Tall Herb Fringe Vegetation on Banks of Montenegrin Rivers as a Habitat Type of European Importance
Abstract
:1. Introduction
2. Materials and Methods
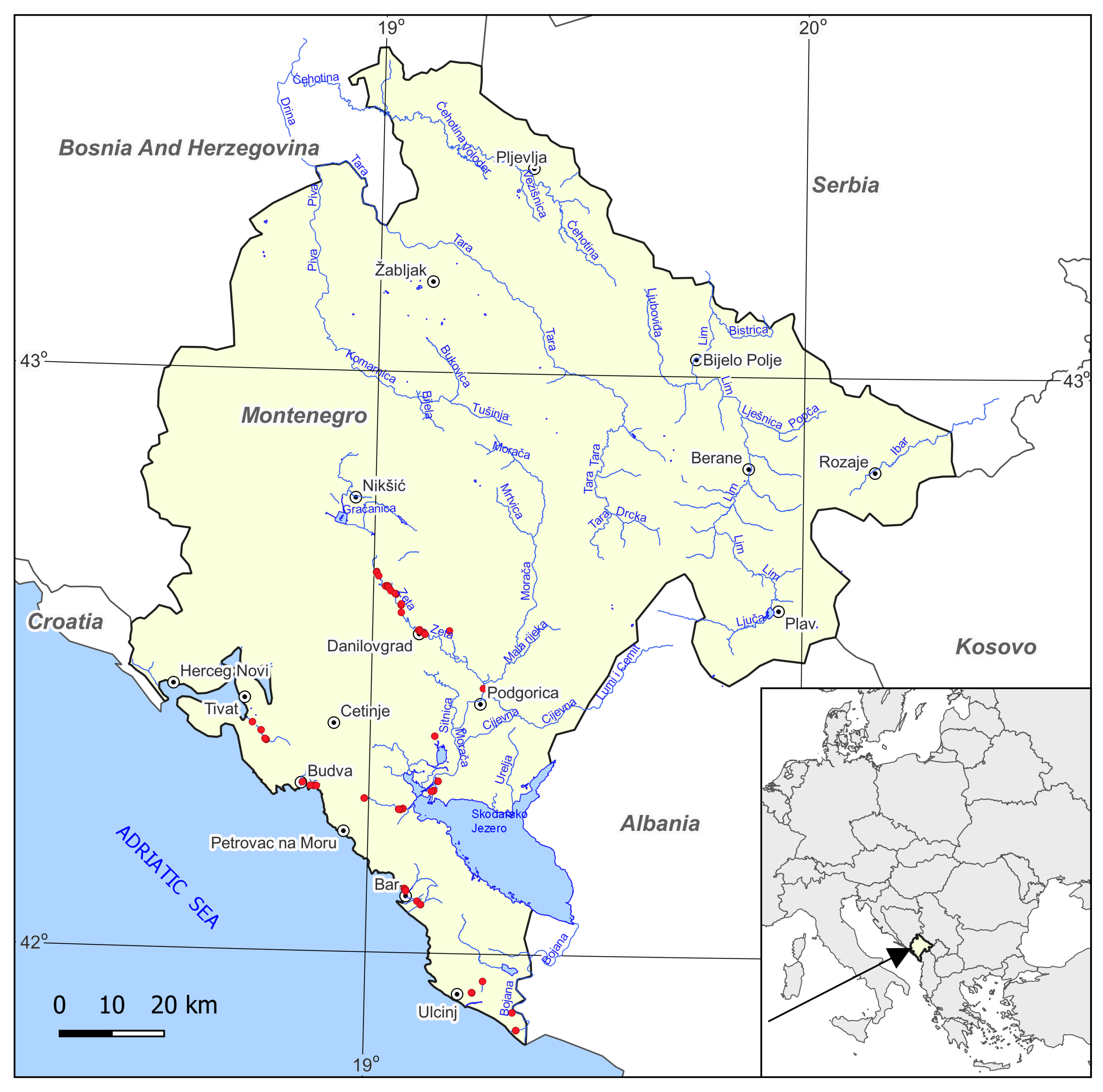
2.1. Study Area
2.2. Data Collection and Analysis
| Column Number | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Cluster | A | B | C1 | C2 | D | E |
| Number of Relevés | 8 | 22 | 11 | 6 | 20 | 3 |
| Diagnostic species | ||||||
| Helianthus ×laetiflorus Pers. | 100 | 14 | 0 | 0 | 0 | 0 |
| Pastinaca sativa L. | 50 | 5 | 0 | 0 | 10 | 0 |
| Rubus sanctus Schreb. | 75 | 73 | 27 | 0 | 15 | 67 |
| Clematis vitalba L. | 38 | 55 | 0 | 0 | 0 | 33 |
| Xanthium orientale subsp. italicum (Moretti) Greuter | 38 | 14 | 0 | 0 | 0 | 0 |
| Cirsium creticum (Lam.) d’Urv. | 13 | 18 | 0 | 0 | 5 | 33 |
| Eupatorium cannabinum L. | 38 | 100 | 36 | 100 | 25 | 0 |
| Rubus caesius L. | 13 | 14 | 100 | 83 | 20 | 0 |
| Bidens frondosa L. | 0 | 9 | 73 | 100 | 30 | 100 |
| Rumex conglomeratus Murray | 0 | 18 | 64 | 50 | 30 | 0 |
| Agrostis capillaris L. | 0 | 9 | 64 | 83 | 30 | 0 |
| Galium palustre L. | 0 | 0 | 45 | 67 | 10 | 0 |
| Aristolochia clematitis L. | 0 | 5 | 36 | 50 | 5 | 0 |
| Pulicaria dysenterica (L.) Bernh. | 0 | 18 | 0 | 67 | 100 | 0 |
| Mentha longifolia (L.) L. | 13 | 18 | 0 | 0 | 40 | 0 |
| Helianthus tuberosus L. | 0 | 0 | 0 | 0 | 0 | 100 |
| Epilobietea angustifolii Tx. et Preising ex von Rochow 1951 | ||||||
| Calystegia sepium (L.) R. Br. | 25 | 32 | 18 | 17 | 55 | 100 |
| Urtica dioica L. | 13 | 9 | 0 | 17 | 15 | 67 |
| Humulus lupulus L. | 13 | 9 | 18 | 17 | 10 | 0 |
| Parietaria officinalis L. | 25 | 5 | 18 | 17 | 5 | 0 |
| Sambucus ebulus L. | 13 | 0 | 9 | 33 | 5 | 0 |
| Epilobium hirsutum L. | 25 | 0 | 0 | 0 | 5 | 0 |
| Torilis japonica (Houtt.) DC. | 0 | 9 | 0 | 0 | 5 | 33 |
| Alno glutinosae-Populetea albae P. Fukarek et Fabijanić 1968 | ||||||
| Hedera helix L. | 13 | 23 | 0 | 17 | 0 | 0 |
| Ulmus minor Mill. | 13 | 9 | 18 | 0 | 0 | 0 |
| Brachypodium sylvaticum (Huds.) P. Beauv. | 25 | 23 | 36 | 50 | 0 | 33 |
| Clematis viticella L. | 0 | 5 | 64 | 33 | 15 | 33 |
| Fraxinus angustifolia Vahl | 0 | 0 | 9 | 33 | 0 | 0 |
| Molinio-Arrhenatheretea Tx. 1937 | ||||||
| Galium mollugo L. | 38 | 50 | 55 | 33 | 20 | 33 |
| Agrostis stolonifera L. | 25 | 18 | 18 | 0 | 20 | 33 |
| Verbena officinalis L. | 13 | 14 | 0 | 0 | 10 | 33 |
| Dactylis glomerata L. | 25 | 14 | 9 | 0 | 0 | 0 |
| Paspalum distichum L. | 13 | 0 | 0 | 0 | 20 | 33 |
| Centaurea jacea L. | 0 | 14 | 0 | 17 | 25 | 0 |
| Ranunculus repens L. | 0 | 14 | 55 | 83 | 20 | 0 |
| Plantago major L. | 0 | 14 | 18 | 33 | 15 | 0 |
| Prunella vulgaris L. | 0 | 18 | 18 | 33 | 25 | 0 |
| Carex hirta L. | 0 | 9 | 27 | 0 | 45 | 0 |
| Potentilla reptans L. | 0 | 0 | 55 | 33 | 45 | 0 |
| Lysimachia nummularia L. | 0 | 0 | 18 | 33 | 30 | 0 |
| Poa trivialis L. | 0 | 9 | 18 | 0 | 15 | 33 |
| Rumex crispus L. | 0 | 5 | 9 | 0 | 10 | 33 |
| Paspalum dilatatum Poir. | 0 | 5 | 0 | 0 | 15 | 33 |
| Epilobium tetragonum L. | 0 | 5 | 9 | 0 | 0 | 33 |
| Trifolium pratense L. | 0 | 0 | 0 | 0 | 15 | 33 |
| Cyperus eragrostis Lam. | 0 | 0 | 0 | 0 | 5 | 33 |
| Phragmito-Magnocaricetea Klika in Klika et Novák 1941 | ||||||
| Lythrum salicaria L. | 13 | 41 | 36 | 83 | 85 | 0 |
| Mentha aquatica L. | 25 | 18 | 36 | 83 | 40 | 33 |
| Lycopus europaeus L. | 13 | 14 | 9 | 33 | 30 | 0 |
| Phragmites australis (Cav.) Steud. | 13 | 5 | 0 | 0 | 5 | 67 |
| Lysimachia vulgaris L. | 0 | 0 | 18 | 17 | 10 | 0 |
| Leersia oryzoides (L.) Sw. | 0 | 0 | 27 | 17 | 0 | 0 |
| Cyperus longus L. | 0 | 5 | 18 | 0 | 20 | 0 |
| Carex acuta L. | 0 | 5 | 9 | 0 | 0 | 33 |
| Gratiola officinalis L. | 0 | 0 | 0 | 0 | 25 | 0 |
| Artemisietea vulgaris Lohmeyer et al. in Tx. ex von Rochow 1951 | ||||||
| Daucus carota L. | 13 | 50 | 18 | 0 | 35 | 33 |
| Equisetum arvense L. | 25 | 18 | 0 | 17 | 10 | 67 |
| Cichorium intybus L. | 38 | 9 | 27 | 17 | 25 | 0 |
| Artemisia verlotiorum Lamotte | 25 | 9 | 9 | 17 | 0 | 0 |
| Melissa officinalis L. | 25 | 32 | 0 | 17 | 0 | 33 |
| Piptatherum miliaceum (L.) Coss. | 38 | 18 | 0 | 0 | 10 | 0 |
| Dittrichia viscosa (L.) Greuter | 25 | 14 | 0 | 0 | 0 | 0 |
| Picris hieracioides L. | 0 | 32 | 9 | 0 | 10 | 33 |
| Artemisia vulgaris L. | 0 | 9 | 0 | 0 | 10 | 33 |
| Elytrigia repens (L.) Nevski | 0 | 5 | 9 | 0 | 10 | 67 |
| Other species | ||||||
| Setaria viridis (L.) P. Beauv. | 25 | 0 | 0 | 0 | 0 | 0 |
| Chenopodium album L. | 13 | 0 | 0 | 0 | 0 | 33 |
| Persicaria maculosa Gray | 25 | 18 | 0 | 0 | 5 | 33 |
| Bidens tripartita L. | 13 | 5 | 9 | 17 | 0 | 0 |
| Cynodon dactylon (L.) Pers. | 13 | 14 | 9 | 0 | 15 | 0 |
| Setaria pumila (Poir.) Roem. & Schult. | 25 | 9 | 0 | 17 | 10 | 0 |
| Cornus sanguinea L. | 0 | 32 | 45 | 17 | 20 | 0 |
| Periploca graeca L. | 0 | 9 | 27 | 0 | 20 | 0 |
| Amorpha fruticosa L. | 0 | 9 | 27 | 0 | 0 | 0 |
| Euphorbia sp. | 0 | 9 | 9 | 0 | 30 | 0 |
| Xanthium strumarium L. | 0 | 9 | 9 | 0 | 15 | 33 |
| Euonymus europaeus L. | 0 | 0 | 18 | 50 | 5 | 0 |
| Persicaria dubia (Stein) Fourr. | 0 | 0 | 9 | 33 | 0 | 0 |
| Column Number | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Relevé Number * | 2 | 64 | 66 | 6 | 39 | 8 | 52 | 18 | 20 | 5 |
| Year | 2010 | 2012 | 2012 | 2010 | 2012 | 2010 | 2012 | 2010 | 2010 | 2010 |
| Month | 10 | 8 | 8 | 10 | 8 | 10 | 8 | 10 | 10 | 10 |
| Day | 8 | 30 | 30 | 8 | 22 | 9 | 27 | 13 | 13 | 8 |
| Relevé area (m2) | 4 | 8 | 6 | 3 | 5 | 4 | 4 | 9 | 4 | 5 |
| Slope (degree) | 40 | 0 | 0 | 0 | 60 | 20 | 0 | 0 | 0 | 50 |
| Aspect | SW | - | - | - | S | NW | - | - | - | W |
| Shrub layer coverage (%) | 20 | 0 | 0 | 1 | 0 | 0 | 1 | 10 | 5 | 5 |
| Herb layer coverage (%) | 100 | 50 | 100 | 85 | 85 | 90 | 80 | 100 | 95 | 100 |
| Moss layer coverage (%) | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 5 | 0 | 0 |
| No. of species | 13 | 18 | 14 | 12 | 10 | 20 | 15 | 17 | 15 | 19 |
| Characteristic and differential* species of association Rubo sancti-Eupatorietum cannabini Myśliwy ass. nov. hoc loco | ||||||||||
| Eupatorium cannabinum L. | 3 | 3 | 4 | 4 | 2b | 3 | 3 | 4 | 2a | 3 |
| Rubus sanctus Schreb. | 2a | . | 2a | + | 2a | 2a | + | 1 | 1 | + |
| * Clematis vitalba L. | 2a | + | 2a | . | . | + | . | + | 2a | + |
| * Cirsium creticum (Lam.) d’Urv. | 1 | . | 1 | . | . | + | . | . | . | + |
| * Xanthium orientale subsp. italicum (Moretti) Greuter | . | . | . | . | . | + | 1 | . | . | + |
| Characteristic species of alliance Dorycnio recti-Rumicion conglomerati Gradstein et Smittenberg 1977 | ||||||||||
| Dittrichia viscosa (L.) Greuter | . | . | . | 2a | . | + | . | . | . | . |
| Pulicaria dysenterica (L.) Bernh. | + | . | . | . | . | 2b | . | . | . | . |
| Scirpoides holoschoenus (L.) Soják | . | + | . | . | . | + | . | . | . | . |
| Poa trivialis L. | . | . | + | . | . | . | . | 2m | . | . |
| Rumex conglomeratus Murray | . | + | . | . | . | . | . | . | . | . |
| Euphorbia hirsuta L. | . | . | . | + | . | . | . | . | . | . |
| Convolvuletalia sepium Tx. ex Moor 1958 et Epilobietea angustifolii Tx. et Preising ex von Rochow 1951 | ||||||||||
| Calystegia sepium (L.) R. Br. | 2a | . | 1 | + | . | 2a | . | . | . | . |
| Lythrum salicaria L. | 2a | 2a | . | . | . | . | . | + | . | 3 |
| Mentha aquatica L. | 2b | 2a | + | . | . | . | . | . | . | . |
| Helianthus ×laetiflorus Pers. | + | . | . | . | . | + | . | . | . | + |
| Humulus lupulus L. | . | . | 2b | . | . | . | . | . | + | . |
| Rubus caesius L. | . | . | . | . | . | . | . | 2a | 2a | . |
| Artemisia verlotiorum Lamotte | . | . | . | . | . | . | + | . | . | 1 |
| Molinio-Arrhenatheretea Tx. 1937 | ||||||||||
| Galium mollugo L. | 1 | . | . | . | + | . | . | + | + | . |
| Agrostis capillaris L. | . | . | . | . | . | . | . | . | + | 2m |
| Agrostis stolonifera L. | . | . | + | . | . | 1 | . | . | . | . |
| Dactylis glomerata L. | . | . | . | . | + | . | . | . | . | + |
| Other species | ||||||||||
| Daucus carota L. | . | + | + | + | . | + | + | . | . | . |
| Brachypodium sylvaticum (Huds.) P. Beauv. | . | . | . | . | 2a | + | . | . | 2a | 1 |
| Cornus sanguinea L. (b) | . | . | . | + | . | . | . | 2a | 2a | 2a |
| Melissa officinalis L. | . | . | . | . | . | + | 2a | 1 | . | + |
| Equisetum ramosissimum Desf. | 1 | . | . | + | . | . | . | . | . | . |
| Robinia pseudoacacia L. (b) | 2b | . | . | . | . | . | + | . | . | . |
| Cynodon dactylon (L.) Pers. | . | 2m | 1 | . | . | . | . | . | . | . |
| Persicaria maculosa Gray | . | + | . | . | . | . | 1 | . | . | . |
| Amorpha fruticosa L. (c) | . | r | . | . | . | . | . | r | . | . |
| Picris hieracioides L. | . | . | + | . | 1 | . | . | . | . | . |
| Piptatherum miliaceum (L.) Coss. | . | . | . | . | . | 2m | + | . | . | . |
| Equisetum arvense L. | . | . | . | . | . | . | 1 | 2a | . | . |
| Vicia sp. | . | . | . | + | . | + | . | . | . | . |
| Hedera helix L. | . | . | . | . | . | . | . | + | + | . |
| Sporadic species: Acer campestre L. (b) 18 (2a), Aristolochia clematitis L. 20 (+), Berteroa mutabilis (Vent.) DC. 8 (+), Bidens frondosa L. 20 (+), Bidens subalternans DC. 8 (+), Bidens tripartita L. 52 (2a), Carduus crispus L. 39 (+), Carex hirta L. 18 (2a), Carex rostrata Stokes 20 (1), Centaurea jacea L. 64 (+), Cichorium intybus L. 52 (+), Clematis flammula L. 39 (3), Clematis viticella L. 6 (+), Convolvulus arvensis L. 6 (+), Cruciata laevipes Opiz 66 (+), Cyperus longus L. 64 (+), Digitaria sanguinalis (L.) Scop. 52 (2m), Dioscorea communis (L.) Caddick & Wilkin 18 (+), Dorycnium pentaphyllum subsp. herbaceum (Vill.) Bonnier & Layens 39 (+), Elytrigia repens (L.) Nevski 5 (1), Equisetum telmateia Ehrh. 66 (2a), Ficus carica L. (c) 64 (1), Galium elongatum C. Presl 18 (+), Galium verum L. 5 (1), Lactuca serriola L. 39 (+), Ligustrum vulgare L. (c) 64 (r), Melilotus albus Medik. 8 (2a), Paspalum dilatatum Poir. 8 (1), Pastinaca sativa L. 2 (+), Periploca graeca L. 64 (+), Persicaria lapathifolia (L.) Delarbre 64 (+), Phragmites australis (Cav.) Steud. 6 (2a), Polygonum aviculare aggr. 52 (+), Prunella vulgaris L. 20 (+), Prunus spinosa L. (b) 18 (+), Rumex crispus L. 52 (+), Salix alba L. (b) 5 (+), Scirpus sylvaticus L. 5 (2a), Setaria pumila (Poir.) Roem. & Schult. 64 (+), Sonchus oleraceus L. 64 (+), Torilis arvensis (Huds.) Link 5 (+), Urtica dioica L. 20 (3), Verbena officinalis L. 5 (2b). | ||||||||||
3. Results
3.1. Characteristics of Plant Communities and the Contribution of IAS
- Helianthus tuberosus community (Convolvuletalia sepium)
- Helianthus × laetiflorus community (Dorycnio recti-Rumicion conglomerati)
- Rubo sancti-Eupatorietum cannabini Myśliwy ass. nov. hoc loco
- Rubus caesius community (Dorycnio recti-Rumicion conglomerati)
- Rubus caesius-Eupatorium cannabinum community (Dorycnio recti-Rumicion conglomerati)
- Mentho longifolii-Pulicarietum dysentericae Slavnić 1958
3.2. The General Pattern of Tall Herb Vegetation and Its Relationships with Land Use Type and River Size
4. Discussion
4.1. The Position of Riparian Tall Herb Communities of Montenegro in the Classification Scheme
4.2. Floristic Composition of Plant Communities and the Contribution of IAS
4.3. Comments Regarding the Definition of Habitat 6430
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A










References
- Ward, J.V.; Tockner, K.; Schiemer, F. Biodiversity of floodplain river ecosystems: Ecotones and connectivity. Regul. Rivers Res. Manag. 1999, 15, 125–139. [Google Scholar] [CrossRef]
- Ward, J.V.; Tockner, K.; Arscott, D.B.; Claret, C. Riverine landscape diversity. Freshw. Biol. 2002, 47, 517–539. [Google Scholar] [CrossRef]
- Leyer, I. Predicting plant species’ responses to river regulation. The role of water level fluctuations. J. Appl. Ecol. 2005, 42, 239–250. [Google Scholar] [CrossRef]
- Janssen, J.A.M.; Rodwell, J.S.; García Criado, M.; Gubbay, S.; Haynes, T.; Nieto, A.; Sanders, N.; Landucci, F.; Loidi, J.; Symank, A.; et al. European Red List of Habitats. Part 2. Terrestrial and Freshwater Habitats; Publications Office of the European Union: Luxembourg, 2016. [Google Scholar] [CrossRef]
- Stępień, E.; Zawal, A.; Buczyński, P.; Buczyńska, E.; Szenejko, M. Effects of dredging on the vegetation in a small lowland river. PeerJ 2019, 7, e6282. [Google Scholar] [CrossRef] [PubMed]
- Evans, D. Interpreting the habitats of Annex 1: Past, present and future. Acta Bot. Gall. 2010, 157, 677–686. [Google Scholar] [CrossRef]
- Interpretation Manual of European Union Habitats—Version EUR 28; European Commission DG-ENV: Brussels, Switzerland, 2013.
- Müller, T. Klasse: Artemisietea vulgaris Lohm., Prsg. et Tx. in Tx. 50. In Süddeutsche Pflanzengesellschaften; Oberdorfer, E., Ed.; Gustav Fischer Verlag: Stuttgart, Germany, 1983; Volume 3, pp. 135–177. [Google Scholar]
- Brzeg, A. Przegląd systematyczny zbiorowisk okrajkowych dotąd stwierdzonych i mogących występować w Polsce. Fragm. Florist. Et Geobot. 1989, 34, 385–423. [Google Scholar]
- Mucina, L. Galio-Urticetea. In Die Pflanzengesellschaften Österreichs; Mucina, L., Grabherr, G., Ellmauer, T., Eds.; I. Anthropogene Vegetation; Fisher: Jena, Germany, 1993; pp. 203–251. [Google Scholar]
- Richardson, D.M.; Holmes, P.M.; Esler, K.J.; Galatowitsch, S.M.; Stromberg, J.C.; Kirkman, S.P.; Pyšek, P.; Hobbs, R.J. Riparian vegetation: Degradation, alien plant invasions, and restoration prospects. Divers. Distrib. 2007, 13, 126–139. [Google Scholar] [CrossRef]
- Medvecká, J.; Jarolímek, I.; Zaliberová, M. Ruderal vegetation of the Horná Orava Region 2. Galio-Urticetea Epilobietea Angustifolii Thaiszia 2010, 20, 17–52. [Google Scholar]
- Pattison, Z.; Minderman, J.; Boon, P.J.; Willby, N. Twenty years of change in riverside vegetation: What role have invasive plants played? Appl. Veg. Sci. 2017, 20, 422–434. [Google Scholar] [CrossRef]
- Myśliwy, M. Diversity and environmental variability of riparian tall herb fringe communities of the order Convolvuletalia sepium in Polish river valleys. Monogr. Bot. 2019, 108, 1–129. [Google Scholar] [CrossRef]
- Myśliwy, M. Senecionetum fluviatilis in Poland from a European perspective—Diversity, distribution and threats. Plant Biosyst. —Int. J. Deal. All Asp. Plant Biol. 2020, 154, 814–826. [Google Scholar] [CrossRef]
- Stanciu, E. Establishment of NATURA 2000 Network. Guidelines for the Management of Natura 2000 Sites—Montenegro. Final Document, Submitted to the Steering Committee. ProPark Foundation for Protected Areas, Romania. 2019. Available online: https://propark.ro/en/publicatii/natura-2000-management-guidelines-for-montenegro-608.html (accessed on 8 August 2023).
- Angiolini, C.; Viciani, D.; Bonari, G.; Lastrucci, L. Habitat conservation prioritization: A floristic approach applied to a Mediterranean wetland network. Plant Biosyst. 2017, 151, 598–612. [Google Scholar] [CrossRef]
- Tzonev, R.T. Two new associations from the herbaceous riparian vegetation in the Central Danubian Plain, Bulgaria. Phytol. Balc. 2017, 23, 271–280. [Google Scholar]
- Dimopoulos, P.; Tsiripidis, I.; Xystrakis, F.; Kallimanis, A.; Panitsa, M. Methodology for Monitoring and Conservation Status Assessment of the Habitat Types in Greece; National Center of Environment and Sustainable Development, Ministry of Environment and Energy: Athens, Greece, 2018.
- Petrović, D.; Hadžiablahović, S.; Vuksanović, S.; Mačić, V.; Lakušić, D. Catalogue of Habitat Types of EU Importance of Montenegro; Version 3; Environmental Protection Agency of Montenegro, Podgorica, Montenegro & Faculty of Forestry, University of Banja Luka, Banja Luka, Bosnia and Herzegovina. 2019. Available online: https://www.ucg.ac.me/skladiste/blog_41660/objava_79477/fajlovi/KATALOG%20NATURA%202000%20STANI%C5%A0TA%20FINAL%202019.pdf (accessed on 25 August 2023).
- Pešić, V.; Paunović, M.; Kostianoy, A.G. The Rivers of Montenegro: Introductory Remarks. In The Rivers of Montenegro. The Handbook of Environmental Chemistry; Pešić, V., Paunović, M., Kostianoy, A.G., Eds.; Springer Nature Switzerland AG: Cham, Switzerland, 2020; Volume 93, pp. 1–12. [Google Scholar]
- Bošković, M.; Bajković, I. Quantitative characteristics of surface runoff of waters in Montenegro. BALWOIS 2006–403. 2006. Available online: https://balwois.com/wp-content/uploads/old_proc/ffp-870.pdf (accessed on 1 August 2023).
- Burić, D.; Ducić, V.; Mihajlović, J. The climate of Montenegro: Modificators and types—Part two. Bull. Serb. Geogr. Soc. 2014, 94, 73–90. [Google Scholar] [CrossRef]
- Braun-Blanquet, J. Pflanzensoziologie, 3rd ed.; Springer: New York, NY, USA, 1964. [Google Scholar]
- Barkman, J.J.; Doing, H.; Segal, S. Kritische Bemerkungen und Vorschläge zur quantitativen Vegetationanalyse. Acta Bot. Neerl. 1964, 13, 394–419. [Google Scholar] [CrossRef]
- Hennekens, S.M.; Schaminee, J.H.J. Turboveg, a comprehensive database management system for vegetation data. J. Veg. Sci. 2001, 12, 589–591. [Google Scholar] [CrossRef]
- van der Maarel, E. Transformation of cover-abundance values for appropriate numerical treatment—Alternatives to the proposals by Podani. J. Veg. Sci. 2007, 18, 767–770. [Google Scholar]
- Kovach, W.L. MVSP—A MultiVariate Statistical Package for Windows. Version 3.2. In Users’ Manual; Kovach Computing Services: Pentraeth, UK, 2010. [Google Scholar]
- ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference manual and CanoDraw for Windows User’s guide. In Software for Canonical Community Ordination (Version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002. [Google Scholar]
- Jongman, R.H.G.; ter Braak, C.J.F.; van Tongeren, O.F.R. (Eds.) Data Analysis in Community and Landscape Ecology; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Euro+Med 2006+ [Continuously Updated]: Euro+Med PlantBase—The Information Resource for Euro-Mediterranean Plant Diversity. Available online: http://www.europlusmed.org (accessed on 17 July 2023).
- Mucina, L.; Bültmann, H.; Dierßen, K.; Theurillat, J.; Raus, T.; Čarni, A.; Šumberová, K.; Willner, W.; Dengler, J.; García, R.G.; et al. Vegetation of Europe: Hierarchical floristic classification system of vascular plant, bryophyte, lichen, and algal communities. Appl. Veg. Sci. 2016, 19 (Suppl. S1), 3–264. [Google Scholar] [CrossRef]
- Dring, J.; Hoda, P.; Mersinllari, M.; Mullaj, A.; Pignatti, S.; Rodwell, J. Plant communities of Albania—A preliminary overview. Ann. Di Bot. N.S. 2002, 2, 7–30. [Google Scholar]
- Petronic, S.; Bratic, N.; Jakisic, T.; Tunguz, V. Habitat types of European importance in the area of wetlands Gromizelj (Bosnia and Hercegovina). AGROFOR Int. J. 2017, 2, 10–18. [Google Scholar] [CrossRef]
- Škvorc, Ž.; Jasprica, N.; Alegro, A.; Kovačić, S.; Franjić, J.; Krstonošić, D.; Vraneša, A.; Čarni, A. Vegetation of Croatia: Phytosociological classification of the high-rank syntaxa. Acta Bot. Croat. 2017, 76, 200–224. [Google Scholar] [CrossRef]
- Tabašević, M.I. Ecological and Syntaxonomic Characterization of Ruderal Vegetation in Serbia. Ph.D. Thesis, University of Belgrade, Faculty of Biology, Belgrade, Serbian, 2022. [Google Scholar]
- Šilc, U.; Čarni, A. Conspectus of vegetation syntaxa in Slovenia. Hacquetia 2012, 11, 113–164. [Google Scholar] [CrossRef]
- Borhidi, A.; Kevey, B.; Varga, Z. Checklist of the higher syntaxa of Hungary. Ann. Bot. 1999, 57, 159–166. [Google Scholar]
- Kovács, A.J. Syntaxonomical checklist of the plant communities of Szeklerland (Eastern Transylvania). Kanitzia 2004, 12, 75–149. [Google Scholar]
- de Foucault, B. Contribution au prodrome des végétations de France: Les Filipendulo ulmariae-Convolvuletea sepium Géhu et Géhu-Franck 1987. J. Bot. 2011, 53, 73–137. [Google Scholar] [CrossRef]
- Biondi, E.; Blasi, C.; Allegrezza, M.; Anzellotti, I.; Azzella, M.M.; Carli, E.; Casavecchia, S.; Copiz, R.; Del Vico, E.; Facioni, L.; et al. Plant communities of Italy: The Vegetation Prodrome. Plant Biosyst. 2014, 148, 728–814. [Google Scholar] [CrossRef]
- Preislerová, Z.; Jiménez-Alfaro, B.; Mucina, L.; Berg, C.; Bonari, G.; Kuzemko, A.; Landucci, F.; Marcenò, C.; Monteiro-Henriques, T.; Novák, P.; et al. Distribution maps of vegetation alliances in Europe. Appl. Veg. Sci. 2022, 25, e12642. [Google Scholar] [CrossRef]
- Gradstein, S.R.; Smittenberg, J.H. The hydrophilous vegetation of western Crete. Vegetatio 1977, 34, 65–86. [Google Scholar] [CrossRef]
- Hadžiablahović, S. The diversity of the flora and vegetation of Lake Skadar/Shkodra. In The Skadar/Shkodra Lake Environment; Pešić, V., Karaman, G., Kostianoy, A.G., Eds.; The Handbook of Environmental Chemistry 80; Springer Nature Switzerland AG: Cham, Switzerland, 2018; pp. 203–238. [Google Scholar]
- Kark, S. Ecotones and ecological gradients. In Ecological Systems: Selected Entries from the Encyclopedia of Sustainability Science and Technology; Leemans, R., Ed.; Springer Science + Business Media: New York, NY, USA, 2013; pp. 147–160. [Google Scholar]
- Stępień, E.; Rosadziński, S. Communities of the Bidentetea class of small coastal river valleys of the Western Pomerania (Poland). Ecol. Montenegrina 2020, 28, 8–19. [Google Scholar] [CrossRef]
- Loidi, J.; Navarro, C. Datos sobre las alianzas Dauco-Melilotion Görs 1966 y Convolvulion sepii R. Tax. 1947 en el País Vasco. Acta Bot. Barcinon. 1988, 37, 257–264. [Google Scholar]
- Loidi, J.; Berastegi, A.; Biurrun, I.; García-Mijangos, I.; Herrera, M. Perennial nitrophilous vegetation of the northerm Iberian Peninsula. J. Veg. Sci. 1996, 7, 575–584. [Google Scholar] [CrossRef]
- Brullo, S.; Spampinato, G. La vegetazione dei corsi d’acqua della Sicilia. Boll. Accad. Gioenia Sci. Nat. 1990, 23, 119–252. [Google Scholar]
- Minissale, P.; Sciandrello, S.; Spampinato, G. Analisi della biodiversità vegetale e relativa cartografia della Riserva Naturale Orientata “Pantalica, Valle dell’Anapo e Torrente Cava Grande ” (Sicilia sudorientale). Quad. Bot. Amb. Appl. 2007, 18, 145–207. [Google Scholar]
- de Foucault, B. Quelques données phytosociologiques sur les ripisylves languedociennes et certains de leurs satellites. Carnets Bot. 2021, 42, 1–14. [Google Scholar] [CrossRef]
- Stešević, D.; Petrović, D. Preliminary list of plant invaders in Montenegro. Biol. Nyssana 2010, 1, 35–42. [Google Scholar]
- Reports on European Red List of Habitats. E5.4 Lowland Moist or Wet Tall-Herb and Fern Fringe. 2016. Available online: https://forum.eionet.europa.eu/european-red-list-habitats/library/terrestrial-habitats/e.-grasslands/e5.4-lowland-moist-or-wet-tall-herb-and-fern-fringe (accessed on 12 August 2023).
- Fois, M.; Bacchetta, G.; Caria, M.C.; Cogoni, D.; Farris, E.; Fenu, G.; Manca, M.; Pinna, M.S.; Pisanu, S.; Rivieccio, G.; et al. Proposals for improvement of Annex I of Directive 92/43/EEC: Sardinia. Plant Sociol. 2021, 58, 65–76. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Myśliwy, M.; Pešić, V. Tall Herb Fringe Vegetation on Banks of Montenegrin Rivers as a Habitat Type of European Importance. Water 2023, 15, 3684. https://doi.org/10.3390/w15203684
Myśliwy M, Pešić V. Tall Herb Fringe Vegetation on Banks of Montenegrin Rivers as a Habitat Type of European Importance. Water. 2023; 15(20):3684. https://doi.org/10.3390/w15203684
Chicago/Turabian StyleMyśliwy, Monika, and Vladimir Pešić. 2023. "Tall Herb Fringe Vegetation on Banks of Montenegrin Rivers as a Habitat Type of European Importance" Water 15, no. 20: 3684. https://doi.org/10.3390/w15203684
APA StyleMyśliwy, M., & Pešić, V. (2023). Tall Herb Fringe Vegetation on Banks of Montenegrin Rivers as a Habitat Type of European Importance. Water, 15(20), 3684. https://doi.org/10.3390/w15203684







