Effects of Environmental Factors on the Distribution and Diversity of Aquatic Oligochaetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Field and Labarotory Work
2.2. Data Analyses
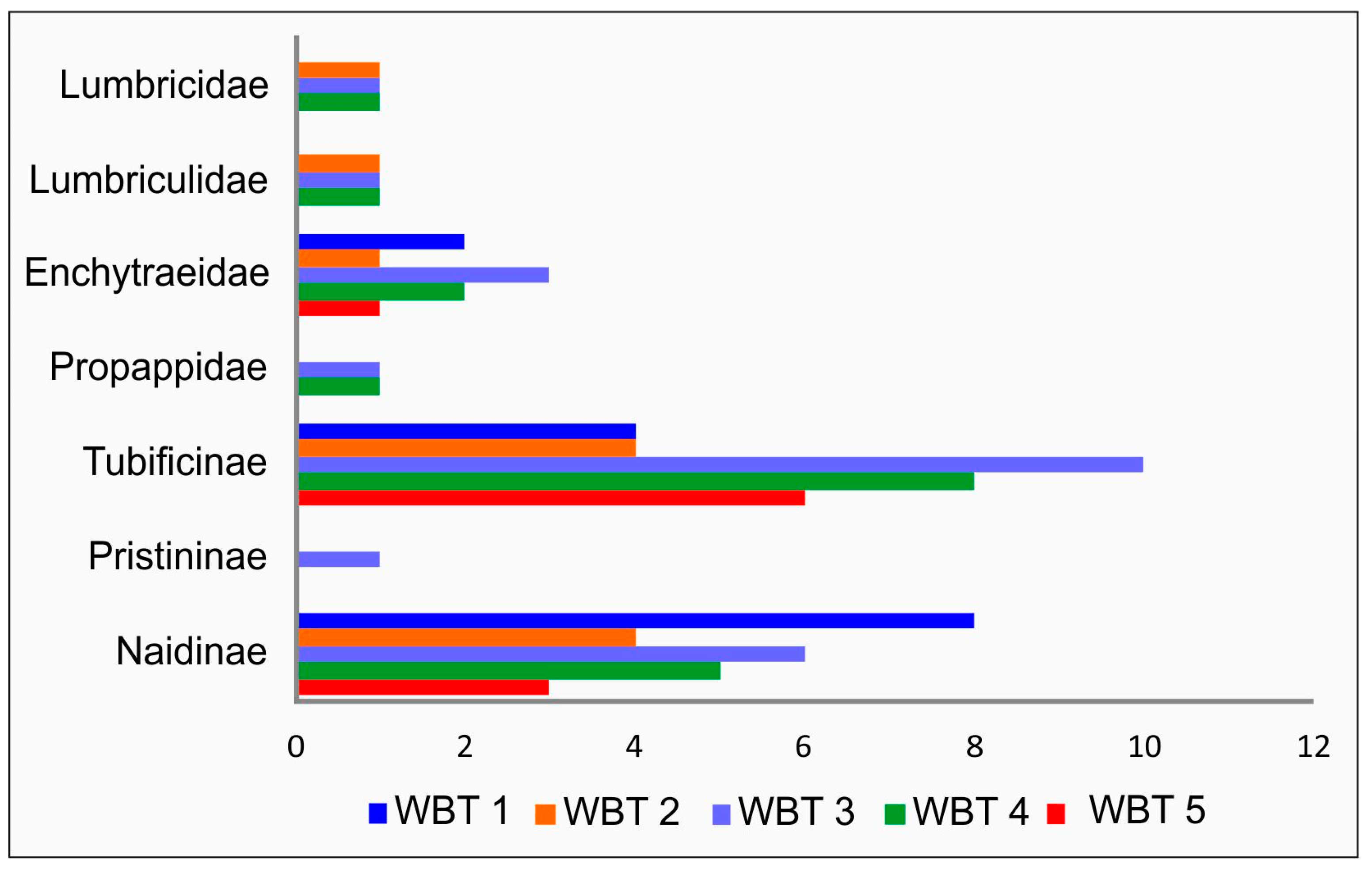
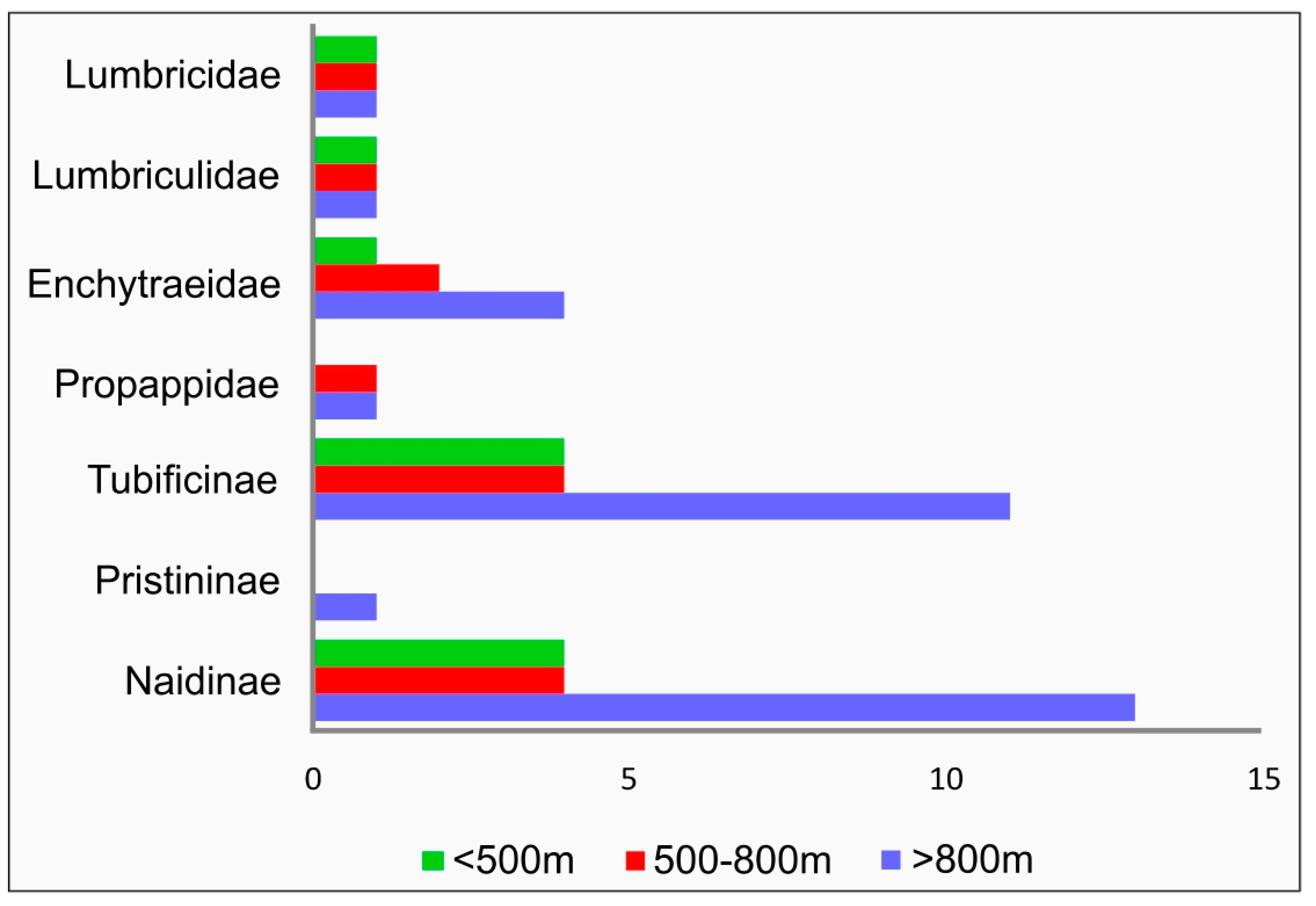
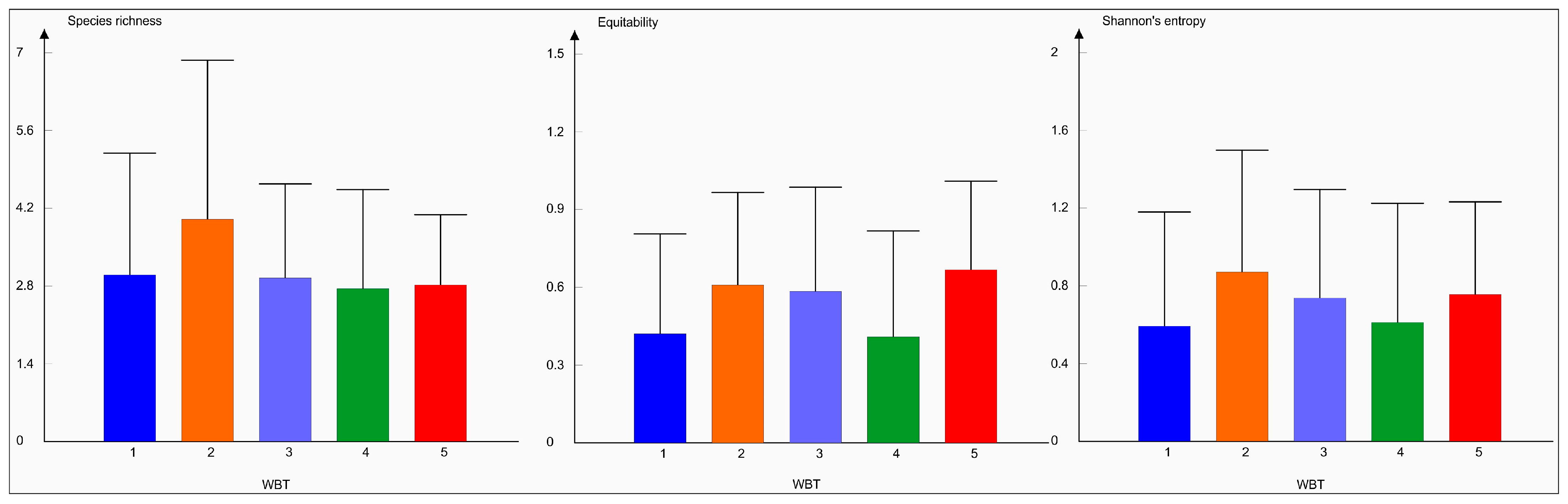
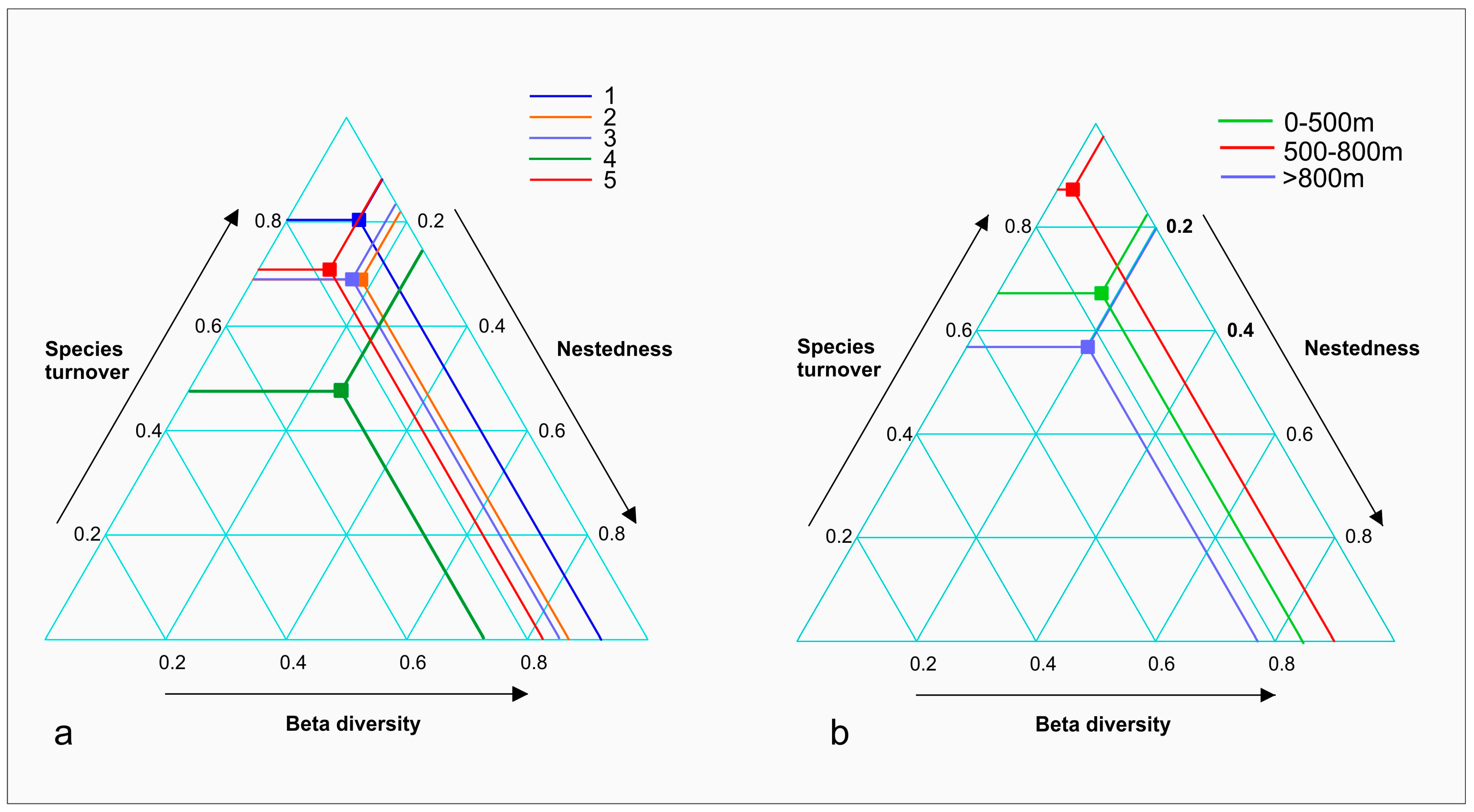
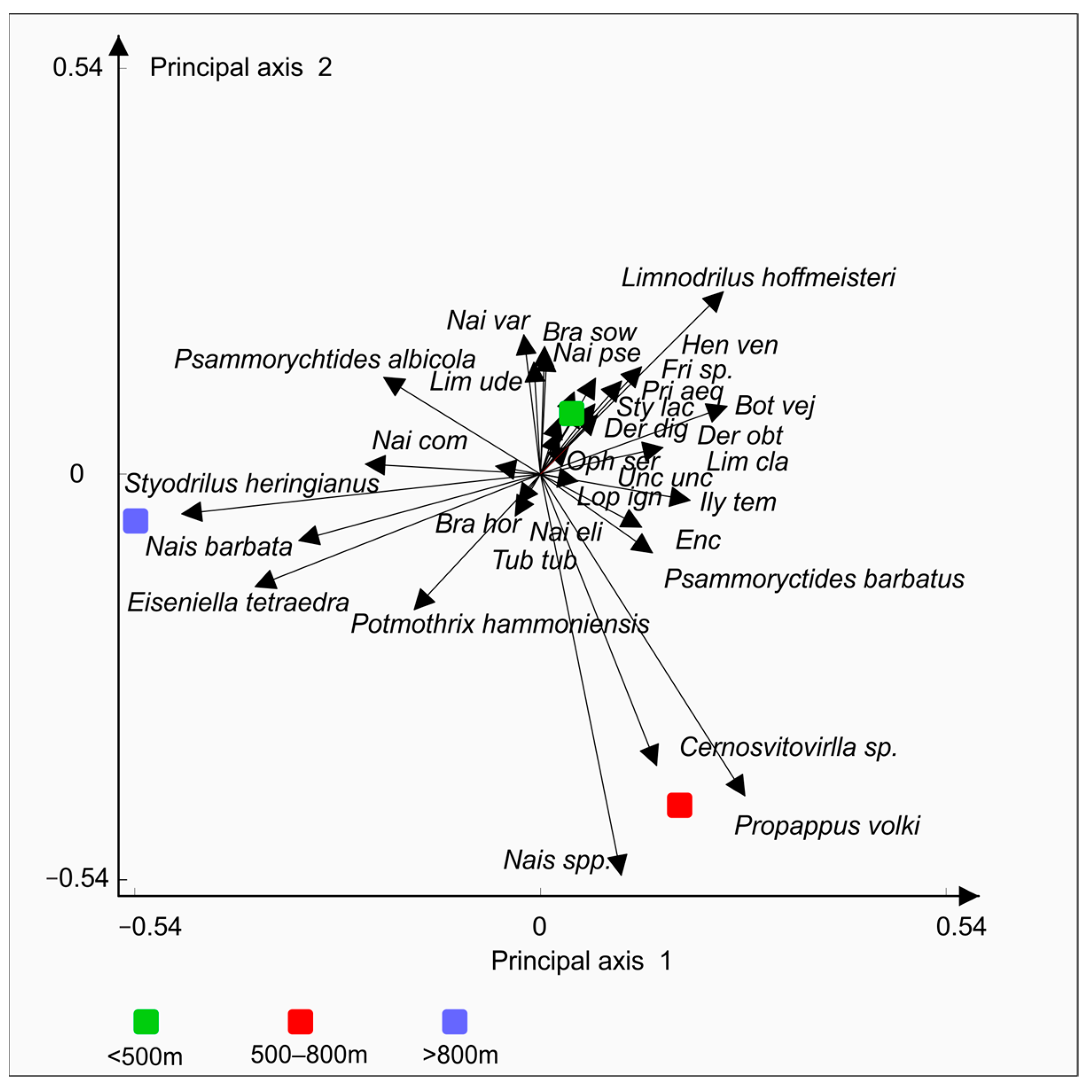
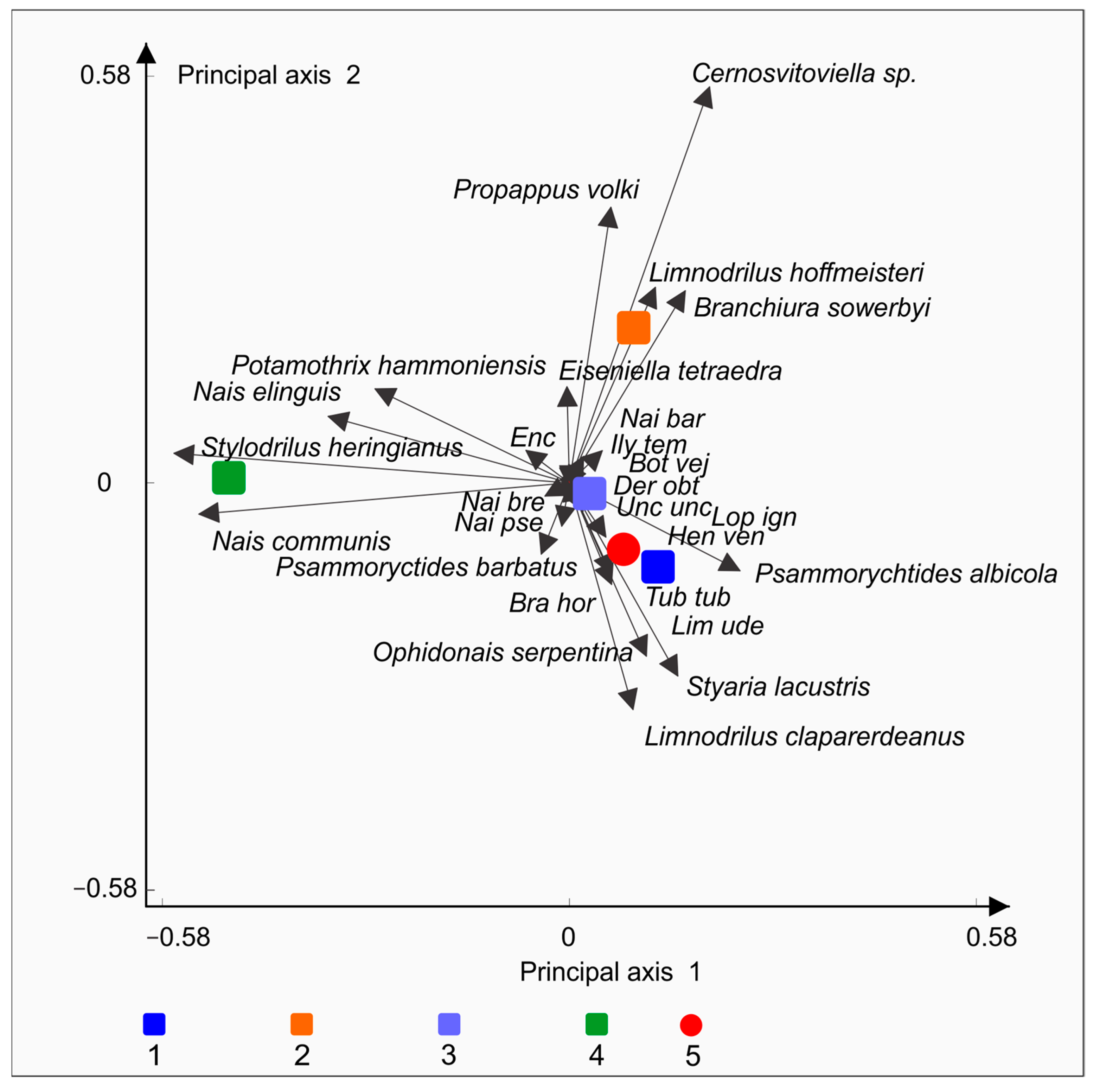
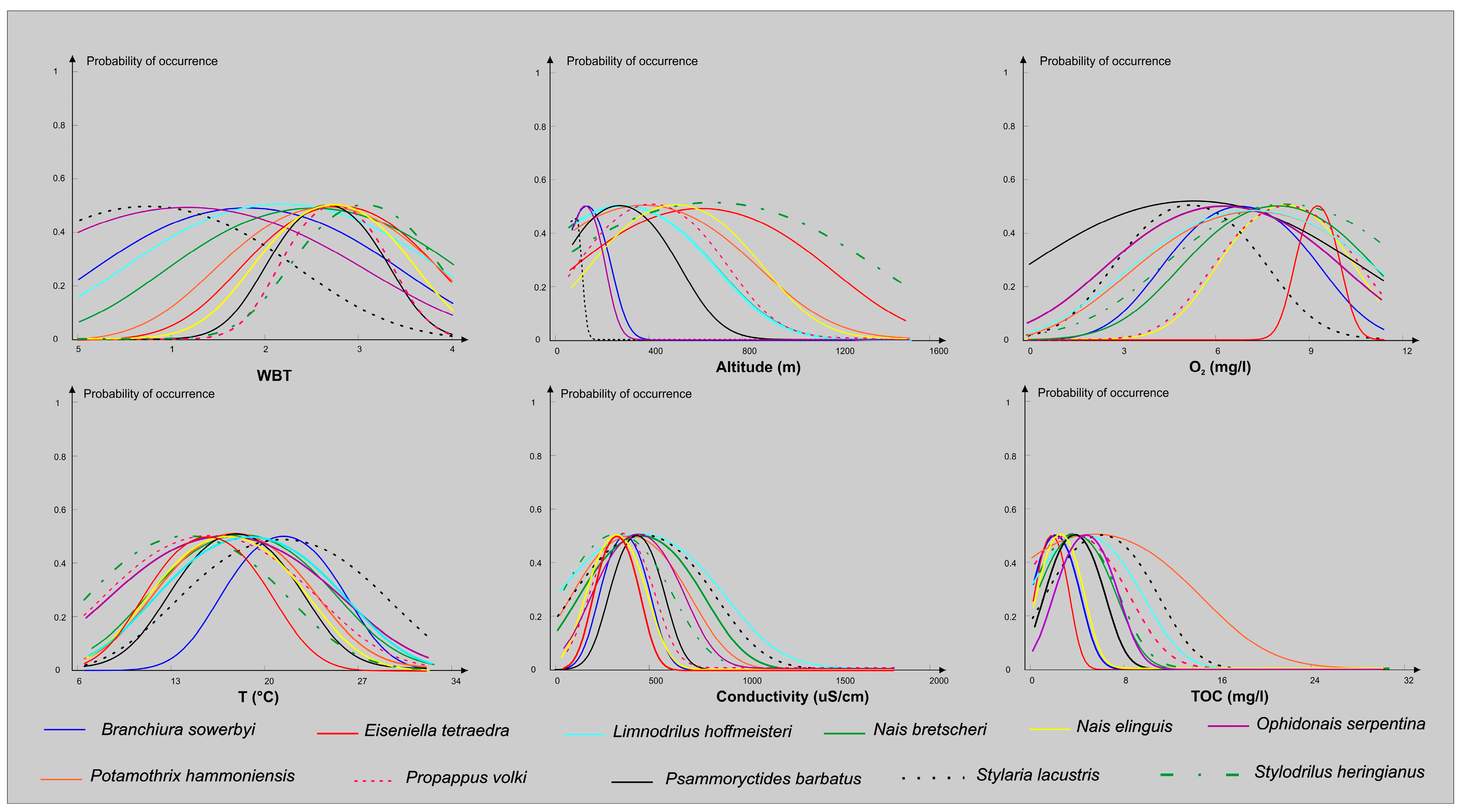
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ilies, J. Limnofauna Europaea; Gustav Fischer Verlag: Stuttgart, Germany, 1978. [Google Scholar]
- Verdonschot, P.F.M. The Role of Oligochaetes in the Management of Waters. Hydrobiologia 1989, 180, 213–227. [Google Scholar] [CrossRef]
- Elexová, E.; Némethová, D. The Effect of Abiotic Environmental Variables on the Danube Macrozoobenthic Communities. Limnologica 2003, 33, 340–354. [Google Scholar] [CrossRef]
- Šporka, F.; Nagy, Š. The Macrozoobenthos of Parapotamon-Type Side Arms of the Danube River in Slovakia and Its Response to Flowing Conditions. Biologia 1998, 53, 633–643. [Google Scholar]
- Moog, O. Fauna Aquatica Austriaca Katalog zur Autökologischen Einstufung Aquatischer Organismen Österreichs; Wasserwirtschaftskataster, Federal Ministry for Agriculture and Forestry: Vienna, Austria, 2002; p. 670. [Google Scholar]
- Timm, T.; Seire, A.; Pall, P. Half a Century of Oligochaeta Research in Estonian Running Waters. Hydrobiologia 2001, 463, 223–234. [Google Scholar] [CrossRef]
- Van Haaren, T.; Soors, J. Aquatic Oligochaetes of The Netherlands and Belgium; KNNV Publishing Zeist: Zeist, The Netherlands, 2012. [Google Scholar]
- Dumnicka, E. New for Poland Tubificid (Oligochaeta) Species from Karstic Springs. Pol. J. Ecol. 2009, 57, 395–401. [Google Scholar]
- Schenková, J.; Pařil, P.; Petřivalská, K.; Bojková, J. Aquatic Oligochaetes (Annelida: Clitellata) of the Czech Republic: Check-List, New Records, and Ecological Remarks. Zootaxa 2010, 44, 29–44. [Google Scholar] [CrossRef]
- Giani, N.; Sambugar, B.; Martínez-Ansemil, E.; Martin, P.; Schmelz, R.M. The Groundwater Oligochaetes (Annelida, Clitellata) of Slovenia. Subterr. Biol. 2011, 9, 85–102. [Google Scholar] [CrossRef][Green Version]
- Uzunov, Y. Aquatic Oligochaets (Oligochaeta Limicola), Annelida: Aphanoneura, Oligochaeta, Branchiobdellea—Catalogus Faunae Bulgaricae 7; Professor Marin Drinov Academic Publishing House: Sofia, Bulgaria, 2008. [Google Scholar]
- Kerovec, M.; Kerovec, M.; Brigić, A. Croatian Freshwater Oligochaetes: Species Diversity, Distribution and Relationship to Surrounding Countries. Zootaxa 2016, 4193, 73–101. [Google Scholar] [CrossRef]
- Šundić, D.; Radujković, B.M.; Krpo-Ćetković, J. Catalogue of Aquatic Oligochaeta (Annelida: Clitellata) of Montenegro, Exclusive of Naidinae and Pristininae. Zootaxa 2011, 18, 1–25. [Google Scholar] [CrossRef]
- Dhora, D. Regjistër i Specieve të Faunës së Shqipërisë; Camaj-Pipa, Botimet: Shkodrës, Albania, 2009; ISBN 9789995602956. [Google Scholar]
- Atanacković, A.; Zorić, K.; Tomović, J.; Vasiljević, B.; Paunović, M. Distributional Patterns of Aquatic Oligochaeta Communities (Annelida: Clitellata) in Running Waters in Serbia. Arch. Biol. Sci. 2020, 72, 359–372. [Google Scholar] [CrossRef]
- Kang, H.; Bae, M.-J.; Lee, D.-S.; Hwang, S.-J.; Moon, J.-S.; Park, Y.-S. Distribution Patterns of the Freshwater Oligochaete Limnodrilus hoffmeisteri Influenced by Environmental Factors in Streams on a Korean Nationwide Scale. Water 2017, 9, 921. [Google Scholar] [CrossRef]
- Schenková, J.; Helešic, J. Habitat Preferences of Aquatic Oligochaeta (Annelida) in the Rokytná River, Czech Republic—A Small Highland Stream. Hydrobiologia 2006, 564, 117–126. [Google Scholar] [CrossRef]
- EN 27828:1994; Water Quality—Methods of Biological Sampling—Guidance on Handnet Sampling of Aquatic Benthic Macro-Invertebrates (ISO 7828:1985). European Committee for Standardisation: Brussels, Belgium, 1994.
- Timm, T. A Guide to the Freshwater Oligochaeta and Polychaeta of Northern and Central Europe. Lauterbornia 2009, 66, 1–235. [Google Scholar]
- Ter Braak, C.J.F. Canonical Correspondence Analysis: A New Eigenvector Technique for Multivariate Direct Gradient Analysis. Ecology 1986, 67, 1167–1179. [Google Scholar] [CrossRef]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning; Springer: New York, NY, USA, 2013; p. 112. [Google Scholar]
- Karadžić, B. FLORA: A Software Package for Statistical Analysis of Ecological Data. Water Res. Manag. 2013, 3, 45–54. [Google Scholar]
- Baselga, A. Partitioning the Turnover and Nestedness Components of Beta Diversity. Glob. Ecol. Biogeogr. 2010, 19, 134–143. [Google Scholar] [CrossRef]
- Bray, J.H.; Maxwell, S.E.; Maxwell, S.E. Multivariate Analysis of Variance; Sage: Thousand Oaks, CA, USA, 1985; ISBN 0803923104. [Google Scholar]
- Timm, T. The Genus Potamothrix (Annelida, Oligochaeta, Tubificidae): A Literature Review. Est. J. Ecol. 2013, 62, 121–136. [Google Scholar] [CrossRef]
- Atanacković, A.; Zorić, K.; Paunović, M. Invading Europe: The Tropical Aquatic Worm Branchiodrilus hortensis (Stephenson, 1910) (Clitellata, Naididae) Extends Its Range. BioInvasions Rec. 2021, 10, 598–604. [Google Scholar] [CrossRef]
- Nesemann, H.; Sharma, G.; Sinha, R.K. Aquatic Annelida (Polychaeta, Oligochaeta, Hirudinea) of the Ganga River and Adjacent Water Bodies in Patna (India: Bihar), with Description of a New Leech Species (Family Salifidae). Ann. Naturhist. Museum Wien 2004, 105, 139–187. [Google Scholar]
- Dumnicka, E. Composition and Abundance of Oligochaetes (Annelida: Oligochaeta) in Springs of Kraków-Czȩstochowa Upland (Southern Poland): Effect of Spring Encasing and Environmental Factors. Pol. J. Ecol. 2006, 54, 231–242. [Google Scholar]
- Dumnicka, E. Distribution of Oligochaeta in Various Littoral Habitats in the Anthropogenic Reservoirs. Int. J. Oceanogr. Hydrobiol. 2007, 36, 13–19. [Google Scholar]
- Marinković, N.; Karadžić, B.; Pešić, V.; Gligorović, B.; Grosser, C.; Paunović, M.; Nikolić, V.; Raković, M. Faunistic Patterns and Diversity Components of Leech Assemblages in Karst Springs of Montenegro. Knowl. Manag. Aquat. Ecosyst. 2019, 420, 26. [Google Scholar] [CrossRef]
- Petsch, D.K.; Ragonha, F.H.; Gimenez, B.C.G.; Barboza, L.G.A.; Takeda, A.M. Partitioning Beta Diversity of Aquatic Oligochaeta in Different Environments of a Neotropical Floodplain. Acta Sci.—Biol. Sci. 2015, 37, 41–49. [Google Scholar] [CrossRef][Green Version]
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The River Continuum Concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Schenková, J.; Kroča, J. Seasonal Changes of an Oligochaetous Clitellata (Annelida) Community in a Mountain Stream. Acta Univ. Carol. Environ. 2007, 21, 143–150. [Google Scholar]
- Von Schiller, D.; Acuña, V.; Aristi, I.; Arroita, M.; Basaguren, A.; Bellin, A.; Boyero, L.; Butturini, A.; Ginebreda, A.; Kalogianni, E.; et al. River Ecosystem Processes: A Synthesis of Approaches, Criteria of Use and Sensitivity to Environmental Stressors. Sci. Total Environ. 2017, 596–597, 465–480. [Google Scholar] [CrossRef]
- Marchand, J. The Influence of Seasonal Salinity and Turbidity Maximum Variations on the Nursery Function of the Loire Estuary (France). Netherl. J. Aquat. Ecol. 1993, 27, 427–436. [Google Scholar] [CrossRef]

| Waterbody Type | Altitude | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Taxon | Abb. | Type 1 | Type 2 | Type 3 | Type 4 | Type 5 | Alt 1 | Alt 2 | Alt 3 |
| f. Naididae | |||||||||
| subf. Naidinae | |||||||||
| Branhiodrilus hortensis (Stephenson, 1910) | Bra hor | 0.2 | 0.09 | 0.04 | |||||
| Chaetogaster diaphanus (Gruithuisen, 1828) | Cha dia | 0.1 | 0.02 | ||||||
| Dero digitata Müller, 1773 | Der dig | 0.09 | 0.02 | ||||||
| Dero dorsalis Ferronière, 1899 | Der dor | 0.09 | 0.02 | ||||||
| Dero obtusa d’Udekem, 1835 | Der obt | 0.18 | 0.04 | ||||||
| Nais alpina Sperber, 1948 | Nai alp | 0.17 | 0.14 | ||||||
| Nais barbata Müller, 1773 | Nai bar | 0.03 | 0.1 | ||||||
| Nais bretscheri Michaelsen,1899 | Nai bre | 0.2 | 0.37 | 0.17 | 0.27 | 0.3 | 0.29 | 0.2 | |
| Nais communis Piguet, 1906 | Nai com | 0.03 | 0.17 | 0.02 | 0.1 | ||||
| Nais elinguis Müller, 1774 | Nai eli | 0.4 | 0.16 | 0.17 | 0.13 | 0.29 | 0.2 | ||
| Nais pseudobtusa Piguet, 1906 | Nai pse | 0.2 | 0.02 | 0.14 | |||||
| Nais variabilis Piguet, 1906 | Nai var | 0.1 | 0.02 | ||||||
| Ophidonais serpentina (Müller, 1773) | Oph ser | 0.2 | 0.05 | 0.27 | 0.11 | ||||
| Uncinais uncinata (Ørsted, 1842) | Unc unc | 0.09 | 0.02 | ||||||
| Stylaria lacustris (Linnaeus, 1767) | Sty lac | 0.4 | 0.05 | 0.64 | 0.21 | ||||
| subf. Pristininae | |||||||||
| Pristina aequiseta Bourne, 1891 | Pri aeq | 0.03 | 0.02 | ||||||
| subf. Tubificinae | |||||||||
| Bothrioneurum vejdovskyanum Štolc, 1888 | Bot vej | 0.1 | 0.03 | 0.04 | |||||
| Branchiura sowerbyi Beddard, 1892 | Bra sow | 0.2 | 0.1 | 0.05 | 0.09 | 0.09 | |||
| Ilyodrilus templetoni (Southern, 1909) | Ily tem | 0.1 | 0.02 | ||||||
| Limnodrilus claparedeanus Ratzel, 1868 | Lim cla | 0.4 | 0.2 | 0.11 | 0.13 | 0.14 | |||
| Limnodrilus hoffmeisteri Claparède, 1862 | Lim hof | 0.8 | 0.6 | 0.55 | 0.33 | 0.55 | 0.62 | 0.43 | 0.3 |
| Limnodrilus udekemianus Claparède, 1862 | Lim ude | 0.2 | 0.1 | 0.13 | 0.09 | 0.15 | |||
| Potamothrix hammoniensis (Michaelsen, 1901) | Pot ham | 0.2 | 0.6 | 0.39 | 0.33 | 0.09 | 0.36 | 0.43 | 0.3 |
| Potamothrix vejdovskyi (Hrabě, 1941) | Pot vej | 0.17 | 0.1 | ||||||
| Psammoryctides albicola (Michaelsen, 1901) | Psa alb | 0.08 | 0.17 | 0.04 | 0.2 | ||||
| Psammoryctides barbatus (Grube, 1861) | Psa bar | 0.1 | 0.05 | 0.04 | 0.14 | ||||
| Lophochaeta ignota (Štolc, 1886) | Lop ign | 0.03 | 0.02 | ||||||
| Tubifex tubifex (Müller, 1774) | Tub tub | 0.2 | 0.08 | 0.08 | |||||
| f. Propappidae | |||||||||
| Propappus volki (Michaelsen, 1916) | Pro vol | 0.1 | 0.08 | 0.04 | 0.29 | ||||
| f. Enchytraeidae | |||||||||
| Enchytraeidae gen. sp. | Enc | 0.2 | 0.1 | 0.18 | 0.17 | 0.18 | 0.15 | 0.29 | 0.2 |
| Fridericia sp. | Fri sp. | 0.03 | 0.09 | 0.04 | |||||
| Henlea ventriculosa (d’Udekem, 1854) | Hen ven | 0.08 | 0.06 | ||||||
| Cernosvitoviella sp. | Cer sp. | 0.2 | 0.02 | 0.14 | |||||
| f. Lumbriculidae | |||||||||
| Stylodrilus heringianus Claparède,1862 | Sty her | 0.2 | 0.26 | 0.5 | 0.15 | 0.14 | 0.6 | ||
| f. Lumbricidae | |||||||||
| Eiseniella tetraedra (Savigny, 1826) | Eis tet | 0.2 | 0.05 | 0.17 | 0.04 | 0.14 | 0.2 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atanacković, A.; Popović, N.; Marinković, N.; Tomović, J.; Đuknić, J.; Stanković, J.; Paunović, M. Effects of Environmental Factors on the Distribution and Diversity of Aquatic Oligochaetes. Water 2023, 15, 3873. https://doi.org/10.3390/w15223873
Atanacković A, Popović N, Marinković N, Tomović J, Đuknić J, Stanković J, Paunović M. Effects of Environmental Factors on the Distribution and Diversity of Aquatic Oligochaetes. Water. 2023; 15(22):3873. https://doi.org/10.3390/w15223873
Chicago/Turabian StyleAtanacković, Ana, Nataša Popović, Nikola Marinković, Jelena Tomović, Jelena Đuknić, Jelena Stanković, and Momir Paunović. 2023. "Effects of Environmental Factors on the Distribution and Diversity of Aquatic Oligochaetes" Water 15, no. 22: 3873. https://doi.org/10.3390/w15223873
APA StyleAtanacković, A., Popović, N., Marinković, N., Tomović, J., Đuknić, J., Stanković, J., & Paunović, M. (2023). Effects of Environmental Factors on the Distribution and Diversity of Aquatic Oligochaetes. Water, 15(22), 3873. https://doi.org/10.3390/w15223873









