Wastewater Hydroponics for Pollutant Removal and Food Production: Principles, Progress and Future Outlook
Abstract
1. Introduction
2. Wastewater Hydroponic Process
2.1. Nutrient Solutions
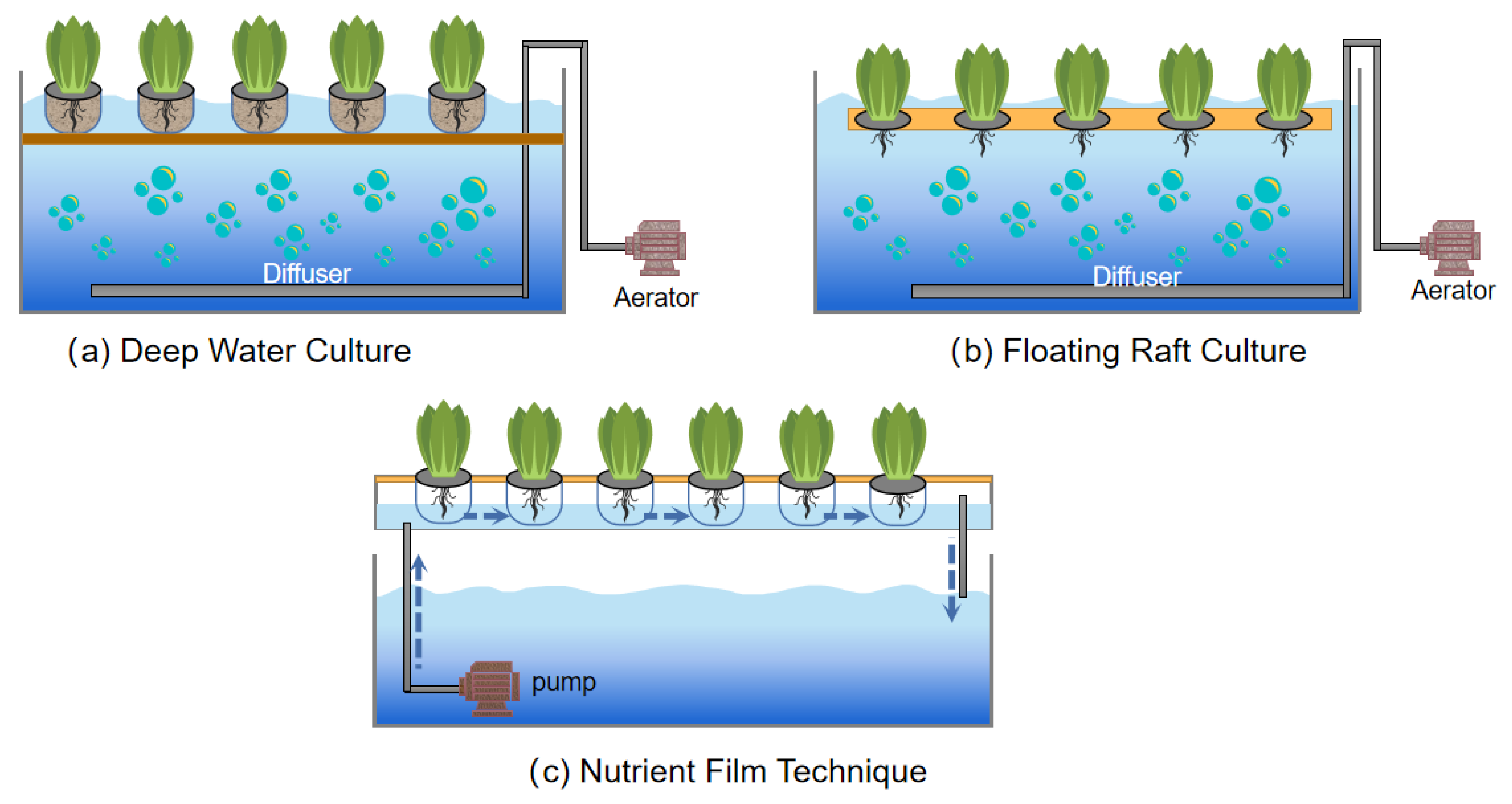
2.2. Wastewater Hydroponics
2.3. Hydroponic Plants
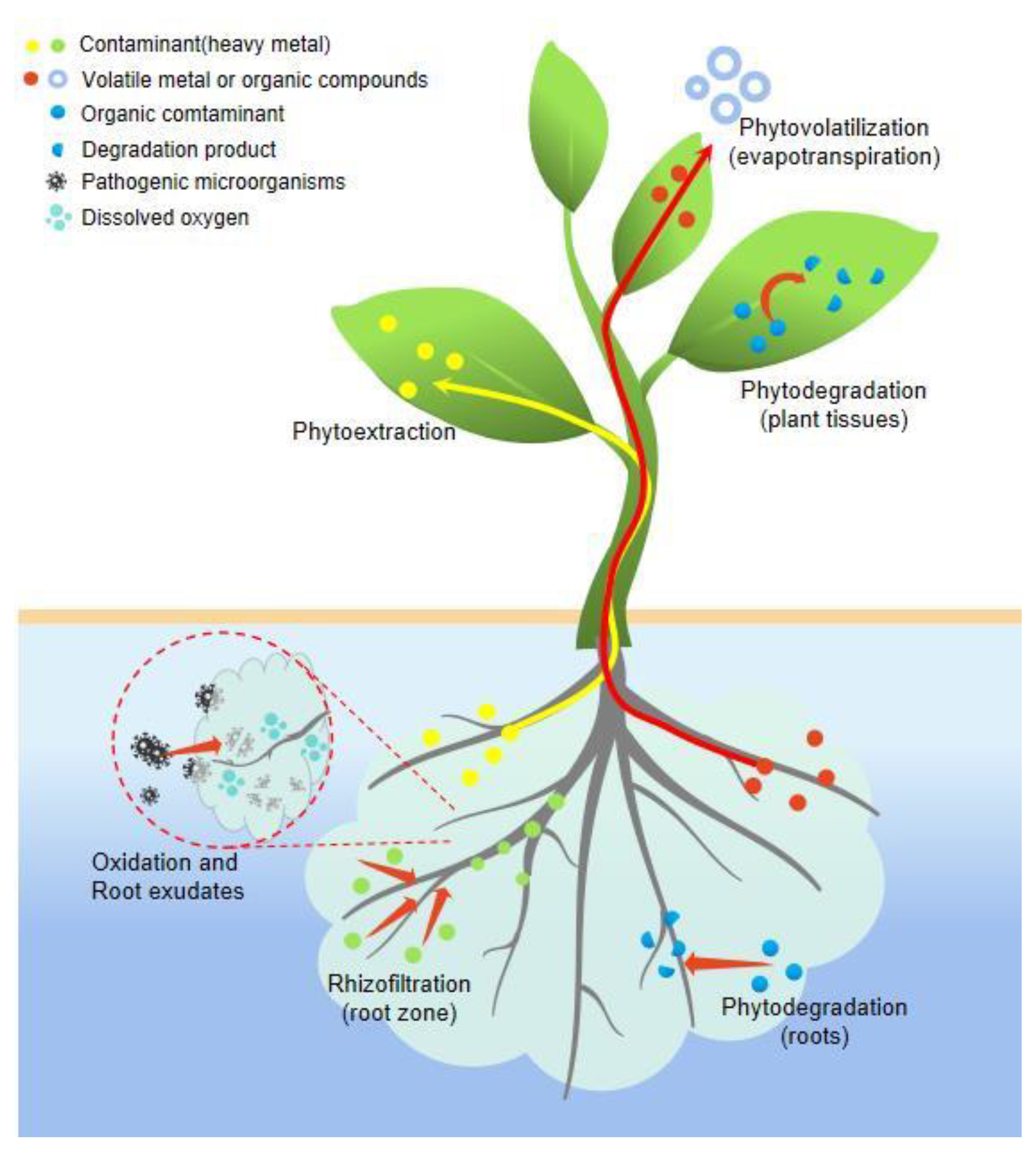
3. Mechanism of Hydroponic Plants to Purify Wastewater
Comparison between Wastewater Hydroponics and Constructed Wetlands
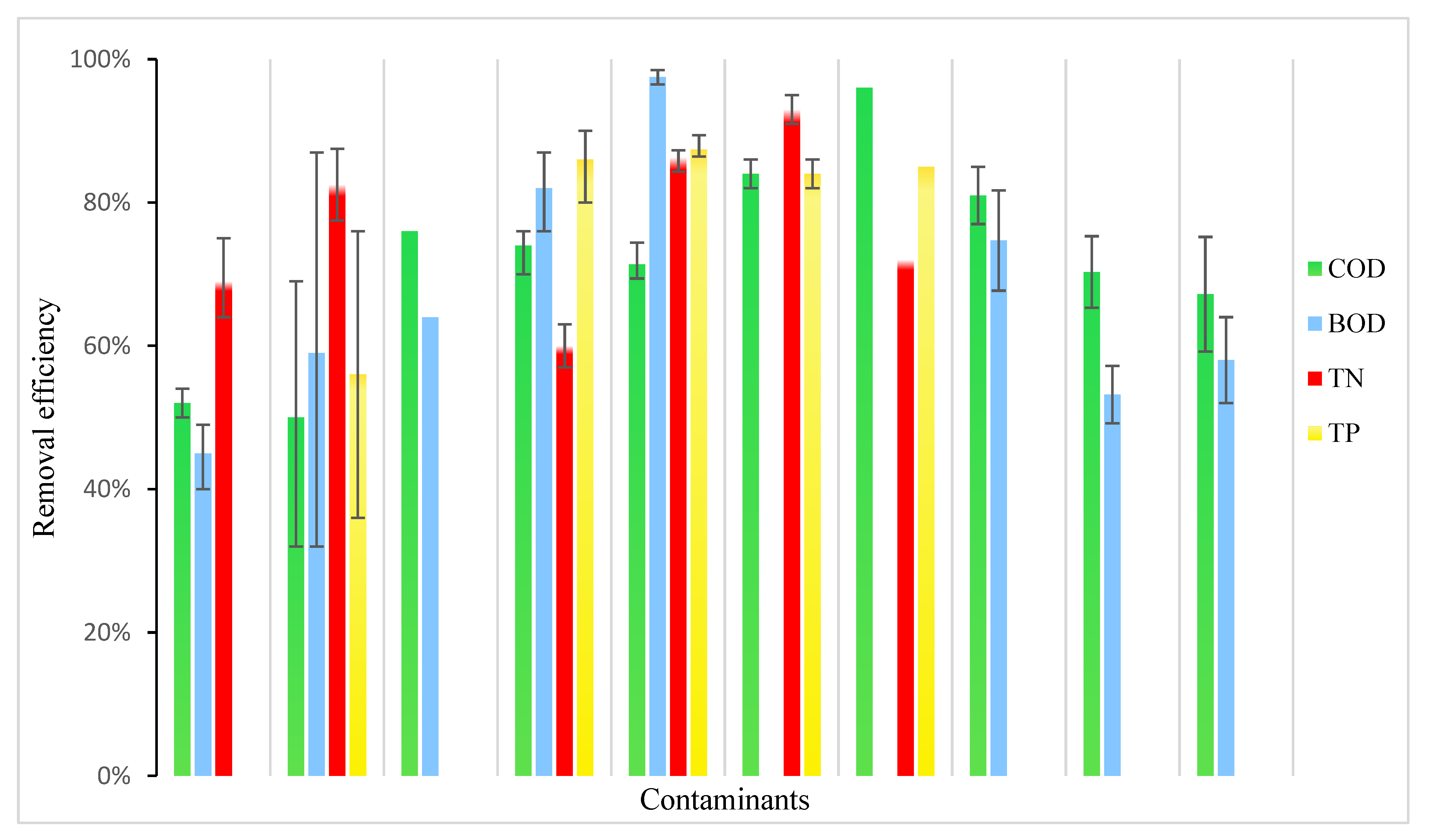
4. Hydroponics Performance in Pollutant Removal from Wastewater
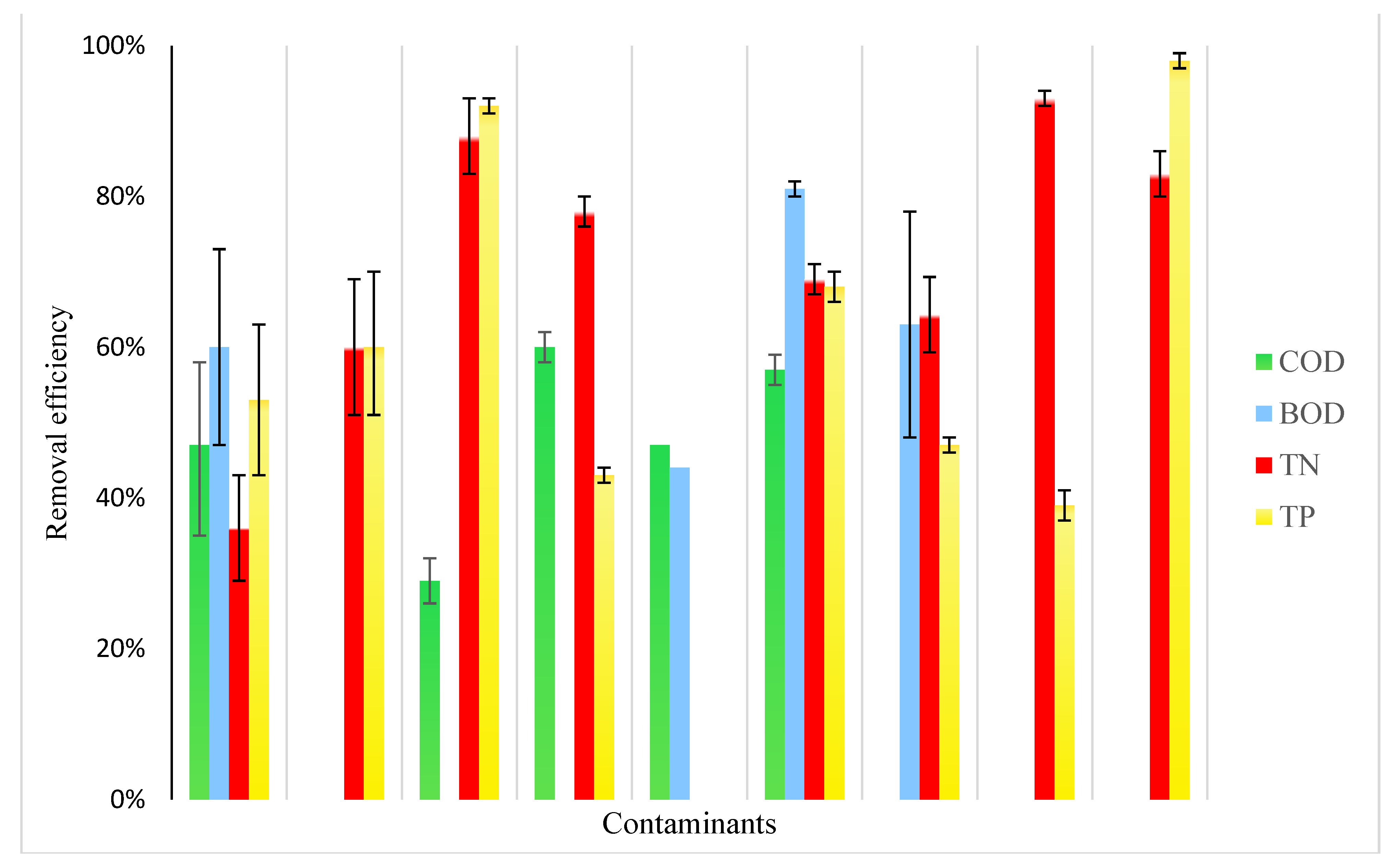
5. Advantages and Limitations of Wastewater Hydroponics
6. Conclusions
7. Future Perspective
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- United Nations Summary Progress Update 2021: SDG 6—Water and Sanitation for All. Available online: https://www.unwater.org/publications/summary-progress-update-2021-sdg-6-water-and-sanitation-all (accessed on 1 July 2023).
- Idris-Nda, A.; Aliyu, H.K.; Dalil, M. The Challenges of Domestic Wastewater Management in Nigeria: A Case Study of Minna, Central Nigeria. Int. J. Dev. Sustain. 2013, 2, 1169–1182. [Google Scholar]
- Saxena, G.; Bharagava, R. Organic and Inorganic Pollutants in Industrial Wastes. In Environmental Pollutants and Their Bioremediation Approaches; CRC Press: Boca Raton, FL, USA, 2017; pp. 23–56. [Google Scholar]
- United Nations Environment Programme 2017 UN World Water Development Report, Wastewater: The Untapped Resource. Available online: https://www.unep.org/resources/publication/2017-un-world-water-development-report-wastewater-untapped-resource (accessed on 1 July 2023).
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead Toxicity: A Review. Interdiscip. Toxicol. 2015, 8, 55–64. [Google Scholar] [CrossRef]
- Choong, T.S.Y.; Chuah, T.G.; Robiah, Y.; Gregory Koay, F.L.; Azni, I. Arsenic Toxicity, Health Hazards and Removal Techniques from Water: An Overview. Desalination 2007, 217, 139–166. [Google Scholar] [CrossRef]
- Costa, M.; Klein, C.B. Toxicity and Carcinogenicity of Chromium Compounds in Humans. Crit. Rev. Toxicol. 2006, 36, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Rafati Rahimzadeh, M.; Rafati Rahimzadeh, M.; Kazemi, S.; Moghadamnia, A.-A. Cadmium Toxicity and Treatment: An Update. Casp. J. Intern. Med. 2017, 8, 135–145. [Google Scholar] [CrossRef]
- Poongothai, S.; Ravikrishnan, R.; Balikrishna Murthy, P. Endocrine Disruption and Perspective Human Health Implications: A Review. Internet J. Toxicol. 2007, 4, 10–1210. [Google Scholar]
- Raghav, M.; Eden, S.; Mitchell, K.; Witte, B. Contaminants of Emerging Concern in Water; The University of Arizona: Tucson, AZ, USA, 2013. [Google Scholar]
- Crini, G.; Lichtfouse, E. Advantages and Disadvantages of Techniques Used for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W. Adsorptive Removal of Antibiotics from Water and Wastewater: Progress and Challenges. Sci. Total Environ. 2015, 532, 112–126. [Google Scholar] [CrossRef]
- Xu, B.; Liu, S.; Zhou, J.L.; Zheng, C.; Weifeng, J.; Chen, B.; Zhang, T.; Qiu, W. PFAS and Their Substitutes in Groundwater: Occurrence, Transformation and Remediation. J. Hazard. Mater. 2021, 412, 125159. [Google Scholar] [CrossRef]
- Palansooriya, K.N.; Yang, Y.; Tsang, Y.F.; Sarkar, B.; Hou, D.; Cao, X.; Meers, E.; Rinklebe, J.; Kim, K.-H.; Ok, Y.S. Occurrence of Contaminants in Drinking Water Sources and the Potential of Biochar for Water Quality Improvement: A Review. Crit. Rev. Environ. Sci. Technol. 2020, 50, 549–611. [Google Scholar] [CrossRef]
- Qiu, B.; Tao, X.; Wang, H.; Li, W.; Ding, X.; Chu, H. Biochar as a Low-Cost Adsorbent for Aqueous Heavy Metal Removal: A Review. J. Anal. Appl. Pyrolysis 2021, 155, 105081. [Google Scholar] [CrossRef]
- Chen, W.-H.; Hoang, A.T.; Nižetić, S.; Pandey, A.; Cheng, C.K.; Luque, R.; Ong, H.C.; Thomas, S.; Nguyen, X.P. Biomass-Derived Biochar: From Production to Application in Removing Heavy Metal-Contaminated Water. Process Saf. Environ. Prot. 2022, 160, 704–733. [Google Scholar] [CrossRef]
- Carra, I.; Sánchez Pérez, J.A.; Malato, S.; Autin, O.; Jefferson, B.; Jarvis, P. Performance of Different Advanced Oxidation Processes for Tertiary Wastewater Treatment to Remove the Pesticide Acetamiprid. J. Chem. Technol. Biotechnol. 2016, 91, 72–81. [Google Scholar] [CrossRef]
- Sun, Y.; Xue, S.; Li, L.; Ding, W.; Liu, J.; Han, Y. Sulfur Dioxide and o -Xylene Co-Treatment in Biofilter: Performance, Bacterial Populations and Bioaerosols Emissions. J. Environ. Sci. 2018, 69, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Antony, A.; Low, J.H.; Gray, S.; Childress, A.E.; Le-Clech, P.; Leslie, G. Scale Formation and Control in High Pressure Membrane Water Treatment Systems: A Review. J. Memb. Sci. 2011, 383, 1–16. [Google Scholar] [CrossRef]
- Sardare, M.D.; Admane, S.V. A Review on Plant without Soil-Hydroponics. Int. J. Res. Eng. Technol. 2013, 02, 299–304. [Google Scholar] [CrossRef]
- Fussy, A.; Papenbrock, J. An Overview of Soil and Soilless Cultivation Techniques—Chances, Challenges and the Neglected Question of Sustainability. Plants 2022, 11, 1153. [Google Scholar] [CrossRef]
- Magwaza, S.T.; Magwaza, L.S.; Odindo, A.O.; Mditshwa, A. Hydroponic Technology as Decentralised System for Domestic Wastewater Treatment and Vegetable Production in Urban Agriculture: A Review. Sci. Total Environ. 2020, 698, 134154. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Fisiologia Vegetal, 5th ed.; Artmed: Joane, Portugal, 2013. [Google Scholar]
- Boyden, B.H.; Rababah, A.A. Recycling Nutrients from Municipal Wastewater. Desalination 1996, 106, 241–246. [Google Scholar] [CrossRef]
- Krishnasamy, K.; Nair, J.; Bäuml, B. Hydroponic System for the Treatment of Anaerobic Liquid. Water Sci. Technol. 2012, 65, 1164–1171. [Google Scholar] [CrossRef] [PubMed]
- Adrover, M.; Moyà, G.; Vadell, J. Use of Hydroponics Culture to Assess Nutrient Supply by Treated Wastewater. J. Environ. Manag. 2013, 127, 162–165. [Google Scholar] [CrossRef] [PubMed]
- da Silva Cuba Carvalho, R.; Bastos, R.G.; Souza, C.F. Influence of the Use of Wastewater on Nutrient Absorption and Production of Lettuce Grown in a Hydroponic System. Agric. Water Manag. 2018, 203, 311–321. [Google Scholar] [CrossRef]
- KHAN, F.A. A Review on Hydroponic Greenhouse Cultivation for Sustainable Agriculture. Int. J. Agric. Environ. Food Sci. 2018, 2, 59–66. [Google Scholar] [CrossRef]
- Norström, A. Treatment of Domestic Wastewater Using Microbiological Processes and Hydroponics in Sweden; Royal Institute of Technology: Stockholm, Sweden, 2005. [Google Scholar]
- Majid, M.; Khan, J.N.; Ahmad Shah, Q.M.; Masoodi, K.Z.; Afroza, B.; Parvaze, S. Evaluation of Hydroponic Systems for the Cultivation of Lettuce (Lactuca sativa L., Var. Longifolia) and Comparison with Protected Soil-Based Cultivation. Agric. Water Manag. 2021, 245, 106572. [Google Scholar] [CrossRef]
- Grewal, H.S.; Maheshwari, B.; Parks, S.E. Water and Nutrient Use Efficiency of a Low-Cost Hydroponic Greenhouse for a Cucumber Crop: An Australian Case Study. Agric. Water Manag. 2011, 98, 841–846. [Google Scholar] [CrossRef]
- Neocleous, D.; Vasilakakis, M. Effects of NaCl Stress on Red Raspberry (Rubus Idaeus L. ‘Autumn Bliss’). Sci. Hortic. 2007, 112, 282–289. [Google Scholar] [CrossRef]
- Munns, R. Comparative Physiology of Salt and Water Stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef]
- Trajkova, F.; Papadantonakis, N.; Savvas, D. Comparative Effects of NaCl and CaCl2 Salinity on Cucumber Grown in a Closed Hydroponic System. HortScience 2006, 41, 437–441. [Google Scholar] [CrossRef]
- Germano, R.P.; Melito, S.; Cacini, S.; Carmassi, G.; Leoni, F.; Maggini, R.; Montesano, F.F.; Pardossi, A.; Massa, D. Sweet Basil Can Be Grown Hydroponically at Low Phosphorus and High Sodium Chloride Concentration: Effect on Plant and Nutrient Solution Management. Sci. Hortic. 2022, 304, 111324. [Google Scholar] [CrossRef]
- Velazquez-Gonzalez, R.S.; Garcia-Garcia, A.L.; Ventura-Zapata, E.; Barceinas-Sanchez, J.D.O.; Sosa-Savedra, J.C. A Review on Hydroponics and the Technologies Associated for Medium- and Small-Scale Operations. Agriculture 2022, 12, 646. [Google Scholar] [CrossRef]
- Damasceno, L.M.O.; de Andrade Júnior, A.S.; Gheyi, H.R. Cultivation of Gerbera Irrigated with Treated Domestic Effluents. Rev. Bras. Eng. Agrícola E Ambient. 2010, 14, 582–588. [Google Scholar] [CrossRef]
- Greipsson, S. Phytoremediation. Nat. Educ. Knowl. 2011, 30, 7. [Google Scholar]
- Chaney, R.L. Phytoextraction and Phytomining of Soil Nickel. In Nickel in Soils and Plants; CRC Press: Boca Raton, FL, USA, 2018; pp. 341–374. [Google Scholar]
- Dinh, T.; Dobo, Z.; Kovacs, H. Phytomining of Noble Metals—A Review. Chemosphere 2022, 286, 131805. [Google Scholar] [CrossRef] [PubMed]
- Dang, P.; Li, C. A Mini-Review of Phytomining. Int. J. Environ. Sci. Technol. 2022, 19, 12825–12838. [Google Scholar] [CrossRef]
- Nagata, T.; Morita, H.; Akizawa, T.; Pan-Hou, H. Development of a Transgenic Tobacco Plant for Phytoremediation of Methylmercury Pollution. Appl. Microbiol. Biotechnol. 2010, 87, 781–786. [Google Scholar] [CrossRef]
- Zayed, A.; Pilon-Smits, E.; deSouza, M.; Lin, Z.-Q.; Terry, N. Remediation of Selenium-Polluted Soils and Waters by Phytovolatilization. In Phytoremediation of Contaminated Soil and Water; CRC Press: Boca Raton, FL, USA, 2020; pp. 61–83. [Google Scholar]
- Wang, L.; Hou, D.; Shen, Z.; Zhu, J.; Jia, X.; Ok, Y.S.; Tack, F.M.G.; Rinklebe, J. Field Trials of Phytomining and Phytoremediation: A Critical Review of Influencing Factors and Effects of Additives. Crit. Rev. Environ. Sci. Technol. 2020, 50, 2724–2774. [Google Scholar] [CrossRef]
- Etim, E.E. Phytoremediation and Its Mechanisms: A Review. Int. J. Environ. Bioenergy 2012, 2, 120–136. [Google Scholar]
- Kristanti, R.A.; Ngu, W.J.; Yuniarto, A.; Hadibarata, T. Rhizofiltration for Removal of Inorganic and Organic Pollutants in Groundwater: A Review. Biointerface Res. Appl. Chem. 2021, 11, 12326–12347. [Google Scholar] [CrossRef]
- Wang, H.; Sheng, L.; Xu, J. Clogging Mechanisms of Constructed Wetlands: A Critical Review. J. Clean. Prod. 2021, 295, 126455. [Google Scholar] [CrossRef]
- Wang, M.; Chen, S.; Jia, X.; Chen, L. Concept and Types of Bioremediation. In Handbook of Bioremediation; Elsevier: Amsterdam, The Netherlands, 2021; pp. 3–8. [Google Scholar]
- Tonelli, F.C.P.; Tonelli, F.M.P.; Lemos, M.S.; Nunes, N.A.d.M. Mechanisms of Phytoremediation. In Phytoremediation; Elsevier: Amsterdam, The Netherlands, 2022; pp. 37–64. [Google Scholar]
- Bedabati Chanu, L.; Gupta, A. Phytoremediation of Lead Using Ipomoea Aquatica Forsk. in Hydroponic Solution. Chemosphere 2016, 156, 407–411. [Google Scholar] [CrossRef]
- Khumanleima Chanu, H.; Gupta, A. Necrosis as an Adaptive Response to Copper Toxicity in Ipomoea Aquatica Forsk. and Its Possible Application in Phytoremediation. Acta Physiol. Plant 2014, 36, 3275–3281. [Google Scholar] [CrossRef]
- Garousi, F.; Kovács, B.; Andrási, D.; Veres, S. Selenium Phytoaccumulation by Sunflower Plants under Hydroponic Conditions. Water Air Soil. Pollut. 2016, 227, 382. [Google Scholar] [CrossRef]
- January, M.C.; Cutright, T.J.; Van Keulen, H.; Wei, R. Hydroponic Phytoremediation of Cd, Cr, Ni, As, and Fe: Can Helianthus Annuus Hyperaccumulate Multiple Heavy Metals? Chemosphere 2008, 70, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.; Pang, J.; Li, Y.; Li, Y.; Zhu, J.; Wang, J.; Chang, M.; Wang, L. Hydroponic Phytoremediation of Ni, Co and Pb by Iris sibirica L. Sustainability 2021, 13, 9400. [Google Scholar] [CrossRef]
- Lee, J.H.; Hossner, L.R.; Attrep, M., Jr.; Kung, K.S. Uptake and Translocation of Plutonium in Two Plant Species Using Hydroponics. Environ. Pollut. 2002, 117, 61–68. [Google Scholar] [CrossRef]
- Dushenkov, S.; Vasudev, D.; Kapulnik, Y.; Gleba, D.; Fleisher, D.; Ting, K.C.; Ensley, B. Removal of Uranium from Water Using Terrestrial Plants. Environ. Sci. Technol. 1997, 31, 3468–3474. [Google Scholar] [CrossRef]
- Lewis, B.G.; MacDonell, M.M. Release of Radon-222 by Vascular Plants: Effect of Transpiration and Leaf Area. J. Environ. Qual. 1990, 19, 93–97. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Xiong, J.; Li, L.; Zhao, B.; Sohail, I.; He, Z. A Constructed Wetland System with Aquatic Macrophytes for Cleaning Contaminated Runoff/Storm Water from Urban Area in Florida. J. Environ. Manag. 2021, 280, 111794. [Google Scholar] [CrossRef]
- Vymazal, J. Removal of Enteric Bacteria in Constructed Treatment Wetlands with Emergent Macrophytes: A Review. J. Environ. Sci. Health Part. A 2005, 40, 1355–1367. [Google Scholar] [CrossRef]
- Dhir, B. Effective Control of Waterborne Pathogens by Aquatic Plants. In Waterborne Pathogens; Elsevier: Amsterdam, The Netherlands, 2020; pp. 339–361. [Google Scholar]
- Shingare, R.P.; Thawale, P.R.; Raghunathan, K.; Mishra, A.; Kumar, S. Constructed Wetland for Wastewater Reuse: Role and Efficiency in Removing Enteric Pathogens. J. Environ. Manag. 2019, 246, 444–461. [Google Scholar] [CrossRef]
- Alufasi, R.; Gere, J.; Chakauya, E.; Lebea, P.; Parawira, W.; Chingwaru, W. Mechanisms of Pathogen Removal by Macrophytes in Constructed Wetlands. Environ. Technol. Rev. 2017, 6, 135–144. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, Y.; Xie, H.; Yang, Z. Constructed Wetlands: A Review on the Role of Radial Oxygen Loss in the Rhizosphere by Macrophytes. Water 2018, 10, 678. [Google Scholar] [CrossRef]
- von Martels, J.Z.H.; Sadaghian Sadabad, M.; Bourgonje, A.R.; Blokzijl, T.; Dijkstra, G.; Faber, K.N.; Harmsen, H.J.M. The Role of Gut Microbiota in Health and Disease: In Vitro Modeling of Host-Microbe Interactions at the Aerobe-Anaerobe Interphase of the Human Gut. Anaerobe 2017, 44, 3–12. [Google Scholar] [CrossRef]
- Wu, S.; Carvalho, P.N.; Müller, J.A.; Manoj, V.R.; Dong, R. Sanitation in Constructed Wetlands: A Review on the Removal of Human Pathogens and Fecal Indicators. Sci. Total Environ. 2016, 541, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Curtis, T.P.; Mara, D.D.; Silva, S.A. Influence of PH, Oxygen, and Humic Substances on Ability of Sunlight to Damage Fecal Coliforms in Waste Stabilization Pond Water. Appl. Environ. Microbiol. 1992, 58, 1335–1343. [Google Scholar] [CrossRef]
- Fernández, A.; Tejedor, C.; Chordi, A. Effect of Different Factors on the Die-off of Fecal Bacteria in a Stabilization Pond Purification Plant. Water Res. 1992, 26, 1093–1098. [Google Scholar] [CrossRef]
- Headley, T.; Nivala, J.; Kassa, K.; Olsson, L.; Wallace, S.; Brix, H.; van Afferden, M.; Müller, R. Escherichia coli Removal and Internal Dynamics in Subsurface Flow Ecotechnologies: Effects of Design and Plants. Ecol. Eng. 2013, 61, 564–574. [Google Scholar] [CrossRef]
- Stefanakis, A.I.; Bardiau, M.; Trajano, D.; Couceiro, F.; Williams, J.B.; Taylor, H. Presence of Bacteria and Bacteriophages in Full-Scale Trickling Filters and an Aerated Constructed Wetland. Sci. Total Environ. 2019, 659, 1135–1145. [Google Scholar] [CrossRef]
- Haichar, F.e.Z.; Santaella, C.; Heulin, T.; Achouak, W. Root Exudates Mediated Interactions Belowground. Soil. Biol. Biochem. 2014, 77, 69–80. [Google Scholar] [CrossRef]
- Seidel, K. Macrophytes and Water Purification. In Biological Control of Water Pollution; University of Pennsylvania Press: Philadelphia, PA, USA, 1970; pp. 109–121. [Google Scholar]
- RodrÃguez-Navarro, D.N.; Dardanelli, M.S.; RuÃz-SaÃnz, J.E. Attachment of Bacteria to the Roots of Higher Plants. FEMS Microbiol. Lett. 2007, 272, 127–136. [Google Scholar] [CrossRef]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The Role of Root Exudates in Rhizosphere Interactions with Plants and other Organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef]
- Huang, X.-F.; Chaparro, J.M.; Reardon, K.F.; Zhang, R.; Shen, Q.; Vivanco, J.M. Rhizosphere Interactions: Root Exudates, Microbes, and Microbial Communities. Botany 2014, 92, 267–275. [Google Scholar] [CrossRef]
- Mustafa, H.M.; Hayder, G. Evaluation of Water Lettuce, Giant Salvinia and Water Hyacinth Systems in Phytoremediation of Domestic Wastewater. H2Open J. 2021, 4, 167–181. [Google Scholar] [CrossRef]
- Lu, S.; Zhang, X.; Wang, J.; Pei, L. Impacts of Different Media on Constructed Wetlands for Rural Household Sewage Treatment. J. Clean. Prod. 2016, 127, 325–330. [Google Scholar] [CrossRef]
- Steer, D.; Fraser, L.; Boddy, J.; Seibert, B. Efficiency of Small Constructed Wetlands for Subsurface Treatment of Single-Family Domestic Effluent. Ecol. Eng. 2002, 18, 429–440. [Google Scholar] [CrossRef]
- Romeo, D.; Vea, E.B.; Thomsen, M. Environmental Impacts of Urban Hydroponics in Europe: A Case Study in Lyon. Procedia CIRP 2018, 69, 540–545. [Google Scholar] [CrossRef]
- Gong, L.; Chen, G.; Li, J.; Zhu, G. Utilization of Rural Domestic Sewage Tailwaters by Ipomoea Aquatica in Different Hydroponic Vegetable and Constructed Wetland Systems. Water Sci. Technol. 2020, 386–400. [Google Scholar] [CrossRef]
- Yadav, S.K.; Rajagopal, K. Hydroponic Treatment System Plant for Canteen Wastewater Treatment in Park College of Technology. In Zero Waste; CRC Press: Boca Raton, FL, USA, 2019; pp. 187–202. [Google Scholar]
- Sangare, D.; Coulibaly, L.S.; Andrianisa, H.A.; Coulibaly, J.Z.; Coulibaly, L. Investigating the Capacity of Hydroponic System Using Lettuce (Lactuca sativa L.) in the Removal of Pollutants from Greywater while Ensuring Food Security. Int. J. Environ. Agric. Biotechnol. 2021, 6, 123–131. [Google Scholar] [CrossRef]
- Kaushal, J.; Mahajan, P. Design and Evaluation of Hydroponic System for Tertiary Treatment of STP Wastewater: An Eco Friendly Approach. Mater. Today Proc. 2021, 45, 4914–4918. [Google Scholar] [CrossRef]
- Nguyen, T.V.D.; Huynh, H.N.T.; Nguyen, M.N.H.; Ngo, T.V.; Vu, N.; Thinh, D. The Use of Water Spinach (Ipomoea Aquatica) in Domestic Wastewater Treatment. J. Agric. Dev. 2018, 17, 49–54. [Google Scholar] [CrossRef]
- Cui, H.; Schröder, P. Uptake, Translocation and Possible Biodegradation of the Antidiabetic Agent Metformin by Hydroponically Grown Typha Latifolia. J. Hazard. Mater. 2016, 308, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Panja, S.; Sarkar, D.; Zhang, Z.; Datta, R. Removal of Antibiotics and Nutrients by Vetiver Grass (Chrysopogon Zizanioides) from a Plug Flow Reactor Based Constructed Wetland Model. Toxics 2021, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Ndulini, S.F.; Sithole, G.M.; Mthembu, M.S. Investigation of Nutrients and Faecal Coliforms Removal in Wastewater Using a Hydroponic System. Phys. Chem. Earth Parts A/B/C 2018, 106, 68–72. [Google Scholar] [CrossRef]
- Goren, A.Y.; Yucel, A.; Sofuoglu, S.C.; Sofuoglu, A. Phytoremediation of Olive Mill Wastewater with Vetiveria zizanioides (L.) Nash and Cyperus alternifolius L. Environ. Technol. Innov. 2021, 24, 102071. [Google Scholar] [CrossRef]
- Eregno, F.; Moges, M.; Heistad, A. Treated Greywater Reuse for Hydroponic Lettuce Production in a Green Wall System: Quantitative Health Risk Assessment. Water 2017, 9, 454. [Google Scholar] [CrossRef]
- Jesse, S.D.; Zhang, Y.; Margenot, A.J.; Davidson, P.C. Hydroponic Lettuce Production Using Treated Post-Hydrothermal Liquefaction Wastewater (PHW). Sustainability 2019, 11, 3605. [Google Scholar] [CrossRef]
- Keller, R.; Perim, K.; Semionato, S.; Zandonade, E.; Cassini, S.; Gonçalves, R.F. Hydroponic Cultivation of Lettuce (Lactuca Sativa) Using Effluents from Primary, Secondary and Tertiary +UV Treatments. Water Supply 2005, 5, 95–100. [Google Scholar] [CrossRef]
- Davamani, V.; Indhu Parameshwari, C.; Arulmani, S.; Ezra John, J.; Poornima, R. Hydroponic Phytoremediation of Paperboard Mill Wastewater by Using Vetiver (Chrysopogon zizanioides). J. Environ. Chem. Eng. 2021, 9, 105528. [Google Scholar] [CrossRef]
- Jin, E.; Cao, L.; Xiang, S.; Zhou, W.; Ruan, R.; Liu, Y. Feasibility of Using Pretreated Swine Wastewater for Production of Water Spinach (Ipomoea Aquatic Forsk.) in a Hydroponic System. Agric. Water Manag. 2020, 228, 105856. [Google Scholar] [CrossRef]
- Cui, L.-H.; Luo, S.-M.; Zhu, X.-Z.; Liu, Y.-H. Treatment and Utilization of Septic Tank Effluent Using Vertical-Flow Constructed Wetlands and Vegetable Hydroponics. J. Environ. Sci 2003, 15, 75–82. [Google Scholar]
- Worku, A.; Tefera, N.; Kloos, H.; Benor, S. Bioremediation of Brewery Wastewater Using Hydroponics Planted with Vetiver Grass in Addis Ababa, Ethiopia. Bioresour. Bioprocess. 2018, 5, 39. [Google Scholar] [CrossRef]
- Gong, Y.; Chen, J.; Pu, R. The Enhanced Removal and Phytodegradation of Sodium Dodecyl Sulfate (SDS) in Wastewater Using Controllable Water Hyacinth. Int. J. Phytoremediation 2019, 21, 1080–1089. [Google Scholar] [CrossRef]
- Ispolnov, K.; Aires, L.M.I.; Lourenço, N.D.; Vieira, J.S. A Combined Vermifiltration-Hydroponic System for Swine Wastewater Treatment. Appl. Sci. 2021, 11, 5064. [Google Scholar] [CrossRef]
- Gebeyehu, A.; Shebeshe, N.; Kloos, H.; Belay, S. Suitability of Nutrients Removal from Brewery Wastewater Using a Hydroponic Technology with Typha Latifolia. BMC Biotechnol. 2018, 18, 74. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Xiong, H.; Chen, J.; Chen, K.; Chen, P.; Yu, C.; Zhu, A. Hydroponic Method for Ramie and Removal of Nitrogen and Phosphorus from Livestock Wastewater. Int. J. Phytoremediation 2018, 20, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Mokuolu, O.A.; Olokoba, S.O.; Aremu, S.A.; Olanlokun, O.K. Fish Pond Wastewater Hydroponic Treatment Potential of Citrullus Colocynthis. J. Res. Wildl. Environ. 2019, 11, 118–126. [Google Scholar]
- Zhang, D.Q.; Gersberg, R.M.; Hua, T.; Zhu, J.; Goyal, M.K.; Ng, W.J.; Tan, S.K. Fate of Pharmaceutical Compounds in Hydroponic Mesocosms Planted with Scirpus Validus. Environ. Pollut. 2013, 181, 98–106. [Google Scholar] [CrossRef]
- Mackuľak, T.; Mosný, M.; Škubák, J.; Grabic, R.; Birošová, L. Fate of Psychoactive Compounds in Wastewater Treatment Plant and the Possibility of Their Degradation Using Aquatic Plants. Environ. Toxicol. Pharmacol. 2015, 39, 969–973. [Google Scholar] [CrossRef]
- Cui, H.; de Angelis, M.H.; Schröder, P. Iopromide Exposure in Typha latifolia L.: Evaluation of Uptake, Translocation and Different Transformation Mechanisms in Planta. Water Res. 2017, 122, 290–298. [Google Scholar] [CrossRef]
- Gahlawat, S.; Gauba, P. Phytoremediation of Aspirin and Tetracycline by Brassica Juncea. Int. J. Phytoremediation 2016, 18, 929–935. [Google Scholar] [CrossRef]
- Dordio, A.V.; Belo, M.; Martins Teixeira, D.; Palace Carvalho, A.J.; Dias, C.M.B.; Picó, Y.; Pinto, A.P. Evaluation of Carbamazepine Uptake and Metabolization by Typha Spp., a Plant with Potential Use in Phytotreatment. Bioresour. Technol. 2011, 102, 7827–7834. [Google Scholar] [CrossRef]
- Yan, Q.; Feng, G.; Gao, X.; Sun, C.; Guo, J.; Zhu, Z. Removal of Pharmaceutically Active Compounds (PhACs) and Toxicological Response of Cyperus Alternifolius Exposed to PhACs in Microcosm Constructed Wetlands. J. Hazard. Mater. 2016, 301, 566–575. [Google Scholar] [CrossRef]
- Enyoh, C.E.; Isiuku, B.O. 2,4,6-Trichlorophenol (TCP) Removal from Aqueous Solution Using Canna Indica L.: Kinetic, Isotherm and Thermodynamic Studies. Chem. Ecol. 2021, 37, 64–82. [Google Scholar] [CrossRef]
- González García, M.; Fernández-López, C.; Pedrero-Salcedo, F.; Alarcón, J.J. Absorption of Carbamazepine and Diclofenac in Hydroponically Cultivated Lettuces and Human Health Risk Assessment. Agric. Water Manag. 2018, 206, 42–47. [Google Scholar] [CrossRef]
- Madikizela, L.M.; Ncube, S.; Chimuka, L. Uptake of Pharmaceuticals by Plants Grown under Hydroponic Conditions and Natural Occurring Plant Species: A Review. Sci. Total Environ. 2018, 636, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Shenker, M.; Harush, D.; Ben-Ari, J.; Chefetz, B. Uptake of Carbamazepine by Cucumber Plants—A Case Study Related to Irrigation with Reclaimed Wastewater. Chemosphere 2011, 82, 905–910. [Google Scholar] [CrossRef]
- Ravichandran, M.K.; Philip, L. Fate of Carbamazepine and Its Effect on Physiological Characteristics of Wetland Plant Species in the Hydroponic System. Sci. Total Environ. 2022, 846, 157337. [Google Scholar] [CrossRef]
- Topal, M. Uptake of Tetracycline and Degradation Products by Phragmites Australis Grown in Stream Carrying Secondary Effluent. Ecol. Eng. 2015, 79, 80–85. [Google Scholar] [CrossRef]
- Gul, H.; Raza, W.; Lee, J.; Azam, M.; Ashraf, M.; Kim, K.-H. Progress in Microbial Fuel Cell Technology for Wastewater Treatment and Energy Harvesting. Chemosphere 2021, 281, 130828. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.K.; Chiranjeevi, P.; Sukrampal; Patil, S.A. Integrated Drip Hydroponics-Microbial Fuel Cell System for Wastewater Treatment and Resource Recovery. Bioresour. Technol. Rep. 2020, 9, 100392. [Google Scholar] [CrossRef]
- Wang, S.; Adekunle, A.; Raghavan, V. Exploring the Integration of Bioelectrochemical Systems and Hydroponics: Possibilities, Challenges, and Innovations. J. Clean. Prod. 2022, 366, 132855. [Google Scholar] [CrossRef]
- Kantanoleon, N.; Zampetakis, L.; Manios, T. Public Perspective towards Wastewater Reuse in a Medium Size, Seaside, Mediterranean City: A Pilot Survey. Resour. Conserv. Recycl. 2007, 50, 282–292. [Google Scholar] [CrossRef]
- Drechsel, P.; Mahjoub, O.; Keraita, B. Social and Cultural Dimensions in Wastewater Use. In Wastewater; Springer: Dordrecht, The Netherlands, 2015; pp. 75–92. [Google Scholar]
- Verhoest, P.; Gaume, B.; Bauwens, J.; te Braak, P.; Huysmans, M. Public Acceptance of Recycled Water: A Survey of Social Attitudes toward the Consumption of Crops Grown with Treated Wastewater. Sustain. Prod. Consum. 2022, 34, 467–475. [Google Scholar] [CrossRef]
- Lahlou, F.Z.; Mackey, H.R.; McKay, G.; Al-Ansari, T. Reuse of Treated Industrial Wastewater and Bio-Solids from Oil and Gas Industries: Exploring New Factors of Public Acceptance. Water Resour. Ind. 2021, 26, 100159. [Google Scholar] [CrossRef]
- Schikora, A.; Virlogeux-Payant, I.; Bueso, E.; Garcia, A.V.; Nilau, T.; Charrier, A.; Pelletier, S.; Menanteau, P.; Baccarini, M.; Velge, P.; et al. Conservation of Salmonella Infection Mechanisms in Plants and Animals. PLoS ONE 2011, 6, e24112. [Google Scholar] [CrossRef] [PubMed]
- Hirneisen, K.A.; Kniel, K.E. Comparative Uptake of Enteric Viruses into Spinach and Green Onions. Food Environ. Virol. 2013, 5, 24–34. [Google Scholar] [CrossRef]
- DiCaprio, E.; Ma, Y.; Purgianto, A.; Hughes, J.; Li, J. Internalization and Dissemination of Human Norovirus and Animal Caliciviruses in Hydroponically Grown Romaine Lettuce. Appl. Environ. Microbiol. 2012, 78, 6143–6152. [Google Scholar] [CrossRef]
- Li, Y.; Chen, Y.-F.; Chen, P.; Min, M.; Zhou, W.; Martinez, B.; Zhu, J.; Ruan, R. Characterization of a Microalga Chlorella Sp. Well Adapted to Highly Concentrated Municipal Wastewater for Nutrient Removal and Biodiesel Production. Bioresour. Technol. 2011, 102, 5138–5144. [Google Scholar] [CrossRef] [PubMed]
- Ji, M.-K.; Kabra, A.N.; Salama, E.-S.; Roh, H.-S.; Kim, J.R.; Lee, D.S.; Jeon, B.-H. Effect of Mine Wastewater on Nutrient Removal and Lipid Production by a Green Microalga Micratinium Reisseri from Concentrated Municipal Wastewater. Bioresour. Technol. 2014, 157, 84–90. [Google Scholar] [CrossRef]
- Zhao, X.; Zhou, Y.; Huang, S.; Qiu, D.; Schideman, L.; Chai, X.; Zhao, Y. Characterization of Microalgae-Bacteria Consortium Cultured in Landfill Leachate for Carbon Fixation and Lipid Production. Bioresour. Technol. 2014, 156, 322–328. [Google Scholar] [CrossRef]
- Sun, Z.; Guo, W.; Yang, J.; Zhao, X.; Chen, Y.; Yao, L.; Hou, H. Enhanced Biomass Production and Pollutant Removal by Duckweed in Mixotrophic Conditions. Bioresour. Technol. 2020, 317, 124029. [Google Scholar] [CrossRef] [PubMed]
- Elshafai, S.; Elgohary, F.; Nasr, F.; Petervandersteen, N.; Gijzen, H. Nutrient Recovery from Domestic Wastewater Using a UASB-Duckweed Ponds System. Bioresour. Technol. 2007, 98, 798–807. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Shen, G. Growing Duckweed in Swine Wastewater for Nutrient Recovery and Biomass Production. Bioresour. Technol. 2011, 102, 848–853. [Google Scholar] [CrossRef] [PubMed]
| Name | Category | Use | Reference |
|---|---|---|---|
| Spinach | Leafy green flowering plant | Food | [92,93] |
| Romaine lettuce | Annual plant | Food | [81,93] |
| Bidens pilosa L | Herbaceous flowering plant | Food and medicine | [86] |
| Amaranthus hybridus | Annual flowering plant | Food, medicine, ornamental | |
| Chrysopogon zizanioides | Perennial bunchgrass | Skincare, animal feed, perfumery, medicine | [75,85,87,91,94] |
| Water lettuce | Aquatic plants | Animal feed, medicine | [75] |
| Giant salvinia | Aquatic fern | Cancer research | |
| Water hyacinth | Perennial aquatic plant | Bioenergy, food, medicine | [75,80,95] |
| Nephthytis podophyllum | Evergreen herbaceous plant | Ornamental | [82] |
| Ipomoea aquatica | Semi-aquatic, tropical plant | Food, animal feed, medicine | [83] |
| Pointed cabbage | Biennial plant | Food | [96] |
| Typha latifolia | Perennial herbaceous plant | Food and medicine | [97] |
| Umbrella palm | Grass-like plant | Ornamental | [87] |
| Ramie | Perennial herbaceous plant | Textile | [98] |
| Citrullus colocynthis | Desert viny plant | Food and medicine | [99] |
| Pharmaceutical Ingredients | Plant | Exposure Concentration | Period (Days) | Removal Efficiency | References |
|---|---|---|---|---|---|
| Antidiabetic II medicine (Metformin) | Typha latifolia | 6.45 mg/L | 28 | 74.0 ± 4.1% | [84] |
| 32.29 mg/L | 81.1 ± 3.3% | ||||
| Blood lipid regulator (Carbamazepine) | Scirpus validus | 0.5 mg/L | 21 | 54–64% | [100] |
| Analgesic (Naproxen) | 80–94% (30–42% by photodegradation) | ||||
| Anti-inflammatory (Diclofenac) | 85–98% (73–77% by photodegradation) | ||||
| Blood lipid regulator (Clofibric acid) | 41–73% | ||||
| Stimulant drug (Caffeine) | 100% | ||||
| Opioid analgesic (Codeine) | I. pseudacorus | 24 × 10−6 mg/L | 2 | 93% | [101] |
| Antidepressant (Citalopram) | 84 × 10−6 mg/L | 87% | |||
| X-ray contrast agent (Iopromide) | Typha latifolia | 15.8 mg/L | 28 | 87% | [102] |
| Anti-inflammatory and analgesic (Aspirin) | Brassica juncea | 4–180 mg/L | 28 | 90% | [103] |
| Antibiotic (Tetracycline) | 5–225 mg/L | 24 | 71% | ||
| Antibiotic (Ciprofloxacin) | Chrysopogon zizanioides | 10 mg/L | 7 | 93% | [85] |
| Antibiotic (Tetracycline) | 97% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mai, C.; Mojiri, A.; Palanisami, S.; Altaee, A.; Huang, Y.; Zhou, J.L. Wastewater Hydroponics for Pollutant Removal and Food Production: Principles, Progress and Future Outlook. Water 2023, 15, 2614. https://doi.org/10.3390/w15142614
Mai C, Mojiri A, Palanisami S, Altaee A, Huang Y, Zhou JL. Wastewater Hydroponics for Pollutant Removal and Food Production: Principles, Progress and Future Outlook. Water. 2023; 15(14):2614. https://doi.org/10.3390/w15142614
Chicago/Turabian StyleMai, Chao, Amin Mojiri, Swaminathan Palanisami, Ali Altaee, Yuhan Huang, and John L. Zhou. 2023. "Wastewater Hydroponics for Pollutant Removal and Food Production: Principles, Progress and Future Outlook" Water 15, no. 14: 2614. https://doi.org/10.3390/w15142614
APA StyleMai, C., Mojiri, A., Palanisami, S., Altaee, A., Huang, Y., & Zhou, J. L. (2023). Wastewater Hydroponics for Pollutant Removal and Food Production: Principles, Progress and Future Outlook. Water, 15(14), 2614. https://doi.org/10.3390/w15142614












