Oil Sands Wetland Ecosystem Monitoring Program Indicators in Alberta, Canada: Transitioning from Pilot to Long-Term Monitoring
Abstract
:1. Introduction
1.1. OSM Wetlands Program History
1.2. Objectives
- Identify high-level pathway–state relationships from oil sands development pressures;
- Present pilot program monitoring data and preliminary high-level wetland indicator observations;
- Assess wetland indicator abilities to measure changes in the ecosystem “state”, relating select indicator data to oil sands development pressures where appropriate;
- Discuss the limitations of the pilot study, proposed improvements, and recommendations for long-term monitoring efforts.
2. Materials and Methods
2.1. Study Site
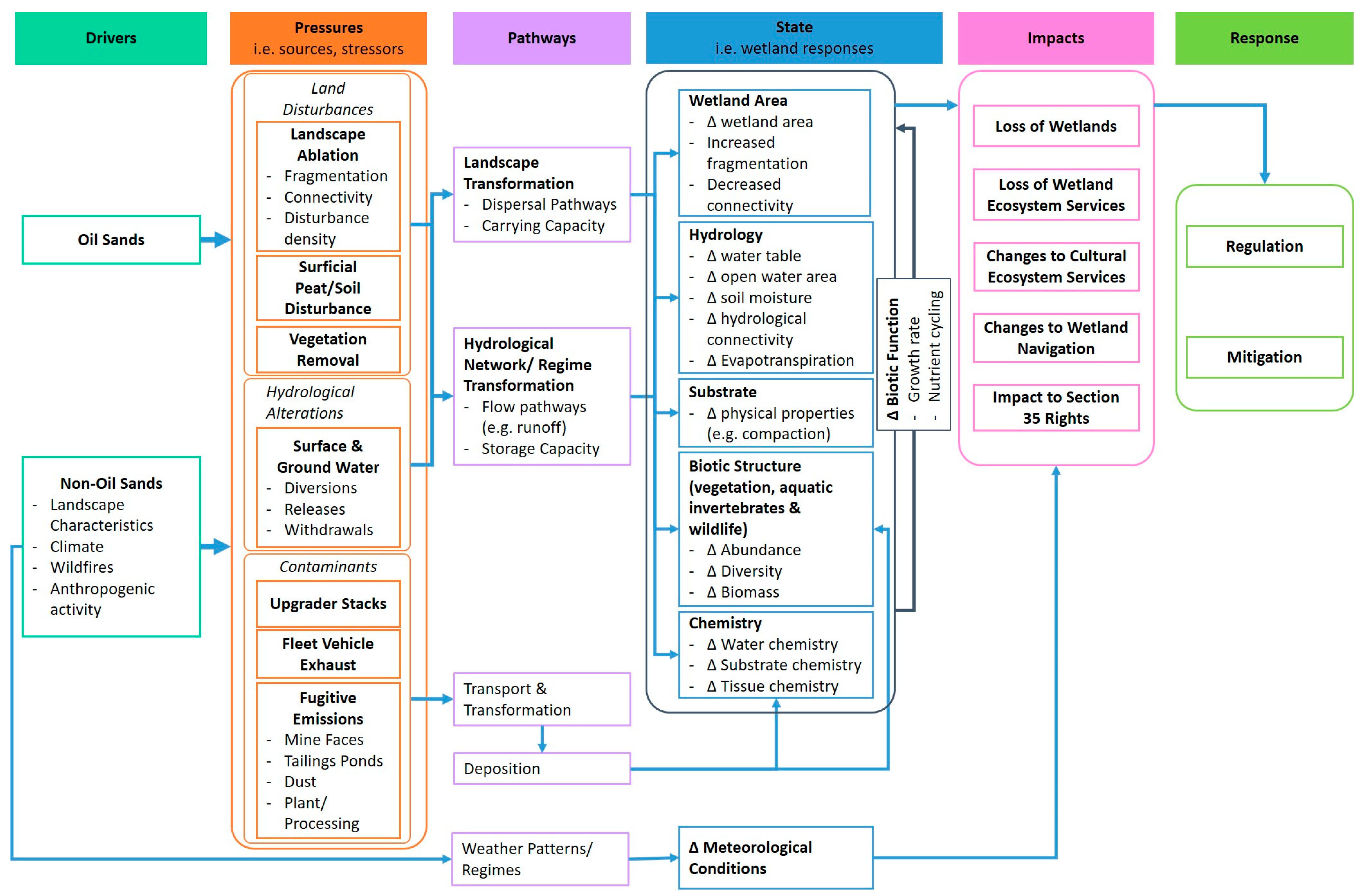
2.2. Wetland Conceptual Model and Indicators
2.3. Data Analysis
2.3.1. Hydrometeorological Data
2.3.2. Water Quality
2.3.3. Benthic Invertebrates
2.3.4. Wildlife
2.3.5. Vegetation
3. Results
3.1. Wetland Conceptual Model and Indicators
3.2. Hydrometeorological Data
3.3. Water Quality
3.4. Benthic Invertebrates
3.5. Wildlife
3.6. Vegetation
4. Discussion
4.1. Addressing OSM Program Objectives
4.2. Wetland Indicators: Implications and Long-Term Monitoring Potential
4.2.1. Hydrometeorological Data
4.2.2. Water Quality
4.2.3. Benthic Invertebrates
4.2.4. Wildlife
4.2.5. Vegetation
4.3. Defining Baseline Conditions and Assessing Variability
4.4. Scaling up Monitoring Data—Remote Sensing Strategy
5. Conclusions and Recommendations
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Name | Location | Elev. (m) | Description |
|---|---|---|---|
| ANZ01 (Bog) | 56.469, −111.043 | 469 | Black spruce-dominant with bog rosemary and cottongrass understory. Adjacent to train tracks in recently burned forest. |
| AOS01 (SOW) | 56.939, −111.662 | 326 | Bog birch-dominant with sedge understory. Adjacent to road and pipeline corridor. |
| ATH04 (Fen) | 56.905, −111.448 | 215 | Dwarf birch-dominant with sedge understory and scattered larch. Adjacent to winter road that transects perpendicular to flow direction. |
| ATH02 (SOW) | 56.913, −111.441 | 212 | Sedge and rush-dominant with common cattail and yellow pond lily near shoreline. Black spruce and poplar in adjacent upland. Adjacent to recent cut block (2019), within the flood plain of the Athabasca River. |
| AUR02 (Bog) | 57.272, −111.261 | 305 | Black spruce-dominant with willow and graminoid understory. Nutrient-rich wetland complex located adjacent to cut block. Complex transitions from bog to swamp. |
| AUR01 (SOW) | 57.258, −111.252 | 305 | Willow-dominant with sedge and common cattail understory. Located on reclaimed exploration area/borrow. |
| HWC02 (SOW) | 56.533, −111.322 | 421 | Willow-dominant with sedge and graminoid understory. Common cattail and rush at shoreline. Disturbed wetland/borrow pit immediately adjacent to Highway 63. |
| HAN02 (Fen) | 56.315, −111.624 | 597 | Black spruce- and larch-dominant with willow and sedge understory. Floating fen located on in situ oil sands lease, adjacent to Highway 63. |
| HEA01 (Fen) | 56.955, −111.541 | 286 | Dwarf birch-dominant with sedge understory. Scattered willow, larch, and black spruce. Wetland complex within oil sands exploration area. Road intersects connected swamp upstream. |
| HOR03 (Bog) | 56.327, −111.591 | 542 | Black spruce-dominant with bog birch, Labrador tea, and other graminoid. Within forest adjacent to pipeline corridor and Highway 63. |
| JCK02 (Bog) | 57.432, −111.254 | 265 | Black spruce-dominant with bog birch, cottongrass, and Labrador tea understory. Pristine bog, located near low-impact exploration. |
| JCK01 (Fen) | 57.442, −111.218 | 265 | Sedge- and pitcher plant-dominant in “flarks” with larch, black spruce, and bog birch in adjacent “strings”. Patterned fen, located near low-impact exploration. Patterned fen is made up of strings and flarks; strings are elevated mounds, and flarks are low-lying areas between strings. |
| JCK03 (SOW) | 57.403, −111.310 | 264 | Black spruce- and willow-dominant with mixed sedge and forb understory. Low-impact SOW complex transitioning to treed swamp and shrubby/graminoid fen. |
| JEN01 (SOW) | 57.136, −111.602 | 209 | Willow, birch, sedge, and other graminoid-dominant with common cattail and horsetail near shoreline. High-impact SOW intersected by Highway 63 and pipeline corridor. Adjacent to industrial yard. |
| JPH04 (Bog) | 57.113, −111.423 | 311 | Black spruce-dominant with cottongrass, bog birch, and Labrador tea understory. Located within sandy substrate. Adjacent to active exploration and access road. |
| MAQ01 (Fen) | 56.369, −111.284 | 695 | Sedge-dominant with scattered dwarf birch. Low-impact graminoid fen hydrologically connected to lake. Large open water area at central zone. |
| MCK01 (Bog) | 57.228, −111.703 | 272 | Black spruce-dominant with bog birch, Labrador tea, and bog rosemary understory. Adjacent to Canadian Natural Resources (CNRL) Horizon Highway and exploration access trail. |
| MCM01 (Bog) | 56.627, −111.196 | 361 | Black spruce-dominant with bog birch, Labrador tea, bog rosemary, and cranberry understory. Elevated bog from surrounding fen. Surrounded by exploration access trail. |
| PAT01 (SOW) | 57.511, −111.402 | 269 | Willow-dominant with scattered poplar and Jack pine. Sedge- and yellow pond lily-dominant near riparian and shoreline. Karst sinkhole within sandy upland. Adjacent to access road. |
| PAU01 (Fen) | 56.375, −111.235 | 715 | Bog birch- and sedge-dominant with scattered larch and black spruce. Located in local valley. Exhibits deep peat deposits. Intersecting road induces water pooling at northern part of fen. |
| POP01 (Fen) | 56.938, −111.549 | 294 | Larch- and black spruce-dominant with willow, bog birch, and sedge understory. Rich treed fen, adjacent to recent fire, access road, and low-impact seismic. |
| SAL01 (Fen) | 56.573, −111.276 | 372 | Saline tolerant grasses-dominant with willow and dwarf birch scattered throughout. Low-impact seismic line at north of fen. Scattered with open water areas. |
| Equipment | Description |
|---|---|
| HOBO USB Micro Station Data Logger-H21-USB https://www.onsetcomp.com/sites/default/files/resources-documents/20875-E%20H21-USB%20Manual.pdf (accessed on 6 May 2023) | Processor, power, and data storage assembly unit for connected sensors. |
| HOBO Rain Gauge Data Logger-RG3 https://www.onsetcomp.com/sites/default/files/resources-documents/10241-M%20MAN-RG3%20and%20RG3-M.pdf (accessed on 6 May 2023) | Records the amount of precipitation as rainfall. |
| Soil Moisture Smart Sensor-EC5 (S-SMC-M005) https://www.onsetcomp.com/sites/default/files/resources-documents/15081-J%20S-SMx%20Manual.pdf (accessed on 6 May 2023) | Records soil moisture content and temperature. |
| HOBO Temperature/RH Smart Sensor (S-THB-M002) https://www.onsetcomp.com/sites/default/files/resources-documents/11427-O%20S-THB%20Manual.pdf (accessed on 6 May 2023) | Records temperature and relative humidity. Sensor is protected from direct radiation with a solar radiation shield (HOBO RS3). |
| Onset HOBO U20 Water Level Logger https://www.onsetcomp.com/sites/default/files/resources-documents/12315-J%20U20%20Manual.pdf (accessed on 6 May 2023) | Records pressure exerted by vertical water column (when submerged) and water temperature. Depth to water is calculated by calibrating pressure measurements against ambient barometric pressure. |
| Reconyx Hyperfire 2 Outdoor Series Camera https://www.reconyx.com/img/file/-HyperFire2UserGuide2018_04_24_v1.pdf (accessed on 6 May 2023) | Digital camera with a passive infrared motion detector and a nighttime infrared illuminator that work in combination to capture photographs of wildlife. |
| Wildlife Acoustics Song Meter SM4 https://www.wildlifeacoustics.com/uploads/user-guides/SM4-USER-GUIDE-EN20220923.pdf (accessed on 6 May 2023) | Programmable audio recorder designed for the periodic, seasonal, and long-term monitoring of wildlife bioacoustics. |
References
- Zoltai, S.C.; Vitt, D.H. Canadian wetlands: Environmental gradients and classification. Vegetatio 1995, 118, 131–137. [Google Scholar] [CrossRef]
- National Wetlands Working Group. Wetlands of Canda/by National Wetlands Working Group, Canada Committee on Ecological Land Classification; Environment Canada; Sustainable Development Branch, Canadian Wildlife Service: Ottawa, ON, Canada, 1988.
- Convention on Wetlands. Global Wetland Outlook: Special Edition 2021; Ramsar: Gland, Switzerland, 2021. [Google Scholar]
- IPBES. Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services (IPBES) Secratariat: Bonn, Germany, 2019. [Google Scholar]
- O’Flaherty, M.; Davidson-Hunt, I. Scoping Exercise for Indigenous Ecological Classification of Wetlands in the Athabasca Oil Sands Region; Cumulative Environmental Management Association: Fort McMurray, AB, Canada, 2008; p. 45. [Google Scholar]
- Dyck, T.; Garibaldi, A. Cultural Importance of Wetland Plants: A Desktop Review of the Athabasca and Peace Oil Sands Region; Alberta Environment and Parks: Edmonton, AB, Canada, 2018; p. 21.
- Uprety, Y.; Asselin, H.; Dhakal, A.; Julien, N. Traditional use of medicinal plants in the boreal forest of Canada: Review and perspectives. J. Ethnobiol. Ethnomed. 2012, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Gardner, R.C.; Finlayson, C. Global Wetland Outlook: State of the World’s Wetlands and Their Services to People; Ramsar Convention Secretariat: Gland, Switzerland, 2018. [Google Scholar]
- Komers, P.E.; Stanojevic, Z. Rates of disturbance vary by data resolution: Implications for conservation schedules using the Alberta boreal forest as a case study. Glob. Chang. Biol. 2013, 19, 2916–2928. [Google Scholar] [CrossRef] [PubMed]
- Helbig, M.; Waddington, J.M.; Alekseychik, P.; Amiro, B.D.; Aurela, M.; Barr, A.G.; Black, T.A.; Blanken, P.D.; Carey, S.K.; Chen, J.; et al. Increasing contribution of peatlands to boreal evapotranspiration in a warming climate. Nat. Clim. Chang. 2020, 10, 555–560. [Google Scholar] [CrossRef]
- Government of Alberta. Alberta Wetland Policy; Alberta Environment and Parks: Edmonton, AB, Canada, 2013.
- Natural Resources Canada. Oil Resources. Available online: https://www.nrcan.gc.ca/our-natural-resources/energy-sources-distribution/fossil-fuels/crude-oil/oil-resources/18085 (accessed on 14 June 2022).
- Alberta Energy Regulator. Statistical Series 39: Alberta Mineable Oil Sands Plant Statistics, Monthly Supplement; Alberta Energy Regulator: Calgary, AB, Canada, 2022.
- Alberta Energy Regulator. ST-53 2022 Alberta In Situ Oil Sands Summary; Alberta Energy Regulator: Calgary, AB, Canada, 2022.
- Cronmiller, J.G.; Noble, B.F. The discontinuity of environmental effects monitoring in the Lower Athabasca region of Alberta, Canada: Institutional challenges to long-term monitoring and cumulative effects management. Environ. Rev. 2018, 26, 169–180. [Google Scholar] [CrossRef]
- Kelly, E.N.; Schindler, D.W.; Hodson, P.V.; Short, J.W.; Radmanovich, R.; Nielsen, C.C. Oil sands development contributes elements toxic at low concentrations to the Athabasca River and its tributaries. Proc. Natl. Acad. Sci. USA 2010, 107, 16178–16183. [Google Scholar] [CrossRef]
- Kelly, E.N.; Short, J.W.; Schindler, D.W.; Hodson, P.V.; Ma, M.; Kwan, A.K.; Fortin, B.L. Oil sands development contributes polycyclic aromatic compounds to the Athabasca River and its tributaries. Proc. Natl. Acad. Sci. USA 2009, 106, 22346–22351. [Google Scholar] [CrossRef]
- Gosselin, P.; Hrudey, S.E.; Naeth, M.A.; Plourde, A.; Therrien, R.; Van Der Kraak, G.; Xu, Z. Environmental and Health Impacts of Canada’s Oil Sands Industry; Royal Society of Canada: Ottawa, ON, Canada, 2010. [Google Scholar]
- Oil Sands Advisory Panel. A Foundation for the Future: Building an Environmental Monitoring System for the Oil Sands; Environment Canada: Ottawa, ON, Canada, 2010.
- Environment Canada. Joint Canada-Alberta Implementation Plan for Oil Sands Monitoring; Environment Canada: Ottawa, ON, Canada, 2012; p. 32.
- Ficken, C.D.; Connor, S.J.; Rooney, R.; Cobbaert, D. Drivers, pressures, and state responses to inform long-term oil sands wetland monitoring program objectives. Wetl. Ecol. Manag. 2021, 30, 47–66. [Google Scholar] [CrossRef]
- Hunsaker, C.T.; Carpenter, D.E. Ecological Indicators for the Environmental Monitoring and Assessment Program; U.S. Environmental Protection Agency: Research Triangle Park, NC, USA, 1990.
- ESRD. Alberta Wetland Classification System; Branch, W.P., Ed.; Policy and Planning Division: Edmonton, AB, Canada, 2015.
- Smeets, E.; Weterings, R. Environmental Indicators: Typology and Overview; 25/1999; European Environmental Agency: Copenhagen, Denmark, 1999.
- Baird, D.J.; Van den Brink, P.J.; Chariton, A.A.; Dafforn, K.A.; Johnston, E.L. New diagnostics for multiply stressed marine and freshwater ecosystems: Integrating models, ecoinformatics and big data. Mar. Freshw. Res. 2016, 67, 391–392. [Google Scholar] [CrossRef]
- Dubé, M.; Cash, K.; Wrona, F.; Enei, G.; Cronmiller, J.; Abel, R.; Andreeff, W.; Berrade, D.; Davidson, C.; Dawson, J. Oil Sands Monitoring Program Letter of Agreement and Operational Framework; Alberta Environment and Parks: Edmonton, AB, Canada, 2018.
- Likens, G.; Lindenmayer, D. Effective Ecological Monitoring; CSIRO Publishing: Clayton South, VIC, Australia, 2018. [Google Scholar]
- Alberta Environment (Ed.) Aquatic Ecosystems Field Sampling Protocols; Alberta Environment: Edmonton, AB, Canada, 2006; p. 142.
- Wrona, F.; di Cenzo, P. Lower Athabasca Water Quality Monitoring Plan: Phase 1; Environment Canada: Ottawa, ON, Canada, 2011.
- Government of Alberta. Environmental Quality Guidelines for Alberta Surface Waters; Water Policy Branch, Ed.; Alberta Environment and Parks: Edmonton, AB, Canada, 2018.
- Government of Alberta. Shapefiles for Land Disturbance due to Oil Sands Projects: 2020 (Detailed); Alberta Environment and Parks: Edmonton, AB, Canada, 2020.
- Government of Alberta. Alberta’s Oil Sands Projects and Upgraders; Alberta Environment and Parks: Edmonton, AB, Canada, 2017.
- Armellin, A.; Baird, D.; Curry, C.; Glozier, N.; Martens, A.; McIvor, E. CABIN Wetland Macroinvertebrate Protocol; Environment and Climate Change Canada, Ed.; Environment and Climate Change Canada: Gatineau, QC, Canada, 2019; p. 65.
- McDermott, H.; Paull, T.; Strachan, S. CABIN Laboratory Methods: Processing, Taxonomy and Quality Control of Benthic Macroinvertebrate Samples; Environment Canada: Ottawa, ON, Canada, 2014.
- Kovalenko, K.E.; Ciborowski, J.J.; Daly, C.; Dixon, D.G.; Farwell, A.J.; Foote, A.L.; Frederick, K.R.; Costa, J.M.; Kennedy, K.; Liber, K.; et al. Food web structure in oil sands reclaimed wetlands. Ecol. Appl. 2013, 23, 1048–1060. [Google Scholar] [CrossRef]
- Hanisch, J.R.; Connor, S.J.; Scrimgeour, G.J.; Cobbaert, D. Bioassessment of benthic macroinvertebrates in wetlands: A paired comparison of two standardized sampling protocols. Wetl. Ecol. Manag. 2020, 28, 199–216. [Google Scholar] [CrossRef]
- Lankau, H. Autonomous Recording Unit Deployment Protocol SM2, SM3, and SM4 Models of Song Meters; Bioacoustic Unit: Edmonton, AB, Canada, 2017. [Google Scholar]
- Chasmer, L.; Mahoney, C.; Millard, K.; Nelson, K.; Peters, D.; Merchant, M.; Hopkinson, C.; Brisco, B.; Niemann, O.; Montgomery, J.; et al. Remote Sensing of Boreal Wetlands 2: Methods for Evaluating Boreal Wetland Ecosystem State and Drivers of Change. Remote Sens. 2020, 12, 1321. [Google Scholar] [CrossRef]
- Chasmer, L.; Lima, E.M.; Mahoney, C.; Hopkinson, C.; Montgomery, J.; Cobbaert, D. Shrub changes with proximity to anthropogenic disturbance in boreal wetlands determined using bi-temporal airborne lidar in the Oil Sands Region, Alberta Canada. Sci. Total Environ. 2021, 780, 146638. [Google Scholar] [CrossRef] [PubMed]
- Rooney, R.C.; Bayley, S.E.; Schindler, D.W. Oil sands mining and reclamation cause massive loss of peatland and stored carbon. Proc. Natl. Acad. Sci. USA 2012, 109, 4933–4937. [Google Scholar] [CrossRef]
- Lower Athabasca Regional Plan Review Panel. Review Panel Report 2015: Lower Athabasca Regional Plan; Alberta Environment and Parks: Edmonton, AB, Canada, 2015; p. 256.
- Devito, K.; Mendoza, C.; Qualizza, C. Conceptualizing Water Movement in the Boreal Plains. Implications for Watershed Reconstruction; Environmental and Reclamation Research Group for the Canadian Oil Sands Network for Research and Development: Fort McMurray, AB, Canada, 2012. [Google Scholar]
- Kompanizare, M.; Petrone, R.M.; Shafii, M.; Robinson, D.T.; Rooney, R.C. Effect of climate change and mining on hydrological connectivity of surficial layers in the Athabasca Oil Sands Region. Hydrol. Process. 2018, 32, 3698–3716. [Google Scholar] [CrossRef]
- Mitsch, J.W.; Gosselink, J.G. Wetlands, 5th ed.; Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Volik, O.; Elmes, M.; Petrone, R.; Kessel, E.; Green, A.; Cobbaert, D.; Price, J. Wetlands in the Athabasca Oil Sands Region: The nexus between wetland hydrological function and resource extraction. Environ. Rev. 2020, 28, 246–261. [Google Scholar] [CrossRef]
- Weston, J.; Kariyeva, J.; Roy, M.-C. Wetland Management and Monitoring Needs. A Review of Alberta’s Legislation, Regulations, and Policies Related to Wetland Management; Alberta Biodiversity Monitoring Institute: Edmonton, AB, Canada, 2016. [Google Scholar]
- Bocking, E.; Cooper, D.J.; Price, J. Using tree ring analysis to determine impacts of a road on a boreal peatland. For. Ecol. Manag. 2017, 404, 24–30. [Google Scholar] [CrossRef]
- Plach, J.M.; Wood, M.E.; Macrae, M.L.; Osko, T.J.; Petrone, R.M. Effect of a semi-permanent road on N, P, and CO2 dynamics in a poor fen on the Western Boreal Plain, Canada. Ecohydrology 2017, 10, e1874. [Google Scholar] [CrossRef]
- Wieder, R.K.; Vile, M.A.; Scott, K.D.; Quinn, J.C.; Albright, C.M.; McMillen, K.J.; Herron, C.; Fillingim, H. Is bog water chemistry affected by increasing N and S deposition from oil sands development in Northern Alberta, Canada? Environ. Monit. Assess. 2021, 193, 766. [Google Scholar] [CrossRef]
- Wieder, R.K.; Vitt, D.H.; Vile, M.A.; Graham, J.A.; Hartsock, J.A.; Fillingim, H.; House, M.; Quinn, J.C.; Scott, K.D.; Petix, M.; et al. Experimental nitrogen addition alters structure and function of a boreal bog: Critical load and thresholds revealed. Ecol. Monogr. 2019, 89, e01371. [Google Scholar] [CrossRef]
- Proemse, B.C.; Mayer, B.; Chow, J.C.; Watson, J.G. Isotopic characterization of nitrate, ammonium and sulfate in stack PM2.5 emissions in the Athabasca Oil Sands Region, Alberta, Canada. Atmos. Environ. 2012, 60, 555–563. [Google Scholar] [CrossRef]
- CCME. Canadian Water Quality Guidelines for the Protection of Aquatic Life: Polycyclic Aromatic Hydrocarbons (PAHs); Environment Canada; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 1999.
- CCME. Canadian Water Quality Guidelines for the Protection of Aquatic Life: Ammonia; Environment Canada; National Guidelines and Standards Office: Gatineau, QC, Canada, 2010.
- Landis, M.S.; Pancras, J.P.; Graney, J.R.; Stevens, R.K.; Percy, K.E.; Krupa, S. Receptor Modeling of Epiphytic Lichens to Elucidate the Sources and Spatial Distribution of Inorganic Air Pollution in the Athabasca Oil Sands Region. In Alberta Oil Sands; Elsevier: Amsterdam, The Netherlands, 2012; Volume 11, pp. 427–467. [Google Scholar]
- Klemt, W.H.; Kay, M.L.; Wiklund, J.A.; Wolfe, B.B.; Hall, R.I. Assessment of vanadium and nickel enrichment in Lower Athabasca River floodplain lake sediment within the Athabasca Oil Sands Region (Canada). Environ. Pollut. 2020, 265, 114920. [Google Scholar] [CrossRef] [PubMed]
- Ficken, C.D.; Cobbaert, D.; Rooney, R.C. Low extent but high impact of human land use on wetland flora across the boreal oil sands region. Sci. Total Environ. 2019, 693, 133647. [Google Scholar] [CrossRef] [PubMed]
- Parsons, B.G.; Watmough, S.A.; Dillon, P.J.; Somers, K.M. A bioassessment of lakes in the Athabasca Oil Sands Region, Alberta, using benthic macroinvertebrates. J. Limnol. 2010, 69, 105. [Google Scholar] [CrossRef]
- Ceballos, G.; Ehrlich, P.R.; Dirzo, R. Biological annihilation via the ongoing sixth mass extinction signaled by vertebrate population losses and declines. Proc. Natl. Acad. Sci. USA 2017, 114, E6089–E6096. [Google Scholar] [CrossRef]
- Roberts, D.R.; Bayne, E.M.; Beausoleil, D.; Dennett, J.; Fisher, J.T.; Hazewinkel, R.O.; Sayanda, D.; Wyatt, F.; Dube, M.G. A synthetic review of terrestrial biological research from the Alberta oil sands region: 10 years of published literature. Integr. Environ. Assess. Manag. 2022, 18, 388–406. [Google Scholar] [CrossRef] [PubMed]
- Venier, L.A.; Thompson, I.D.; Fleming, R.; Malcolm, J.; Aubin, I.; Trofymow, J.A.; Langor, D.; Sturrock, R.; Patry, C.; Outerbridge, R.O.; et al. Effects of natural resource development on the terrestrial biodiversity of Canadian boreal forests. Environ. Rev. 2014, 22, 457–490. [Google Scholar] [CrossRef]
- You, Y.; Staebler, R.M.; Moussa, S.G.; Beck, J.; Mittermeier, R.L. Methane emissions from an oil sands tailings pond: A quantitative comparison of fluxes derived by different methods. Atmos. Meas. Tech. 2021, 14, 1879–1892. [Google Scholar] [CrossRef]
- Vile, M.A.; Wieder, R.K.; Živković, T.; Scott, K.D.; Vitt, D.H.; Hartsock, J.A.; Iosue, C.L.; Quinn, J.C.; Petix, M.; Fillingim, H.M.; et al. N2-fixation by methanotrophs sustains carbon and nitrogen accumulation in pristine peatlands. Biogeochemistry 2014, 121, 317–328. [Google Scholar] [CrossRef]
- Makar, P.A.; Akingunola, A.; Aherne, J.; Cole, A.S.; Aklilu, Y.-a.; Zhang, J.; Wong, I.; Hayden, K.; Li, S.-M.; Kirk, J.; et al. Estimates of exceedances of critical loads for acidifying deposition in Alberta and Saskatchewan. Atmos. Chem. Phys. 2018, 18, 9897–9927. [Google Scholar] [CrossRef]
- Bartels, S.F.; Gendreau-Berthiaume, B.; Macdonald, S.E. The impact of atmospheric acid deposition on tree growth and forest understory vegetation in the Athabasca Oil Sands Region. Sci. Total Environ. 2019, 696, 133877. [Google Scholar] [CrossRef] [PubMed]
- Watmough, S.A.; Whitfield, C.J.; Fenn, M.E. The importance of atmospheric base cation deposition for preventing soil acidification in the Athabasca Oil Sands Region of Canada. Sci. Total Environ. 2014, 493, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kirk, J.L.; Muir, D.C.; Gleason, A.; Wang, X.; Lawson, G.; Frank, R.A.; Lehnherr, I.; Wrona, F. Atmospheric deposition of mercury and methylmercury to landscapes and waterbodies of the Athabasca oil sands region. Environ. Sci. Technol. 2014, 48, 7374–7383. [Google Scholar] [CrossRef] [PubMed]
- Cooke, C.A.; Kirk, J.L.; Muir, D.C.G.; Wiklund, J.A.; Wang, X.; Gleason, A.; Evans, M.S. Spatial and temporal patterns in trace element deposition to lakes in the Athabasca oil sands region (Alberta, Canada). Environ. Res. Lett. 2017, 12, 124001. [Google Scholar] [CrossRef]
- Wasiuta, V.; Kirk, J.L.; Chambers, P.A.; Alexander, A.C.; Wyatt, F.R.; Rooney, R.C.; Cooke, C.A. Accumulating Mercury and Methylmercury Burdens in Watersheds Impacted by Oil Sands Pollution. Environ. Sci. Technol. 2019, 53, 12856–12864. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shotyk, W.; Zaccone, C.; Noernberg, T.; Pelletier, R.; Bicalho, B.; Froese, D.G.; Davies, L.; Martin, J.W. Airborne Petcoke Dust is a Major Source of Polycyclic Aromatic Hydrocarbons in the Athabasca Oil Sands Region. Environ. Sci. Technol. 2016, 50, 1711–1720. [Google Scholar] [CrossRef]
- Wentworth, G.R.; Aklilu, Y.A.; Landis, M.S.; Hsu, Y.M. Impacts of a large boreal wildfire on ground level atmospheric concentrations of PAHs, VOCs and ozone. Atmos. Environ. 2018, 178, 19–30. [Google Scholar] [CrossRef]
- Government of Canada. Canadian Environmental Protection Act; Government of Canada, Ed.; Minister of Justice: Ottawa, ON, Canada, 1999.
- Landis, M.S.; Studabaker, W.B.; Pancras, J.P.; Graney, J.R.; Puckett, K.; White, E.M.; Edgerton, E.S. Source apportionment of an epiphytic lichen biomonitor to elucidate the sources and spatial distribution of polycyclic aromatic hydrocarbons in the Athabasca Oil Sands Region, Alberta, Canada. Sci. Total Environ. 2019, 654, 1241–1257. [Google Scholar] [CrossRef]
- Brix, K.V.; Schlekat, C.E.; Garman, E.R. The mechanisms of nickel toxicity in aquatic environments: An adverse outcome pathway analysis. Environ. Toxicol. Chem. 2017, 36, 1128–1137. [Google Scholar] [CrossRef]
- Donner, M.W.; Bicalho, B.; Sinn, C.; Shotyk, W. Selenium and sulphur in Athabasca bituminous sands mineral and bitumen fractions. Fuel 2018, 224, 718–725. [Google Scholar] [CrossRef]
- Lemly, A.D. Environmental implications of excessive selenium: A review. Biomed. Environ. Sci. 1997, 10, 415–435. [Google Scholar] [PubMed]
- Gensemer, R.W.; Playle, R.C. The Bioavailability and Toxicity of Aluminum in Aquatic Environments. Crit. Rev. Environ. Sci. Technol. 1999, 29, 315–450. [Google Scholar] [CrossRef]
- Cardwell, A.S.; Adams, W.J.; Gensemer, R.W.; Nordheim, E.; Santore, R.C.; Ryan, A.C.; Stubblefield, W.A. Chronic toxicity of aluminum, at a pH of 6, to freshwater organisms: Empirical data for the development of international regulatory standards/criteria. Environ. Toxicol. Chem. 2018, 37, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Oil Sands Monitoring Program. Oil Sands Monitoring Program: Annual Report for 2018–2019; Alberta Environment and Parks: Edmonton, ON, Canada, 2019.
- Rosa, L.; Davis, K.F.; Rulli, M.C.; D’Odorico, P. Environmental consequences of oil production from oil sands. Earth’s Future 2017, 5, 158–170. [Google Scholar] [CrossRef]
- Dabros, A.; Pyper, M.; Castilla, G. Seismic lines in the boreal and arctic ecosystems of North America: Environmental impacts, challenges, and opportunities. Environ. Rev. 2018, 26, 214–229. [Google Scholar] [CrossRef]
- Alexander, A.C.; Chambers, P.A. Assessment of seven Canadian rivers in relation to stages in oil sands industrial development, 1972–2010. Environ. Rev. 2016, 24, 484–494. [Google Scholar] [CrossRef]
- Fennell, J.; Arciszewski, T.J. Current knowledge of seepage from oil sands tailings ponds and its environmental influence in northeastern Alberta. Sci. Total Environ. 2019, 686, 968–985. [Google Scholar] [CrossRef]
- Barthelmes, A.; Couwenberg, J.; Risager, M.; Tegetmeyer, C.; Joosten, H. Peatlands and Climate in a Ramsar Context: A Nordic-Baltic Perspective; Nordic Council of Ministers: Copenhagen, Denmark, 2015. [Google Scholar]
- Elmes, M.C.; Price, J.S. Hydrologic function of a moderate-rich fen watershed in the Athabasca Oil Sands Region of the Western Boreal Plain, northern Alberta. J. Hydrol. 2019, 570, 692–704. [Google Scholar] [CrossRef]
- Natural Regions Committee. Natural Regions and Subregions of Alberta; Downing, D.J., Pettapiece, W.W., Eds.; Government of Alberta: Edmonton, AB, Canada, 2006.
- Devito, K.; Creed, I.; Gan, T.; Mendoza, C.; Petrone, R.; Silins, U.; Smerdon, B. A framework for broad-scale classification of hydrologic response units on the Boreal Plain: Is topography the last thing to consider? Hydrol. Process. 2005, 19, 1705–1714. [Google Scholar] [CrossRef]
- Tudhope, A.W.; Chilcott, C.P.; McCulloch, M.T.; Cook, E.R.; Chappell, J.; Ellam, R.M.; Lea, D.W.; Lough, J.M.; Shimmield, G.B. Variability in the El Nino-Southern Oscillation through a glacial-interglacial cycle. Science 2001, 291, 1511–1517. [Google Scholar] [CrossRef]
- Alexander, A.C.; Chambers, P.A.; Jeffries, D.S. Episodic acidification of 5 rivers in Canada’s oil sands during snowmelt: A 25-year record. Sci. Total Environ. 2017, 599–600, 739–749. [Google Scholar] [CrossRef]
- Campbel, P.G.C.; Stokes, P.M. Acidification and Toxicity of Metals to Aquatic Biota. Can. J. Fish. Aquat. Sci. 1985, 42, 2034–2049. [Google Scholar] [CrossRef]
- Kullberg, A.; Hargeby, A.; Petersen, R., Jr.; Bishop, K.; Jansson, M. The ecological significance of dissolved organic carbon in acidified waters. Ambio 1993, 22, 331–337. [Google Scholar]
- Burton, T.M.; Allan, J.W. Influence of pH, Aluminum, and Organic Matter on Stream Invertebrates. Can. J. Fish. Aquat. Sci. 1986, 43, 1285–1289. [Google Scholar] [CrossRef]
- Schindler, D.W. Evolution of phosphorus limitation in lakes. Science 1977, 195, 260–262. [Google Scholar] [CrossRef]
- Wassenaar, P.N.H.; Verbruggen, E.M.J. Persistence, bioaccumulation and toxicity-assessment of petroleum UVCBs: A case study on alkylated three-ring PAHs. Chemosphere 2021, 276, 130113. [Google Scholar] [CrossRef]
- Culp, J.M.; Glozier, N.E.; Baird, D.J.; Wrona, F.J.; Brua, R.B.; Ritcey, A.L.; Peters, D.L.; Casey, R.; Choung, C.B.; Curry, C.J. Assessing Ecosystem Health in Benthic Macroinvertebrate Assemblages of the Athabasca River Main Stem, Tributaries and Peace-Athabasca Delta; Government of Alberta: Edmonton, AB, Canada, 2018.
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef]
- De Lancey, E.R.; Brisco, B.; McLeod, L.J.T.; Hedley, R.; Bayne, E.M.; Murnaghan, K.; Gregory, F.; Kariyeva, J. Modelling, Characterizing, and Monitoring Boreal Forest Wetland Bird Habitat with RADARSAT-2 and Landsat-8 Data. Water 2021, 13, 2327. [Google Scholar] [CrossRef]
- McLeod, L.J.T.; DeLancey, E.R.; Bayne, E.M. Spatially Explicit Abundance Modeling of a Highly Specialized Wetland Bird Using Sentinel-1 and Sentinel-2 Modélisation spatialement explicite de l’abondance d’un oiseau très spécifique aux zones humides à l’aide de Sentinel-1 et de Sentinel-2. Can. J. Remote Sens. 2022, 48, 37–54. [Google Scholar] [CrossRef]
- Government of Alberta. Alberta Merged Wetland Inventory; Alberta Environment and Parks: Edmonton, AB, Canada, 2020.
- Ducks Unlimited Canada. Alberta Wetland Classification System Field Guide; Ducks Unlimited Canada: Edmonton, AB, Canada, 2021. [Google Scholar]
- Devito, K.J.; Hokanson, K.J.; Moore, P.A.; Kettridge, N.; Anderson, A.E.; Chasmer, L.; Hopkinson, C.; Lukenbach, M.C.; Mendoza, C.A.; Morissette, J.; et al. Landscape controls on long-term runoff in subhumid heterogeneous Boreal Plains catchments. Hydrol. Process. 2017, 31, 2737–2751. [Google Scholar] [CrossRef]
- Chasmer, L.; Cobbaert, D.; Mahoney, C.; Millard, K.; Peters, D.; Devito, K.; Brisco, B.; Hopkinson, C.; Merchant, M.; Montgomery, J.; et al. Remote Sensing of Boreal Wetlands 1: Data Use for Policy and Management. Remote Sens. 2020, 12, 1320. [Google Scholar] [CrossRef]
- Vose, R.S.; Arguez, A. The Definition of the Standard WMO Climate Normal: The Key to Deriving Alternative Climate Normals. Bull. Am. Meteorol. Soc. 2011, 92, 699–704. [Google Scholar] [CrossRef]
- Environment Canada. Metal Mining Technical Guidance for Environmental Effects Monitoring; Environment Canada; Government of Canada: Ottawa, ON, Canada, 2012; p. 550.
- Ficken, C.D.; Rooney, R.C. Oil Sands Mining Wetland Monitoring Program: 2018 Pilot Year Report; University of Waterloo: Waterloo, ON, Canada, 2019; p. 81. [Google Scholar]
- Montgomery, J.S.; Hopkinson, C.; Brisco, B.; Patterson, S.; Rood, S.B. Wetland hydroperiod classification in the western prairies using multitemporal synthetic aperture radar. Hydrol. Process. 2018, 32, 1476–1490. [Google Scholar] [CrossRef]
- Merchant, M.A. Classifying open water features using optical satellite imagery and an object-oriented convolutional neural network. Remote Sens. Lett. 2020, 11, 1127–1136. [Google Scholar] [CrossRef]
- Abib, T.H.; Chasmer, L.; Hopkinson, C.; Mahoney, C.; Rodriguez, L.C.E. Seismic line impacts on proximal boreal forest and wetland environments in Alberta. Sci. Total Environ. 2019, 658, 1601–1613. [Google Scholar] [CrossRef]
- Adam, E.; Mutanga, O.; Rugege, D. Multispectral and hyperspectral remote sensing for identification and mapping of wetland vegetation: A review. Wetl. Ecol. Manag. 2009, 18, 281–296. [Google Scholar] [CrossRef]
- Mahoney, C.; Hall, R.; Hopkinson, C.; Filiatrault, M.; Beaudoin, A.; Chen, Q. A Forest Attribute Mapping Framework: A Pilot Study in a Northern Boreal Forest, Northwest Territories, Canada. Remote Sens. 2018, 10, 1338. [Google Scholar] [CrossRef]
- Matasci, G.; Hermosilla, T.; Wulder, M.A.; White, J.C.; Coops, N.C.; Hobart, G.W.; Zald, H.S.J. Large-area mapping of Canadian boreal forest cover, height, biomass and other structural attributes using Landsat composites and lidar plots. Remote Sens. Environ. 2018, 209, 90–106. [Google Scholar] [CrossRef]
- Wan, Z. New refinements and validation of the MODIS Land-Surface Temperature/Emissivity products. Remote Sens. Environ. 2008, 112, 59–74. [Google Scholar] [CrossRef]
- Wan, Z.; Li, Z.L. Radiance-based validation of the V5 MODIS land-surface temperature product. Int. J. Remote Sens. 2010, 29, 5373–5395. [Google Scholar] [CrossRef]
- Hopkinson, C.; Crasto, N.; Marsh, P.; Forbes, D.; Lesack, L. Investigating the spatial distribution of water levels in the Mackenzie Delta using airborne LiDAR. Hydrol. Process. 2011, 25, 2995–3011. [Google Scholar] [CrossRef]
- Wulder, M.A.; White, J.C.; Nelson, R.F.; Næsset, E.; Ørka, H.O.; Coops, N.C.; Hilker, T.; Bater, C.W.; Gobakken, T. Lidar sampling for large-area forest characterization: A review. Remote Sens. Environ. 2012, 121, 196–209. [Google Scholar] [CrossRef]
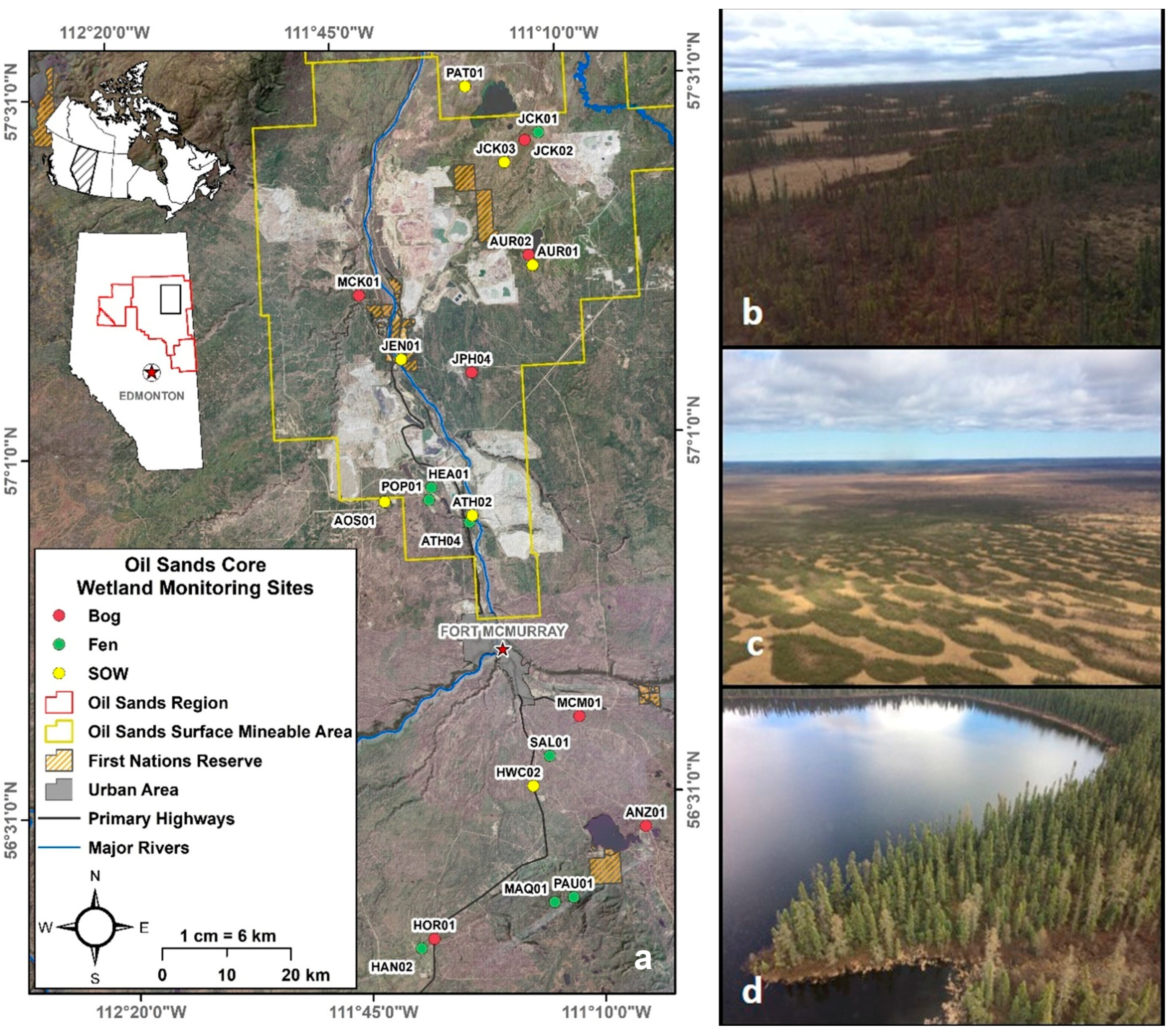




| Wetland Class | Position in the Landscape | Soil and Vegetation | Water Regime and Chemistry |
|---|---|---|---|
| Fen | Flat to gentle slopes. Often part of wetland complexes. | >40 cm depth peat. Hummocky microtopography. High floristic species diversity. Wetland forms include wooded, shrubby, graminoid. | Minerotrophic (water inputs from surface runoff, groundwater, and precipitation). Surface and groundwater flow with near-surface water table. Generally freshwater but can be slightly brackish. Ranges from nutrient-poor to extremely rich. Wide range of pH (from neutral to slightly alkaline). |
| Bog | Flat elevated terraces. | >40 cm depth peat. Hummocky microtopography. Low floristic species diversity. Wetland forms include wooded, shrubby, open. | Ombrotrophic (water input primarily from precipitation). Low groundwater flow with a stable water table. Acidic pH and low nutrients. |
| SOW | Natural and anthropogenic topographic depressions or lake margins. | Mineral wetlands with <40 cm organic soil. Characterized by floating and aquatic vegetation in <2 m of open water. Wetland forms include floating or submersed aquatic, bare. | Minerotrophic (water inputs from surface runoff, groundwater, and precipitation). Permanent open water bodies in the oil sands region with dynamic seasonal water levels. Nutrient-rich freshwater or saline. Typically, neutral pH. |
| Wetland Indicators | Indicator Rationale | Predicted Responses to Priority Oil Sands Development Pressures |
|---|---|---|
| Wetland area. (Changes in wetland area, fragmentation, loss of connectivity). | Wetland area status and trends are critical indicators of wetland health and condition [8]. Northern Alberta has one of the fastest rates of land disturbance [9]. Local Indigenous communities are concerned about land use change. | Land disturbances result in direct wetland loss, increased fragmentation, and decreased connectivity [39,40]. Changes in the habitat and abundance of traditional plant areas, wetland-reliant species at risk, and biodiversity [41]. |
| Meteorology. (Precipitation, temperature, relative humidity, and wind speed and direction). | Contextualize the influence of local climate on wetland hydrological conditions versus anthropogenic development. Contextualize wetland hydrological functioning as related to 10–15-year wet–dry climate cycles that characterize the OSR [42]. | Climate change is predicted to affect the duration of wetland hydrological connectivity in the region [43]. |
| Hydrology. (Water table depth, soil moisture levels) | Wetlands provide hydrological ecosystem services [44]. Hydrology is sensitive to local land disturbances and anthropogenic hydrological alterations [43,45]. Water table position and open water area are proxies for assessing change in wetland function [46]. Local Indigenous communities are concerned about access routes to harvesting and occupancy sites. | OS water management may result in abnormal water table positions [45], resulting in terrestrialization and changes to runoff. Localized infrastructure development may obstruct wetland natural subsurface flow, changing hydrodynamics [47,48]. |
| Surface water quality. (Full suite of OS SWQ parameters of concern for shallow open water wetlands; reduced suite of parameters in peatlands). | SWQ parameters provide a measure of aquatic habitat condition relative to the needs of flora and fauna (e.g., habitat, drinking water, etc.) [29]. Deposited contaminants can be transported large distances through the hydrological network (e.g., surface water and/or groundwater). Multiple OS contaminants can modify wetland function [49,50,51]. | Directly deposited or transported contaminants may cause eutrophication/nitrification [49,50]. Contaminant concentrations may change in relation to established guidelines [30,52,53]. Potential to change specific conductance and pH [54]. |
| Sediment quality. (Shallow open water wetlands only; full suite of OS sediment parameters of concern). | Sediments are contaminant sinks and a major exposure route for plants, invertebrates, amphibians, and birds. | Contaminant concentrations may change in relation to established guidelines (e.g., CCME PAL Guidelines). Shallow lake sediments near the OS mining center are enriched in vanadium and nickel [55]. |
| Vegetation. (Community composition and structure; culturally important plants; high disturbance indicator species; obligate wetland species). | Plant communities are sensitive to natural and anthropogenic drivers. Culturally important plants are a proxy of wetland health and change [6]. Local Indigenous communities are concerned about changes in vegetation communities reducing biodiversity [5,41]. | High disturbance indicator species are more common in wetlands nearer to land disturbances [56]. Change in vegetation community composition resulting from contaminant deposition. |
| Benthic invertebrates. (Shallow open water wetlands only; community composition). | Benthic invertebrates are small, aquatic organisms commonly used to assess the environmental condition of freshwaters (rivers, lakes, wetlands) across Canada [57]. | Benthic invertebrate communities are sensitive to the extent of land disturbance in wetland buffers and associated changes in surface water quality. |
| Wildlife. (Remote cameras and acoustic recorders). | Wildlife are sensitive to land disturbances and human activity. Local Indigenous communities are concerned that fewer wildlife are using wetlands [41]. | Potential for negative ecological and socioeconomic impacts on wildlife due to anthropogenic activity [58]. Increased human noise and activity has potential to reduce wildlife habitat and presence [59,60]. |
| Parameter | Potential Oil Sands-Related Source | Importance |
|---|---|---|
| Total Nitrogen | Industrial emissions (stack, fleet) of NOX; emissions of NH3 from tailings [61]; microbial fixation [62]. | Eutrophication of low-nutrient habitats (i.e., bogs and poor fens); shift from bryophyte-dominated to vascular-dominated communities [50]. Potential ammonia toxicity to fish and other aquatic life but dependent on pH and temperature [53]. |
| Sulfate | Industrial emissions of SO2 and H2S [51]. | Acidifying deposition [63]; sulfate toxicity is hardness dependent [30]. |
| Σ Base Cations | Deposition of fugitive dust from surface mining/surficial erosion [54]. | Some evidence of neutralizing acid deposition [64,65]; potential to increase pH in bogs. |
| Total and Methylmercury | Industrial emissions [66]; global deposition [67]; in situ fixation [68]. | Bioaccumulation and biomagnification in aquatic food web (Lavoie et al., 2013); human health concerns associated with wild food sources. |
| Σ Alk-PAHs | Raw bitumen, petroleum coke [69]; wildfire [70]. | Known mutagens and carcinogens; classified as toxic substances in Canada under Schedule 1 of the Canadian Environmental Protection Act [71]. |
| Vanadium | Petrogenic in origin; associated with stack emissions and fugitive dust from raw bitumen. | Mostly used as tracer for oil sands impacts on site; Alberta WQ guidelines only exist for irrigation and livestock water [30]. |
| Nickel | Oil sands and petroleum coke [72]. | Mostly used as tracer for oil sands impacts on site; essential metal but toxic at higher concentrations [73]. |
| Selenium | Associated with the organic (i.e., bitumen) fraction of ores [74]. | Bioaccumulation in aquatic food webs; toxic effects include deformed embryos and reproductive failure in wildlife [75]. |
| Aluminum | Crustal element associated with fugitive dust and mining activities [72]. | Known toxicity to aquatic organisms but dependent on pH, dissolved organic carbon (DOC), and hardness [76,77]. |
| Wetland Class | Richness | Diversity | Most Abundant Taxon * | |
|---|---|---|---|---|
| 2017 | ||||
| MAQ01 | Fen | 40 | 2.95 | Caenis (0.21) |
| AOS01 | SOW | 29 | 2.57 | Hyalella (0.34) |
| HAN01 | SOW | 39 | 2.13 | Caenis (0.50) |
| PAT01 | SOW | 33 | 2.77 | Hyalella (0.18) |
| 2018 | ||||
| MAQ01 | Fen | 32 | 2.67 | Hydrozetidae (0.22) |
| HWC02 | SOW | 36 | 2.16 | Caenis (0.48) |
| HAN01 | SOW | 34 | 2.84 | Chaoborus (0.17) |
| ATH02 | SOW | 40 | 2.68 | Caenis (0.30) |
| AOS01 | SOW | 26 | 2.56 | Dicrotendipes (0.18) |
| JEN01 | SOW | 27 | 1.97 | Caenis (0.50) |
| AUR01 | SOW | 35 | 3.10 | Leptoceridae (0.10) |
| PAT01 | SOW | 50 | 2.79 | Hyalella (0.18) |
| 2019 | ||||
| MAQ01 | Fen | 35 | 2.47 | Stylaria lacustris (0.33) |
| HWC02 | SOW | 49 | 2.90 | Tanytarsus (0.14) |
| HAN02 | Fen | 39 | 2.48 | Chaoborus (0.25) |
| ATH02 | SOW | 31 | 1.80 | Hyalella (0.56) |
| AOS01 | SOW | 48 | 2.90 | Hyalella (0.14) |
| JEN01 | SOW | 36 | 2.80 | Stylaria lacustris (0.16) |
| JCK03 | SOW | 37 | 2.32 | Hyalella (0.36) |
| AUR01 | SOW | 38 | 3.17 | Psectrocladius (0.10) |
| PAT01 | SOW | 59 | 2.99 | Baetidae (0.10) |
| Site | Class | Remote Cameras | Acoustic Recorder | ||||
|---|---|---|---|---|---|---|---|
| Richness | Species at Risk | Richness | Species at Risk | ||||
| 2018 | 2019 | 2018 | 2019 | 2018 | 2018 | ||
| ANZ01 | Bog | 6 | 6 | - | - | 27 | CONI 2 |
| AUR02 | 4 | 6 | - | - | 20 | - | |
| HOR03 | 4 | 2 | - | - | 22 | CONI 2 | |
| JCK02 | - | 3 | - | - | - | - | |
| JPH04 | 8 | 8 | - | WOCA 3 | 24 | CONI 2 | |
| MCK01 | 4 | 3 | - | - | 21 | CONI 2 | |
| MCM01 | 6 | - | WOCA 1 | - | 16 | CONI 2 | |
| ATH04 | Fen | 3 | 2 | - | - | 17 | - |
| HAN02 | - | 3 | - | STGR 2 | - | - | |
| HEA01 | 4 | 2 | - | - | 27 | CONI 2 | |
| JCK01 | - | 5 | - | STGR 2 | - | - | |
| MAQ01 | 2 | 1 | - | - | - | CONI 2 | |
| PAU01 | 5 | - | - | - | 14 | CONI 2 | |
| POP01 | 6 | 1 | - | - | 20 | CATO 4 | |
| SAL01 | 7 | 5 | STGR 2 | - | 19 | OSFL 4 | |
| AOS01 | SOW | 5 | 4 | - | - | 21 | CONI 2 |
| ATH02 | 10 | 11 | - | - | 22 | CONI 2 | |
| AUR01 | 8 | 3 | - | - | 21 | CONI 2, OSFL 4 | |
| HAN01 | 3 | - | - | - | - | - | |
| HWC02 | 5 | 5 | - | - | 19 | - | |
| JCK03 | - | 6 | - | - | - | - | |
| JEN01 | 10 | 10 | - | - | 13 | CONI 2, OSFL 4 | |
| PAT01 | 11 | 8 | STGR 2 | - | 21 | CONI 2 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahoney, C.; Montgomery, J.; Connor, S.; Cobbaert, D. Oil Sands Wetland Ecosystem Monitoring Program Indicators in Alberta, Canada: Transitioning from Pilot to Long-Term Monitoring. Water 2023, 15, 1914. https://doi.org/10.3390/w15101914
Mahoney C, Montgomery J, Connor S, Cobbaert D. Oil Sands Wetland Ecosystem Monitoring Program Indicators in Alberta, Canada: Transitioning from Pilot to Long-Term Monitoring. Water. 2023; 15(10):1914. https://doi.org/10.3390/w15101914
Chicago/Turabian StyleMahoney, Craig, Joshua Montgomery, Stephanie Connor, and Danielle Cobbaert. 2023. "Oil Sands Wetland Ecosystem Monitoring Program Indicators in Alberta, Canada: Transitioning from Pilot to Long-Term Monitoring" Water 15, no. 10: 1914. https://doi.org/10.3390/w15101914





