Photocatalytic Degradation of Methylene Blue from Aqueous Solutions by Using Nano-ZnO/Kaolin-Clay-Based Nanocomposite
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
2.2.1. Synthesis of Zinc Oxide Nanoparticles
2.2.2. Development of Clay Nanocomposite
2.3. Preparation of Methylene Blue Dye Aqueous Solution and Dye Removal
3. Characterization of Nanoparticles and Nanocomposites
4. Results and Discussion
4.1. Chemical Composition of Garlic Peel Extracts
4.2. Mechanism of Formation of ZnONP/Kaolin Composite
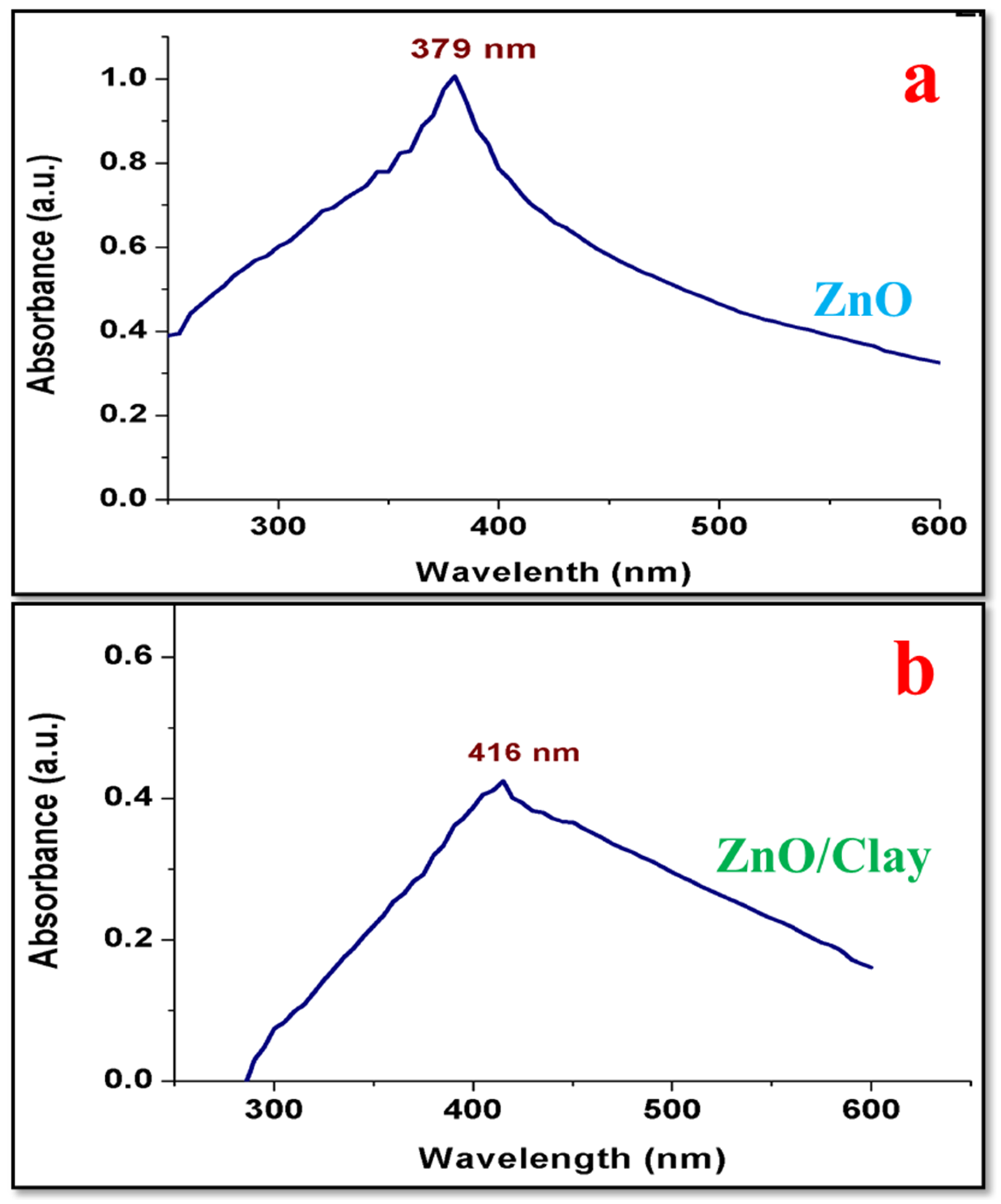
4.3. UV–Visible Spectroscopy
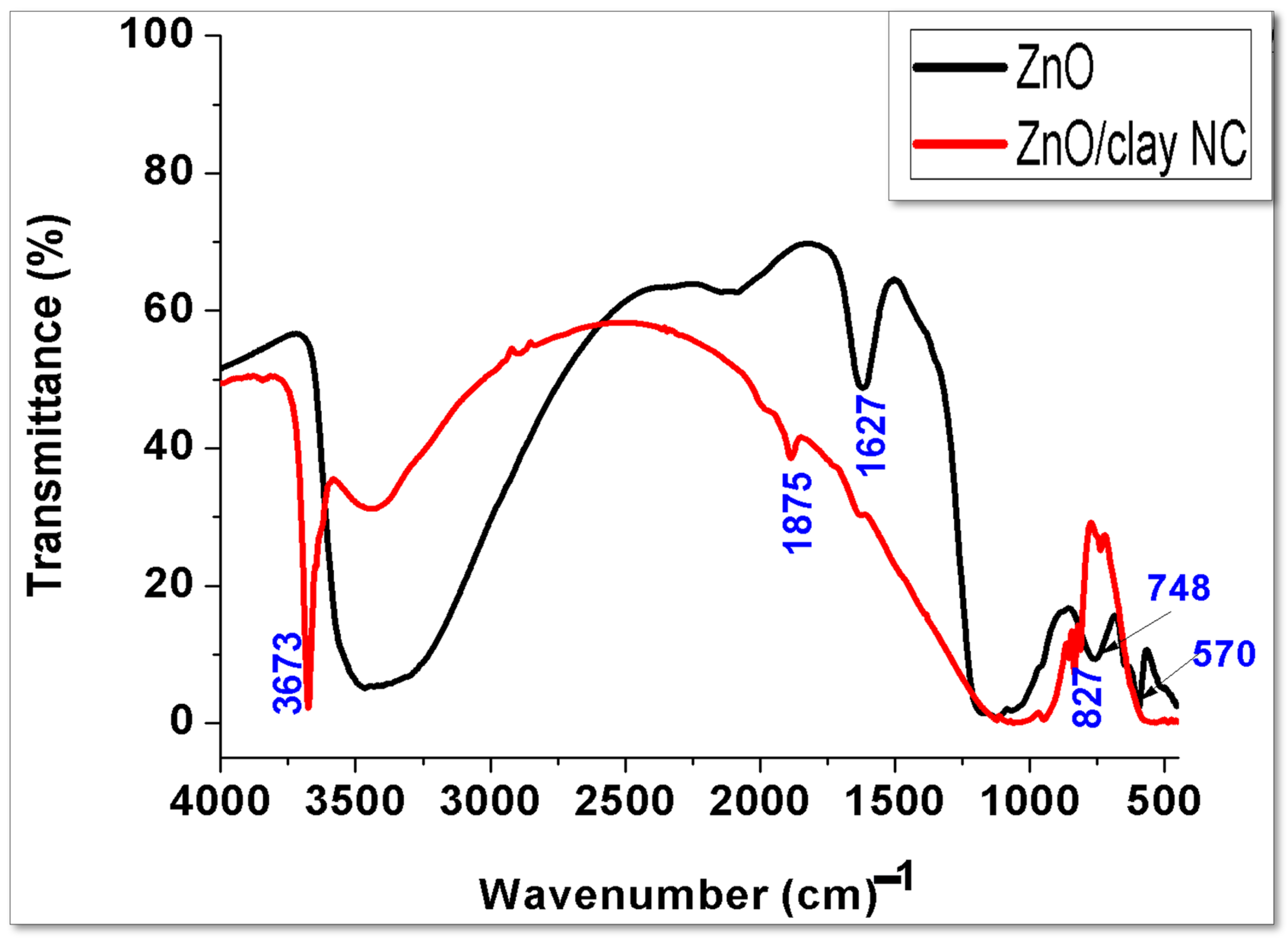
4.4. FTIR Analysis of ZnONPs and ZnO/Clay NC
4.5. Phase Identification of ZnONPs, Kaolin, and Clay NC by XRD
4.6. Morphological Analysis of NC by TEM
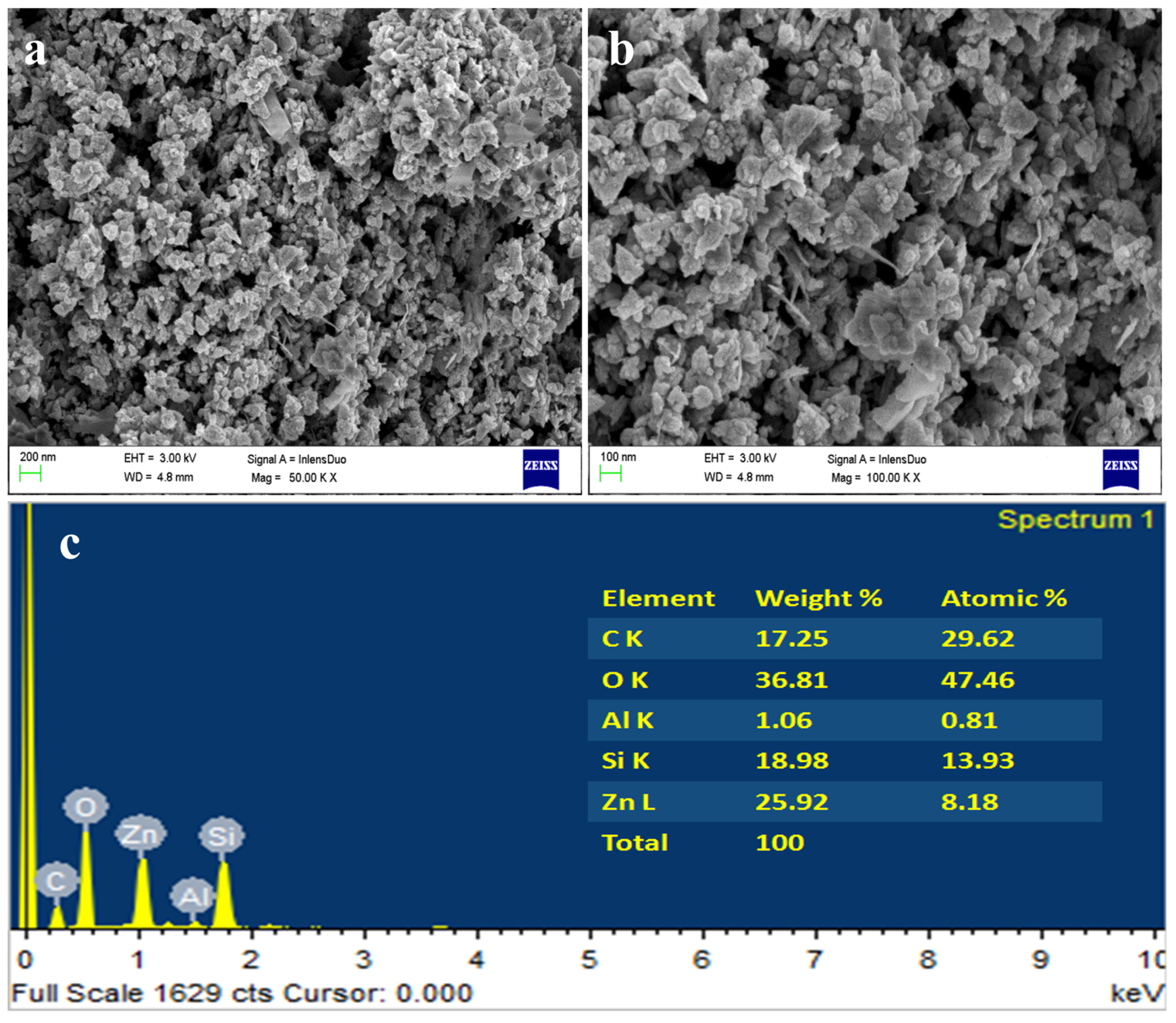
4.7. Morphological Analysis of NC by FESEM
4.8. Photocatalytic Degradation of Dye
4.8.1. Optimization of Variables in Photocatalytic Degradation
4.8.2. Effect of Catalyst Dose
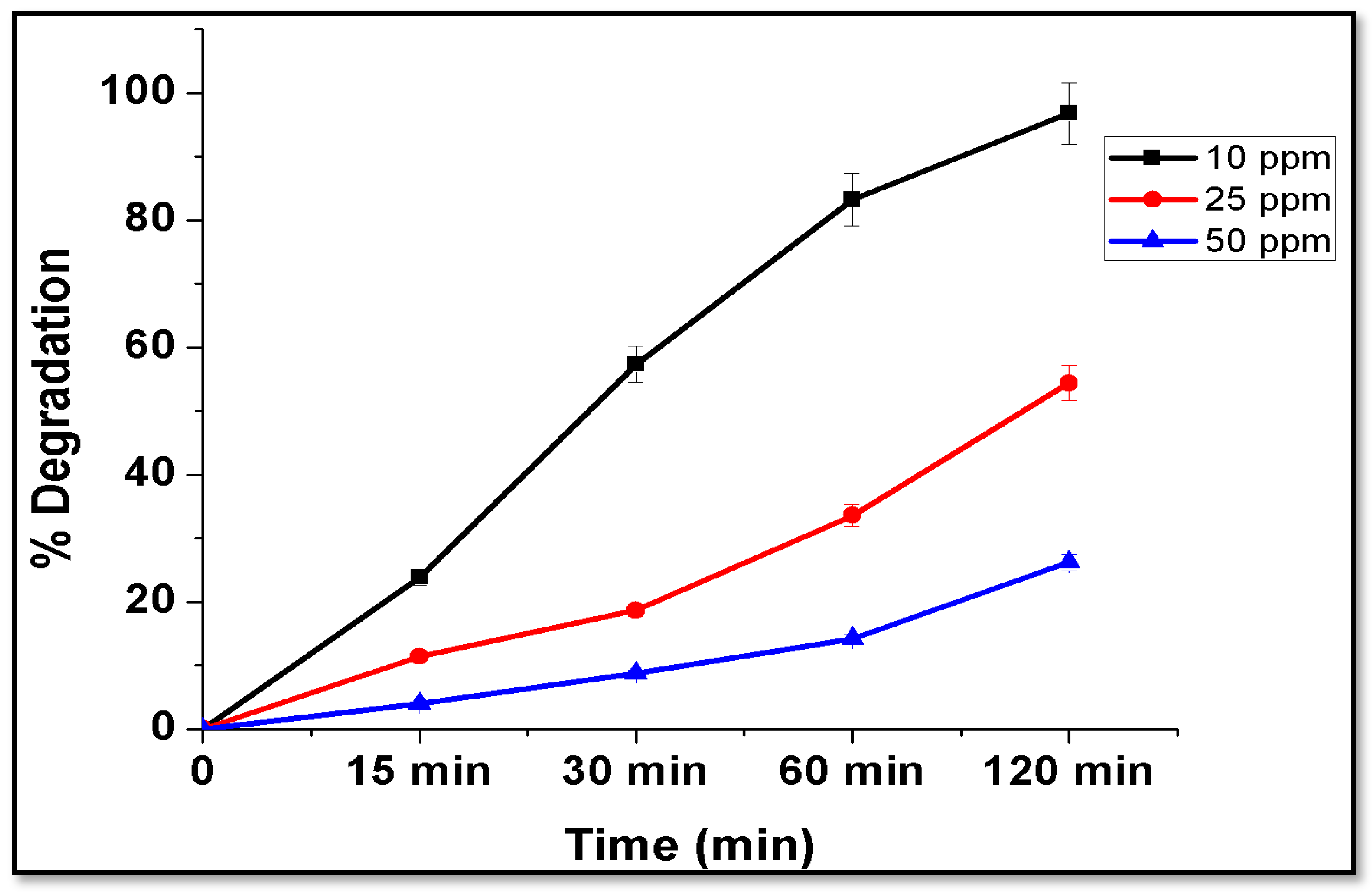
4.8.3. Effect of Initial Concentration of Dye
4.8.4. Effect of pH
4.8.5. Photocatalytic Activity and Mechanism
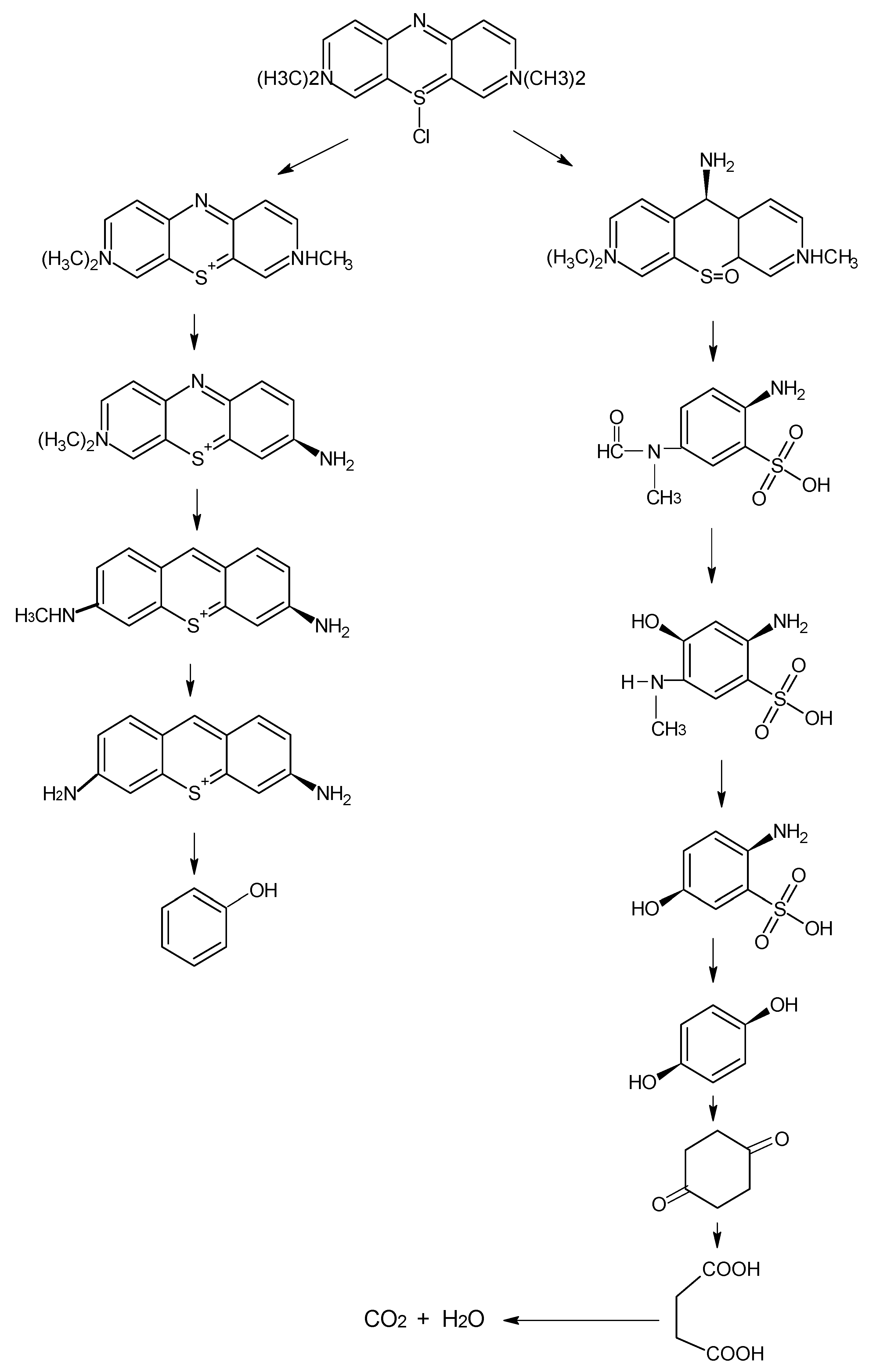
4.8.6. Chromophoric Group Degradation: First Degradation Pathway
4.8.7. Auxochrome Group Degradation: Second Degradation Pathway
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Islam, T.; Repon, M.R.; Islam, T.; Sarwar, Z.; Rahman, M.M. Impact of Textile Dyes on Health and Ecosystem: A Review of Structure, Causes, and Potential Solutions. Environ. Sci. Pollut. Res. 2023, 30, 9207–9242. [Google Scholar] [CrossRef] [PubMed]
- Al-Tohamy, R.; Ali, S.S.; Li, F.; Okasha, K.M.; Mahmoud, Y.A.-G.; Elsamahy, T.; Jiao, H.; Fu, Y.; Sun, J. A Critical Review on the Treatment of Dye-Containing Wastewater: Ecotoxicological and Health Concerns of Textile Dyes and Possible Remediation Approaches for Environmental Safety. Ecotoxicol. Environ. Saf. 2022, 231, 113160. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.B.; Thombre, N.V. Coagulation, Flocculation, and Precipitation in Water and Used Water Purification. In Handbook of Water and Used Water Purification; Lahnsteiner, J., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–25. ISBN 978-3-319-66382-1. [Google Scholar]
- Sreelekshmi, P.B.; Pillai, R.R.; Unnimaya, S.; Anju, A.L.; Meera, A.P. Biofabrication of Novel ZnO Nanoparticles for Efficient Photodegradation of Industrial Dyes. Clean. Technol. Environ. Policy 2023. [Google Scholar] [CrossRef]
- Choudhary, N.; Chaudhari, J.; Mochi, V.; Patel, P.; Ali, D.; Alarifi, S.; Sahoo, D.K.; Patel, A.; Kumar Yadav, V. Phytonanofabrication of Copper Oxide by Albizia Saman and Their Potential as an Antimicrobial Agent and Remediation of Congo Red Dye from Wastewater. Water 2023, 15, 3787. [Google Scholar] [CrossRef]
- Gorbatsevich, O.B.; Kholodkov, D.N.; Kurkin, T.S.; Malakhova, Y.N.; Strel’tsov, D.R.; Buzin, A.I.; Kazakova, V.V.; Muzafarov, A.M.; Enikolopov, N.S. Synthesis and Properties of Watersoluble Silica Nanoparticles. Russ. Chem. Bull. 2017, 66, 409–417. [Google Scholar] [CrossRef]
- Mohan, A.; Jaison, A.; Lee, Y.-C. Emerging Trends in Mesoporous Silica Nanoparticle-Based Catalysts for CO2 Utilization Reactions. Inorg. Chem. Front. 2023, 10, 3171–3194. [Google Scholar] [CrossRef]
- Tang, T.; Zhou, M.; Lv, J.; Cheng, H.; Wang, H.; Qin, D.; Hu, G.; Liu, X. Sensitive and Selective Electrochemical Determination of Uric Acid in Urine Based on Ultrasmall Iron Oxide Nanoparticles Decorated Urchin-like Nitrogen-Doped Carbon. Colloids Surf. B Biointerfaces 2022, 216, 112538. [Google Scholar] [CrossRef]
- VafaeiAsl, M.; Keshavarz, I.; Shemirani, F.; Jamshidi, P. Green Synthesis of a Novel Magnetic Fe3O4@SiO2/TiO2@WO3 Nanocomposite for Methylene Blue Removal under UV and Visible Light Irradiations. Res. Chem. Intermed. 2023, 49, 1909–1924. [Google Scholar] [CrossRef]
- Liang, Y.; Li, J.; Xue, Y.; Tan, T.; Jiang, Z.; He, Y.; Shangguan, W.; Yang, J.; Pan, Y. Benzene Decomposition by Non-Thermal Plasma: A Detailed Mechanism Study by Synchrotron Radiation Photoionization Mass Spectrometry and Theoretical Calculations. J. Hazard. Mater. 2021, 420, 126584. [Google Scholar] [CrossRef]
- Sun, S.; Liu, H.; Zhang, J.; Wang, W.; Xu, P.; Zhu, X.; Wang, Y.; Wan, S. Application of a Novel Coagulant in Reservoir Water Treatment in Qingdao. Desalination Water Treat. 2023, 284, 49–60. [Google Scholar] [CrossRef]
- Ansari, M.A.H.; Khan, M.E.; Mohammad, A.; Baig, M.T.; Chaudary, A.; Tauqeer, M. 15—Application of Nanocomposites in Wastewater Treatment. In Nanocomposites-Advanced Materials for Energy and Environmental Aspects; Khan, M.E., Aslam, J., Verma, C., Eds.; Woodhead Publishing: Sawston, UK, 2023; pp. 297–319. ISBN 978-0-323-99704-1. [Google Scholar]
- Zhang, J.; Zhong, A.; Huang, G.; Yang, M.; Li, D.; Teng, M.; Han, D. Enhanced Efficiency with CDCA Co-Adsorption for Dye-Sensitized Solar Cells Based on Metallosalophen Complexes. Sol. Energy 2020, 209, 316–324. [Google Scholar] [CrossRef]
- Li, H.; Si, S.; Yang, K.; Mao, Z.; Sun, Y.; Cao, X.; Yu, H.; Zhang, J.; Ding, C.; Liang, H.; et al. Hexafluoroisopropanol Based Silk Fibroin Coatings on AZ31 Biometals with Enhanced Adhesion, Corrosion Resistance and Biocompatibility. Prog. Org. Coat. 2023, 184, 107881. [Google Scholar] [CrossRef]
- Yan, F.; Jiang, J.; Li, K.; Liu, N.; Chen, X.; Gao, Y.; Tian, S. Green Synthesis of Nanosilica from Coal Fly Ash and Its Stabilizing Effect on CaO Sorbents for CO2 Capture. Environ. Sci. Technol. 2017, 51, 7606–7615. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, C.; Liu, H.; Hrynshpan, D.; Savitskaya, T.; Chen, J.; Chen, J. Enhanced Denitrification Performance of Alcaligenes SpTB by Pd Stimulating to Produce Membrane Adaptation Mechanism Coupled with Nanoscale Zero-Valent Iron. Sci. Total Environ. 2020, 708, 135063. [Google Scholar] [CrossRef] [PubMed]
- Baig, N.; Kammakakam, I.; Falath, W. Nanomaterials: A Review of Synthesis Methods, Properties, Recent Progress, and Challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar] [CrossRef]
- Tariq, A.; Bhawani, S.A.; Asaruddin, M.R.; Alotaibi, K.M. 2—Introduction to Nanocomposites. In Polysaccharide-Based Nanocomposites for Gene Delivery and Tissue Engineering; Bhawani, S.A., Karim, Z., Jawaid, M., Eds.; Woodhead Publishing: Sawston, UK, 2021; pp. 15–37. ISBN 978-0-12-821230-1. [Google Scholar]
- Chen, D.; Wang, Q.; Li, Y.; Li, Y.; Zhou, H.; Fan, Y. A General Linear Free Energy Relationship for Predicting Partition Coefficients of Neutral Organic Compounds. Chemosphere 2020, 247, 125869. [Google Scholar] [CrossRef]
- Zsirka, B.; Vágvölgyi, V.; Horváth, E.; Juzsakova, T.; Fónagy, O.; Szabó-bárdos, E.; Kristóf, J. Halloysite-Zinc Oxide Nanocomposites as Potential Photocatalysts. Minerals 2022, 12, 476. [Google Scholar] [CrossRef]
- Chitra, M.; Mangamma, G.; Uthayarani, K.; Neelakandeswari, N.; Girija, E.K. Band Gap Engineering in ZnO Based Nanocomposites. Phys. E Low. Dimens. Syst. Nanostruct. 2020, 119, 113969. [Google Scholar] [CrossRef]
- Shalan, A.E.; Makhlouf, A.S.H.; Lanceros-Méndez, S. Nanocomposites Materials and Their Applications: Current and Future Trends. In Advances in Nanocomposite Materials for Environmental and Energy Harvesting Applications; Shalan, A.E., Hamdy Makhlouf, A.S., Lanceros-Méndez, S., Eds.; Springer International Publishing: Cham, Switzerland, 2022; pp. 3–14. ISBN 978-3-030-94319-6. [Google Scholar]
- Zahmatkesh, S.; Hajiaghaei-Keshteli, M.; Bokhari, A.; Sundaramurthy, S.; Panneerselvam, B.; Rezakhani, Y. Wastewater Treatment with Nanomaterials for the Future: A State-of-the-Art Review. Environ. Res. 2023, 216, 114652. [Google Scholar] [CrossRef]
- Gnanamoorthy, G.; Yadav, V.K.; Ali, D.; Ramar, K.; Ali, H.; Narayanan, V. New Designing (NH4)2SiP4O13 Nanowires and Effective Photocatalytic Degradation of Malachite Green. Chem. Phys. Lett. 2022, 803, 139817. [Google Scholar] [CrossRef]
- Kong, L.; Liu, Y.; Dong, L.; Zhang, L.; Qiao, L.; Wang, W.; You, H. Enhanced Red Luminescence in CaAl12O19:Mn4+ via Doping Ga3+ for Plant Growth Lighting. Dalton Trans. 2020, 49, 1947–1954. [Google Scholar] [CrossRef]
- Liu, W.; Zhao, C.; Zhou, Y.; Xu, X. Modeling of Vapor-Liquid Equilibrium for Electrolyte Solutions Based on COSMO-RS Interaction. J. Chem. 2022, 2022, 9070055. [Google Scholar] [CrossRef]
- Al-Attar, H.M.; Hussein, H.T.; Zamel, R.S.; Addie, A.J.; Mohammed, M.K.A. Methylene Blue Degradation Using ZnO:CuO:Al2O3 Nanocomposite Synthesized by Liquid Laser Ablation. Opt. Quantum. Electron. 2023, 55, 309. [Google Scholar] [CrossRef]
- Shan, L.; Tan, C.Y.; Shen, X.; Ramesh, S.; Zarei, M.S.; Kolahchi, R.; Hajmohammad, M.H. The Effects of Nano-Additives on the Mechanical, Impact, Vibration, and Buckling/Post-Buckling Properties of Composites: A Review. J. Mater. Res. Technol. 2023, 24, 7570–7598. [Google Scholar] [CrossRef]
- Ghazanlou, S.I.; Ghazanlou, S.I.; Ashraf, W. Improvement in the Physical and Mechanical Properties of the Cement-Based Composite with the Addition of Nanostructured BN–Fe3O4 Reinforcement. Sci. Rep. 2021, 11, 19358. [Google Scholar] [CrossRef]
- Sharma, D.; Kumar, M.; Jain, V.P.; Chaudhary, S.; Jaiswar, G. Bio-Based Polyamide Nanocomposites of Nanoclay, Carbon Nanotubes and Graphene: A Review. Iran. Polym. J. 2023, 32, 773–790. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, Z.; Yin, X.-A.; Zhu, Y. Impacts of Biochars on Bacterial Community Shifts and Biodegradation of Antibiotics in an Agricultural Soil during Short-Term Incubation. Sci. Total Environ. 2021, 771, 144751. [Google Scholar] [CrossRef] [PubMed]
- Temga, J.P.; Mache, J.R.; Madi, A.B.; Nguetnkam, J.P.; Bitom, D.L. Ceramics Applications of Clay in Lake Chad Basin, Central Africa. Appl. Clay Sci. 2019, 171, 118–132. [Google Scholar] [CrossRef]
- Giese, R.F. Kaolin Group Minerals. In Encyclopedia of Sediments and Sedimentary Rocks; Middleton, G.V., Church, M.J., Coniglio, M., Hardie, L.A., Longstaffe, F.J., Eds.; Springer: Dordrecht, The Netherlands, 2003; pp. 398–400. ISBN 978-1-4020-3609-5. [Google Scholar]
- Murray, H.; Kogel, J.E. Engineered Clay Products for the Paper Industry. Appl. Clay Sci. 2005, 29, 199–206. [Google Scholar] [CrossRef]
- Hadia, N.J.; Ng, Y.H.; Stubbs, L.P.; Torsæter, O. High Salinity and High Temperature Stable Colloidal Silica Nanoparticles with Wettability Alteration Ability for EOR Applications. Nanomaterials 2021, 11, 707. [Google Scholar] [CrossRef]
- Mochizuki, C.; Nakamura, J.; Nakamura, M. Development of Non-Porous Silica Nanoparticles towards Cancer Photo-Theranostics. Biomedicines 2021, 9, 73. [Google Scholar] [CrossRef]
- Kloprogge, J. The Kaolin Group: Octahedral and Tetrahedral Sheets. In Spectroscopic Methods in the Study of Kaolin Minerals and Their Modifications; Kloprogge, J., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 97–159. ISBN 978-3-030-02373-7. [Google Scholar]
- Caponi, N.; Collazzo, G.C.; Jahn, S.L.; Dotto, G.L.; Mazutti, M.A.; Foletto, E.L. Use of Brazilian Kaolin as a Potential Low-Cost Adsorbent for the Removal of Malachite Green from Colored Effluents. Mater. Res. 2017, 20, 14–22. [Google Scholar]
- Ibrahim, H.; Mahmoud, T.; Hegazy, E. Application of Zeolite Prepared from Egyptian Kaolin for the Removal of Heavy Metals: II. Isotherm Models. J. Hazard. Mater. 2010, 182, 842–847. [Google Scholar] [CrossRef]
- Valášková, M.; Blahůšková, V.; Vlček, J. Effects of Kaolin Additives in Fly Ash on Sintering and Properties of Mullite Ceramics. Minerals 2021, 11, 887. [Google Scholar] [CrossRef]
- Gougazeh, M.; Buhl, J.-C. Synthesis and Characterization of Zeolite A by Hydrothermal Transformation of Natural Jordanian Kaolin. J. Assoc. Arab. Univ. Basic. Appl. Sci. 2014, 15, 35–42. [Google Scholar] [CrossRef]
- Babayevska, N.; Przysiecka, Ł.; Iatsunskyi, I.; Nowaczyk, G.; Jarek, M.; Janiszewska, E.; Jurga, S. ZnO Size and Shape Effect on Antibacterial Activity and Cytotoxicity Profile. Sci. Rep. 2022, 12, 8148. [Google Scholar] [CrossRef]
- Peres, L.G.S.; Malafatti, J.O.D.; Bernardi, B.; Mattoso, L.H.C.; Paris, E.C. Biodegradable Starch Sachets Reinforced with Montmorillonite for Packing ZnO Nanoparticles: Solubility and Zn2+ Ions Release. J. Polym. Environ. 2023, 31, 2388–2398. [Google Scholar] [CrossRef]
- Shu, Z.; Zhang, Y.; Yang, Q.; Yang, H. Halloysite Nanotubes Supported Ag and ZnO Nanoparticles with Synergistically Enhanced Antibacterial Activity. Nanoscale Res. Lett. 2017, 12, 135. [Google Scholar] [CrossRef]
- Peng, H.; Liu, X.; Tang, W.; Ma, R. Facile Synthesis and Characterization of ZnO Nanoparticles Grown on Halloysite Nanotubes for Enhanced Photocatalytic Properties. Sci. Rep. 2017, 7, 2250. [Google Scholar] [CrossRef]
- Choudhary, N.; Yadav, V.K.; Ali, H.; Ali, D.; Almutairi, B.O.; Cavalu, S.; Patel, A. Remediation of Methylene Blue Dye from Wastewater by Using Zinc Oxide Nanoparticles Loaded on Nanoclay. Water 2023, 15, 1427. [Google Scholar] [CrossRef]
- Meigoli Boushehrian, M.; Esmaeili, H.; Foroutan, R. Ultrasonic Assisted Synthesis of Kaolin/CuFe2O4 Nanocomposite for Removing Cationic Dyes from Aqueous Media. J. Environ. Chem. Eng. 2020, 8, 103869. [Google Scholar] [CrossRef]
- Choudhary, N.; Yadav, V.K.; Yadav, K.K.; Almohana, A.I.; Almojil, S.F.; Gnanamoorthy, G.; Kim, D.-H.; Islam, S.; Kumar, P.; Jeon, B.-H. Application of Green Synthesized MMT/Ag Nanocomposite for Removal of Methylene Blue from Aqueous Solution. Water 2021, 13, 3206. [Google Scholar] [CrossRef]
- Gnanamoorthy, G.; Karthikeyan, V.; Ali, D.; Kumar, G.; Yadav, V.K.; Narayanan, V. Global Popularization of CuNiO2 and Their RGO Nanocomposite Loveabled to the Photocatalytic Properties of Methylene Blue. Environ. Res. 2022, 204, 112338. [Google Scholar] [CrossRef]
- Bayomie, O.S.; Kandeel, H.; Shoeib, T.; Yang, H.; Youssef, N.; El-Sayed, M.M.H. Novel Approach for Effective Removal of Methylene Blue Dye from Water Using Fava Bean Peel Waste. Sci. Rep. 2020, 10, 7824. [Google Scholar] [CrossRef]
- Massaro, M.; Casiello, M.; D’Accolti, L.; Lazzara, G.; Nacci, A.; Nicotra, G.; Noto, R.; Pettignano, A.; Spinella, C.; Riela, S. One-Pot Synthesis of ZnO Nanoparticles Supported on Halloysite Nanotubes for Catalytic Applications. Appl. Clay Sci. 2020, 189, 105527. [Google Scholar] [CrossRef]
- Mustapha, S.; Tijani, J.O.; Ndamitso, M.M.; Abdulkareem, S.A.; Shuaib, D.T.; Mohammed, A.K.; Sumaila, A. The Role of Kaolin and Kaolin/ZnO Nanoadsorbents in Adsorption Studies for Tannery Wastewater Treatment. Sci. Rep. 2020, 10, 13068. [Google Scholar] [CrossRef] [PubMed]
- Mamulová Kutláková, K.; Tokarský, J.; Peikertová, P. Functional and Eco-Friendly Nanocomposite Kaolinite/ZnO with High Photocatalytic Activity. Appl. Catal. B 2015, 162, 392–400. [Google Scholar] [CrossRef]
- Modi, S.; Yadav, V.K.; Amari, A.; Osman, H.; Igwegbe, C.A.; Fulekar, M.H. Nanobioremediation: A Bacterial Consortium-Zinc Oxide Nanoparticle-Based Approach for the Removal of Methylene Blue Dye from Wastewater. Environ. Sci. Pollut. Res. 2023, 30, 72641–72651. [Google Scholar] [CrossRef]
- Modi, S.; Yadav, V.K.; Amari, A.; Alyami, A.Y.; Gacem, A.; Harharah, H.N.; Fulekar, M.H. Photocatalytic Degradation of Methylene Blue Dye from Wastewater by Using Doped Zinc Oxide Nanoparticles. Water 2023, 15, 2275. [Google Scholar] [CrossRef]
- Modi, S.; Fulekar, M.H. Green Synthesis of Zinc Oxide Nanoparticles Using Garlic Skin Extract and Its Characterization. J. Nanostruct. 2020, 10, 20–27. [Google Scholar] [CrossRef]
- Melguizo-Rodríguez, L.; García-Recio, E.; Ruiz, C.; De Luna-Bertos, E.; Illescas-Montes, R.; Costela-Ruiz, V.J. Biological Properties and Therapeutic Applications of Garlic and Its Components. Food Funct. 2022, 13, 2415–2426. [Google Scholar] [CrossRef]
- dos Santos, P.C.M.; da Silva, L.M.R.; Magalhaes, F.E.A.; Cunha, F.E.T.; Ferreira, M.J.G.; de Figueiredo, E.A.T. Garlic (Allium sativum L.) Peel Extracts: From Industrial by-Product to Food Additive. Appl. Food Res. 2022, 2, 100186. [Google Scholar] [CrossRef]
- Hernández-Montesinos, I.Y.; Carreón-Delgado, D.F.; Ocaranza-Sánchez, E.; Ochoa-Velasco, C.E.; Suárez-Jacobo, Á.; Ramírez-López, C. Garlic (Allium sativum) Peel Extracts and Their Potential as Antioxidant and Antimicrobial Agents for Food Applications: Influence of Pretreatment and Extraction Solvent. Int. J. Food Sci. Technol. 2023. [Google Scholar] [CrossRef]
- Rahaman, M.Z.; Akther Hossain, A.K.M. Effect of Metal Doping on the Visible Light Absorption, Electronic Structure and Mechanical Properties of Non-Toxic Metal Halide CsGeCl3. RSC Adv. 2018, 8, 33010–33018. [Google Scholar] [CrossRef]
- Amendola, V.; Pilot, R.; Frasconi, M.; Maragò, O.M.; Iatì, M.A. Surface Plasmon Resonance in Gold Nanoparticles: A Review. J. Phys. Condens. Matter 2017, 29, 203002. [Google Scholar] [CrossRef]
- Khan, S.H.; Pathak, B.; Fulekar, M.H. Synthesis, Characterization and Photocatalytic Degradation of Chlorpyrifos by Novel Fe: ZnO Nanocomposite Material. Nanotechnol. Environ. Eng. 2018, 3, 13. [Google Scholar] [CrossRef]
- Aluvihara, S.; Kalpage, C.S.; Lemle, K.L. Elementary Chemical Analysis of Different Clay Types. J. Phys. Conf. Ser. 2021, 1781, 012007. [Google Scholar] [CrossRef]
- Bordeepong, S.; Bhongsuwan, D.; Pungrassami, T.; Bhongsuwan, T. Mineralogy, Chemical Composition and Ceramic Properties of Clay Deposits in Southern Thailand. Agric. Nat. Resour. 2012, 46, 485–500. [Google Scholar]
- Biswas, B.; Labille, J.; Prelot, B. Clays and Modified Clays in Remediating Environmental Pollutants. Environ. Sci. Pollut. Res. 2020, 27, 38381–38383. [Google Scholar] [CrossRef] [PubMed]
- Amar, A.; Loulidi, I.; Kali, A.; Boukhlifi, F.; Hadey, C.; Jabri, M. Physicochemical Characterization of Regional Clay: Application to Phenol Adsorption. Appl. Environ. Soil. Sci. 2021, 2021, 8826063. [Google Scholar] [CrossRef]
- Mustapha, S.; Tijani, J.O.; Ndamitso, M.M.; Abdulkareem, A.S.; Shuaib, D.T.; Mohammed, A.K. A Critical Review on Geosmin and 2-Methylisoborneol in Water: Sources, Effects, Detection, and Removal Techniques. Environ. Monit. Assess. 2021, 193, 204. [Google Scholar] [CrossRef]
- Modi, S.; Fulekar, M.H. Synthesis and Characterization of Zinc Oxide Nanoparticles and Zinc Oxide/Cellulose Nanocrystals Nanocomposite for Photocatalytic Degradation of Methylene Blue Dye under Solar Light Irradiation. Nanotechnol. Environ. Eng. 2020, 5, 18. [Google Scholar] [CrossRef]
- Saied, E.; Salem, S.S.; Al-Askar, A.A.; Elkady, F.M.; Arishi, A.A.; Hashem, A.H. Mycosynthesis of Hematite (α-Fe2O3) Nanoparticles Using Aspergillus Niger and Their Antimicrobial and Photocatalytic Activities. Bioengineering 2022, 9, 397. [Google Scholar] [CrossRef] [PubMed]
- Ahamad Khan, M.; Lone, S.A.; Shahid, M.; Zeyad, M.T.; Syed, A.; Ehtram, A.; Elgorban, A.M.; Verma, M.; Danish, M. Phytogenically Synthesized Zinc Oxide Nanoparticles (ZnO-NPs) Potentially Inhibit the Bacterial Pathogens: In Vitro Studies. Toxics 2023, 11, 452. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Wang, Y.; Chen, C.; Su, Y.; Li, L.; Miao, L.; Gu, H.; Zhao, W.; Ding, L.; Hu, D. Oriented Plate-like KNbO3 Polycrystals: Topochemical Mesocrystal Conversion and Piezoelectric and Photocatalytic Responses. Inorg. Chem. 2023, 62, 10408–10419. [Google Scholar] [CrossRef]
- Zheng, Y.; Liu, Y.; Guo, X.; Chen, Z.; Zhang, W.; Wang, Y.; Tang, X.; Zhang, Y.; Zhao, Y. Sulfur-Doped g-C3N4/RGO Porous Nanosheets for Highly Efficient Photocatalytic Degradation of Refractory Contaminants. J. Mater. Sci. Technol. 2020, 41, 117–126. [Google Scholar] [CrossRef]
- Yu, H.; Zhu, J.; Qiao, R.; Zhao, N.; Zhao, M.; Kong, L. Facile Preparation and Controllable Absorption of a Composite Based on PMo12/Ag Nanoparticles: Photodegradation Activity and Mechanism. ChemistrySelect 2022, 7, e202103668. [Google Scholar] [CrossRef]
- Meky, A.I.; Hassaan, M.A.; Fetouh, H.A.; Ismail, A.M.; El Nemr, A. Cube-Shaped Cobalt-Doped Zinc Oxide Nanoparticles with Increased Visible-Light-Driven Photocatalytic Activity Achieved by Green Co-Precipitation Synthesis. Sci. Rep. 2023, 13, 19329. [Google Scholar] [CrossRef] [PubMed]
- Tsade Kara, H.; Anshebo, S.T.; Sabir, F.K.; Adam Workineh, G. Removal of Methylene Blue Dye from Wastewater Using Periodiated Modified Nanocellulose. Int. J. Chem. Eng. 2021, 2021, 9965452. [Google Scholar] [CrossRef]
- Behnajady, M.A.; Modirshahla, N.; Hamzavi, R. Kinetic Study on Photocatalytic Degradation of C.I. Acid Yellow 23 by ZnO Photocatalyst. J. Hazard. Mater. 2006, 133, 226–232. [Google Scholar] [CrossRef]
- Badeenezhad, A.; Azhdarpoor, A.; Bahrami, S.; Yousefinejad, S. Removal of Methylene Blue Dye from Aqueous Solutions by Natural Clinoptilolite and Clinoptilolite Modified by Iron Oxide Nanoparticles. Mol. Simul. 2019, 45, 564–571. [Google Scholar] [CrossRef]
- Albarakaty, F.M.; Alzaban, M.I.; Alharbi, N.K.; Bagrwan, F.S.; Abd El-Aziz, A.R.M.; Mahmoud, M.A. Zinc Oxide Nanoparticles, Biosynthesis, Characterization and Their Potent Photocatalytic Degradation, and Antioxidant Activities. J. King Saud Univ. Sci. 2023, 35, 102434. [Google Scholar] [CrossRef]
- Yadav, V.K.; Choudhary, N.; Ali, D.; Kumar, G.; Gnanamoorthy, G.; Khan, A.U.; Kumar, P.; Hari Kumar, S.; Tizazu, B.Z. Determination of Adsorption of Methylene Blue Dye by Incense Stick Ash Waste and Its Toxicity on RTG-2 Cells. Adsorpt. Sci. Technol. 2022, 2022, 8565151. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, M.; Huang, J.; Zhao, N.; Yu, H. Controllable Synthesis, Photocatalytic Property, and Mechanism of a Novel POM-Based Direct Z-Scheme Nano-Heterojunction α-Fe2O3/P2Mo18. Molecules 2023, 28, 6671. [Google Scholar] [CrossRef] [PubMed]
- Ai, L.; Jiang, J. Removal of Methylene Blue from Aqueous Solution with Self-Assembled Cylindrical Graphene–Carbon Nanotube Hybrid. Chem. Eng. J. 2012, 192, 156–163. [Google Scholar] [CrossRef]
- Mir, I.A.; Singh, I.; Birajdar, B.; Rawat, K. A Facile Platform for Photocatalytic Reduction of Methylene Blue Dye by CdSe-TiO2 Nanoparticles. Water Conserv. Sci. Eng. 2017, 2, 43–50. [Google Scholar] [CrossRef]
- Tran, H.V.; Bui, L.T.; Dinh, T.T.; Le, D.H.; Huynh, C.D.; Trinh, A.X. Graphene Oxide/Fe3O4/Chitosan Nanocomposite: A Recoverable and Recyclable Adsorbent for Organic Dyes Removal. Application to Methylene Blue. Mater. Res. Express 2017, 4, 035701. [Google Scholar] [CrossRef]
- Bangari, R.S.; Yadav, A.; Bharadwaj, J.; Sinha, N. Boron Nitride Nanosheets Incorporated Polyvinylidene Fluoride Mixed Matrix Membranes for Removal of Methylene Blue from Aqueous Stream. J. Environ. Chem. Eng. 2022, 10, 107052. [Google Scholar] [CrossRef]
- Tarekegn, M.M.; Balakrishnan, R.M.; Hiruy, A.M.; Dekebo, A.H. Removal of Methylene Blue Dye Using Nano Zerovalent Iron, Nanoclay and Iron Impregnated Nanoclay—A Comparative Study. RSC Adv. 2021, 11, 30109–30131. [Google Scholar] [CrossRef]
- Fouda, A.; Salem, S.S.; Wassel, A.R.; Hamza, M.F.; Shaheen, T.I. Optimization of Green Biosynthesized Visible Light Active CuO/ZnO Nano-Photocatalysts for the Degradation of Organic Methylene Blue Dye. Heliyon 2020, 6, e04896. [Google Scholar] [CrossRef]
- Fan, J.; Zhang, B.; Zhu, B.; Shen, W.; Chen, Y.; Zeng, F. New Insight into the Mechanism for the Removal of Methylene Blue by Hydrotalcite-Supported Nanoscale Zero-Valent Iron. Water 2023, 15, 183. [Google Scholar] [CrossRef]
- Nazir, M.A.; Khan, N.A.; Cheng, C.; Shah, S.S.A.; Najam, T.; Arshad, M.; Sharif, A.; Akhtar, S.; Rehman, A. Ur Surface Induced Growth of ZIF-67 at Co-Layered Double Hydroxide: Removal of Methylene Blue and Methyl Orange from Water. Appl. Clay Sci. 2020, 190, 105564. [Google Scholar] [CrossRef]
- Khan, S.B.; Khan, M.I.; Nisar, J. Microwave-Assisted Green Synthesis of Pure and Mn-Doped ZnO Nanocomposites: In Vitro Antibacterial Assay and Photodegradation of Methylene Blue. Front. Mater. 2022, 8, 710155. [Google Scholar] [CrossRef]
- Jamshaid, M.; Nazir, M.A.; Najam, T.; Shah, S.S.A.; Khan, H.M.; Rehman, A. ur Facile Synthesis of Yb3+-Zn2+ Substituted M Type Hexaferrites: Structural, Electric and Photocatalytic Properties under Visible Light for Methylene Blue Removal. Chem. Phys. Lett. 2022, 805, 139939. [Google Scholar] [CrossRef]
- Jana, T.K.; Chatterjee, K. Hybrid Nanostructures Exhibiting Both Photocatalytic and Antibacterial Activity—A Review. Environ. Sci. Pollut. Res. 2023, 30, 95215–95249. [Google Scholar] [CrossRef] [PubMed]
- Cinelli, G.; Cuomo, F.; Ambrosone, L.; Colella, M.; Ceglie, A.; Venditti, F.; Lopez, F. Photocatalytic Degradation of a Model Textile Dye Using Carbon-Doped Titanium Dioxide and Visible Light. J. Water Process Eng. 2017, 20, 71–77. [Google Scholar] [CrossRef]
- Ablikim, M.; Achasov, M.N.; Ahmed, S.; Albrecht, M.; Alekseev, M.; Amoroso, A.; An, F.F.; An, Q.; Bai, J.Z.; Bai, Y.; et al. Measurement of the Absolute Branching Fraction of the Inclusive Decay Λc+ → Λ+X. Phys. Rev. Lett. 2018, 121, 062003. [Google Scholar] [CrossRef]



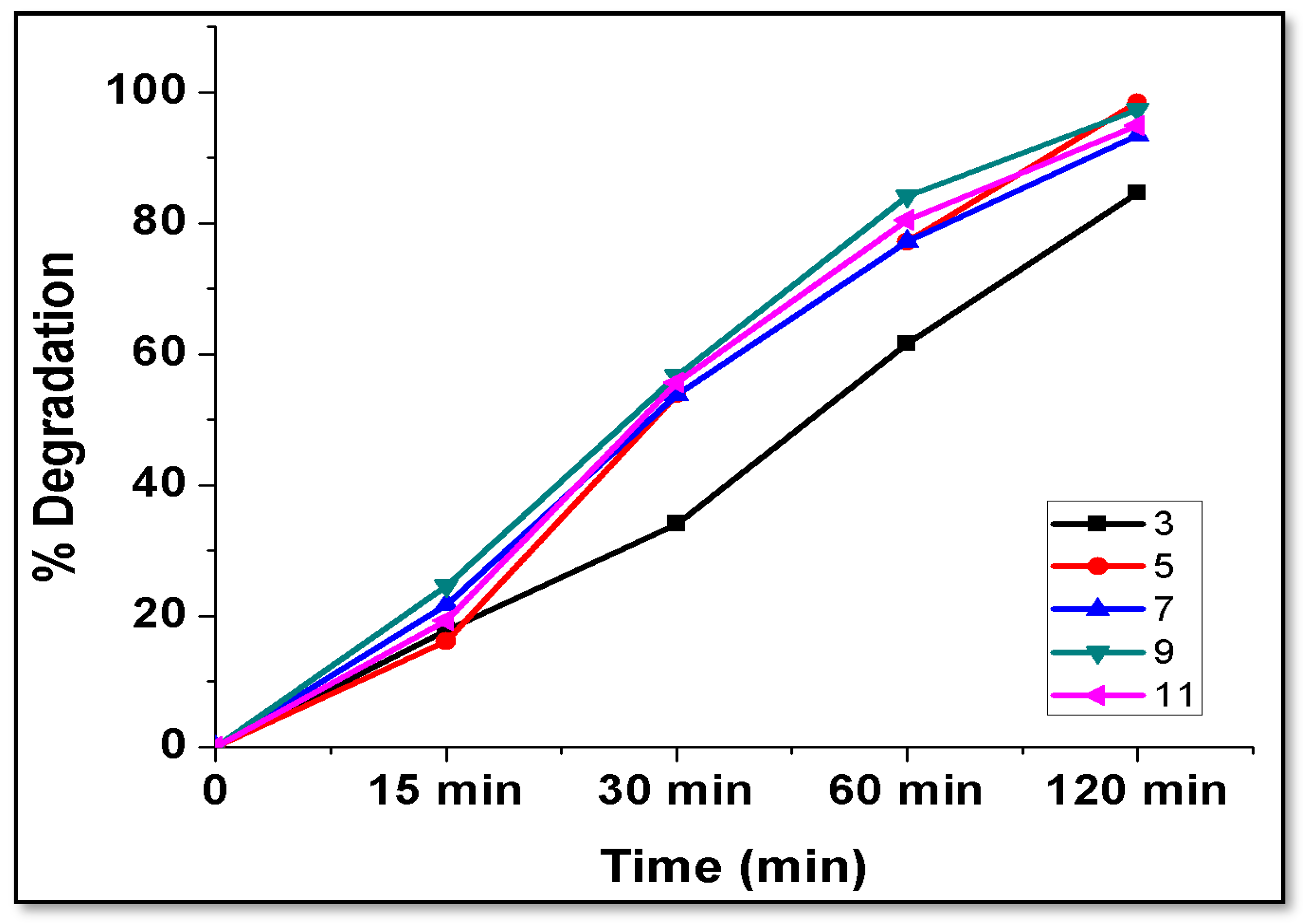
| Nanocomposite/Nanomaterials | Size (nm) | Removal Percentage | Concentration of Dye (ppm) | Time (Minutes) | References |
|---|---|---|---|---|---|
| MMT/Ag | 1–2 microns | 99.9% | 25 | 30 | [48] |
| Halloysite/ZnONPs | 40–200 (diameter) | 97.5 | 25 | 15 | [46] |
| ZnONPs | 7 | 82.01 | 10 | - | [55] |
| W-doped ZnONPs | 18 | 88.21 | 10 | - | |
| Sb-doped ZnONPs | 9.55 | 90.06 | 10 | 120 | |
| ZnONPs | 40–100 | 100 | 300 | 60 | [78] |
| NaOH-activated incense sticks, ash | 69.74 | 200 | 60 | [79] | |
| Fe3O4@SiO2/TiO2@WO3 | 92.77 | 60 | [9] | ||
| Na periodate-modified nanocellulose (NaIO4 NC) from Eichhornia crassipes | Several microns | 90.91 mg·g−1 78.1% | 30 | 60 | [80] |
| Self-assembled cylindrical graphene–CNTs | Width: 20–40 nm Length: several microns | 81.97 mg/g 97% | 10 | [81] | |
| CdSe-TiO2 NPs | TiO2: 20 ± 1 nm CdSe: 3.0 ± 0.2 nm | 67% | 10 | 60 | [82] |
| Chitosan/Fe3O4/graphene oxide (CS/Fe3O4/GO) NC | IONPs: 30–40 nm | 30.10 mg/g (by Langmuir) 90–100% | 10–40 | [83] | |
| Boron nitride nanosheets @ polyvinylidene fluoride | 142.86 mg/g 100% | 10 | [84] | ||
| NZVI-impregnated nanoclay | 72.4 nm | ~100 | 40 | 120 | [85] |
| Clinoptilolite modified by iron oxide NPs | Microns | 98% | 200 | 45 (optimum) | [77] |
| CuO/ZnO nanophotocatalysts | 10–55 nm | 97% | 85 | [86] | |
| Hydrotalcite-supported NZVI | 90 nm to several microns |
81 mg/g 99.6% | 40 | 30 | [87] |
| ZnONP loaded on bacterial consortium | 95.46% | 10 | 4320 | [54] | |
| Zeolite imidazolate frameworks (ZIF-67) @ co-layered double hydroxide | 1 μm diameter, thickness ~40 nm |
57.24 mg/g 79.9% | 25 | 100 | [88] |
| Mn-doped ZnO nanocomposites | 50 nm | 70% | 0.1 mM | 60 | [89] |
| Yb3+-Zn2+ substituted M-type hexaferrites | 16–17 nm | 96.1 % | 90 | [90] | |
| ZnONPs@cellulose nanocrystals NC | 59.51 | 88.62% | 10 | 120 | [68] |
| X-CuTiAP (en, trien, ETA, and DMA) nanospheres | Below 100 | 33–73 | 1 mM in 100 mL | 50–60 | [91] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Modi, S.; Yadav, V.K.; Ali, D.; Choudhary, N.; Alarifi, S.; Sahoo, D.K.; Patel, A.; Fulekar, M.H. Photocatalytic Degradation of Methylene Blue from Aqueous Solutions by Using Nano-ZnO/Kaolin-Clay-Based Nanocomposite. Water 2023, 15, 3915. https://doi.org/10.3390/w15223915
Modi S, Yadav VK, Ali D, Choudhary N, Alarifi S, Sahoo DK, Patel A, Fulekar MH. Photocatalytic Degradation of Methylene Blue from Aqueous Solutions by Using Nano-ZnO/Kaolin-Clay-Based Nanocomposite. Water. 2023; 15(22):3915. https://doi.org/10.3390/w15223915
Chicago/Turabian StyleModi, Shreya, Virendra Kumar Yadav, Daoud Ali, Nisha Choudhary, Saud Alarifi, Dipak Kumar Sahoo, Ashish Patel, and Madhusudan Hiraman Fulekar. 2023. "Photocatalytic Degradation of Methylene Blue from Aqueous Solutions by Using Nano-ZnO/Kaolin-Clay-Based Nanocomposite" Water 15, no. 22: 3915. https://doi.org/10.3390/w15223915








