Physicochemical and Sorption Characteristics of Carbon Biochars Based on Lignin and Industrial Waste Magnetic Iron Dust
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
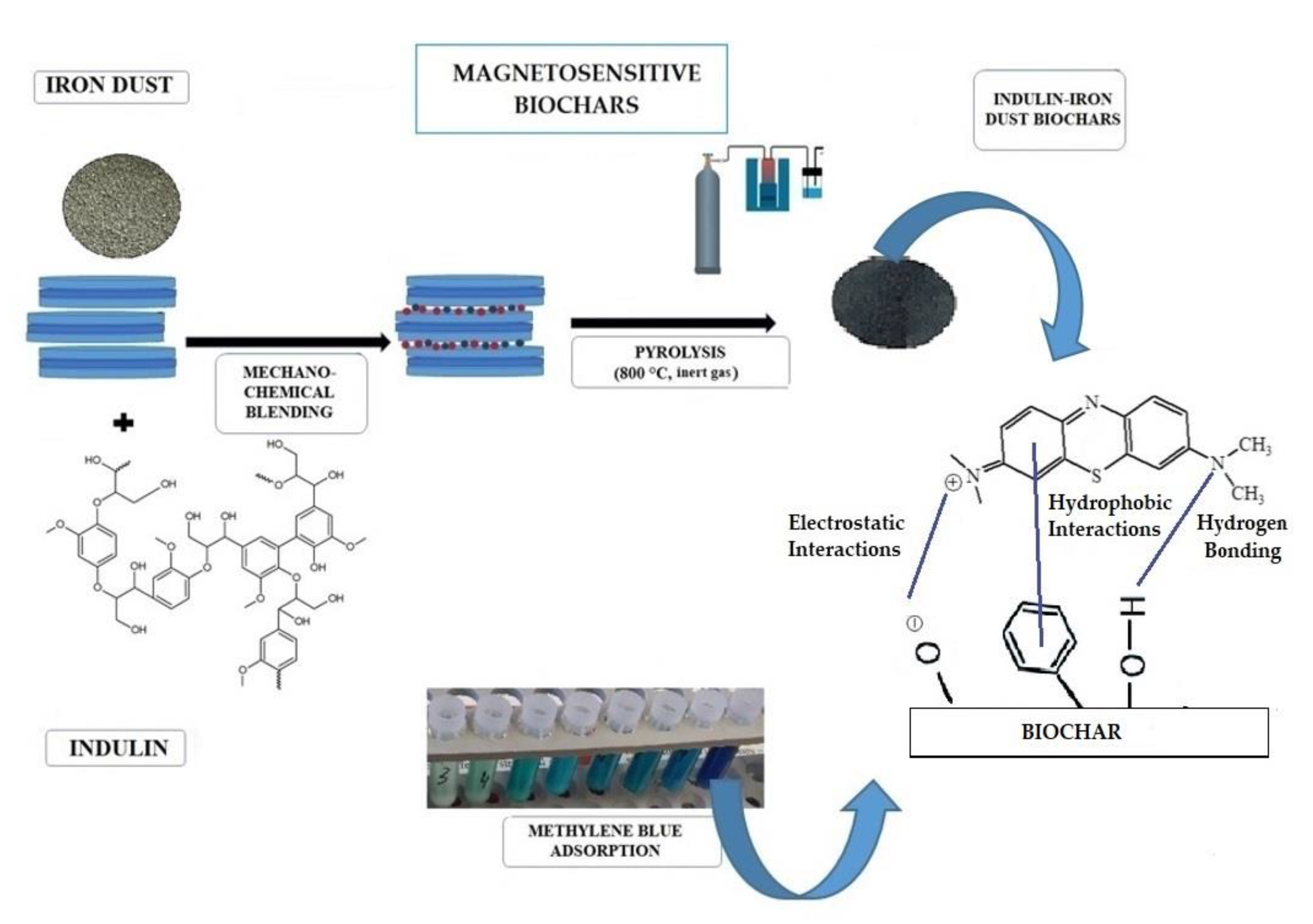
2.2. Materials Preparation
2.3. Methods
2.3.1. Low-Temperature Nitrogen Adsorption-Desorption
2.3.2. Potentiometric Titration
2.3.3. X-ray Diffraction (XRD)
2.3.4. X-ray Fluorescence (XRF)
2.3.5. Raman Spectroscopy
2.3.6. Thermal Analysis (TA)
2.3.7. Equilibrium Adsorption Studies
3. Results and Discussion
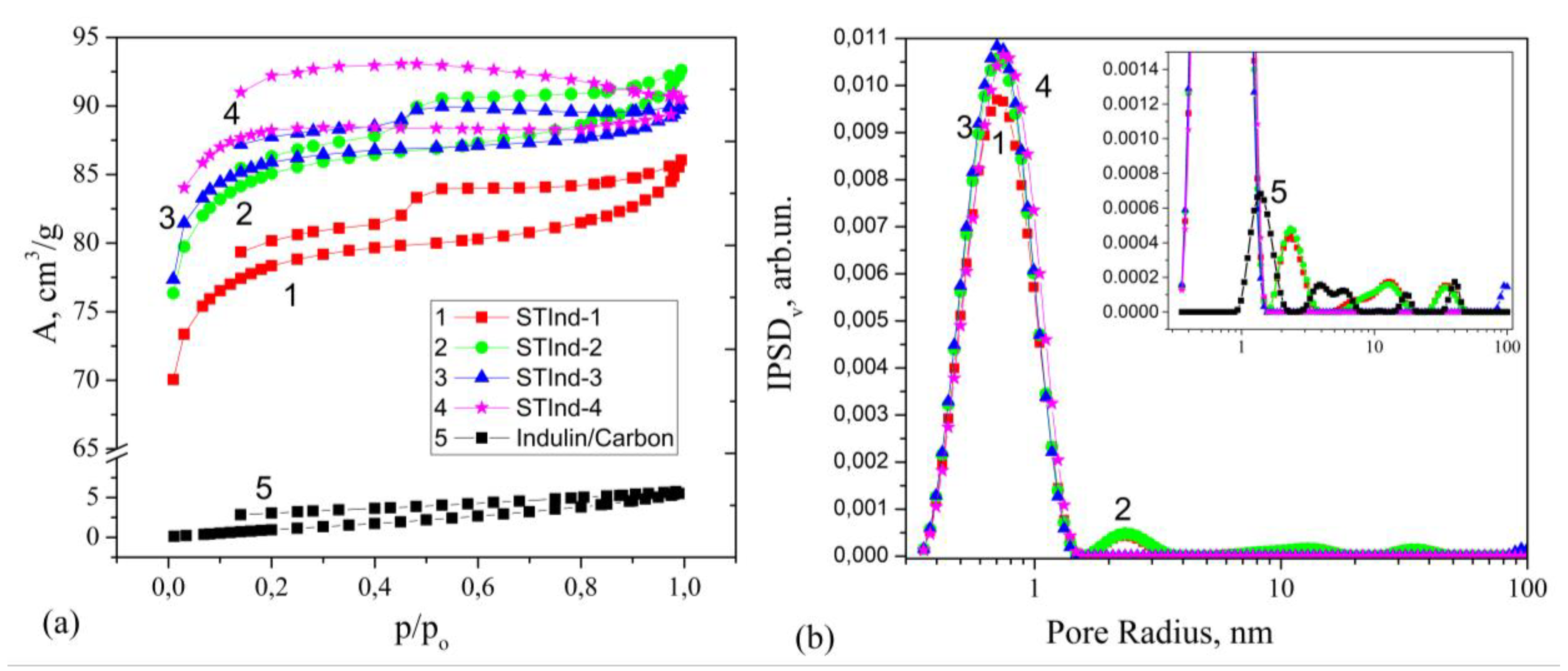
3.1. Low-Temperature Nitrogen Adsorption-Desorption
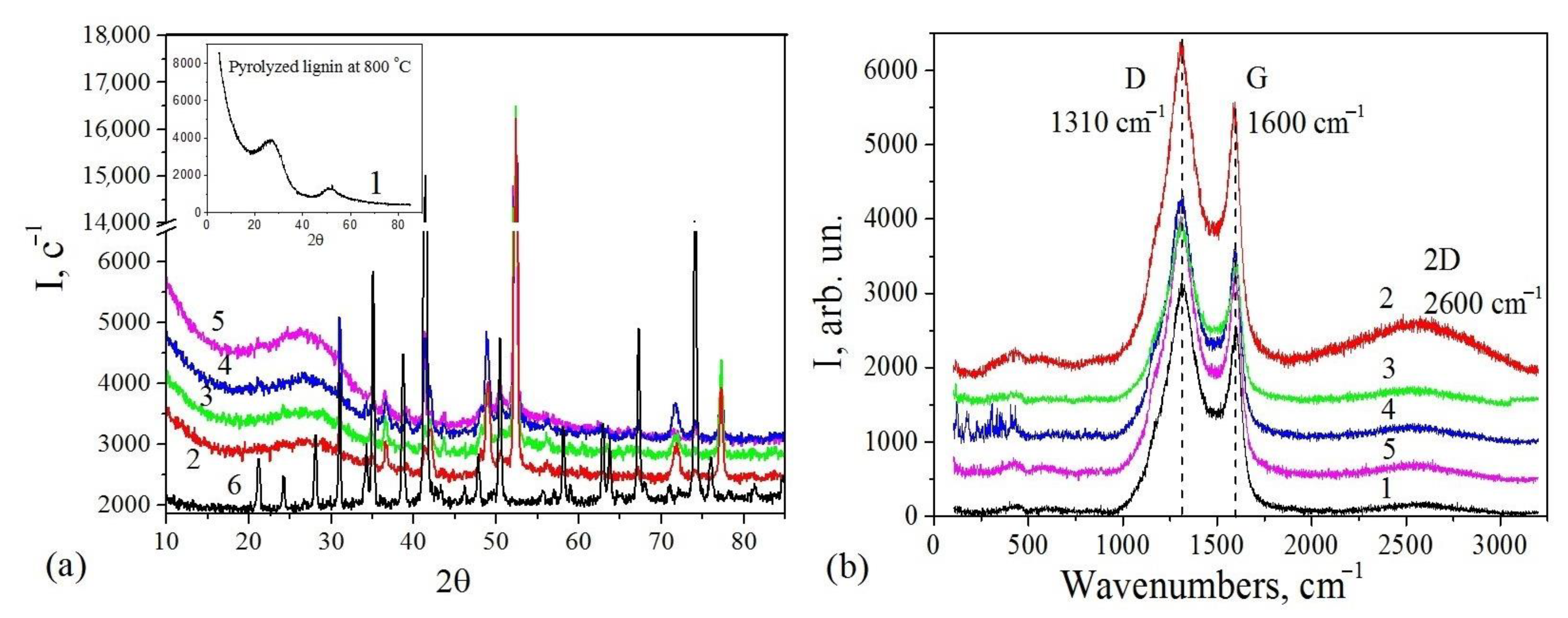
3.2. X-ray Investigations
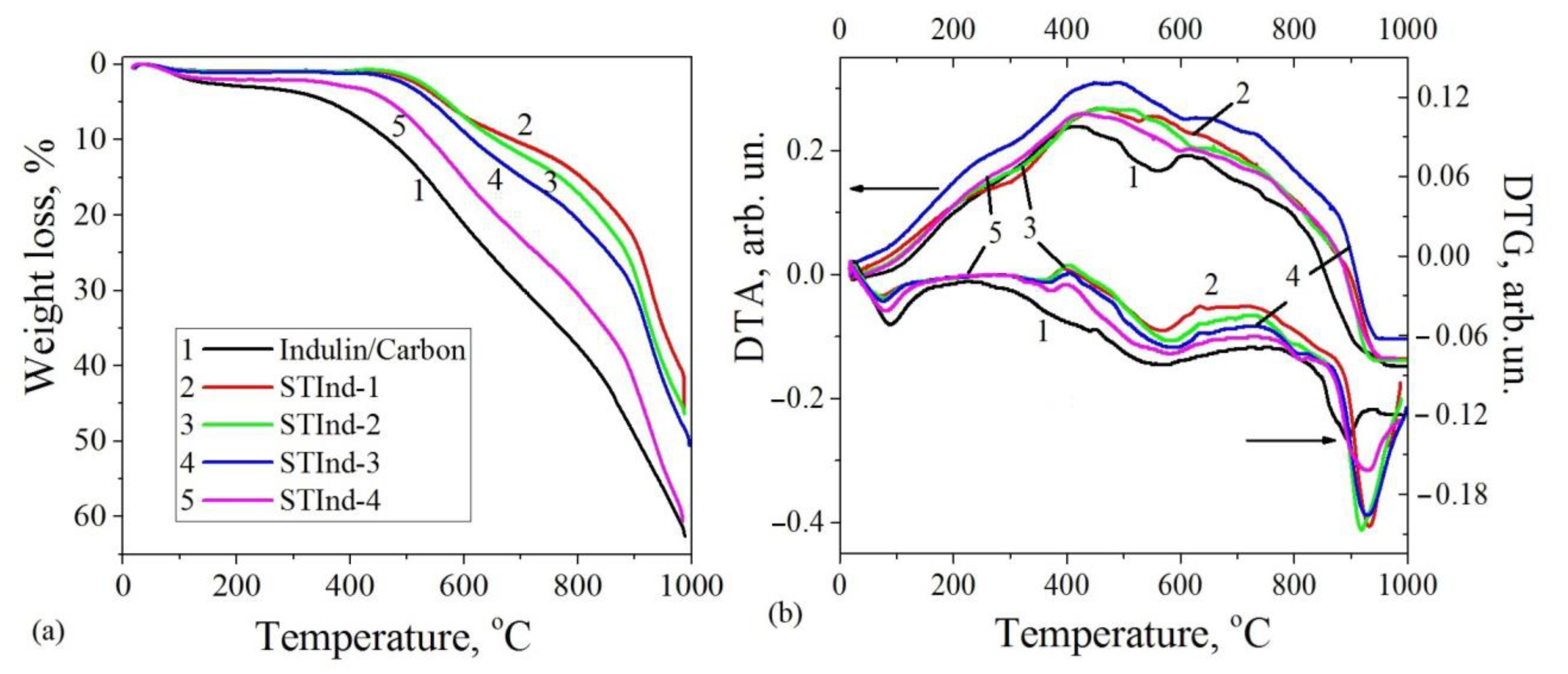
3.3. Thermal Analysis
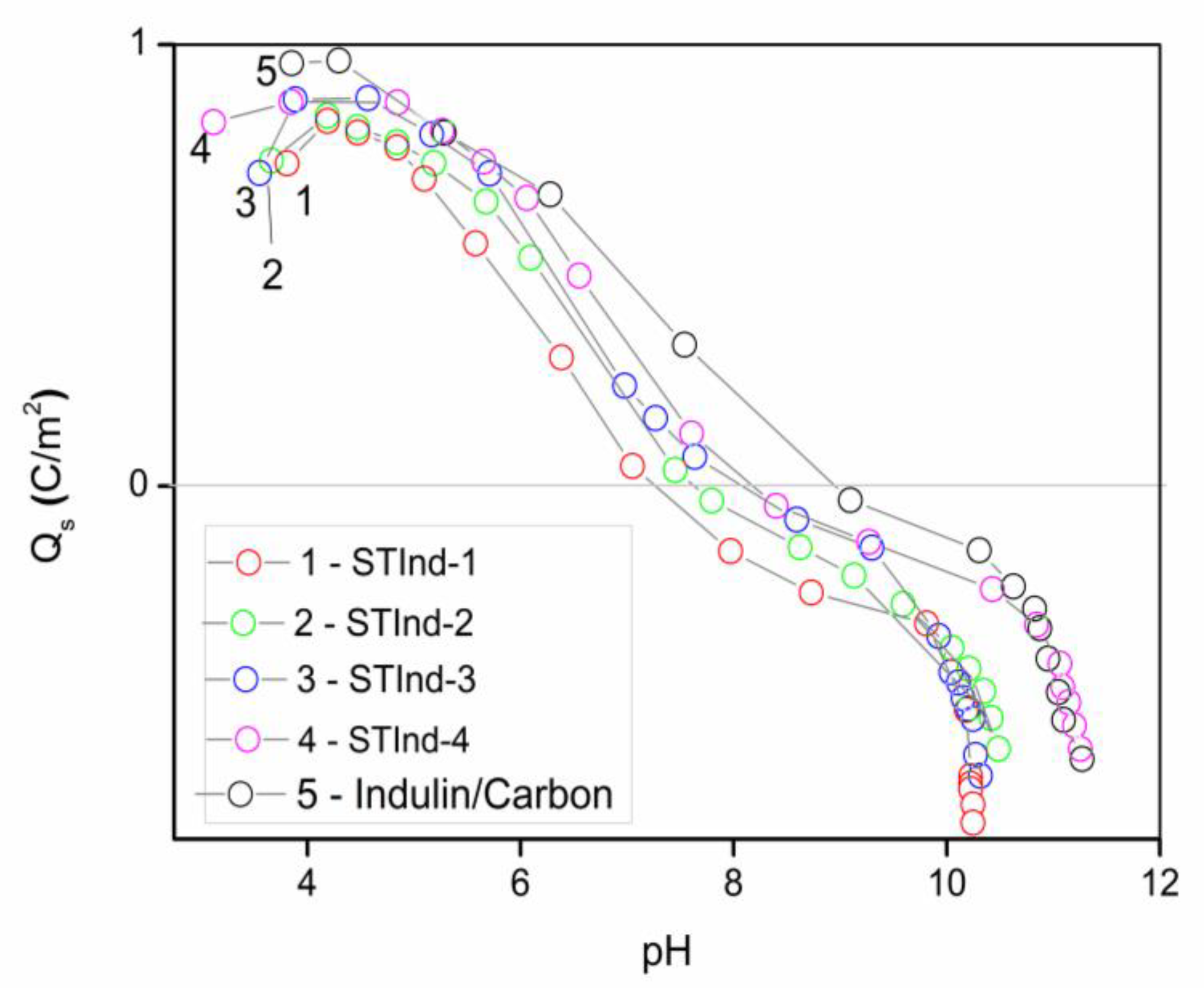
3.4. Potentiometric Titration
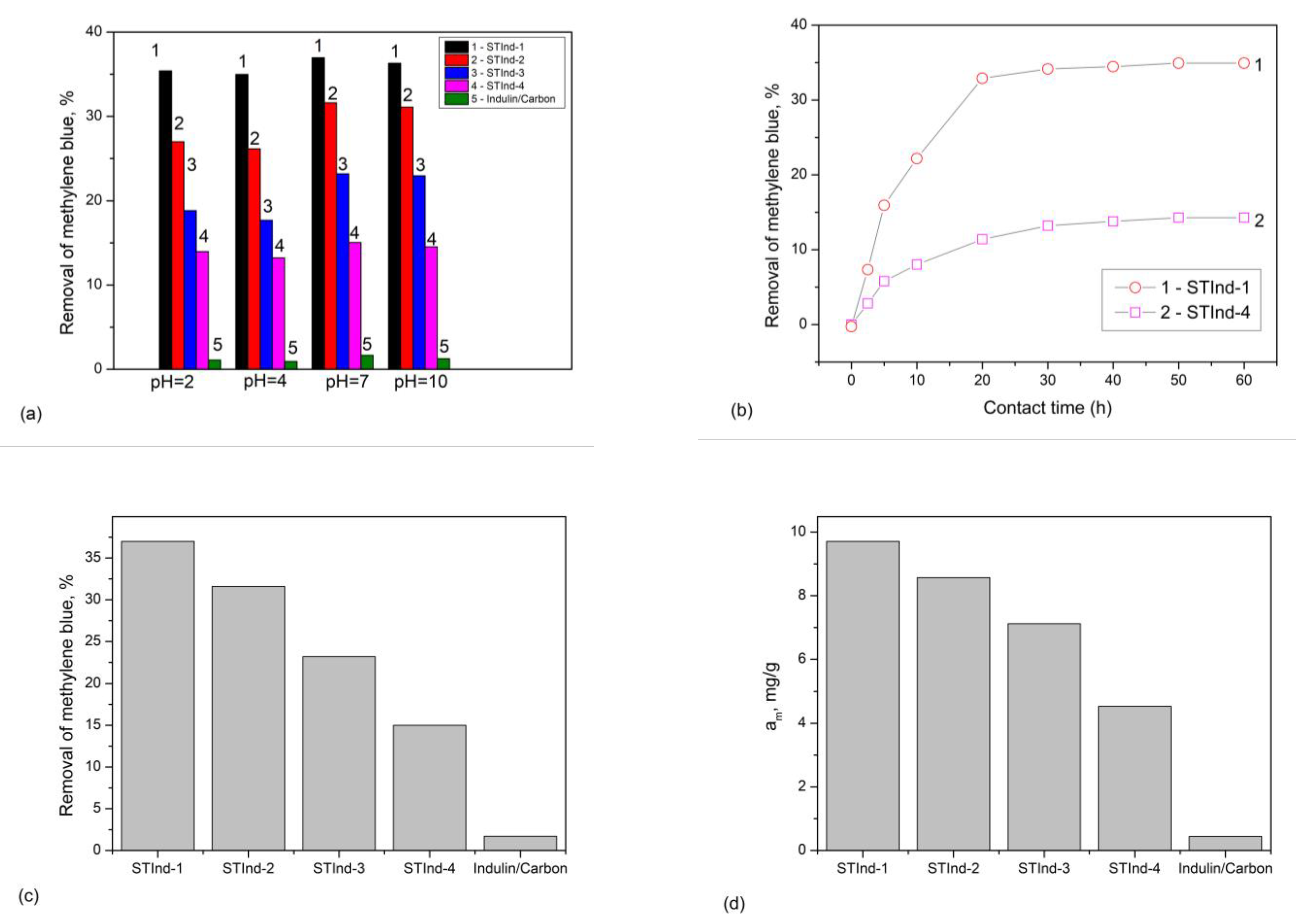
3.5. Adsorption Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Budarin, H.L.V.; Clark, J.H. Rapid and Efficient Adsorption of Methylene Blue Dye From Aqueous Solution by Hierarchically Porous, Activated Starbons®: Mechanism and Porosity Dependence. J. Hazard. Mater. 2022, 436, 129174. [Google Scholar] [CrossRef]
- Chen, J.; Tang, C.; Li, X.; Sun, J.; Liu, Y.; Huang, W.; Wang, A.; Lu, Y. Preparation and Modification of Rape Straw Biochar and Its Adsorption Characteristics for Methylene Blue in Water. Water 2022, 14, 3761. [Google Scholar] [CrossRef]
- Imessaoudene, A.; Cheikh, S.; Bollinger, J.C.; Belkhiri, L.; Tiri, A.; Bouzaza, A.; El Jery, A.; Assadi, A.; Amrane, A.; Mouni, L. Zeolite Waste Characterization and Use as Low-Cost, Ecofriendly, and Sustainable Material for Malachite Green and Methylene Blue Dyes Removal: Box–Behnken Design, Kinetics, and Thermodynamics. Appl. Sci. 2022, 12, 7587. [Google Scholar] [CrossRef]
- Zamouchea, M.; Mouni, L.; Ayachia, A.; Merniza, I. Use of commercial activated carbon for the purification of synthetic water polluted by a pharmaceutical product. Desalination Water Treat. 2019, 172, 86–95. [Google Scholar] [CrossRef]
- Zhu, S.; Khan, M.A.; Kameda, T.; Xu, H.; Wang, F.; Xia, M.; Yoshioka, T. New insights into the capture performance and mechanism of hazardous metals Cr3+ and Cd2+ onto an effective layered double hydroxide based material. J. Hazard. Mater. 2022, 426, 128062. [Google Scholar] [CrossRef]
- Bosacka, A.; Zienkiewicz-Strzalka, M.; Wasilewska, M.; Derylo-Marczewska, A.; Podkoscielna, B. Physicochemical and Adsorption Characteristics of Divinylbenzene-co-Triethoxyvinylsilane Microspheres as Materials for the Removal of Organic Compounds. Molecules 2021, 26, 2396. [Google Scholar] [CrossRef]
- Bosacka, A.; Zienkiewicz-Strzalka, M.; Derylo-Marczewska, A.; Chrzanowska, A.; Wasilewska, M.; Sternik, D. Physicochemical, structural, and adsorption properties of chemically and thermally modified activated carbons. Colloids Surf. A Physicochem. Eng. Asp. 2022, 647, 129130. [Google Scholar] [CrossRef]
- Derylo-Marczewska, A.; Sternik, D.; Swiatkowski, A.; Kusmierek, K.; Gac, W.; Buczek, B. Adsorption of phenol from aqueous and cyclohexane solutions on activated carbons with differentiated surface chemistry. Thermochim. Acta 2022, 715, 179299. [Google Scholar] [CrossRef]
- Galaburda, M.; Bosacka, A.; Sternik, D.; Bogatyrov, V.; Oranska., O.; Gun’ko, V.M.; Derylo-Marczewska, A. Development, Synthesis and Characterization of Tannin/Bentonite-Derived Biochar for Water and Wastewater Treatment from Methylene Blue. Water 2022, 14, 2407. [Google Scholar] [CrossRef]
- Norgren, M.; Edlund, H. Lignin: Recent advances and emerging applications. Curr. Opin. Colloid Interface Sci. 2014, 19, 409–416. [Google Scholar] [CrossRef]
- Kakroodi, A.R.; Sain, M. Lignin-Reinforced Rubber Composites. In Lignin in Polymer Composites, 1st ed.; Faruk, O., Sain, M., Eds.; William Andrew Elsevier: Amsterdam, The Netherlands, 2016; pp. 195–206. [Google Scholar] [CrossRef]
- Mammar, A.C.; Mouni, L.; Bollinger, J.C.; Belkhiri, L.; Bouzaza, A.; Assadi, A.A.; Belkacemi, H. Modeling and optimization of process parameters in elucidating the adsorption mechanism of Gallic acid on activated carbon prepared from date stones. Sep. Sci. Technol. 2020, 55, 3113–3125. [Google Scholar] [CrossRef]
- Bouchelkia, N.; Mouni, L.; Belkhiri, L.; Bouzaza, A.; Bollinger, J.C.; Madani, K.; Dahmoune, F. Removal of lead(II) from water using activated carbon developed from jujube stones, a low-cost sorbent. Sep. Sci. Technol. 2016, 51, 1645–1653. [Google Scholar] [CrossRef]
- Chatterjee, S.; Saito, T. Lignin-Derived Advanced Carbon Materials. ChemSusChem 2015, 8, 3941–3958. [Google Scholar] [CrossRef] [PubMed]
- Kane, S.; Ulrich, R.; Harrington, A.; Stadie, N.P.; Ryan, C. Physical and chemical mechanisms that influence the electrical conductivity of lignin-derived biochar. Carbon Trends 2021, 5, 100088. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, X.; Wu, Y.; Wang, Q.; Wang, H.; Li, D. Fast-pyrolysis lignin-biochar as an excellent precursor for high-performance capacitors. Renew. Energy 2022, 198, 1318–1327. [Google Scholar] [CrossRef]
- Demir, M.; Tessema, T.; Farghaly, A.A.; Nyankson, E.; Saraswat, S.K.; Aksoy, B.; Islamoglu, T.; Collinson, M.M.; El-Kaderi, H.M.; Gupta, R. Lignin-derived heteroatom-doped porous carbons for supercapacitor and CO2 capture applications. Int. J. Energy Res. 2018, 42, 2686–2700. [Google Scholar] [CrossRef]
- Demir, M.; Kahveci, Z.; Aksoy, B.; Palapati, N.K.R.; Subramanian, A.; Cullinan, H.T.; El-Kaderi, H.M.; Harris, C.T.; Gupta, R.B. Graphitic Biocarbon from Metal-Catalyzed Hydrothermal Carbonization of Lignin. Ind. Eng. Chem. Res. 2015, 54, 10731–10739. [Google Scholar] [CrossRef]
- Yi, Z.; Li, C.; Zhang, L.; Zhang, S.; Gao, W.; Wang, S.; Li, B.; Hu, X. Impacts of CO2 atmosphere on property of the biochar from pyrolysis of lignin. J. Anal. Appl. Pyrolysis 2022, 167, 105689. [Google Scholar] [CrossRef]
- Dai, Q.; Liu, Q.; Zhang, X.; Cao, L.; Hu, B.; Shao, J.; Ding, F.; Guo, X.; Gao, B. Synergetic effect of co-pyrolysis of sewage sludge and lignin on biochar production and adsorption of methylene blue. Fuel 2022, 324, 124587. [Google Scholar] [CrossRef]
- Wu, F.; Chen, L.; Hu, P.; Zhou, X.; Zhou, H.; Wang, D.; Lu, X.; Mi, B. Comparison of properties, adsorption performance and mechanisms to Cd(II) on lignin-derived biochars under different pyrolysis temperatures by microwave heating. Environ. Technol. Innov. 2022, 25, 102196. [Google Scholar] [CrossRef]
- Liu, D.; Gu, W.; Zhou, L.; Lei, J.; Wang, L.; Zhang, J.; Liu, Y. From biochar to functions: Lignin induced formation of Fe3C in carbon/Fe composites for efficient adsorption of tetracycline from wastewater. Sep. Purif. Technol. 2023, 304, 122217. [Google Scholar] [CrossRef]
- Liu, X.J.; Li, M.F.; Singh, S.K. Manganese-modified lignin biochar as adsorbent for removal of methylene blue. J. Mater. Res. Technol. 2021, 12, 1434–1445. [Google Scholar] [CrossRef]
- Li, Y.; Wang, F.; Miao, Y.; Mai, Y.; Li, H.; Chen, X.; Chen, J. A lignin-biochar with high oxygen-containing groups for adsorbing lead ion prepared by simultaneous oxidization and carbonization. Bioresour. Technol. 2020, 307, 123165. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, T.; Sun, X.; Bai, L.; Han, C.; Zhang, P. The potential of biochar and lignin-based adsorbents for wastewater treatment: Comparison, mechanism, and application—A review. Ind. Crops Prod. 2021, 166, 113473. [Google Scholar] [CrossRef]
- World Steel Association AISBL. Available online: https://worldsteel.org/steel-topics/statistics/world-steel-in-figures-2022 (accessed on 10 November 2022).
- World Steel Association AISBL. Available online: https://worldsteel.org/publications/policy-papers/co-product-position-paper/ 2022 (accessed on 10 November 2022).
- Nayak, N.P. Characterization of blast furnace flue dust- an assessment for its utilization. Mater. Today. Proc. 2022, 50, 2078–2083. [Google Scholar] [CrossRef]
- Lepa, V.V.; Prognimak, O.D. Blast Furnace Slag Disposal Problems in the Context of the Transition to a Circular Economy. Econ. Her. Donbass 2021, 1, 129–145. [Google Scholar] [CrossRef]
- Özbay, E.; Erdemir, M.; Durmu, H. Utilization and efficiency of ground granulated blast furnace slag on concrete properties—A review. Constr. Build. Mater. 2016, 105, 423–434. [Google Scholar] [CrossRef]
- Das, B.; Prakash, S.; Reddy, P.S.R.; Biswal, S.K.; Mohapatra, B.K.; Misra, V.N. Effective utilization of blast furnace flue dust of integrated steel plants. Eur. J. Miner. Process. Environ. Prot. 2002, 2, 61–68. [Google Scholar]
- Li, Y.; Liu, Y.; Gong, X.; Nie, Z.; Cui, S.; Wang, Z.; Chen, W. Environmental impact analysis of blast furnace slag applied to ordinary Portland cement production. J. Clean. Prod. 2016, 120, 221–230. [Google Scholar] [CrossRef]
- Drachuk, Y.; Stalinskaya, E.; Snitko, E.; Zavgorodnyaya, E.; Jaworska, M.; Savyuk, L.; Cheylyakh, D. Slag waste of metallurgical production. Environmental and economic justification of their use in industry in Ukraine. Polityka Energetyczna Energy Policy J. 2021, 24, 169–182. [Google Scholar] [CrossRef]
- Galaburda, M.; Kovalska, E.; Hogan, B.T.; Baldycheva, A.; Nikolenko, A.; Dovbeshko, G.I.; Oranska, O.I.; Bogatyrov, V.M. Mechanochemical synthesis of carbon-stabilized Cu/C, Co/C and Ni/C nanocomposites with prolonged resistance to oxidation. Sci. Rep. 2019, 9, 17435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galaburda, V.; Bogatyrov, V.M.; Skubiszewska-ZiĿba, J.; Oranska, O.I.; Sternik, D.; Gun’ko, V.M. Synthesis and structural features of resorcinol formaldehyde resin chars containing nickel nanoparticles. Appl. Surf. Sci. 2016, 360, 722–730. [Google Scholar] [CrossRef]
- Galaburda, M.; Bogatyrov, V.; Sternik, D.; Oranska, O.; Borysenko, M.; Škorvánek, I.; Skwarek, E.; Deryło-Marczewska, A.; Gun’ko, V. Magneto-sensitive carbon-inorganic composites based on particleboard and plywood wastes. Chem. J. Mold. 2021, 16, 68–78. [Google Scholar] [CrossRef]
- Gregg, S.J.; Sing, K.S.W. Adsorption, Surface Area and Porosity. In Berichte der Bunsengesellschaft für Physikalische Chemie, 2nd ed.; Auflage, Academic Press: London, UK, 1982; Volume 86, p. 957. [Google Scholar]
- Gun’ko, V.M.; Mikhalovsky, S.V. Evaluation of slitlike porosity of carbon adsorbents. Carbon 2004, 42, 843–849. [Google Scholar] [CrossRef]
- Neimark, A.V.; Ravikovitch, P.I.; Vishnyakov, A. Adsorption hysteresis in nanopores. Phys. Rev. E 2000, 62, R1493(R). [Google Scholar] [CrossRef] [Green Version]
- Ravikovitch, P.I.; Domhnaill, S.Ó.; Neimark, A.V. Capillary hysteresis in nanopores: Theoretical and experimental studies of nitrogen adsorption on MCM-41. Langmuir 1995, 11, 4765–4772. [Google Scholar] [CrossRef]
- Li, J.; Feng, Y.; Li, H.; Zhang, Q. Study of recycling blast furnace dust by magnetization roasting with straw charcoal as reductant. Physicochem. Probl. Miner. Process. 2022, 58, 149265. [Google Scholar] [CrossRef]
- Ferrari, A.C. Raman spectroscopy of graphene and graphite: Disorder, electron–phonon coupling, doping and nonadiabatic effects. Solid State Commun. 2007, 143, 47–57. [Google Scholar] [CrossRef]
- Galaburda, M.V.; Bogatyrov, V.M.; Borysenko, M.V.; Oranska, O.I.; Sternik, D.; Sidorenko Voitko, I.; Derylo-Marczewska, A. Magnetosensitive C/Fe adsorbents synthesized from lignin. In Proceedings of the Ukrainian Conference with International Participation “Chemistry, Physics and Technology of Surface” Dedicated to the 90th Birthday of Aleksey Chuiko, Academician of NAS of Ukraine, Kyiv, Ukraine, 21–22 October 2020. [Google Scholar] [CrossRef]
- Wang, Y.; Alsmeyer, D.C.; McCreery, R.L. Raman spectroscopy of carbon materials: Structural basis of observed spectra. Chem. Mater. 1990, 2, 557–563. [Google Scholar] [CrossRef]
- Gul, E.; Alrawashdeh, K.A.B.; Masek, O.; Skreiberg, O.; Corona, A.; Zampilli, M.; Wang, L.; Samaras, P.; Yang, Q.; Zhou, H.; et al. Production and use of biochar from lignin and lignin-rich residues (such as digestate and olive stones) for wastewater treatment. J. Anal. Appl. Pyrolysis 2021, 158, 105263. [Google Scholar] [CrossRef]
- Szymański, G.S.; Karpinski, Z.; Biniak, S.; Swiątkowski, A. The effect of the gradual thermal decomposition of surface oxygen species on the chemical and catalytic properties of oxidized activated carbon. Carbon 2022, 40, 2627–2639. [Google Scholar] [CrossRef]
- Derylo-Marczewska, A.; Miroslaw, K.; Marczewski, A.W.; Sternik, D. Studies of adsorption equilibria and kinetics of o-, m-, p-nitro- and chlorophenols on microporous carbons from aqueous solutions. Adsorption 2010, 16, 359–375. [Google Scholar] [CrossRef] [Green Version]
- Bogatyrov, V.M.; Borysenko, M.V.; Oranska, O.I.; Galaburda, M.V.; Makhno, S.M.; Gorbyk, P.P. Synthesis and properties of metal-carbon nanocomposites Ni/C, Co/C and Cu/C with high metal content. Poverhn. 2017, 9, 136–144. [Google Scholar] [CrossRef] [Green Version]
- Azizian, S.; Eris, S.; Wilson, L.D. Re-evaluation of the century-old Langmuir isotherm for modeling adsorption phenomena in solution. Chem. Phys. 2018, 513, 99–104. [Google Scholar] [CrossRef]
- Kujawska, J.; Wasag, H. Biochar: A low-cost adsorbent of Methylene Blue from aqueous solutions. J. Phys. Conf. Ser. 2021, 1736, 012002. [Google Scholar] [CrossRef]
- Ouedrhiri, A.; Lghazi, Y.; Bahar, J.; Ait Himi, M.; El Haimer, C.; Youbi, B.; Khoukhi, M.; Bimaghra, Y. Adsorption of the Methylene Blue Dye in Environmental Water Samples by Biochar Obtained from the Valorization of Argan Shells. Phys. Chem. Res. 2022, 10, 301–313. [Google Scholar] [CrossRef]
- Bordoloi, N.; Deep Dey, M.; Mukhopadhyay, R.; Kataki, R. Adsorption of Methylene blue and Rhodamine B by using biochar derived from Pongamia glabra seed cover. Water Sci. Technol. 2018, 77, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Limaa, R.S.; Paiva e Silva Zanta, C.L.; Meilib, L.; Santos Lins, P.V. Souza dos Santos, G.E.; Tonholo, J. Fenton-based processes for the regeneration of biochar from Syagrus coronata biomass used as dye adsorbent. Desalination Water Treat. 2019, 162, 391–398. [Google Scholar] [CrossRef]
- Sun, L.; Wan, S.; Luo, W. Biochars prepared from anaerobic digestion residue, palm bark, and eucalyptus for adsorption of cationic methylene blue dye: Characterization, equilibrium, and kinetic studies. Bioresour. Technol. 2013, 140, 406–413. [Google Scholar] [CrossRef]
- Lesbani, A.; Asri, F.; Palapa, N.R.; Taher, T.; Rachmat, A. Efficient Removal of Methylene Blue by Adsorption Using Composite Based Ca/Al Layered Double Hydroxide-Biochar. Glob. Nest J. 2020, 22, 250–257. [Google Scholar] [CrossRef]
- Sáenz-Alanís, C.A.; García-Reyes, R.B.; Soto-Regalado, E.; García-González, A. Phenol and methylene blue adsorption on heat-treated activated carbon: Characterization, kinetics, and equilibrium studies. Adsorpt. Sci. Technol. 2017, 35, 789–805. [Google Scholar] [CrossRef] [Green Version]
- Kannan, N.; Sundaram, M. Kinetics and mechanism of removal of methylene blue by adsorption on various carbons—A comparative study. Dyes Pigments 2001, 51, 25–40. [Google Scholar] [CrossRef]
| Samples | Components Ratios | |||
|---|---|---|---|---|
| Kraft Lignin (g) | Kraft Lignin (%) | Magnetic Iron Dust (g) | Magnetic Iron Dust (%) | |
| STInd–1 | 12.5 | 71.0 | 5.0 | 29.0 |
| STInd–2 | 12.5 | 76.0 | 4.0 | 24.0 |
| STInd–3 | 12.5 | 81.0 | 3.0 | 19.0 |
| STInd–4 | 12.5 | 91.0 | 1.3 | 9.0 |
| Indulin/Carbon | 12.5 | 100.0 | 0.0 | 0.0 |
| Sample | SBET (m2/g) | Vmicro (cm3/g) | Vmeso (cm3/g) | Vp (cm3/g) | Vmicro/Vp | Vmeso/Vp |
|---|---|---|---|---|---|---|
| STInd–1 | 290 | 0.118 | 0.014 | 0.132 | 0.894 | 0.106 |
| STInd–2 | 315 | 0.128 | 0.014 | 0.142 | 0.901 | 0.099 |
| STInd–3 | 313 | 0.129 | 0.011 | 0.140 | 0.921 | 0.079 |
| STInd–4 | 330 | 0.131 | 0.008 | 0.139 | 0.942 | 0.058 |
| Indulin/Carbon | 7 | - | 0.007 | 0.007 | - | - |
| Component | Fe | Ca | Si | Cl | S | Mg | Mn | Pb | Zn | Al |
|---|---|---|---|---|---|---|---|---|---|---|
| Content (wt%) | 79.763 (±0.100) | 9.965 (±0.031) | 3.951 (±0.080) | 3.843 (±0.050) | 2.166 (±0.058) | 0.134 (±0.010) | 0.089 (±0.017) | 0.045 (±0.006) | 0.027 (±0.004) | 0.015 (±0.003) |
| Sample | Phase Composition | Center (cm−1) | FWHM (cm−1) | ID/IG | Carbon Content (%) | ||
|---|---|---|---|---|---|---|---|
| D | G | D | G | ||||
| DustST | Fe3O4, α-Fe2O3, SiO2, CaCO3 | - | - | - | - | - | - |
| STInd–1 | Fe, FeO, Fe3O4, SiO2, Camorph | 1310 | 1600 | 84.8 | 57.2 | 0.9 | 46.0 |
| STInd–2 | Fe, FeO, Fe3O4, C SiO2, Camorph | 1312 | 1598 | 120.3 | 64.6 | 1.0 | 51.0 |
| STInd–3 | Fe, FeO, Fe3O4, C SiO2, Camorph | 1314 | 1598 | 120.8 | 64.7 | 1.2 | 56.0 |
| STInd–4 | Fe, Fe3O4, FeO, SiO2, Camorph | 1316 | 1598 | 121.5 | 63.2 | 1.3 | 66.0 |
| Indulin/Carbon | Camorph | 1319 | 1586 | 90.5 | 58.1 | 1.2 | 100.0 |
| Sample | Modified Langmuir Isotherm | Freundlich Isotherm | ||||
|---|---|---|---|---|---|---|
| R2 | KML (Dimensionless) | am (mg/g) | R2 | KF (mg/g) | N | |
| STInd–1 | 0.911 | 4336 | 9.71 | 0.933 | 0.949 | 1.49 |
| STInd–2 | 0.980 | 5139 | 8.57 | 0.953 | 1.045 | 1.66 |
| STInd–3 | 0.905 | 4285 | 7.12 | 0.951 | 0.838 | 1.72 |
| STInd–4 | 0.979 | 5960 | 4.53 | 0.972 | 0.669 | 1.89 |
| Indulin/Carbon | 0.988 | 5,889,834 | 0.44 | 0.184 | 0.361 | 12.71 |
| Lp. | Sample | am (mg/g) | Literature |
|---|---|---|---|
| 1 | Zeolite-Waste Materials | 10 | [3] |
| 2 | Tannin/Bentonite-Derived Biochar (TBC-3) | 5 | [9] |
| 3 | Compost Biochar | 13 | [50] |
| 4 | Biomass Biochar | 12 | [50] |
| 5 | Optimal Biochar from Argan Shells Powder | 31 | [51] |
| 6 | Biochar from Pongamia Glabra Seed Cover | 2 | [52] |
| 7 | Ouricuri Endocarp Biochar | 2 | [53] |
| 8 | Biochar from Anaerobic Digestion Residue (BC-R) | 10 | [54] |
| 9 | Biochar from Palm Bark (BC-PB) | 3 | [54] |
| 10 | Biochar from Eucalyptus (BC-E) | 2 | [54] |
| 11 | Composite Based on Ca/Al Layered Double Hydroxide-biochar | 19 | [55] |
| 12 | GAC | 131 | [56] |
| 13 | CAC | 980 | [57] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galaburda, M.; Bosacka, A.; Sternik, D.; Oranska, O.; Borysenko, M.; Gun’ko, V.; Derylo-Marczewska, A. Physicochemical and Sorption Characteristics of Carbon Biochars Based on Lignin and Industrial Waste Magnetic Iron Dust. Water 2023, 15, 189. https://doi.org/10.3390/w15010189
Galaburda M, Bosacka A, Sternik D, Oranska O, Borysenko M, Gun’ko V, Derylo-Marczewska A. Physicochemical and Sorption Characteristics of Carbon Biochars Based on Lignin and Industrial Waste Magnetic Iron Dust. Water. 2023; 15(1):189. https://doi.org/10.3390/w15010189
Chicago/Turabian StyleGalaburda, Mariia, Alicja Bosacka, Dariusz Sternik, Olena Oranska, Mykola Borysenko, Volodymyr Gun’ko, and Anna Derylo-Marczewska. 2023. "Physicochemical and Sorption Characteristics of Carbon Biochars Based on Lignin and Industrial Waste Magnetic Iron Dust" Water 15, no. 1: 189. https://doi.org/10.3390/w15010189
APA StyleGalaburda, M., Bosacka, A., Sternik, D., Oranska, O., Borysenko, M., Gun’ko, V., & Derylo-Marczewska, A. (2023). Physicochemical and Sorption Characteristics of Carbon Biochars Based on Lignin and Industrial Waste Magnetic Iron Dust. Water, 15(1), 189. https://doi.org/10.3390/w15010189









