Comparison of the Meiofauna and Marine Nematode Communities before and after Removal of Spartina alterniflora in the Mangrove Wetland of Quanzhou Bay, Fujian Province
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Stations
2.2. Sample Handling and Sorting
2.3. Data Processing and Analysis
3. Results
3.1. Composition, Abundance, and Biomass
3.2. Marine Nematode Abundance and Biomass
3.3. Marine Nematode Dominant Genus
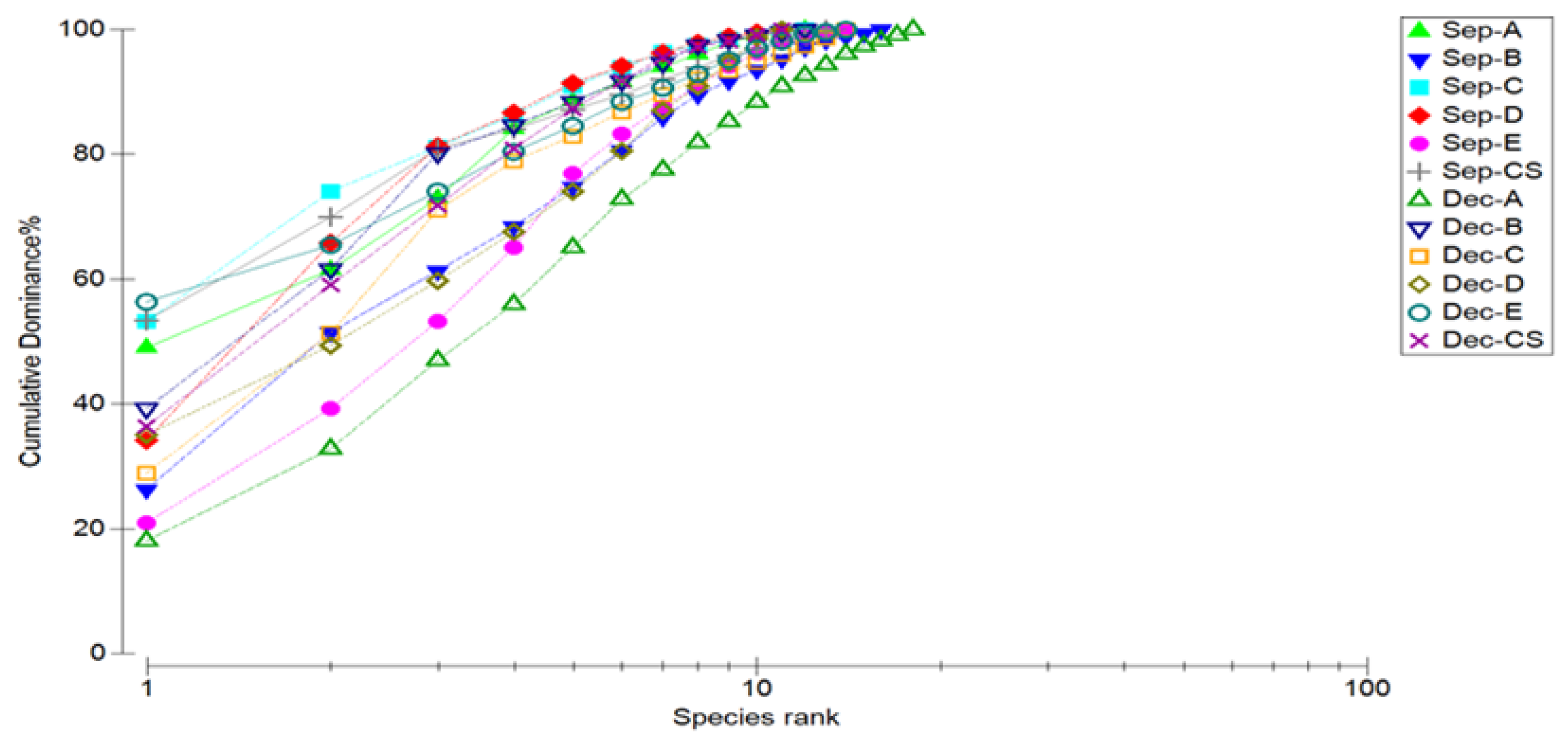
3.4. Marine Nematode Diversity and K-Dominance Analysis
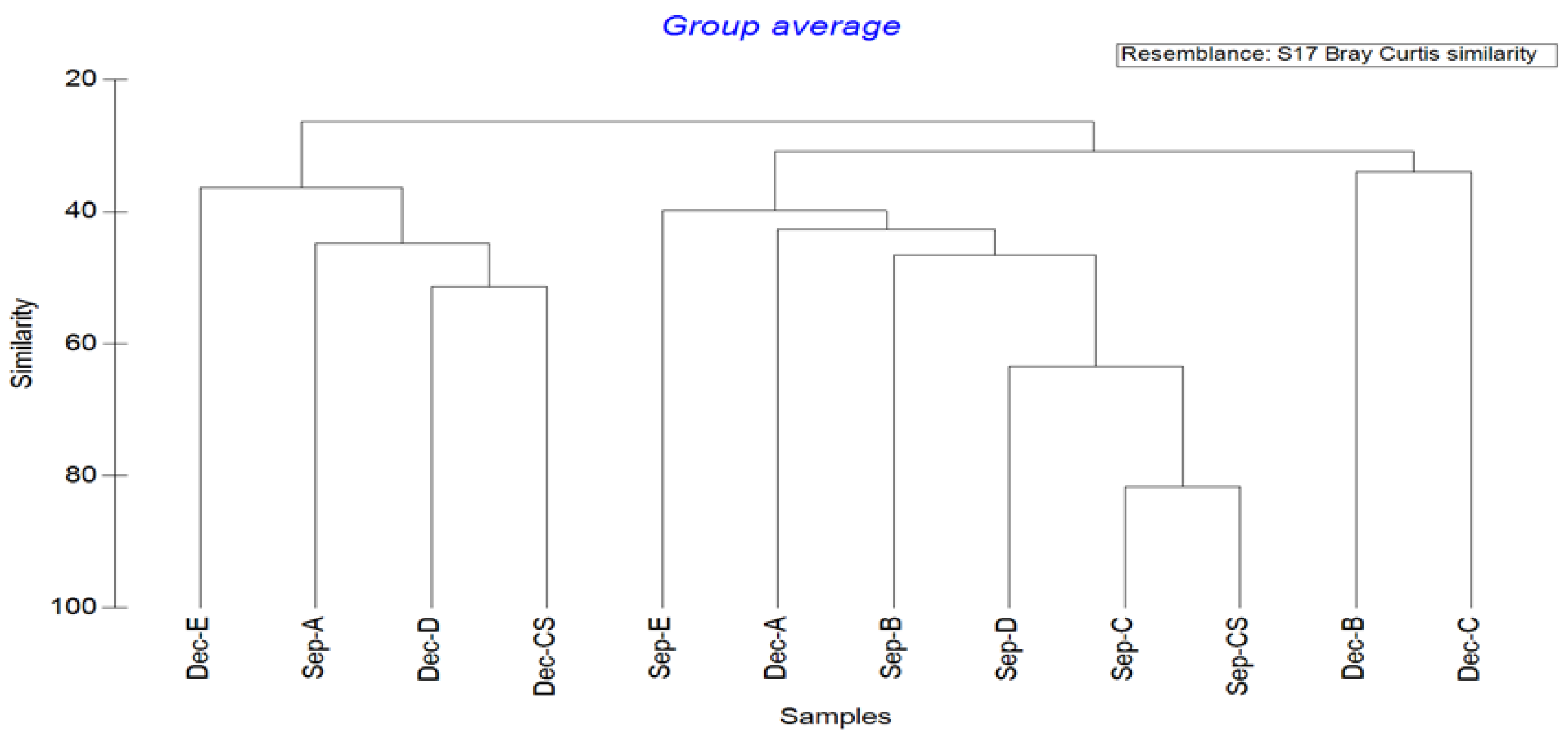
3.5. Cluster Analysis
4. Discussion
4.1. Composition, Abundance, and Biomass of Meiofauna before and after Removal of Spartina alterniflora
4.2. A Comparison of Marine Nematode Diversity and Dominant Genera between S. alterniflora and Mangrove Habitats
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, J.L.; Huang, B. Progress in the studies of the meiofauna in mangrove ecosystem. Mar. Sci. 2012, 36, 118–122. (In Chinese) [Google Scholar]
- Valiela, L.; Bowen, J.L.; York, J.K. Mangrove forests: One of the World’s threatened major tropical environments. Bioscience 2001, 51, 807–815. [Google Scholar] [CrossRef]
- Polidoro, B.A.; Carpenter, K.E.; Collins, L.; Duke, N.C.; Ellison, A.M.; Ellison, J.C.; Farnsworth, E.J.; Fernando, E.S.; Kathiresan, K.; Koedam, N.E.; et al. The loss of species: Mangrove extinction risk and geographic areas of global concern. PLoS ONE 2010, 5, e10095. [Google Scholar] [CrossRef]
- Zheng, J.Y.; Wei, H.Y.; Chen, R.D.; Liu, J.M.; Wang, L.K.; Gu, W. Invasive Trends of Spartina alterniflora in the Southeastern Coast of China and Potential Distributional Impacts on Mangrove Forests. Plants 2023, 12, 1923. [Google Scholar] [CrossRef]
- Cao, M.M.; Cui, L.N.; Sun, H.M.; Zhang, X.M.; Zheng, X.; Jiang, J. Effects of Spartina alterniflora invasion on soil microbial community structure and ecological functions. Microorganisms 2021, 9, 138. [Google Scholar] [CrossRef]
- An, S.Q.; Gu, B.H.; Zhou, C.F.; Wang, Z.S.; Deng, Z.F.; Zhi, Y.B.; Li, H.L.; Chen, L.; Yu, D.H. Spartina invasion in China: Implications for invasive species management and future research. Weed. Res. 2007, 47, 183–191. [Google Scholar] [CrossRef]
- Wang, H.H.; Wang, Y.H.; Pang, J.L.; Zhang, H.M.; Wang, J.N.; Tong, Y.W.; Guo, H.Y. Effects of nitrogen input increase on composition and diversity of culturable soil bacterial and fungal communities under Spartina alterniflora invasion in coastal wetland. Acta Sci. Nat. Univ. Nankaiensis 2022, 55, 96–105. (In Chinese) [Google Scholar]
- Wang, J.; Gu, Y.F.; You, H.P. Research Progress on the Control Measures and Utilization Status of Spartina alterniflora. Genom. Appl. Biol. 2017, 36, 3152–3156. (In Chinese) [Google Scholar]
- Lu, F.; Wang, X. General situation of the introduction and genetic diversity of Spartina alterniflora. Shandong For. Sci. Technol. 2017, 47, 107–112. (In Chinese) [Google Scholar]
- Wang, Y.M.; Gao, X.Y.; Xie, S.J.; Ma, Y.T.; Ye, T.H.; Liu, J.K.; Lin, M.Z.; Feng, J.X. Effects of Spartina alterniflora invasion on the functional groups and niche of benthic crustaceans and fishes in mangrove wetland. China J. Appl. Ecol. 2022, 33, 3016–3026. (In Chinese) [Google Scholar]
- Zhuo, W. Effects of Interspecific Competition on the Chlorophyll Inversion of Phragmites australis and Spartina alterniflora in the Coastal Wetland of the Yangtze River Estuary. Ph.D. Thesis, East China Normal University, Shanghai, China, 2022. (In Chinese). [Google Scholar]
- Zhang, Y.H.; Huang, G.M.; Wang, W.Q.; Chen, L.Z.; Lin, G.H. Interactions between mangroves and exotic Spartina in an anthropogenically disturbed estuary in southern China. Ecology 2012, 93, 588–597. [Google Scholar] [CrossRef]
- Cai, L.Z.; Li, H.M.; Zou, C.Z. Species composition and seasonal variation of marine nematodes on Futian mudflat in Shenzhen estuary. China Biodivers. 2000, 8, 385–390. (In Chinese) [Google Scholar] [CrossRef]
- Zhu, H.L.; Guo, Y.Q.; Wu, C.Y.; Zhang, L.Y. Study on meiofauna abundance in mangrove of Xien Island, Zhejiang Province. J. Jimei Univ. Nat. Sci. 2020, 25, 241–247. (In Chinese) [Google Scholar]
- Liu, J.L.; Huang, B.; Liang, Z.W. Study on abundance and biomass of benthic meiofauna in mangrove of Dongzhai Bay. Acta Oceanol. Sin. 2013, 35, 187–192. (In Chinese) [Google Scholar]
- Du, Y.F.; Gao, S.; Warwick, R.M.; Hua, E. Research progress in the ecological functions of free-living marine nematodes in coastal wetlands. Bull. Sci. Technol. 2014, 59, 3043–3060. (In Chinese) [Google Scholar]
- Guo, Y.Q.; Warwick, R.M.; Zhang, Z.N.; Mu, F.H. Freeliving marine nematodes as a pollution indicator of the Bohai Sea. J. Environ. Sci. 2022, 14, 558–562. [Google Scholar]
- Coull, B.C.; Chandler, G.T. Pollution and meiofauna: Field, laboratory and mesocosm studies. Oceanogr. Mar. Biol. Annu. Rev. 1992, 30, 191–271. [Google Scholar]
- Feng, J.X.; Huang, Q.; Chen, H.; Shen, Q.Q.; Huang, J.R.; Li, Z.F. Effects of Spartina alterniflora invasion on benthic faunal community in saltmarsh and mangrove wetland. China J. Ecol. 2018, 37, 943–951. (In Chinese) [Google Scholar]
- Pan, C.; Wu, C.Y.; Guo, Y.Q.; Yang, P.P.; Wang, H. Preliminary study on meiofauna and marine nematode community in the mangrove wetland of Techeng Island, Zhanjiang in winter. J. Fish. Res. 2020, 42, 97–104. (In Chinese) [Google Scholar]
- Peng, J.X. Ecological restoration strategy of Quanzhou Bay Estuary Wetland. For. Surv. Des. 2021, 41, 74–77. (In Chinese) [Google Scholar]
- Fang, M.J. Distribution of Spartina alterniflora in Fujian coast waters. J. Oceanogr. Taiwan Strait 2012, 31, 100–104. (In Chinese) [Google Scholar]
- Xuan, Q.N.; Smol, A.N.; Vanreusel, A.; Thanh, N.V. Biodiversity of meiofauna in the intertidal Khe Nhan mudflat, Can Gio mangrove forest, Vietnam with special emphasis on free living nematodes. Ocean Sci. J. 2007, 42, 135–152. [Google Scholar] [CrossRef]
- Olafsson, E. Meiobenthos in mangrove areas in eastern Africa with emphasis on assemblage structure of free-living marine nematodes. Hydrobiologia 1995, 312, 47–57. [Google Scholar] [CrossRef]
- Gwyther, J. Nematode assemblages from Avicennia marina leaf litter in a temperate mangrove forest in south-eastern Australia. Mar. Biol. 2003, 142, 289–297. [Google Scholar] [CrossRef]
- Xiao, Y.P.; Guo, Y.Q. Two new and one known free-living marine nematode species of the family Axonolaimidae from coastal wetlands in western Taiwan. Reg. Stud. Mar. Sci. 2023, 60, 102830. [Google Scholar] [CrossRef]
- Fu, S.J.; Cai, L.Z.; Cao, J.; Chen, X.W. Nematode Responses to the Invasion of Exotic Spartina in Mangrove Wetlands in Southern China. Estuaries Coasts 2017, 40, 1437–1449. [Google Scholar] [CrossRef]
- Cao, X.L. Impacts of Spartina alterniflora Invasions on Benthic Nematode Communities in Coastal Wetlands in China. Masters’ Thesis, Hangzhou Normal University, Hangzhou, China, 2016. (In Chinese). [Google Scholar]
- Chen, H.L. Effect of Spartina alterniflora Invasions on Nematode Communities in Salt Marshes of the Yangtze River Estuary: Patterns and Mechanisms. Ph.D. Thesis, Fudan University, Shanghai, China, 2009. (In Chinese). [Google Scholar]
- Somerfield, P.J.; Warwick, R.M. Meiofauna in Marine Pollution Monitoring Programmes: A Laboratory Manual; Directorate of Fisheries Research Technical Series; Ministry of Agriculture, Fisheries and Food: London, UK, 1996. [Google Scholar]
- Platt, H.M.; Warwick, R. Freeliving Marine Nematodes: Part II. British Chromadorida; The Linnean Society of London and The Estuarine and Coastal Sciences Association: London, UK, 1988; 502p. [Google Scholar]
- Higgins, R.P.T.H. Introduction to the Study of Meiofauna; Smithsonian Institution Press: London, UK, 1988. [Google Scholar]
- Warwick, R.; Platt, H.; Somerfield, P. Freeliving Marine Nematodes: Part III Monhysterida; Cambridge University Press: London, UK, 1998. [Google Scholar]
- Mu, F.H.; Zhang, Z.N.; Guo, Y.Q. Abundance and Biomass of the Benthic Meiofauna in the Bohai Sea. J. Ocean Univ. Qingdao 2001, 31, 897–905. (In Chinese) [Google Scholar]
- Liu, M.D.; Chen, J.C.; Guo, Y.Q.; Lu, Z.Q.; Li, X.S.; Chang, Y. Study on the individual dry weight of marine nematodes in mangrove sediments. Acta Oceanol. Sin. 2018, 40, 89–96. (In Chinese) [Google Scholar]
- McIntyre, A.D. The meiofauna and macrofauna of some tropical beaches. J. Zool. 1968, 156, 377–392. [Google Scholar] [CrossRef]
- Widbom, B. Determination of average individual dry weights and ash-free dry weights in different sieve fractions of marine meiofauna. Mar. Biol. 1984, 84, 101–108. [Google Scholar] [CrossRef]
- Chang, Y.; Guo, Y.Q. Study on Meiofauna Abundance and Nematode Diversity in Mangrove of Luoyang River, Fujian Province. J. Jimei Univ. Nat. Sci. 2014, 19, 7–12. (In Chinese) [Google Scholar]
- Yu, W.Y.; Li, C.H.; Hua, C.; Shen, Y.M.; Ma, L.; Guo, N.; Li, Z.X. The diversity analysis of macrobenthic fauna on the tidal flat with Spartina alterniflora in Dongtai, Yangcheng in Jiangsu. Trans. Oceanol. Limnol. 2009, 01, 123–128. (In Chinese) [Google Scholar]
- Wu, S.J.; Liang, S.C. Impacts of human activities on ecosystem in mangroves. Mar. Environ. Sci. 2008, 27, 537–542. (In Chinese) [Google Scholar]
- Shi, B.Z. Taxonomy of Nematodes and Community Structure of Meiofauna in Various Marine Habitats. Ph.D. Thesis, Institute of Oceanology Chinese Academy of Sciences, Qingdao, China, 2016. (In Chinese). [Google Scholar]
- Sahoo, G.; Suchiang, S.R.; Ansari, Z.A. Meiofauna-mangrove interaction: A pilot study from a tropical mangrove habitat. Cah. Biol. Mar. 2012, 54, 349–358. [Google Scholar]
- Gee, J.M.; Somerfield, P.J. Do mangrove diversity and leaf litter decay promote meiofaunal diversity? J. Exp. Mar. Biol. Ecol. 1997, 218, 13–33. [Google Scholar]
- Zou, M.M.; Guo, Y.Q.; Gan, J.C. Studies on the Community Structure of Xinying Port (Hainan Province) Mangrove Wetland Marine Nematodes in Autumn. Periodical Ocean Univ. China 2022, 52, 44–54. (In Chinese) [Google Scholar]
- Chang, Y. Study on the Biodiversity and Taxonomy of Marine Nematodes at Mangroves in Fujian Province. Master’s Thesis, Jimei University, Xiamen, China, 2015. (In Chinese). [Google Scholar]
- Nicholas, W.L.; Elek, J.A.; Stewart, A.C.; Marples, T.G. The nematode fauna of a temperate Australian mangrove mudflat: Its population, density, diversity and distribution. Hydrobiologia 1991, 209, 13–27. [Google Scholar] [CrossRef]
- Mokievsky, V.O.; Tchesunov, A.V.; Udalov, A.A.; Toam, N.D. Quantitative Distribution of Meiobenthos and the Structure of the Free-Living Nematode Community of the Mangrove Intertidal Zone in Nha Trang Bay (Vietnam) in the South China Sea. Russ. J. Mar. Biol. 2011, 37, 272–283. [Google Scholar] [CrossRef]
- Yang, P.P.; Chang, Y.; Guo, Y.Q.; Chen, J.C. Dominant genera of marine nematodes and their feeding types in several mangrove wetlands of Fujian Province. Haiyang Xuebao 2019, 41, 90–102. (In Chinese) [Google Scholar]
- Xiao, Y.P.; Guo, Y.Q.; Shi, Y.J. Study on marine nematodes community structure in mangrove wetland of Jinmen Island. J. Appl. Oceanogr. 2023, 42, 402–408. (In Chinese) [Google Scholar]
- Bianchelli, S.; Buschi, E.; Danovaro, R.; Pusceddu, A. Nematode biodiversity and benthic trophic state are simple tools for the assessment of the environmental quality in coastal marine ecosystems. Ecol. Indic. 2018, 95, 270–287. [Google Scholar] [CrossRef]
- Moreno, M.; Semprucci, F.; Vezzulli, L.; Balsamo, M.; Fabiano, M.; Albertelli, G. The use of nematodes in assessing ecological quality status in the Mediterranean coastal ecosystems. Ecol. Indic. 2010, 11, 328–336. [Google Scholar] [CrossRef]
- Semprucci, F.; Balsamo, M.; Frontalini, F. The nematode assemblage of a coastal lagoon (Lake Varano, southern Italy): Ecology and biodiversity patterns. Sci. Mar. 2014, 78, 579–588. [Google Scholar] [CrossRef]
- Hua, E.; Cui, C.Y.; Xu, H.L.; Liu, X.S. Study on the Community Characteristics of Marine Nematodes in Futian Mangrove Reserve, Shenzhen. Periodical Ocean Univ. China 2020, 50, 46–63. (In Chinese) [Google Scholar]
- Chen, Z.Z. Study on Bioremediation Technology of Spartina alterniflora in mudflat of South Fujian Coast. Anhui Agric. Sci. Bull. 2016, 22, 117–119. (In Chinese) [Google Scholar]
- Chen, F.T.; Liu, L.N.; Li, Q.X.; Yu, L.C. Impact of Spartina alterniflora Invasion on Wetland Ecological Environment and Its Control-Taking Shandong Province as an Example. China Resour. Compr. Util. 2023, 41, 109–113. (In Chinese) [Google Scholar]
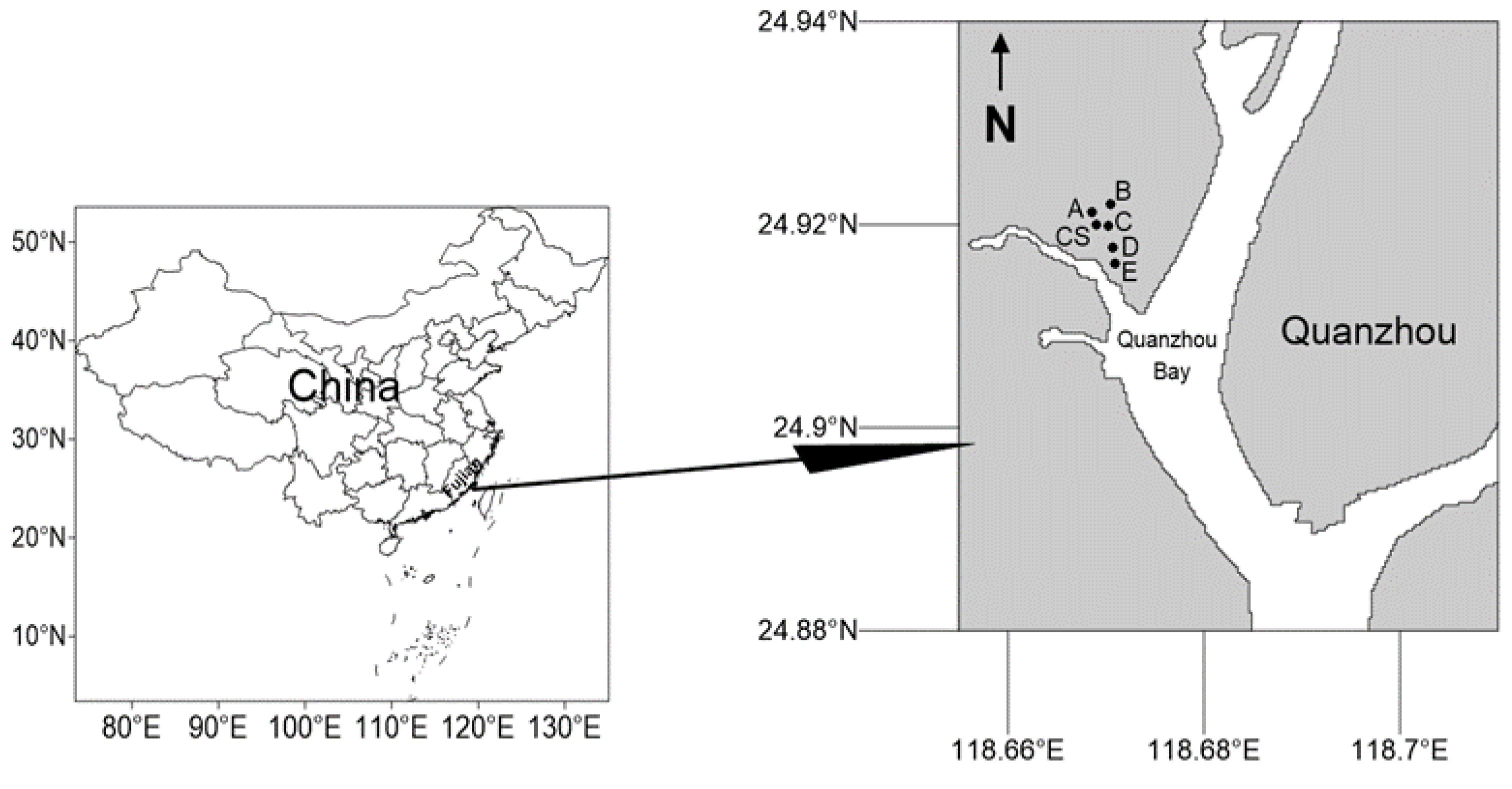
| Sampling Station | Latitude | Longitude |
|---|---|---|
| A | 24.9213 | 118.6686 |
| B | 24.9222 | 118.6699 |
| C | 24.9210 | 118.6696 |
| D | 24.9184 | 118.6702 |
| E | 24.9177 | 118.6703 |
| CS | 24.9211 | 118.6687 |
| Date | Group | Station A | Station B | Station C | Station D | Station E | Station CS | Mean |
|---|---|---|---|---|---|---|---|---|
| 2022.09 | Nematoda | 127.17 ± 155.58 | 179.91 ± 245.15 | 130.96 ± 102.41 | 327.52 ± 329.01 | 233.15 ± 107.05 | 78.98 ± 18.91 | 179.61 ± 159.69 |
| Copepoda | 0.5 ± 0 | 11.35 ± 15.34 | 12.87 ± 16.77 | 0 ± 0 | 30.03 ± 2.5 | 30.28 ± 27.83 | 14.17 ± 10.41 | |
| Oligochaeta | 0.5 ± 0.71 | 0.5 ± 0.71 | 1.51 ± 1.43 | 0 ± 0 | 0.76 ± 1.07 | 0.25 ± 0.36 | 0.59 ± 0.71 | |
| Polychaeta | 0 ± 0 | 0 ± 0 | 1.51 ± 1.43 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.25 ± 0.24 | |
| Turbellaria | 1.01 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.17 ± 0 | |
| Total | 129.19 ± 156.3 | 191.77 ± 259.78 | 146.85 ± 122.04 | 327.52 ± 329.01 | 263.93 ± 103.48 | 109.51 ± 46.39 | 194.8 ± 169.5 | |
| 2022.10 | Nematoda | 14.89 ± 10.35 | 17.92 ± 12.49 | 31.79 ± 17.13 | 126.92 ± 57.45 | 97.9 ± 44.25 | 3.03 ± 2.14 | 48.74 ± 23.97 |
| Turbellaria | 0.76 ± 1.07 | 1.01 ± 0.71 | 0.25 ± 0.36 | 9.08 ± 12.85 | 8.83 ± 3.93 | 0.5 ± 0.71 | 3.41 ± 3.27 | |
| Copepoda | 0 ± 0 | 0.76 ± 0.36 | 0 ± 0 | 0.25 ± 0.36 | 0 ± 0 | 4.04 ± 5.71 | 0.84 ± 1.07 | |
| Oligochaeta | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.25 ± 0.36 | 0 ± 0 | 0.04 ± 0.06 | |
| Total | 15.64 ± 11.42 | 19.68 ± 11.42 | 32.05 ± 17.49 | 136.26 ± 70.66 | 106.99 ± 47.82 | 7.57 ± 4.28 | 53.03 ± 27.18 | |
| 2022.12 | Nematoda | 118.59 ± 5 | 167.54 ± 42.82 | 59.55 ± 6.42 | 50.47 ± 28.55 | 110.27 ± 119.54 | 67.88 ± 19.63 | 95.72 ± 36.99 |
| Turbellaria | 4.54 ± 5 | 0 ± 0 | 1.26 ± 1.78 | 0 ± 0 | 0 ± 0 | 0.5 ± 0.71 | 1.05 ± 1.25 | |
| Copepoda | 1.01 ± 0.71 | 4.79 ± 2.5 | 17.41 ± 22.48 | 0.25 ± 0.36 | 0.25 ± 0.36 | 0.25 ± 0.36 | 4 ± 4.46 | |
| Oligochaeta | 7.07 ± 4.28 | 1.77 ± 0.36 | 1.01 ± 0 | 4.29 ± 6.07 | 3.53 ± 5 | 1.77 ± 1.07 | 3.24 ± 2.8 | |
| Total | 131.21 ± 3.57 | 174.11 ± 40.68 | 79.23 ± 27.12 | 55.01 ± 34.97 | 114.05 ± 124.18 | 70.40 ± 21.77 | 104.00 ± 42.05 | |
| 2023.03 | Nematoda | 357.8 ± 189.84 | 679.77 ± 220.53 | 811.48 ± 0.71 | 695.16 ± 335.79 | 1629.52 ± 479.6 | 1339.1 ± 246.58 | 918.8 ± 245.51 |
| Turbellaria | 1.77 ± 0.36 | 0 ± 0 | 3.53 ± 4.28 | 0.25 ± 0.36 | 2.02 ± 2.85 | 6.06 ± 2.85 | 2.27 ± 1.78 | |
| Copepoda | 74.18 ± 24.98 | 86.8 ± 27.12 | 33.56 ± 1.78 | 183.19 ± 122.75 | 219.52 ± 13.56 | 275.79 ± 23.19 | 145.51 ± 35.57 | |
| Oligochaeta | 2.52 ± 1.43 | 0 ± 0 | 0.76 ± 0.36 | 0 ± 0 | 0 ± 0 | 13.37 ± 3.93 | 2.78 ± 0.95 | |
| Polychaeta | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.25 ± 0.36 | 0 ± 0 | 0 ± 0 | 0.04 ± 0.06 | |
| Total | 436.27 ± 216.6 | 766.57 ± 247.65 | 849.33 ± 5 | 878.85 ± 459.26 | 1851.07 ± 463.18 | 1634.32 ± 230.16 | 1069.40 ± 270.31 |
| Date | Group | Station A | Station B | Station C | Station D | Station E | Station CS | Mean |
|---|---|---|---|---|---|---|---|---|
| 2022.09 | Nematoda | 101.74 ± 124.47 | 143.93 ± 196.12 | 104.77 ± 81.93 | 262.02 ± 263.21 | 186.52 ± 85.64 | 63.18 ± 15.13 | 143.69 ± 127.75 |
| Copepoda | 0.94 ± 0 | 21.12 ± 28.54 | 23.94 ± 31.2 | 0 ± 0 | 55.85 ± 4.65 | 56.32 ± 51.77 | 26.36 ± 19.36 | |
| Oligochaeta | 7.06 ± 9.98 | 7.06 ± 9.98 | 21.17 ± 19.95 | 0 ± 0 | 10.58 ± 14.97 | 3.53 ± 4.99 | 8.23 ± 9.98 | |
| Polychaeta | 0 ± 0 | 0 ± 0 | 21.2 ± 19.98 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 3.53 ± 3.33 | |
| Turbellaria | 3.53 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0.59 ± 0 | |
| Total | 113.26 ± 134.44 | 172.1 ± 214.68 | 171.06 ± 153.06 | 262.02 ± 263.21 | 252.95 ± 66.03 | 123.03 ± 61.91 | 182.4 ± 148.89 | |
| 2022.10 | Nematoda | 11.91 ± 8.28 | 14.33 ± 9.99 | 25.43 ± 13.7 | 101.54 ± 45.96 | 78.32 ± 35.4 | 2.42 ± 1.71 | 38.99 ± 19.17 |
| Turbellaria | 2.65 ± 3.75 | 3.53 ± 2.5 | 0.88 ± 1.25 | 31.79 ± 44.96 | 30.91 ± 13.74 | 1.77 ± 2.5 | 11.92 ± 11.45 | |
| Copepoda | 0 ± 0 | 1.41 ± 0.66 | 0 ± 0 | 0.47 ± 0.66 | 0 ± 0 | 7.51 ± 10.62 | 1.56 ± 1.99 | |
| Oligochaeta | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 3.53 ± 4.99 | 0 ± 0 | 0.59 ± 0.83 | |
| Total | 14.56 ± 12.03 | 19.27 ± 6.83 | 26.32 ± 14.95 | 133.8 ± 91.59 | 112.76 ± 44.15 | 11.7 ± 11.4 | 53.07 ± 30.16 | |
| 2022.12 | Nematoda | 94.87 ± 4 | 134.04 ± 34.26 | 47.64 ± 5.14 | 40.37 ± 22.84 | 88.21 ± 95.63 | 54.3 ± 15.7 | 76.57 ± 29.59 |
| Turbellaria | 15.9 ± 17.49 | 0 ± 0 | 4.42 ± 6.24 | 0 ± 0 | 0 ± 0 | 1.77 ± 2.5 | 3.68 ± 4.37 | |
| Copepoda | 1.88 ± 1.33 | 8.92 ± 4.65 | 32.38 ± 41.81 | 0.47 ± 0.66 | 0.47 ± 0.66 | 0.47 ± 0.66 | 7.43 ± 8.3 | |
| Oligochaeta | 98.77 ± 59.86 | 24.69 ± 4.99 | 14.11 ± 0 | 59.97 ± 84.81 | 49.39 ± 69.84 | 24.69 ± 14.97 | 45.27 ± 39.08 | |
| Total | 211.42 ± 45.05 | 167.65 ± 34.6 | 98.55 ± 40.71 | 100.81 ± 108.31 | 138.07 ± 164.81 | 81.23 ± 33.83 | 132.59 ± 71.22 | |
| 2023.03 | Nematoda | 286.24 ± 151.87 | 543.81 ± 176.42 | 649.19 ± 0.57 | 556.13 ± 268.63 | 1303.62 ± 383.68 | 1071.28 ± 197.26 | 735.04 ± 196.41 |
| Turbellaria | 6.18 ± 1.25 | 0 ± 0 | 12.36 ± 14.99 | 0.88 ± 1.25 | 7.07 ± 9.99 | 21.2 ± 9.99 | 7.95 ± 6.24 | |
| Copepoda | 137.98 ± 46.46 | 161.45 ± 50.44 | 62.42 ± 3.32 | 340.73 ± 228.32 | 408.31 ± 25.22 | 512.97 ± 43.14 | 270.65 ± 66.15 | |
| Oligochaeta | 35.28 ± 19.95 | 0 ± 0 | 10.58 ± 4.99 | 0 ± 0 | 0 ± 0 | 186.96 ± 54.88 | 38.8 ± 13.3 | |
| Polychaeta | 0 ± 0 | 0 ± 0 | 0 ± 0 | 3.53 ± 5 | 0 ± 0 | 0 ± 0 | 0.59 ± 0.83 | |
| Total | 465.68 ± 219.54 | 705.26 ± 226.87 | 734.55 ± 12.75 | 901.27 ± 503.2 | 1719 ± 348.46 | 1792.41 ± 218.99 | 1053.03 ± 254.97 |
| Dominant Genera | Percentage of Dominances (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Station A | Station B | Station C | Station D | Station E | Station CS | |||||||
| Sep. | Dec. | Sep. | Dec. | Sep. | Dec. | Sep. | Dec. | Sep. | Dec. | Sep. | Dec. | |
| Ptycholaimellus | 2.00 | 7.76 | 6.43 | 22.31 | 53.30 | 22.37 | 34.22 | 1.30 | 11.83 | 1.13 | 53.37 | 4.55 |
| Parodontophora | 11.00 | 18.10 | 26.32 | 4.55 | 20.81 | 2.63 | 31.55 | 14.29 | 11.83 | 8.65 | 16.56 | 22.73 |
| Terschellingia | 49.00 | - | 9.94 | - | 2.54 | 1.32 | 5.35 | 35.06 | 6.45 | 9.02 | 2.45 | 36.36 |
| Halichoanolaimus | - | 2.59 | 25.15 | - | 5.08 | - | 2.14 | 6.49 | 3.23 | 1.13 | 1.84 | - |
| Metachromadora | 4.50 | 4.74 | 3.51 | - | 3.05 | - | - | 7.79 | 18.28 | 6.39 | 1.84 | - |
| Parasphaerolaimus | 1.00 | - | 5.26 | - | 0.51 | 1.32 | - | - | 20.97 | 2.26 | 3.07 | - |
| Daptonema | - | - | - | - | - | - | - | - | - | 56.39 | - | 10.00 |
| Admirandus | - | 3.02 | - | 39.26 | 0.51 | 28.95 | - | - | 4.30 | - | 1.23 | - |
| Anoplostoma | - | 14.66 | 2.34 | 18.60 | 7.11 | 3.95 | 2.67 | 3.90 | 0.54 | - | 11.04 | - |
| Station | Species Number (S) | Margalef’s Species Richness Index (D) | Pielou’s Evenness Index (J’) | Shannon–Wiener Index (H’) | Dominance Index (1 − λ) |
|---|---|---|---|---|---|
| Sep-A | 12 | 2.08 | 0.68 | 1.69 | 0.72 |
| Sep-B | 16 | 2.92 | 0.78 | 2.17 | 0.84 |
| Sep-C | 12 | 2.08 | 0.61 | 1.51 | 0.66 |
| Sep-D | 11 | 1.91 | 0.7 | 1.67 | 0.76 |
| Sep-E | 14 | 2.49 | 0.84 | 2.21 | 0.87 |
| Sep-CS | 13 | 2.36 | 0.63 | 1.61 | 0.68 |
| Dec-A | 18 | 3.12 | 0.86 | 2.49 | 0.9 |
| Dec-B | 12 | 2 | 0.69 | 1.72 | 0.76 |
| Dec-C | 14 | 3 | 0.76 | 2 | 0.83 |
| Dec-D | 11 | 2.3 | 0.85 | 2.05 | 0.83 |
| Dec-E | 14 | 2.32 | 0.63 | 1.66 | 0.66 |
| Dec-CS | 11 | 2.12 | 0.76 | 1.82 | 0.79 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, M.-C.; Guo, Y.-Q.; Shih, Y.-J.; Liu, K.; Li, C.-X.; Ji, F.-F.; Chu, T.-J. Comparison of the Meiofauna and Marine Nematode Communities before and after Removal of Spartina alterniflora in the Mangrove Wetland of Quanzhou Bay, Fujian Province. Water 2023, 15, 3829. https://doi.org/10.3390/w15213829
Hu M-C, Guo Y-Q, Shih Y-J, Liu K, Li C-X, Ji F-F, Chu T-J. Comparison of the Meiofauna and Marine Nematode Communities before and after Removal of Spartina alterniflora in the Mangrove Wetland of Quanzhou Bay, Fujian Province. Water. 2023; 15(21):3829. https://doi.org/10.3390/w15213829
Chicago/Turabian StyleHu, Ming-Cheng, Yu-Qing Guo, Yi-Jia Shih, Kai Liu, Chun-Xue Li, Fen-Fen Ji, and Ta-Jen Chu. 2023. "Comparison of the Meiofauna and Marine Nematode Communities before and after Removal of Spartina alterniflora in the Mangrove Wetland of Quanzhou Bay, Fujian Province" Water 15, no. 21: 3829. https://doi.org/10.3390/w15213829





