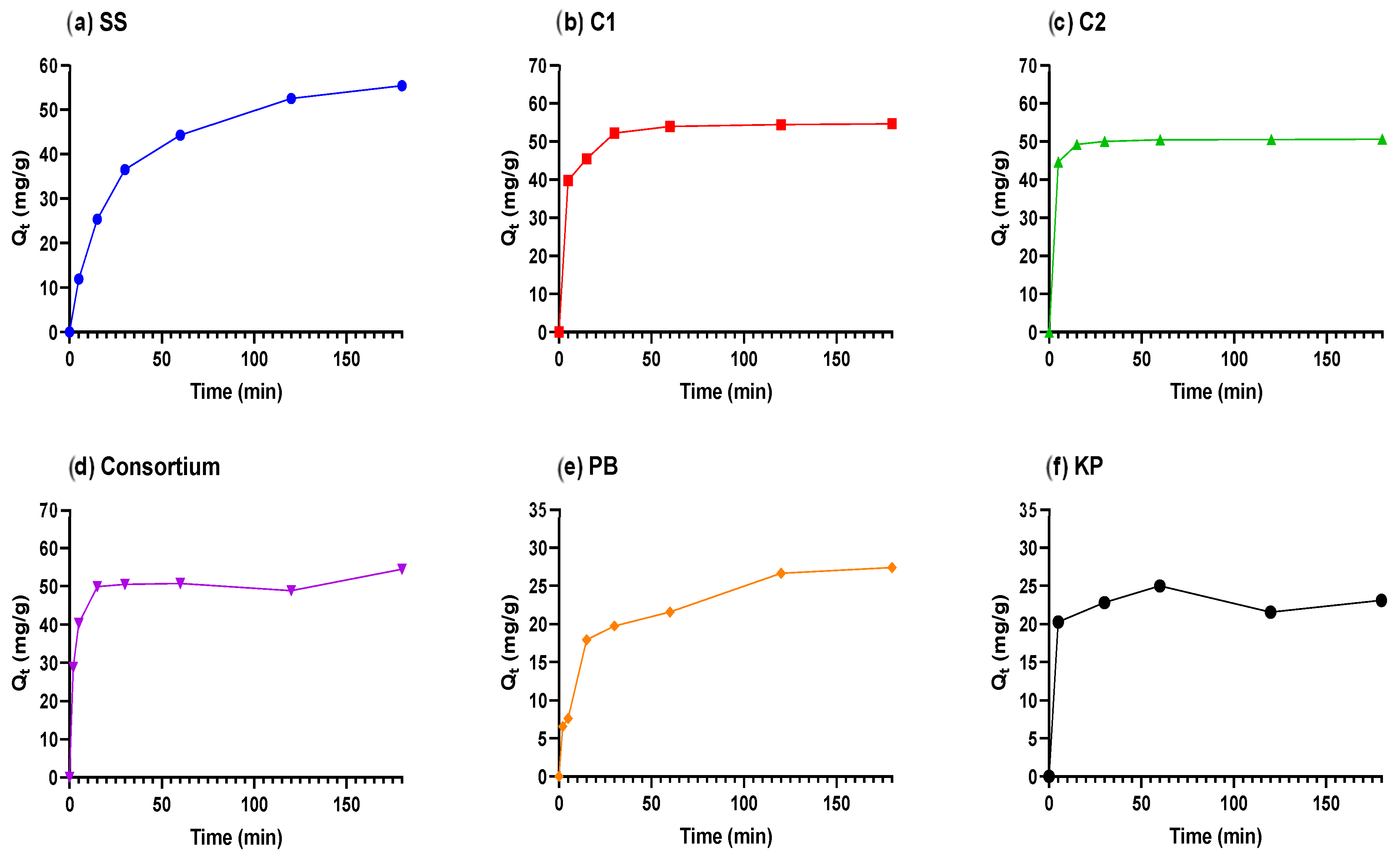
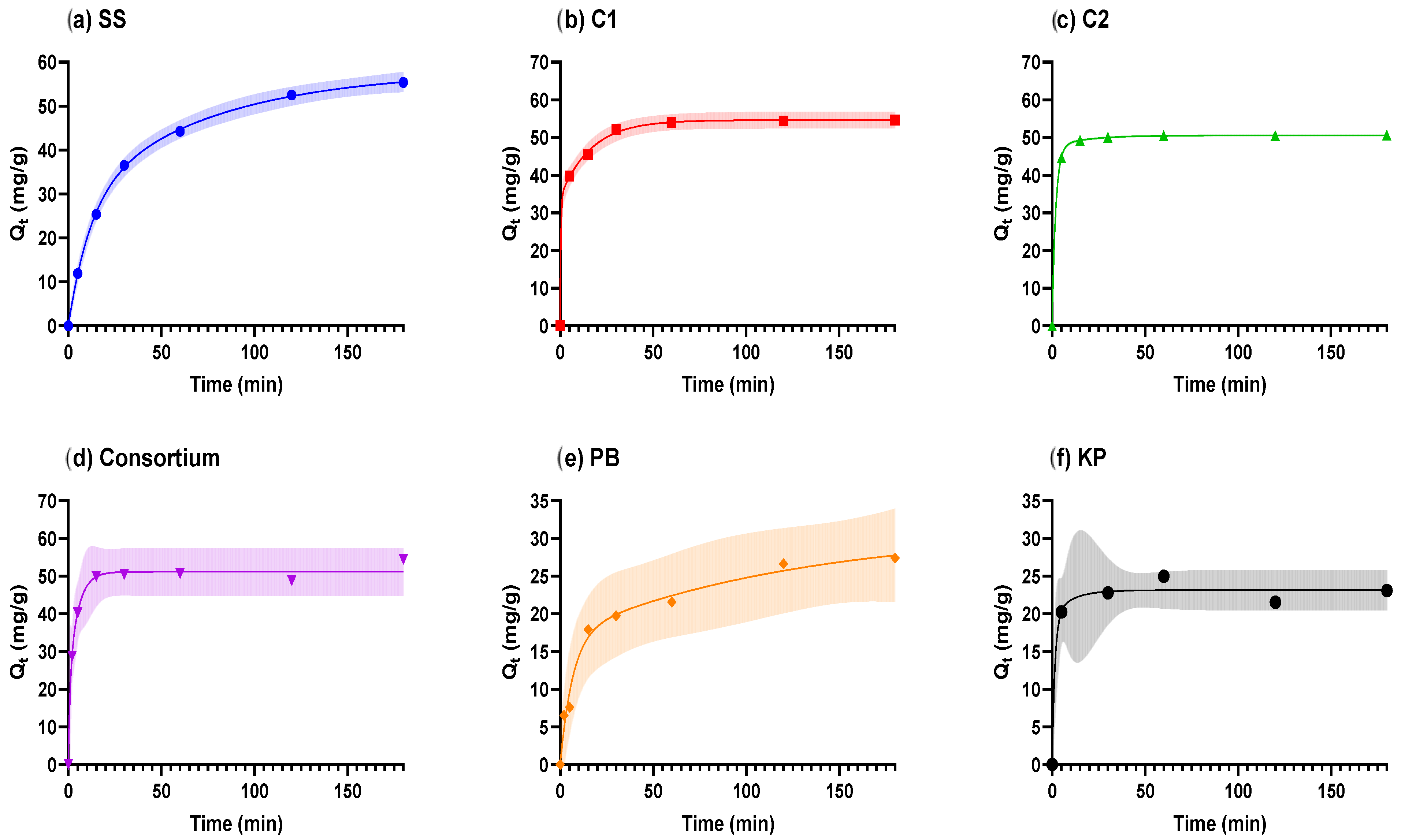



| Adsorbents | SS | C1 | C2 | Cons | PB | KP |
|---|---|---|---|---|---|---|
| Two-Phase Pseudo-First-Order | ||||||
| (mg/g) | 58.04 | 54.62 | 50.55 | 51.16 | 31.15 | 23.14 |
| PercentFast | 48.71 | 62.60 | 94.02 | 40.31 | 54.99 | 84.33 |
| mg/g) | 28.27 | 34.19 | 47.53 | 20.62 | 17.13 | 19.51 |
| (mg/g) | 29.77 | 20.43 | 3.02 | 30.54 | 14.02 | 3.63 |
| (1/min) | 0.084 | 2.491 | 0.513 | 1.113 | 0.149 | 0.662 |
| (1/min) | 0.014 | 0.060 | 0.057 | 0.208 | 0.008 | 0.102 |
| 0.999 | 0.999 | 1 | 0.993 | 0.985 | 0.986 | |
| SSE | 0.800 | 1.835 | 0.013 | 16.53 | 10.33 | 5.946 |
| Sy.x | 0.516 | 0.677 | 0.066 | 2.033 | 1.607 | 1.408 |
| RMSE | 0.365 | 0.553 | 0.046 | 1.537 | 1.215 | 1.09 |
| Pseudo-Second-Order | ||||||
| (mg/g) | 61.58 | 55.13 | 51.01 | 52.82 | 27.94 | 23.33 |
| (g/mg·min) | 0.0008 | 0.0009 | 0.0284 | 0.0120 | 0.0034 | 0.0589 |
| 0.999 | 0.996 | 0.999 | 0.991 | 0.977 | 0.985 | |
| SSE | 1.781 | 10.47 | 0.372 | 20.52 | 16.25 | 6.408 |
| Sy.x | 0.597 | 1.447 | 0.273 | 1.849 | 1.646 | 1.266 |
| RMSE | 0.545 | 1.321 | 0.249 | 1.712 | 1.524 | 1.132 |
| Pseudo-First-Order | ||||||
| (mg/g) | 52.96 | 52.53 | 50.17 | 50.71 | 24.92 | 23.10 |
| (1/min) | 0.040 | 0.264 | 0.439 | 0.373 | 0.075 | 0.420 |
| 0.989 | 0.979 | 0.999 | 0.988 | 0.949 | 0.986 | |
| SSE | 28.51 | 48.88 | 1.147 | 28.88 | 35.79 | 6.002 |
| Sy.x | 2.388 | 3.127 | 0.479 | 2.194 | 2.442 | 1.225 |
| RMSE | 2.18 | 2.854 | 0.437 | 2.031 | 2.261 | 1.096 |
| Crank Mass Transfer Model | ||||||
| 58.16 | 53.42 | 50.21 | 51.18 | 27 | 23.10 | |
| k | 2.6 × 10−5 | 2.4 × 10−4 | 5.7 × 10−4 | 3.5 × 10−4 | 4.5 × 10−5 | 5.4 × 10−4 |
| 2.6 × 10−13 | 2.4 × 10−12 | 5.7 × 10−12 | 3.5 × 10−12 | 4.5 × 10−13 | 5.4 × 10−12 | |
| 0.992 | 0.988 | 0.999 | 0.993 | 0.977 | 0.986 | |
| SSE | 22.11 | 28.28 | 0.947 | 16.68 | 16.63 | 6.002 |
| Sy.x | 2.103 | 2.378 | 0.435 | 1.667 | 1.665 | 1.225 |
| RMSE | 1.92 | 2.171 | 0.397 | 1.544 | 1.541 | 1.096 |
| Adsorbents | SS | C1 | C2 | Cons | PB | KP |
|---|---|---|---|---|---|---|
| Two-Surface Langmuir | ||||||
| (mg/g) | 72.46 | 86.07 | 52.61 | 45.82 | 46.13 | |
| (mg/g) | 83.83 | 144.20 | 156.70 | 221.40 | 430.80 | 223.60 |
| L/mg) | ∞ | ∞ | ∞ | ∞ | ∞ | ∞ |
| L/mg) | 0.024 | 0.035 | 0.022 | 0.025 | 0.0013 | 0.015 |
| 0.827 | 0.833 | 0.737 | 0.788 | 0.648 | 0.664 | |
| SSE | 7446 | 23818 | 33143 | 26332 | 19166 | 47731 |
| Sy.x | 13.31 | 21.61 | 25.49 | 25.98 | 21.36 | 30.59 |
| RMSE | 13.01 | 21.20 | 25.01 | 25.34 | 20.87 | 30.01 |
| Langmuir | ||||||
| (mg/g) | 141.20 | 208.50 | 193.80 | 220.40 | 153.20 | 217.70 |
| (L/mg) | 3.68 | 0.258 | 0.058 | 0.025 | 0.022 | 0.043 |
| 0.003–0.0005 | 0.04–0.006 | 0.145–0.028 | 0.29–0.064 | 0.48–0.11 | 0.32–0.06 | |
| 0.648 | 0.644 | 0.681 | 0.788 | 0.350 | 0.568 | |
| SSE | 15116 | 52666 | 40210 | 26282 | 35371 | 61358 |
| Sy.x | 18.75 | 31.82 | 27.81 | 25.63 | 28.68 | 34.03 |
| RMSE | 18.54 | 31.52 | 27.54 | 25.32 | 28.35 | 34.03 |
| Freundlich | ||||||
| (L/mg) | 82.96 | 89.76 | 54.55 | 33.39 | 21.17 | 34.39 |
| n | 10.11 | 6.172 | 4.516 | 3.295 | 2.973 | 2.804 |
| 0.802 | 0.826 | 0.707 | 0.703 | 0.496 | 0.628 | |
| SSE | 8504 | 25686 | 36942 | 36805 | 27459 | 52938 |
| Sy.x | 14.06 | 22.23 | 26.65 | 30.33 | 25.27 | 31.91 |
| RMSE | 13.90 | 22.01 | 26.40 | 29.96 | 24.98 | 31.60 |
| Adsorbents | BET Surface Area (m2/g) | Sauter Equivalent Diameter (μm) |
|---|---|---|
| SS | 0.20 | 31 |
| C1 | 0.083 | 72 |
| C2 | 0.021 | 290 |
| Cons | 0.065 | 92 |
| PB | 0.025 | 240 |
| KP | 0.017 | 350 |
| Wavenumber (1/cm) | Bond | Functional Group | Adsorbent | Reference |
|---|---|---|---|---|
| 553 | C–C, C–N, -CH2 | C–C and C–N ring deformation; CH2 wagging and rocking | SS, C1, C2 | [59] |
| 1034–1080 | C–O–C, S=O | Ethers, symmetric stretching of S=O | SS, C1, C2 | [50,60,61] |
| 1240 | C=O, C=S | Alkyl ketone, thioketone stretching | SS, C1, C2 | [62] |
| 1375 | -CH2, S=O | CH2 wagging, sulphonic ester stretching | SS, C1, C2 | [59,61] |
| 1525 | C=O | Stretching vibration in ketones and esters | SS, C1, C2 | [62] |
| 1575 | C=C | Stretching vibration in alkenes | SS, C1, C2 | [62,63] |
| 1635 | C=O, C–C, N–H, C=N, C–N, | Stretching vibration in aldehydes and ketones. N –H vibration coupled with C=N bending vibration or C–N stretching | SS, C1, C2, Cons, PB, KP | [50,62,64,65] |
| 1665 | C=O, C=S | Stretching vibration in ketones, aldehydes, and esters/thioesters | SS | [61,62] |
| 2000-2200 | C≡C, C≡N, C=S | Alkynes, nitrile, isothiocyanate asymmetric stretching | Cons, PB, KP | [61,66] |
| 2924–2934 | -CH2 | Stretching vibration of C–H | SS, C1, C2 | [62,65] |
| Circa 3400 | C–OH | Hydroxyl group | SS, C1, C2, Cons, PB, KP | [62,65] |
| Circa 3790 | O–H stretching | Alcohol | SS, C1, C2 | [66] |
| Adsorbents | Pb(II) Desorbed 1st Cycle | Pb(II) Desorbed 2nd Cycle | ||
|---|---|---|---|---|
| (%) | (mg/g) | (%) | (mg/g) | |
| SS | 72.35 | 64.97 | 14.03 | 13.45 |
| C1 | 68.62 | 70.64 | 2.32 | 3.26 |
| C2 | 69.73 | 71.46 | 3.35 | 4.08 |
| Cons | 69.58 | 66.76 | 0.87 | 2.11 |
| PB | 60.99 | 50.16 | 0.74 | 2.43 |
| KP | 72.38 | 79.53 | 4.94 | 12.32 |
| Adsorbent Description | (mg/g) | Reference |
|---|---|---|
| Rhodococcus sp. HX-2 | 88.74 | [75] |
| Streptomyces rimosus | 135 | [76] |
| Ion-imprinted magnetic biosorbent | 116.28 | [77] |
| Magnetic sewage sludge biochar | 99.90 | [78] |
| Sludge-derived biochar | 40.80 | [79] |
| Pyrolyzed sewage sludge | 40.30 | [80] |
| Raw WAS | 307 | [6] |
| -activated WAS | 274 | [6] |
| SS | 141.20 | This study |
| C1 | 208.50 | This study |
| C2 | 193.80 | This study |
| Cons | 220.40 | This study |
| PB | 153.20 | This study |
| KP | 217.70 | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kpai, P.Y.; Nel, J.; Haneklaus, N.; Chirwa, E.M.N.; Brink, H.G. Comparative Screening Study on the Adsorption of Aqueous Pb(II) Using Different Metabolically Inhibited Bacterial Cultures from Industry. Water 2023, 15, 4259. https://doi.org/10.3390/w15244259
Kpai PY, Nel J, Haneklaus N, Chirwa EMN, Brink HG. Comparative Screening Study on the Adsorption of Aqueous Pb(II) Using Different Metabolically Inhibited Bacterial Cultures from Industry. Water. 2023; 15(24):4259. https://doi.org/10.3390/w15244259
Chicago/Turabian StyleKpai, Patrick Y., Jaco Nel, Nils Haneklaus, Evans M. N. Chirwa, and Hendrik G. Brink. 2023. "Comparative Screening Study on the Adsorption of Aqueous Pb(II) Using Different Metabolically Inhibited Bacterial Cultures from Industry" Water 15, no. 24: 4259. https://doi.org/10.3390/w15244259
APA StyleKpai, P. Y., Nel, J., Haneklaus, N., Chirwa, E. M. N., & Brink, H. G. (2023). Comparative Screening Study on the Adsorption of Aqueous Pb(II) Using Different Metabolically Inhibited Bacterial Cultures from Industry. Water, 15(24), 4259. https://doi.org/10.3390/w15244259











