Tuning the Photocatalytic Performance of Ni-Zn Ferrite Catalyst Using Nd Doping for Solar Light-Driven Catalytic Degradation of Methylene Blue
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Catalysts
2.3. Characterization Details
2.4. Photo Catalytic Degradation of Methylene Blue
3. Results and Discussions
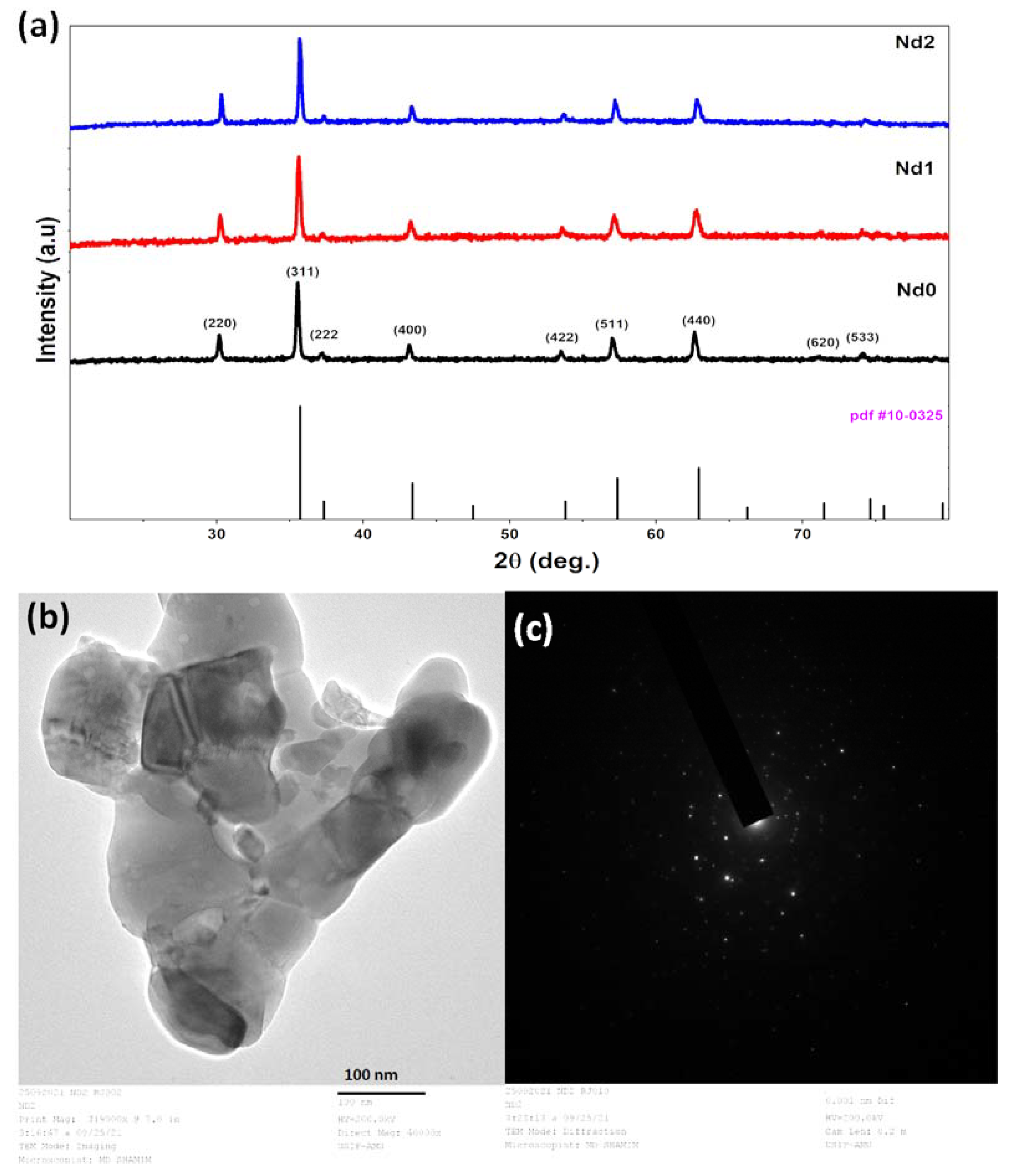
3.1. Characterization of the Catalysts
3.2. SEM Analysis and Elemental Mapping
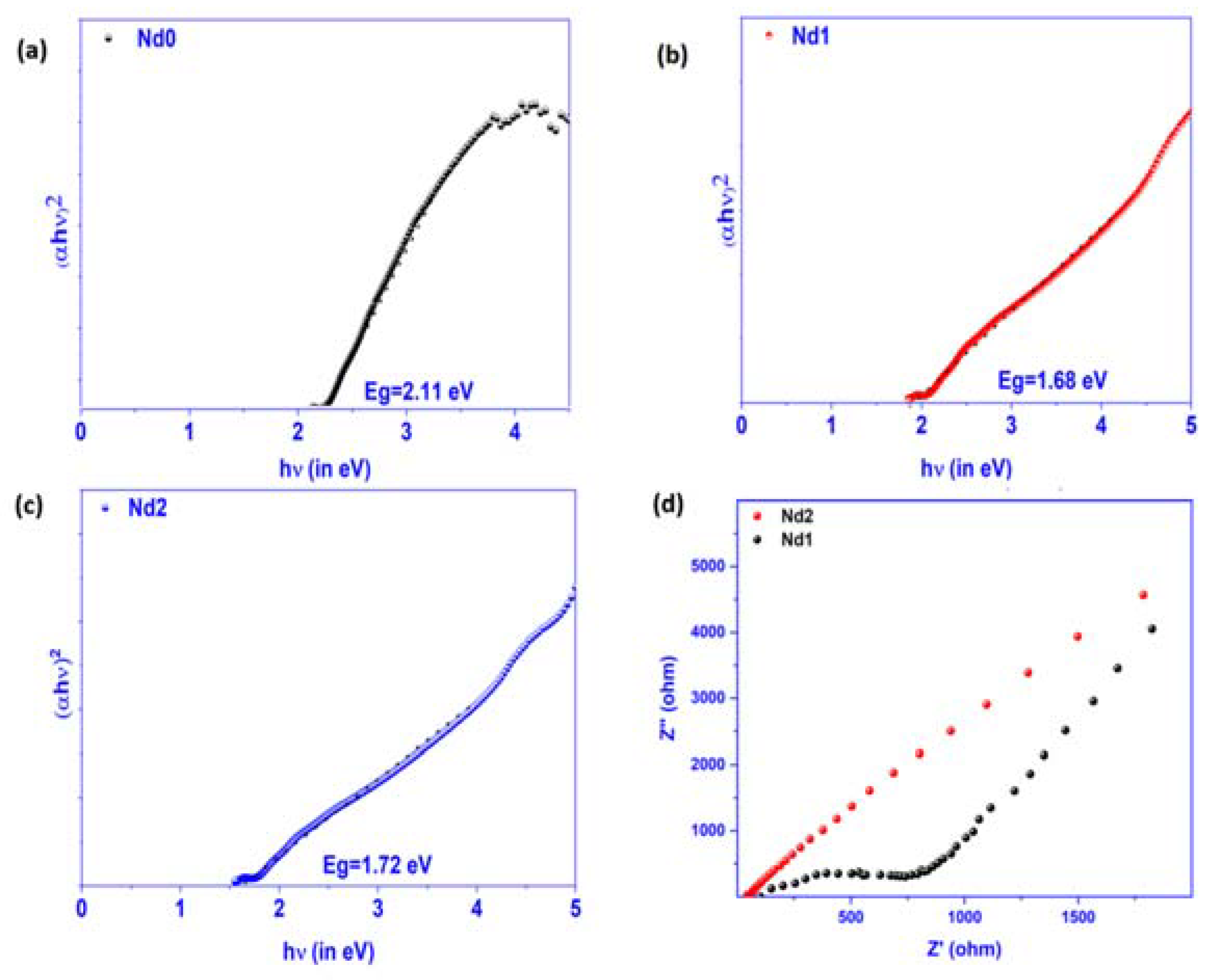
3.3. Band Gap Estimation and Electrochemical Response Studies
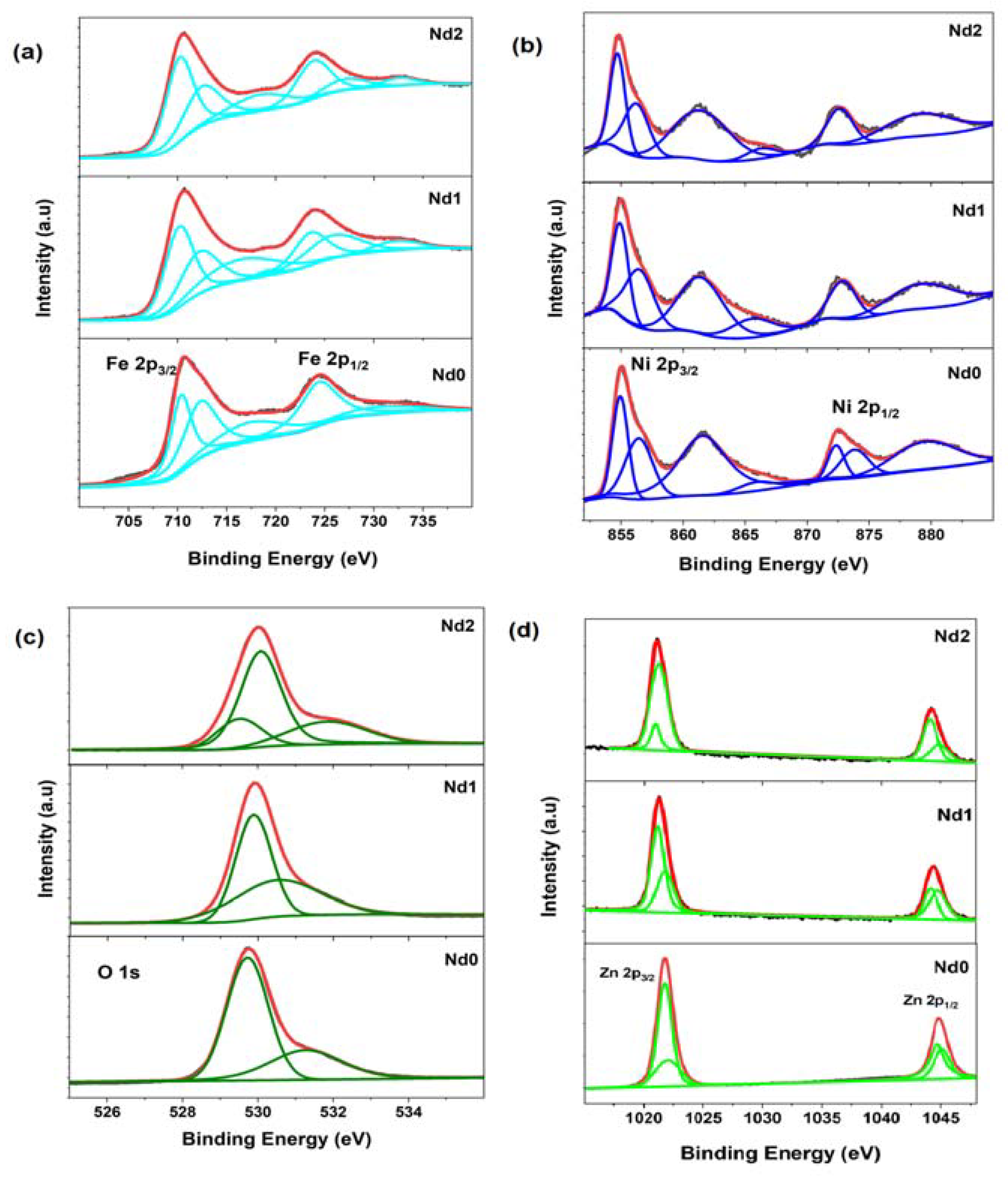
3.4. X-ray Photo Electron Spectroscopy Studies
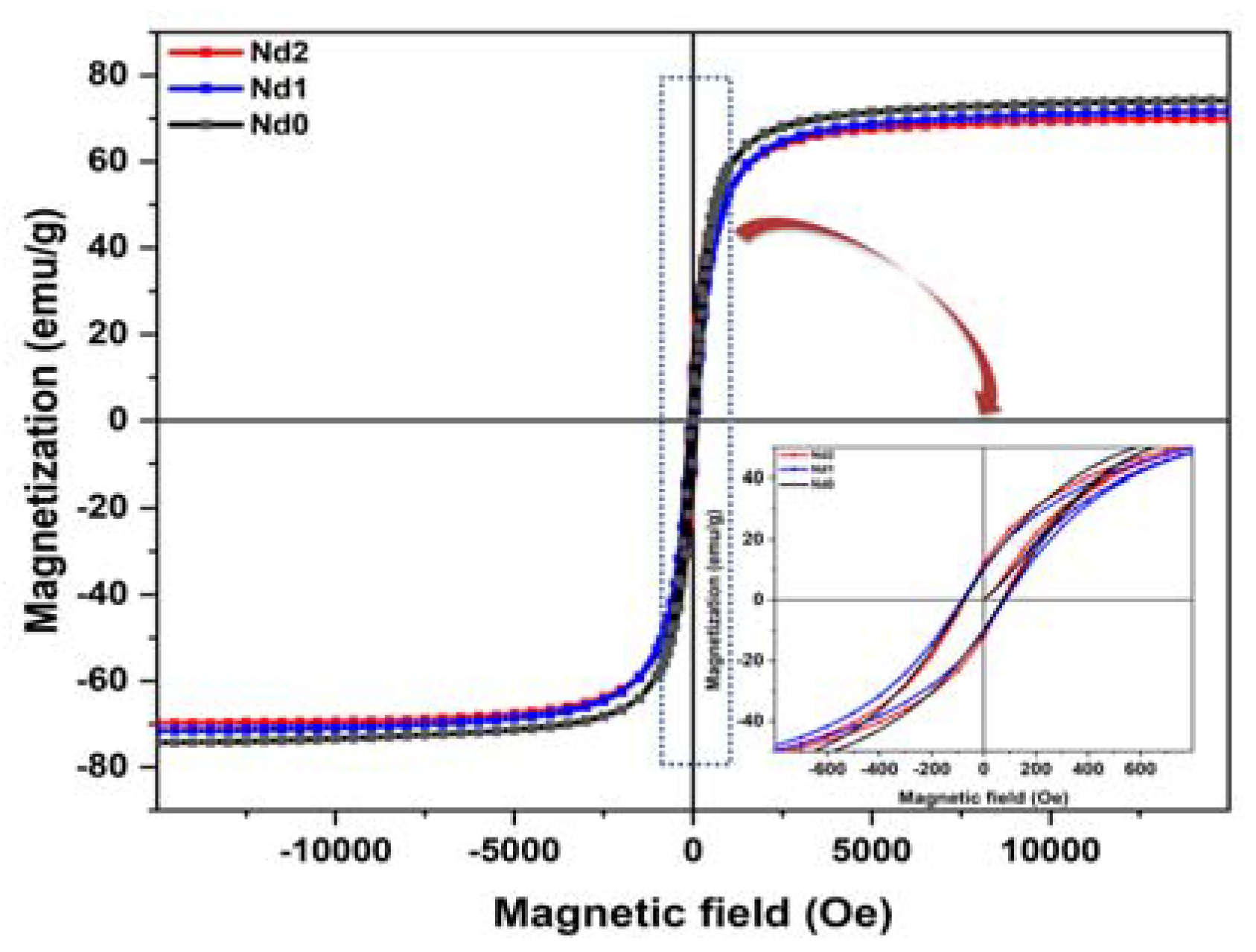
3.5. Magnetic Study of the Catalyst
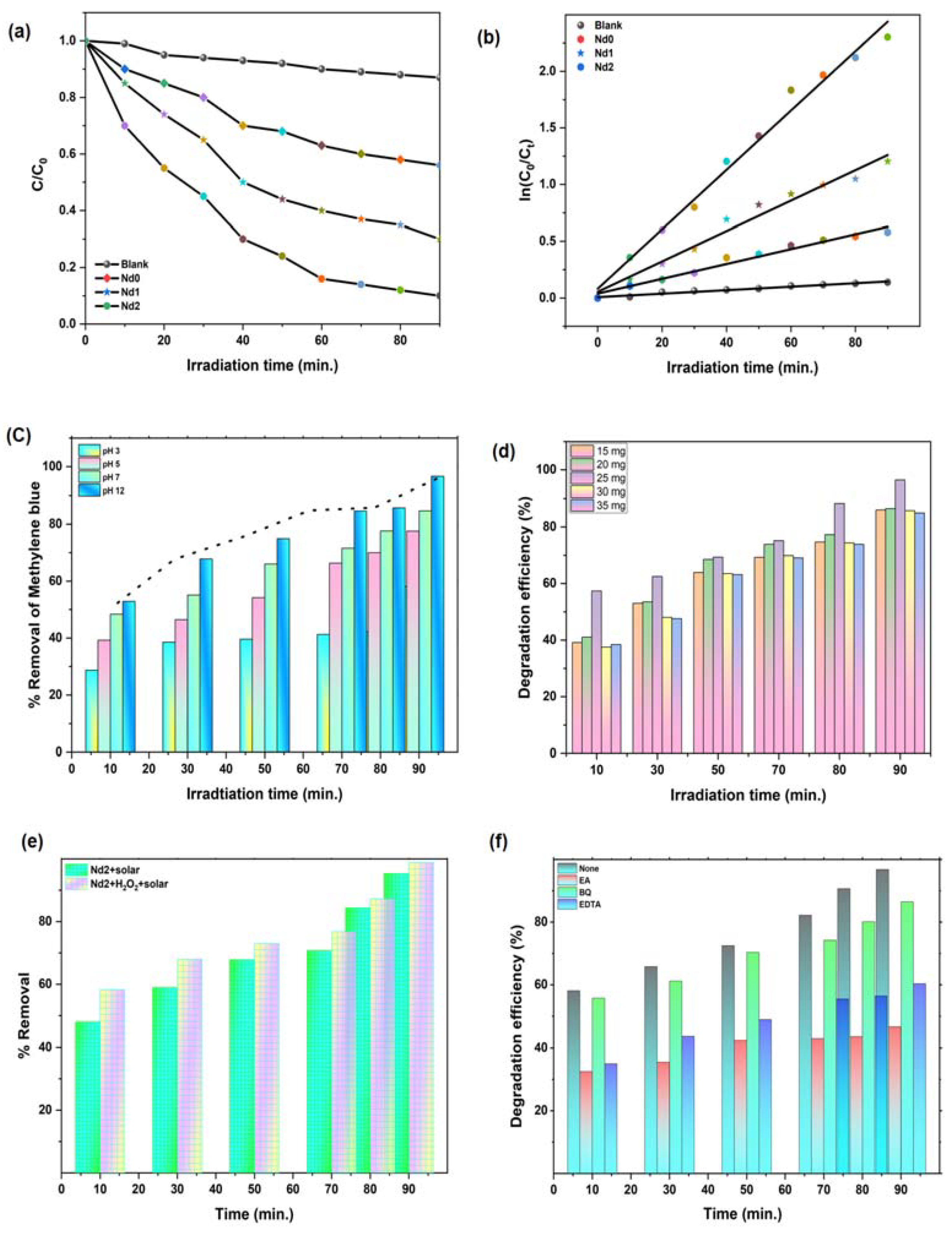
4. Photo Catalytic Activity against Methylene Blue Dye
4.1. Effect of Nd Doping on Ni-Zn Ferrite Catalyst Performance
| Sr. No. | Photocatalyst | Light Source | Dye Concentration | Catalyst Dosage | Removal Efficiency (%) (Reaction Time) | Reference |
|---|---|---|---|---|---|---|
| 1 | MnFe2O4-GSC | Sunlight | 10 mg L−1 | 0.25 g L−1 | 100 % (180 min) | [59] |
| 2 | ZnFe2O4 | UV-Visible, sunlight | 10 mg L−1 | 0.1 g L−1 | 96% 85% (180 min) | [60] |
| 3 | NiCeyFe2-yO4/rGO | Visible light | 5 mg L−1 | 0.5 g L−1 | 94.67 % (70 min) | [61] |
| 4 | CoFe2O4-CNT | visible light lamp | 10 mg L−1 | 0.50 g L−1 | 97% (180 min) | [62] |
| 5 | CoFe2O4-rGO | visible light | 10 mg L−1 | 0.50 g L−1 | 58% (180 min) | |
| 6 | NiCe0.05Fe1.95O4@rGO | Xenon (200 W) | 5 mg L−1 | - | 94.67% (70 min) | [61] |
| 7 | Ni0.96Cd0.04Gd0.04 Fe1.96O4/rGO | visible light | 10 mg L−1 | 0.10 g L−1 | 92.27% (160 min) | [63] |
| 8 | NiFe2O4 | Halogen lamp (200 W) | 10 mg L−1 | - | 98.23% (60 min) | [64] |
| 9 | Co0.1Mg0.9Fe2O4, MgFe2O4 | halogen lamp, sunlight | 10 mg L−1 | 0.10 g g L−1 | 74.50%, 82% (240 min) | [65] |
| 10 | Ni0.5Zn0.5Nd0.05Fe1.95O4 (Nd2) | Solar-light | 20 mg L−1 | 0.25 g L−1 | 98% (90 min) | This work |
4.2. Effect of Reaction Parameters
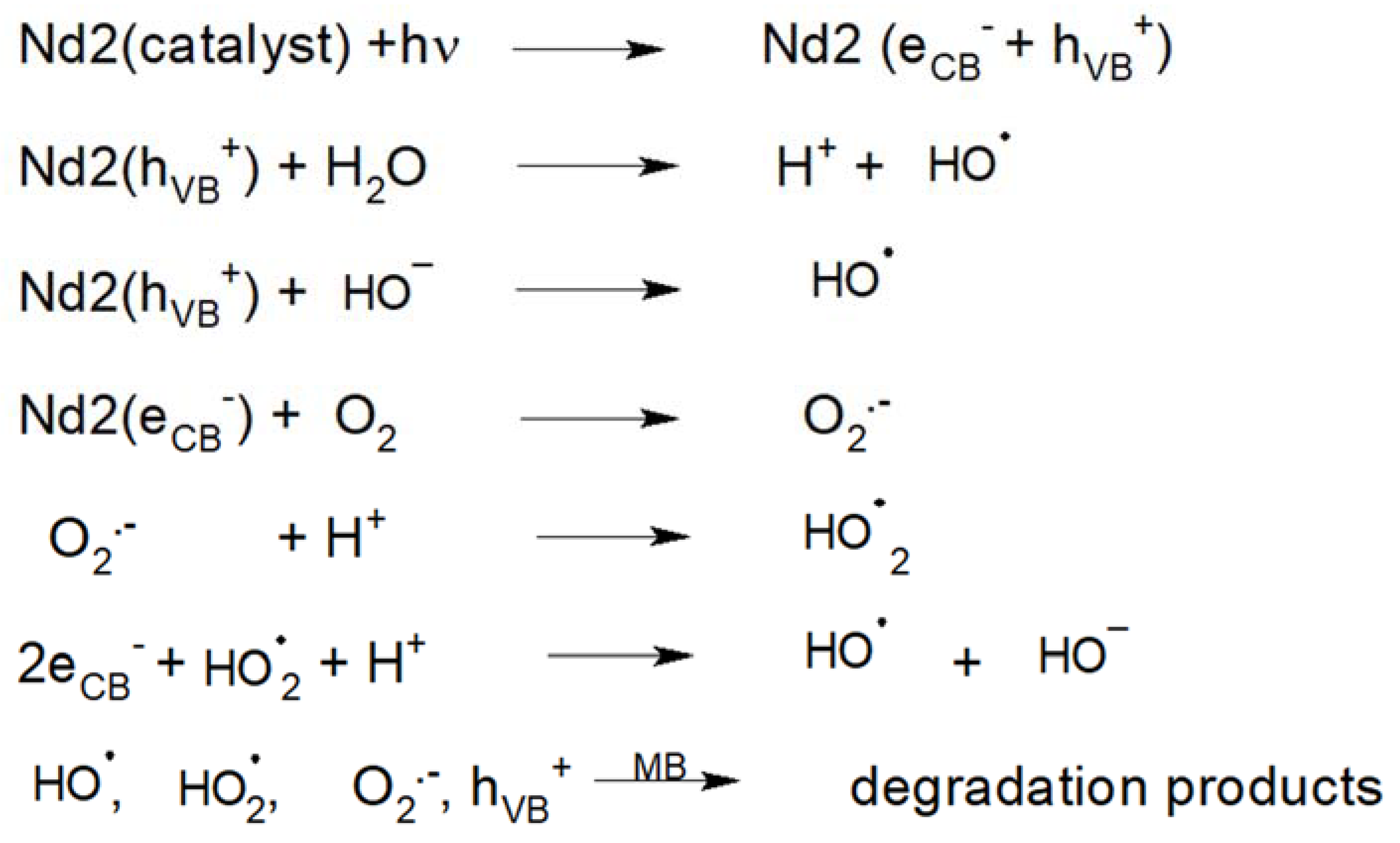
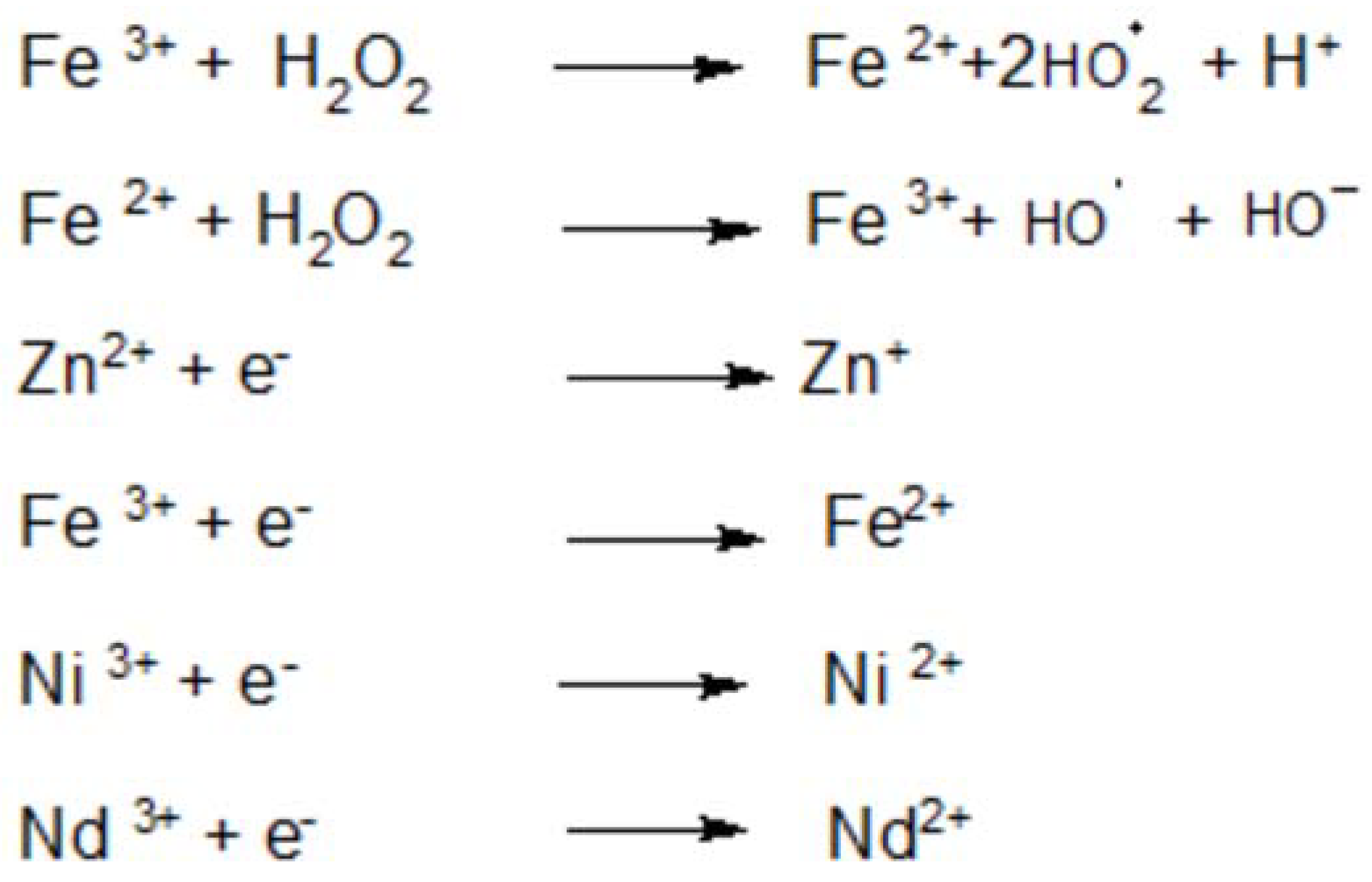
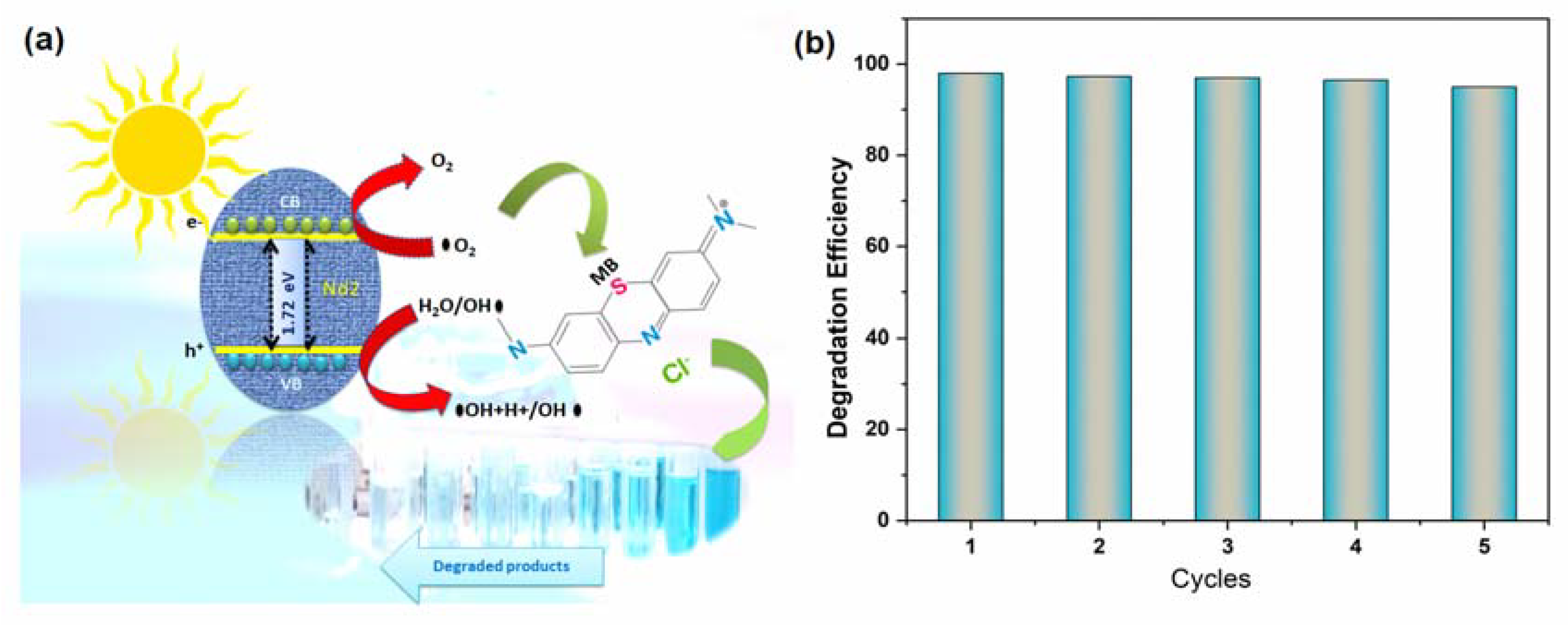
4.3. Degradation Mechanism Insight
4.4. Stability and Reusability Studies
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumar, A.; Kumar, A.; Sharma, G.; Al-Muhtaseb, A.a.H.; Naushad, M.; Ghfar, A.A.; Guo, C.; Stadler, F.J. Biochar-templated g-C3N4/Bi2O2CO3/CoFe2O4 nano-assembly for visible and solar assisted photo-degradation of paraquat, nitrophenol reduction and CO2 conversion. Chem. Eng. J. 2018, 339, 393–410. [Google Scholar] [CrossRef]
- Jain, R.; Sikarwar, S. Photocatalytic and adsorption studies on the removal of dye Congo red from wastewater. Int. J. Environ. Pollut. 2006, 27, 158. [Google Scholar] [CrossRef] [Green Version]
- Alshorifi, F.T.; Alswat, A.A.; Salama, R.S. Gold-selenide quantum dots supported onto cesium ferrite nanocomposites for the efficient degradation of rhodamine B. Heliyon 2022, 8, e09652. [Google Scholar] [CrossRef]
- Muthuraman, G.; Teng, T.T. Extraction and recovery of rhodamine B, methyl violet and methylene blue from industrial wastewater using D2EHPA as an extractant. J. Ind. Eng. Chem. 2009, 15, 841–846. [Google Scholar] [CrossRef]
- Kant, S.; Pathania, D.; Singh, P.; Dhiman, P.; Kumar, A. Removal of malachite green and methylene blue by Fe0.01Ni0.01Zn0.98O/polyacrylamide nanocomposite using coupled adsorption and photocatalysis. Appl. Catal. B Environ. 2014, 147, 340–352. [Google Scholar] [CrossRef]
- Li, S.; Lin, Q.; Liu, X.; Yang, L.; Ding, J.; Dong, F.; Li, Y.; Irfan, M.; Zhang, P. Fast photocatalytic degradation of dyes using low-power laser-fabricated Cu 2 O–Cu nanocomposites. RSC Adv. 2018, 8, 20277–20286. [Google Scholar] [CrossRef] [Green Version]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A.; et al. Review on Methylene Blue: Its Properties, Uses, Toxicity and Photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Sharma, S.; Sharma, G.; Kumar, A.; Dhiman, P.; AlGarni, T.S.; Naushad, M.; Alothman, Z.A.; Stadler, F.J. Controlled synthesis of porous Zn/Fe based layered double hydroxides: Synthesis mechanism, and ciprofloxacin adsorption. Sep. Purif. Technol. 2021, 278, 119481. [Google Scholar] [CrossRef]
- Natarajan, S.; Bajaj, H.C.; Tayade, R.J. Recent advances based on the synergetic effect of adsorption for removal of dyes from waste water using photocatalytic process. J. Environ. Sci. 2018, 65, 201–222. [Google Scholar] [CrossRef]
- Gupta, V.K.; Kumar, R.; Nayak, A.; Saleh, T.A.; Barakat, M.A. Adsorptive removal of dyes from aqueous solution onto carbon nanotubes: A review. Adv. Colloid Interface Sci. 2013, 193, 24–34. [Google Scholar] [CrossRef]
- Dhiman, P.; Patial, M.; Kumar, A.; Alam, M.; Naushad, M.; Sharma, G.; Vo, D.-V.N.; Kumar, R. Environmental friendly and robust Mg0.5-xCuxZn0.5Fe2O4 spinel nanoparticles for visible light driven degradation of Carbamazepine: Band shift driven by dopants. Mater. Lett. 2020, 284, 129005. [Google Scholar] [CrossRef]
- Dhiman, P.; Kumar, A.; Shekh, M.; Sharma, G.; Rana, G.; Vo, D.-V.N.; AlMasoud, N.; Naushad, M.; Alothman, Z.A. Robust magnetic ZnO-Fe2O3 Z-scheme hetereojunctions with in-built metal-redox for high performance photo-degradation of sulfamethoxazole and electrochemical dopamine detection. Environ. Res. 2021, 197, 111074. [Google Scholar] [CrossRef]
- Gupta, V.K.; Jain, R.; Mittal, A.; Saleh, T.A.; Nayak, A.; Agarwal, S.; Sikarwar, S. Photo-catalytic degradation of toxic dye amaranth on TiO2/UV in aqueous suspensions. Mater. Sci. Eng. C 2012, 32, 12–17. [Google Scholar] [CrossRef]
- Cevallos-Mendoza, J.; Amorim, C.G.; Rodríguez-Díaz, J.M.; Montenegro, M.D.C.B.S.M. Removal of Contaminants from Water by Membrane Filtration: A Review. Membranes 2022, 12, 570. [Google Scholar] [CrossRef]
- Kour, S.; Jasrotia, R.; Puri, P.; Verma, A.; Sharma, B.; Singh, V.P.; Kumar, R.; Kalia, S. Improving photocatalytic efficiency of MnFe2O4 ferrites via doping with Zn2+/La3+ ions: Photocatalytic dye degradation for water remediation. Environ. Sci. Pollut. Res. 2021, 1–16. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Vo, D.-V.N.; Yaashikaa, P.R.; Karishma, S.; Jeevanantham, S.; Gayathri, B.; Bharathi, V.D. Photocatalysis for removal of environmental pollutants and fuel production: A review. Environ. Chem. Lett. 2021, 19, 441–463. [Google Scholar] [CrossRef]
- Im, J.K.; Sohn, E.J.; Kim, S.; Jang, M.; Son, A.; Zoh, K.-D.; Yoon, Y. Review of MXene-based nanocomposites for photocatalysis. Chemosphere 2020, 270, 129478. [Google Scholar] [CrossRef]
- Kumar, A.; Sharma, G.; Naushad, M.; Al-Muhtaseb, A.H.; García-Peñas, A.; Mola, G.T.; Si, C.; Stadler, F.J. Bio-inspired and biomaterials-based hybrid photocatalysts for environmental detoxification: A review. Chem. Eng. J. 2019, 382, 122937. [Google Scholar] [CrossRef]
- Mudhoo, A.; Paliya, S.; Goswami, P.; Singh, M.; Lofrano, G.; Carotenuto, M.; Carraturo, F.; Libralato, G.; Guida, M.; Usman, M.; et al. Fabrication, functionalization and performance of doped photocatalysts for dye degradation and mineralization: A review. Environ. Chem. Lett. 2020, 18, 1825–1903. [Google Scholar] [CrossRef]
- Naushad, M.; Vasudevan, S.; Sharma, G.; Kumar, A.; Alothman, Z.A. Adsorption kinetics, isotherms, and thermodynamic studies for Hg2+ adsorption from aqueous medium using alizarin red-S-loaded amberlite IRA-400 resin. Desalination Water Treat. 2016, 57, 18551–18559. [Google Scholar] [CrossRef]
- Dhiman, P.; Kumar, G.; Batoo, K.; Kumar, A.; Sharma, G.; Singh, M. Effective Degradation of Methylene Blue using ZnO: Fe: Ni Nanocomposites. Mater. Res. Found. 2018, 29. [Google Scholar]
- Dhiman, P.; Dhiman, N.; Kumar, A.; Sharma, G.; Naushad, M.; Ghfar, A.A. Solar active nano-Zn1−xMgxFe2O4 as a magnetically separable sustainable photocatalyst for degradation of sulfadiazine antibiotic. J. Mol. Liq. 2019, 294, 111574. [Google Scholar] [CrossRef]
- Akhtar, M.N.; Nazir, M.S.; Khan, M.A.; Ullah, S.; Assiri, M.A. Preparations and characterizations of Ca doped Ni–Mg–Mn nanocrystalline ferrites for switching field high-frequency applications. Ceram. Int. 2021, 48, 3833–3840. [Google Scholar] [CrossRef]
- Dhiman, P.; Rana, G.; Alshgari, R.A.; Kumar, A.; Sharma, G.; Naushad, M.; Alothman, Z.A. Magnetic Ni–Zn ferrite anchored on g-C3N4 as nano-photocatalyst for efficient photo-degradation of doxycycline from water. Environ. Res. 2023, 216, 114665. [Google Scholar] [CrossRef] [PubMed]
- Alshorifi, F.T.; Ali, S.L.; Salama, R.S. Promotional Synergistic Effect of Cs–Au NPs on the Performance of Cs–Au/MgFe2O4 Catalysts in Catalysis 3,4-Dihydropyrimidin-2(1H)-Ones and Degradation of RhB Dye. J. Inorg. Organomet. Polym. Mater. 2022, 32, 3765–3776. [Google Scholar] [CrossRef]
- Mugutkar, A.B.; Gore, S.K.; Patange, S.M.; Mane, R.S.; Raut, S.D.; Shaikh, S.F.; Ubaidullah, M.; Pandit, B.; Jadhav, S.S. Ammonia gas sensing and magnetic permeability of enhanced surface area and high porosity lanthanum substituted Co–Zn nano ferrites. Ceram. Int. 2022, 48, 15043–15055. [Google Scholar] [CrossRef]
- Jamir, M.; Alam, A.; Borah, J.P. Advancement of Spinel Ferrites for Biomedical Application. In Nanoscale Engineering of Biomaterials: Properties and Applications; Pandey, L.M., Hasan, A., Eds.; Springer Nature Singapore: Singapore, 2022; pp. 227–253. [Google Scholar]
- Tobbala, D.E.; Rashed, A.S.; Salama, R.S.; Ahmed, T.I. Performance enhancement of reinforced concrete exposed to electrochemical magnesium chloride using nano-ferrite zinc-rich epoxy. J. Build. Eng. 2022, 57, 104869. [Google Scholar] [CrossRef]
- Dhiman, P. Hexagonal Ferrites, Synthesis, Properties and Their Applications. Mater. Res. Found. 2021, 112, 336. [Google Scholar]
- Miri, A.; Sarani, M.; Najafidoust, A.; Varma, R.S. Evaluation of photocatalytic performance and cytotoxic activity of green synthesized nickel ferrite nanoparticles. Environ. Prog. Sustain. Energy 2021, 41, e13757. [Google Scholar] [CrossRef]
- Hong, D.; Yamada, Y.; Nagatomi, T.; Takai, Y.; Fukuzumi, S. Catalysis of nickel ferrite for photocatalytic water oxidation using [Ru (bpy)3]2+ and S2O82–. J. Am. Chem. Soc. 2012, 134, 19572–19575. [Google Scholar] [CrossRef]
- Kumari, P.S.; Charan, G.V.; Kumar, D.R. Synthesis, structural, photocatalytic and anti-cancer activity of Zn doped Ni nano chromites by citrate gel auto combustion method. Inorg. Chem. Commun. 2022, 139, 109393. [Google Scholar] [CrossRef]
- Mosleh, M. Synthesis, characterization and optical properties of neodymium doped nickel ferrite nanoparticles prepared by novel sol–gel method. J. Mater. Sci. Mater. Electron. 2016, 27, 6703–6707. [Google Scholar] [CrossRef]
- Chaudhari, N.D.; Nadargi, D.Y.; Kabbur, S.M.; Kambale, R.C.; Das, A.; Suryavanshi, S.S. Investigation of Structural, Morphological and Elastic Properties of Ni-Zn Ferrite Grown with an Oxalate Precursor. J. Electron. Mater. 2022, 51, 2732–2740. [Google Scholar] [CrossRef]
- Chehade, W.; Basma, H.; Abdallah, A.; Hassan, R.S.; Awad, R. Synthesis and magneto-optical studies of novel Ni0.5Zn0.5Fe2O4/ Zn0.95Co0.05O nanocomposite as a candidate for photocatalytic applications. Ceram. Int. 2021, 48, 1238–1255. [Google Scholar] [CrossRef]
- Kumar, N.H.; Ravinder, D.; Babu, T.A.; Venkatesh, N.; Swathi, S.; Prasad, N.K. Development of Cu2+ substituted Ni–Zn ferrite nano-particles and their high-temperature semiconductor behaviour. J. Indian Chem. Soc. 2022, 99, 100362. [Google Scholar] [CrossRef]
- Upadhyay, C.; Mishra, D.; Verma, H.; Anand, S.; Das, R. Effect of preparation conditions on formation of nanophase Ni–Zn ferrites through hydrothermal technique. J. Magn. Magn. Mater. 2003, 260, 188–194. [Google Scholar] [CrossRef]
- Dhiman, P.; Rana, G.; Kumar, A.; Sharma, G.; Vo, D.-V.N.; AlGarni, T.S.; Naushad, M.; Alothman, Z.A. Nanostructured magnetic inverse spinel Ni–Zn ferrite as environmental friendly visible light driven photo-degradation of levofloxacin. Chem. Eng. Res. Des. 2021, 175, 85–101. [Google Scholar] [CrossRef]
- Vergis, B.R.; Kottam, N.; Krishna, R.H.; Kumar, G.A. Comparison of magnetic and dielectric properties of transition metal nanospinel ferrites, MFe2O4, (M = Co, Cu, Ni, Zn) synthesized by one-pot combustion route. Mater. Today Proc. 2021, 49, 870–877. [Google Scholar] [CrossRef]
- Dhiman, P.; Sharma, G.; Alodhayb, A.N.; Kumar, A.; Rana, G.; Sithole, T.; Alothman, Z.A. Constructing a Visible-Active CoFe2O4@Bi2O3/NiO Nanoheterojunction as Magnetically Recoverable Photocatalyst with Boosted Ofloxacin Degradation Efficiency. Molecules 2022, 27, 8234. [Google Scholar] [CrossRef]
- Zhang, S.; Wu, J.; Li, F.; Li, L. Enhanced photocatalytic performance of spinel ferrite (MFe2O4, M = Zn, Mn, Co, Fe, Ni) catalysts: The correlation between morphology–microstructure and photogenerated charge efficiency. J. Environ. Chem. Eng. 2022, 10, 107702. [Google Scholar] [CrossRef]
- Hammouche, J.; Gaidi, M.; Columbus, S.; Omari, M. Enhanced Photocatalytic Performance of Zinc Ferrite Nanocomposites for Degrading Methylene Blue: Effect of Nickel Doping Concentration. J. Inorg. Organomet. Polym. Mater. 2021, 31, 3496–3504. [Google Scholar] [CrossRef]
- Harish, K.N.; Bhojya Naik, H.S.; Prashanth kumar, P.N.; Viswanath, R. Optical and Photocatalytic Properties of Solar Light Active Nd-Substituted Ni Ferrite Catalysts: For Environmental Protection. ACS Sustain. Chem. Eng. 2013, 1, 1143–1153. [Google Scholar] [CrossRef]
- Adam, M.S.S.; Hafez, A.M.; Khalaf, M.M. Rare earth Ce- and Nd-doped spinel nickel ferrites as effective heterogeneous catalysts in the (ep)oxidation of alkenes. J. Iran. Chem. Soc. 2020, 17, 3237–3250. [Google Scholar] [CrossRef]
- Vosoughifar, M.; Kimiay, A. Neodymium-doped copper ferrite: Auto-combustion synthesis, characterization and photocatalytic properties. J. Mater. Sci. Mater. Electron. 2016, 27, 10031–10035. [Google Scholar] [CrossRef]
- Kaur, A.; Salunke, D.B.; Umar, A.; Mehta, S.K.; Sinha, A.S.K.; Kansal, S.K. Visible light driven photocatalytic degradation of fluoroquinolone levofloxacin drug using Ag2O/TiO2 quantum dots: A mechanistic study and degradation pathway. New J. Chem. 2017, 41, 12079–12090. [Google Scholar] [CrossRef]
- Dhiman, P.; Mehta, T.; Kumar, A.; Sharma, G.; Naushad, M.; Ahamad, T.; Mola, G.T. Mg0.5NixZn0.5−xFe2O4 spinel as a sustainable magnetic nano-photocatalyst with dopant driven band shifting and reduced recombination for visible and solar degradation of Reactive Blue-19. Adv. Powder Technol. 2020, 31, 4585–4597. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, V.; Aggarwal, S.; Mandal, U.K.; Kotnala, R. Synthesis of nanocrystalline Ni0.5Zn0.5Fe2O4 ferrite and study of its magnetic behavior at different temperatures. Mater. Sci. Eng. B 2010, 166, 76–82. [Google Scholar] [CrossRef]
- Ateia, E.E.; Ahmed, M.; Salah, L.; El-Gamal, A. Effect of rare earth oxides and La3+ ion concentration on some properties of Ni–Zn ferrites. Phys. B Condens. Matter 2014, 445, 60–67. [Google Scholar] [CrossRef]
- Jadhav, S.A.; Somvanshi, S.B.; Khedkar, M.V.; Patade, S.R.; Jadhav, K.M. Magneto-structural and photocatalytic behavior of mixed Ni–Zn nano-spinel ferrites: Visible light-enabled active photodegradation of rhodamine B. J. Mater. Sci. Mater. Electron. 2020, 31, 11352–11365. [Google Scholar] [CrossRef]
- Mariosi, F.R.; Venturini, J.; Viegas, A.D.C.; Bergmann, C.P. Lanthanum-doped spinel cobalt ferrite (CoFe2O4) nanoparticles for environmental applications. Ceram. Int. 2020, 46, 2772–2779. [Google Scholar] [CrossRef]
- Gupta, N.K.; Ghaffari, Y.; Kim, S.; Bae, J.; Kim, K.S.; Saifuddin, M. Photocatalytic Degradation of Organic Pollutants over MFe2O4 (M = Co, Ni, Cu, Zn) Nanoparticles at Neutral pH. Sci. Rep. 2020, 10, 4942. [Google Scholar] [CrossRef] [Green Version]
- Dhiman, P.; Chand, J.; Verma, S.; Sarveena; Singh, M.P. Ni, Fe Co-doped ZnO nanoparticles synthesized by solution combustion method. AIP Conf. Proc. 2014, 1591, 1443–1445. [Google Scholar]
- Torchynska, T.; Vega Macotela, L.; Khomenkova, L.; Gourbilleau, F.; Lartundo Rojas, L. Annealing impact on emission and phase varying of Nd-doped Si-rich-HfO2 films prepared by RF magnetron sputtering. J. Mater. Sci. Mater. Electron. 2020, 31, 4587–4594. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Kou, Q.; Wang, Z.; Han, D.; Sun, Y.; Yang, J.; Liu, Y.; Yang, L. Effects of Nd concentration on structural and magnetic properties of ZnFe2O4 nanoparticles. J. Mater. Sci. Mater. Electron. 2017, 29, 3665–3671. [Google Scholar] [CrossRef]
- Qian, K.; Yao, Z.; Lin, H.; Zhou, J.; Haidry, A.A.; Qi, T.; Chen, W.; Guo, X. The influence of Nd substitution in Ni–Zn ferrites for the improved microwave absorption properties. Ceram. Int. 2019, 46, 227–235. [Google Scholar] [CrossRef]
- Somnath; Sharma, I.; Kotnala, R.K.; Singh, M.; Kumar, A.; Dhiman, P.; Singh, V.P.; Verma, K.; Kumar, G. Structural, magnetic and Mössbauer studies of Nd-doped Mg-Mn ferrite nanoparticles. J. Magn. Magn. Mater. 2017, 444, 77–86. [Google Scholar] [CrossRef]
- Dalal, M.; Mallick, A.; Mahapatra, A.; Mitra, A.; Das, A.; Das, D.; Chakrabarti, P. Effect of cation distribution on the magnetic and hyperfine behaviour of nanocrystalline Co doped Ni–Zn ferrite (Ni0.4Zn0.4Co0.2Fe2O4). Mater. Res. Bull. 2016, 76, 389–401. [Google Scholar] [CrossRef]
- Luciano, A.J.R.; Soletti, L.D.S.; Ferreira, M.E.C.; Cusioli, L.F.; de Andrade, M.B.; Bergamasco, R.; Yamaguchi, N.U. Manganese ferrite dispersed over graphene sand composite for methylene blue photocatalytic degradation. J. Environ. Chem. Eng. 2020, 8, 104191. [Google Scholar] [CrossRef]
- Ali, N.; Zada, A.; Zahid, M.; Ismail, A.; Rafiq, M.; Riaz, A.; Khan, A. Enhanced photodegradation of methylene blue with alkaline and transition-metal ferrite nanophotocatalysts under direct sun light irradiation. J. Chin. Chem. Soc. 2018, 66, 402–408. [Google Scholar] [CrossRef]
- Rahman, A.; Warsi, M.F.; Shakir, I.; Shahid, M.; Zulfiqar, S. Fabrication of Ce3+ substituted nickel ferrite-reduced graphene oxide heterojunction with high photocatalytic activity under visible light irradiation. J. Hazard. Mater. 2020, 394, 122593. [Google Scholar] [CrossRef]
- Jelokhani, F.; Sheibani, S.; Ataie, A. Adsorption and photocatalytic characteristics of cobalt ferrite-reduced graphene oxide and cobalt ferrite-carbon nanotube nanocomposites. J. Photochem. Photobiol. A Chem. 2020, 403, 112867. [Google Scholar] [CrossRef]
- Rahman, A.; Zulfiqar, S.; Haq, A.U.; Alsafari, I.A.; Qazi, U.Y.; Warsi, M.F.; Shahid, M. Cd-Gd-doped nickel spinel ferrite nanoparticles and their nanocomposites with reduced graphene oxide for catalysis and antibacterial activity studies. Ceram. Int. 2020, 47, 9513–9521. [Google Scholar] [CrossRef]
- Jadhav, S.A.; Khedkar, M.V.; Somvanshi, S.B.; Jadhav, K. Magnetically retrievable nanoscale nickel ferrites: An active photocatalyst for toxic dye removal applications. Ceram. Int. 2021, 47, 28623–28633. [Google Scholar] [CrossRef]
- Dojcinovic, M.P.; Vasiljevic, Z.Z.; Pavlovic, V.P.; Barisic, D.; Pajic, D.; Tadic, N.B.; Nikolic, M.V. Mixed Mg–Co spinel ferrites: Structure, morphology, magnetic and photocatalytic properties. J. Alloy. Compd. 2021, 855, 157429. [Google Scholar] [CrossRef]
- Sharma, G.; Kumar, A.; Naushad, M.; Dhiman, P.; Thakur, B.; García-Peñas, A.; Stadler, F.J. Gum Acacia-Crosslinked-Poly (Acrylamide) Hydrogel Supported C3N4/BiOI Heterostructure for Remediation of Noxious Crystal Violet Dye. Materials 2022, 15, 2549. [Google Scholar] [CrossRef]
- Dhiman, P.; Naushad, M.; Batoo, K.M.; Kumar, A.; Sharma, G.; Ghfar, A.A.; Kumar, G.; Singh, M. Nano FexZn1−xO as a tuneable and efficient photocatalyst for solar powered degradation of bisphenol A from aqueous environment. J. Clean. Prod. 2017, 165, 1542–1556. [Google Scholar] [CrossRef]
- Sharma, R.; Bansal, S.; Singhal, S. Tailoring the photo-Fenton activity of spinel ferrites (MFe2O4) by incorporating different cations (M= Cu, Zn, Ni and Co) in the structure. Rsc Adv. 2015, 5, 6006–6018. [Google Scholar] [CrossRef]
- Abdo, M.; El-Daly, A. Sm-substituted copper-cobalt ferrite nanoparticles: Preparation and assessment of structural, magnetic and photocatalytic properties for wastewater treatment applications. J. Alloy. Compd. 2021, 883, 160796. [Google Scholar] [CrossRef]



| Sample | Nd0 | Nd1 | Nd2 |
|---|---|---|---|
| Bragg R-Factor | 5.58 | 11.00 | 6.00 |
| Rf Factor | 4.45 | 9.36 | 4.48 |
| Volume | 581.107 | 588.942 | 584.982 |
| Rp | 62.00 | 83.8 | 77.7 |
| Rwp | 28.00 | 39.8 | 34.0 |
| Re | 26.00 | 27.00 | 31.7 |
| ꭓ2 | 1.123 | 2.181 | 1.147 |
| De-Convoluted Peak Positions of Fe 2p Spectra (in eV) | |||||
| Nd0 | 710.45 | 712.33 | 717.46 | 724.39 | 731.6 |
| Nd1 | 710.30 | 712.31 | 716.72 | 723.75 | 726.17, 732.18 |
| Nd2 | 710.35 | 712.88 | 718.49 | 724.01 | 727.21, 732.7 |
| De-convoluted peak positions of Ni 2p spectra (in eV) | |||||
| Nd0 | 854.80, 856.57 | 861.46 | 866.20 | 872.34, 873.97 | 879.67 |
| Nd1 | 854.87, 856.4 | 861.16 | 865.75 | 872.86 | 879.46 |
| Nd2 | 854.58, 856.35 | 861.24 | 866.65 | 872.65 | 879.09 |
| De-convoluted peak positions of O 1s spectra (in eV) | |||||
| Nd0 | 529.68 | - | 531.22 | ||
| Nd1 | 529.87 | - | 530.60 | ||
| Nd2 | 529.46 | 530.14 | 531.85 | ||
| De-convoluted peak positions of Zn 2p spectra (in eV) | |||||
| Nd0 | 1121.70 | 1122.10 | 1044.62 | 1045.36 | |
| Nd1 | 1021.31 | 1021.95 | 1044.10 | 1044.92 | |
| Nd2 | 1020.78 | 1021.35 | 1044.37 | 1045.31 | |
| De-Convoluted Peak Positions of Nd 3d Spectra (in eV) | |||||
| Nd1 | 974.48, 978.34 | 982.67 | 994.58 | 1001.82, 1005.35, 1009.70 | |
| Nd2 | 976.04, 980.10 | 982.64 | 995.02 | 1001.46, 1005.16, 1009.34 | |
| Catalyst | Regression Coefficient (R2) | Rate Constant (min.−1) | Kinetics Equation |
|---|---|---|---|
| Nd0 | 0.9720 | 0.0065 | 0.0065*x + 0.0390 |
| Nd1 | 0.9755 | 0.0133 | 0.0133*x + 0.0546 |
| Nd2 | 0.9869 | 0.0262 | 0.0263*x + 0.0814 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhiman, P.; Rana, G.; Dawi, E.A.; Kumar, A.; Sharma, G.; Kumar, A.; Sharma, J. Tuning the Photocatalytic Performance of Ni-Zn Ferrite Catalyst Using Nd Doping for Solar Light-Driven Catalytic Degradation of Methylene Blue. Water 2023, 15, 187. https://doi.org/10.3390/w15010187
Dhiman P, Rana G, Dawi EA, Kumar A, Sharma G, Kumar A, Sharma J. Tuning the Photocatalytic Performance of Ni-Zn Ferrite Catalyst Using Nd Doping for Solar Light-Driven Catalytic Degradation of Methylene Blue. Water. 2023; 15(1):187. https://doi.org/10.3390/w15010187
Chicago/Turabian StyleDhiman, Pooja, Garima Rana, Elmuez A. Dawi, Amit Kumar, Gaurav Sharma, Arun Kumar, and Jayati Sharma. 2023. "Tuning the Photocatalytic Performance of Ni-Zn Ferrite Catalyst Using Nd Doping for Solar Light-Driven Catalytic Degradation of Methylene Blue" Water 15, no. 1: 187. https://doi.org/10.3390/w15010187
APA StyleDhiman, P., Rana, G., Dawi, E. A., Kumar, A., Sharma, G., Kumar, A., & Sharma, J. (2023). Tuning the Photocatalytic Performance of Ni-Zn Ferrite Catalyst Using Nd Doping for Solar Light-Driven Catalytic Degradation of Methylene Blue. Water, 15(1), 187. https://doi.org/10.3390/w15010187








