Allometric Relationships between Dry Matter Weights of Maize Organs and Their Responses to Drought
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Methods
2.2.1. Calculation of Irrigation Volume
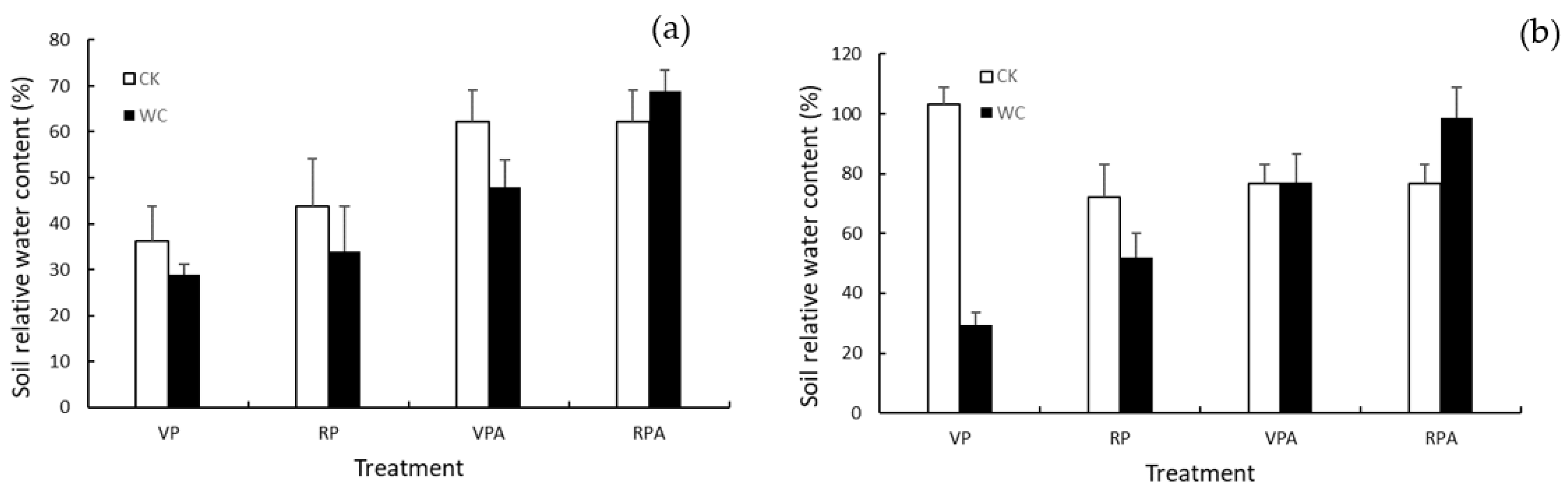
2.2.2. Soil Water Content Measurement
2.2.3. Parameter Expression and Allometric Method
2.3. Data Statistics
3. Results
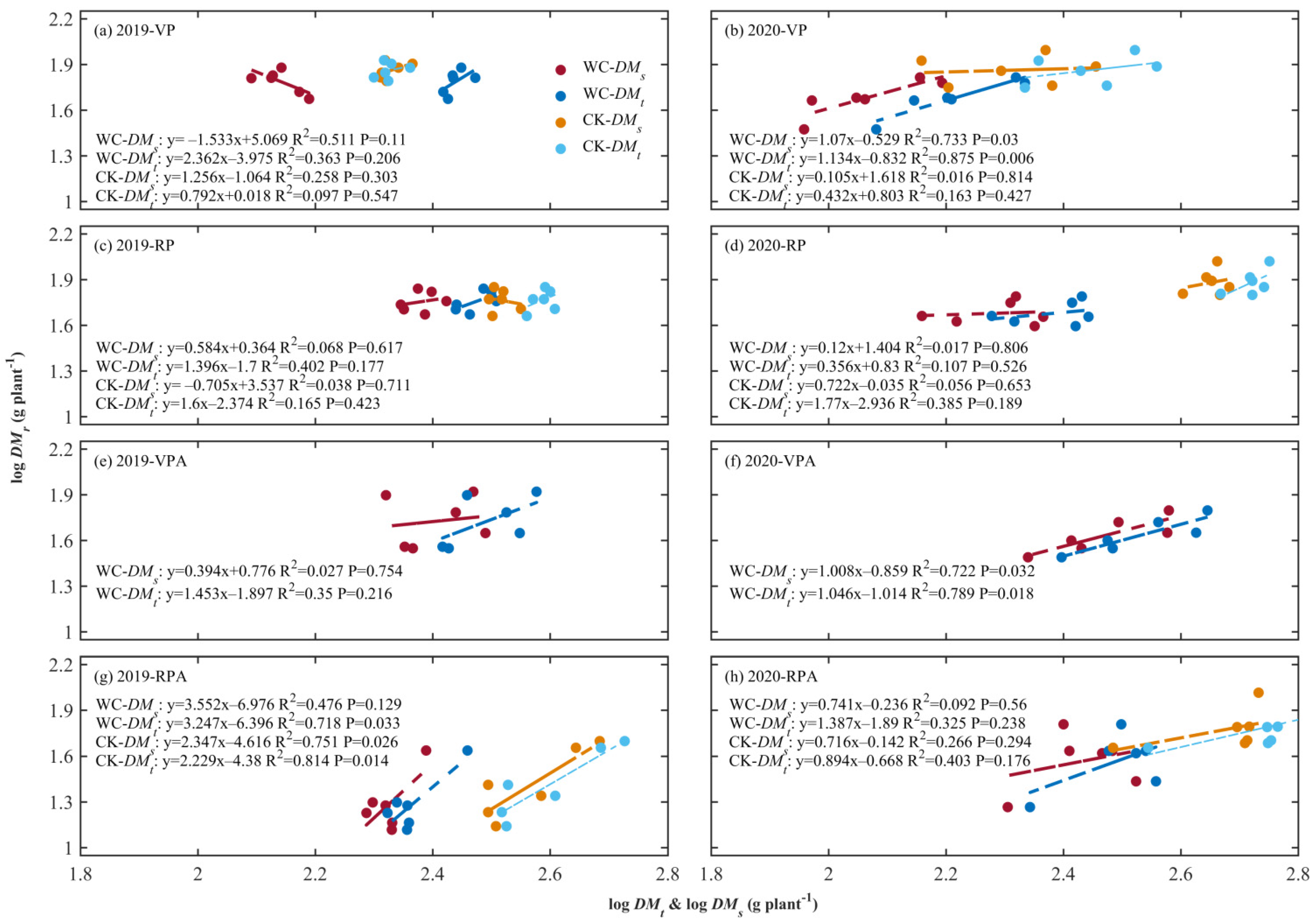
3.1. ARs between Roots, Aboveground, and Total Biomass
3.2. ARs between Ear and Various Parts of Maize
3.3. Size-Dependence of HI and Its Response to Drought and Rewatering
4. Discussion
4.1. ARs between Maize Root and Shoot and Total Plant and Their Response to Drought
4.2. ARs between Ear and Different Biomass and Their Drought Responses
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weiner, J.; Campbell, L.G.; Pino, J.; Echarte, L. The allometry of reproduction within plant populations. J. Ecol. 2009, 97, 1220–1233. [Google Scholar] [CrossRef]
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: Meta-analyses of interspecific variation and environmental control. New Phycol. 2012, 193, 30–50. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Sun, Z.X.; Zheng, J.M.; Wang, W.B.; Bai, W.; Feng, L.S.; Yang, N.; Xiang, W.Y.; Zhang, Z.; Feng, C. Dry Matter Accumulation, Allocation, Yield and Productivity of Maize Soybean Intercropping Systems in the Semi-Arid Region of Western Liaoning Province. Sci. Agric. Sin. 2021, 54, 909–920. [Google Scholar]
- Lizaso, J.; Ruiz-Ramos, M.; Rodríguez, L.; Gabaldon-Leal, C.; Oliveira, J.; Lorite, I.; Sánchez, D.; García, E.; Rodríguez, A. Impact of high temperatures in maize: Phenology and yield components. Field Crops Res. 2018, 216, 129–140. [Google Scholar] [CrossRef] [Green Version]
- Kruse, J.; Hänsch, R.; Mendel, R.R.; Renneberg, H. The role of root nitrate reduction in the systemic control of biomass partitioning between leaves and roots in accordance to the C/N-status of tobacco plants. Plant Soil. 2010, 332, 387–403. [Google Scholar] [CrossRef]
- Weiner, J. Allocation, plasticity and allometry in plants. Perspect. Plant Ecol. 2004, 6, 207–215. [Google Scholar] [CrossRef]
- Shipley, B.; Meziane, D. The balanced-growth hypothesis and the allometry of leaf and root biomass allocation. Funct. Ecol. 2002, 16, 326–331. [Google Scholar] [CrossRef]
- Qin, X.L.; Weiner, J.; Qi, L.; Xiong, Y.C.; Li, F.M. Allometric analysis of the effects of density on reproductive allocation and Harvest Index in 6 varieties of wheat (Triticum). Field Crops Res. 2013, 144, 162–166. [Google Scholar] [CrossRef]
- Ratjen, A.M.; Lemaire, G.; Kage, H.D.; Justes, P.E. Key variables for simulating leaf area and N status: Biomass based relations versus phenology driven approaches. Eur. J. Agron. 2018, 100, 110–117. [Google Scholar] [CrossRef]
- Zhang, Z.X.; Yu, K.L.; Jin, X.L.; Nan, Z.B.; Wang, J.F.; Niu, X.L.; Whish, J.P.M.; Bell, L.W.; Siddique, K.H.M. Above- and belowground dry matter partitioning of four warm-season annual crops sown on different dates in a semiarid region. Eur. J. Agron. 2019, 109, 125918. [Google Scholar] [CrossRef]
- Kage, H.; Kochler, M.; Stützel, H. Root growth and dry matter partitioning of cauliflower under drought stress conditions: Measurement and simulation. Eur. J. Agron. 2004, 20, 379–394. [Google Scholar] [CrossRef]
- Kumar, R.; Sarawgi, A.K.; Ramos, C.; Amarante, S.T.; Ismail, A.M.; Wade, L.J. Partitioning of dry matter during drought stress in rainfed lowland rice. Field Crops Res. 2006, 96, 455–465. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, S.; Hasan, W.; Ul-Allah, S.; Tanveer, M.; Farooq, M.; Nawaz, A. Drought stress in sunflower: Physiological effects and its management through breeding and agronomic alternatives. Agric. Water Manag. 2018, 201, 152–166. [Google Scholar] [CrossRef]
- Cai, F.; Zhang, Y.S.; Mi, N.; Ming, H.Q.; Zhang, S.J.; Zhang, H.; Zhao, X.L. Maize (Zea mays L.) physiological responses to drought and rewatering, and the associations with water stress degree. Agric. Water Manag. 2020, 241, 106479. [Google Scholar] [CrossRef]
- Djaman, K.; Irmak, S.; Rathje, W.R.; Martin, D.L.; Eisenhauer, D.E. Maize evapotranspiration, yield production functions, biomass, grain yield, harvest index, and yield response factors under full and limited irrigation. Trans. ASABE 2013, 56, 373–393. [Google Scholar] [CrossRef]
- Cai, F.; Zhang, Y.S.; Mi, N.; Ming, H.Q.; Zhang, S.J.; Zhang, H.; Zhao, X.L.; Zhang, B.B. The Effect of Drought and Sowing Date on Dry Matter Accumulation and Partitioning in the Above-Ground Organs of Maize. Atmosphere 2022, 13, 677. [Google Scholar] [CrossRef]
- Mi, N.; Cai, F.; Zhang, Y.S.; Ji, R.P.; Zhang, S.J.; Wang, Y. Differential responses of maize yield to drought at vegetative and reproductive stages. Plant Soil Environ. 2018, 64, 260–267. [Google Scholar]
- Wang, X.C.; Yang, W.Y.; Deng, X.Y.; Zhang, Q.; Yong, T.W.; Liu, W.G.; Yang, F.; Mao, S.M. Differences of dry matter accumulation and distribution of maize and their responses to nitrogen fertilization in maize/soybean and maize/sweet potato relay intercropping systems. Plant Nutr. Fertil. Sci. 2015, 21, 46–57. [Google Scholar]
- Wei, T.B.; Hu, F.L.; Zhao, C.; Feng, F.X.; Yu, A.Z.; Liu, C.; Chai, Q. Response of dry matter accumulation and yield components of maize under n-fertilizer postponing application in oasis irrigation areas. Sci. Agric. Sin. 2017, 50, 2916–2927. [Google Scholar]
- Anothai, J.; Soler, C.M.T.; Green, A. Evaluation of two evapotranspiration approaches simulated with the CSM-CERES Maize model under different irrigation strategies and the impact on maize growth, development and soil moisture content for semi-arid conditions. Agric. For. Meteorol. 2013, 176, 64–76. [Google Scholar] [CrossRef]
- Brett, M.T. When is a correlation between non-independent variables “spurious”? Oikos 2004, 105, 647–656. [Google Scholar] [CrossRef]
- McCarthy, M.C.; Enquist, B.J. Consistency between an allometric approach and optimal partitioning theory in global patterns of plant biomass allocation. Funct. Ecol. 2007, 21, 713–720. [Google Scholar] [CrossRef]
- Maddonni, G.A. Analysis of the climatic constraints to maize production in the current agricultural region of Argentina—A probabilistic approach. Theor. Appl. Climatol. 2012, 107, 325–345. [Google Scholar] [CrossRef]
- Bonelli, L.E.; Monzon, J.P.; Cerrudo, A.; Rizzalli, R.H. Maize grain yield components and source-sink relationship as affected by the delay in sowing date. Field Crops Res. 2016, 198, 215–225. [Google Scholar] [CrossRef]
- Cerrudo, A.; Di Matteo, J.; Fernandez, E.; Robles, M.; Pico, L.O.; Andrade, F.H. Yield components of maize as affected by short shading periods and thinning. Crop Pasture Sci. 2013, 64, 580–587. [Google Scholar] [CrossRef]



| Abbreviation | Description |
|---|---|
| AR | Allometric relationship |
| DMP | Dry matter partitioning |
| DMW | Dry matter weight |
| RSR | Root-shoot ratio |
| HI | Harvest index |
| SRWC | Soil relative water content |
| CK | The control treatment |
| WC | Water control treatment |
| VP | Vegetative period of maize |
| RP | Reproductive period of maize |
| VPWC, VPCK | WC during VP and its CK |
| RPWC, RPCK | WC during RP and its CK |
| VPAWC, RPAWC | VPWC and RPWC irrigated until maturity |
| CKA | The CK for VPAWC and RPAWC |
| DMr, DMe, DMs, and DMt | DMWs of root, ear, aboveground organ, and total plant |
| DMv0, DMv1 | DMWs of total plant minus ear and shoot minus ear |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ming, H.; Cai, F.; Mi, N.; Zhang, Y.; Zhang, H.; Zhang, S.; Zhao, X.; Feng, Y. Allometric Relationships between Dry Matter Weights of Maize Organs and Their Responses to Drought. Water 2023, 15, 99. https://doi.org/10.3390/w15010099
Ming H, Cai F, Mi N, Zhang Y, Zhang H, Zhang S, Zhao X, Feng Y. Allometric Relationships between Dry Matter Weights of Maize Organs and Their Responses to Drought. Water. 2023; 15(1):99. https://doi.org/10.3390/w15010099
Chicago/Turabian StyleMing, Huiqing, Fu Cai, Na Mi, Yushu Zhang, Hui Zhang, Shujie Zhang, Xianli Zhao, and Yanrui Feng. 2023. "Allometric Relationships between Dry Matter Weights of Maize Organs and Their Responses to Drought" Water 15, no. 1: 99. https://doi.org/10.3390/w15010099





