Phenol Removal from Wastewater Using Tyrosinase Enzyme Immobilized in Granular Activated Carbon and Activated Chitosan Beads
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Tyrosinase Enzyme Obtaining Methodology
2.3. Supports and Immobilization of Enzymes in the Crude Extract
2.4. Enzymatic Oxidation of Phenol Using Immobilized Enzymes
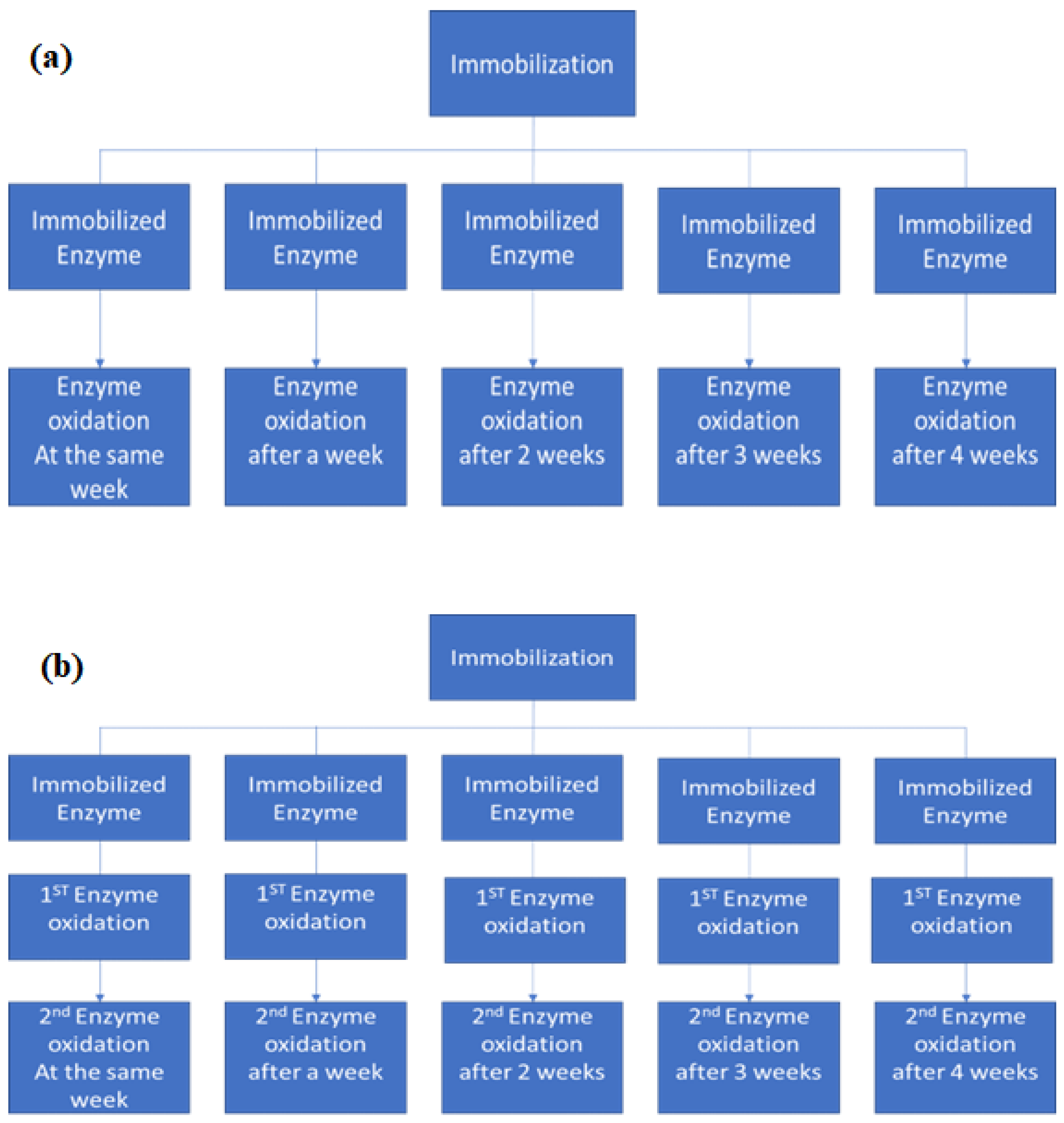
2.5. Reuse and Post-Storage Stability of the Materials
3. Results and Discussion
3.1. Materials’ Characterization
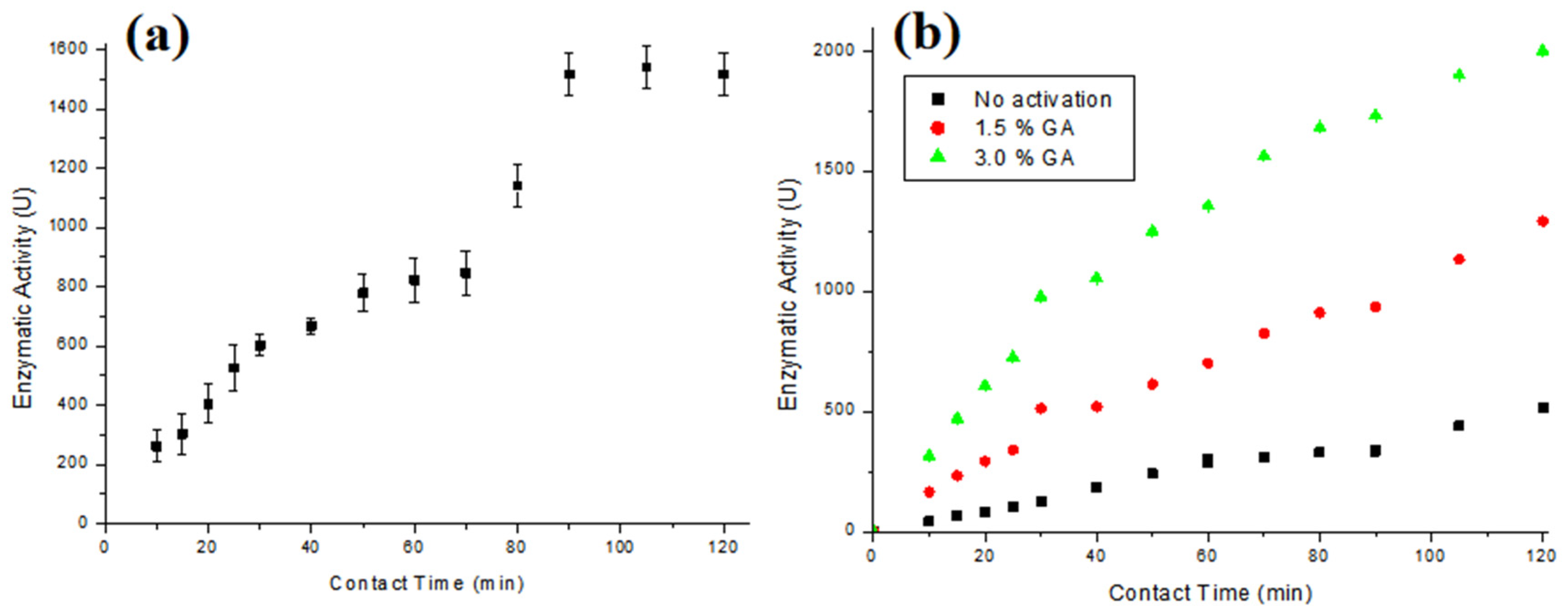
3.2. Enzyme Immobilization
3.3. Enzymatic Oxidation Assays
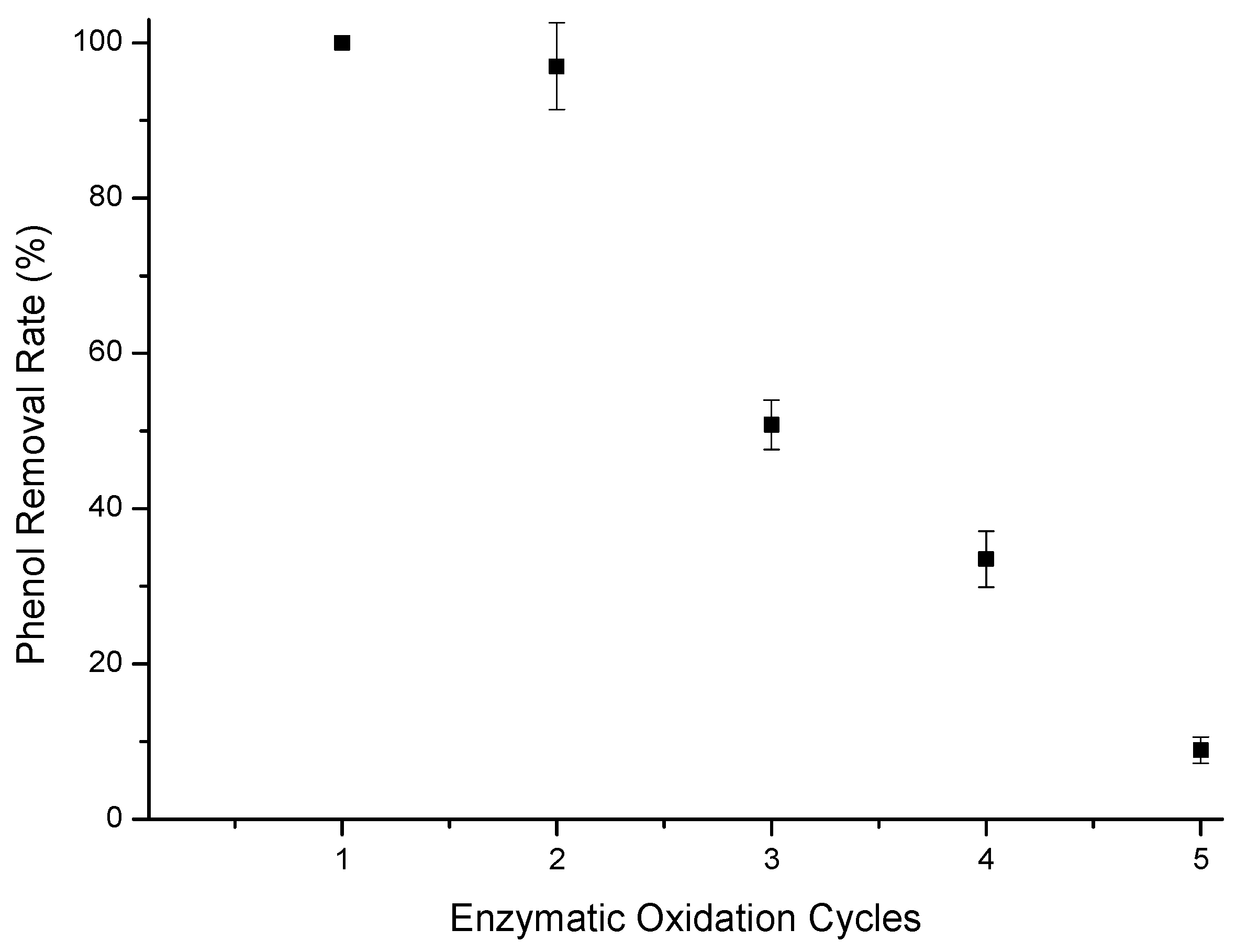
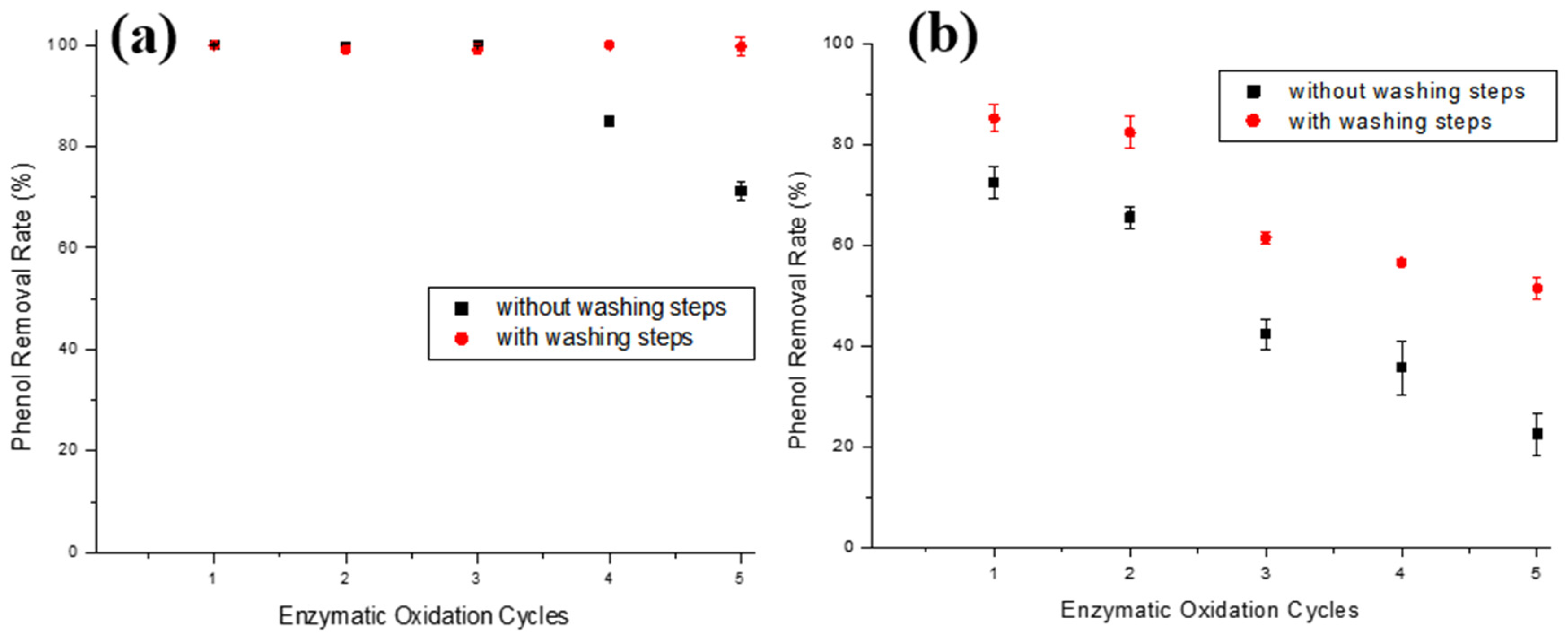
3.4. Reuse of Materials and Supports
3.4.1. Immobilized Enzyme Reuse
3.4.2. Support Reuse
3.5. Immobilized Enzyme Storage
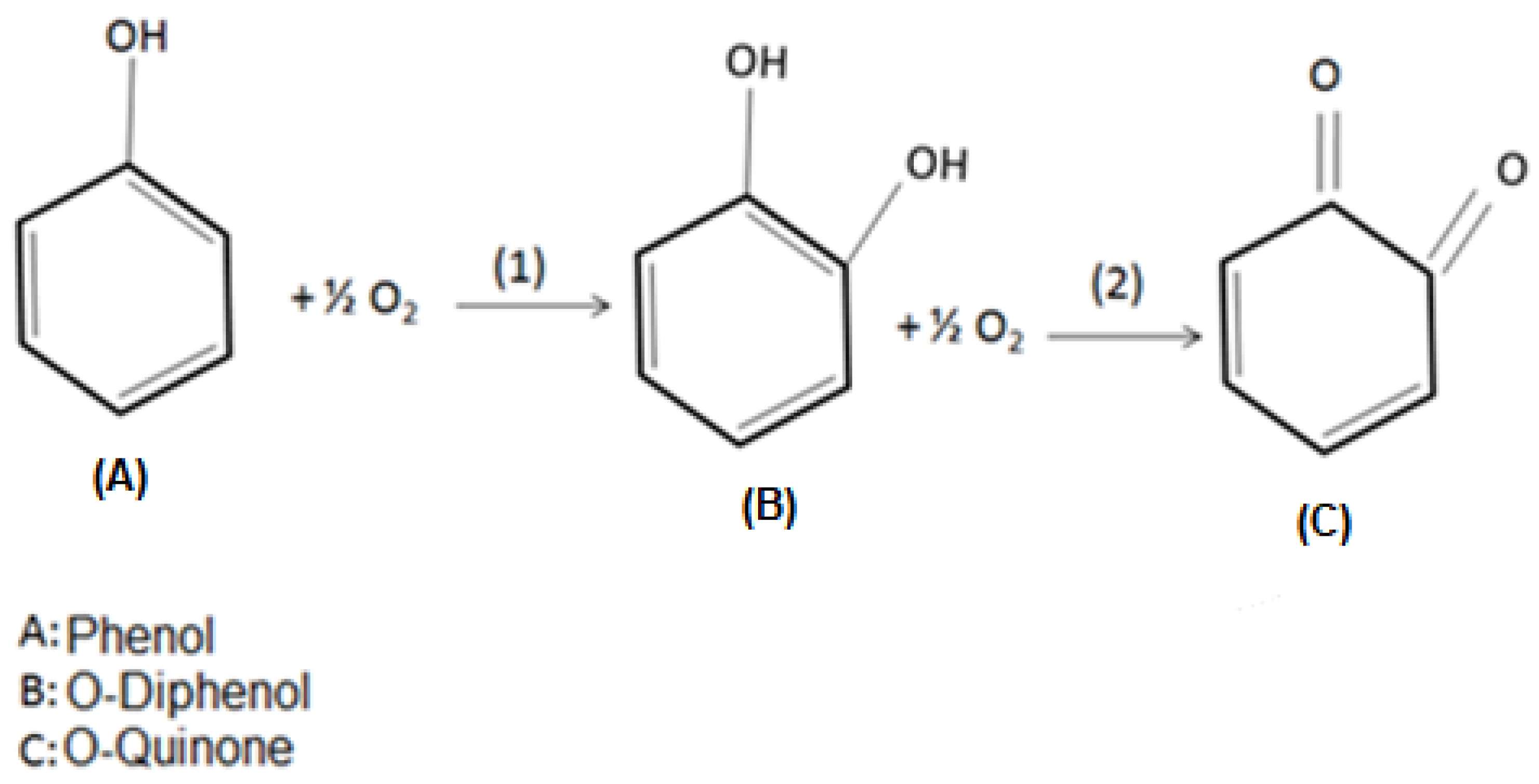
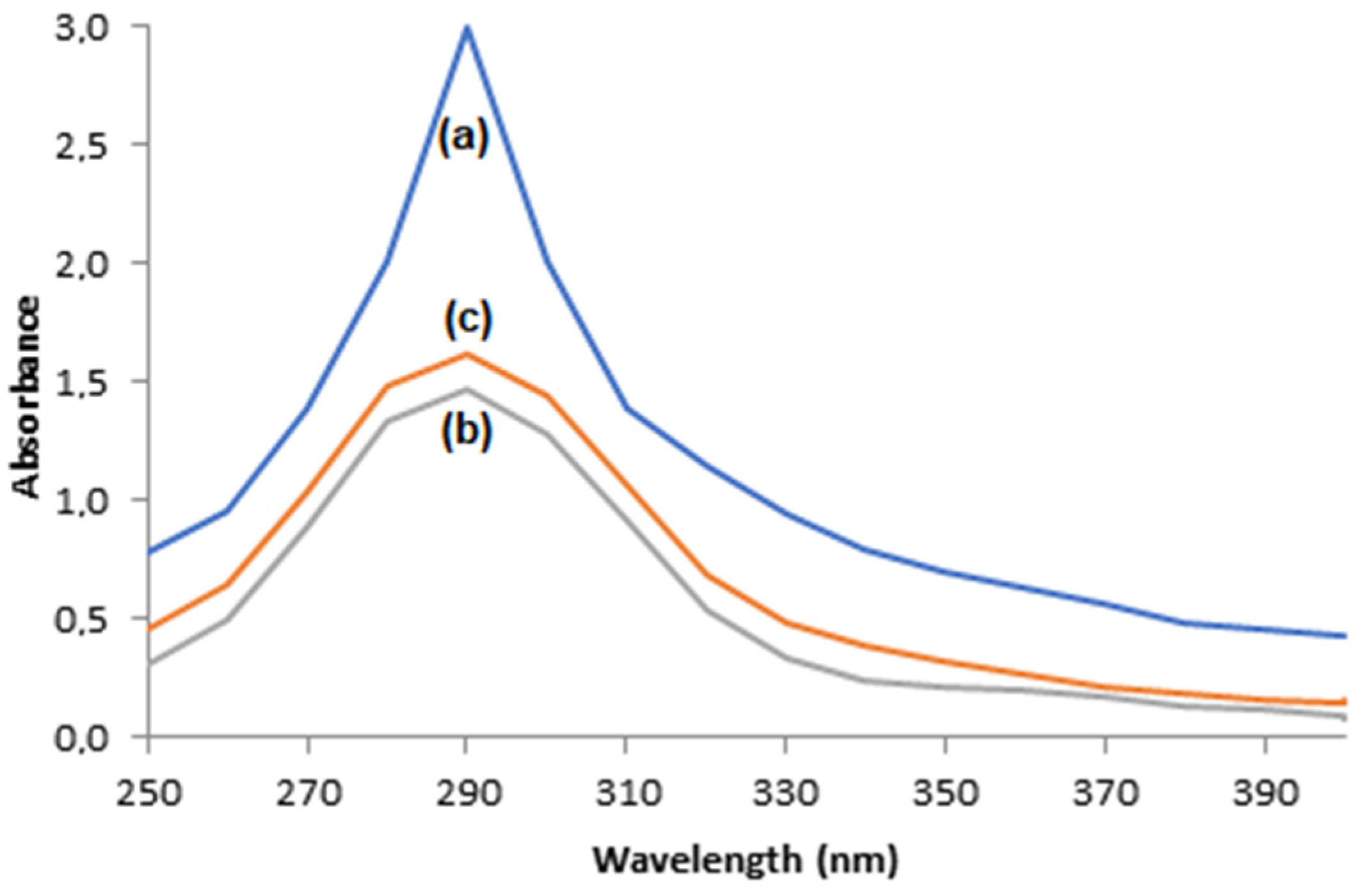
3.6. O-Quinone Production and Adsorption via the Supports
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hasan, M.K.; Shahriar, A.; Jim, K.U. Water pollution in Bangladesh and its impact on public health. Heliyon 2019, 5, e02145. [Google Scholar] [CrossRef] [PubMed]
- WWAP. The United Nations World Water Development Report 4: Managing Water under Uncertainty and Risk; WWAP: Paris, France, 2012. [Google Scholar]
- Motamedi, M.; Yerushalmi, L.; Haghighat, F.; Chen, Z. Recent developments in photocatalysis of industrial effluents ։ A review and example of phenolic compounds degradation. Chemosphere 2022, 296, 133688. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, S.; Kargari, A.; Sanaeepur, H.; Abbassian, K.; Najafi, A.; Mofarrah, E. Phenol removal from industrial wastewaters: A short review. Desalin. Water Treat. 2013, 53, 2215–2234. [Google Scholar] [CrossRef]
- Hansch, C.; McKarns, S.C.; Smith, C.J.; Doolittle, D.J. Comparative QSAR evidence for a free-radical mechanism of phenol-induced toxicity. Chem. Biol. Interact. 2000, 127, 61–72. [Google Scholar] [CrossRef]
- Boyd, E.M.; Killham, K.; Meharg, A. Toxicity of mono-, di- and tri-chlorophenols to lux marked terrestrial bacteria, Burkholderia species Rasc c2 and Pseudomonas fluorescens. Chemosphere 2001, 43, 157–166. [Google Scholar] [CrossRef]
- Ertürk, M.D.; Saçan, M.T.; Novic, M.; Minovski, N. Quantitative structure–activity relationships (QSARs) using the novel marine algal toxicity data of phenols. J. Mol. Graph. Model. 2012, 38, 90–100. [Google Scholar] [CrossRef]
- Said, K.A.M.; Ismail, A.F.; Karim, Z.A.; Abdullah, M.S.; Hafeez, A. A review of technologies for the phenolic compounds recovery and phenol removal from wastewater. Process. Saf. Environ. Prot. 2021, 151, 257–289. [Google Scholar] [CrossRef]
- Ilavský, J.; Hrivnák, J.; Barlokov, D. Analysis of chlorinated phenols in water. Food Environ. Saf. 2012, 11, 5–14. [Google Scholar]
- Chae, Y.; Kim, L.; Kim, D.; Cui, R.; Lee, J.; An, Y.-J. Deriving hazardous concentrations of phenol in soil ecosystems using a species sensitivity distribution approach. J. Hazard. Mater. 2020, 399, 123036. [Google Scholar] [CrossRef]
- Li, C.; Deng, W.; Gao, C.; Xiang, X.; Feng, X.; Batchelor, B.; Li, Y. Membrane distillation coupled with a novel two-stage pretreatment process for petrochemical wastewater treatment and reuse. Sep. Purif. Technol. 2019, 224, 23–32. [Google Scholar] [CrossRef]
- USEPA. Federal Register; USEPA: Washington, DC, USA, 1987. [Google Scholar]
- CONAMA. Brazilian National Council for the Environment, Resolution #430; CONAMA: Brasília, Brazil, 2011; 9p. (In Portuguese) [Google Scholar]
- Pandey, K.; Singh, B.; Pandey, A.K.; Badruddin, I.J.; Pandey, S.; Mishra, V.K.; Jain, P.A. Application of Microbial Enzymes in Industrial Waste Water Treatment. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1243–1254. [Google Scholar] [CrossRef]
- Xu, D.-Y.; Yang, Z. Cross-linked tyrosinase aggregates for elimination of phenolic compounds from wastewater. Chemosphere 2013, 92, 391–398. [Google Scholar] [CrossRef]
- Erhan, E.; Keskinler, B.; Akay, G.; Algur, O. Removal of phenol from water by membrane-immobilized enzymes: Part I. Dead-end filtration. J. Membr. Sci. 2002, 206, 361–373. [Google Scholar] [CrossRef]
- Singh, S.; Mishra, R.; Sharma, R.S.; Mishra, V. Phenol remediation by peroxidase from an invasive mesquite: Turning an environmental wound into wisdom. J. Hazard. Mater. 2017, 334, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Villegas, L.G.C.; Mashhadi, N.; Chen, M.; Mukherjee, D.; Taylor, K.E.; Biswas, N. A Short Review of Techniques for Phenol Removal from Wastewater. Curr. Pollut. Rep. 2016, 2, 157–167. [Google Scholar] [CrossRef]
- Eş, I.; Vieira, J.D.G.; Amaral, A.C. Principles, techniques, and applications of biocatalyst immobilization for industrial application. Appl. Microbiol. Biotechnol. 2015, 99, 2065–2082. [Google Scholar] [CrossRef]
- Yamada, K.; Inoue, T.; Akiba, Y.; Kashiwada, A.; Matsuda, K.; Hirata, M. Removal of p-Alkylphenols from Aqueous Solutions by Combined Use of Mushroom Tyrosinase and Chitosan Beads. Biosci. Biotechnol. Biochem. 2006, 70, 2467–2475. [Google Scholar] [CrossRef]
- Wu, J.C.Y.; Hutchings, C.H.; Lindsay, M.J.; Werner, C.J.; Bundy, B.C. Enhanced Enzyme Stability Through Site-Directed Covalent Immobilization. J. Biotechnol. 2015, 193, 83–90. [Google Scholar] [CrossRef]
- Abdollahi, K.; Yazdani, F.; Panahi, R. Covalent immobilization of tyrosinase onto cyanuric chloride crosslinked amine-functionalized superparamagnetic nanoparticles: Synthesis and characterization of the recyclable nanobiocatalyst. Int. J. Biol. Macromol. 2017, 94, 396–405. [Google Scholar] [CrossRef]
- Mollaei, M.; Abdollahpour, S.; Atashgahi, S.; Abbasi, H.; Masoomi, F.; Rad, I.; Lotfi, A.S.; Zahiri, H.S.; Vali, H.; Noghabi, K.A. Enhanced phenol degradation by Pseudomonas sp. SA01: Gaining insight into the novel single and hybrid immobilizations. J. Hazard. Mater. 2010, 175, 284–292. [Google Scholar] [CrossRef]
- El-aziz, S.M.A.; Hassan, A.; Faraag, I. Tyrosinase Enzyme Purification and Immobilization from Pseudomonas sp. EG22 Using Cellulose Coated Magnetic Nanoparticles : Characterization of Bioactivity in Melanin Product. World J. Microbiol. Biotechnol. 2023; preprints. [Google Scholar] [CrossRef]
- Liu, D.-M.; Dong, C. Recent advances in nano-carrier immobilized enzymes and their applications. Process. Biochem. 2020, 92, 464–475. [Google Scholar] [CrossRef]
- Kameda, E.; Langone, M.A.P.; Coelho, M.A.Z. Tyrosinase Extract from Agaricus bisporus Mushroom and itsin Natura Tissue for Specific Phenol Removal. Environ. Technol. 2006, 27, 1209–1215. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, V.P.S.; Silva, L.M.C.; Salgado, A.M.; Pereira, K.S. Application of agaricus bisporus extract for benzoate sodium detection based on tyrosinase inhibition for biosensor development. Chem. Eng. Trans. 2013, 32, 1831–1836. [Google Scholar] [CrossRef]
- Silvestre, M.P.C.; Silva, V.D.M.; de Marco, L.M.; Delvivo, F.M.; Coelho, J.V. Imobilização da pancreatina em carvão ativado e em alumina para o preparo de hidrolisados de soro de leite. Acta Sci. Health Sci. 2005, 27, 163–169. [Google Scholar] [CrossRef]
- Kennedy, L.J.; Selvi, P.; Padmanabhan, A.; Hema, K.; Sekaran, G. Immobilization of polyphenol oxidase onto mesoporous activated carbons-isotherm and kinetic studies. Chemosphere 2007, 69, 262–270. [Google Scholar] [CrossRef]
- Kumar, A.G.; Perinbam, K.; Kamatchi, P.; Nagesh, N.; Sekaran, G. In situ immobilization of acid protease on mesoporous activated carbon packed column for the production of protein hydrolysates. Bioresour. Technol. 2010, 101, 1377–1379. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, Y.; Pan, S.; Wu, T.; Tang, X. Optimal immobilization of β-glucosidase into chitosan beads using response surface methodology. Electron. J. Biotechnol. 2013, 16. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 22nd ed.; APHA: Washington, DC, USA, 2005. [Google Scholar]
- Tuncay, F.O.; Cakmak, U.; Kolcuoğlu, Y. Rhaphiolepis indica (L.) Lindl. Fruit: LC-HRMS-based phytochemical profile, FTIR spectral, in vitro enzyme inhibition and antioxidant analysis. Food Biosci. 2023, 56, 103228. [Google Scholar] [CrossRef]
- Romero, G.; Contreras, L.M.; Céspedes, C.A.; Wilkesman, J.; Clemente-Jiménez, J.M.; Rodríguez-Vico, F.; Heras-Vázquez, F.J.L. Efficiency Assessment between Entrapment and Covalent Bond Immobilization of Mutant β-Xylosidase onto Chitosan Support. Polymers 2023, 15, 3170. [Google Scholar] [CrossRef]
- Chavita, A.C. Estudo da Remoção de Fenóis de Soluções aquosas através da Adsorção em Quitosana, Degradação Enzimática por Tirosinase e Imobilização de Tirosinase em Matriz de Quitosana. Master’s Thesis, University of São Paulo, São Paulo, Brazil, 2010. (In Portuguese). [Google Scholar]
- Miyaguti, R.M. Oxidação Enzimática de Soluções Fenólicas com Tirosinase Imobilizada em Quitosana Oxidação Enzimática de Soluções Fenólicas com Tirosinase Imobilizada em Quitosana. Master’s Thesis, University of São Paulo, São Paulo, Brazil, 2011. (In Portuguese). [Google Scholar]
- Colombié, S.; Gaunand, A.; Lindet, B. Lysozyme inactivation under mechanical stirring: Effect of physical and molecular interfaces. Enzym. Microb. Technol. 2001, 28, 820–826. [Google Scholar] [CrossRef]
- Gikanga, B.; Hui, A.; Maa, Y.-F. Mechanistic Investigation on Grinding-Induced Subvisible Particle Formation during Mixing and Filling of Monoclonal Antibody Formulations. PDA J. Pharm. Sci. Technol. 2017, 72, 117–133. [Google Scholar] [CrossRef] [PubMed]
- Menoncin, S.; Domingues, N.M.; Freire, D.M.G.; Oliveira, J.V.; Di Luccio, M.; Treichel, H.; de Oliveira, D. Imobilização de lipases produzidas por fermentação em estado sólido utilizando Penicillium verrucosum em suportes hidrofóbicos. Food Sci. Technol. 2009, 29, 440–443. [Google Scholar] [CrossRef]
- Wiesbauer, J.; Cardinale, M.; Nidetzky, B. Shaking and stirring: Comparison of controlled laboratory stress conditions applied to the human growth hormone. Process. Biochem. 2013, 48, 33–40. [Google Scholar] [CrossRef]
- Bevilaqua, J.; Cammarota, M.; Freire, D.M.G.; Sant’Anna, G.L. Phenol removal through combined biological and enzymatic treatments. Braz. J. Chem. Eng. 2002, 19, 151–158. [Google Scholar] [CrossRef]
- Wada, S.; Ichikawa, H.; Tatsumi, K. Removal of phenols from wastewater by soluble and immobilized tyrosinase. Biotechnol. Bioeng. 1993, 42, 854–858. [Google Scholar] [CrossRef]
- Pigatto, G. Study on Tyrosinase Application in the Treatment of Plant Effluent and Verification of Genotoxicity of the Treated Effluent into Plant Cells. Ph.D. Thesis, University of São Paulo, São Paulo, Brazil, 2013. (In Portuguese). [Google Scholar]
- García-Molina, P.; García-Molina, F.; Teruel-Puche, J.A.; Rodríguez-López, J.N.; García-Cánovas, F.; Muñoz-Muñoz, J.L. Considerations about the kinetic mechanism of tyrosinase in its action on monophenols: A review. Mol. Catal. 2022, 518, 112072. [Google Scholar] [CrossRef]
- Choi, S.; Ahn, H.; Kim, S. Tyrosinase-mediated hydrogel crosslinking for tissue engineering. J. Appl. Polym. Sci. 2021, 139, 51887. [Google Scholar] [CrossRef]
- Lee, P.-G.; Lee, S.-H.; Hong, E.Y.; Lutz, S.; Kim, B.-G. Circular permutation of a bacterial tyrosinase enables efficient polyphenol-specific oxidation and quantitative preparation of orobol. Biotechnol. Bioeng. 2018, 116, 19–27. [Google Scholar] [CrossRef]
- Gulaboski, R.; Bogeski, I.; Mirčeski, V.; Saul, S.; Pasieka, B.; Haeri, H.H.; Stefova, M.; Stanoeva, J.P.; Mitrev, S.; Hoth, M.; et al. Hydroxylated derivatives of dimethoxy-1,4-benzoquinone as redox switchable earth-alkaline metal ligands and radical scavengers. Sci. Rep. 2013, 3, srep01865. [Google Scholar] [CrossRef]
- Ito, S.; Sugumaran, M.; Wakamatsu, K. Chemical Reactivities of ortho-Quinones Produced in Living Organisms: Fate of Quinonoid Products Formed by Tyrosinase and Phenoloxidase Action on Phenols and Catechols. Int. J. Mol. Sci. 2020, 21, 6080. [Google Scholar] [CrossRef]
- Qu, Y.; Zhan, Q.; Du, S.; Ding, Y.; Fang, B.; Du, W.; Wu, Q.; Yu, H.; Li, L.; Huang, W. Catalysis-based specific detection and inhibition of tyrosinase and their application. J. Pharm. Anal. 2020, 10, 414–425. [Google Scholar] [CrossRef] [PubMed]







| Stirring (rad/s) | Enzymatic Activity (U) | Immobilization Degree |
|---|---|---|
| 10.5 | 900 | 0.30 |
| 15.7 | 1650 | 0.55 |
| 20.9 | 1590 | 0.53 |
| 31.4 | 1200 | 0.40 |
| T-GAC | |||
|---|---|---|---|
| Enzymatic Activity (U) | Initial Phenol Concentration (mg/L) | Final Phenol Concentration (mg/L) | Phenol Removal (%) |
| ~1500 | 10.0 | ~0 | 100 |
| ~1500 | 20.0 | 2.5 | 87.5 |
| ~1500 | 40.0 | 6.7 | 83.3 |
| ~1500 | 60.0 | 19.5 | 67.5 |
| ~1500 | 100.0 | 45.6 | 54.4 |
| T-ACB | |||
| Enzymatic activity (U) | Initial phenol concentration (mg/L) | Final phenol concentration (mg/L) | Phenol removal (%) |
| ~1500 | 10.0 | ~0 | 100 |
| ~1500 | 20.0 | ~0 | 100 |
| ~1500 | 40.0 | 0.5 | 97.5 |
| ~1500 | 60.0 | 1.7 | 95.8 |
| ~1500 | 100.0 | 32.1 | 67.9 |
| Support Usage | Initial Enzymatic Activity (U) | Final Enzymatic Activity (U) | Immobilization Degree |
|---|---|---|---|
| 1 | 3000 | 1440.0 | 0.52 |
| 2 | 3015 | 1581.7 | 0.48 |
| 3 | 3002 | 1701.2 | 0.43 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Mello, A.C.C.; da Silva, F.P.; Gripa, E.; Salgado, A.M.; da Fonseca, F.V. Phenol Removal from Wastewater Using Tyrosinase Enzyme Immobilized in Granular Activated Carbon and Activated Chitosan Beads. Water 2023, 15, 3778. https://doi.org/10.3390/w15213778
de Mello ACC, da Silva FP, Gripa E, Salgado AM, da Fonseca FV. Phenol Removal from Wastewater Using Tyrosinase Enzyme Immobilized in Granular Activated Carbon and Activated Chitosan Beads. Water. 2023; 15(21):3778. https://doi.org/10.3390/w15213778
Chicago/Turabian Stylede Mello, Ana Carina Cruz, Felipe Pereira da Silva, Everton Gripa, Andrea Medeiros Salgado, and Fabiana Valéria da Fonseca. 2023. "Phenol Removal from Wastewater Using Tyrosinase Enzyme Immobilized in Granular Activated Carbon and Activated Chitosan Beads" Water 15, no. 21: 3778. https://doi.org/10.3390/w15213778





