1. Introduction
The collection and analysis of soil solution are informative for quantifying the concentration and speciation of trace elements (TEs) to predict their behavior in soil, including their bioavailability, toxicity, mobility, and biogeochemical cycling [
1,
2,
3]. Measuring total TE concentrations in soils to interpret their behavior is not adequate, as the chemical form determines their behavior [
4]. The portion of TEs that exhibit the highest mobility in soils is found in the soil solution. This soil solution demonstrates significant diversity in concentration and speciation across different soils, owing to the variety of solid phases and the varying chemistry of the soil solution (e.g., pH, EC). Classes of TE species in soil solution include the following: hydrated ions, labile complexes with simple inorganic ligands (e.g., Zn(OH)
2, ZnSO
4 (aq), and ZnCO
3 (aq)), organic complexes and metal bearing organic compounds (e.g., humic substances and porphyrins), and suspended inorganic solids (e.g., hydroxides, oxides) [
5]. The bioavailability, toxicity, mobility, and biogeochemical cycling of these classes are not necessarily similar. The first step in understanding the actual concentration, speciation, and distribution of TEs in soil solutions is to measure the solution with minimum alteration. Soil solution sampling methods can be either ex situ or in situ. Every method collects soil solution containing slightly different classes and concentrations of TEs. Thus, the method should match the specific research question.
In ex situ methods or laboratory-based methods, soil is moved from the field to the laboratory before the extraction of soil solution. Several laboratory extraction techniques, such as column displacement, compression, and centrifugation, saturation extracts, and water extracts, can be used [
6,
7,
8,
9]. Selective extraction methods have been used to target specific chemical species in soil, or elements that are bound to or associated with particular soil phases or compounds. Thus, selective extraction methods can assess the amount of potentially mobile TEs. While they could respond as mobile species in the extraction, similar conditions are required in the soil for actual mobilization. There are several selective extraction methods which use different reagents to extract operationally defined geochemical fractions. However, this method depends on the reagents used. There are several criticisms of this extraction approach, such as the lack of selectivity, sorption, and redistribution of metals solubilized during the extraction, as well as changes during the sample pretreatment and storage [
10]. The specificity of many reagents can be increased by combining them in a sequential extraction scheme in which the residue from the first extraction is utilized as the material for the second extraction, and so on through several steps. It is challenging to ensure the representative and consistent partitioning and speciation of TEs in soils when applying extraction techniques [
9,
10]. Chemical extractants remove the entire reservoir of reactive metal which may contain several orders of magnitude of metals greater than that found in soil solution [
9]. The authors reported the approximate percentages and mobility/availability of metals extracted using different techniques (column leaching–mobile phase, 5%; single extraction–plant available metal content, 10%; sequential extraction–exchangeable, acid, and water soluble, 20%). Another major limitation in extraction techniques is that they alter the partitioning of TEs in soil solution, making it difficult to determine the actual in situ speciation [
8]. Sequential extraction and column leaching tests in the laboratory were compared to element concentrations in pore water directly sampled from the field using rhizon pore water samplers [
11]. The results indicated that sequential extraction would be more rigorous as it extracts higher concentrations of TEs. Different factors such as soil-to-solution ratio, duration of extraction, solutes used, and both shaking and centrifugation methods employed determined the partitioning of TEs. Column leaching tests extracted larger amounts of Cd compared to the pore water. Water flows through the pathways of least resistance in the soil. Thus, the homogenization of soil prior to extraction in the laboratory could change extracted TE concentrations. Raudina et al. conducted a study to compare soil solution sampling methods: squeezing/pressing and vacuum filtration with porous ceramic cups, to select the optimal method for soils in frozen swamps. Significantly higher concentrations of Ca and Mg were obtained in the soil solutions collected using the vacuum method. When soils are exposed to pressure, the natural equilibrium in the gas–solid–liquid phase can be disrupted. Consequently, the natural composition and properties of the collected soil solution can be changed [
12]. Centrifugation can also compact and reduce the pore size which can have a significant effect on soils with a fine texture. Also, during the centrifugation process, water moves down through the sample resulting in the saturation of the sample at its base [
6]. This could homogenize the chemical composition of the sample compared to the heterogeneous field conditions. Centrifuged soil solutions may have had a longer time of contact with the soil matrix and with the roots as they may have originated from smaller pores compared to lysimetric soil solutions. Frequently, greater concentrations and strong seasonal changes in properties are found In centrifuged solutions compared to lysimeter solutions [
13,
14].
In in situ or field-based methods, the soil solution is collected on-site and subsequently analyzed in the laboratory. Compared to ex situ methods, these methods are less destructive and allow for repeated sampling at the same location [
15]. Ex situ methods disturb the natural soil structure or composition during the sampling process. Thus, the collected sample may not accurately represent the true composition and distribution of soil components at the sampling site. In situ sampling methods are grouped under the general term “lysimetry”, which comprises a range of sampler types: monolith, filled-in, tension, passive, and ebermayer [
16]. Porous cups, porous plates, capillary wicks, resin boxes, and lysimeters are widely used methods for in situ soil solution sampling [
17]. Lysimeters which are made from different materials have been used to better understand the bioavailability and bioaccessibility of TEs through the analysis of different forms of TEs in soil solution samples collected using lysimeters. Lysimeter sampling has been combined with AF4-ICPMS to measure the distribution of TEs among major colloidal forms in soil solution [
18].
Different types of lysimeters have been used in both agricultural and natural settings to collect soil solution with or without applying tension. These tension and zero-tension lysimeters collect water from different compartments, which have differing mobility through different pathways. Zero-tension lysimeters only collect saturated flow or macropore flow which drain through the soil under the force of gravity. Tension lysimeters collect both the freely draining water and the capillary water which makes up the primary source of water for plants, and it is the most bioaccessible compartment for the uptake of TEs [
19]. In general, relatively mobile water pertains to the quantity of water in the soil between saturation and field capacity (also known as gravitational water). This water is attributed to the recent infiltration from the most recent precipitation or irrigation event. The relatively immobile pool of soil water can be characterized as the water existing in the soil before the latest precipitation or irrigation event. This water is held within the pore spaces of the soil matrix through capillary action, making it less mobile compared to water from recent events. This pool of water contributes to the flux of TEs into plants [
20,
21,
22]. Landon et al. [
23] observed different stable isotopic compositions in soil solutions collected from a tension lysimeter, a wick sampler, and soil cores, and explained that each method collected water from a different pool in the total soil–water reservoir. The results showed that wick samplers collected the relatively mobile soil water pool, but the tension lysimeters collected both relatively more and less mobile water.
There are several challenges associated with using lysimeters to collect representative soil solution samples for TE analysis. Due to the low concentrations of TEs in soil solutions and small sample volume, the sampling process is highly susceptible to errors. One of the major challenges is the sorption and release of TEs by lysimeter materials, which could, respectively, underestimate or overestimate the actual concentration of TEs [
24,
25]. Sorption and release depend on the lysimeter material and the chemistry of the soil solution. A significant sorption of TEs was observed using conventional aluminum oxide and ceramic suction cups [
26]. Under some conditions, contaminants may also be released into the collected sample from the lysimeter [
27]. Stainless steel lysimeter samplers were reliable for collecting soil solution samples for the analysis of most TEs due to their low adsorption capacity and desorption characteristics [
18]. The inertness of lysimeter material and its ability to provide good contact between the lysimeter and the soil improve the reliability of sampling. McGuire et al. [
28] reported that the adsorption of TEs onto silica components of the lysimeter was directly related to the solution pH. Hence, determining the best lysimeter type to use for collecting a given soil solution to measure TEs is not an easy task. Due to the varying performance of lysimeter materials with different soil solution chemistry and TEs, it is important to assess the suitability of any given lysimeter type and material for collecting soil solution to measure the particular TE concentrations of interest.
There is thus no universally accepted lysimeter or standard sampling protocol to collect soil solution for the analysis of TEs. Each of the abovementioned methods has advantages and disadvantages. Therefore, optimal lysimeter selection depends upon the research question for a particular experiment, their associated advantages and disadvantages, and the available resources including time.
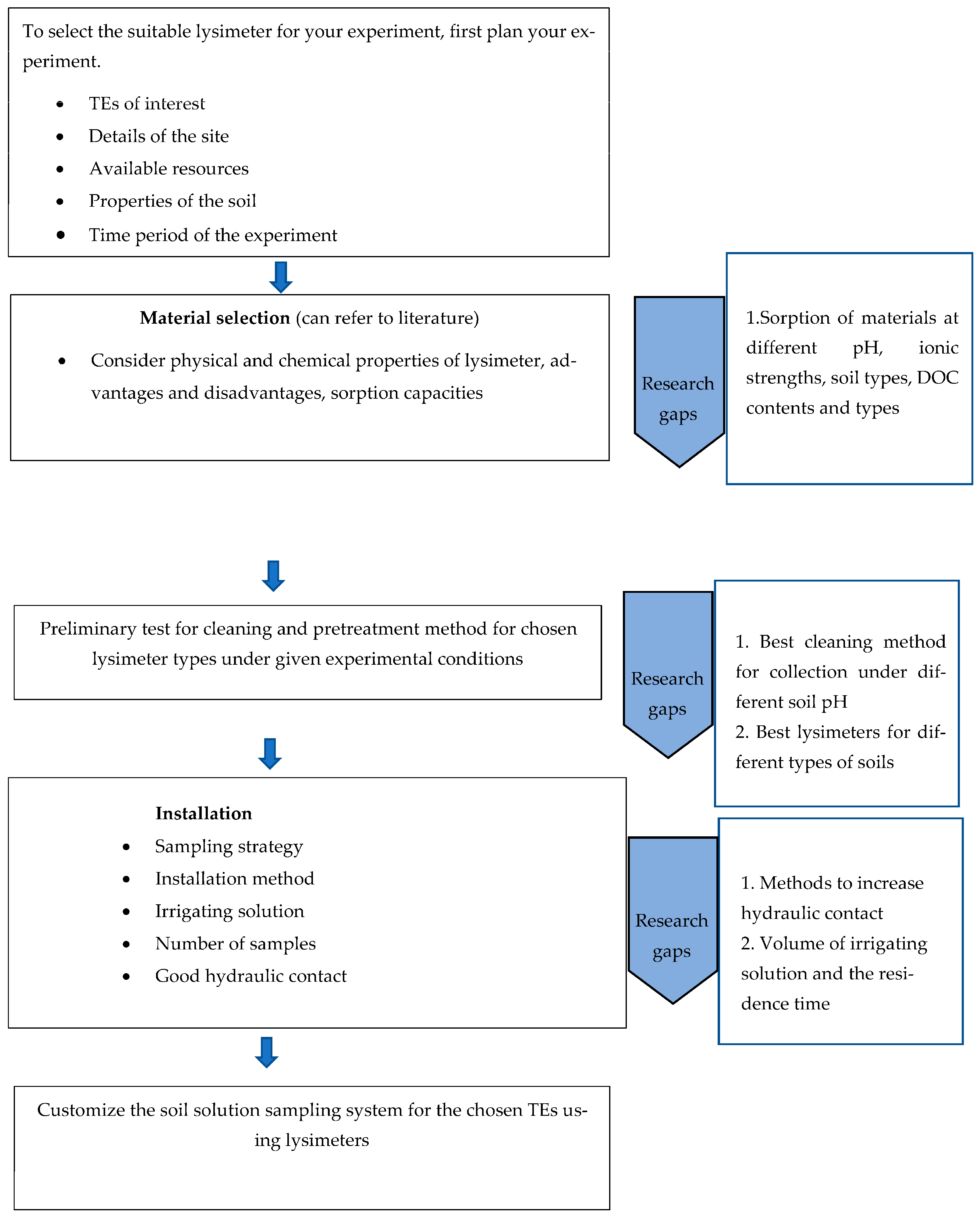
Overall, the reliable collection and analysis of soil solution that reflects natural conditions is extremely important for understanding the bioavailability, toxicity, mobility, pollution risk, and biogeochemical cycling of TEs. Multiple analytical techniques can be used to determine TE speciation, concentration, and distribution amongst different colloidal forms. To obtain accurate results, the collected soil solution should closely resemble the natural soil solution. Therefore, the sampling of soil solution should be carried out in a way that minimally alters the natural condition of the soil. Lysimeters are capable of minimally impacting soil solution, and have been widely used in hydrological, agricultural, and environmental research. This review provides an overview of the key variables that should be measured to ensure that the impact of sample collection is minimized, including the following: different lysimeter designs and materials and their associated advantages and disadvantages, limitations in using lysimeters for TE studies, cleaning and preconditioning methods, the effect of soil properties, and the details associated with lysimeter selection and installation. This provides a thorough background and guidelines to support researchers in planning their approach to collect soil solutions for TE analysis.
3. Sorption and Release of TEs from Lysimeters
Sorption and release are identified as two of the major sources of sample bias in soil solution sampling for nutrient analysis [
38]. The impacts of sorption and release may be considerable for solutes with a lower concentration, making them major limitations for the collection of samples for the analysis of TEs.
The sorption effect of a lysimeter can be affected by various factors such as the sorption capacity of the suction cup material, the composition and pH of the soil solution, and the sampling rate, as well as the volume of sample collected [
24]. Depending on the lysimeter material, the sorption of TEs may differ. Wenzel et al. [
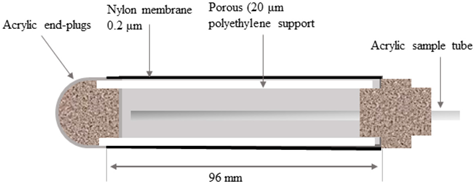
37] compared the adsorption of As, Cd, Co, Cr, Cu, Mn, Ni, Pb, and Zn on lysimeters made from a nylon membrane, and from a porous ceramic cup. The As, Cr, Cu, and Pb were completely removed from the soil solution when using the ceramic cup. Other TEs were also significantly adsorbed by ceramic lysimeters compared to the nylon membrane lysimeters, in a pH range from 4 to 5.
The TE sorption properties of ceramic, aluminum oxide, and plastic suction cups were assessed by [
42] who found that sorption was highly dependent on the TE and pH. There was no notable retention of TEs at pH 4, except for Pb on the aluminum oxide and ceramic cups. However, when the pH values were around 8, Cd and Zn also showed strong sorption in the aluminum oxide and ceramic cups. Conversely, Be, Cu, and Pb exhibited significant sorption across all cup types at this pH.
New lysimeters generally release different TEs into the sample due to contaminants left over from the production process. Also, TEs may be desorbed from the surfaces of the lysimeters or through contact with the soil solution. Any TEs that were previously adsorbed or bound to lysimeter surfaces may thus be released back into the solution during sampling. Any TEs that have penetrated into the lysimeter cup or other components over time may also be leached out during the sampling process. Some impurities in the lysimeter cup or other materials used in its fabrication may also be soluble in the soil solution. If these materials contain TEs or have absorbed TEs from previous experiments, they can release TEs into the newly collected soil solution. Precautions can be taken to minimize the potential release of TEs, such as rinsing or cleaning to remove any loosely bound or soluble TEs. Additionally, using inert and high-purity materials for lysimeter construction can help to reduce the chance of contamination.
Sorption behavior depends on the element. Wenzel et al. [
37] tested the adsorption properties of nylon and ceramic lysimeters by sequentially pumping test soil solutions with low and high metal concentrations. They reported three different patterns of change in solute concentrations in ceramic lysimeters: (1) TEs completely removed from soil solution during the pumping of low-concentration test solution, but with a concentration of the effluent close to that of the test solution at a high concentration (Co, Ni, and Mn); (2) TEs having a similar pattern to the group, but with more metals adsorbed during the pumping of a high-concentration solution (Cd, Zn); and (3) oxyanions and metal cations which were almost completely removed from the solution (As(V), Cr (III), Cu, and Pb). This grouping was consistent with their environmental behavior: Group 1 and 2 elements are among the most mobile TEs, and their adsorption to oxide surfaces is less than that of Group 3 elements. McGuire et al. [
28] also evaluated the adsorption of TEs on ceramic, PTFE, fritted glass, and stainless steel lysimeters as a function of TE concentration at pH 6 to 8. They observed greater adsorption of TEs in the highly concentrated solution compared to the less concentrated solution where the pattern of TE adsorption on lysimeter material was Zn >> Co > Cr > Cd.
3.1. Effect of Dissolved Organic Carbon (DOC) on Adsorption
Dissolved organic carbon (DOC) could be a reason for this adsorption of TEs by the lysimeter materials, due to the complexation of TEs to humic acids. The DOC may influence the adsorption of TEs by lysimeter materials in two ways: (1) DOC can form a complex with TEs, preventing retention if the complexes are not sorbed to the lysimeter, and (2) the sorption of DOC by the lysimeter material may increase the sorption of TEs by creating more cation exchange sites [
25]. The DOC can be leached from lysimeter materials if they are not inert. The DOC in soils can increase or decrease the sorption of TEs to lysimeter cups and other components of the sampling pathway. If DOC-TE complexes are not sorbed, then TEs will not sorb to cups, and previously sorbed TEs may form a complex with DOC and will be released in the soil solution. Rais et al. [
25] reported that 67% of Cu, 4% of Zn and Cd, and 52% of Pb were predicted to be present as complexes with fulvic acids in an acidic synthetic soil solution containing DOC, at pH 4.5. The sorption of most TEs was reduced due to the formation of soluble complexes with DOC. The DOC adsorbed on the surface of cups may increase the CEC of the cup, ultimately increasing the adsorption of TEs. In most cases, the sorption of TEs was reduced by DOC; however, in some cases, the opposite effect was observed for ceramic materials. That said, the impact of DOC also depends on the type of DOC.
3.2. Effect of pH on Adsorption
The pH of soil solution affects the adsorption of TEs to lysimeters. Under certain pH conditions, TEs may undergo redox reactions, leading to changes in their oxidation state. The oxidation state of TEs can affect their solubility. Thus, pH variations can influence the speciation of TEs, altering their chemical forms and subsequent solubility. For example, at acidic pH, some TEs may become more soluble, increasing their mobility and potentially reducing their sorption into lysimeter materials. The oxidation state and associated speciation of TEs can exhibit different affinities for different ligands. For instance, certain TEs may form stronger complexes with ligands under specific pH conditions, reducing their capability for sorption into lysimeter cup materials. The changes in oxidation state, mineralization, complex formation, and solubility of TEs ultimately influence their sorption into lysimeter materials. At low pH levels, the competition between protons and TEs for binding sites on the cup materials is also important due to the limiting of TE sorption. Wenzel et al. [
37] tested the effect of pH on TE adsorption using three replicates of lysimeters with standard solutions adjusted to pH 4, 5, and 6. Increasing pH from 4 to 5 did not significantly affect TE adsorption by nylon lysimeters; however, increasing pH from 5 to 6 significantly affected the concentration of soluble As, Cr, Cu, and Zn. The adsorption of Co, Mn, and Ni using ceramic lysimeters was significantly different at pH 5 and 6. For ceramic lysimeters, little difference in adsorption was observed at pH 4 and 5 [
24].
Grossmann and Udluft [
26] tested the effect of pH on TE sorption with nylon, ceramic, and aluminum oxide (Al
2O
3) lysimeters using a test solution with different ion concentrations. No sorption of Ni and Mn was observed at pH 4 or 8. At pH 4, 100% of the Cd, Cu, Pb, and Zn was recovered in the outflow from the Al
2O
3 and nylon lysimeters, while at pH 8, less than 10% of these elements was recovered from the Al
2O
3 and ceramic lysimeters. More than 80% of these elements were recovered from nylon lysimeters at pH 8, except for 52% recovery of Pb.
It is not easy to group suitable lysimeter materials for various TEs according to pH. Differences in experiment conditions, cleaning methods, and preconditioning contribute to variable results. However, a general scheme for selecting the optimal lysimeter material for particular TEs in soil solution at different pHs is summarized in
Table 2.
3.3. Effect of Ionic Strength on Adsorption
The ionic strength of the soil solution (measured as electrical conductivity) changes the adsorption of TEs via lysimeters. Andersen et al. [
24] conducted an experiment using no electrolyte, 0.01 M CaCl
2, and 0.01 M NaCl solutions. However, the ionic strengths of these solutions were higher than the general ionic strengths in soil solutions. They observed a significant decrease in the adsorption of Cd, Ni, and Zn at higher concentrations of Ca and Na. This was explained by at least some of the adsorption being due to ion exchange reactions or competition at specific adsorption sites on the lysimeter material. At higher concentrations of Ca and Na, there is an increased likelihood of ion exchange reactions and increased competition for adsorption sites on the lysimeter material, reducing the capability of TE sorption. This competition effect can contribute to the decreased adsorption observed for Cd, Ni, and Zn. However, this effect was non-significant for Cu, indicating that Cu ions were more strongly adsorbed on the lysimeter surface. This suggests that the sorption of Cu onto the lysimeter material may be less influenced by the presence of other ions or ion exchange reactions compared to Cd, Ni, and Zn.
Table 3 provides some physical and physiochemical characteristics of the tension lysimeters with their cross-sectional views.
4. Methods of Lysimeter Sampling
After determining the best lysimeter type, many factors should also be considered in the process of installation. Firstly, disturbance to the soil profile should be minimized. Generally, it is recommended to install the lysimeters one year prior to sampling to facilitate equilibration with the soil solution [
17]. The ions present in the soil solution interact with the surface of the lysimeter, leading to the exchange of ions between the lysimeter material and the soil solution. This process helps to establish equilibrium between the lysimeter and the soil solution, ensuring that the measured soil solution represents the surrounding soil conditions. Over time, the movement of water within the soil profile affects the distribution of solutes. Installing lysimeters one year before sampling allows sufficient time for water to percolate through the soil layers, carrying solutes and facilitating their redistribution. This helps in achieving a more representative sampling of the soil solution as the water reaches equilibrium with the soil matrix. The physical properties of the soil, such as soil texture, structure, and porosity, can also undergo equilibration with the lysimeter. Over time, the surrounding soil structure may adjust, leading to changes in water flow and solute transport. Allowing for an extended period of equilibration ensures that the lysimeters capture these changes and provide a representative measurement of the soil solution. Overall, the equilibration process in lysimeters involves multiple aspects, including ion exchange, water movement, microbial activity, and soil structural dynamics. Allowing sufficient time for these processes to occur ensures that lysimeters accurately represent the soil conditions and provide reliable measurements related to the soil solution.
Before installation, each lysimeter should be tested for the conductance of distilled water under a constant vacuum, and the bubbling pressure of each lysimeter should be tested to determine the range of functionality. It is recommended to soak lysimeters in distilled water before installation to completely fill the lysimeter walls with water [
44]. Specifically with regard to the soil solution sampling for TE analysis, the risk of contamination should be minimized, including the sorption of solutes by the lysimeter. For this purpose, the proper cleaning of lysimeters is very important. Lysimeters should be rinsed with dilute acids, followed by washing with ultra-pure water to remove the contaminants and reduce the sorption. Cleaning methods are further discussed in
Section 4.2.
To install tension lysimeters, an auger hole is generally drilled to the desired depth with a diameter slightly larger than the suction cup. Care should be taken to ensure that soils from the upper horizons do not fall into the hole. After inserting the lysimeter, it is essential to gradually backfill the surrounding space with local soil and tamp it after each backfilling step. This process enhances contact between the soil and the cup. An alternative approach involves filling the hole with a slurry before installing the cup, in order to obtain good hydraulic contact between the soil and the cup (see
Section 4.1). After installing, the remainder of the core is backfilled with the local soil [
7]. Pore spaces that allow the seeping of water down through the space between the lysimeter and the surrounding soil should be prevented to avoid hydraulic short circuits.
It is important to minimize the risk of TE contamination during sampling. Milli Q water (≥18.2 MΩ cm at 25 °C, EMD Millipore, Bedford, Massachusetts, Darmstadt, Germany) was used by Du et al. [
18] to irrigate the soil to achieve the desired moisture level for extracting the soil solution. After installation, the lysimeter surface should be stabilized by conditioning with soil solution, for example by collecting and discarding excess solution (see
Section 4.3).
Installation of suction cup lysimeters is straight forward compared to other sampling systems. Abdulkareem et al. [
45] reported four possible installation methods for lysimeters: (1) horizontal, (2) vertical without shaft, (3) vertical with shaft, and (4) vertical at 45° (
Figure 2). Similar installation methods were described by [
26]. For a horizontal installation, it is also advantageous to install the lysimeter at a slight angle to provide a route for water to percolate away from the lysimeter cup.
Even though the solution chemistry results of lysimeters are generally consistent and reliable, there are other challenges during installation. One major problem is mechanical damage of the suction cup during installation and due to frost-heave in the soil. The mechanical resistance of the suction cup of a lysimeter is thus very important. Nylon cups mantled with two layers of nylon membranes were more mechanically stable and resistant compared to cups mantled with single layers [
37].
4.1. Hydraulic Contact
Good contact between the lysimeter cup and soil is necessary for the continuous movement of soil pore water into the cup. Usually, good hydraulic contact is achieved by backfilling the hole around the sampler with the soil that was removed, as mentioned in the previous section. Soil removed from the augured hole to pour around the lysimeter cup is sieved to remove coarse sand and gravel particles. Large void spaces that can reduce the contact area of the cup can be eliminated using this method [
44].
Silica flour or very fine silica sand can also be used to make a silica slurry which is poured into the open auger hole [
28]. The sampler is then pressed firmly into the slurry. Using silica as the backfilling material for TE analysis should be carefully considered, since silica can absorb TEs such as Fe
3+, Co
2+, and Cr
3+ [
46]. McGuire et al. [
28] reported that the adsorption of TEs on silica highly depends on solution pH with significant adsorption at a pH of 8, reporting that the order of sorption was Zn >> Co > Cr > Cd.
After installing the lysimeters, the auger hole should be refilled carefully with continuous tamping, to minimize the preferential flow of surface water. Creating mounds of soil around the exposed sampling tube will minimize this direct channeling. In some ceramic samplers, a plug of bentonite sealer is installed in the hole to eliminate this potential source of error [
44].
4.2. Cleaning of Lysimeters
Cleaning with acids reduces sample contamination by inorganic constituents. The flushing and rinsing of lysimeters with dilute acid and distilled water can reduce the release of TEs from lysimeters [
33,
34]. Fresh lysimeters which are used to sample soil solution for the first time may underestimate the concentration of some TEs due to sorption on or within the cup. Saturating the CEC would minimize the sorption of TEs. Flushing with dilute acid would quickly saturate the exchange sites with H
+ ions; however, rinsing the cups with distilled water afterward would not remove the H
+ ions from exchange sites as there would be no other cations to replace them. Rinsing with distilled water would also remove the residual acid and other contaminants. Porous ceramic cups were washed with 250–500 mL of 0.1 N HCl followed by deionized water to remove contaminants deposited in the cup during manufacturing. Calcium, sodium, and magnesium were removed from the cup via this cleaning procedure [
44].
Du et al. [
18] tested three cleaning methods for minimizing the background concentrations of TE in lysimeters constructed from surgical (316 L) stainless steel. The three methods were the following: (1) lysimeters soaked in a 10% reagent-grade HNO
3 acid bath for 2 weeks and rinsed with ASTM type 1 water; (2) soaked in a 10% reagent-grade HNO
3 acid bath for a week, rinsed with Milli-Q water (MQW), and soaked in an MQW bath for 7 days; and (3) lysimeters soaked in a 10% reagent-grade HNO
3 acid bath for a week and rinsed with MQW. Cleaning method 3 was sufficient to clean the lysimeters to work with most TEs.
Two cleaning methods were compared for ceramic, PTFE, and fritted-glass lysimeters: (1) rinse with 1 M HCl followed by deionized water and (2) rinse with distilled, deionized water. Stainless steel lysimeters were also cleaned with 3 M HNO
3 followed by deionized water. Acid-washed lysimeters were rinsed with deionized water until the pH of the discharge solution was similar to the input water. The results showed that all of the lysimeter types cleaned with water had a greater adsorption of TEs compared to the acid-cleaned lysimeters. Wenzel et al. [
37] rinsed nylon lysimeters with distilled water only, as preliminary tests indicated that no TEs were released at detectable levels.
Table 4 summarizes different cleaning methods for lysimeters.
4.3. Conditioning the Lysimeter
The sorption of TEs onto a cleaned lysimeter is considerable during initial contact with soil solution. Lysimeters should thus be conditioned to avoid bias, via equilibration with a solute having a more or less similar composition to the soil solution. Conditioning involves rinsing the lysimeter with a solution similar to the soil solution which will be collected. The quantity of solution required depends on several factors, including the pH, the CEC of the cup, and the concentration of TE to be investigated [
26]. For instance, for metals with concentrations below 10
−5 mol/L and pHs above 6, several liters of solution were required for the adequate conditioning of cadmium (≈1 L), nickel (3 L), and aluminum, copper, lead, and zinc (>3 L). Discarding the first and second samples after field installation is another way to condition the surfaces of the lysimeter and sampling pathway.
4.4. Sampling Strategies
The sampling volume should be as small as possible to minimize disturbance to the soil system and corresponding deviations from natural conditions. This disturbance can have a marked effect on soil solution chemistry, for instance by stimulating nitrification. This can ultimately change the speciation of TEs in soil solution. The period of sampling should also be short as the soil solution is not sterile and microbial activity may alter the concentration and speciation of TEs. This may occur indirectly, for example through changes in the redox potential and pH. The elevation of pH by one unit (a 10-fold difference in H
3O
+ concentration) due to microbial activity was observed [
26].
In general, sampling with tension lysimeters occurs either in continuous or discontinuous modes. For continuous operation, a pressure gradient is applied, which depends on the actual potential in the undisturbed soil as measured using a tensiometer. The advantages of continuous sampling include continuous collection of pore water, an accurate assessment of the water drainage front, and a low withdrawal rate; thus, a minimal change in the natural water flow pattern is achieved [
26,
28]. A low withdrawal rate of water from the soil and minimizing the disturbance to the natural water flow pattern help to maintain natural hydrological conditions, allowing for more measurements of the natural soil solution composition.
The disadvantages of continuous sampling include potentially creating a preferential flow path to the cup, which can bypass certain areas of the soil and result in non-representative samples. The cup shape may also become deformed, and the area of soil that is bypassed cannot be defined. Discontinuous sample collection operates over several short-term intervals. This operation mode can be used to collect solutes at specific time intervals if the solution is removed between sampling times. A more temporary disturbance to the natural flow system and low required time for sustained suction are advantages of this mode. However, nonpermanent flow through the cup material can result in high sorption. Therefore, it is recommended to discharge the first part of each sample that is collected. Short-term sampling intervals during periods of rapidly changing solute concentrations can also be recorded (e.g., after heavy rainfall).
Grossmann and Udluft [
26] define the following three strategies: (1) discontinuously episodically; (2) discontinuously regularly; and (3) continuously. Episodic sampling is undertaken according to the frequency of a certain season or event. Regular sampling is conducted periodically at long-term intervals to determine changes in concentration. Continuous sampling is used to determine short-term differences in concentration and speciation.
6. Summary and Outlook
There is currently no agreement as to the best lysimeter sampling system for collecting soil solution for TE analysis under most soil conditions. Different types and materials of lysimeters have their own strengths and weaknesses with regard to representing the inherent concentrations and speciation of TEs in soil solution. In spite of having both strengths and weaknesses, the majority of research has used tension lysimeters to collect soil solution for TE analysis, likely because the pool of soil solution which is collected via tension lysimeters provides a better idea of bioavailable TE concentration due to the inclusion of capillary water. The selection of lysimeter material has important implications for the sorption and release of TEs. However, sorption behavior may differ according to the chemical properties of the soil solution, such as DOC, pH, and ionic strength. Therefore, the optimal design and material for lysimeters are heavily reliant on the type of experiment, research questions, and available resources. Appropriate cleaning and pretreatment procedures are also required to obtain accurate results. Although this review has outlined various cleaning and pretreatment approaches, it remains necessary to conduct prior research to test the best cleaning and pretreatment methods for each lysimeter type under specific experimental conditions. Additionally, accurate installation is also required for optimal lysimeter performance. This involves maintaining effective hydraulic contact and irrigating the soil using a solution with minimum contamination, as well as properties similar to the soil solution.
Developing a predictive model to understand the sorption and release patterns of trace elements (TEs) on different lysimeter materials under varying pHs, ionic strengths, soil types, and dissolved organic carbon (DOC) contents and types holds great promise for future research endeavors. Such a model could offer a systematic and cost-effective approach to explore complex interactions between TEs and lysimeter materials in different environmental scenarios. However, developing such a model would require a substantial amount of high-quality experimental data encompassing a wide range of environmental conditions and lysimeter materials. Rigorous validation of the model using independent datasets would also be necessary to ensure its reliability and applicability across various scenarios. The geochemical behavior of TEs is highly related to their colloidal behavior, and further investigation of the behavior of colloids in the sampling system will contribute to the development of more reliable sampling protocols, enabling accurate assessments of TEs’ mobility, bioavailability, and potential environmental risks. Exploring the volume of irrigating solution and the residence time is another future aspect, contributing to the advancement of knowledge in the field.