Review of Slow Sand Filtration for Raw Water Treatment with Potential Application in Less-Developed Countries
Abstract
1. Introduction
2. Contemporary Raw Water Purification Methods
2.1. Mechanical Filtration
2.2. Reverse Osmosis
2.3. Ion Exchange
2.4. Electrochemical Purification
2.5. Distillation
2.6. Sorption
2.7. Coagulation and Flocculation
2.8. Disinfection
3. Slow Sand Filtration
3.1. History
3.2. SSF Requirements
3.3. SSF Biological Processes
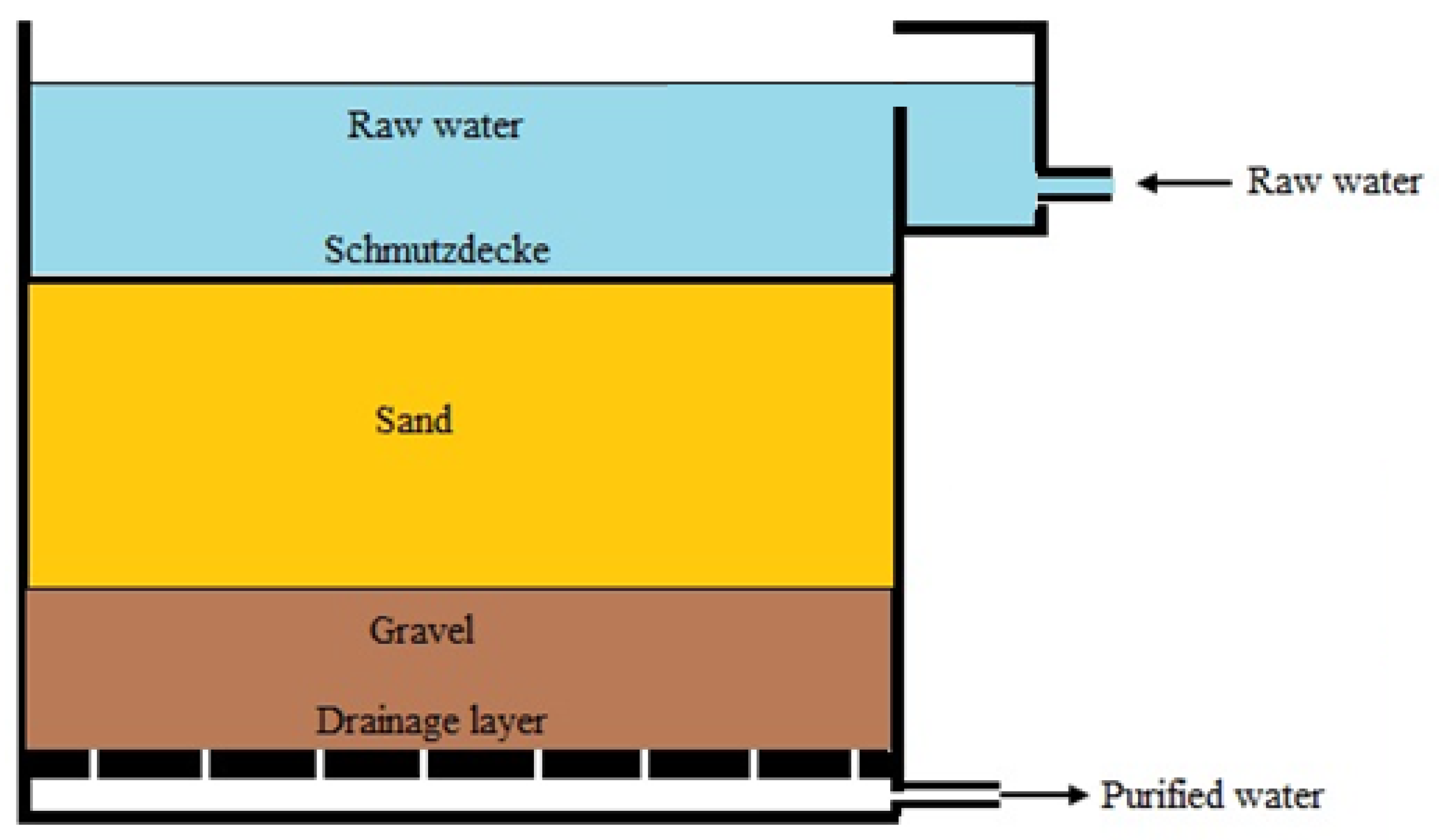
3.4. General Design of SSF
3.5. SSF Regeneration
3.6. SSF Speed Mode
3.7. Influence of Filter Media and Hydraulic Residence Time
3.8. Purification of Water of Ions, Bacteria, and Microbes
3.9. Temperature Effects
3.10. Modifications to the Filter Media
4. Conclusions
5. Future Perspectives
- SSF functions well as bio-filters that are especially important for developing and less-developed countries. However, to further continue to develop methods for SSF, combined use with disinfection techniques for additional water purification from microbiological pollutants such as bacteria, microbes, viruses, and parasites can be performed. These techniques can be applied before or after the SSF application. It is important to develop these techniques using local materials.
- Studies are needed to investigate SSF using other types of basic material in areas where suitable sand and gravel are not readily available. Possible local material may be constituted by inert or semi-inert material from processes such as ash from energy use and biomaterial from agricultural waste.
- It is becoming important, especially for developing and less-developed countries, to test and advance SSF methods to reduce the contents of emerging environmental pollutants such as protozoa, cyanobacteria, surfactants, and microplastics. Thus, further studies are needed to determine design criteria (particle size distribution, depth of media, residence time, temperature, etc.) for different types of pollutants, existing and emerging.
- Further research is needed to advise on life-cycle time, operation (e.g., batch or continuous flow), and maintenance procedures (cleaning of media, backflushing, etc.) for used porous media in SSF. This is especially important in developing countries where intermittent raw water input may affect the function of the SSF due to drought and wet periods.
- Surprisingly few people in developing and less-developed countries still have no access to SSF to obtain safe drinking and household water. This is noteworthy in view of SSF’s simplicity and efficiency in preventing typical WASH diseases. Obviously, SSF has a much larger role to play in helping to reach the UN Sustainable Development Goals. Further research is needed on experiences with SSF in developing countries such as SFF media materials, process removal efficiency, regeneration time, etc.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATL | Atenolol |
| ATZ | Atrazine |
| BSF | Bio-Sand Filters |
| BOD | Biological Oxygen Demand |
| CAWST | Center for Accessible Water Supply and Sanitation Technology |
| Ce | Aqueous antimicrobial concentration mg/L after 24 h equilibration |
| Cs | Sorbed antimicrobial per kilogram of solid, mg/kg |
| C-HSSF | Continuous Household SSF |
| COD | Chemical Oxygen Demand |
| d | Diameter for typical particles in filter media |
| d60 | Diameter at which 60% (by weight) of sand passes through the sieve |
| d10 | Diameter at which 10% (by weight) of sand passes through the sieve |
| foc | Mass fraction of organic carbon matter in the schmutzdecke layer |
| fom | Mass fraction of organic matter in the schmutzdecke layer |
| GO | Graphene Oxide |
| HRT | Hydraulic Residence Time (V × n/Q) |
| HSSF | Household SSF |
| HSSF-CC | Household SSF Continuous Compact |
| HSSF-CT | Household SSF Continuous Traditional |
| HSSF-ID | Household SSF Intermittent Diffusor |
| HSSF-IF | Household SSF Intermittent Float |
| I-HSSF | Intermittent Household SSF |
| K60/10 | Homogeneity coefficient of sand (d60/d10) |
| Kd | Sorption coefficient |
| Koc | Normalized sorption coefficient to the share of organic carbon (Kd/foc) |
| Kom | Normalized sorption coefficient to the share of organic matter (Kd/fom) |
| LIN | Lincomycin |
| MS2 virus | Bacteriophage Emesvirus zinderi |
| n | Sand porosity |
| NTU | Nephelometric Turbidity Unit |
| PCR | Polymerase Chain Reaction |
| POU | Point Of Use |
| PVC | Polyvinyl Chloride |
| Q | Water flow rate (m3/h) |
| SCI | Science Citation Index |
| SMX | Sulfamethoxazole |
| SMZ | Sulfamethazine |
| SODIS | Solar Water Disinfection |
| SSCI | Social Sciences Citation Index |
| SSF | Slow Sand Filtration |
| STP | Sewage Treatment Plant |
| Tams | Triactinomyxon actinospores |
| TOK | Total Organic Carbon |
| TRI | Trimethoprim |
| TYL | Tylosin |
| UASB-RALF | Upflow Anaerobic Sludge Bed Reactor |
| UN SDGs | United Nations Sustainable Development Goals |
| USEPA | U.S. Environmental Protection Agency |
| V | Total sand volume (m3) |
| WASH | Water, sanitation, and hygiene |
| WHO | World Health Organization |
| ZVI | Zero-Valent Iron |
References
- Erazo-Oliveras, A.; May Ol-Bracero, O.L.; Ríos-Dávila, R.A. Improving slow sand filters for water-limited communities. Opflow 2012, 38, 24–27. [Google Scholar] [CrossRef]
- WHO. World Health Report; World Health Organization: Geneva, Switzerland, 2004; Volume 1, pp. 1–540.
- WHO. WHO Guidelines for Drinking Water Quality, 3rd ed.; World Health Organization: Geneva, Switzerland, 2004; p. 143.
- WHO. Preventing Diarrhoea through Better Water, Sanitation and Hygiene: Exposures and Impacts in Low- and Middle-Income Countries; World Health Organization: Geneva, Switzerland, 2014.
- Slezak, L.A.; Sims, R.C. The Application and Effectiveness of slow sand filtration in the United States. J. Am. Water Work. Assoc. 1984, 76, 38. [Google Scholar] [CrossRef]
- Agrawal, A.; Sharma, N.; Sharma, P. Designing an economically slow sand filter for households to improve water quality parameters. Mater. Today Proc. 2021, 43, 1582–1586. [Google Scholar] [CrossRef]
- Elliott, M.A.; Stauber, C.E.; Koksal, F.; DiGiano, F.A.; Sobsey, M.D. Reductions of E-coli, echovirus type 12 and bacteriophages in an intermittently operated household-scale slow sand filter. Water Res. 2008, 42, 2662–2670. [Google Scholar] [CrossRef]
- Haig, S.J.; Collins, G.; Davies, R.L.; Dorea, C.C.; Quince, C. Biological aspects of slow sand filtration: Past, present and future. Water Sci. Technol. Water Supply 2011, 11, 468–472. [Google Scholar] [CrossRef]
- Maiyo, J.K.; Dasika, S.; Jafvert, C.T. Slow Sand Filters for the 21st Century: A Review. Int. J. Environ. Res. Public Health 2023, 20, 1019. [Google Scholar] [CrossRef]
- Barkay-Arbel, Y.; Hatukai, S.; Asheri, T.; Vaizel-Ohayon, D.; Rebhun, M. Performance and process mechanisms of a high-rate direct filtration plant targeting 0.1 ntu. J. Am. Water Work. Assoc. 2012, 104, E653–E663. [Google Scholar] [CrossRef]
- Wen-Yong, W.; Yan, H.; Hong-Lu, L.; Shi-Yang, Y.; Yong, N. Reclaimed Water Filtration Efficiency and Drip Irrigation Emitter Performance with Different Combinations of Sand and Disc Filters. Irrig. Drain. 2015, 64, 362–369. [Google Scholar] [CrossRef]
- Zielina, M. Particle Shapes in the Drinking Water Filtration Process. CLEAN Soil Air Water 2011, 39, 941–946. [Google Scholar] [CrossRef]
- Schraer, A. Amiad Water Systems Filtration Technologies. Water Purif. Water Treat. Water Supply 2021, 4, 36–41. (In Russian) [Google Scholar]
- Lior, N.; El-Nashar, A.; Sommariva, C. Advanced Instrumentation, Measurement, Control, and Automation (IMCA) in Multistage Flash (MSF) and Reverse-Osmosis (RO) Water Desalination. In Advances in Water Desalination; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 453–658. [Google Scholar] [CrossRef]
- Uragami, T. Reverse Osmosis. In Science and Technology of Separation Membranes, 1st ed.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2017; Chapter 9; pp. 259–295. [Google Scholar] [CrossRef]
- Zargar, M.; Jin, B.; Dai, S. Development and application of reverse osmosis for separation. In Membrane Processing for Dairy Ingredient Separation, 1st ed.; Kang, H., James, M., Eds.; Dickson. John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; Chapter 6; pp. 139–175. [Google Scholar] [CrossRef]
- SenGupta, A.K. Ion Exchange Fundamentals. In Ion Exchange in Environmental Processes: Fundamentals, Applications and Sustainable Technology, 1st ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; Chapter 2; pp. 50–129. [Google Scholar] [CrossRef]
- Berrios, M.; Siles, J.A.; Martín, M.A.; Martín, A. Ion Exchange. In Separation and Purification Technologies in Biorefineries, 1st ed.; Ramaswamy, S., Ramarao, B.V., Huang, H.-J., Eds.; Wiley Online Library: Hoboken, NJ, USA, 2013; Chapter 6; pp. 149–156. [Google Scholar] [CrossRef]
- Bornak, B. Desalination by Ion Exchange. In Desalination, 1st ed.; Kucera, J., Ed.; Scrivener Publishing LLC: Beverly, MA, USA, 2014; Chapter 11; pp. 503–520. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, T. Ion Exchange Membranes. In Encyclopedia of Membrane Science and Technology; Eric, M.V., Hoek, V., Tarabara, V., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 1–58. [Google Scholar] [CrossRef]
- Ichihashi, K.; Konno, D.; Maryunina, K.Y.; Inoue, K.; Toyoda, K.; Kawaguchi, S.; Nishihara, S. Selective Ion Exchange in Supramolecular Channels in the Crystalline State. Angew. Chem. Int. Ed. 2019, 58, 4169–4172. [Google Scholar] [CrossRef]
- Baker, R.W. Ion Exchange Membrane Processes—Electrodialysis. In Membrane Technology and Applications, 3rd ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; Chapter 10. [Google Scholar] [CrossRef]
- Conforti, K.M.; Bazant, M.Z. Continuous ion-selective separations by shock electrodialysis. AIChE J. 2019, 66, e16751. [Google Scholar] [CrossRef]
- Zhu, H.; Yang, B.; Gao, C.; Wu, Y. Ion transfer modeling based on Nernst–Planck theory for saline water desalination during electrodialysis process. Asia Pac. J. Chem. Eng. 2020, 15, e2410. [Google Scholar] [CrossRef]
- Gong, F.; Li, H.; Yuan, X.; Huang, J.; Xia, D.; Papavassiliou, V.P.; Xiao, R.; Yamauchi, Y.; Kevin, C.-W.; Wu, K.C.-W.; et al. Recycling Polymeric Solid Wastes for Energy-Efficient Water Purification, Organic Distillation, and Oil Spill Cleanup. Nano Micro Small 2021, 17, 2102459. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Wang, Y.; Zhang, Y.; Song, X.; Peng, H.; Jiang, H. Bilayer rGO-Based Photothermal Evaporator for Efficient Solar-Driven Water Purification. Chem. A Eur. J. 2021, 27, 17428–17436. [Google Scholar] [CrossRef] [PubMed]
- Taraskin, K.A.; Orlov, D.S.; Kanaev, B.A.; Shcherbakov, D.A.; Vorobyov, M.V. Adsorption treatment of wastewater from the production of noise-insulating composite materials from sulfur-containing compounds. Water Purif. Water Treat. Water Supply 2021, 5, 32–38. (In Russian) [Google Scholar]
- Zubkov, A.A.; Bagrov, V.V.; Kamrukov, A.S.; Kostritsa, V.N.; Krylov, V.I. Natural sorbents and their use for wastewater treatment. Water Purif. Water Treat. Water Supply 2020, 2, 36–44. (In Russian) [Google Scholar]
- Korosteleva, Y.A.; Fetyukhina, E.G.; Ignarina, L.M. Sorbent based on diatomite Diamix Aqua is an effective and profitable alternative to carbon filters in power plant water treatment. Water Purif. Water Treat. Water Supply 2020, 6, 44–51. [Google Scholar]
- Yap, P.L.; Nine, M.J.; Hassan, K.; Tung, T.T.; Tran, D.N.H.; Losic, D. Graphene-Based Sorbents for Multipollutants Removal in Water: A Review of Recent Progress. Adv. Funct. Mater. 2020, 31, 2007356. [Google Scholar] [CrossRef]
- Liu, J.; Yang, H.; Liu, K.; Miao, R.; Fang, Y. Gel-Emulsion Templated Polymeric Aerogels for Water Treatment through Organic Liquid Removing and Solar Vapor Generation. ChemSusChem 2019, 13, 749–755. [Google Scholar] [CrossRef]
- Eremeev, D.N.; Voropaev, S.V. The use of cationic flocculants to improve the efficiency of dehydration of urban sewage sludge. Water Purif. Water Treat. Water Supply 2021, 3, 40–45. (In Russian) [Google Scholar]
- Abdiyev, K.Z.; Maric, M.; Orynbayev, B.Y.; Toktarbay, Z.; Zhursumbaeva, M.B.; Seitkaliyeva, N.Z. Flocculating properties of 2-acrylamido-2-methyl-1-propane sulfonic acid-co-allylamine polyampholytic copolymers. Polym. Bull. 2022, 79, 10741–10756. [Google Scholar] [CrossRef]
- Shachneva, E.Y.; Khentov, V.Y.; Kudinova, D.E. Removal of zinc ions using H-600 flocculant. Water Purif. Water Treat. Water Supply 2020, 4, 20–27. (In Russian) [Google Scholar]
- Dauletov, Y.; Abdiyev, K.Z.; Toktarbay, Z.; Nuraje, N.; Zhursumbaeva, M.; Kenzhaliyev, B. Radical Polymerization and Kinetics of N,N-diallyl-N,N-dimethylammonium Chloride and Vinyl Ether of Monoethanolamine. Fibers Polym. 2018, 19, 2023–2029. [Google Scholar] [CrossRef]
- Courtney, M.; Weijue, G.; Pedram, F. Cationic lignin polymers as flocculants for municipal wastewater. Water Environ. J. 2023, 37, 95–102. [Google Scholar]
- Xiwen, L.; Qiaoxia, G.; Shenyong, R.; Junkang, G.; Chongbin, W.; Jiaxin, C.; Baojian, S. Synthesis of starch-based flocculant by multi-component grafting copolymerization and its application in oily wastewater treatment. J. Appl. Polym. Sci. 2023, 140, e53356. [Google Scholar]
- Bouras, B.; Tennouga, H. Flocculation of Clay Suspensions Using Copolymers Based on Acrylamide and Biopolymer. Phys. Chem. Res. 2023, 11, 221–230. [Google Scholar]
- Abdiyev, K.Z.; Maric, M.; Orynbayev, B.; Zhursumbaeva, M.; Seitkaliyeva, N.; Toktarbay, Z. Novel Cationic Polymer Surfactant for Regulation of the Rheological and Biocidal Properties of the Water-Based Drilling Muds. Polymers 2023, 15, 330. [Google Scholar] [CrossRef]
- Shaikhutdinov, E.M.; Khussain, S.K.; Abdiyev, K.Z.; Seitkaliyeva, N.Z. Complexation of Sodium 2-Acrylamido-2-Methylpropanesulfonate-Monoethanolamine Vinyl Ether Copolymer with Polyelectrolytes in Aqueous Medium. Polym. Sci. Ser. A 2007, 49, 584–592. [Google Scholar] [CrossRef]
- Abdiyev, K.Z.; Shaikhutdinov, E.M.; Zhursumbaeva, M.B.; Khussain, S.K. Effect of polymer concentration on the surface properties of polyacid-poly(N-vinylpyrrolidone) complexes. Colloid J. 2003, 65, 399–402. [Google Scholar] [CrossRef]
- Shanahan, P. Water and Wastewater Treatment Engineering. Massachusetts Institute of Technology: MIT OpenCourseWare. 2006. Available online: https://ocw.mit.edu (accessed on 3 January 2023).
- Brief History during the Snow Era. Available online: https://www.ph.ucla.edu/epi/snow/1859map/Chelsea_waterworks_a2.html (accessed on 24 February 2023).
- Christman, K. The history of chlorine. Waterworld 1998, 14, 66–67. [Google Scholar]
- Huisman, L.; Wood, W.E. Slow Sand Filtration; World Health Organization: Geneva, Switzerland, 1974. Available online: https://apps.who.int/iris/bitstream/handle/10665/38974/9241540370.pdf?sequence=1&isAllowed=y (accessed on 23 January 2023).
- Buchan, J. Crowed with Genius: The Scottish Enlightenment: Edinburgh’s Moment of the Mind; Harper Collins: New York, NY, USA, 2003. [Google Scholar]
- Crittenden, J.; Trussell, R.R.; Hand, D.W.; Howe, K.; Tchobanoglous, G. Water Treatment: Principles and Design, 2nd ed.; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Johnson, G. Present Day Water Filtration Practice. J. Am. Water Work. Assoc. 1914, 1, 31–80. [Google Scholar] [CrossRef]
- National Drinking Water Clearinghouse. Slow sand filtration. Municipal Water Supply. In Water Encyclopedia; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar] [CrossRef]
- History|Poughkeepsies Water Treatment Facility. Retrieved 18 May 2017. Available online: www.pokwater.com (accessed on 13 February 2023).
- Lacey, M. Filtration to privatization, utility issues are universal. J. Am. Water Work. Assoc. 2009, 101, 2. [Google Scholar] [CrossRef]
- Manz, D. New horizons for slow sand filtration. In Proceedings of the Eleventh Canadian National Conference and Second Policy Forum on Drinking Water and the Biennial Conference of the Federal-Provincial-Territorial Committee on Drinking Water, Promoting Public Health through Safe Drinking Water, Calgary, AB, Canada, 3–6 April 2004; pp. 682–692. [Google Scholar]
- Gunn, S.; William, A.; Masellis, M. Concepts and Practice of Humanitarian Medicine. 23 October 2007. Available online: https://books.google.com/books?id=t1exE1cfKXIC&pg=PA87 (accessed on 19 January 2023).
- Wegelin, M. Roughing gravel filters for suspended solids removal. In Slow Sand Filtration: Recent Developments in Water Treatment Technology; Graham, N.J.D., Ed.; Ellis Horwood Ltd.: England, UK, 1988; p. 86. [Google Scholar]
- Logsdon, G.S.; Lippy, E.C. The role of filtration in preventing waterborne disease. J. Am. Water Work. Assoc. 1982, 74, 649–655. [Google Scholar] [CrossRef]
- Collins, M.R. Experiences introducing “new” technology: Slow sand filtration. In Providing Safe Drinking Water in Small Systems: Technology, Operations, and Economics; Cotruvo, J.A., Craun, G.F., Hearn, N., Eds.; CRC Press: Boca Raton, FL, USA, 1998; pp. 213–224. [Google Scholar]
- Letterman, R.D.; Cullen, T.R. Slow Sand Filter Maintenance: Costs and Effects on Water Quality; Report 600/S2-85/056; U.S. Environmental Protection Agency: Washington, DC, USA, 1985.
- Al-Ani, M.; McElroy, J.M.; Hibler, C.P.; Hendricks, D.W. Filtration of Giardia Cysts and Other Substances: Volume 3, Rapid-Rate Filtration; Report 600/S2-85/027; U.S. Environmental Protection Agency: Washington, DC, USA, 1985.
- Droste, R.L. The Theory and Practice of Water and Wastewater Treatment; Wiley: New York, NY, USA, 1997. [Google Scholar]
- Arndt, R.; Wagner, E. Rapid and slow sand filtration Techniques and Their Efficacy at Filtering Triactinomyxons of Myxobolus cerebralis from Contaminated Water. N. Am. J. Aquac. 2004, 66, 261–270. [Google Scholar] [CrossRef]
- Bellamy, W.D.; Silverman, G.P.; Hendricks, D.W.; Logsdon, G.S. Removing Giardia cysts with slow sand filtration. J. Am. Water Work. Assoc. 1985, 77, 52–60. [Google Scholar] [CrossRef]
- Frederick, W. Drinking Water Regulation and Health, 1st ed.; Pontius, Wiley and Science: New York, NY, USA, 2003; ISBN 13: 978-0471415541/10: 0471415545. [Google Scholar]
- Yildiz, B.S. Water and wastewater treatment: Biological processes. In Metropolitan Sustainability; Woodhead Publishing Limited: Cambridge, UK, 2012; pp. 406–428. [Google Scholar]
- Lubarsky, H.; Fava, N.D.M.N.; Souza Freitas, B.L.; Terin, U.C.; Oliveira, M.; Lamon, A.W.; Pichel, N.; Byrne, J.A.; Sabogal-Paz, L.P.; Fernandez-Ibañez, P. Biological Layer in Household Slow Sand Filters: Characterization and Evaluation of the Impact on Systems Efficiency. Water 2022, 14, 1078. [Google Scholar] [CrossRef]
- Islam, M.M.M.; Iqbal, M.S.; D’Souza, N.; Islam, M.A. A review on present and future microbial surface water quality worldwide. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100523. [Google Scholar] [CrossRef]
- Liang, L.; Goh, S.G.; Vergara, G.G.R.V.; Fang, H.M.; Rezaeinejad, S.; Chang, S.Y.; Bayen, S.; Lee, W.A.; Sobsey, M.D.; Rose, J.B.; et al. Alternative fecal indicators and their empirical relationships with Enteric Viruses, Salmonella enterica, and Pseudomonas aeruginosa in surface waters of a tropical urban catchment. Appl. Environ. Microbiol. 2015, 81, 850–860. [Google Scholar] [CrossRef]
- Clark, P.A.; Pinedo, C.A.; Fadus, M.; Capuzzi, S. Slow-sand water filter: Design, implementation, accessibility and sustainability in developing countries. Med. Sci. Monit. 2012, 18, RA105–RA117. [Google Scholar] [CrossRef]
- Visscher, J.T. Slow sand filtration: Design, Operation, and Maintenance. J. Am. Water Work. Assoc. 1990, 82, 67–71. [Google Scholar] [CrossRef]
- Logsdon, G.S.; Sorg, T.J.; Clark, R.M. Capability and Cost of Treatment Technologies for Small Systems. J. Am. Water Work. Assoc. 1990, 82, 60–66. [Google Scholar] [CrossRef]
- Verma, S.; Daverey, A.; Sharma, A. Slow sand filtration for water and wastewater treatment—A review. Environ. Technol. Rev. 2017, 6, 47–58. [Google Scholar] [CrossRef]
- Logsdon, G.S.; Kohne, R.; Abel, S.; LaBonde, S. Slow sand filtration for small water systems. J. Environ. Eng. Sci. 2002, 68, 100–108. [Google Scholar] [CrossRef]
- Cleary, S. Sustainable Drinking Water Treatment for Small Communities using Multistage Slow Sand Filtration. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2005. [Google Scholar]
- Gottinger, A.M.; McMartin, D.W.; Price, D.; Hanson, B. The effectiveness of slow sand filters to treat Canadian rural prairie water. Can. J. Civ. Eng. 2011, 38, 455–463. [Google Scholar] [CrossRef]
- Collins, M.R.; Eighmy, T.T.; Malley, J.P. Evaluating Modifications to Slow Sand Filters. J. Am. Water Work. Assoc. 1991, 83, 62–70. [Google Scholar] [CrossRef]
- Guchi, E. Review on slow sand filtration in removing microbial contamination and particles from drinking water. Research 2015, 3, 47–55. [Google Scholar] [CrossRef]
- Weber-Shirk, M.; Dick, R. Physical–chemical mechanisms in slow sand filters. J. Am. Water Work. Assoc. 1997, 89, 87–100. [Google Scholar] [CrossRef]
- Montgomery, J.M. Water Treatment: Principles and Design; John Wiley & Sons: Hoboken, NJ, USA, 1985; pp. 1–432. [Google Scholar]
- Galvis, G.; Latorre, J.; Visscher, J.T. Multi-Stage Filtration: An Innovative Water Treatment Technology; TP Series; IRC International Water and Sanitation Center: The Hague, The Netherlands, 1998; Volume 34. [Google Scholar]
- Mc Connell, L.J. Evaluation of the Slow Rate Sand Filtration Process for Treatment of Drinking Water Containing Virus and Bacteria. Master’s Thesis, Utah State University, Logan, UT, USA, 1984. [Google Scholar]
- Ellis, K.V.; Wood, W.E. Slow sand filtration. CRC Crit. Rev. Environ. Control. 1985, 15, 315–354. [Google Scholar] [CrossRef]
- Van Dijk, J.C.; Ooman, J.H.C. Slow Sand Filtration for Community Water Supply in Developing Countries: A Design and Construction manual; WHO International Reference Center for Community Water Supply: The Hague, Netherlands, 1978; Chapter 4. [Google Scholar]
- Troyan, J.J.; Hansen, S.P. Treatment of Microbial Contaminants in Potable Water Supplies; Noyes Data Corporation: Park Ridge, IL, USA, 1989; pp. 5–54. [Google Scholar]
- Yahya, M.T.; Cluff, C.B.; Gerba, C.P. Virus removal by slow sand filtration and nanofiltration. Water Sci. Technol. 1993, 27, 445–448. [Google Scholar] [CrossRef]
- Cullen, T.R.; Letterman, R.D. The Effect of Slow Sand Filter Maintenance on Water Quality. J. Am. Water Work. Assoc. 1985, 77, 48–55. [Google Scholar] [CrossRef]
- Campos, L.C.; Su, M.F.; Graham, N.J.; Smith, S.R. Biomass development in slow sand filters. Water Res. 2002, 18, 4543–4551. [Google Scholar] [CrossRef]
- Poynter, S.F.B.; Slade, J.S. The removal of viruses by slow sand filtration. Prog. Water Technol. 1977, 9, 75–88. [Google Scholar]
- Bellamy, W.D.; Silverman, G.P.; Hendricks, D.W. Filtration of Giardia Cysts and Other Substances: Volume 2, Slow Sand Filtration; U.S. Environmental Protection Agency, Water Engineering Research Laboratory: Cincinnati, OH, USA, 1985. [Google Scholar]
- Slow Sand Filtration; Tech Brief Fourteen; National Drinking Water Clearinghouse (U.S.): Morgantown, WV, USA, 2000.
- Eighmy, T.T.; Collins, M.R. Modifications to the slow rate Filtration Process for Improved Trihalometane precursor Removal. In Slow Sand Filtration: Recent Developments in Water Treatment Technology; Graham, N.J.D., Ed.; Ellis Horwood Ltd.: England, UK, 1988; pp. 1–97. [Google Scholar]
- Edzwald, J.K. American Water Works Association. Water Quality & Treatment, a Handbook on Drinking Water, 6th ed.; McGraw-Hill: New York, NY, USA, 2010. [Google Scholar]
- Biosand Filter Manual: Design, Construction, & Installation; Centre for Affordable Water and Sanitation Technology: Calgary, AB, Canada, 2007.
- Haarhoff, J.; Cleasby, J.L. Biological and Physical Mechanisms in Slow Sand Filtration; Logsdon, G.S., Ed.; SSF American Society of Civil Engineers: New York, NY, USA, 1991; pp. 19–68. [Google Scholar]
- Weber-Shirk, M.; Dick, R. Biological mechanisms in slow sand filters. J. Am. Water Work. Assoc. 1997, 89, 72–83. [Google Scholar] [CrossRef]
- Weber-Shirk, M.; Dick, R. Bacterivory by a chrysophyte in slow sand filters. Water Res. 1999, 33, 631–638. [Google Scholar] [CrossRef]
- Bauer, R.; Dizer, H.; Graeber, I.; Rosenwinkel, K.-H.; López-Pilaa, J.M. Removal of bacterial fecal indicators, coliphages and enteric adenoviruses from waters with high fecal pollution by slow sand filtration. Water Res. 2011, 45, 439–452. [Google Scholar] [CrossRef]
- Kennedy, T.J.; Anderson, T.A.; Hernandez, E.A.; Morse, A.N. Determining the operational limits of the biosand filter. Water Sci. Technol.-Water Supply 2013, 13, 56–65. [Google Scholar] [CrossRef]
- Fewster, E.; Mol, A.; Wiessent-Brandsma, C. The Bio-sand Filter. Long term sustainability: User habits and technical performance evaluated. In Proceedings of the Presentation Given at the 2003 International Symposium on Household Technologies for Safe Water, Nairobi, Kenya, 16–17 June 2004. [Google Scholar]
- Mahmood, Q.; Baig, S.A.; Nawab, B.; Shafqat, M.N.; Pervez, A.; Zeb, B.S. Development of low cost household drinking water treatment system for the earthquake affected communities in Northern Pakistan. Desalination 2011, 273, 316–320. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (EPA). Technologies for Upgrading Existing or Designing New Drinking Water Treatment Facilities. Document No. EPA/625/4-89/023. 1990. Available online: https://nepis.epa.gov/Exe/ZyNET.exe/300048WU.TXT (accessed on 2 March 2023).
- HDR Engineering. Handbook of Public Water Systems; John Wiley and Sons: New York, NY, USA, 2001; p. 353. ISBN 978-0-471-29211-1. Available online: https://www.wiley.com/en-ie/Handbook+of+Public+Water+Systems,+2nd+Edition-p-9780471292111 (accessed on 28 March 2023).
- Bellamy, W.D.; Hendricks, D.W.; Logsdon, G.S. Slow Sand Filtration: Influences of Selected Process Variables. J. Am. Water Work. Assoc. 1985, 77, 62–66. [Google Scholar] [CrossRef]
- Burman, N.P. Biological control of slow sand filtration. Effl. Water Treat. J. 1962, 2, 674. [Google Scholar]
- Weber-Shirk, M.L. Enhancing slow sand filter performance with an Acid- Soluble Seston Extract. Water Res. 2002, 36, 4753–4756. [Google Scholar] [CrossRef] [PubMed]
- Rooklidge, S.J.; Miner, J.R.; Kassim, T.A.; Nelson, P.O. Antimicrobial contaminant removal by multistage slow sand filtration. J. Am. Water Work. Assoc. 2005, 97, 92–100. [Google Scholar] [CrossRef]
- Lesikar, B. Sand Filters for Home Use—Texas Agricultural Extension Service. Scribd. Available online: http://www.scribd.com/doc/34621075/Sand-filters-for-home-use-Texas-Agricultural-Extension-Service (accessed on 25 January 2023).
- Logan, A.J.; Stevik, T.K.; Siegrist, R.L.; Rønn, R.M. Transport and fate of Cryptosporidium parvum oocysts in intermittent sand filters. Water Res. 2001, 35, 4359–4369. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.G. A study of bacteria reduction by slow sand filtration. In Paper Presented at the 1987 IWPC Biennial Conference, Port Elizabeth, South Africa, 12–15 May 1987; National Institute for water Research: Pretoria, South Africa, 1987. [Google Scholar]
- Peterson, H.; Corkal, D. Biological Treatment of Ground Water. Prairie Farm Rehabilitation Administration. Water Quality Matters. Available online: http://www5.agr.gc.ca/resources/prod/doc/pfra/pdf/bio_treat_groundwater_e.pdf (accessed on 25 January 2023).
- Peterson, H.; Broley, T.; Sketchell, J.; Corkal, D. ADD Board 15 and 43 Project Report: Biological Treatment of Ground Water; Publication No. R-1640-6-E-97; Saskatchewan Research Council: Saskatoon, SK, Canada, 1997. [Google Scholar]
- Kennedy, T.J.; Hernandez, E.A.; Morse, A.N.; Anderson, T.A. Hydraulic Loading Rate Effect on Removal Rates in a BioSand Filter: A Pilot Study of Three Conditions. Water Air Soil Pollut. 2012, 223, 4527–4537. [Google Scholar] [CrossRef]
- Bradley, I.; Straub, A.; Maraccini, P.; Markazi, S.; Nguyen, T.H. Iron oxide amended biosand filters for virus removal. Water Res. 2011, 45, 4501–4510. [Google Scholar] [CrossRef]
- Hussain, G.; Haydar, S.; Bari, A.J.; Aziz, J.A.; Anis, M.; Asif, Z. Evaluation of Plastic Household Biosand Filter (BSF) In Combination with Solar Disinfection (SODIS) For Water Treatment. J. Chem. Soc. Pak. 2015, 37, 352–362. [Google Scholar]
- Nitzsche, K.S.; Weigold, P.; Losekann-Behrens, T.; Kappler, A.; Behrens, S. Microbial community composition of a household sand filter used for arsenic, iron, and manganese removal from groundwater in Vietnam. Chemosphere 2015, 138, 47–59. [Google Scholar] [CrossRef]
- Lukasheva, G.N.; Yurovsky, A.V. Slow self-cleaning filter for deferrization of natural waters. Bull. Assoc. Univ. Tour. Serv. 2010, 4, 56–63. (In Russian) [Google Scholar]
- Yurovsky, A.V.; Lukasheva, G.N. Study of the efficiency of a slow self-cleaning filter. In Materials of the All-Russian Scientific Conference of Graduate Students and Young Scientists “Modern Problems of Tourism and Service”; FGSHUVPO “RGUTiS”: Moscow, Russia, 2010; pp. 270–274. (In Russian) [Google Scholar]
- Yurovsky, A.V.; Lukasheva, G.N. Formation of a chemisorption layer of the filter bed of a slow self-cleaning iron removal filter. In Materials of the All-Russian Scientific Conference of Graduate Students and Young Scientists “Modern Problems of Tourism and Service”; FGSHUVPO “RGUTiS”: Moscow, Russia, 2010; pp. 274–278. (In Russian) [Google Scholar]
- Sultanov, F.; Daulbayev, C.; Azat, S.; Kuterbekov, K.; Bekmyrza, K.; Bakbolat, B.; Bigaj, M.; Mansurov, Z. Influence of Metal Oxide Particles on Bandgap of 1D Photocatalysts Based on SrTiO3/PAN Fibers. Nanomaterials 2020, 10, 1734. [Google Scholar] [CrossRef]
- Rakhmanin, Y.A.; Cheskis, A.B. Drinking Water. Quality Standards. Handbook; VINITI: Moscow, Russia, 1993; pp. 1–7, 19, 26, 46. [Google Scholar]
- Grützmacher, G.; Böttcher, G.; Chorus, I.; Bartel, H. Removal of microcystins by slow sand filtration. Environ. Toxicol. 2002, 17, 386–394. [Google Scholar] [CrossRef]
- Onan, L.; LaPara, T. Tylosin-resistant Bacteria Cultivated from Agricultural Soil. FEMS Toxicol. Lett. 2003, 220, 15–24. [Google Scholar] [CrossRef] [PubMed]
- De Liguoro, M.; Cibin, V.; Capolongo, F.; Halling-Sørensen, B.; Montesissa, C. Use of oxytetracycline and tylosin in intensive calf farming: Evaluation of transfer to manure and soil. Chemosphere 2003, 52, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Inglezakis, V.J.; Azat, S.; Tauanov, Z.; Mikhalovsky, S.V. Functionalization of biosourced silica and surface reactions with mercury in aqueous solutions. Chem. Eng. J. 2021, 423, 129745. [Google Scholar] [CrossRef]
- Pachocka, M. Intermittent Slow Sand Filters: Improving their Design for Developing World Applications. Master’s Thesis, University of Delaware, Newark, DE, USA, 2010. [Google Scholar]
- Partinoudi, V.; Collins, M.R.; Dwyer, P.L.; Martin-Doole, M. Assessing Temperature Influences on Slow Sand Filtration Treatment Performance. Project Summary, New England Water Treatment Technology Assistance Center, Univ. of New Hampshire. 2007. Available online: http://www.unh.edu/wttac/Project_Summaries/assessing_temperature_slow_sand.pdf (accessed on 14 January 2023).
- Fogel, D.; Isaac-Renton, J.; Guasparini, R.; Moorehead, W.; Ongerth, J. Removing giardia and cryptosporidium by slow sand filtration. J. Am. Water Work. Assoc. 1993, 85, 77–84. [Google Scholar] [CrossRef]
- Liu, J.; Cao, X.; Meng, X. Effects of temperature on performances of a slow sand filter used for advanced wastewater treatment. Chin. J. Environ. Eng. 2010, 4, 2437–2440. [Google Scholar]
- Schijven, J.F.; van den Berg, H.H.J.L.; Colin, M.; Dullemont, Y.; Hijnen, W.A.M.; Magic-Knezev, A.; Oorthuizen, W.A.; Wubbels, G. A mathematical model for removal of human pathogenic viruses and bacteria by slow sand filtration under variable operational conditions. Water Res. 2013, 47, 2592–2602. [Google Scholar] [CrossRef]
- Jabur, H.S.; Gimbel, R.; Graham, N.J.D.; Collins, M.R. The Effect of Water Temperature on the Slow Sand Filter Process. Recent Progress in Slow Sand and Alternative Biofiltration Processes; IWA: London, UK, 2006; Volume 5, p. 582. [Google Scholar]
- Unger, M.; Collins, M.R. Assessing Escherichia coli removal in the schmutzdecke of slow-rate biofilters. J. Am. Water Works Ass. 2008, 100, 60–73. [Google Scholar] [CrossRef]
- Welte, B.; Montiel, A. Removal of BDOC by Slow Sand Filtration: Comparison with granular activated carbon and effect of temperature. In Advances in Slow Sand and Alternative Biological Filtration; Graham, N., Collins, R., Eds.; John Wiley & Sons Ltd.: England, UK, 1996; p. 60. [Google Scholar]
- Fitriani, N.; Wahyudianto, F.E.; Salsabila, N.F.; Mohamed, R.M.S.R.; Kurniawan, S.B. Performance of modified slow sand filter to reduce turbidity, total suspended solids, and iron in river water as water treatment in disaster areas. J. Ecol. Eng. 2023, 24, 1–18. [Google Scholar] [CrossRef]
- Escola Casas, M.; Guivernau, M.; Vinas, M.; Fernandez, B.; Caceres, R.; Biel, C.; Matamoros, V. Use of wood and cork in biofilters for the simultaneous removal of nitrates and pesticides from groundwater. Chemosphere 2023, 313, 137502. [Google Scholar] [CrossRef]
- Holmes, E.B.; Oza, H.H.; Bailey, E.S.; Sobsey, M.D. Evaluation of chitosans as coagulants—Flocculants to improve sand filtration for drinking water treatment. Inter. J. Mol. Sci. 2023, 24, 1295. [Google Scholar] [CrossRef]
- Liu, H.-L.; Li, X.; Li, N. Application of bio-slow sand filters for drinking water production: Linking purification performance to bacterial community and metabolic functions. J. Water Process Eng. 2023, 53, 103622. [Google Scholar] [CrossRef]
- Fava, N.M.N.; Terin, U.C.; Freitas, B.L.S.; Sabogal-Paz, L.P.; Fernandez-Ibanez, P.; Byrne, J.A. Household slow sand filters in continuous and intermittent flows and their efficiency in microorganism’s removal from river water. Environ. Technol. 2022, 43, 1583–1592. [Google Scholar] [CrossRef] [PubMed]
- Lamon, A.W.; Faria Maciel, P.M.; Campos, J.R.; Corbi, J.J.; Dunlop, P.S.M.; Fernandez-Ibanez, P.; Byrne, J.A.; Sabogal-Paz, L.P. Household slow sand filter efficiency with schmutzdecke evaluation by microsensors. Environ. Technol. 2022, 43, 4042–4053. [Google Scholar] [CrossRef]
- Vu, C.T.; Wu, T. Enhanced slow sand filtration for the removal of micropollutants from groundwater. Sci. Total Environ. 2022, 809, 152161. [Google Scholar] [CrossRef]
- Zhang, M.; He, L.; Zhang, X.; Wang, S.; Zhang, B.; Hsieh, L.; Yang, K.; Tong, M. Improved removal performance of Gram-negative and Gram-positive bacteria in sand filtration system with arginine modified biochar amendment. Water Res. 2022, 211, 118006. [Google Scholar] [CrossRef]
- Rosa e Silva, G.O.; Loureiro, H.O.; Soares, L.G.; de Andrade, L.H.; Santos, R.G.L. Evaluation of an alternative household water treatment system based on slow filtration and solar disinfection. J. Water Health 2022, 20, 157–166. [Google Scholar] [CrossRef]
- Ribeiro, M.P.; Botari, A. Evaluation of effluent post-treatment by slow filtration and adsorption with activated carbon produced from spent coffee grounds in surfactant removal in sewage treatment. Rev. Ambiente Agua 2022, 17, E2756. [Google Scholar] [CrossRef]
- Mohammed, M.O.A.; Solumon, A.A.M. Two models of household sand filters for small scale water purification. Pol. J. Environ. Stud. 2022, 31, 2737–2748. [Google Scholar] [CrossRef]
| Category | Group/Name | Size (μm) |
|---|---|---|
| Mineral | Clays (colloidal) | 0.001–1 |
| Silicates | No data | |
| Non-Silicates | No data | |
| Biological | Viruses | 0.001–0.1 |
| Bacteria | 0.3–10 | |
| Algae, unicellular | 30–50 | |
| Giardia cysts | 10 | |
| Parasite eggs | 10–50 | |
| Nematode eggs | 10 | |
| Cryptosporidium oocysts | 4–5 | |
| Other particles | Amorphous debris, small | 1–5 |
| Organic colloids | No data |
| Temperature Change | Treatment Efficiency | Reference |
|---|---|---|
| Decrease from 20 °C to <4 °C | Microcystins were eliminated >85%, decreasing to <60% due to slowing down of bacterial biodegradation at low temperature. | [119] |
| Decrease from 21 °C to 5.5 °C | Higher temperature had 2.5 times more efficient microbial removal rates for Bacillus spores and E. coli due to biological respiration. | [124] |
| Decrease from 16–18 °C to 5–8 °C | Virus removal was reduced from an average of 99.997% to 99.68%. Bacteriophages appeared not to be significantly affected. Coliform bacteria removal decreased from >99.5% to 97.6% while E. coli concentration increased from >88.0% to >94.6%. | [86] |
| Decrease from about 20 °C to 0.5 °C | Findings suggested that Cryptosporidium may not be adequately removed from a contaminated source water under very cold operating conditions or if the filtration plant does not comply with accepted design standards. | [125] |
| Decrease from 17 °C to 2 °C | Giardia was not affected, while coliform bacteria increased 100 times. | [87] |
| Decrease from 23–25 °C to 10–14 °C | Removal rates of turbidity, COD, color, and total bacterial counts decreased by 12.5%, 26.5%, 22.9%, and 5.8% (advanced wastewater treatment). | [126] |
| Decrease from 17 °C to 5(2) °C | Removal of Total coliform bacteria decreased from 97% to 87%. Standard plate bacteria increased 100 times. | [101] |
| Decrease from 19.5 °C to 4 °C | Experiments and modeling showed that removal of microorganisms (Bacteriophage, Escherichia coli) is most sensitive to changes in temperature and age of the schmutzdecke. Change in filtration rate had small effect on microorganism removal. | [127] |
| Decrease from 14 °C to 2 °C | Temperature has effect on pH, BOD, COD, and TOC removal by about 50% decrease. | [128] |
| Decrease from 24 °C to 8 °C | E. coli Log removal rate decreased from 2.2–2.5 to 1.6–1.7. | [129] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdiyev, K.; Azat, S.; Kuldeyev, E.; Ybyraiymkul, D.; Kabdrakhmanova, S.; Berndtsson, R.; Khalkhabai, B.; Kabdrakhmanova, A.; Sultakhan, S. Review of Slow Sand Filtration for Raw Water Treatment with Potential Application in Less-Developed Countries. Water 2023, 15, 2007. https://doi.org/10.3390/w15112007
Abdiyev K, Azat S, Kuldeyev E, Ybyraiymkul D, Kabdrakhmanova S, Berndtsson R, Khalkhabai B, Kabdrakhmanova A, Sultakhan S. Review of Slow Sand Filtration for Raw Water Treatment with Potential Application in Less-Developed Countries. Water. 2023; 15(11):2007. https://doi.org/10.3390/w15112007
Chicago/Turabian StyleAbdiyev, Kaldibek, Seitkhan Azat, Erzhan Kuldeyev, Darkhan Ybyraiymkul, Sana Kabdrakhmanova, Ronny Berndtsson, Bostandyk Khalkhabai, Ainur Kabdrakhmanova, and Shynggyskhan Sultakhan. 2023. "Review of Slow Sand Filtration for Raw Water Treatment with Potential Application in Less-Developed Countries" Water 15, no. 11: 2007. https://doi.org/10.3390/w15112007
APA StyleAbdiyev, K., Azat, S., Kuldeyev, E., Ybyraiymkul, D., Kabdrakhmanova, S., Berndtsson, R., Khalkhabai, B., Kabdrakhmanova, A., & Sultakhan, S. (2023). Review of Slow Sand Filtration for Raw Water Treatment with Potential Application in Less-Developed Countries. Water, 15(11), 2007. https://doi.org/10.3390/w15112007









