The Taxon-Specific Species Sensitivity and Aquatic Ecological Risk Assessment of Three Heavy Metals in Songhua River Water, China
Abstract
:1. Introduction
2. Materials and Methods
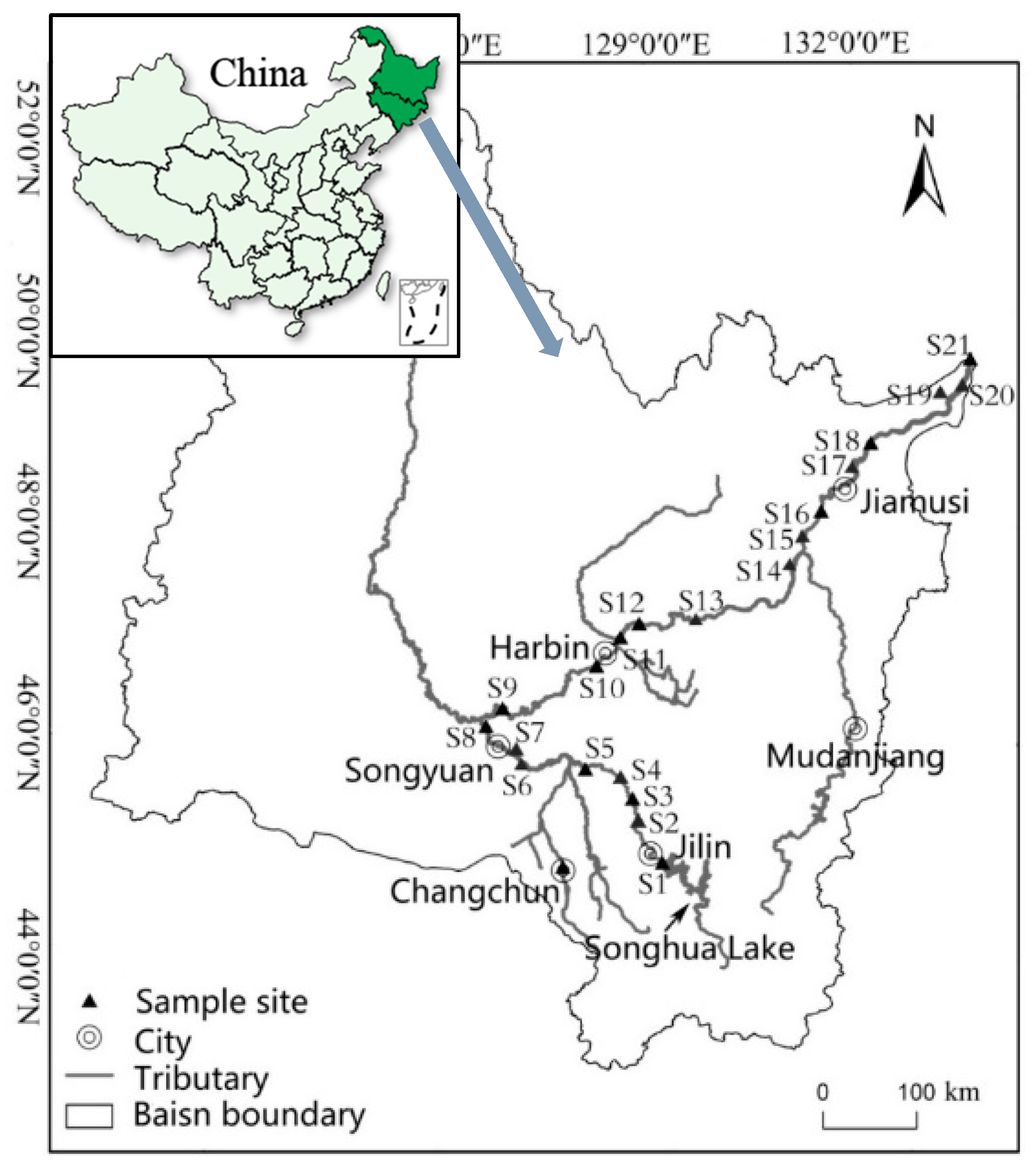
2.1. Exposure Concentration of Three Metals
2.2. Toxicity Data Collection and Species Sensitivity Analysis
2.3. Ecological Risk Assessment of Three Metals
3. Results and Discussion
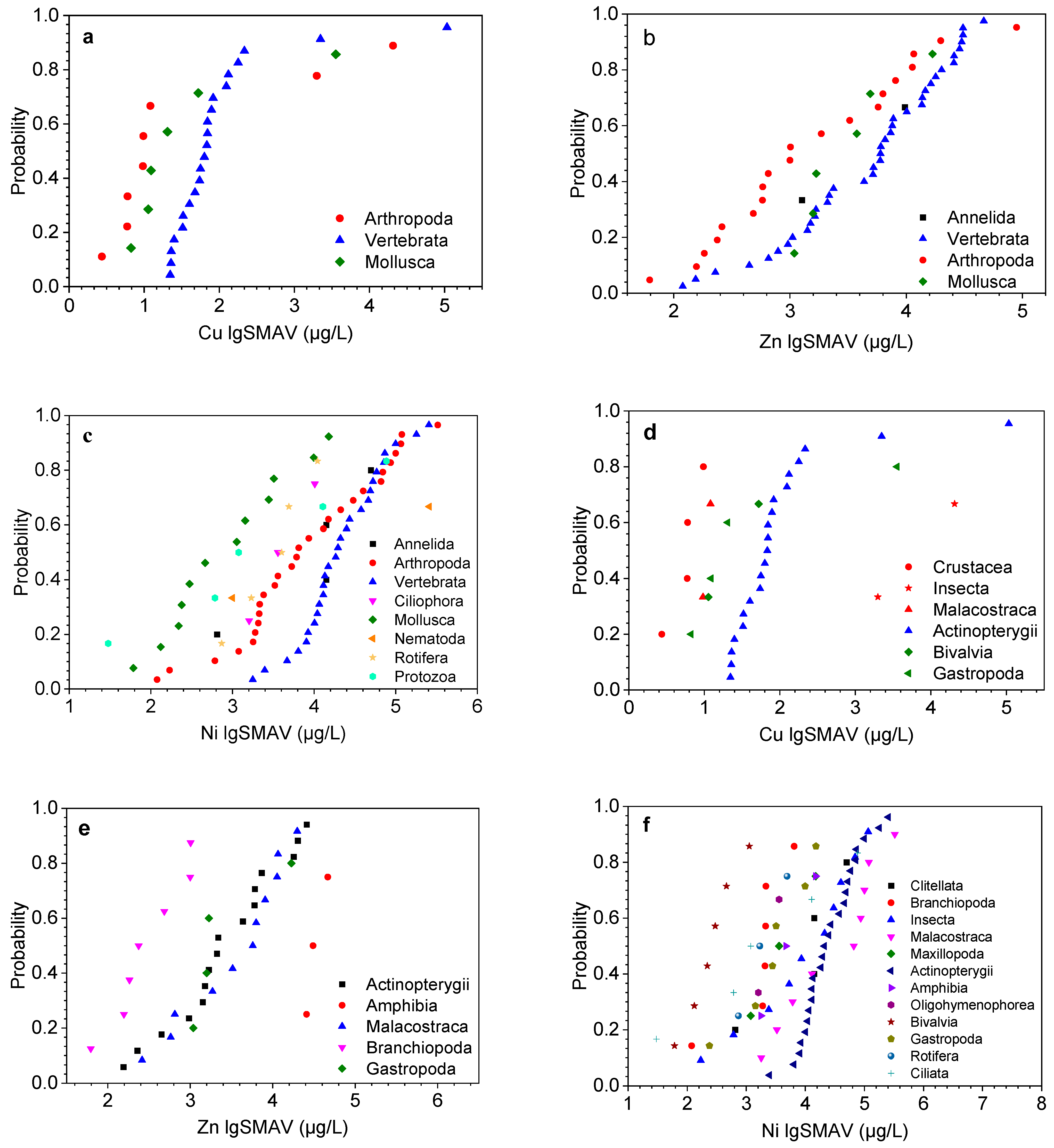
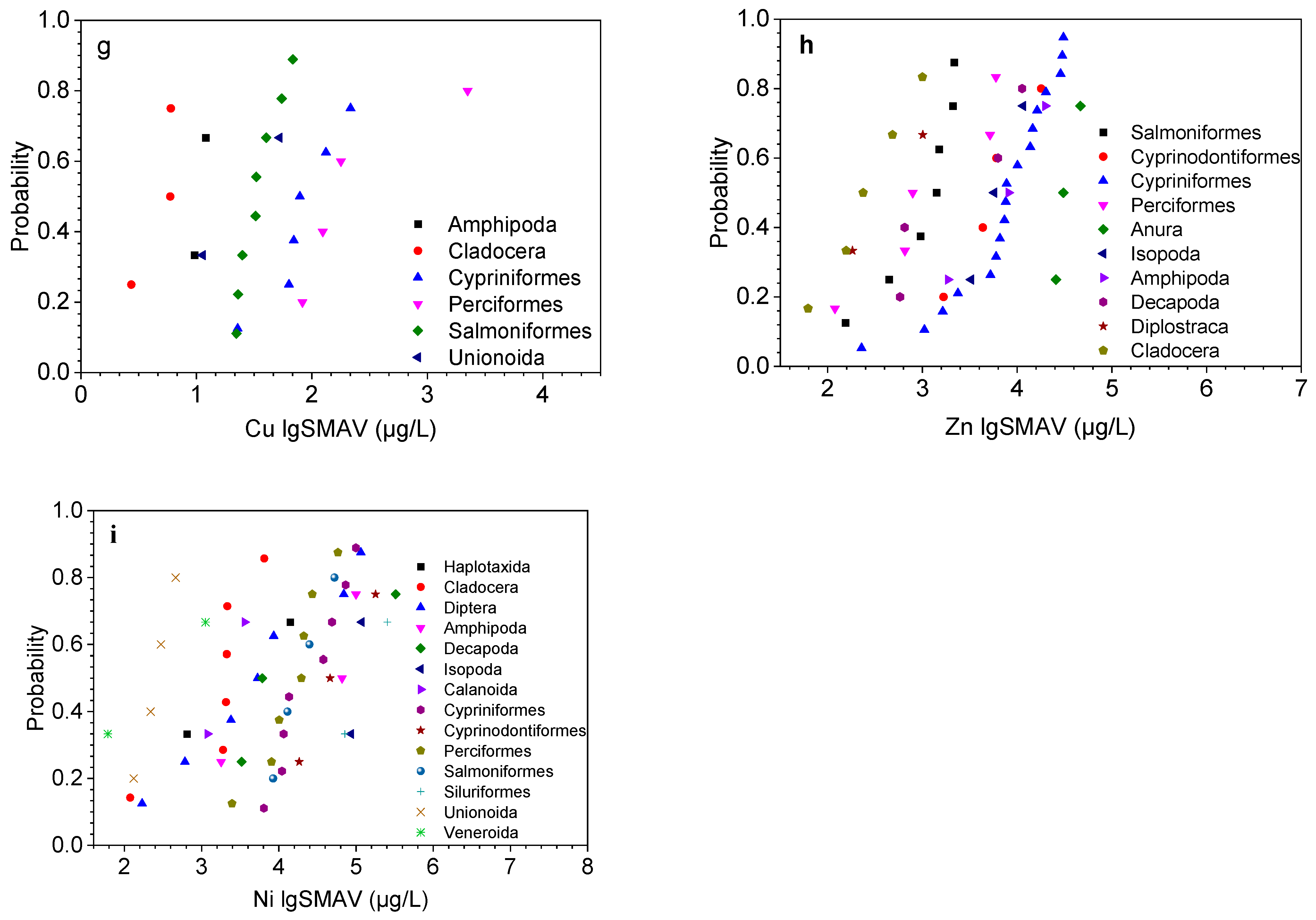
3.1. Species Sensitivity Analysis and Effect Assessment
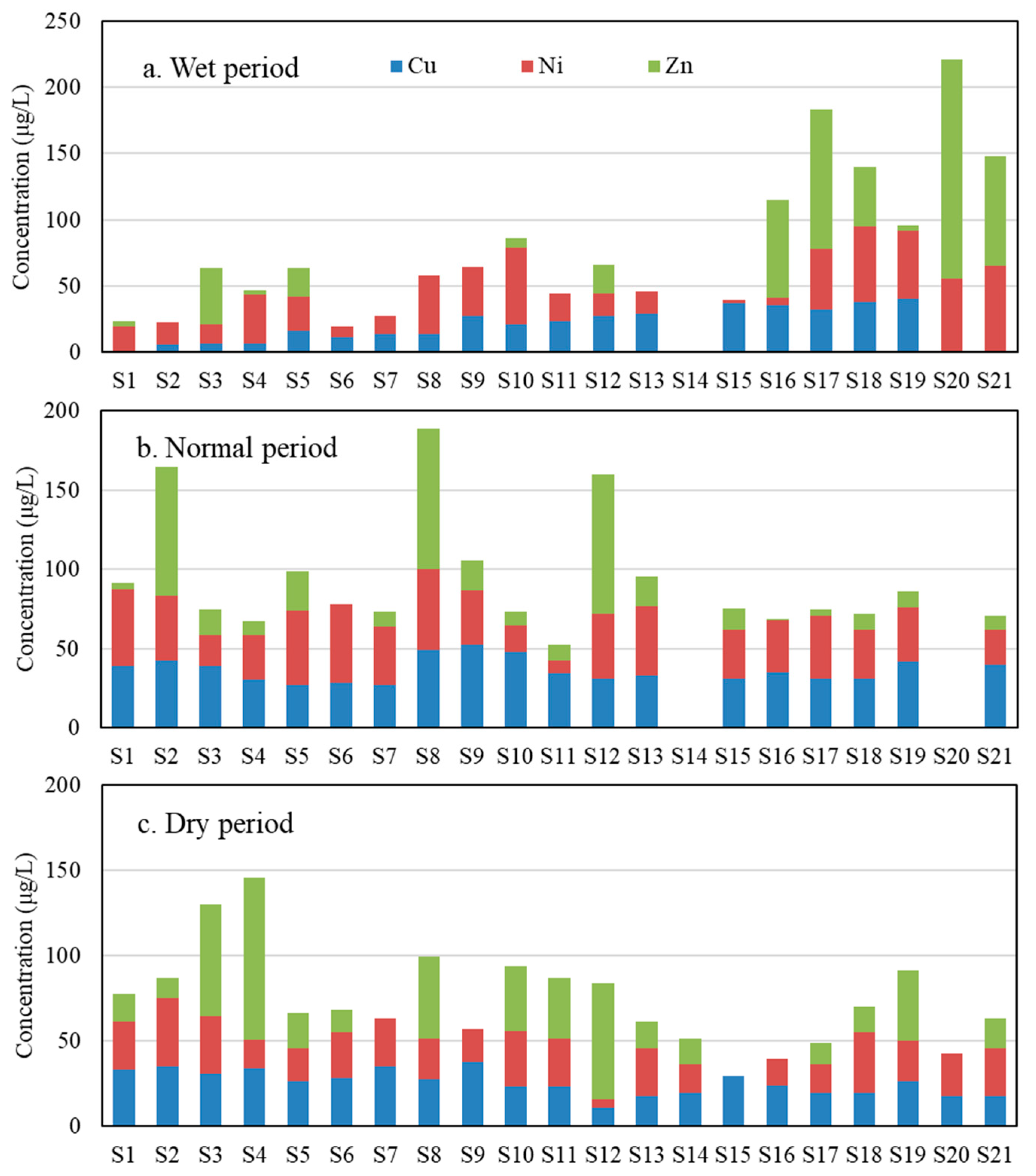
3.2. Exposure Assessment in Songhua River Water
3.3. Probabilistic Ecological Risk Assessment
3.4. Limitations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wu, Q.H.; Tam, N.F.Y.; Leung, J.Y.S.; Zhou, X.Z.; Fu, J.; Yao, B.; Huang, X.Z.; Xia, L.H. Ecological risk and pollution history of metals in Nansha mangrove, South China. Ecotoxicol. Environ. Saf. 2014, 104, 143–151. [Google Scholar] [CrossRef]
- Pilehvar, A.; Town, R.M.; Blust, R. The effect of copper on behaviour, memory, and associative learning ability of zebrafish (Danio rerio). Ecotoxicol. Environ. Saf. 2020, 188, 109900. [Google Scholar] [CrossRef]
- Martin, C.W. Persistence of trace metal contamination in a fluvial system: Lahn river, central Germany. Geomorphology 2023, 426, 108603. [Google Scholar] [CrossRef]
- Yang, J.; Chen, L.; Liu, L.Z.; Shi, W.L.; Meng, X.Z. Comprehensive risk assessment of metals in lake sediment from public parks in Shanghai. Ecotoxicol. Environ. Saf. 2014, 102, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Shou, Y.; Zhao, J.; Zhu, Y.; Qiao, J.; Shen, Z.; Zhang, W.; Han, N.; Núñez-Delgado, A. Heavy metals pollution characteristics and risk assessment in sediments and waters: The case of Tianjin, China. Environ. Res. 2022, 212, 113162. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, N.J.; Parr, T.B.; Gray, E.; Iburg, J.; Capps, K.A. Trophic Strategies Influence Metal Bioaccumulation in Detritus-Based, Aquatic Food Webs. Environ. Sci. Technol. 2018, 52, 11886–11894. [Google Scholar] [CrossRef]
- Xie, X.; Wang, F.; Wang, G.; Mei, R.; Wang, C. Study on Heavy Metal Pollution in Surface Water in China. Environ. Sci. Manag. 2017, 42, 31–34. [Google Scholar]
- Islam, M.S.; Ahmed, M.K.; Raknuzzaman, M.; Habibullah-Al-Mamun, M.; Islam, M.K. Heavy metal pollution in surface water and sediment: A preliminary assessment of an urban river in a developing country. Ecol. Indic. 2015, 48, 282–291. [Google Scholar] [CrossRef]
- Islam, M.S.; Han, S.; Masunaga, S. Assessment of trace metal contamination in water and sediment of some rivers in Bangladesh. J. Water Environ. Technol. 2014, 12, 109–121. [Google Scholar] [CrossRef]
- Liao, X.; Zhang, C.; Sun, G.; Li, Z.; Shang, L.; Fu, Y.; He, Y.; Yang, Y. Assessment of Metalloid and Metal Contamination in Soils from Hainan, China. Int. J. Environ. Res. Public Health 2018, 15, 454. [Google Scholar] [CrossRef]
- Morton-Bermea, O.; Hernández-Alvarez, E.; González-Hernández, G.; Romero, F.; Lozano, R.; BeramendiOrosco, L.E. Assessment of heavy metal pollution in urban top soils from the metropolitan area of Mexico City. J. Geochem. Explor. 2009, 101, 218–224. [Google Scholar] [CrossRef]
- Macklin, M.G.; Thomas, C.J.; Mudbhatkal, A.; Brewer, P.A.; Hudson-Edwards, K.A.; Lewin, J.; Scussolini, P.; Eilander, D.; Lechner, A.; Owen, J.; et al. Impacts of metal mining on river systems: A global assessment. Science 2023, 381, 1345–1350. [Google Scholar] [CrossRef]
- Baldantoni, D.; Alfani, A.; Tommasi, P.D.; Bartoli, G.; De Santo, A.V. Assessment of macro and microelement accumulation capability of two aquatic plants. Environ. Pollut. 2004, 130, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Rozentsvet, O.A.; Nesterov, V.N.; Sinyutina, N.F. The effect of copper ions on the lipid composition of subcellular membranes in Hydrilla verticillata. Chemosphere 2012, 89, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Hall, L.W.; Scott, M.C.; Killen, W.D. Ecological risk assessment of copper and cadmium in surface waters of Chesapeake Bay watershed. Environ. Toxicol. Chem. 1998, 17, 1172–1189. [Google Scholar] [CrossRef]
- Zhao, J.; Wu, E.; Zhang, B.; Bai, X.; Lei, P.; Qiao, X.; Li, Y.; Li, B.; Wu, G.; Gao, Y. Pollution characteristics and ecological risks associated with heavy metals in the Fuyang river system in north China—ScienceDirect. Environ. Pollut. 2021, 281, 116994. [Google Scholar] [CrossRef]
- Al-Halani, A.A.; Soliman, A.; Monier, M.N. The seasonal assessment of heavy metals pollution in water, sediments, and fish of grey mullet, red seabream, and sardine from the mediterranean coast, damietta, north egypt. Reg. Stud. Mar. Sci. 2022, 57, 102744. [Google Scholar]
- Wu, F.; Feng, C.; Cao, Y.; Zhang, R.; Li, H.; Zhao, X. Aquatic Life Ambient Freshwater Quality Criteria for Copper in China. Asian J. Ecotoxicol. 2011, 6, 617–628. [Google Scholar]
- Zhang, H. A Study on the Long-Term Effects of Heavy Metal Stress on the Growth of Chlorella in Aquatic Environments. Master’s Thesis, Chongqing Three Gorges University, Chongqing, China, 2020. [Google Scholar]
- Liu, N.; Jin, X.; Zhou, J.; Wang, Y.; Yang, Q.; Wu, F.; Giesy, G.P.; Johnson, A.C. Predicted no-effect concentration (PNEC) and assessment of risk for the fungicide, triadimefon based on reproductive fitness of aquatic organisms. Chemosphere 2018, 207, 682–689. [Google Scholar] [CrossRef]
- Jin, X.; Wang, Z.; Wang, Y.; Lv, Y.; Jin, W.; Rao, K.; Giesy, J.; Leung, K.M.Y. Do water quality criteria based on nonnative species provide appropriate protection for native species. Environ. Toxicol. Chem. 2015, 34, 1793–1798. [Google Scholar] [CrossRef]
- Slooff, W. Benthic macroinvertebrates and water quality assessment, some toxicological considerations. Aquat. Toxicol. 1983, 4, 73–82. [Google Scholar] [CrossRef]
- Forbes, V.E.; Calow, P. Species sensitivity distributions revisited: A critical appraisal. Hum. Ecol. Risk Assess. 2002, 3, 473–492. [Google Scholar] [CrossRef]
- Vaal, M.; van der Wall, J.T.; Hermens, J.; Hoekstra, J. Pattern analysis of the variation in the sensitivity of aquatic species to toxicants. Chemosphere 1997, 35, 1291–1309. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Qin, H.W.; Su, L.M. Interspecies correlations of toxicity to eight aquatic organisms: Theoretical considerations. Sci. Total. Environ. 2010, 408, 4549–4555. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jin, X.W.; Wang, Z.J. Taxon-specific sensitivity differences of copper to aquatic organisms. Asian J. Ecotoxicol. 2014, 9, 640–646. [Google Scholar]
- Wang, Y.Y.; Zhang, L.S.; Meng, F.S.; Zhou, Y.X.; Jin, X.W.; Giesy, J.P.; Liu, F. Improvement on species sensitivity distribution methods for deriving site-specific water quality criteria. Environ. Sci. Pollut. Res. 2015, 22, 5271–5282. [Google Scholar] [CrossRef]
- Jin, X.W.; Zha, J.M.; Xu, Y.P. Derivation of aquatic predicted no-effect concentration (PNEC) for 2,4-dichlorophenol: Comparing native species data with non-native species data. Chemosphere 2011, 84, 1506–1511. [Google Scholar] [CrossRef]
- Hou, D.; He, J.; Lv, C.; Ren, L.; Fan, Q.; Wang, J.; Xie, Z. Distribution characteristics and potential ecological risk assessment of metals (Cu, Pb, Zn, Cd) in water and sediments from Lake Dalinouer, China. Ecotoxicol. Environ. Saf. 2013, 93, 135–144. [Google Scholar] [CrossRef]
- Ke, X.; Gui, S.; Huang, H.; Zhang, H.; Wang, C.; Guo, W. Ecological risk assessment and source identification for heavy metals in surface sediment from the Liaohe River protected area, China. Chemosphere 2017, 175, 473–481. [Google Scholar] [CrossRef]
- Johnson, A.C.; Jürgens, M.D.; Su, C.; Zhang, M.; Zhang, Y.; Shi, Y.; Lu, Y. Which commonly monitored chemical contaminant in the Bohai region and the Yangtze and pearl rivers of China poses the greatest threat to aquatic wildlife? Environ. Toxicol. Chem. 2018, 37, 1115–1121. [Google Scholar] [CrossRef]
- Liu, B.; Mei, Y.; Gao, X.; Yin, Q.; Li, M.; Hu, Z.; Wang, X.; Dou, W.; Shao, Z.; Li, Z. Pollution characteristics and health risk assessment of heavy metals in fishes collected from the Songhua River. J. Northeast. Norm. Univ. Nat. Sci. Ed. 2018, 50, 142–147. [Google Scholar]
- GB7475-87; Water Quality-Determination of Copper, Zinc, Lead and Cadmium-Atomic Absorption Spectrometry. State Environmental Protection Administration of China: Beijing, China, 1987.
- GB11912-89; Water Quality-Determination of Nickel-Flame Atomic Absorption Spectrometric Method. State Bureau of Technical Supervision: Beijing, China, 1989.
- Wheeler, J.R.; Grist, E.P.M.; Leung, K.M.Y.; Morritt, D.; Crane, M. Species sensitivity distributions: Data and model choice. Mar. Pollut. Bull. 2002, 45, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.C.; Yang, X.L.; Wang, M.X. Prediction Model for Setting Long-Term Water Quality Criteria of Copper in Chinese River Basins: MLR vs. BLM. Res. Environ. Sci. 2023, 36, 1236–1244. [Google Scholar]
- Liu, N.; Li, Y.B.; Liu, H.L. The water quality criteria and ecological risks of copper under the influence of multiple factors. China Environ. Sci. 2022, 42, 3353–3361. [Google Scholar]
- Solomon, K.R.; Sibley, P. New concepts in ecological risk assessment: Where do we go fromhere? Mar. Pollut. Bull. 2002, 44, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Jin, X.; Yan, Z.; Luo, Y.; Feng, C.; Fu, Z.; Tang, Z.; Wu, F.; Giesy, J.P. Occurrence and multiple-level ecological risk assessment of pharmaceuticals and personal care products (PPCPs) in two shallow lakes of China. Environ. Sci. Eur. 2020, 32, 69. [Google Scholar] [CrossRef]
- Hou, L.; Jin, X.; Liu, N.; Luo, Y.; Yan, Z.; Chen, M.; Xu, J. Triadimefon in aquatic environments: Occurrence, fate, toxicity, and ecological risk. Environ. Sci. Eur. 2022, 34, 12. [Google Scholar] [CrossRef]
- Jin, X.; Wang, Y.; Jin, W.; Rao, K.; Giesy, J.P.; Hollert, H.; Richardson, K.L.; Wang, Z. Ecological risk of nonylphenol in China surface waters based on reproductive fitness. Environ. Sci. Technol. 2014, 48, 1256–1262. [Google Scholar] [CrossRef]
- Chen, M.; Hong, Y.; Jin, X.; Guo, C.; Zhao, X.; Liu, N.; Lu, H.; Liu, Y.; Xu, J. Ranking the risks of eighty pharmaceuticals in surface water of a megacity: A multilevel optimization strategy. Sci. Total Environ. 2023, 878, 163184. [Google Scholar] [CrossRef]
- Jin, X.; Gao, J.; Zha, J.; Xu, Y.; Wang, Z.; Giesy, J.P.; Richardson, K.L. A tiered ecological risk assessment of three chlorophenols in Chinese surface waters. Environ. Sci. Pollut. Res. 2012, 19, 1544–1554. [Google Scholar] [CrossRef]
- Moore, D.R.J.; Teed, R.S.; Greer, C.D.; Solomon, K.R.; Giesy, J.P. Refined avian risk assessment for chlorpyrifos in the United States. Rev. Environ. Contam. Toxicol. 2014, 231, 163–217. [Google Scholar] [PubMed]
- Barlas, N.; Akbulut, N.; Aydoğan, M. Assessment of metal residues in the sediment and water samples of Uluabat Lake, Turkey. Bull. Environ. Contam. Toxicol. 2005, 74, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Strong, J.A.; Andonegi, E.; Bizsel, K.C.; Danovaro, R.; Elliott, M.; Franco, A.; Garces, E.; Little, S.; Mazik, K.; Moncheva, S.; et al. Marine biodiversity and ecosystem function relationships: The potential for practical monitoring applications. Estuar. Coast. Shelf. 2015, 161, 46–64. [Google Scholar] [CrossRef]
- Sun, C.; Zhang, Z.; Cao, H.; Xu, M.; Xu, L. Concentrations, speciation, and ecological risk of heavy metals in the sediment of the songhua river in an urban area with petrochemical industries. Chemosphere 2019, 219, 538–545. [Google Scholar] [CrossRef]
- GB3838-2002; Environmental Quality Standards for Surface Water. State Environmental Protection Administration of China: Beijing, China, 2002.
- USEPA. National Recommended Water Quality Criteria; U.S. Environmental Protection Agency, Office of Research and Development: Washington, DC, USA, 2015. [Google Scholar]
- Li, J.; Zheng, C. Environmental Background Data Handbook; China Environmental Science Press: Beijing, China, 1989. [Google Scholar]
- Li, K.Y.; Cui, S.; Zhang, F.X. Concentrations, Possible Sources and Health Risk of Heavy Metals in Multi-media Environment of the Songhua River, China. Int. J. Environ. Res. Public Health 2020, 17, 1766. [Google Scholar] [CrossRef]
- Lu, J.L.; Hao, L.B.; Zhao, Y.Y.; Bai, R.J.; Sun, S.M.; Fang, C.C. Contents and Potential Ecological Risk of Metals in Middle and Lower Reaches of Second Songhua River. Environ. Sci. Technol. 2009, 32, 168–172. [Google Scholar]
- Liu, N.; Jin, X.W.; Feng, C.L. Ecological risk assessment of fifty Pharmaceuticals and Personal Care Products (PPCPs) in Chinese surface waters: A proposed multiple-level system. Environ. Int. 2020, 136, 105454. [Google Scholar] [CrossRef] [PubMed]
- Lao, C.; Luo, L.; Shen, Y.; Zhu, S. Progress in the Study of Interaction Process and Mechanism between Microorganism and Heavy Metal. Res. Environ. Sci. 2020, 33, 1929–1937. [Google Scholar]
- Strotmann, U.; Flores, D.P.; Konrad, O.; Gendig, C. Bacterial Toxicity Testing: Modification and Evaluation of the Luminescent Bacteria Test and the Respiration Inhibition Test. Processes 2020, 8, 1349. [Google Scholar] [CrossRef]
- Xie, B. The Effect of Cu and Zinc ions on Activated Sludge Microbes and Its Analysis with Molecular Biological Technique. Doctoral Dissertation, Donghua University, Shanghai, China, 2002. [Google Scholar]
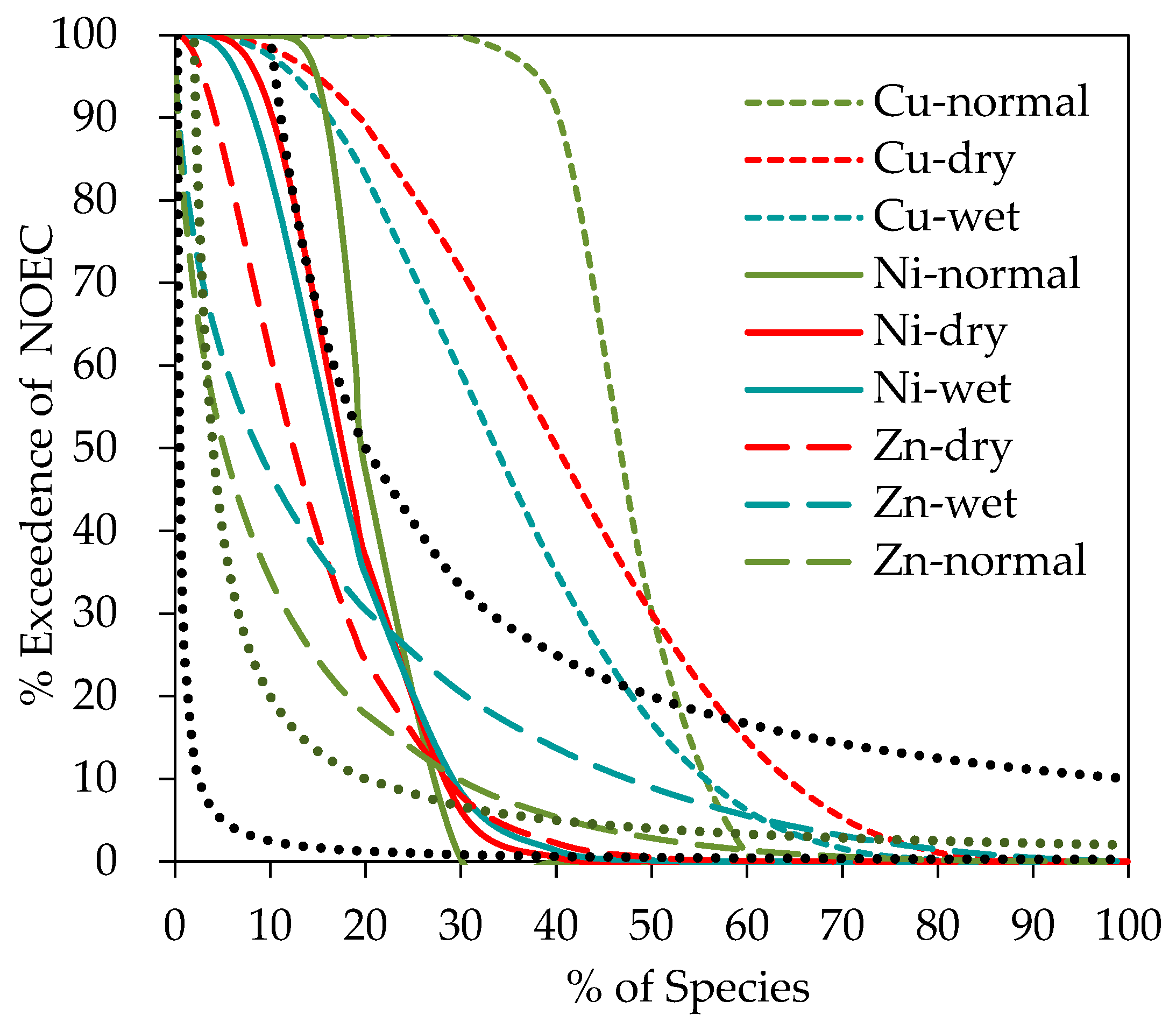
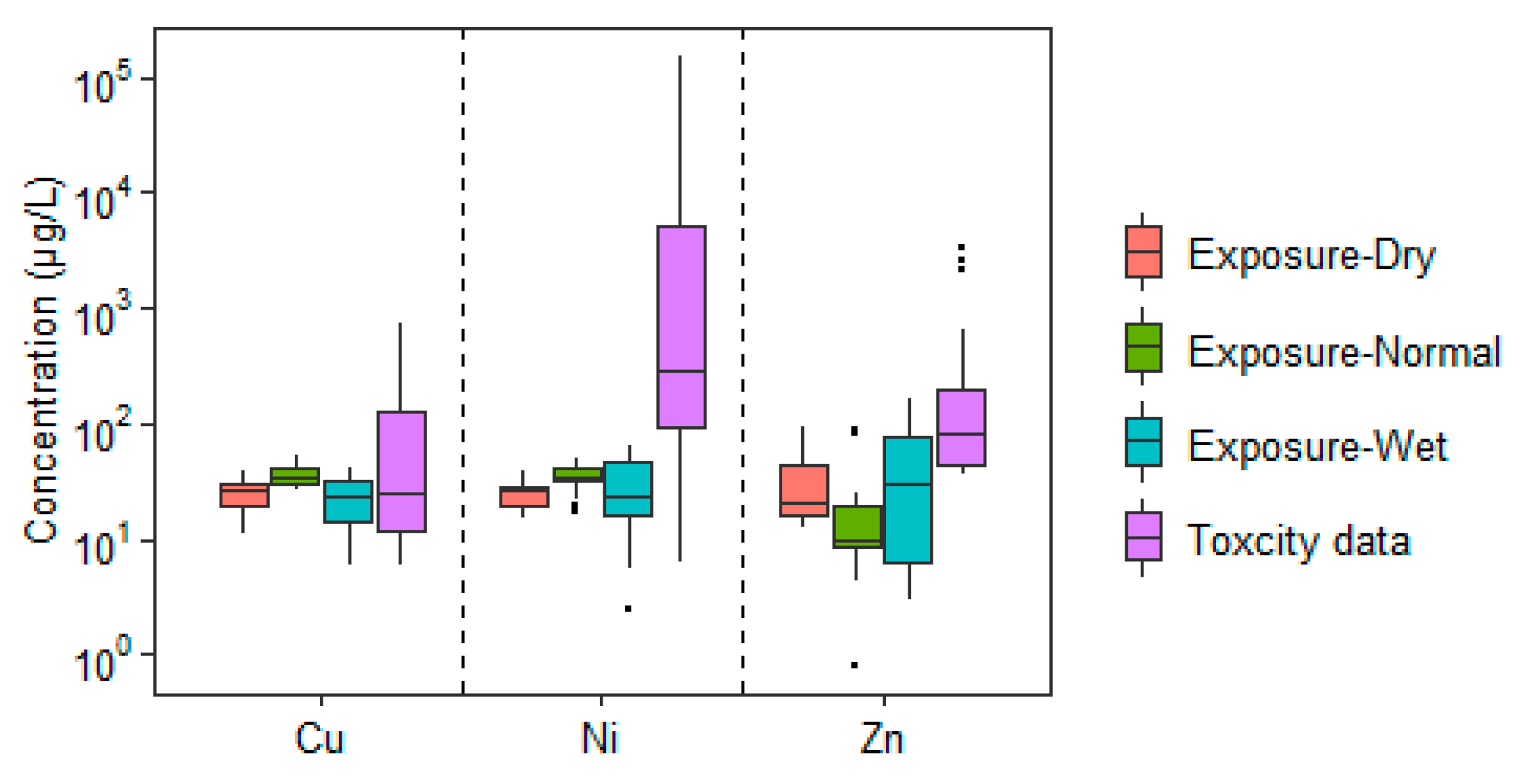
| Location | Metal | Number of Points | Minimum | Median | Maximum | References |
|---|---|---|---|---|---|---|
| Bohai Region Rivers | Copper | 816 | 0.00071 | 4.7 | 2755 | [31] |
| Nickel | 118 | 0.8 | 9.3 | 571 | [31] | |
| Zinc | 838 | 0.035 | 35 | 25,370 | [31] | |
| Pearl River | Copper | 187 | n.d | 8 | 2169 | [31] |
| Nickel | 125 | n.d | 12 | 128 | [31] | |
| Zinc | 156 | n.d | 27 | 37,500 | [31] | |
| Yangtze River | Copper | 1314 | n.d | 2.44 | 343 | [31] |
| Nickel | 44 | 0.4 | 2.7 | 18 | [31] | |
| Zinc | 1307 | n.d | 10.8 | 500 | [31] | |
| UK | Copper | 89,604 | 0.1 | 1.66 | 5320 | [31] |
| Nickel | 14,981 | 0.25 | 1.69 | 270 | [31] | |
| Zinc | 11,679 | 0.702 | 6.5 | 6900 | [31] | |
| Songhua River | Copper | 57 | 5.8 | 29.1 | 52.7 | Present study |
| Nickel | 59 | 2.5 | 28.2 | 65.3 | Present study | |
| Zinc | 46 | 0.8 | 16.85 | 166 | Present study |
| Metal | N | Mean (μg/L) | SD | Coefficients of Variation | Shapiro–Wilk Test for Log-Normal Distribution |
|---|---|---|---|---|---|
| Exposure data set | |||||
| Cu—normal | 19 | 36.4 | 7.8 | 0.21 | 0.207 |
| Cu—wet | 17 | 22.9 | 11.9 | 0.52 | 0.060 |
| Cu—dry | 21 | 25.7 | 7.3 | 0.28 | 0.179 |
| Ni—normal | 17 | 35.1 | 9.7 | 0.28 | 0.011 |
| Ni—wet | 20 | 30.5 | 19.8 | 0.65 | 0.068 |
| Ni—dry | 19 | 24.6 | 6.9 | 0.28 | 0.225 |
| Zn—normal | 18 | 23.6 | 29.2 | 1.24 | 0.098 |
| Zn—wet | 12 | 48.1 | 50.5 | 1.05 | 0.270 |
| Zn—dry | 15 | 34.5 | 25.1 | 0.73 | 0.069 |
| Toxicity data set | |||||
| Cu | 23 | 124 | 201 | 1.62 | 0.071 |
| Ni | 21 | 10,630 | 32,092 | 3.02 | 0.621 |
| Zn | 21 | 511 | 906 | 1.77 | 0.011 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Meng, F.; Liu, N.; Zhang, J.; Xue, H. The Taxon-Specific Species Sensitivity and Aquatic Ecological Risk Assessment of Three Heavy Metals in Songhua River Water, China. Water 2023, 15, 3694. https://doi.org/10.3390/w15203694
Zhang L, Meng F, Liu N, Zhang J, Xue H. The Taxon-Specific Species Sensitivity and Aquatic Ecological Risk Assessment of Three Heavy Metals in Songhua River Water, China. Water. 2023; 15(20):3694. https://doi.org/10.3390/w15203694
Chicago/Turabian StyleZhang, Lingsong, Fansheng Meng, Na Liu, Jiasheng Zhang, and Hao Xue. 2023. "The Taxon-Specific Species Sensitivity and Aquatic Ecological Risk Assessment of Three Heavy Metals in Songhua River Water, China" Water 15, no. 20: 3694. https://doi.org/10.3390/w15203694
APA StyleZhang, L., Meng, F., Liu, N., Zhang, J., & Xue, H. (2023). The Taxon-Specific Species Sensitivity and Aquatic Ecological Risk Assessment of Three Heavy Metals in Songhua River Water, China. Water, 15(20), 3694. https://doi.org/10.3390/w15203694






