The Role of Biocomposites and Nanocomposites in Eliminating Organic Contaminants from Effluents
Abstract
:1. Introduction
2. Different Pollutants in Water
2.1. Dyes
2.2. Drugs, Pesticides and Insecticides
2.3. Other Organic Pollutants
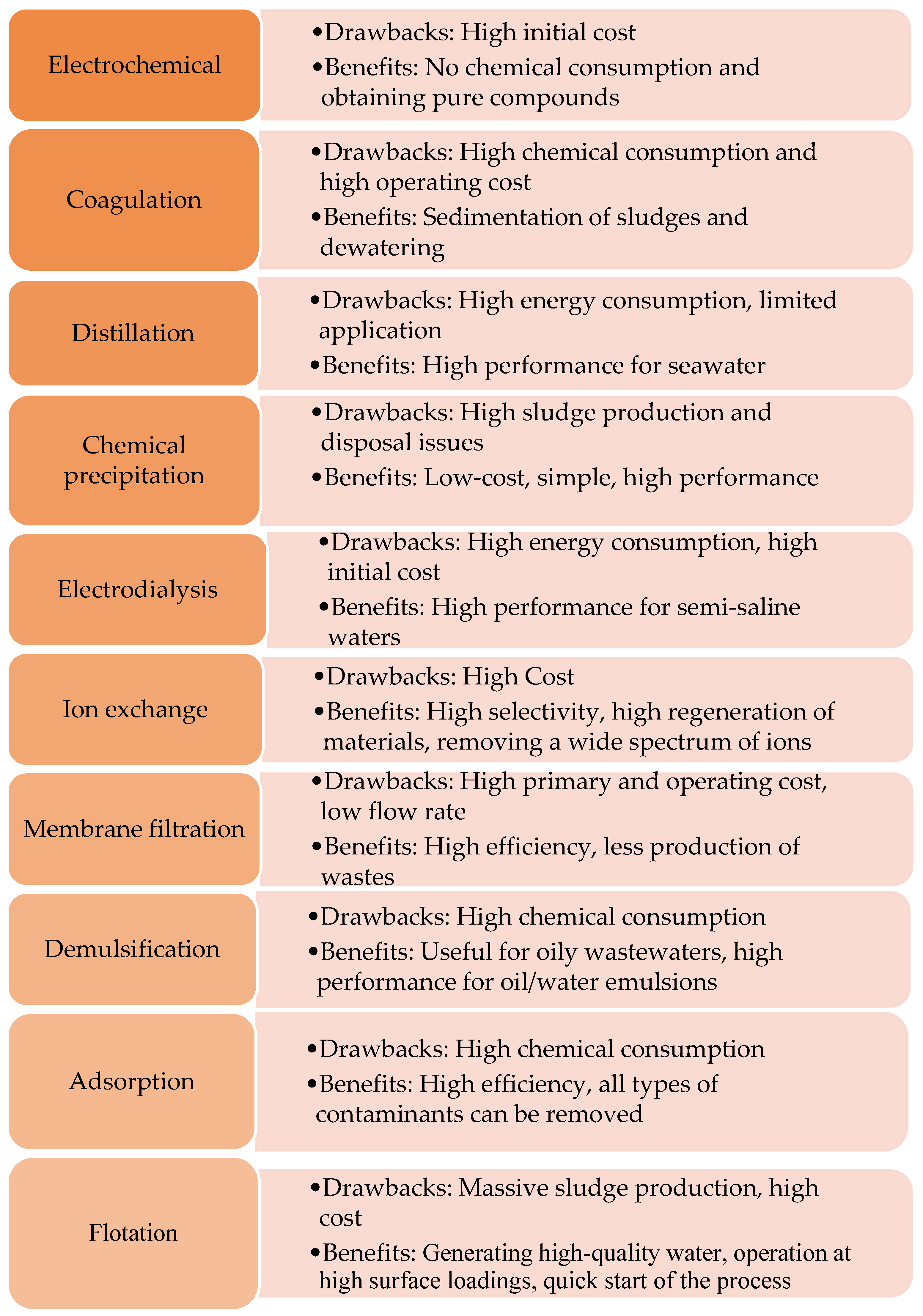
3. Wastewater Treatment Approaches
4. Sorption Technique
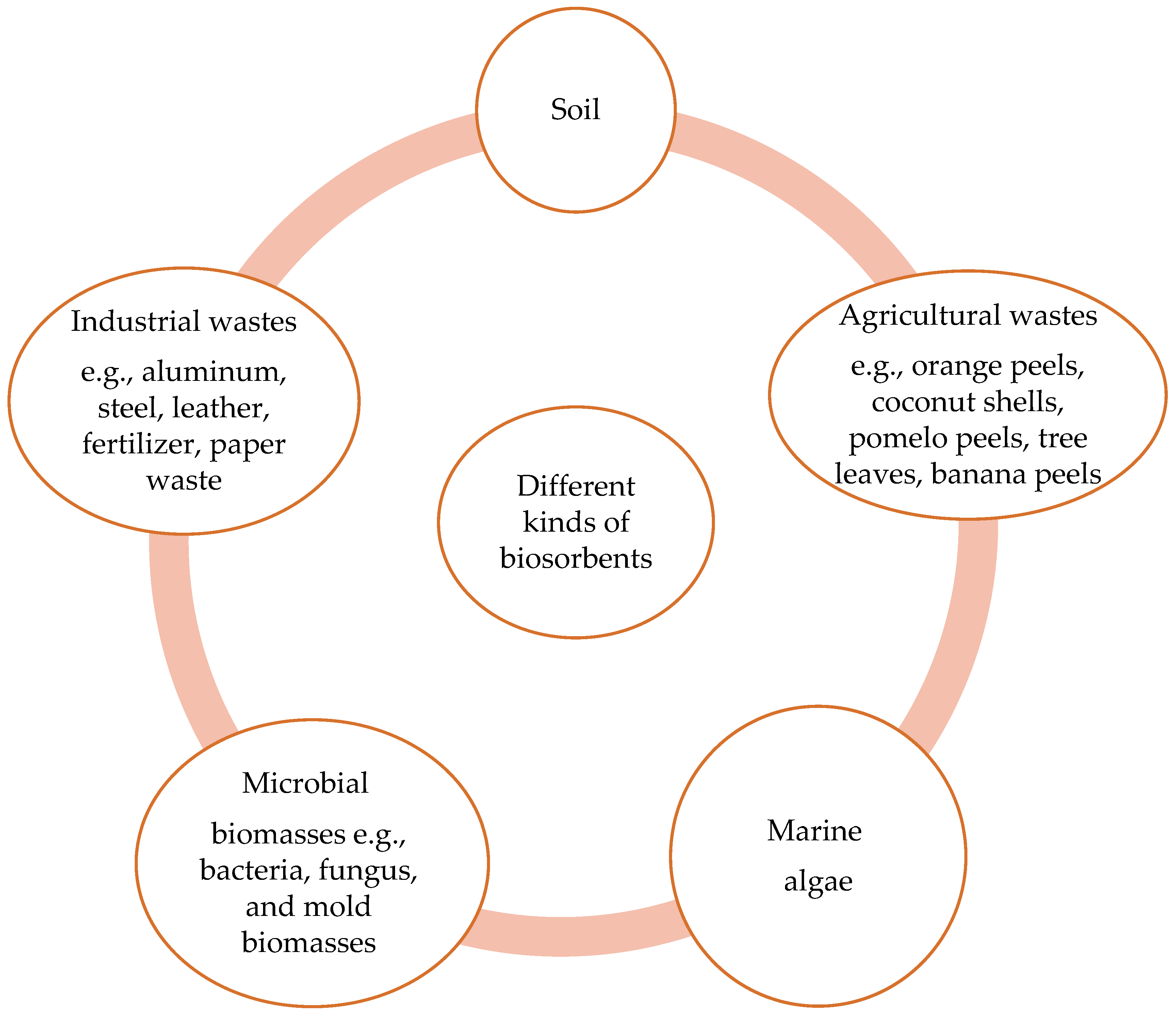
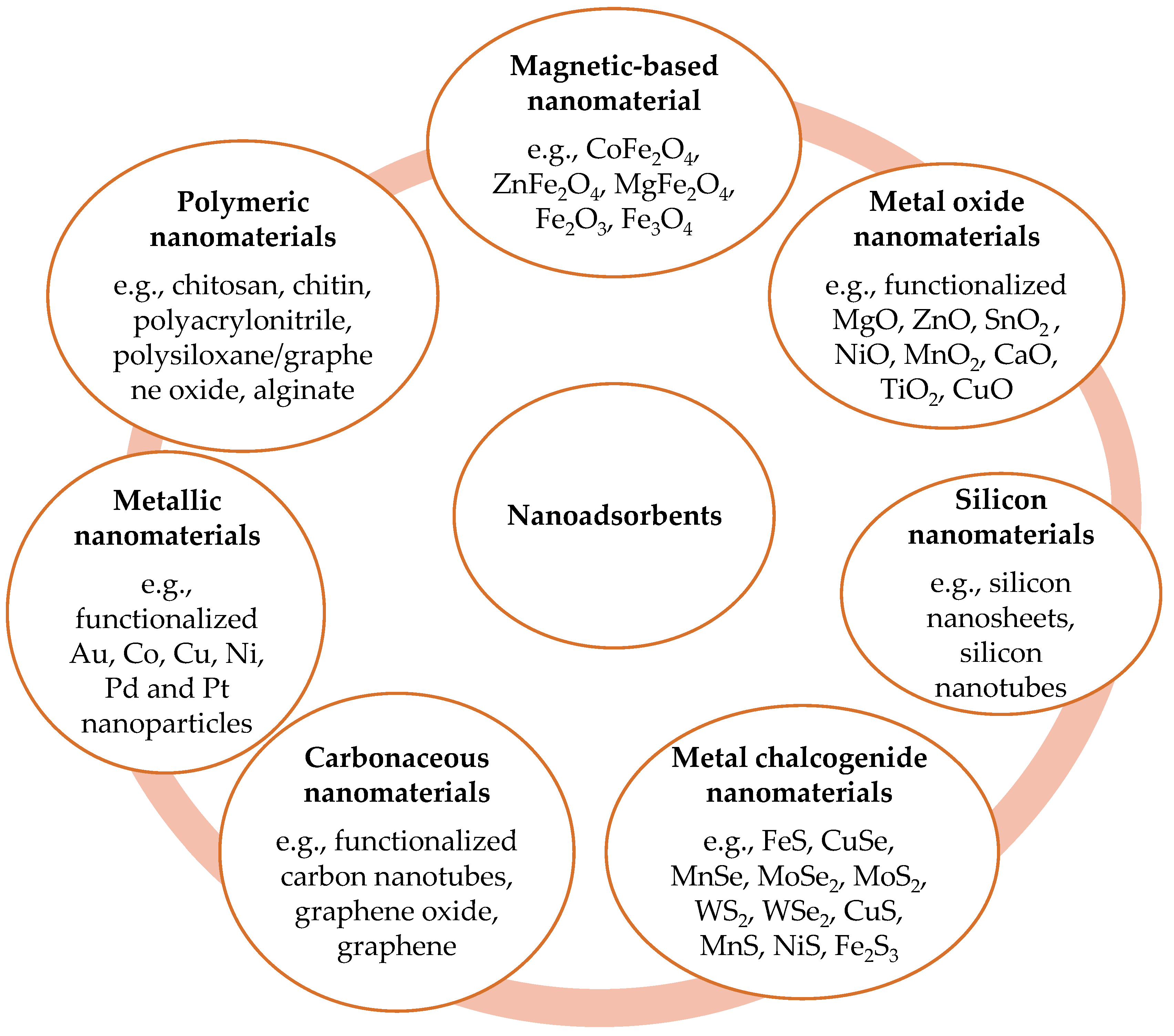
5. Different Kinds of Sorbents
5.1. Biosorbents
5.2. Nanocomposites
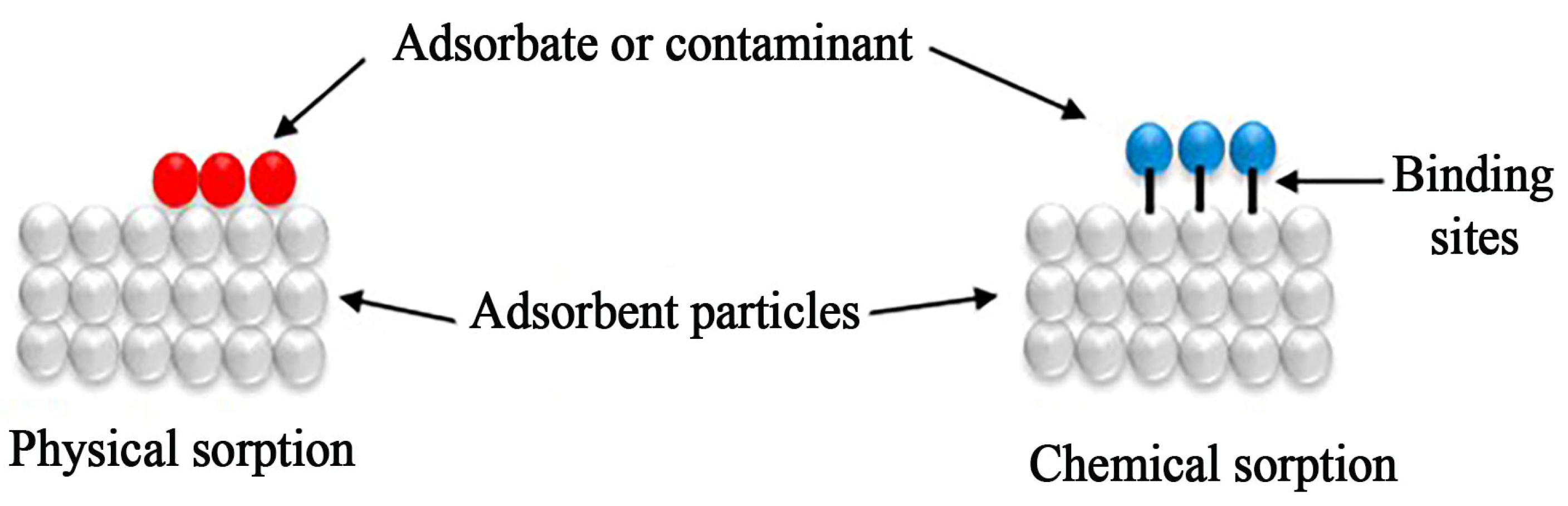
6. Sorption Mechanism
7. Effective Variables on Sorption
8. Reusability
9. Sorption Kinetics
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, Y.; Chen, Y. Impact of population growth and land-use change on water resources and ecosystems of the arid Tarim River Basin in Western China. Int. J. Sustain. Dev. World Ecol. 2006, 13, 295–305. [Google Scholar] [CrossRef]
- Dilpazeer, F.; Munir, M.; Baloch, M.Y.J.; Shafiq, I.; Iqbal, J.; Saeed, M.; Abbas, M.M.; Shafique, S.; Aziz, K.H.H.; Mustafa, A.; et al. A Comprehensive Review of the Latest Advancements in Controlling Arsenic Contaminants in Groundwater. Water 2023, 15, 478. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and non-conventional adsorbents for wastewater treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Esmaeili, H.; Tamjidi, S. Ultrasonic-assisted synthesis of natural clay/Fe3O4/graphene oxide for enhance removal of Cr (VI) from aqueous media. Environ. Sci. Pollut. Res. 2020, 27, 31652–31664. [Google Scholar] [CrossRef]
- Santhosh, C.; Velmurugan, V.; Jacob, G.; Jeong, S.K.; Grace, A.N.; Bhatnagar, A. Role of nanomaterials in water treatment applications: A review. Chem. Eng. J. 2016, 306, 1116–1137. [Google Scholar] [CrossRef]
- Liang, H.; Esmaeili, H. Application of nanomaterials for demulsification of oily wastewater: A review study. Environ. Technol. Innov. 2021, 22, 101498. [Google Scholar] [CrossRef]
- Tamjidi, S.; Ameri, A. A review of the application of sea material shells as low cost and effective bio-adsorbent for removal of heavy metals from wastewater. Environ. Sci. Pollut. Res. 2020, 27, 31105–31119. [Google Scholar] [CrossRef]
- Shreya; Verma, A.K.; Dash, A.K.; Bhunia, P.; Dash, R.R. Removal of surfactants in greywater using low-cost natural adsorbents: A review. Surf. Interfaces 2021, 27, 101532. [Google Scholar] [CrossRef]
- Suresh, S. Removal of Basic Violet 14 from aqueous solution using sulphuric acid activated materials. Springerplus 2016, 5, 633. [Google Scholar] [CrossRef]
- Hussain, S.; Anjali, K.P.; Hassan, S.T.; Dwivedi, P.B. Waste tea as a novel adsorbent: A review. Appl. Water Sci. 2018, 8, 165. [Google Scholar] [CrossRef]
- Erbaş, O. Performance Analysis of Combined Cycle Power Plant with Multi-Effect Distillation (MED) Desalination Process. Bilecik Şeyh Edebali ÜNiversitesi Fen Bilim. Derg. 2019, 6, 85–90. [Google Scholar] [CrossRef]
- Li, X.; Liu, J.; Zheng, C.; Han, G.; Hoff, H. Energy for water utilization in China and policy implications for integrated planning. Int. J. Water Resour. Dev. 2016, 32, 477–494. [Google Scholar] [CrossRef]
- Akpor, O.B.; Otohinoyi, D.A.; Olaolu, D.T.; Aderiye, B.I. Pollutants in wastewater effluents: Impacts and remediation processes. Int. J. Environ. Res. Earth Sci. 2014, 3, 50–59. [Google Scholar]
- Farhadi, A.; Ameri, A.; Tamjidi, S. Application of Agricultural Wastes as a Low-cost Adsorbent for Removal of Heavy Metals and Dyes from Wastewater: A Review Study. Phys. Chem. Res. 2021, 9, 211–226. [Google Scholar] [CrossRef]
- Muthuraman, G.; Teng, T.T. Extraction and recovery of rhodamine B, methyl violet and methylene blue from industrial wastewater using D2EHPA as an extractant. J. Ind. Eng. Chem. 2009, 15, 841–846. [Google Scholar] [CrossRef]
- López-Ahumada, E.; Salazar-Hernández, M.; Talavera-López, A.; Solis-Marcial, O.J.; Hernández-Soto, R.; Ruelas-Leyva, J.P.; Hernández, J.A. Removal of Anionic and Cationic Dyes Present in Solution Using Biomass of Eichhornia crassipes as Bioadsorbent. Molecules 2022, 27, 6442. [Google Scholar] [CrossRef] [PubMed]
- Hisarlı, G.; Atun, G.; Ortaboy, S.; Tüzün, E. A systematic study for the removal of anionic dyes by sepiolites modified with a homologous series of trimethylammonium-surfactants from single and binary component solutions. Sep. Sci. Technol. 2022, 57, 1304–1326. [Google Scholar] [CrossRef]
- Chacca, D.E.M.; Maldonado, I.; Vilca, F.Z. Environmental and ecotoxicological effects of drugs used for the treatment of COVID-19. Front. Environ. Sci. 2022, 10, 1287. [Google Scholar] [CrossRef]
- Ruiz, M.; Font, G. Ecotoxicological Effects of Pharmaceuticals in the Environment; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2011; pp. 227–246. [Google Scholar]
- Taghizade Firozjaee, T.; Mehrdadi, N.; Baghdadi, M.; Nabi Bidhendi, G.R. Application of nanotechnology in pesticides removal from aqueous solutions-A review. Int. J. Nanosci. Nanotechnol. 2018, 14, 43–56. [Google Scholar]
- Mnif, W.; Hassine, A.I.H.; Bouaziz, A.; Bartegi, A.; Thomas, O.; Roig, B. Effect of endocrine disruptor pesti-cides: A review. Int. J. Environ. Res. Public Health 2011, 8, 2265–2303. [Google Scholar] [CrossRef]
- Bretveld, R.W.; Thomas, C.M.; Scheepers, P.T.; Zielhuis, G.A.; Roeleveld, N. Pesticide exposure: The hormonal function of the female reproductive system disrupted? Reprod. Biol. Endocrinol. 2006, 4, 30. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; Saxena, M.; Lochab, A. Recent progress in nanomaterials for adsorptive removal of organic con-taminants from wastewater. ChemistrySelect 2020, 5, 335–353. [Google Scholar] [CrossRef]
- Yao, L.; Selmi, A.; Esmaeili, H. A review study on new aspects of biodemulsifiers: Production, features and their application in wastewater treatment. Chemosphere 2021, 284, 131364. [Google Scholar] [CrossRef]
- Renu; Agarwal, M.; Singh, K. Methodologies for removal of heavy metal ions from wastewater: An overview. Interdiscip. Environ. Rev. 2017, 18, 124–142. [Google Scholar] [CrossRef]
- Obijole, A.O.; Ogungbemi, S.T.; Adekunbi, A.E.; Sani, B.S.; Idi, M.D.; Oke, I.A. Electrochemical Treatment of Water as an Effective and Emerging Technology. In Handbook of Research on Water Sciences and Society; IGI Global: Hershey, PA, USA, 2022; pp. 293–314. [Google Scholar]
- Gao, R.; Mosquera-Romero, S.; Ntagia, E.; Wang, X.; Rabaey, K.; Bonin, L. Electrochemical separation of or-ganic and inorganic contaminants in wastewater. J. Electrochem. Soc. 2022, 169, 033505. [Google Scholar] [CrossRef]
- Fernandes, A.; Pacheco, M.; Ciríaco, L.; Lopes, A. Review on the electrochemical processes for the treatment of sanitary landfill leachates: Present and future. Appl. Catal. B Environ. 2015, 176, 183–200. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.A.; Naeem, H.T.; Hadi, R.T. A comparative study of chemical material additives on poly-acrylamide to treatment of waste water in refineries. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; Volume 518, p. 062003. [Google Scholar]
- Teh, C.Y.; Budiman, P.M.; Shak, K.P.Y.; Wu, T.Y. Recent Advancement of Coagulation–Flocculation and Its Application in Wastewater Treatment. Ind. Eng. Chem. Res. 2016, 55, 4363–4389. [Google Scholar] [CrossRef]
- Ezugbe, E.O.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef]
- Pohl, A. Removal of Heavy Metal Ions from Water and Wastewaters by Sulfur-Containing Precipitation Agents. Water, Air, Soil Pollut. 2020, 231, 503. [Google Scholar] [CrossRef]
- Kumar, K.V.; Gadipelli, S.; Wood, B.; Ramisetty, K.A.; Stewart, A.A.; Howard, C.A.; Brett, D.J.L.; Rodriguez-Reinoso, F. Characterization of the adsorption site energies and heterogeneous surfaces of porous materials. J. Mater. Chem. A 2019, 7, 10104–10137. [Google Scholar] [CrossRef]
- Tamjidi, S.; Esmaeili, H.; Moghadas, B.K. Application of magnetic adsorbents for removal of heavy metals from wastewater: A review study. Mater. Res. Express 2019, 6, 102004. [Google Scholar] [CrossRef]
- Costanzo, F.; Silvestrelli, P.L.; Ancilotto, F. Physisorption, diffusion, and chemisorption pathways of H2 molecule on graphene and on (2, 2) carbon nanotube by first principles calculations. J. Chem. Theory Comput. 2012, 8, 1288–1294. [Google Scholar] [CrossRef]
- Tang, X.; Zhou, Y.; Xu, Y.; Zhao, Q.; Zhou, X.; Lu, J. Sorption of polycyclic aromatic hydrocarbons from aqueous solution by hexadecyltrimethylammonium bromide modified fibric peat. J. Chem. Technol. Biotechnol. 2010, 85, 1084–1091. [Google Scholar] [CrossRef]
- Kanamarlapudi, S.L.R.K.; Chintalpudi, V.K.; Muddada, S. Application of Biosorption for Removal of Heavy Metals from Wastewater. Biosorption 2018, 18, 69. [Google Scholar] [CrossRef]
- De Gisi, S.; Lofrano, G.; Grassi, M.; Notarnicola, M. Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: A review. Sustain. Mater. Technol. 2016, 9, 10–40. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Yun, Y.-S. Bacterial biosorbents and biosorption. Biotechnol. Adv. 2008, 26, 266–291. [Google Scholar] [CrossRef] [PubMed]
- Ameri, A.; Tamjidi, S.; Dehghankhalili, F.; Farhadi, A.; Saati, M.A. Application of algae as low cost and effective bio-adsorbent for removal of heavy metals from wastewater: A review study. Environ. Technol. Rev. 2020, 9, 85–110. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, V.; Singh, J. Role of fungi in the removal of heavy metals and dyes from wastewater by biosorption processes. In Recent Advancement in White Biotechnology Through Fungi: Volume 3: Perspective for Sustainable Environments; Springer: Cham, Switzerland, 2019; pp. 397–418. [Google Scholar]
- Maqbool, M.; Bhatti, H.N.; Sadaf, S.; Al-Anazy, M.M.; Iqbal, M. Biocomposite of polyaniline and sodium alginate with Oscillatoria biomass: A potential adsorbent for the removal of basic blue 41. J. Mater. Res. Technol. 2020, 9, 14729–14741. [Google Scholar] [CrossRef]
- Nair, V.; Panigrahy, A.; Vinu, R. Development of novel chitosan–lignin composites foradsorption of dyes and metal ions from wastewater. Chem. Eng. J. 2014, 254, 491–502. [Google Scholar] [CrossRef]
- Sivashankar, R.; Susheeba, O.K.; Sivasubramanian, V. Adsorption of organic dye on to novel magnetic bio-composite: Kinetics and equilibrium studies. Res. J. Chem. Environ. 2015, 19, 48–57. [Google Scholar]
- Oyekanmi, A.A.; Alshammari, M.B.; Ibrahim, M.N.M.; Hanafiah, M.M.; Elnaggar, A.Y.; Ahmad, A.; Oyediran, A.T.; Rosli, M.A.; Setapar, S.H.M.; Daud, N.N.N.; et al. Highly Effective Cow Bone Based Biocomposite for the Sequestration of Organic Pollutant Parameter from Palm Oil Mill Effluent in a Fixed Bed Column Adsorption System. Polymers 2021, 14, 86. [Google Scholar] [CrossRef] [PubMed]
- TAĞAÇ, A.A.; YURDAKOÇ, M.K. SULU ÇÖZELTİDEKİ REMAZOL MAVİSİ (RB)’NIN KİTOSAN/SEP BİYOKOMPOZİTİ ÜZERİNE ADSORPSİYONU. Dokuz Eylül Üniversitesi Mühendislik Fakültesi Fen Ve Mühendislik Derg. 2018, 20, 286–298. [Google Scholar]
- Zeb, M.A.; Shah, J.; Jan, M.R. Alginate magnetic graphene oxide biocomposite synthesis for removal of aromatic amines from aqueous samples. Environ. Sci. Pollut. Res. 2023, 30, 51099–51112. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, Z.R.; Wen, Y.; Xu, C.; Li, L.; Zhou, Q. Removal of Reactive Blue (RB2) from Aqueous Solution by Chitosan/Diatomite Biocomposite. Adv. Mater. Res. 2011, 393, 1394–1397. [Google Scholar] [CrossRef]
- Metin, A.; Çiftçi, H.; Alver, E. Efficient Removal of Acidic Dye Using Low-Cost Biocomposite Beads. Ind. Eng. Chem. Res. 2013, 52, 10569–10581. [Google Scholar] [CrossRef]
- Moosa, A.A.; Ridha, A.M.; Kadhim, N.A. Use of biocomposite adsorbents for the removal of methylene blue dye from aqueous solution. Am. J. Mater. Sci. 2016, 6, 135–146. [Google Scholar]
- Ferkous, H.; Rouibah, K.; Hammoudi, N.-E.; Alam, M.; Djilani, C.; Delimi, A.; Laraba, O.; Yadav, K.K.; Ahn, H.-J.; Jeon, B.-H.; et al. The Removal of a Textile Dye from an Aqueous Solution Using a Biocomposite Adsorbent. Polymers 2022, 14, 2396. [Google Scholar] [CrossRef]
- Ozdemir, O.; Armagan, B.; Turan, M.; Çelik, M.S. Comparison of the adsorption characteristics of azo-reactive dyes on mezoporous minerals. Dye. Pigment. 2004, 62, 49–60. [Google Scholar] [CrossRef]
- Santos, S.C.; Boaventura, R.A. Treatment of a simulated textile wastewater in a sequencing batch reactor (SBR) with addition of a low-cost adsorbent. J. Hazard. Mater. 2015, 291, 74–82. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Tenhu, H.; Khattab, S.A.; Eldeeb, A.A.; Azaam, M.M. Highly efficient adsorbent material for removal of methylene blue dye based on functionalized polyacrylonitrile. Eur. Polym. J. 2022, 169, 111138. [Google Scholar] [CrossRef]
- Prabakaran, E.; Pillay, K.; Brink, H. Hydrothermal synthesis of magnetic-biochar nanocomposite derived from avocado peel and its performance as an adsorbent for the removal of methylene blue from wastewater. Mater. Today Sustain. 2022, 18, 100123. [Google Scholar] [CrossRef]
- Md Nurus, S.; Nanami, H.; Makoto, T.; Shoeb, A. Preparation of chitosan/laterite/iron oxide-based biocomposite and its application as a potential adsorbent for the removal of methylene blue from aqueous solution. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100658. [Google Scholar]
- Jiwon, C.; Neha, S.; Minyoung, K.; Kyusik, Y. Activated carbon derived from sucrose and melamine as low-cost adsorbent with fast adsorption rate for removal of methylene blue in wastewaters. J. Water Process Eng. 2022, 47, 102763. [Google Scholar]
- Thekkudan, V.N.; Ponnusamy, S.K.; Charles, C.; Sundar, S.L.; Vishnu, D.; Anbalagan, S.; Vaithyanathan, V.K.; Subramanian, S. Review on nanoadsorbents: A solution for heavy metal removal from wastewater. IET Nanobiotechnol. 2016, 11, 213–224. [Google Scholar] [CrossRef]
- Channegowda, M. Recent advances in environmentally benign hierarchical inorganic nano-adsorbents for the removal of poisonous metal ions in water: A review with mechanistic insight into toxicity and adsorption. Nanoscale Adv. 2020, 2, 5529–5554. [Google Scholar] [CrossRef] [PubMed]
- Bruckmann, F.S.; Schnorr, C.; Oviedo, L.R.; Knani, S.; Silva, L.F.O.; Silva, W.L.; Dotto, G.L.; Rhoden, C.R.B. Adsorption and Photocatalytic Degradation of Pesticides into Nanocomposites: A Review. Molecules 2022, 27, 6261. [Google Scholar] [CrossRef]
- El-Sayed, M.E. Nanoadsorbents for water and wastewater remediation. Sci. Total. Environ. 2020, 739, 139903. [Google Scholar] [CrossRef]
- Dil, E.A.; Ghaedi, M.; Asfaram, A. The performance of nanorods material as adsorbent for removal of azo dyes and heavy metal ions: Application of ultrasound wave, optimization and modeling. Ultrason. Sonochem. 2017, 34, 792–802. [Google Scholar] [CrossRef]
- Rahdar, S.; Rahdar, A.; Sattari, M.; Hafshejani, L.D.; Tolkou, A.K.; Kyzas, G.Z. Barium/Cobalt@Polyethylene Glycol Nanocomposites for Dye Removal from Aqueous Solutions. Polymers 2021, 13, 1161. [Google Scholar] [CrossRef]
- Nyankson, E.; Adjasoo, J.; Efavi, J.K.; Amedalor, R.; Yaya, A.; Manu, G.P.; Asare, K.; Amartey, N.A. Characterization and evaluation of zeolite A/Fe3O4 nanocomposite as a potential adsorbent for removal of organic molecules from wastewater. J. Chem. 2019, 2019, 8090756. [Google Scholar] [CrossRef]
- Boushehrian, M.M.; Esmaeili, H.; Foroutan, R. Ultrasonic assisted synthesis of Kaolin/CuFe2O4 nanocompo-site for removing cationic dyes from aqueous media. J. Environ. Chem. Eng. 2020, 8, 103869. [Google Scholar] [CrossRef]
- Mojarad, A.A.; Tamjidi, S.; Esmaeili, H. Clay/starch/Fe3O4 nanocomposite as an efficient adsorbent for the removal of methyl violet dye from aqueous media. Int. J. Environ. Anal. Chem. 2020, 102, 8159–8180. [Google Scholar] [CrossRef]
- Delnavaz, M.; Kazemimofrad, Z. Nano zerovalent iron (NZVI) adsorption performance on acidic dye 36 removal: Optimization of effective factors, isotherm and kinetic study. Environ. Prog. Sustain. Energy 2020, 39, e13349. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Arjmand, O.; Amani, A.M.; Babapoor, A.; Fateh, M.A.; Fateh, H.; Mojoudi, F.; Esmaeili, H.; Jahandideh, S. Erythrosine Adsorption from Aqueous Solution via Decorated Graphene Oxide with Magnetic Iron Oxide Nano Particles: Kinetic and Equilibrium Studies. Acta Chim. Slov. 2018, 65, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Khaleghi, H.; Esmaeili, H.; Jaafarzadeh, N.; Ramavandi, B. Date seed activated carbon decorated with CaO and Fe3O4 nanoparticles as a reusable sorbent for removal of formaldehyde. Korean J. Chem. Eng. 2022, 39, 146–160. [Google Scholar] [CrossRef]
- Khaleghi, H.; Jaafarzadeh, N.; Esmaeili, H.; Ramavandi, B. Alginate@Fe3O4@Bentonite nanocomposite for formaldehyde removal from synthetic and real effluent: Optimization by central composite design. Environ. Sci. Pollut. Res. 2022, 30, 29566–29580. [Google Scholar] [CrossRef]
- Ahmadipouya, S.; Haris, M.H.; Ahmadijokani, F.; Jarahiyan, A.; Molavi, H.; Moghaddam, F.M.; Rezakazemi, M.; Arjmand, M. Magnetic Fe3O4@UiO-66 nanocomposite for rapid adsorption of organic dyes from aqueous solution. J. Mol. Liq. 2020, 322, 114910. [Google Scholar] [CrossRef]
- Alsaiari, N.S.; Osman, H.; Amari, A.; Tahoon, M.A. The Synthesis of Metal–Organic-Framework-Based Ternary Nanocomposite for the Adsorption of Organic Dyes from Aqueous Solutions. Magnetochemistry 2022, 8, 133. [Google Scholar] [CrossRef]
- Abdullah, T.A.; Juzsakova, T.; Rasheed, R.T.; Salman, A.D.; Sebestyen, V.; Domokos, E.; Sluser, B.; Cretescu, I. Polystyrene-Fe3O4-MWCNTs nanocomposites for toluene removal from water. Materials 2021, 14, 5503. [Google Scholar] [CrossRef]
- Zangiabadi, M.; Saljooqi, A.; Shamspur, T.; Mostafavi, A. Evaluation of GO nanosheets decorated by CuFe2O4 and CdS nanoparticles as photocatalyst for the degradation of dinoseb and imidacloprid pesticides. Ceram. Int. 2020, 46, 6124–6128. [Google Scholar] [CrossRef]
- Naghizadeh, M.; Taher, M.A.; Tamaddon, A. Facile synthesis and characterization of magnetic nanocom-posite ZnO/CoFe2O4 hetero-structure for rapid photocatalytic degradation of imidacloprid. Heliyon 2019, 5, 02870. [Google Scholar] [CrossRef] [PubMed]
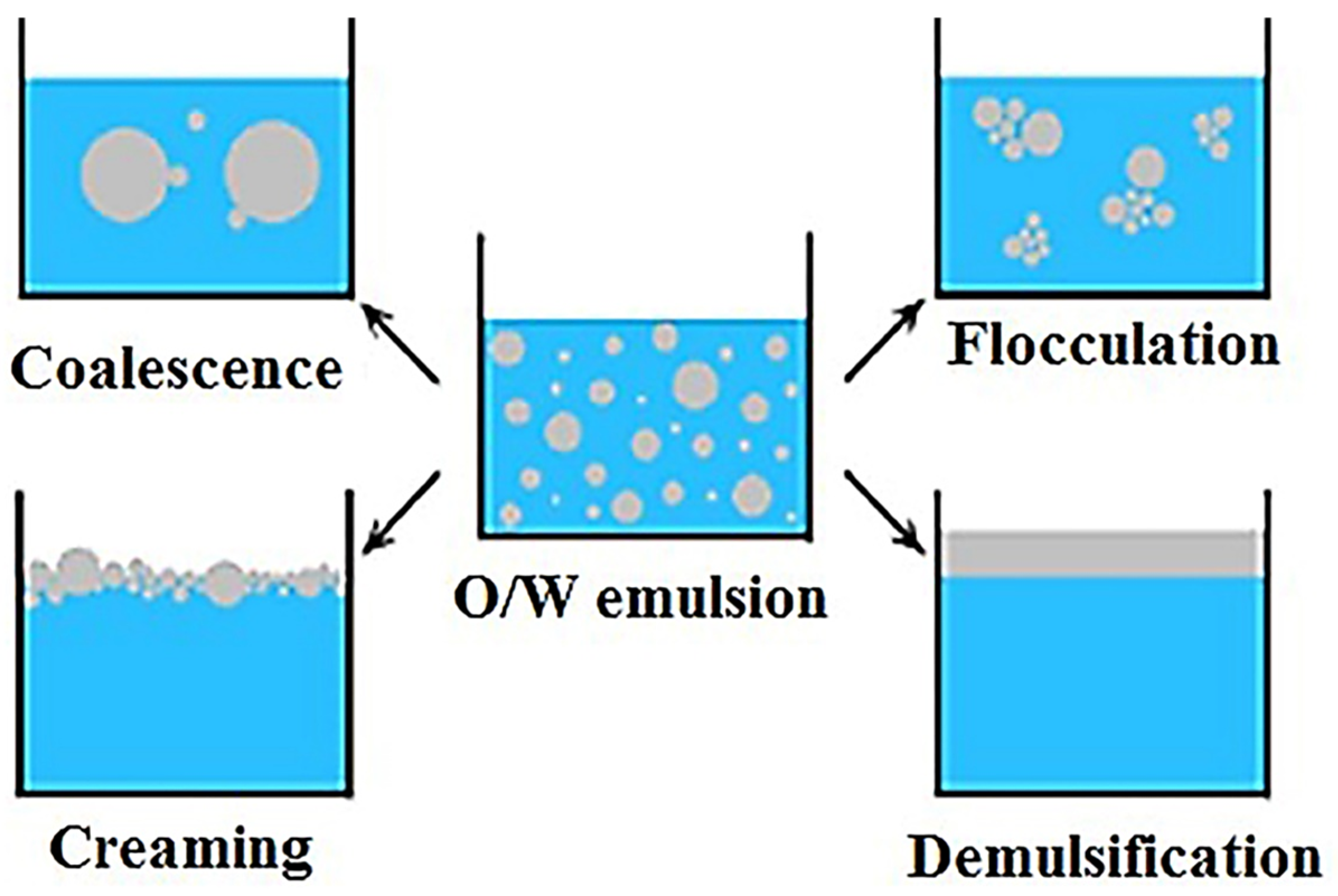
- Elanchezhiyan, S.S.; Prabhu, S.M.; Meenakshi, S. Effective adsorption of oil droplets from oil-in-water emul-sion using metal ions encapsulated biopolymers: Role of metal ions and their mechanism in oil removal. Int. J. Biol. Macromol. 2018, 112, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Xu, G.; Liu, Y. Oil sorption mechanism and capability of cattail fiber assembly. J. Ind. Text. 2012, 43, 330–337. [Google Scholar] [CrossRef]
- Saad, M.A.; Kamil, M.; Abdurahman, N.H.; Yunus, R.M.; Awad, O.I. An Overview of Recent Advances in State-of-the-Art Techniques in the Demulsification of Crude Oil Emulsions. Processes 2019, 7, 470. [Google Scholar] [CrossRef]
- Connors, S.; Lanza, R.; Sirocki, A.; Bergendahl, J. Removal of ibuprofen from drinking water using adsorption. In Major Qualifying Project Completed in Partial Fulfillment of the Bachelor of Science Degree at Worcester Polytechnic Institute; Worcester Polytechnic Institute: Worcester, MA, USA, 2013. [Google Scholar]
- Wang, F.; Zhang, M.; Sha, W.; Wang, Y.; Hao, H.; Dou, Y.; Li, Y. Sorption Behavior and Mechanisms of Organic Contaminants to Nano and Microplastics. Molecules 2020, 25, 1827. [Google Scholar] [CrossRef]
- Xu, B.; Liu, F.; Brookes, P.C.; Xu, J. Microplastics play a minor role in tetracycline sorption in the presence of dissolved organic matter. Environ. Pollut. 2018, 240, 87–94. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, K.; Huang, X.; Liu, J. Sorption of pharmaceuticals and personal care products to polyethylene debris. Environ. Sci. Pollut. Res. 2016, 23, 8819–8826. [Google Scholar] [CrossRef]
- Razanajatovo, R.M.; Ding, J.; Zhang, S.; Jiang, H.; Zou, H. Sorption and desorption of selected pharmaceuticals by polyethylene microplastics. Mar. Pollut. Bull. 2018, 136, 516–523. [Google Scholar] [CrossRef]
- El-Sayed, H.E.; El-Sayed, M.M. Assessment of food processing and pharmaceutical industrial wastes as potential biosorbents: A review. BioMed Res. Int. 2014, 2014, 146769. [Google Scholar] [CrossRef]
- Tamjidi, S.; Moghadas, B.K.; Esmaeili, H.; Khoo, F.S.; Gholami, G.; Ghasemi, M. Improving the surface properties of adsorbents by surfactants and their role in the removal of toxic metals from wastewater: A review study. Process. Saf. Environ. Prot. 2021, 148, 775–795. [Google Scholar] [CrossRef]
- Iftekhar, S.; Ramasamy, D.L.; Srivastava, V.; Asif, M.B.; Sillanpää, M. Understanding the factors affecting the adsorption of Lanthanum using different adsorbents: A critical review. Chemosphere 2018, 204, 413–430. [Google Scholar] [CrossRef] [PubMed]
- Gunatilake, S.K. Methods of removing heavy metals from industrial wastewater. J. Multidis. Eng. Sci. Stud. 2015, 1, 12–18. [Google Scholar]
- Zahoor, M. Effect of agitation speed on adsorption of imidacloprid on activated carbon. J. Chem. Soc. Pak. 2011, 33, 305. [Google Scholar]
- Lü, T.; Zhang, S.; Qi, D.; Zhang, D.; Zhao, H. Thermosensitive poly(N-isopropylacrylamide)-grafted magnetic nanoparticles for efficient treatment of emulsified oily wastewater. J. Alloys Compd. 2016, 688, 513–520. [Google Scholar] [CrossRef]
- Xing, W.; Liu, Q.; Wang, J.; Xia, S.; Ma, L.; Lu, R.; Zhang, Y.; Huang, Y.; Wu, G. High Selectivity and Reusability of Biomass-Based Adsorbent for Chloramphenicol Removal. Nanomaterials 2021, 11, 2950. [Google Scholar] [CrossRef]
- Nasiri, A.; Rajabi, S.; Amiri, A.; Fattahizade, M.; Hasani, O.; Lalehzari, A.; Hashemi, M. Adsorption of tetra-cycline using CuCoFe2O4@ Chitosan as a new and green magnetic nanohybrid adsorbent from aqueous solutions: Isotherm, kinetic and thermodynamic study. Arab. J. Chem. 2022, 15, 104014. [Google Scholar] [CrossRef]
- Akhayere, E.; Vaseashta, A.; Kavaz, D. Novel Magnetic Nano Silica Synthesis Using Barley Husk Waste for Removing Petroleum from Polluted Water for Environmental Sustainability. Sustainability 2020, 12, 10646. [Google Scholar] [CrossRef]
- Azizi, A.; Moniri, E.; Hassani, A.H.; Ahmad Panahi, H. Reusability, optimization, and adsorption studies of modified graphene oxide in the removal of Direct Red 81 using response surface methodology. Adv. Environ. Technol. 2020, 6, 175–185. [Google Scholar]
- Badran, I.; Khalaf, R. Adsorptive removal of alizarin dye from wastewater using maghemite nanoadsorbents. Sep. Sci. Technol. 2019, 55, 2433–2448. [Google Scholar] [CrossRef]
- Ghani, N.R.N.A.; Jami, M.S.; Alam, M.Z. The role of nanoadsorbents and nanocomposite adsorbents in the removal of heavy metals from wastewater: A review and prospect. Pollution 2021, 7, 153–179. [Google Scholar] [CrossRef]
- Li, H.; Cheng, Y.; Li, J.; Li, T.; Zhu, J.; Deng, W.; Zhu, J.; He, D. Preparation and Adsorption Performance Study of Graphene Quantum Dots@ZIF-8 Composites for Highly Efficient Removal of Volatile Organic Compounds. Nanomaterials 2022, 12, 4008. [Google Scholar] [CrossRef] [PubMed]
- Nyankson, E.; Adjasoo, J.; Efavi, J.K.; Yaya, A.; Manu, G.; Kingsford, A.; Abrokwah, R.Y. Synthesis and kinetic adsorption characteristics of Zeolite/CeO2 nanocomposite. Sci. Afr. 2019, 7, e00257. [Google Scholar] [CrossRef]
- Chowdhury, A.; Khan, A.A.; Kumari, S.; Hussain, S. Superadsorbent Ni–Co–S/SDS nanocomposites for ul-trahigh removal of cationic, anionic organic dyes and toxic metal ions: Kinetics, isotherm and adsorption mechanism. ACS Sustain. Chem. Eng. 2019, 7, 4165–4176. [Google Scholar] [CrossRef]
- Li, P.; Yang, C.; Xu, X.; Miao, C.; He, T.; Jiang, B.; Wu, W. Preparation of Bio-Based Aerogel and Its Ad-sorption Properties for Organic Dyes. Gels 2022, 8, 755. [Google Scholar] [CrossRef] [PubMed]


| Adsorbent | Contaminants | Conditions | RE (%) | qmax (m/g) | Ref. |
|---|---|---|---|---|---|
| Polyaniline/sodium alginate/Oscillatoria biomass | Basic Blue 41 | pH = 9, AD = 0.1 g/L, t = 2 h, T = 30 °C | - | 786.6 | [43] |
| Polyaniline/sodium alginate/Oscillatoria biomass | Basic Blue 41 | pH = 9, AD = 0.1 g/L, t = 2 h, T = 30 °C | - | 700.1 | [43] |
| Polyaniline/sodium alginate/Oscillatoria biomass | Basic Blue 41 | pH = 9, AD = 0.1 g/L, t = 2 h, T = 30 °C | - | 634.4 | [43] |
| Chitosan–alkali lignin composite | Remazol Brilliant Blue R | pH = 2, PC = 10 ppm, AD = 2 g/L, t = 1 h | - | 111.1 | [44] |
| Non-immobilized magnetic biocomposite | MB | pH = 7, PC = 10 ppm, AD = 5 g/L, t = 140 min | 99.12 | 232.6 | [45] |
| Cow bones/zeolite/coconut biocomposite | Palm oil | pH = 5.28 | 99.47 | - | [46] |
| Chitosan/sepiolite biocomposite | Remazol Blue | pH = 5, PC = 3 ppm, AD = 0.8 g/L, t = 24 h, T = 45 °C | 99 | 256 | [47] |
| Alginate/magnetic/graphene oxide biocomposite | Aniline | - | 98.82 | 18.39 | [48] |
| Alginate/magnetic/graphene oxide biocomposite | p-chloroaniline | - | 96.65 | 17.13 | [48] |
| Alginate/magnetic/graphene oxide biocomposite | p-nitroaniline | - | 93.55 | 15.24 | [48] |
| Chitosan/diatomite biocomposite | Reactive Blue | pH = 4, AD = 0.75 g/L, T = 30 °C, t = 50 min | 96.36 | - | [49] |
| Chitosan/zeolite biocomposite | Acid Black 194 | pH = 3, AD = 5 g/L, T = 22 °C | - | 2140 | [50] |
| Chitosan | MB | pH = 6, PC = 105 ppm, AD = 1.6 g/L, t = 6 h, T = 25 °C | 85.85 | 11.04 | [51] |
| 10% chitosan/90% activated carbon | MB | pH = 6, PC = 105 ppm, AD = 1.6 g/L, t = 6 h, T = 25 °C | 91.7 | 11.92 | [51] |
| 20% chitosan/80% activated carbon | MB | pH = 6, PC = 105 ppm, AD = 1.6 g/L, t = 6 h, T = 25 °C | 95.81 | 11.99 | [51] |
| Olive waste/KOH | MB | pH = 11, t = 0.5 h, T = 40 °C | 504.9 | [52] | |
| Olive waste/KOH/PEG silica gel | MB | pH = 12, t = 0.5 h, T = 50 °C | 161.44 | [52] | |
| Modified sepiolite | Everzol Yellow | pH = 2, PC = 25 ppm, AD = 50 g/L, t = 2 h | - | 169.1 | [53] |
| Modified zeolite | Everzol red | pH = 2, PC = 25 ppm, AD = 50 g/L, t = 2 h | - | 111.1 | [53] |
| Metal hydroxide sludge | Direct Blue 85 | pH = 7, T = 25 °C | - | 339 | [54] |
| Aminated polyacrylonitrile | MB | pH = 8, t = 20 min | 99.7 | 227.2 | [55] |
| Magnetic/biochar nanocomposite | MB | - | - | 62.1 | [56] |
| Chitosan/laterite/Fe3O4 | MB | - | - | 16 | [57] |
| Sucrose and melamine-derived AC | MB | t = 13 min | - | 454.57 | [58] |
| Nanocomposite | Contaminant | Conditions | RE (%) | qmax (mg/g) | Ref. |
|---|---|---|---|---|---|
| Zinc (II) oxide nanorods loaded on AC | Crystal violet | PC = 14 ppm, AD = 0.025 g/L, t = 0.085 h | 98.66 | 81.6 | [63] |
| Zinc (II) oxide nanorods loaded on AC | MB | PC = 14 ppm, AD = 0.025 g/L, t = 0.085 h | 99.46 | 83.9 | [63] |
| Barium/Cobalt@PEG | MB | pH = 2, PC = 20 ppm, t = 1 h | 78.6 | 215.08 | [64] |
| Zeolite/Fe3O4 | MB | pH = 7, PC = 2 ppm, AD = 2.8 g/L, t = 3 h | 97.5 | 2.57 | [65] |
| Kaolin/CuFe2O4 | MB | pH = 8, PC = 10 ppm, AD = 1.5 g/L, t = 0.5 h, T = 25 °C | 98.64 | 120.48 | [66] |
| Kaolin/CuFe2O4 | MV | pH = 8, PC = 10 ppm, AD = 1.5 g/L, t = 0.5 h, T = 25 °C | 99.25 | 117.65 | [66] |
| Clay/starch/ Fe3O4 | MV | pH = 9, PC = 10 ppm, AD = 1.5 g/L, t = 2.5 h | 99.73 | 29.67 | [67] |
| Nano zerovalent iron | Acidic dye | pH = 5.5, PC = 30 ppm, AD = 0.5 g/L, t = 0.025 h | 94.29 | 125.8 | [68] |
| Graphene oxide/Fe3O4 | Erythrosine | pH = 7, PC = 50 ppm, AD = 4 g/L, t = 2 min, T = 60 °C | 97 | 149.25 | [69] |
| Date seed AC/CaO/Fe3O4 | Formaldehyde | pH = 7, PC = 5 ppm, AD = 2.5 g/L, t = 80 min, T = 25 °C | 98.22 | 24.01 | [70] |
| AC/Fe3O4 | Formaldehyde | pH = 7, PC = 5 ppm, AD = 3 g/L, t = 100 min, T = 25 °C | 95.67 | 24.21 | [70] |
| CaO/Fe3O4 | Formaldehyde | pH = 7, PC = 5 ppm, AD = 3 g/L, t = 100 min, T = 25 °C | 95.14 | 21.28 | [70] |
| Alginate@Fe3O4@bentonite | Formaldehyde | pH = 9.96, PC = 10.69 ppm, AD = 1.28 g/L, t = 16.53 min, T = 25 °C | 94.56 | 50.25 | [71] |
| Fe3O4@UiO-66 | MO | pH = 4, PC = 20 ppm, AD = 0.2 g/L, t = 3 h, T = 25 °C | - | 243.9 | [72] |
| Fe3O4@UiO-66 | MB | pH = 8, PC = 20 ppm, AD = 0.2 g/L, t = 3 h, T = 25 °C | - | 769.23 | [72] |
| SiO2/MnFe2O4/ZIF-8 MOF | Malachite green | pH = 3, PC = 100 ppm, AD = 0.2 g/L, t = 1 h, T = 25 °C | - | 1000.03 | [73] |
| SiO2/MnFe2O4/ZIF-8 MOF | Methyl red | pH = 3, PC = 100 ppm, AD = 0.2 g/L, t = 0.5 h, T = 25 °C | - | 1111.12 | [73] |
| Magnetite/MWCNTs | Toluene | pH = 5, PC = 50 ppm, AD = 0.02 g/L, t = 1 h, T = 35 °C | 85 | 1113 | [74] |
| GO/CuFe2O4/CdS | Imidacloprid and Dinoseb | pH = 7, AD = 0.15 g/L, t = 140 min | 91–94 | - | [75] |
| ZnO@CoFe2O4 | Imidacloprid | pH = 10, AD = 0.1 g/L, t = 45 min | 79.3 | - | [76] |
| Adsorbent | Contaminant | RE (%) | RE (%) after n Reuse Cycles | Ref. |
|---|---|---|---|---|
| Poly(N-isopropylacrylamide)/Fe3O4 | Oil/water emulsion | About 97 | n = 7; RE = about 90% | [90] |
| Corn stalk biomass/chitosan/Fe3O4 biocomposite | Chloramphenicol | 95.49 | n = 5; RE = 88.52% | [91] |
| CuCoFe2O4@Chitosan nanocompoiste | Tetracycline | 93.07 | n = 4; RE = 82.16% | [92] |
| Magnetic nano-silica | Oily wastewater | 85 | n = 5; RE = 80% | [93] |
| Poly methyl vinyl ketone/aniline/GO | Direct Red 81 | 89 | n = 7; RE = 70% | [94] |
| SiO2/MnFe2O4/ZIF-8 | Malachite green | 99 | n = 5; RE = 93.5% | [73] |
| SiO2/MnFe2O4/ZIF-8 | Methyl red | 93 | n = 5; RE = 86.5% | [73] |
| Magnetic modified raw aquatic weed biocomposite | MB | 99.12 | n = 6; RE = 65.6% | [45] |
| Sodium alginate/magnetic modified raw aquatic weed biocomposite | MB | 89 | n = 6; RE = 38% | [45] |
| Adsorbent | Pollutant | PFO Constants | PSO Constants | Elovich Constants | ID Constants | Ref. |
|---|---|---|---|---|---|---|
| SiO2/MnFe2O4/ZIF-8 | Malachite green | k1 = 0.062 R2 = 0.991 | k2 = 0.001 R2 = 0.999 | - | - | [73] |
| SiO2/MnFe2O4/ZIF-8 | Methyl red | k1 = 0.160 R2 = 0.941 | k2 = 0.001 R2 = 0.999 | - | - | [73] |
| Zeolite/cerium oxide | MB | k1 = 0.00076 R2 = 0.44513 | k2 = 1.14655 R2 = 0.99276 | - | ki1 = 0.10233 ki2 = 0.05229 ki3 = 0.02049 | [97] |
| Graphene quantum dots/zeolitic imidazolate framework | Toluene | k1 = 0.038 R2 = 0.98 | k2 = 0.000093 R2 = 0.95 | - | - | [98] |
| Ni-Co-S/SDS | Nile blue | k1 = 0.0499 R2 = 0.9614 | k2 = 0.0011 R2 = 0.9998 | - | - | [99] |
| CaCO3@starch/polyacrylamide /TEMPO-oxidized | Congo red | - | - | - | ki1 = 13.84 ki2 = 3.54 | [100] |
| CaCO3@starch/polyacrylamide /TEMPO-oxidized | MB | - | - | - | ki1 = 5.21 ki2 = 1.19 | [100] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayet, A.M.; Hijji, M.; Saleh, E.A.M.; Reza, A.; Kadhim, S.I.; Abdullaev, S.S.; Alsalamy, A.; Hassan, Z.F.; Gomez, C.V.; Tene, T. The Role of Biocomposites and Nanocomposites in Eliminating Organic Contaminants from Effluents. Water 2023, 15, 3093. https://doi.org/10.3390/w15173093
Mayet AM, Hijji M, Saleh EAM, Reza A, Kadhim SI, Abdullaev SS, Alsalamy A, Hassan ZF, Gomez CV, Tene T. The Role of Biocomposites and Nanocomposites in Eliminating Organic Contaminants from Effluents. Water. 2023; 15(17):3093. https://doi.org/10.3390/w15173093
Chicago/Turabian StyleMayet, Abdulilah Mohammad, Mohammad Hijji, Ebraheem Abdu Musad Saleh, Arif Reza, Sokaina Issa Kadhim, Sherzod Shukhratovich Abdullaev, Ali Alsalamy, Zahraa F. Hassan, Cristian Vacacela Gomez, and Talia Tene. 2023. "The Role of Biocomposites and Nanocomposites in Eliminating Organic Contaminants from Effluents" Water 15, no. 17: 3093. https://doi.org/10.3390/w15173093