Abstract
Resource recovery from wastewater is very important in view of a circular economy approach in the water field. Among the different technologies applied to realize circular economy, an attractive option is the use of nutrient-enriched media that can be utilized as slow-release fertilizers. Zeolites have been re-discovered for their key role in ammonium (NH4+) adsorption from treated wastewater. Although many studies have been carried out to assess the ability of zeolites to adsorb NH4+, only few papers concerning NH4+ desorption from zeolites are available in the literature. Therefore, this study investigated NH4+ desorption from mineralogically different zeolites, before (ZNS and ZNC) and after (ZSS and ZSC) their treatment with sodium chloride. The zeolites differed in mordenite content. The amount of the desorbed NH4+ varied from 78 to 84% of the total NH4+ adsorbed. In particular, the NaCl-treated materials showed the largest desorption (27.6 ± 0.2 mg L−1, and 27.9 ± 0.7 mg L−1, ZSS, and ZSC, respectively) as compared to the untreated zeolites (22.9 ± 0.3 mg L−1, and 24.2 ± 0.3 mg L−1, ZNS, and ZNC, respectively) because of the different affinity of the cations for the zeolite surface. A monomodal pseudo-first-order model best approximated the desorption kinetics, suggesting only one mechanism of NH4+ desorption from zeolites. Such a mechanism is based on the ion exchange between dissolved Na+ and adsorbed NH4+. The desorption kinetics also showed that NH4+ desorbed slower from the NaCl-treated zeolites than the untreated ones. This effect was explained by the different affinity of Na+ and NH4+ for the zeolite surfaces as due to the diverse sizes of the Na+ and NH4+ hydration spheres. By revealing the effect of zeolite mineralogy and surface treatments in the desorption of NH4+, this study can suggest new and effective synthetic strategies for the achievement of cheap new materials to be applied in environmental remediation within a circular economy perspective.
1. Introduction
The efficient removal of nitrogen from wastewater is crucial for reducing the eutrophication caused by the excessive use of fertilizers. The Water Framework Directive 2000/60/EC and Council Directive 91/271/EEC have established remarkably stringent permissible thresholds (ammonium-N < 1 mg L−1 and nitrate-N 10–30 mg L−1 for freshwater discharges, and <50 mg L−1 for seawater) governing the release of N-containing wastewater into the environment [1].
Various conventional removal methods, including biological, physical, and chemical processes, can contribute significantly to making wastewater treatment more sustainable. These methods play a crucial role in reducing nutrient removal costs and increasing the supply of fertilizers for food production [2,3]. Among these different physical methods, adsorption emerges as one of the most applied techniques. Compared to biological processes, adsorption offers several advantages: (i) adsorption leads to shorter process times, accelerating the removal of contaminants from wastewater; (ii) this method directly removes contaminants from wastewater without generating unwanted reaction by-products; (iii) adsorption allows for the easy regeneration of the adsorbent in situ, improving its longevity and sustainability; (iv) it also opens up the possibility of using NH4+-enriched adsorbents as slow-release fertilizers, promoting environmentally friendly nutrient management; and (v) adsorption boasts low operating costs, making it a cost-effective choice for wastewater treatment [4].
Despite the aforementioned promises and potential benefits, the main challenges are rooted in the complex tasks for the identification of suitable adsorbent materials that meet the following essential criteria: being readily available, inexpensive, and possessing a high degree of efficiency [5]. In this regard, zeolites emerge as the most promising candidates for nitrogen removal and represent a good option for achieving environmentally sustainable solutions for resource recovery from wastewater [5,6,7].
Zeolites, fascinating in their nature, are naturally occurring volcanogenic sedimentary minerals that originated from the interaction of complex chemical and physical processes within rocks undergoing various transformations due to atmospheric phenomena [8]. These minerals exhibit three-dimensional structures composed of tetrahedral aluminosilicates in which silicon and aluminum atoms form covalent bonds with oxygen atoms. This arrangement leads to the creation of interconnected cages and channels, which play a key role in the adsorption properties of zeolites [9]. Depending on their geological origin and geological formation processes, zeolites exhibit specific physical and chemical properties that play a key role in influencing their ion-exchange capacity as well as their ability to absorb various gases and vapors [10,11]. Due to these distinct physico-chemical characteristics, such as a remarkably high pore volume, low bulk density, and excellent cation exchange capacity (CEC), zeolites find wide applications in the field of agriculture [12]. Indeed, they are often used to improve a multitude of soil properties, including nutrient retention and water-holding capacity, soil structure refinement, permeability improvement, and increased infiltration rates [13]. Consequently, the strategic integration of zeolites into soil ecosystems produces multiple benefits, resulting in improved plant growth, reduced water leaching, and increased nutrient utilization efficiency [7,14].
Natural zeolites, as well as their modified or synthesized forms, emerged as formidable adsorbents in the realm of nutrient removal from wastewater. This acclaim is rooted in their remarkable cation exchange capacity [1,6,11]. Notably, recent research has yielded compelling evidence affirming zeolites’ proficiency in adsorbing NH4+ ions from diverse aqueous solutions, thereby encompassing the complex milieu of wastewater.
Inizio Modulo
Guida et al. [1] investigated the NH4+ adsorption capacity of six synthetic zeolites, one natural (clinoptilolite) and one engineered in adsorbing NH4+ from a 1 g NH4+ L−1 solution. They observed that NH4+ adsorption ranged from 65 to 85 mg NH4+ g−1 depending on the pre-treatment and shape of the tested zeolites. In a previous study, Langwaldt [15], investigating the NH4+ adsorption capacity of eight different zeolites with different mineralogy, found that the amount of adsorbed NH4+ ranged from 16 to 48 mg NH4+ g−1. Such results suggested that zeolite adsorption ability also depends on the mineralogy composition. The investigation of the complex mechanisms that regulate the adsorption by zeolites represents a crucial point in scientific research. However, in the context of a circular economy perspective oriented towards efficient nutrient recovery, the deepening of the complexities of the nutrient desorption process from zeolites assumes paramount importance [16]. This perspective is underlined by the fact that the NH4+-enriched zeolite has immense potential as a soil conditioner capable of improving various physical and chemical characteristics of the soil.
At the same time, the NH4+-enriched zeolite, in its role as a soil conditioner, exhibits a unique ability to gradually release the adsorbed NH4+. This slow-release characteristic effectively transforms it into a sustainable fertilizer, ensuring a constant supply of accessible nutrients for plant uptake [17]. Furthermore, zeolites offer a further advantage in the form of reusability. After application, these zeolite materials can be regenerated through the application of a salt solution, facilitating the recovery of desorbed nutrients. These recovered nutrients can then be reapplied to the soil, completing a beneficial cycle of nutrient management and sustainability [11].
To the best of our knowledge, and according to Saliu and Oladoja [18], only Guaya et al. [7] have evaluated the application to the soil of Al-, Fe-, and Mn-functionalized zeolites, enriched with NH4+ recovered from urban wastewater, as soil conditioners. In particular, these authors [7] observed that N and P content in sunflower plant tissues increased from 5 to 45% and from 54 to 210%, respectively. The highest nutrient uptake led to an increase in sunflower biomass from 50 to 269%. However, the authors did not provide any information about the timing of NH4+ release in either soil or water. Different methods can carry out the regeneration of zeolites. The most popular regeneration technique commonly consists of using ionic brines, e.g., sodium chloride (NaCl), where the Na+ ions replace the adsorbed NH4+, thus releasing it into the liquid phase according to the following chemical exchange reaction [19,20]:
NaCl + NH4-Zeolite ⇌ NH4Cl + Na-Zeolite
According to Sengupta et al. [4], NH4+ can be recovered from an aqueous solution by raising the pH of the solution to above 9.3. This value corresponds to the pKa of the NH4+-NH3 conjugated acid-base pair. The NH3 gas can be further absorbed into a solution of H2SO4 or HNO3 and used as a fertilizer [4]. The chemical regeneration of zeolites is one of the major issues when dealing with these materials for the NH4+ removal strategy. This is because the process involves large amounts of NaCl and water to detach the absorbed cations from zeolite [21]. Furthermore, it is worth noting that the existing data on the frequency with which zeolites can undergo regeneration and be recycled efficiently reveal a considerable lack of uniformity. This variability depends mainly on a confluence of factors, including the inherent mineralogical properties of each zeolite sample and the particular ionic composition present in the wastewater matrix [21,22].
Given this intricate interplay of variables, there is a need for more comprehensive and meticulous investigations. In particular, further studies are needed to clarify the intricate relationship between zeolite mineralogy and the resulting NH4+ desorption efficiency. Such research is likely to provide valuable insights into the dynamics governing the sustainable use and regeneration of zeolitic materials in wastewater treatment, thus contributing to the continuous advancement of environmental science and resource management. This study aims to evaluate the desorption kinetics of NH4+, one of the most important plant nutrients, from mineralogically different zeolites already used to recover this ion from synthetic wastewaters, as reported in Muscarella et al. [23]. In particular, two natural zeolites with different mineralogy were firstly loaded with NH4+ and then treated with a 1 M NaCl solution in order to allow NH4+ desorption. Different kinetics models were applied to understand the chemical mechanism behind NH4+ desorption. The results revealed that both mineralogy and NaCl treatment affect the NH4+ desorption rate. Therefore, this study can be considered a pivotal study to produce new and more efficient strategies for the synthesis of new materials to be applied in environmental remediation and soil amendment in the perspective of the circular economy.
2. Materials and Methods
2.1. Tested Zeolites
Two different mixtures of natural zeolites, indicated here as ZNS and ZNC, with a diameter of 0.5–1 mm were used. Prior to their utilization, the zeolites were washed with distilled water (3 times × 100 mL of water × 10 g of zeolite) to eliminate possible impurities. Then, they were dried at 105 °C for 2 h. Subsequently, 1 g of each dried zeolite was added with 50 mL of 1 M NaCl (Sigma-Aldrich, Milan, Italy, 97% purity) and shaken for 24 h [23]. Finally, to eliminate the excess Na+, the zeolites were washed with distilled water, as reported a few lines above.
The specific surface area of the untreated zeolite mixtures was determined by using the nitrogen gas adsorption technique with automated equipment (Nova touch LX1, Quantachrome Instrument, Boynton Beach, FL, USA). The multipoint Brunauer-Emmett-Teller (BET) isotherm adsorption data were used for analysis. The morphology and elemental composition of both treated and untreated zeolite mixtures were examined by using a scanning electron microscope (SEM) in combination with an energy-dispersive X-ray (EDX) analyzer (Phenom Pharos and Phenom XL, Phenom-World Thermo Scientific, Waltham, MA, USA).
The structure and phase of the zeolite mixtures were identified by using powder X-ray diffraction (XRD) with an X’PERT PRO X-ray diffractometer (Pan Analytical, Malvern, UK). To assess the functional groups present in the zeolite mixtures, Attenuated-Total-Reflectance (ATR) Fourier Transformed (FT) Infrared (IR) spectroscopy was applied (Spectrum Two, PerkinElmer, Waltham, Massachusetts, USA). The spectra were acquired in the wavenumber range from 4000 to 400 cm−1, with a spectral resolution of 16 cm−1, and 100 scans on samples that were dried for 2 h at 105 °C and finely ground in an agate mortar [23]. Origin software (Version 7.5) was used to analyze all the ATR-FTIR spectra.
2.2. NH4+ Adsorption-Desorption by Zeolitic Mixtures
The described zeolites were NH4+-enriched according to the procedure reported in Muscarella et al. [23]. Briefly, 1 g of each dry zeolite was placed in an orbital shaker and shaken continuously for 24 h at a speed of 80 rpm, while immersed in a solution containing 100 mL of NH4+ at a concentration of 20 g L−1, all maintained at a constant temperature of 25 °C. After 24 h, the samples underwent three washes with 200 mL of distilled water to remove excess NH4+, and were then dried at 105 °C for 2 h. Total NH4+ adsorption by the zeolites was determined using Kjeldahl distillation, which involved a 6 min process by using 30 mL of a 33% (w/v) NaOH solution with high purity (99%) in pearl form, sourced from Sigma-Aldrich [23]. To determine the desorption capacity of zeolite, 1 g of each NH4+-enriched zeolite was shaken with 100 mL of 1 M NaCl on a horizontal shaker for 48 h at 80 rpm at 25 °C. After this step, the material underwent a thorough rinsing process and was washed three times with 200 mL of distilled water to ensure the removal of any residual impurities. It was then subjected to a drying process in an oven, with the temperature maintained at 105 °C for a duration of 2 h in order to prepare it for subsequent analysis. The analytical investigation involved the determination of retained NH4+, using the established Kjeldahl distillation method, as illustrated above. The quantification of NH4+ desorption was carried out on the basis of the fundamental relationship expressed in Equation (1), which involves calculating the difference between the total amount of NH4+ adsorbed and the portion that remained retained and not exchanged by the Na+, thus providing valuable insights into the dynamic process under investigation [1].
NH4+desorbed = NH4+adsorbed − NH4+retained
All the experiments described in the lines above were conducted in triplicate.
Data on the amounts of NH4+ adsorbed by zeolitic mixtures were analyzed by using a two-way ANOVA procedure. In the aforementioned analysis, mineralogy and NaCl treatment were used as factors. Finally, the Tukey test was carried out in order to assess significant differences at p < 0.05.
2.3. Desorption Kinetics
Desorption kinetics were evaluated, during 48 h, by placing in contact 1 g of each sample (ZNC, ZSC, ZNS, ZSS) with 100 mL of a 1 M NaCl solution on a horizontal shaker at 80 rpm at 25 °C. The concentration of NH4+ in solution was determined after 15, 30, 45 min, 1, 2, 4, 8, 24, and 48 h using the Berthelot colorimetric method [24]. The NH4+ kinetics desorption data were mathematically examined by using the Origin (Version 7.5) software program.
2.4. Theory: Kinetic Models
The study of the kinetics of NH4+ desorption, through the application of different kinetic models, allows the evaluation of the efficiency of zeolite regeneration. In this study, two different models were tested.
The monomodal pseudo-first-order model given in Equation (2) is known as physisorption. This model is based on attractive forces between the sorbent and adsorbate molecules and assumes that the desorption rate is exclusively influenced by the amount of adsorbate that is in contact with the porous surface of the zeolite [25].
qt = qe (1 − e−k1et)
In Equation (2), qt is the amount of NH4+ desorbed at time t, qe is the amount of NH4+ desorbed at equilibrium, and k1 is the pseudo-first-order kinetic constant. The latter is directly related to the sample desorption rate of NH4+. The higher the value, the faster the desorption rate is.
The bimodal pseudo-first-order model given in Equation (3) presupposes the existence of a polymodality in the desorption process. This means that a simultaneous presence of multiple pseudo-first-order monomodal desorption patterns occurs, thereby arising from distinct exchange mechanisms.
qt = qe1 (1 − e−k1t) + qe2 (1 − e−k2t)
In Equation (3), qt is the amount of NH4+ desorbed at time t, qe1 and qe2 are the amount of NH4+ desorbed at equilibrium, respectively, and k1 and k2 are the respective pseudo-first-order monomodal kinetic constants. The larger the kinetic constant values, the faster the adsorption rates are led by k1 and k2.
3. Results and Discussion
3.1. BET, XRD, SEM-EDX, and ATR-FTIR Characterization of Tested Zeolites
The BET, XRD, SEM-EDX, and ATR-FTIR data have been already reported and discussed in Muscarella et al. [23]. Briefly, the specific surface areas of the zeolitic mixtures were 40 ± 2 m2 g−1 for ZNS and 47 ± 2 m2 g−1 for ZNC, respectively. The XRD analyses showed that ZNS was a mixture of heulandite, mordenite, clinoptilolite, and stellerite, while ZNC was composed only of heulandite and mordenite.
While the analysis of XRD spectra provided results showing no changes in the mineralogical composition within the zeolite mixtures after NaCl treatment, a closer examination of the samples using scanning electron microscopy (SEM) revealed significant transformations in the surface characteristics of the treated zeolite mixtures. In particular, the untreated samples exhibited a discrete disorder in crystal orientation, a feature that contrasted the treated ones. SEM images of the treated samples showed a distinct and clearly more ordered pattern, characterized by a finer grain structure and well-defined edges. These observations provide convincing evidence of the profound impact of NaCl treatment on the surface properties of zeolite mixtures, offering insight into the surface structural alterations induced by this specific modification process.
Such changes are analogous to those observed by Wijesinghe et al. [26] for the NaCl-treated zeolite.
SEM-EDX elemental analysis indicated that the treatment with NaCl caused a decrease in oxygen, silicon, and aluminium, while increasing Na concentration. In addition, the presence of NaCl was confirmed by its non-uniform distribution on the surfaces of the treated zeolitic mixtures, as evaluated by SEM-EDX analysis. However, as expected, the application of NaCl treatment did not change the Si/Al, Si/O and Al/O ratios. This lack of alteration suggested no lattice structure modification of the two zeolitic mixtures. The two investigated zeolites had similar ATR-FTIR spectra. Byrappa and Kumar [27] reported that the infrared spectrum in the 1500–400 cm−1 region can be employed to analyze and understand the structural characteristics of the zeolite frameworks. The bending of the bonds between tetrahedral (Si, Al) and octahedral species (Al, Fe, Mg) was related by the increase in absorbance in the range between 500 and 420 cm−1; the peaks observed in the intervals 720–650 cm−1 and 1250–950 cm−1 were due to symmetrical and asymmetrical stretching, which are typical of internal tetrahedra linkages. Other peaks attributed to typical external tetrahedra linkages were observed in the ranges of 650–500 cm−1 (double ring), 420–300 cm−1 (pore opening), 820–750 cm−1 (symmetrical stretching), and 1150–1050 cm−1 (asymmetrical stretching).
3.2. NH4+ Desorption
After 48 h of contact between the NH4+-enriched zeolites and the NaCl solution, the amount of the desorbed NH4+ varied from 78 to 84% of the total NH4+ adsorbed on the zeolites, as already described in Muscarella et al. [23]. Notably, the larger the amount of NH4+ adsorbed, the higher the amount of NH4+ desorbed (Figure 1).
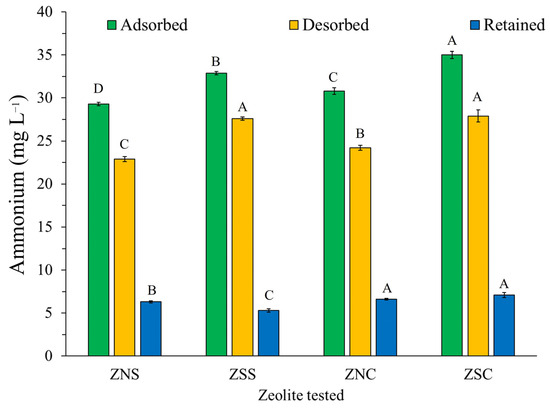
Figure 1.
Amount of NH4+ desorbed by the tested zeolites during 24 h from a 1 M NaCl solution. Different letters indicate significant differences among zeolites for the same parameter (adsorbed, desorbed, and retained). Values are mean ± standard deviation of three replicates.
The amount of NH4+ desorbed in the present study was slightly lower than that reported in Rahmani et al. [28]. In fact, these authors found a desorption rate between 95 and 98% for the natural Chinese clinoptilolite. Such a difference may be due to the diverse mineralogical composition of their zeolites, as compared to those used here. Moreover, the different particle sizes of their zeolites [28] can also cause the different results that have been observed here.
It is noteworthy that the zeolites in the present study that desorbed the highest amount of NH4+ (27.6 ± 0.2 mg L−1and 27.9 ± 0.7 mg L−1, respectively) were those treated with NaCl, i.e., ZSS and ZSC, with no significant difference between them. The higher desorption of these zeolites can be conceivably associated with the larger amount of NH4+ that is already adsorbed on each of them. As described in the discussion below, the mechanism of the desorption process is primarily linked to the ion exchange between the dissolved Na+ and adsorbed NH4+. Therefore, it is possible to suggest that the NH4+ desorption capacity of zeolites is primarily affected by NaCl treatment rather than mineralogical composition.
3.3. NH4+ Desorption Kinetics
NH4+ desorbed from zeolites was assessed at various time intervals during 48 h (Figure 2). The total amount of desorbed NH4+ decreased according to the following order ZSC > ZSS > ZNC = ZNS (Figure 2). Kinetics data were similar to that obtained by the static desorption experiment.
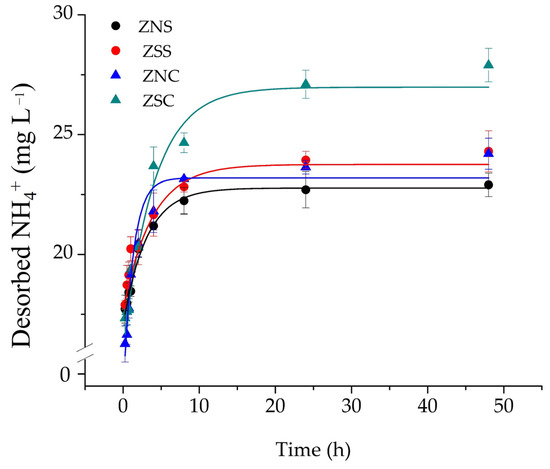
Figure 2.
Monomodal pseudo-first-order NH4+ desorption kinetics by treated clinoptilolite. Values are mean ± standard deviation of three replicates.
The rate of NH4+ desorption was faster during the first 8 h from contact with the NaCl solution, but then slowed down by reaching a plateau after about 24 h (Figure 2). After 24 h, all the zeolites desorbed about 80% of the adsorbed NH4+. These results disagree with the paper from Deng et al. [29], who achieved 85% of desorption within 2 h of reaction time by using a lower NaCl concentration. In fact, Deng et al. used a NaCl concentration ranging from 0.2 up to 0.7 M. Conversely, our results accord with those of Neag et al. [30], who used not only a zeolite particle size similar to that used here (0.5–1.25 mm vs. 0.5–1.5 mm, respectively), but also the same NaCl concentration (i.e., 1 M). The kinetics of NH4+ desorption by zeolite was mathematically analyzed by applying eqs. 1 and 2, respectively. Although the bimodal pseudo-first-order kinetic model returned more reasonable R2 and χ2 values than the monomodal first-order kinetic model, the parameter errors were far larger than those found in the application of the monomodal pseudo-first-order model (Table 1).

Table 1.
Parameter values obtained from the application of monomodal and bimodal pseudo-first- order kinetic models. Values are mean ± standard deviation of three replicates.
This is due to the mathematical model overestimation caused by the larger number of parameters as compared to the number of experimental data.
Based on the above considerations, to describe the desorption process, we referred to the parameters obtained by applying the monomodal pseudo-first-order kinetic model. Hence, a single NH4+ desorption mechanism, which involves the ion exchange between Na+ in solution and NH4+ adsorbed onto zeolites, could account for the process (Figure 3).
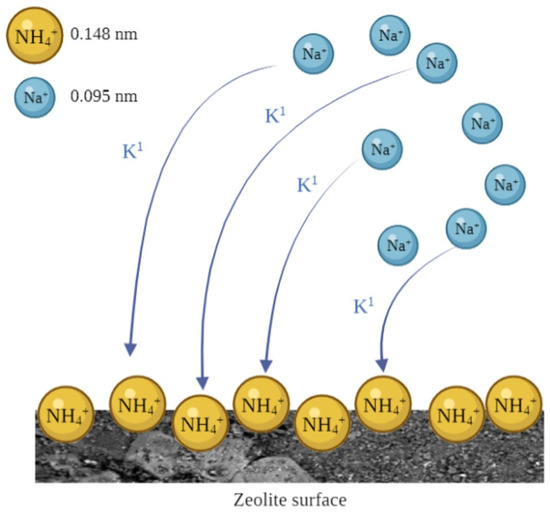
Figure 3.
Monomodality of NH4+ substitution on the surface of zeolites by Na+ in relation to cations’ size.
The parameter qe1 refers to the amount of NH4+ in solution when the equilibrium is reached. Based on the parameters reported in Table 1, ZNC and ZSC desorbed more NH4+ than ZNS and ZSS. Moreover, the kinetic data suggested that NH4+ desorption was faster for ZNC than all the other tested zeolites. Such results can be explained by the different hydration radii of NH4+ and Na+. In fact, given that the former cation is larger than the latter, its hydration sphere is smaller than that of the second one [31]. Therefore, the affinity of NH4+ for the zeolite surface results is the largest, as compared to Na+. Finally, the results from NH4+ desorption kinetic studies provide useful information on the most efficient use of zeolites for practical applications. As an example, we may state that the slower releasing NH4+ zeolite should be more efficient as a long-term soil fertilizer.
4. Conclusions
The results reported in the present study proved that NH4+ desorption from zeolites may depend on both mineralogical composition and NaCl treatment. In particular, the amount of the desorbed NH4+ varied in the range of 78–84% of the total NH4+ adsorbed on the zeolites already described in our previous paper [23]. Moreover, here, it was evidenced that the largest amount of desorbed NH4+ occurred from the NaCl-treated zeolites, which were also involved in a larger adsorption of NH4+. The comparison between the untreated zeolites with different mineralogical compositions revealed that this later affects the amount of desorbed NH4+. The possible explanation was related to the cation exchange capacity of the minerals present in each investigated zeolite (i.e., CEC mordenite > CEC heulandite/clinoptilolite). However, we could also state that, when the zeolites were NaCl-treated, the main desorption effect was due to the larger surface affinity of NH4+ (as compared to Na+) because of the different hydration sphere sizes of the two cations in solution. In our opinion, the importance of the present study relies on the fact that we introduced knowledge about the microscopic chemical mechanisms involved in NH4+ desorption. This can introduce the possibility to better address the synthesis of new, cheaper, and more efficient zeolitic materials in the perspective of the circular economy.
Author Contributions
Conceptualization, V.A.L., L.B., P.C., and G.M.; methodology, V.A.L., P.C., and S.M.M.; formal analysis, S.M.M.; investigation, V.A.L. and S.M.M.; data curation, V.A.L., P.C., and S.M.M.; writing—original draft preparation, V.A.L., P.C., and S.M.M.; writing—review and editing, V.A.L., L.B., P.C., and G.M.; supervision, V.A.L. and G.M. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the project “Achieving wider uptake of water-smart solutions—WIDER UPTAKE” (grant agreement number: 869283), financed by the European Union’s Horizon 2020 Research and Innovation Programme. Website https://wideruptake.unipa.it/, https://www.sintef.no/projectweb/wider-uptake/. Principal investigator for the University of Palermo Prof. Giorgio Mannina; https://wideruptake.unipa.it/.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author.
Acknowledgments
The authors would like to thank Cosimo Masini and Zeocel Italia by Dnd Biotech for providing zeolite samples.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Guida, S.; Potter, C.; Jefferson, B.; Soares, A. Preparation and Evaluation of Zeolites for Ammonium Removal from Municipal Wastewater through Ion Exchange Process. Sci. Rep. 2020, 10, 12426. [Google Scholar] [CrossRef] [PubMed]
- Hojjati-Najafabadi, A.; Esfahani, P.N.; Davar, F.; Aminabhavi, T.M.; Vasseghian, Y. Adsorptive removal of malachite green using novel GO@ZnO-NiFe2O4-αAl2O3 nanocomposites. Chem. Engin. J. 2023, 471, 144485. [Google Scholar] [CrossRef]
- Xia, C.; Joo, S.W.; Hojjati-Najafabadi, A.; Xie, H.; Wu, Y.; Mashifana, T.; Vasseghian, Y. Latest advances in layered covalent organic frameworks for water and wastewater treatment. Chemosphere 2023, 329, 138580. [Google Scholar] [CrossRef]
- Sengupta, S.; Nawaz, T.; Beaudry, J. Nitrogen and Phosphorus Recovery from Wastewater. Curr. Pollut. Rep. 2015, 1, 155–166. [Google Scholar] [CrossRef]
- Han, B.; Butterly, C.; Zhang, W.; He, J.; Chen, D. Adsorbent Materials for Ammonium and Ammonia Removal: A Review. J. Clean. Prod. 2021, 283, 124611. [Google Scholar] [CrossRef]
- Muscarella, S.M.; Badalucco, L.; Laudicina, V.A.; Mannina, G. Chapter 5—Zeolites for the Nutrient Recovery from Wastewater. In Current Developments in Biotechnology and Bioengineering; Mannina, G., Pandey, A., Sirohi, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 95–114. [Google Scholar]
- Guaya, D.; Mendoza, A.; Valderrama, C.; Farran, A.; Sauras-Yera, T.; Cortina, J.L. Use of Nutrient-Enriched Zeolite (NEZ) from Urban Wastewaters in Amended Soils: Evaluation of Plant Availability of Mineral Elements. Sci. Total Environ. 2020, 727, 138646. [Google Scholar] [CrossRef]
- Nguyen, M.L.; Tanner, C.C.; Lahav, O.; Green, M.; Cyrus, J.S.; Reddy, G.B.; Abdusattar, T.; Hadi, A.; Kurniawan, T.; Dimova, G.; et al. The Application Effects of Natural Zeolite on Soil Runoff, Soil Drainage and Some Chemical Soil Properties in Arid Land Area. Desalination 2011, 34, 45–52. [Google Scholar]
- Guaya, D.; Valderrama, C.; Farran, A.; Sauras, T.; Cortina, J.L. Valorisation of N and P from Waste Water by Using Natural Reactive Hybrid Sorbents: Nutrients (N,P,K) Release Evaluation in Amended Soils by Dynamic Experiments. Sci. Total Environ. 2018, 612, 728–738. [Google Scholar] [CrossRef]
- Widiastuti, N.; Wu, H.; Ang, H.M.; Zhang, D. Removal of Ammonium from Greywater Using Natural Zeolite. Desalination 2011, 277, 15–23. [Google Scholar] [CrossRef]
- Chowdhury, S.; Khan, N.; Kim, G.H.; Harris, J.; Longhurst, P.; Bolan, N.S. Zeolite for Nutrient Stripping from Farm Effluents. In Environmental Materials and Waste: Resource Recovery and Pollution Prevention; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 569–589. [Google Scholar]
- Nakhli, S.A.A.; Delkash, M.; Bakhshayesh, B.E.; Kazemian, H. Application of Zeolites for Sustainable Agriculture: A Review on Water and Nutrient Retention. Water Air Soil Pollut. 2017, 228, 464. [Google Scholar] [CrossRef]
- Ramesh, K.; Reddy, D.D. Zeolites and Their Potential Uses in Agriculture. Adv. Agron. 2011, 113, 219–241. [Google Scholar]
- Polat, E.; Karaca, M.; Demir, H.; Onus, A.N. Use of Natural Zeolite (Clinoptilolite) in Agriculture. J. Fruit Ornam. Plant Res. 2004, 12, 183–189. [Google Scholar]
- Langwaldt, J. Ammonium Removal from Water by Eight Natural Zeolites: A Comparative Study. Sep. Sci. Technol. 2008, 43, 2166–2182. [Google Scholar] [CrossRef]
- Mannina, G.; Alduina, R.; Badalucco, L.; Barbara, L.; Capri, F.C.; Cosenza, A.; Di Trapani, D.; Gallo, G.; Laudicina, V.A.; Muscarella, S.M.; et al. Water Resource Recovery Facilities (WRRFs): The Case Study of Palermo University (Italy). Water 2021, 13, 3413. [Google Scholar] [CrossRef]
- De Campos Bernardi, A.C.; Anchão Oliviera, P.P.; de Melo Monte, M.B.; Souza-Barros, F. Brazilian Sedimentary Zeolite Use in Agriculture. Microporous Mesoporous Mater. 2013, 167, 16–21. [Google Scholar] [CrossRef]
- Saliu, T.D.; Oladoja, N.A. Nutrient Recovery from Wastewater and Reuse in Agriculture: A Review. Environ. Chem. Lett. 2021, 19, 2299–2316. [Google Scholar] [CrossRef]
- Canellas, J. Tertiary Ammonium Removal with Zeolites. Ph.D. Thesis, Cranfield University, Cranfield, UK, 2018. [Google Scholar]
- Koon, J.H.; Kaufman, W.J. Ammonia Removal from Municipal Wastewaters by Ion Exchange. J. Water Pollut. Control Fed. 1975, 47, 448–465. [Google Scholar]
- Castro, C.J.; Shyu, H.Y.; Xaba, L.; Bair, R.; Yeh, D.H. Performance and Onsite Regeneration of Natural Zeolite for Ammonium Removal in a Field-Scale Non-Sewered Sanitation System. Sci. Total Environ. 2021, 776, 145938. [Google Scholar] [CrossRef]
- Guo, X.; Zeng, L.; Li, X.; Park, H. Removal of Ammonium from RO Permeate of Anaerobically Digested Wastewater by Natural Zeolite. Sep. Sci. Technol. 2007, 42, 3169–3185. [Google Scholar] [CrossRef]
- Muscarella, S.M.; Laudicina, V.A.; Cano, B.; Badalucco, L.; Conte, P.; Mannina, G. Recovering Ammonium by Treated and Untreated Zeolitic Mixtures: A Comprehensive Experimental and Modelling Study. Microporous Mesoporous Mater. 2023, 349, 112434. [Google Scholar] [CrossRef]
- Mulvaney, R.L.; Sparks, D.L. Methods of Soil Analysis. Part 3. Chemical Methods; John Wiley & Sons: Hoboken, NJ, USA, 1996; pp. 1123–1184. [Google Scholar]
- Largitte, L.; Pasquier, R. A Review of the Kinetics Adsorption Models and Their Application to the Adsorption of Lead by an Activated Carbon. Chem. Eng. Res. Des. 2016, 109, 495–504. [Google Scholar] [CrossRef]
- Wijesinghe, D.T.N.; Dassanayake, K.B.; Sommer, S.G.; Jayasinghe, G.Y.; Scales, P.J.; Chen, D. Ammonium Removal from High-Strength Aqueous Solutions by Australian Zeolite. J. Environ. Sci. Health. A Tox. Hazard. Subst. Environ. Eng. 2016, 51, 614–625. [Google Scholar] [CrossRef]
- Byrappa, K.; Kumar, B.V.S. Characterization of Zeolites by Infrared Spectroscopy. Asian J. Chem. 2007, 19, 4933–4935. [Google Scholar]
- Rahmani, A.R.; Mahvi, A.H. Use of Ion Exchange for Removal of Ammonium: A Biological Regeneration of Zeolite. Pak. J. Biol. Sci. 2004, 8, 146–150. [Google Scholar]
- Deng, Q.; Dhar, B.R.; Elbeshbishy, E.; Lee, H.S. Ammonium Nitrogen Removal from the Permeates of Anaerobic Membrane Bioreactors: Economic Regeneration of Exhausted Zeolite. Environ. Technol. 2014, 35, 2008–2017. [Google Scholar] [CrossRef] [PubMed]
- Neag, E.; Șenilă, M.; Török, A.I.; Roman, M.; Puskás, F. Regeneration and reuse of natural zeolite for ammonium removal. In Proceedings of the International Multidisciplinary Scientific GeoConference: SGEM, Albena, Bulgaria, 30 June–6 July 2019; Volume 19, pp. 651–656. [Google Scholar]
- Conte, P. Effects of Ions on Water Structure: A Low-Field 1H T1 NMR Relaxometry Approach. Magn. Reson. Chem. 2015, 53, 711–718. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).