Hydrogeochemical Characteristics Refine the Conceptual Model of Groundwater Flow in Wood Buffalo National Park, Canada
Abstract
:1. Introduction
2. Geographical, Geological and Hydrogeological Setting
2.1. Physiography and Climate
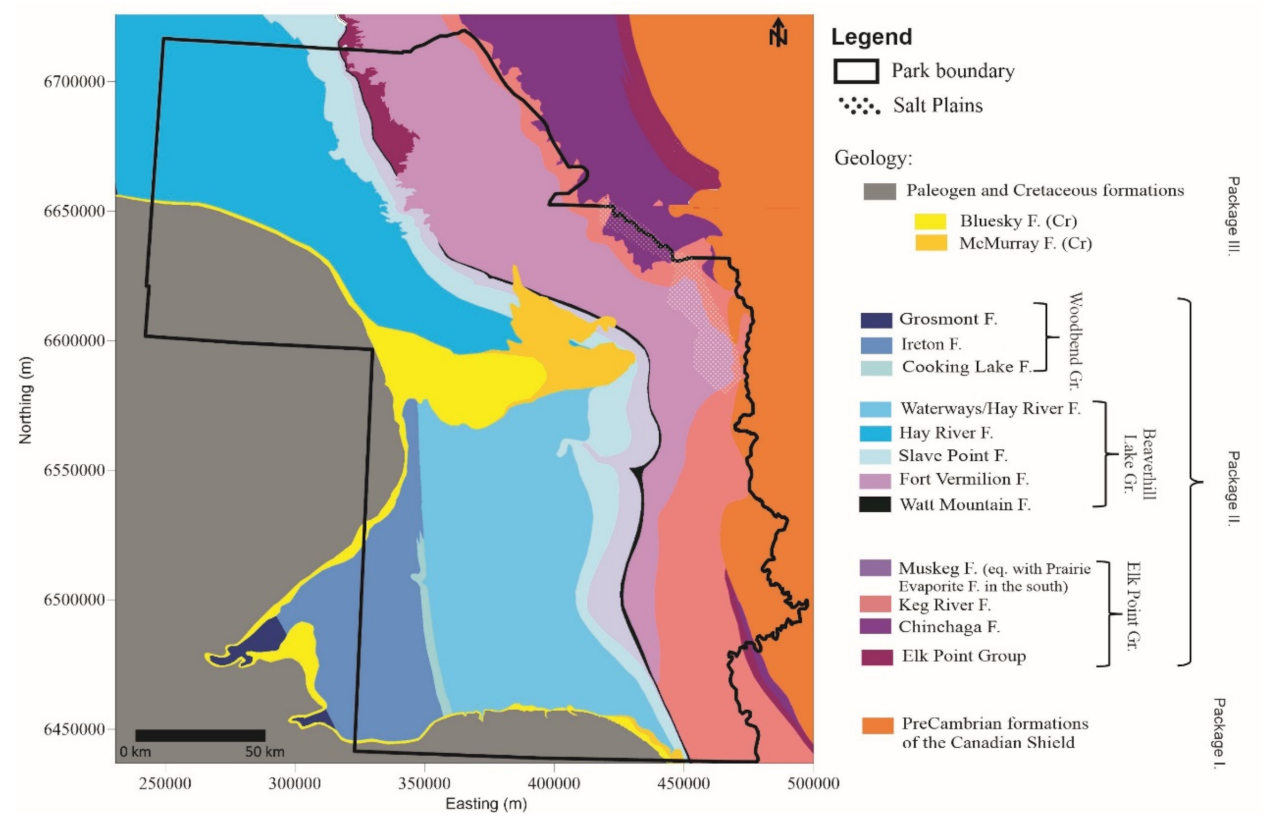
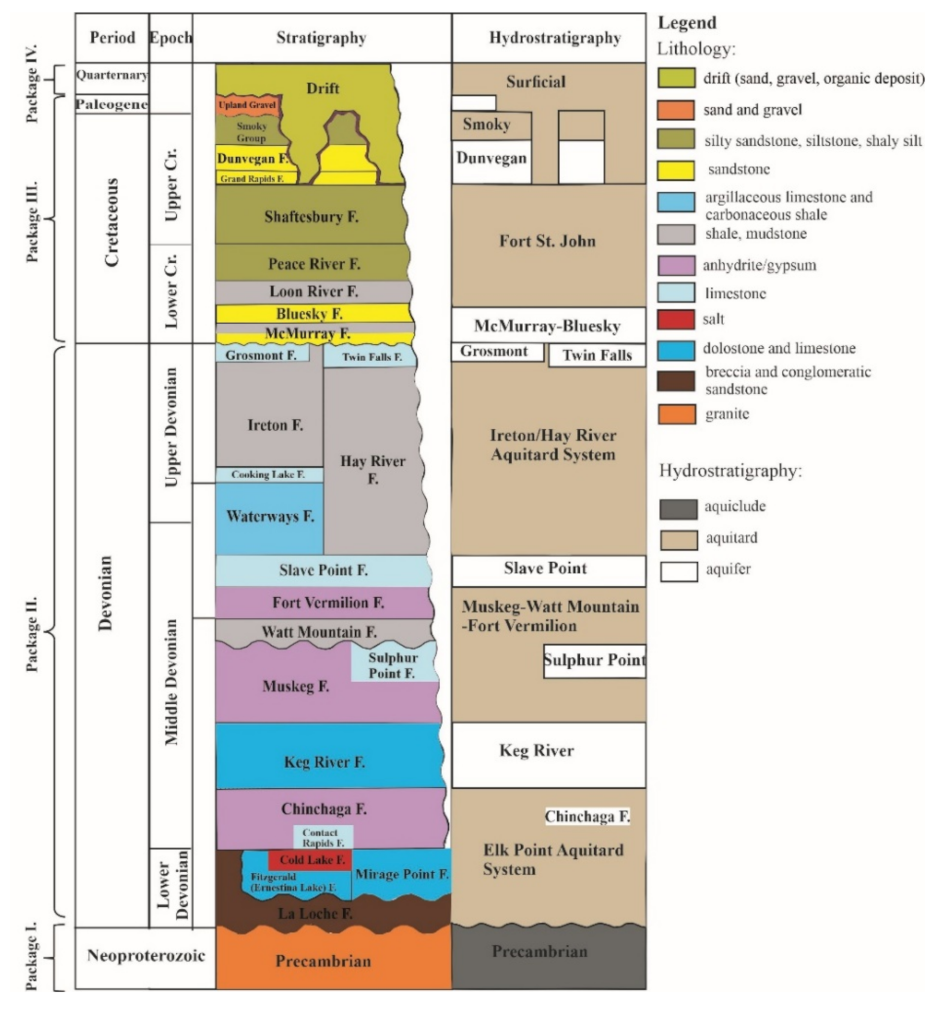
2.2. Geology and Hydrostratigraphy
3. Data and Methods
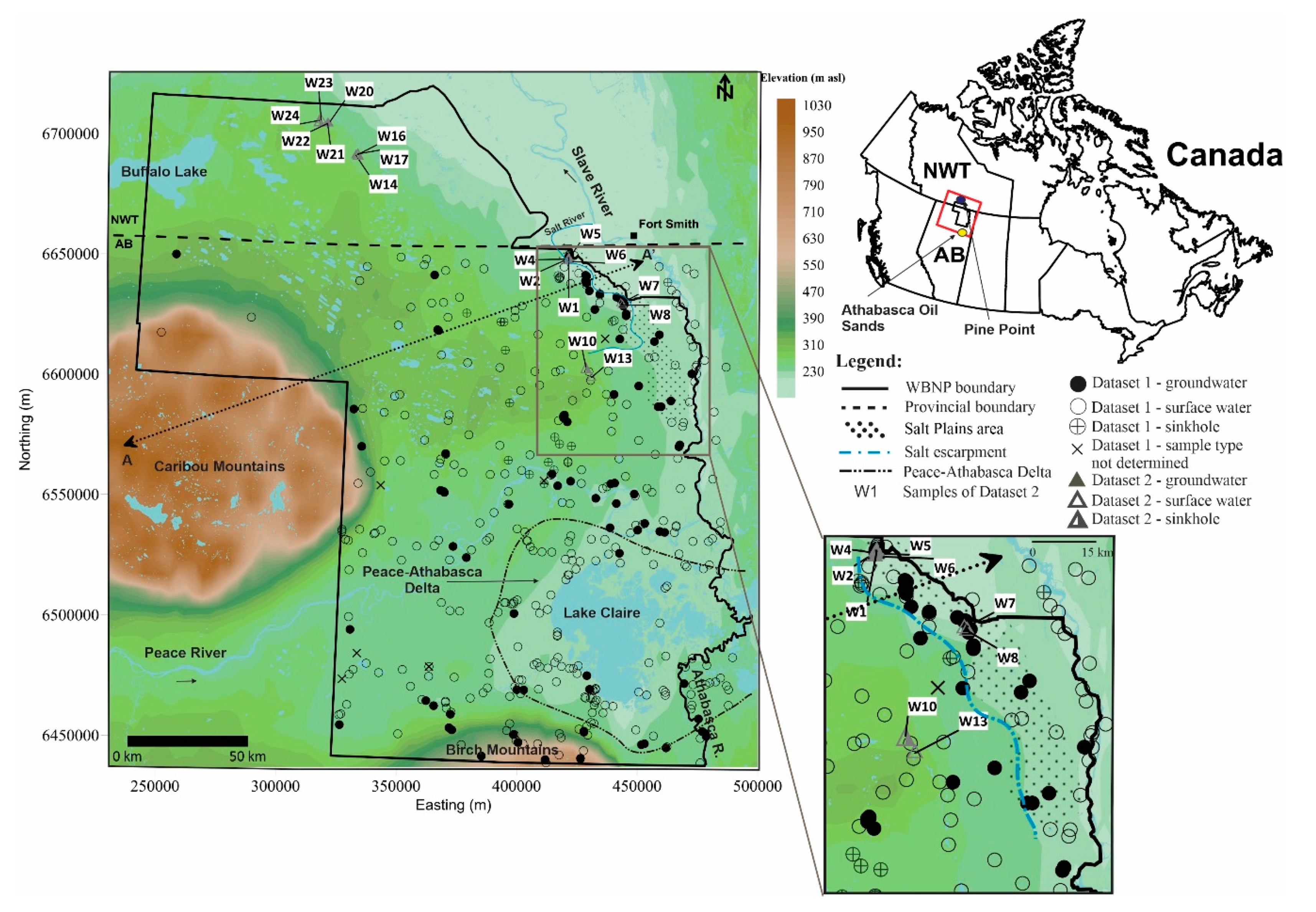
3.1. Data
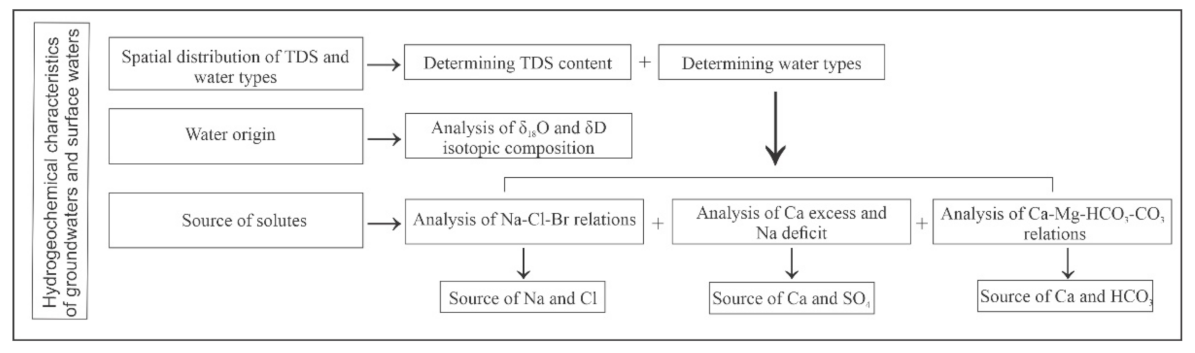
3.2. Data Validation and Evaluation Methods
4. Results
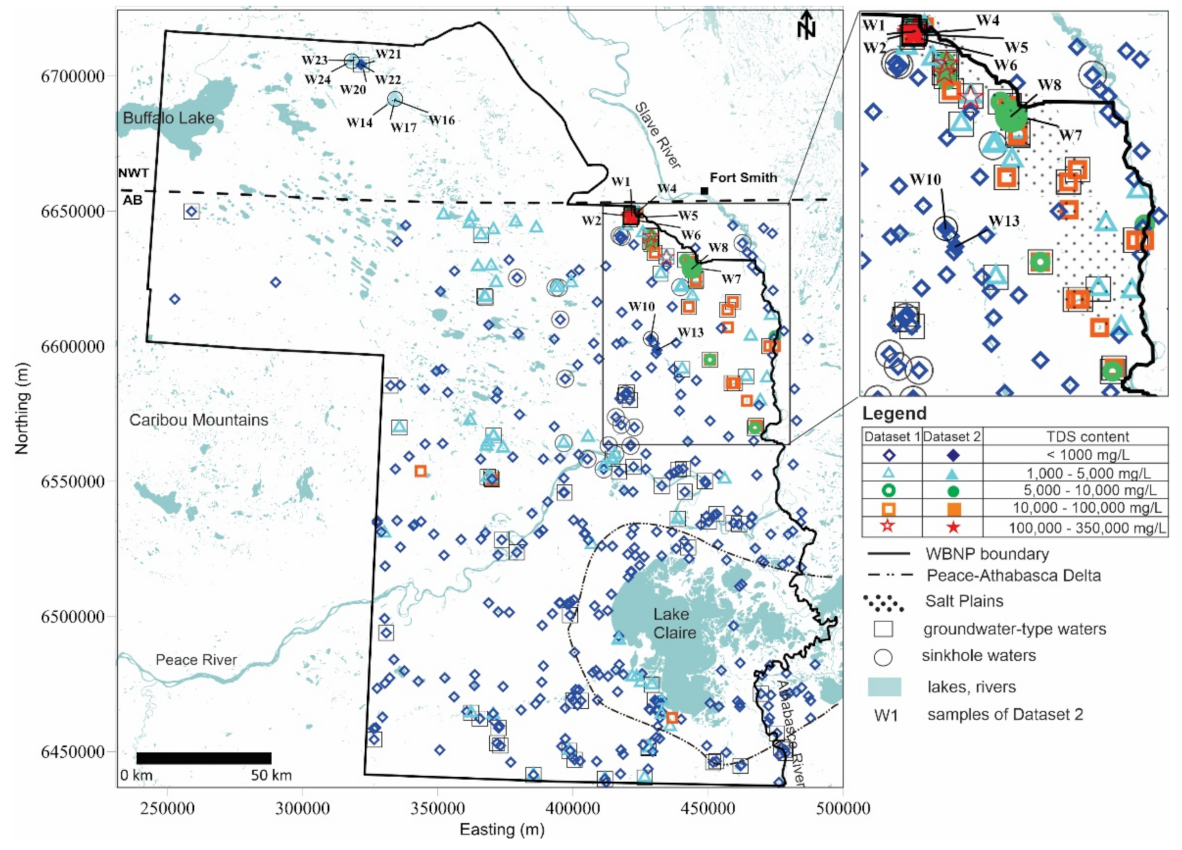
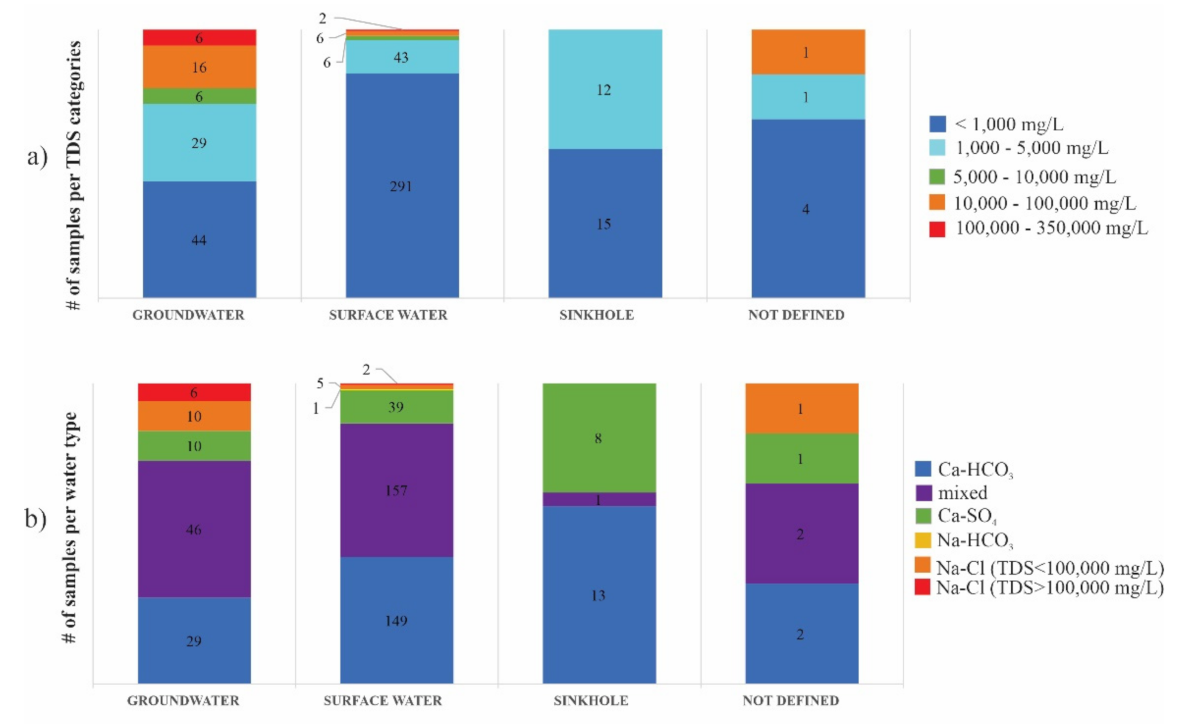
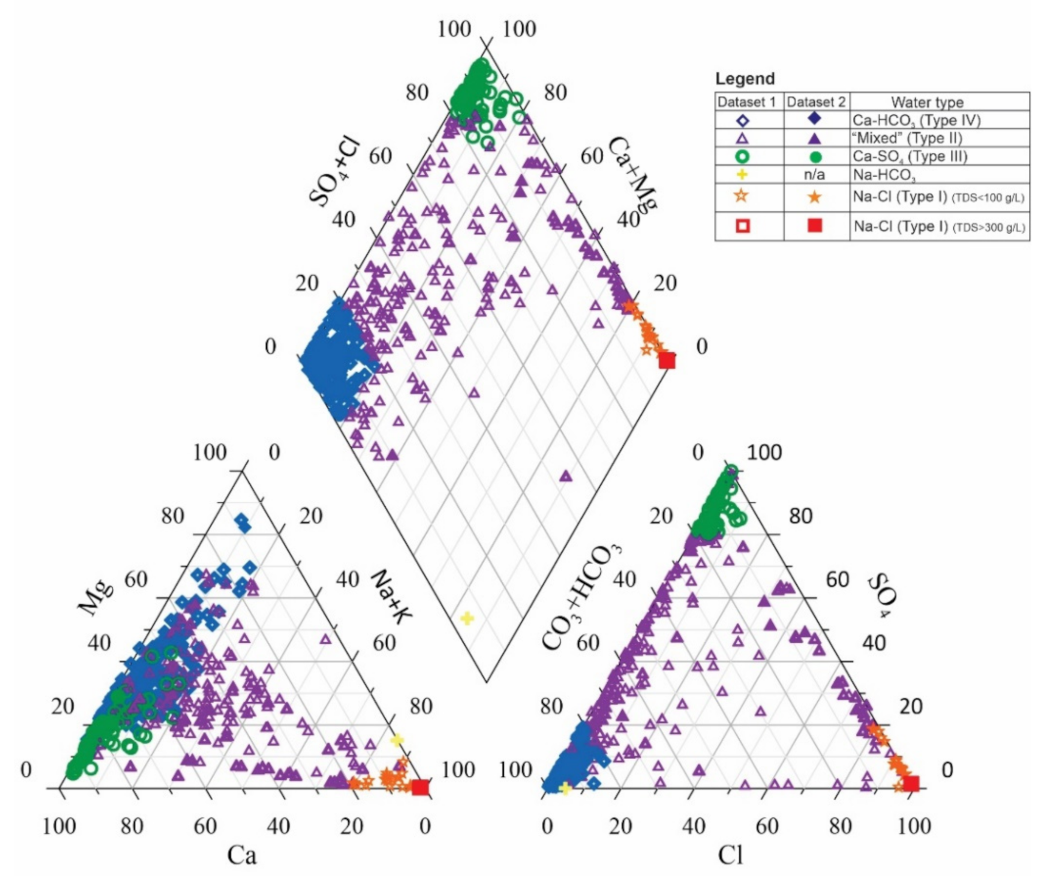
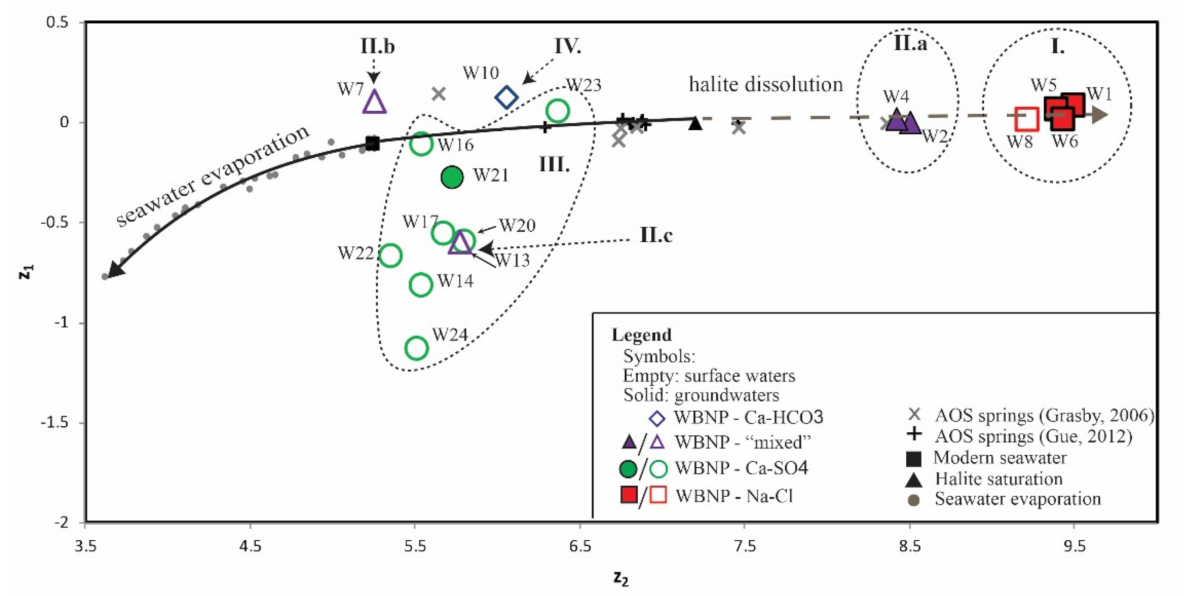
4.1. Total Dissolved Solids Content and Hydrochemical Water Types
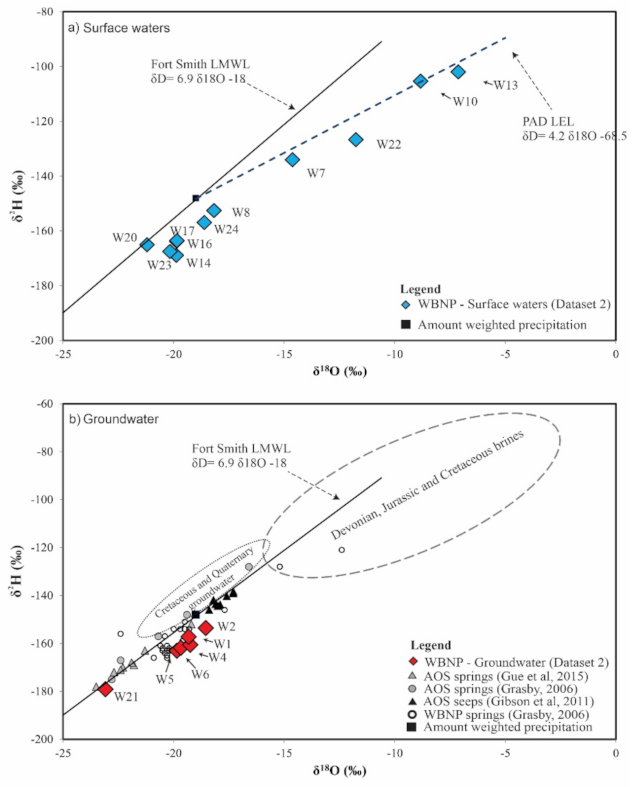
4.2. Isotopic Composition
4.3. Solute Ratios
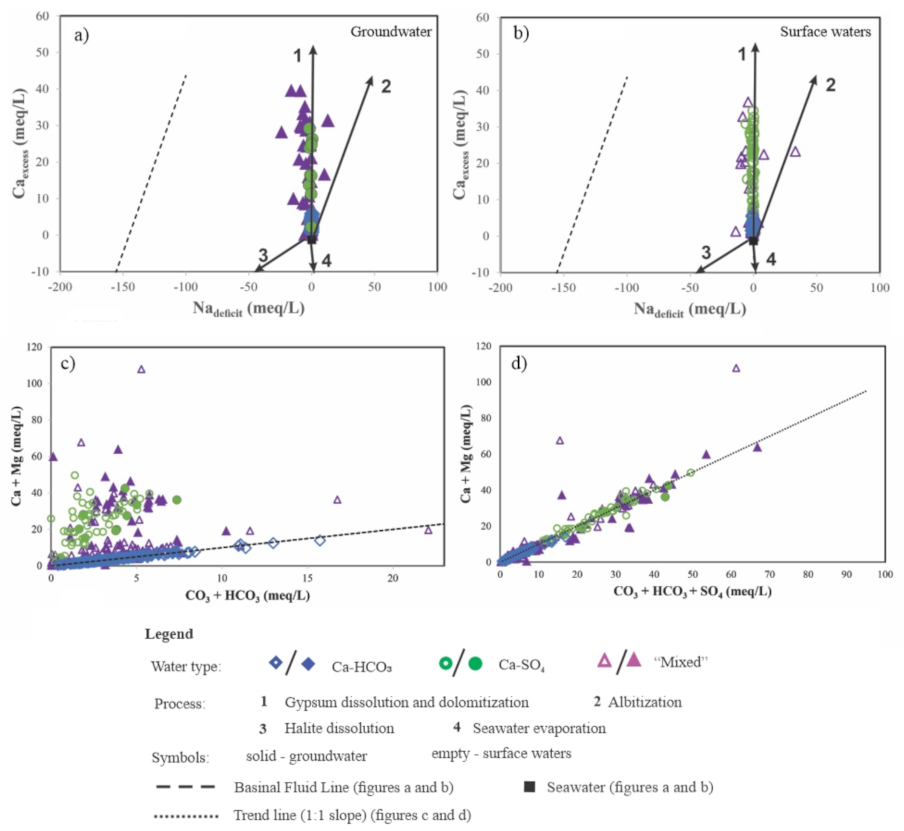
4.3.1. Sodium and Chloride in Type I and Type II Waters
4.3.2. Calcium in Type II and Type III Waters
4.3.3. Calcium and Bicarbonate in Type IV Waters
5. Discussion
5.1. Total Dissolved Solids Content and Hydrochemical Water Types
5.2. Isotopic Composition
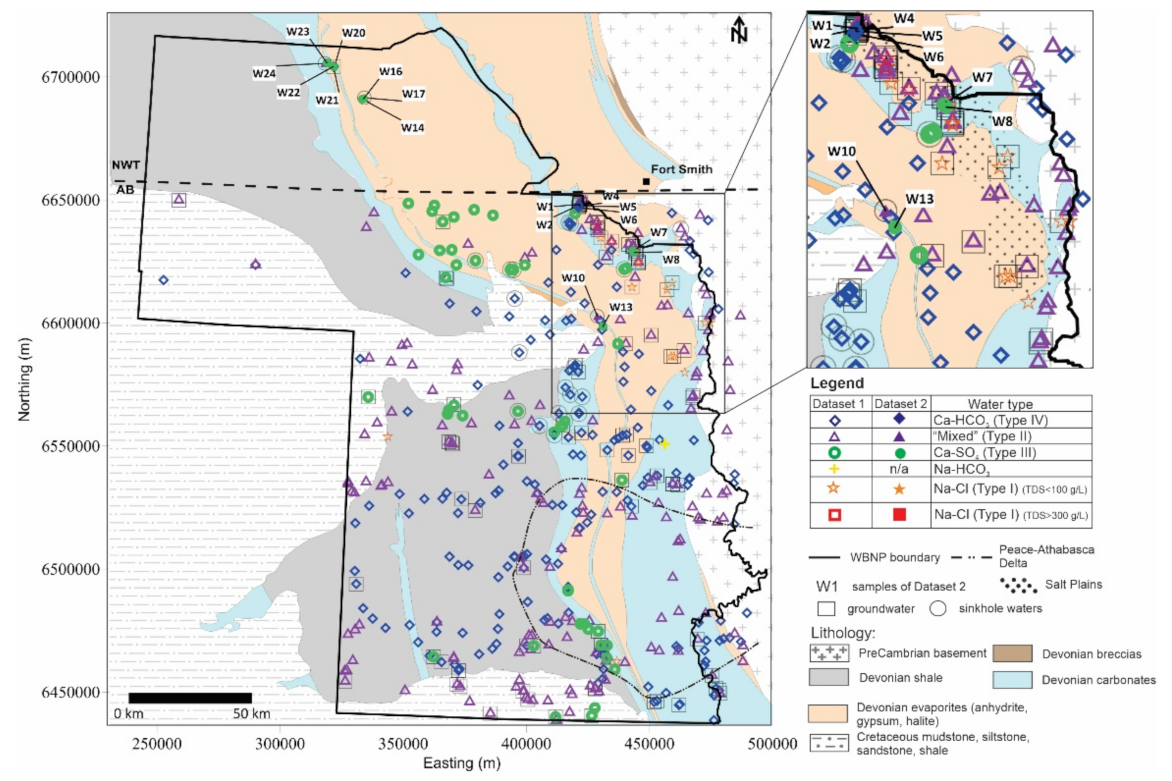
5.3. Source of Solutes
5.4. Updated Conceptual Models
6. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Heritage Committee (WHC), United Nations Educational, Scientific and Cultural Organization (UNESCO), 1983 Report SC/83/CONF.009/8. Available online: http://whc.unesco.org/archive/1983/sc-83-conf009-2e.pdf (accessed on 2 May 2017).
- Canadian Wildlife Service and U.S. Fish and Wildlife Service. International Recovery Plan for the Whooping Crane, Ottawa: Recovery of Nationally Endangered Wildlife (RENEW); U.S. Fish and Wildlife Service: Albuquerque, Mexico, 2007; 162p.
- Ramsar List—The List of Wetlands of International Importance. Available online: https://www.ramsar.org/sites/default/files/documents/library/sitelist.pdf (accessed on 28 March 2018).
- Lemay, T.G.; Guha, S. Compilation of Alberta Groundwater Information from Existing Maps and Data Sources; ERCB/AGS Open File Report 2009-02; Energy Resources Conservation Board: Edmonton, AB, Canada, 2009; 43p. [Google Scholar]
- Ozoray, G. Hydrogeology of the Seen River—Whites and River Area, Alberta; Earth Sciences Report 80-2; Alberta Research Council: Edmonton, AB, Canada, 1980. [Google Scholar]
- World Heritage Centre (WHC)—International Union for Conservation of Nature (IUCN). Reactive Monitoring Mission to Wood Buffalo National Park, Canada. Mission Report. Available online: http://whc.unesco.org/en/documents/156893 (accessed on 5 January 2018).
- Drake, J.J. The Geomorphic Implications of the Geo-Hydrology of Gypsum-Karst Areas. Master’s Thesis, McMaster University, Hamilton, ON, USA, 1970, unpublished. [Google Scholar]
- Stein, R. Significance of Observed Field Features in the Peace Point and Fitzgerald Map Areas of Wood Buffalo National Park; Unpublished Preliminary Report; Alberta Research Council: Edmonton, AB, Canada, 1979. [Google Scholar]
- Tsui, P.C. Deformation, Ground Subsidence, and Slope Movements along the Salt River Escarpment in Wood Buffalo National Park. Master’s Thesis, University of Alberta, Edmonton, AB, Canada, 1982. [Google Scholar]
- Weyer, K.U. Salt Dissolution, Karst Geology, Glacial Events and Groundwater Flow in the Pine Point Region, NWT; Research Report on Joint Project between Cominco Ltd. and NHRI (National Hydrology Research Institute); National Hydrology Research Institute: Calgary, AB, Canada, 1983; p. 461. [Google Scholar]
- Tsui, P.C.; Cruden, D.M. Deformation associated with gypsum karst in the Salt River Escarpment, northeastern Alberta. Can. J. Earth Sci. 1984, 21, 949–959. [Google Scholar] [CrossRef]
- Grasby, S.E.; Chen, Z. Subglacial recharge into the Western Canadian Sedimentary Basin. Impact of Pleistocene glaciation on basin hydrodynamics. Geol. Soc. Am. Bull. 2005, 117, 500–514. [Google Scholar] [CrossRef]
- Grasby, S.E. Brine springs of northern Alberta. In Potential for Carbonate-Hosted Lead-Zinc Mississippi Valley-Type Mineralization in Northern Alberta and Southern Northwest Territories: Geoscience Contributions, Targeted Geoscience Initiative; Hannigan, P.K., Ed.; Geological Survey of Canada Bulletin: Ottawa, ON, Canada, 2006; Volume 591, pp. 241–254. [Google Scholar] [CrossRef]
- Weyer, K.U.; Krouse, H.R.; Horwood, W.C. Investigation of regional geohydrology south of Great Slave Lake, Canada, utilizing natural sulphur and hydrogen isotope variations. In Proceedings of the International Symposium on Isotope Hydrology, Neuherberg, Germany, 19–23 June 1978; IAEA: Vienna, Austria, 1979; pp. 251–264. [Google Scholar]
- Nielsen, G. Groundwater investigations in Peace-Athabasca Delta; Alberta Department of Environment, Water Resources Division, Soils, Geology and Groundwater Branch: Edmonton, AB, Canada, 1972. [Google Scholar]
- Nobert, M.; Barrie, W. Groundwater Conditions in Wood Buffalo National Park, NWT. Interim Report; National Hydrology Research Institute, Environment Canada: Saskatoon, SK, USA, 1986. [Google Scholar]
- McNaughton, D. Hydrological Investigations in Wood Buffalo National Park 1985–1990, Final Report; Environment Canada: Edmonton, AB, Canada, 1991. [Google Scholar]
- Peters, D.L.; Prowse, T.D.; Marsh, P.; Lafleur, P.M.; Buttle, J.M. Persistence of water within perched basins of the Peace-Athabasca Delta, Northern Canada. Wetl. Econ. Manag. 2006, 14, 221–243. [Google Scholar] [CrossRef]
- Natural Regions Committee. Natural Regions and Subregions of Alberta; Downing, D.J., Pettapiece, W.W., Eds.; Pub. No. T/85; Government of Alberta: Edmonton, AB, Canada, 2006; 264p. [Google Scholar]
- Turchenek, L.W.; Pigot, M.E. Peatland Distribution in Alberta, Map 212; Alberta Research Council: Edmonton, AB, Canada, 1988. [Google Scholar]
- Heginbottom, J.A.; Dubreuil, M.A.; Harker, P.A. Canada—Permafrost, MCR 4177. In National Atlas of Canada, 5th ed.; National Resources Canada: Ottawa, ON, Canada, 1995. [Google Scholar]
- Meijer Drees, N.C. Devonian Elk Point Group of the Western Canada Sedimentary Basin. In Mossop GD and Shetsen I (comp.): Geological Atlas of the Western Canada Sedimentary Basin; Canadian Society of Petroleum Geologists and Alberta Research Council: Calgary, AB, Canada, 1994; pp. 129–149. [Google Scholar]
- Oldale, H.S.; Munday, R.J. Devonian Beaverhill Lake Group of the Western Canada Sedimentary Basin. In Mossop GD and Shetsen I (comp): Geological Atlas of the Western Canada Sedimentary Basin; Canadian Society of Petroleum Geologists and Alberta Research Council: Calgary, AB, Canada, 1994; pp. 149–165. [Google Scholar]
- Switzer, S.B.; Holland, W.G.; Christie, D.S.; Graf, G.C.; Hedinger, A.S.; McAuley, R.J.; Wierzbicki, R.A.; Packard, J.J. Devonian Woodbend-Winterburn Strata of the Western Canada Sedimentary Basin. In Geological Atlas of the Western Canada Sedimentary Basin; Mossop, G.D., Shetsen, I., Eds.; Canadian Society of Petroleum Geologists and Alberta Research Council: Calgary, AB, Canada, 1994; pp. 165–203. [Google Scholar]
- Norris, A.W. Paleozoic (Devonian) geology of northeastern Alberta and northwestern Saskatchewan. In Guide to the Athabasca Oil Sands Area, Information Series 65; Carrigy, M.A., Ed.; Alberta Research: Edmonton, AB, Canada, 1973; pp. 15–76. [Google Scholar]
- Richmond, W.O. Paleozoic Stratigraphy and Sedimentation of the Slave Point Formation, Southern Northwest Territories and Northern Alberta. Ph.D. Thesis, Stanford University, Stanford, CA, USA, 1965. [Google Scholar]
- Park, D.G.; Jones, B. Brecciation in the Devonian Keg River Formation of northern Wood Buffalo National Park, Northeast Alberta; Bulletin of Canadian Petroleum Geology: Calgary, AB, USA, 1987; pp. 416–429. [Google Scholar]
- Okulitch, A.V. Phanerozoic Bedrock Geology, Slave River, District of Mackenzie, Northwest Territories; Open File 5281 (National Earth Science Series, Geological Atlas, Map NP-11/12-G), Scale 1:1,000,000; Geological Survey of Canada: Ottawa, ON, Canada, 2006. [Google Scholar]
- Okulitch, A.V.; Fallas, K.M. GIS Dataset of Phanerozoic Geological Features, Slave River, Peace River, and Lake Athabasca Map Areas, Northwest Territories, Alberta, and Saskatchewan; Open File 5338, scale 1:1 000 000, 1 CD-ROM; Geological Survey of Canada: Ottawa, ON, Canada, 2007. [Google Scholar]
- Fenton, M.M.; Waters, E.J.; Pawley, S.M.; Atkinson, N.; Utting, D.J.; Mckay, K. Surficial geology of Alberta; AER/AGS Map 601, Scale 1:1 000 000; Alberta Energy Regulator: Calgary, AB, Canada, 2013. [Google Scholar]
- Prior, G.J.; Hathway, B.; Glombick, P.M.; Pana, D.I.; Banks, C.J.; Hay, D.C.; Schneider, C.L.; Grobe, M.; Elgr, E.; Weiss, J.A. Bedrock Geology of Alberta; ERCB/AGS Map 600, Scale 1:1 000 000; Energy Resources Conservation Board: Edmonton, AB, Canada, 2013. [Google Scholar]
- Bayrock, L. Surficial Geology, Fort Chipewyan, NTS 74L. Map 141; Scale 1:250,000; Research Council of Alberta: Edmonton, AB, Canada, 1972. [Google Scholar]
- Bayrock, L. Surficial Geology, Lake Claire, NTS 84I. Map 144; Scale 1:250,000; Research Council of Alberta: Edmonton, AB, Canada, 1972. [Google Scholar]
- Bayrock, L. Surficial Geology, Peace Point and Fitzgerald, NTS 84P-74M. Map 145; Scale 1:250,000; Research Council of Alberta: Edmonton, AB, Canada, 1972. [Google Scholar]
- Lemmen, D.S. Surficial Geology, Klewi River, District of Mackenzie; Northwest Territories; Map 1905A, Scale 1:250,000; Geological Survey of Canada: Ottawa, ON, Canada, 1998. [Google Scholar]
- Lemmen, D.S. Surficial Geology, Buffalo Lake, District of Mackenzie; Northwest Territories; Map 1906A, Scale 1:250,000; Geological Survey of Canada: Ottawa, ON, Canada, 1998. [Google Scholar]
- Mougeot, C.; Fenton, M.M. Surficial Geology of the Whitesand River Area, Alberta (NTS 84O); ERCB/AGS Map 541, Scale 1:250 000; Energy Resources Conservation Board: Edmonton, AB, Canada, 2010. [Google Scholar]
- Bachu, S.; Underschultz, J.R. Hydrogeology of formation waters, northeastern Alberta basin. Am. Assoc. Pet. Geol. Bull. 1993, 77, 1745–1768. [Google Scholar]
- Bachu, S. Flow of formation waters, aquifer characteristics, and their relation to hydrocarbon accumulations in the northern part of the Alberta basin. Am. Assoc. Pet. Geol. Bull. 1997, 81, 712–733. [Google Scholar] [CrossRef]
- Grobe, M. Distribution and Thickness of Salt within the Devonian Elk Point Group, Western Canada Sedimentary Basin. In Alberta Energy and Utilities Board, EUB/AGS Earth Sciences Report 2000–02; Alberta Geological Survey: Edmonton, AB, Canada, 2000. [Google Scholar]
- Pawlowicz, J.P.; Fenton, M.M. Drift Thickness of Alberta, Alberta Energies and Utilities Board; Map 227, Scale 1:2,000,000; Alberta Geological Survey: Edmonton, AB, Canada, 1995. [Google Scholar]
- Alberta Energy Regulator/Alberta Geological Survey (AER/AGS). Alberta Table of Formations; Alberta Energy Regulator. 2015. Available online: http://ags.aer.ca/document/Table-of-Formations.pdf (accessed on 18 July 2016).
- Core Laboratories Petroleum Services, Stratigraphic Correlation Chart; Core Laboratories Petroleum Services, Geological Sciences Department: Calgary, AB, Canada, 2017. Available online: https://www.spec2000.net/downloads/StratChart.pdf (accessed on 1 February 2020).
- Stewart, S.A. Field Measurements and Water Chemistry Analyses of Water Samples from the Wood Buffalo National Park area, Alberta, Canada: 1974 to 1978 (Tabular Data, Tab Delimited Format), Alberta Energy Regulator/Alberta Geological Survey, AER/AGS Digital Data 2021–0002. Available online: https://ags.aer.ca/publication/dig-2021-0002 (accessed on 1 March 2021).
- APHA. Standard Methods for the Examination of Water and Waste Water, 20th ed.; American Public Health Association: Washington, DC, USA, 1999. [Google Scholar]
- Engle, M.A.; Rowan, E.L. Interpretation of Na–Cl–Br systematics in sedimentary basin brines: Comparison of concentration, element ratio, and isometric log-ratio approaches. Math. Geosci. 2013, 45, 87–101. [Google Scholar] [CrossRef]
- Davisson, M.L.; Criss, R.E. Na-Ca-Cl relations in basinal fluids. Geochim. Et Cosmochim. Acta 1996, 60, 2743. [Google Scholar] [CrossRef]
- Qian, H.; Li, P.; Wu, J. Isotopic characteristics of precipitation, surface and ground waters in the Yinchuan Plain, Northwest China. Environ. Earth Sci. 2013, 70, 57–70. [Google Scholar] [CrossRef]
- Gue, A.E.; Mayer, B.; Grasby, S.E. Origin and geochemistry of saline spring waters in the Athabasca oil sands region, Alberta, Canada. Appl. Geochem. 2015, 61, 132–145. [Google Scholar] [CrossRef]
- IAEA/WMO International Atomic Energy Agency and World Meteorological Organization, Global Network of Isotopes in Precipitation. 2006. Available online: http://www.iaea.org/water (accessed on 8 August 2015).
- Birks, S.J.; Edwards, T.W.D.; Gibson, J.J.; Drimmie, R.J.; Michel, F.A. Canadian Network for Isotopes in Precipitation. 2004. Available online: http://www.science.uwaterloo.ca/twdedwar/cnip/cniphome.html (accessed on 1 January 2017).
- Davisson, M.L.; Presser, T.S.; Criss, R.E. Geochemistry of Tectonically Expelled Fluids from the Northern Coast Ranges, Rumsey Hills, California, USA. Geochim. Et Cosmochim. Acta 1994, 58, 1687. [Google Scholar] [CrossRef]
- Ford, D.; Williams, P. Karst Hydrogeology and Geomorphology; John Wiley & Sons Ltd.: West Sussex, UK, 2007. [Google Scholar]
- Wolfe, B.B.; Karst-Riddoch, T.L.; Hall, R.I.; Edwards, T.W.D.; English, M.C.; Palmini, R.; McGowan, S.; Leawitt, P.R.; Vardy, S.R. Classification of hydrological regimes of northern floodplain basins (Peace–Athabasca Delta, Canada) from analysis of stable isotopes (δ18O, δ2H) and water chemistry. Hydrol. Processes 2007, 21, 151–168. [Google Scholar] [CrossRef]
- Connolly, C.A.; Walter, L.M.; Baadsgard, H.; Longstaffe, F.J. Origin and evolution of formation waters, Alberta Basin, Western Canada Sedimentary Basin. II. Isotope systematics and water mixing. Appl. Geochem. 1990, 5, 397–413. [Google Scholar] [CrossRef] [Green Version]
- Lemay, T. Geochemical and Isotope Data for Formation Water from Selected Wells, Cretaceous to Quaternary Succession, Athabasca Oil Sands (In Situ) Area, Alberta. EUB/AGS Geo-Note 2002–02; Alberta Energy and Utilities Board/Alberta Geological Survey: Edmonton, AB, Canada, 2002; pp. 1–71. [Google Scholar]
- Gibson, J.J.; Birks, S.J.; Moncur, M.; Yi, Y.; Tattrie, K.; Jaschecko, S.; Richardson, K.; Eby, P. Isotopic and Geochemical Tracers for Fingerprinting Process-Affected Waters in the Oil Sands Industry: A Pilot Study, Oil Sands Research and Information Network; OSRIN Report No. TR-12. 2011; University of Alberta, School of Energy and the Environment: Edmonton, AB, Canada, 2011; 109p. [Google Scholar]
- Gue, A.E. The Geochemistry of Saline Springs in the Athabasca Oil Sands Region and Their Impact on the Clearwater and Athabasca Rivers. Master’s Thesis, University of Calgary, Calgary, AB, Canada, 2012, unpublished. [Google Scholar]
- McCaffrey, M.A.; Lazar, B.; Holland, H.D. The evaporation path of seawater and the coprecipitation of Br− and K+ with halite. J. Sediment. Petrol. 1987, 57, 928–937. [Google Scholar] [CrossRef] [PubMed]
- Sharp, Z. Principles of Stable Isotope Geochemistry; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2007. [Google Scholar]
- Michael, K.; Bachu, S. Origin, chemistry and flow of formation waters in the Mississippian–Jurassic sedimentary succession in the west-central part of the Alberta Basin, Canada. J. Mar. Pet. Geol. 2002, 19, 289–306. [Google Scholar] [CrossRef]


| Data Source | Description | Total # of Samples Before Culling | Simplified Sample Type | Type of Samples | # of Samples by Sample Type | # of Samples after Culling | Total # of Samples after Culling |
|---|---|---|---|---|---|---|---|
| Dataset 1 | Archive 1974–78 (Stewart, 2021) | 489 | Groundwater (GW) | springs | 98 | 95 | 465 |
| seeps | |||||||
| cave | |||||||
| auger | |||||||
| well | |||||||
| Surface water (SW) | stream, river, creek, ditch, channel | 365 | 345 | ||||
| lake, pond, pool, hole, wallow, slump | |||||||
| muskeg, oxbow, cutoff, slough | |||||||
| Both (GW-SW) | sinks | 20 | 19 | ||||
| not defined | 6 | 6 | |||||
| Dataset 2 | Collected 2013 | 17 | Groundwater (GW) | springs | 6 | 6 | 17 |
| Surface water (SW) | lake, pond | 8 | 8 | ||||
| Both (GW-SW) | sinks | 3 | 3 |
| Sample ID a | Sample Type | Northing (m) | Easting (m) | pH b | T (°C) b | Na+ (mg/L) | K+ (mg/L) | Mg2+ (mg/L) | Ca2+ (mg/L) | HCO3 (mg/L) | CO32− (mg/L) | SO42− (mg/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| W1 | spring | 421,734 | 6,648,210 | 5.6 | 8.9 | 138,000 | 0 | 210 | 1400 | 198 | 0 | 3879 |
| W2 | spring | 421,632 | 6,648,028 | 6.57 | 5.3 | 373 | 0 | 57 | 644 | 385 | 0 | 1521 |
| W4 | spring | 421,580 | 6,647,737 | 7.22 | 7.1 | 399 | 0 | 56 | 642 | 399 | 0 | 1170 |
| W5 | spring | 421,450 | 6,647,575 | 5.89 | 10.6 | 133,000 | 0 | 200 | 1600 | 209 | 0 | 3712 |
| W6 | spring | 421,173 | 6,646,928 | - | 4.7 | 25,300 | 0 | 80 | 890 | 250 | 0 | 2353 |
| W7 | pond | 443,246 | 6,629,257 | 7.36 | 18 | 588 | 0 | 34 | 428 | 156 | 0 | 1129 |
| W8 | lake | 443,955 | 6,628,644 | 7.7 | 20.1 | 2900 | 0 | 30 | 599 | 175 | 0 | 1383 |
| W10 | sinkhole | 428,794 | 6,602,760 | 8.45 | 17.3 | 14 | 9 | 85 | 13.8 | 416 | 48 | 16 |
| W13 | lake | 431,103 | 6,598,556 | 8.02 | 20.1 | 5 | 8 | 34 | 33.2 | 217 | 0 | 57 |
| W14 | lake | 664,740 | 6,690,842 | 6.9 | 9.9 | 4 | 2 | 75 | 572 | 289 | 0 | 1340 |
| W16 | lake | 665,271 | 6,691,113 | 7.1 | 13.7 | 5 | 2 | 64 | 631 | 318 | 0 | 1269 |
| W17 | sinkhole | 665,526 | 6,691,534 | 7.2 | 15.9 | 5 | 2 | 83 | 654 | 297 | 0 | 1336 |
| W20 | lake | 652,280 | 6,702,992 | 6.78 | 12.1 | 8 | 2 | 86 | 562 | 261 | 0 | 1377 |
| W21 | spring | 651,832 | 6,703,002 | 6.66 | 5.5 | 10 | 2 | 86 | 528 | 306 | 0 | 1367 |
| W22 | lake | 651,847 | 6,702,985 | 7.5 | 15.3 | 4 | 2 | 43 | 169 | 122 | 0 | 486 |
| W23 | lake | 649,044 | 6,704,257 | 6.41 | 6 | 36 | 3 | 106 | 620 | 351 | 0 | 1496 |
| W24 | sinkhole | 648,260 | 6,703,939 | 7.26 | 14.6 | 2 | 1 | 45 | 477 | 221 | 0 | 1160 |
| Sample ID a | Cl− (mg/L) | Br− (mg/L) | δ18 (‰) c | δ2 (‰) c | TDS (mg/L) | CBE (%) d | Br/Cl | z1 e | z2 e | Caexcess (meq/L) f | Nadeficit (meq/L) f | Water Type |
| W1 | 188,000 | 4.0 | −19.3 | −161 | 331,000 | 6.2 | 0.00002 | 0.09 | 9.5 | −141.3 | −1410.0 | Na-Cl |
| W2 | 571 | 0.0 | −18.5 | −154 | 3550 | −1.0 | 0.00007 | 0.00 | 8.5 | 31.5 | −2.2 | “mixed” |
| W4 | 596 | 0.0 | −19.3 | −157 | 3260 | 6.2 | 0.00008 | 0.02 | 8.4 | 31.4 | −2.8 | “mixed” |
| W5 | 186,000 | 4.5 | −19.9 | −163 | 324,000 | 5.0 | 0.00002 | 0.07 | 9.4 | −129.1 | −1239.9 | Na-Cl |
| W6 | 37,900 | 0.8 | −19.7 | −162 | 66,800 | 1.2 | 0.00002 | 0.02 | 9.4 | 1.7 | −172.0 | Na-Cl |
| W7 | 778 | 3.0 | −14.6 | −134 | 3110 | 1.8 | 0.004 | 0.11 | 5.3 | 20.5 | −6.5 | “mixed” |
| W8 | 4360 | 0.1 | −18.2 | −153 | 9450 | 1.2 | 0.00003 | 0.02 | 9.2 | 25.0 | −19.4 | Na-Cl |
| W10 | 18 | 0.0 | −8.8 | −105 | 619 | −4.0 | 0.001 | 0.13 | 6.1 | 0.7 | −0.2 | Ca-HCO3 |
| W13 | 17 | 0.0 | −7.1 | −102 | 370 | −3.3 | 0.001 | −0.60 | 5.8 | 1.6 | 0.2 | “mixed” |
| W14 | 19 | 0.0 | −19.9 | −169 | 2300 | 2.5 | 0.001 | −0.81 | 5.5 | 28.5 | 0.3 | Ca-SO4 |
| W16 | 9 | 0.0 | −19.8 | −164 | 2300 | 7.5 | 0.002 | −0.11 | 5.5 | 31.5 | 0.0 | Ca-SO4 |
| W17 | 16 | 0.0 | −19.9 | −164 | 2390 | 9.0 | 0.001 | −0.55 | 5.7 | 32.6 | 0.2 | Ca-SO4 |
| W20 | 27 | 0.0 | −21.2 | −165 | 2320 | 2.6 | 0.001 | −0.59 | 5.8 | 28.0 | 0.3 | Ca-SO4 |
| W21 | 23 | 0.0 | −23.1 | −179 | 2320 | −0.3 | 0.002 | −0.27 | 5.7 | 26.3 | 0.1 | Ca-SO4 |
| W22 | 15 | 0.0 | −11.8 | −127 | 840 | −1.7 | 0.002 | −0.66 | 5.4 | 8.4 | 0.2 | Ca-SO4 |
| W23 | 51 | 0.0 | −20.2 | −167 | 2660 | 3.7 | 0.001 | 0.06 | 6.4 | 30.9 | −0.3 | Ca-SO4 |
| W24 | 17 | 0.0 | −18.6 | −157 | 1920 | −1.2 | 0.001 | −1.13 | 5.5 | 23.8 | 0.3 | Ca-SO4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Déri-Takács, J.; Rostron, B.J.; Mendoza, C.; Mádl-Szőnyi, J. Hydrogeochemical Characteristics Refine the Conceptual Model of Groundwater Flow in Wood Buffalo National Park, Canada. Water 2022, 14, 965. https://doi.org/10.3390/w14060965
Déri-Takács J, Rostron BJ, Mendoza C, Mádl-Szőnyi J. Hydrogeochemical Characteristics Refine the Conceptual Model of Groundwater Flow in Wood Buffalo National Park, Canada. Water. 2022; 14(6):965. https://doi.org/10.3390/w14060965
Chicago/Turabian StyleDéri-Takács, Judit, Benjamin J. Rostron, Carl Mendoza, and Judit Mádl-Szőnyi. 2022. "Hydrogeochemical Characteristics Refine the Conceptual Model of Groundwater Flow in Wood Buffalo National Park, Canada" Water 14, no. 6: 965. https://doi.org/10.3390/w14060965
APA StyleDéri-Takács, J., Rostron, B. J., Mendoza, C., & Mádl-Szőnyi, J. (2022). Hydrogeochemical Characteristics Refine the Conceptual Model of Groundwater Flow in Wood Buffalo National Park, Canada. Water, 14(6), 965. https://doi.org/10.3390/w14060965