Cumulative Effects of Physical, Chemical, and Biological Measures on Algae Growth Inhibition
Abstract
:1. Introduction
2. Materials and Methods
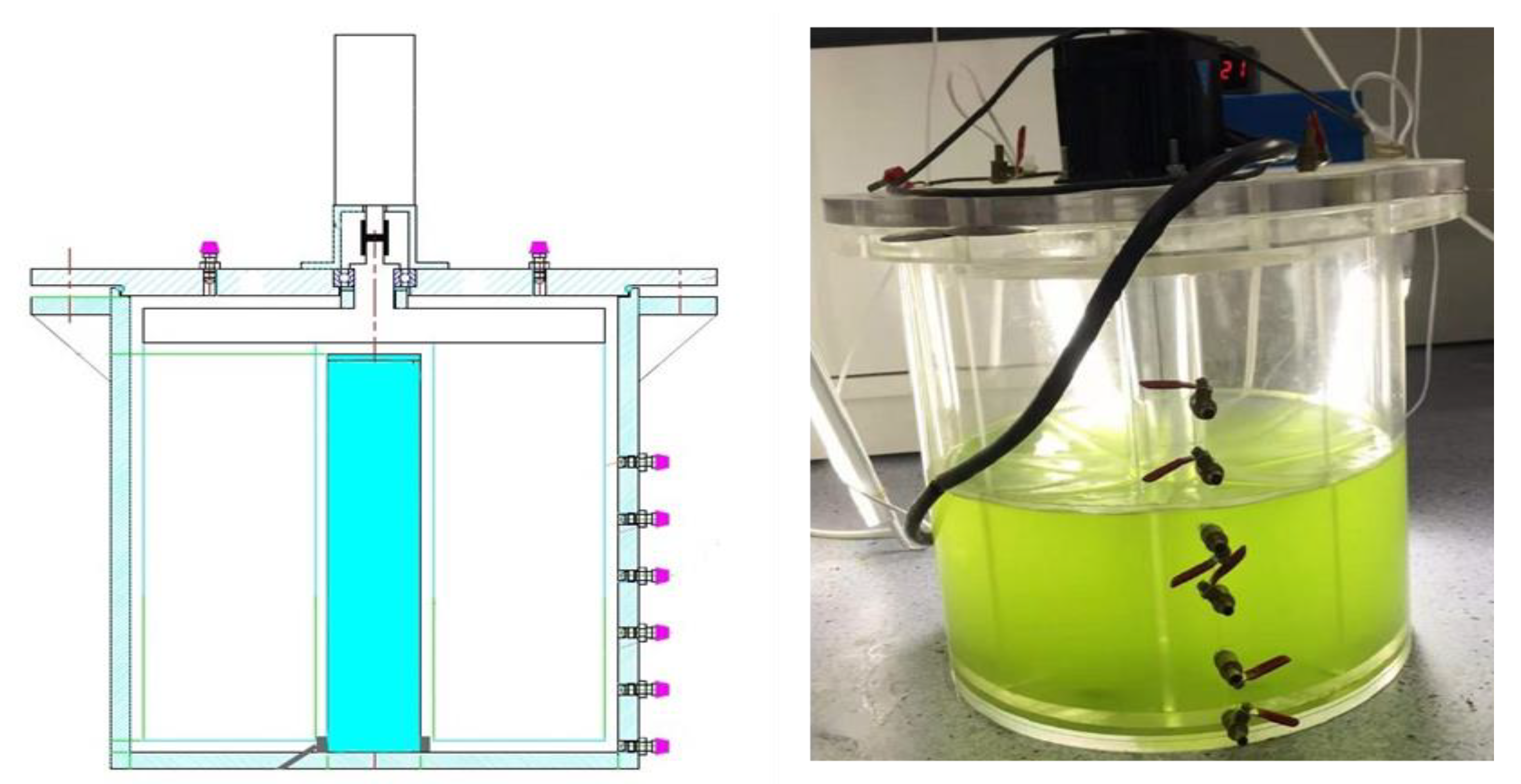
2.1. Experimental Method
2.1.1. Tested Algae and Culture Conditions
2.1.2. Allelochemical and Chemicals
2.1.3. Nutrient
2.1.4. Flow Velocity
2.1.5. Experiment and Sampling
2.2. Calculation of the Cumulative Effect (CE)
2.3. Simulation of the Cumulative Effect Rate
3. Results
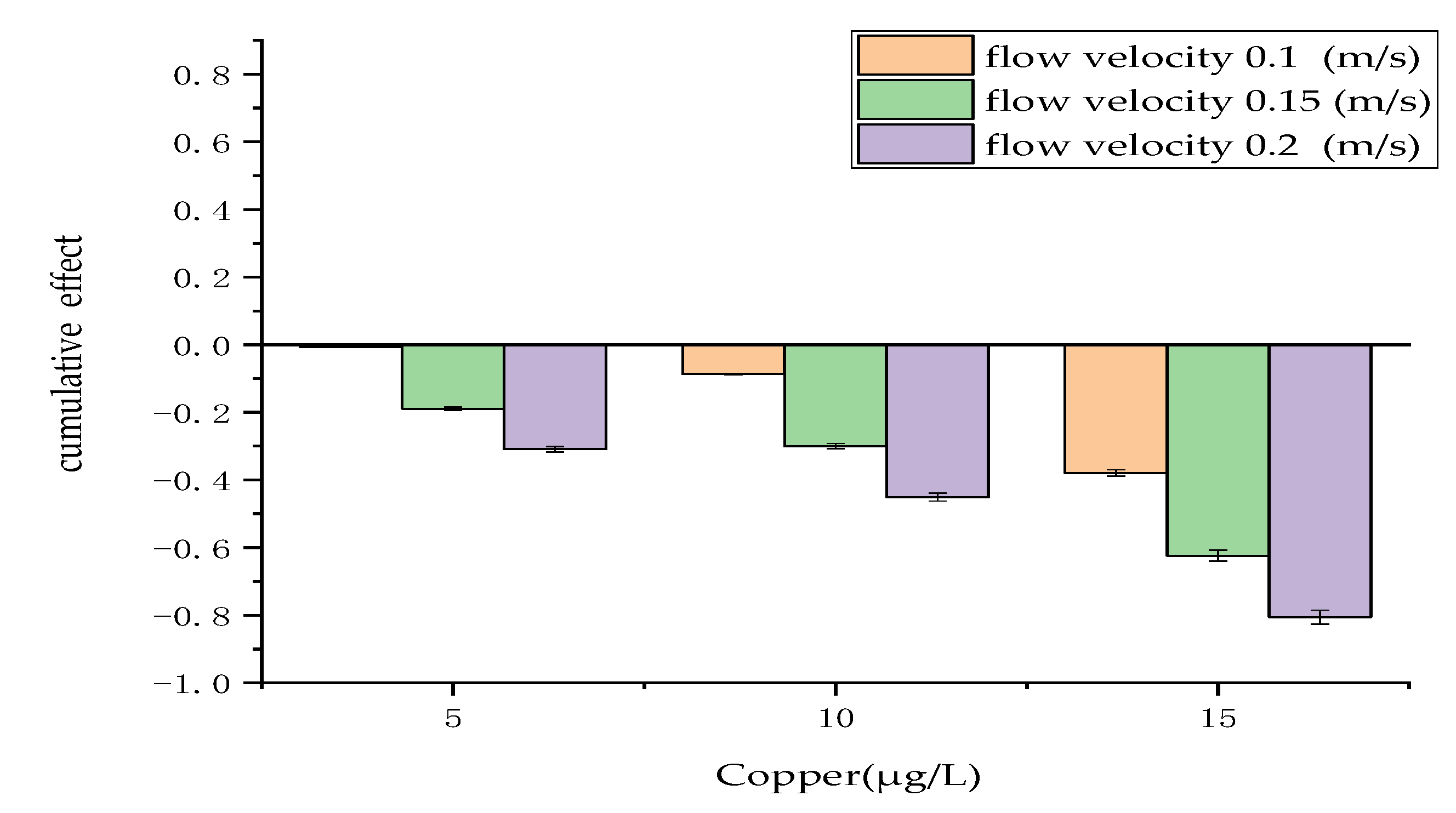
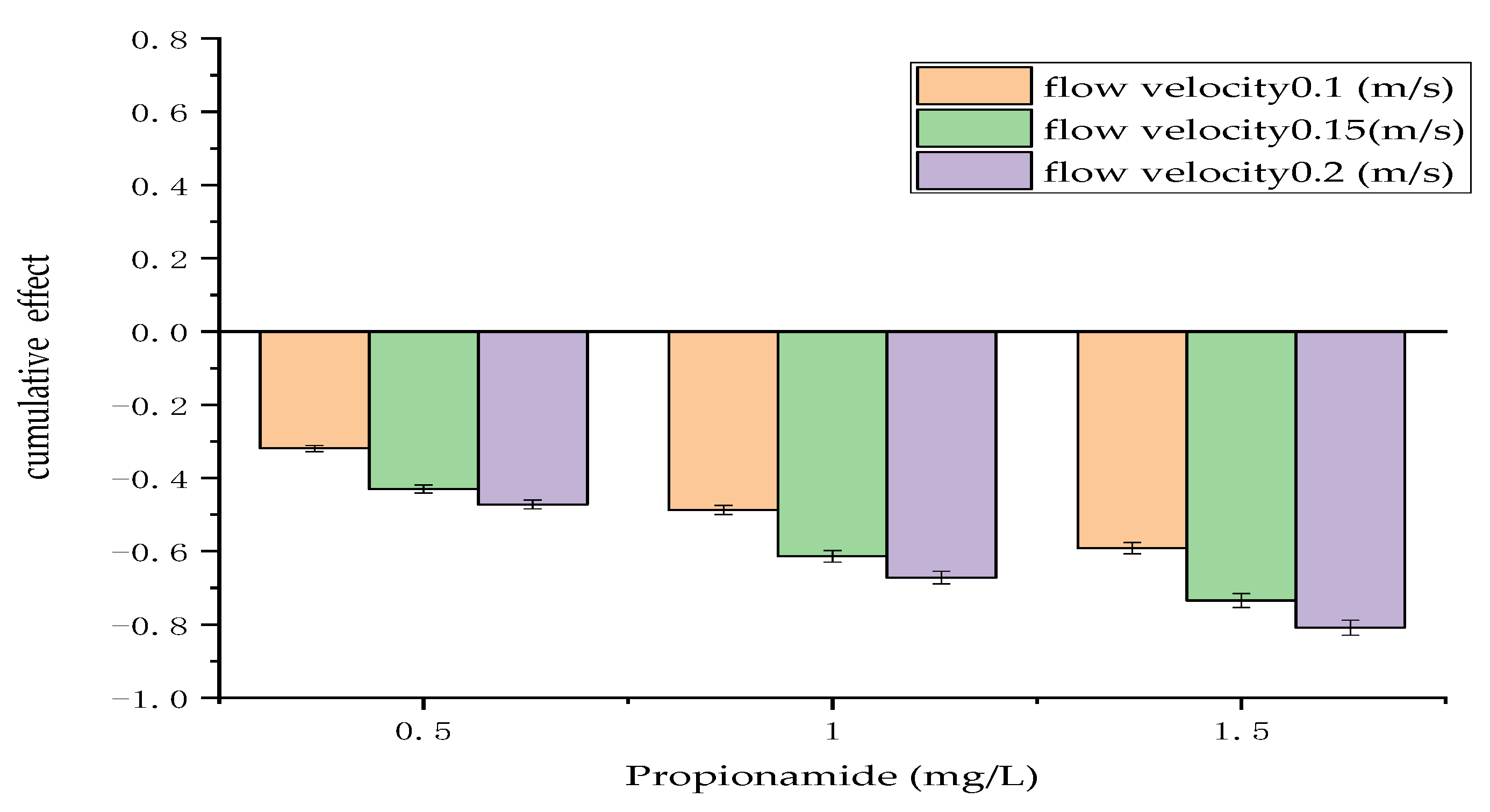
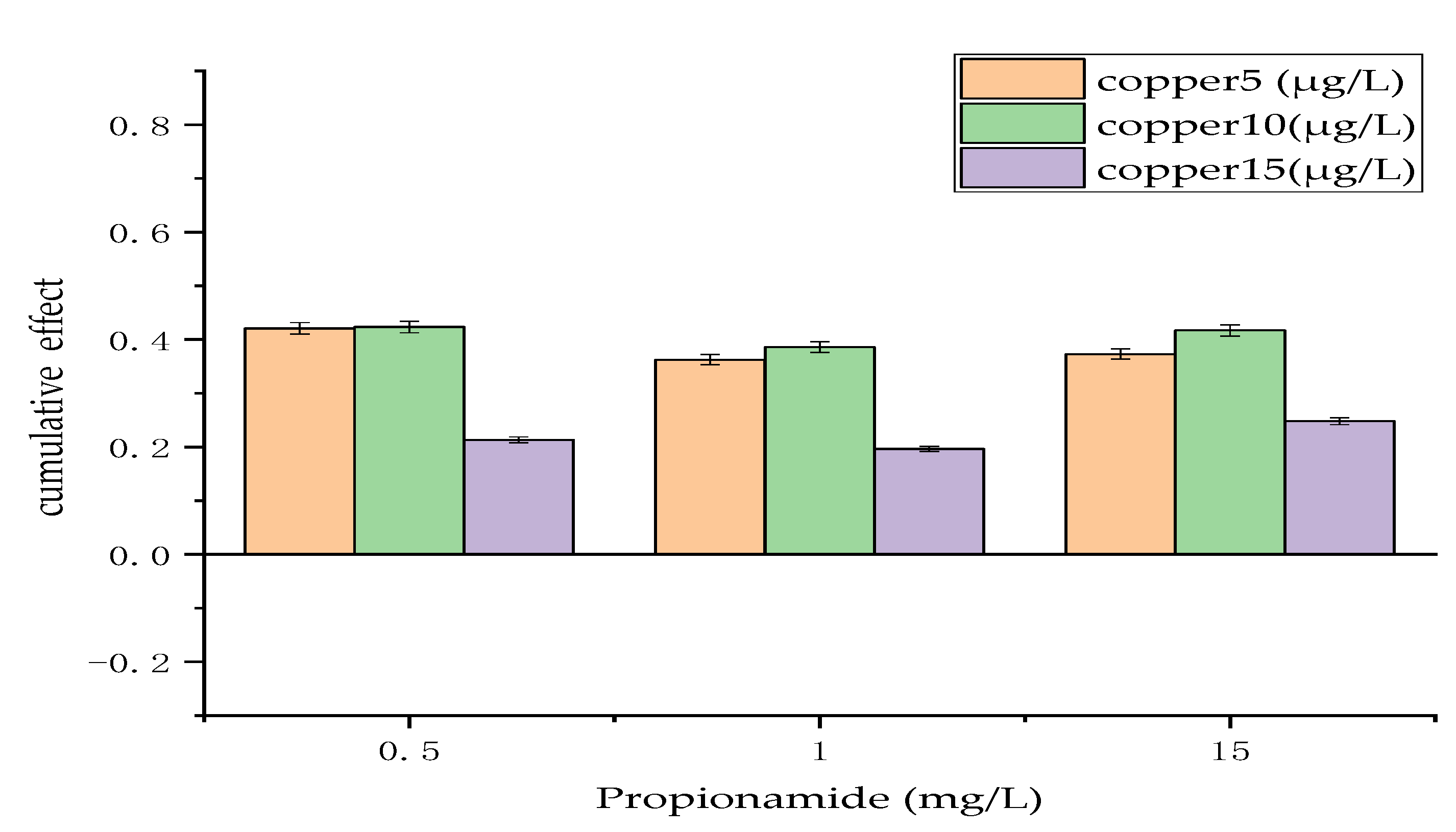
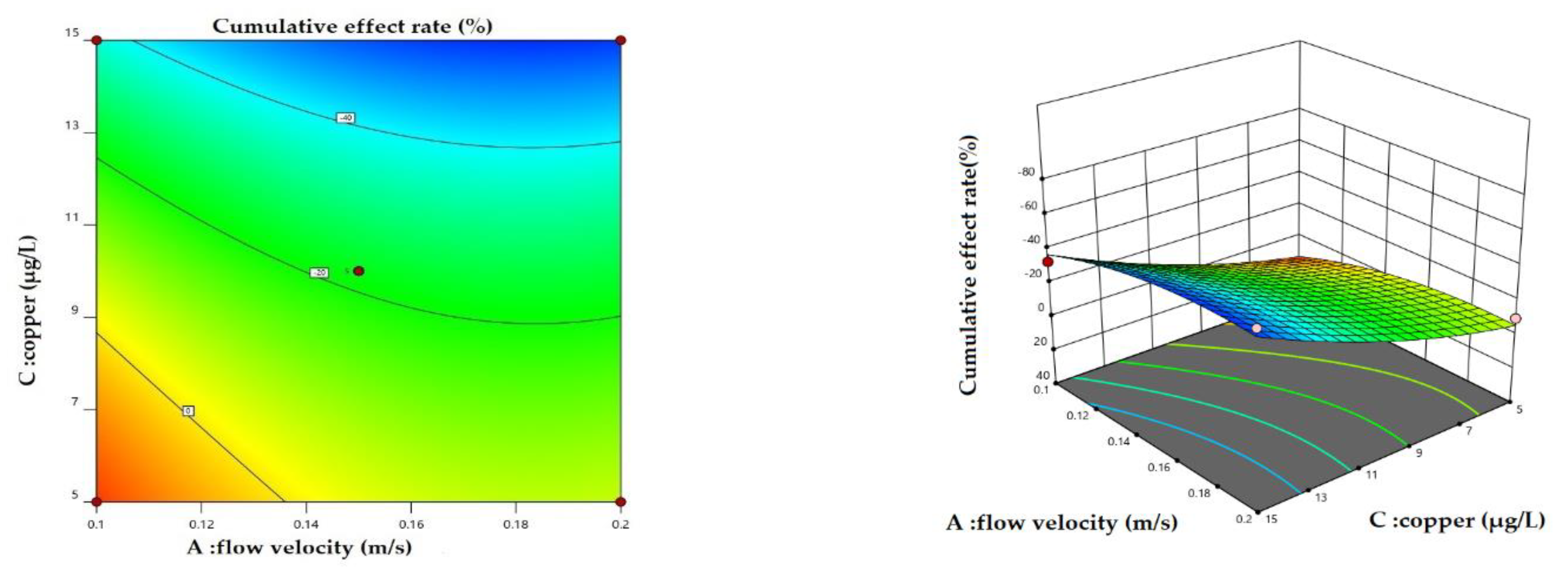
3.1. Cumulative Effect of Physical and Chemical Measures
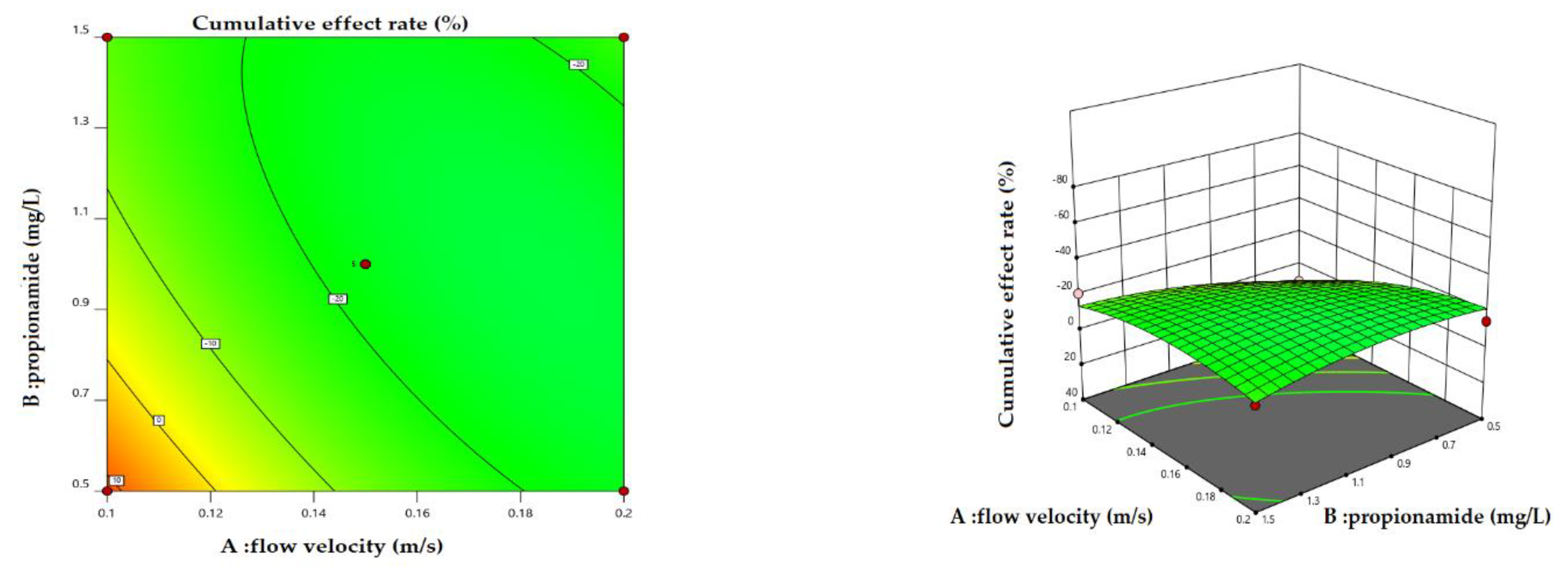
3.2. Cumulative Effect of Physical and Biological Measures
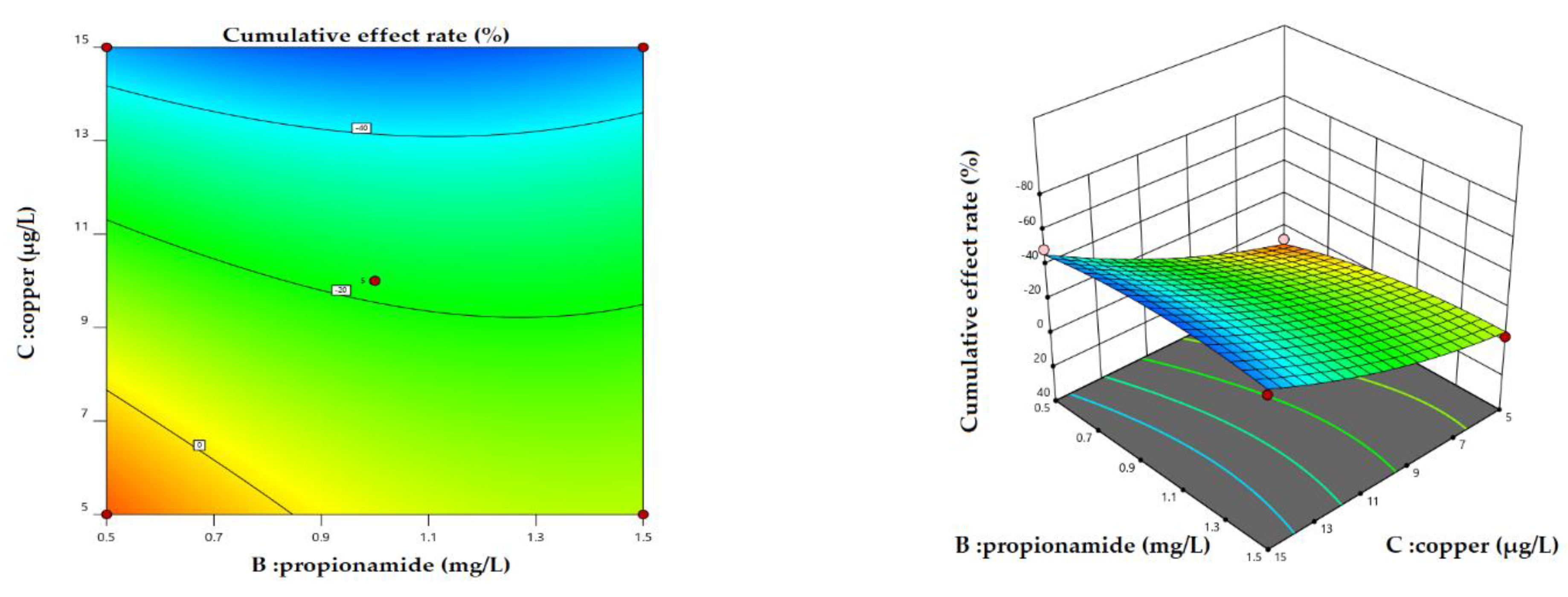
3.3. Cumulative Effect of Chemical and Biological Measures
3.4. Cumulative Effect of Physical, Chemical and Biological Measures
4. Discussion
4.1. Variation in Cumulative Effect Rate under Different Scenarios
4.2. Comparison with Previous Results or Theories
4.3. Limitations of This Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, H.; Zhang, Z.; Liang, D.; Du, H.; Pang, Y.; Hu, K.; Wang, J. Separation of wind’s influence on harmful cyanobacterial blooms. Water Res. 2016, 98, 280–292. [Google Scholar] [CrossRef] [PubMed]
- Van de Waal, D.B.; Verspagen, J.M.; Lurling, M.; Van Donk, E.; Visser, P.M.; Huisman, J. The ecological stoichiometry of toxins produced by harmful cyanobacteria: An experimental test of the carbon-nutrient balance hypothesis. Ecol. Lett. 2009, 12, 1326–1335. [Google Scholar] [CrossRef] [PubMed]
- Amorim, C.A.; Dantas, E.W.; Moura, A.D.N. Modeling cyanobacterial blooms in tropical reservoirs: The role of physicochemical variables and trophic interactions. Sci. Total Environ. 2020, 744, 140659. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.C.; Creed, I.F.; Jones, B.; Bergstrom, A.K. Global changes may be promoting a rise in select cyanobacteria in nutrient-poor northern lakes. Glob. Change Biol. 2020, 26, 4966–4987. [Google Scholar] [CrossRef]
- Jones, M.R.; Pinto, E.; Torres, M.A.; Dörr, F.; Mazur-Marzec, H.; Szubert, K.; Tartaglione, L.; Dell’Aversano, C.; Miles, C.O.; Beach, D.G. CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria. Water Res. 2021, 196, 117017. [Google Scholar] [CrossRef]
- Ji, X.; Verspagen, J.M.H.; Stomp, M.; Huisman, J. Competition between cyanobacteria and green algae at low versus elevated CO2: Who will win, and why? J. Exp. Bot. 2017, 68, 3815–3828. [Google Scholar] [CrossRef] [Green Version]
- Gu, P.; Sun, Y.; Xue, L.; Zhu, L.; Shan, J.; Li, X.; Ni, Z.; Zhang, W.; Zheng, Z. Predicting cyanobacterial decomposition response to multiple environmental factors through Central Composite Design method. Environ. Technol. Inno. 2021, 22, 101515. [Google Scholar] [CrossRef]
- Pearson, L.; Mihali, T.; Moffitt, M.; Kellmann, R.; Neilan, B. On the chemistry, toxicology and genetics of the cyanobacterial toxins, microcystin, nodularin, saxitoxin and cylindrospermopsin. Mar. Drugs 2010, 8, 1650–1680. [Google Scholar] [CrossRef] [Green Version]
- Kang, L.; He, Y.; Dai, L.; He, Q.; Ai, H.; Yang, G.; Liu, M.; Jiang, W.; Li, H. Interactions between suspended particulate matter and algal cells contributed to the reconstruction of phytoplankton communities in turbulent waters. Water Res. 2019, 149, 251–262. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, L.; You, Q.; Pan, Y.; Liu, T.; Zhou, Y.; Zhang, J.; Pang, W.; Wang, Q. Influence of cyanobacterial blooms and environmental variation on zooplankton and eukaryotic phytoplankton in a large, shallow, eutrophic lake in China. Sci. Total Environ. 2021, 773, 145421. [Google Scholar] [CrossRef]
- Park, J.; Son, Y.; Lee, W.H. Variation of efficiencies and limits of ultrasonication for practical algal bloom control in fields. Ultrason. Sonochem. 2019, 55, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Jančula, D.; Maršálek, B. Critical review of actually available chemical compounds for prevention and management of cyanobacterial blooms. Chemosphere 2011, 85, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, E. A coupled sampling design for parameter estimation in microalgae growth experiment: Maximizing the benefits of uniform and non-uniform sampling. Water 2021, 13, 2996. [Google Scholar] [CrossRef]
- Park, K.Y.; Kweon, J.; Chantrasakdakul, P.; Lee, K.; Cha, H.Y. Anaerobic digestion of microalgal biomass with ultrasonic disintegration. Int. Biodeter. Biodegr. 2013, 85, 598–602. [Google Scholar] [CrossRef]
- Li, F.; Zhang, H.; Zhu, Y.; Xiao, Y.; Chen, L. Effect of flow velocity on phytoplankton biomass and composition in a freshwater lake. Sci. Total Environ. 2013, 447, 64–71. [Google Scholar] [CrossRef]
- Batista, M.D.; Anhê, A.C.B.M.; de Souza Inácio Gonçalves, J.C. Use of hydrodynamic cavitation for Algae Removal: Effect on the inactivation of microalgae belonging to genus Scenedesmus. Water Air Soil Pollut. 2017, 228, 443. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Hardwick, L.; Dorani, F. Use of flow management to mitigate cyanobacterial blooms in the Lower Darling River, Australia. J. Plankton Res. 2010, 33, 229–241. [Google Scholar] [CrossRef]
- Wang, Z.; Li, D.; Qin, H.; Li, Y. An integrated method for removal of harmful cyanobacterial blooms in eutrophic lakes. Environ. Pollut. 2012, 160, 34–41. [Google Scholar] [CrossRef]
- Zuo, S.; Zhou, S.; Ye, L.; Ding, Y.; Jiang, X. Antialgal effects of five individual allelochemicals and their mixtures in low level pollution conditions. Environ. Sci. Pollut. R 2016, 23, 15703–15711. [Google Scholar] [CrossRef]
- Greenfield, D.I.; Duquette, A.; Goodson, A.; Keppler, C.J.; Williams, S.H.; Brock, L.M.; Stackley, K.D.; White, D.; Wilde, S.B. The effects of three chemical algaecides on cell numbers and toxin content of the cyanobacteria Microcystis aeruginosa and Anabaenopsis sp. J. Environ. Manag. 2014, 54, 1110–1120. [Google Scholar] [CrossRef]
- Jin, X.; Bi, L.; Lyu, T.; Chen, J.; Zhang, H.; Pan, G. Amphoteric starch-based bicomponent modified soil for mitigation of harmful algal blooms (HABs) with broad salinity tolerance: Flocculation, algal regrowth, and ecological safety. Water Res. 2019, 165, 115005. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; Huang, Z.; Ji, R.; Qiu, Y.; Wang, Z.; Liu, J. A review of allelopathy on microalgae. Microbiology 2019, 165, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; He, Y.; Li, H.; Wei, Y.; Zhao, L.; Yang, G.; Chen, X. Using flocculation and subsequent biomanipulation to control microcystis blooms: A laboratory study. Harmful Algae 2020, 99, 101917. [Google Scholar] [CrossRef] [PubMed]
- Kibuye, F.A.; Zamyadi, A.; Wert, E.C. A critical review on operation and performance of source water control strategies for cyanobacterial blooms: Part II-mechanical and biological control methods. Harmful Algae 2021, 109, 102119. [Google Scholar] [CrossRef]
- Bacellar Mendes, L.B.; Vermelho, A.B. Allelopathy as a potential strategy to improve microalgae cultivation. Biotechnol. Biofuels 2013, 6, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.; Dao, G.; Tao, Y.; Zhan, X.; Hu, H. A review on control of harmful algal blooms by plant-derived allelochemicals. J. Hazard Mater. 2021, 401, 123403. [Google Scholar] [CrossRef]
- Li, B.; Yin, Y.; Kang, L.; Feng, L.; Liu, Y.; Du, Z.; Tian, Y.; Zhang, L. A review: Application of allelochemicals in water ecological restoration—algal inhibition. Chemosphere 2021, 267, 128869. [Google Scholar] [CrossRef]
- Wu, X.; Wu, H.; Ye, J.; Zhong, B. Study on the release routes of allelochemicals from Pistia stratiotes Linn., and its anti-cyanobacteria mechanisms on Microcystis aeruginosa. Environ. Sci. Pollut. R 2015, 22, 18994–19001. [Google Scholar] [CrossRef]
- Jiang, Z.; Xinyuan, Z. Treatment and utilization of wastewater in the Beijing Zoo by an aquatic macrophyte system. J. Ecol. Eng. 1998, 11, 101–110. [Google Scholar] [CrossRef]
- Liu, G.T.; Zhou, C.F.; Sun, L.F.; Zhu, W.W.; Jiang, H.; Wang, H.X.; An, S.Q. Effects of Eichhornia crassipes allelochemicals on the growth of two mono- and co-cultured algae Microcystis aeruginosa and Scenedesmus obliquus. Acta Sci. Circumst. 2011, 31, 2303–2311. (In Chinese) [Google Scholar]
- Wu, X.; Wu, H.; Wang, S.; Wang, Y.; Zhang, R.; Hu, X.; Ye, J. Effect of propionamide on the growth of Microcystis flos-aquae colonies and the underlying physiological mechanisms. Sci. Total Environ. 2018, 630, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Rui, J.L.; Li, H.L.; Zhao, D.H.; Fu, J.J.; An, S.Q. Research on Algal bloom control for environment Engineering. Appl. Mech. Mater. 2013, 340, 971–974. [Google Scholar] [CrossRef]
- Furlan, E.; Torresan, S.; Critto, A.; Lovato, T.; Solidoro, C.; Lazzari, P.; Marcomini, A. Cumulative Impact Index for the Adriatic Sea: Accounting for interactions among climate and anthropogenic pressures. Sci. Total Environ. 2019, 670, 379–397. [Google Scholar] [CrossRef] [PubMed]
- Crain, C.M.; Halpern, B.S.; Beck, M.W.; Kappel, C.V. Understanding and managing human threats to the coastal marine environment. Ann. N. Y. Acad. Sci. 2009, 1162, 39–62. [Google Scholar] [CrossRef] [PubMed]
- Sabater, S.; Navarro, E.; Guasch, H. Effects of copper on algal communities at different current velocities. J. Appl. Phycol 2002, 14, 391–398. [Google Scholar] [CrossRef]
- Wang, X.; Huang, K.; Gao, J.; Szeto, Y.T.; Jiang, C.; Zhu, J.; Zhang, J.; Liu, J. Effects on photosynthetic and antioxidant systems of harmful cyanobacteria by nanocrystalline Zn-MOF-FA. Sci. Total Environ. 2021, 792, 148247. [Google Scholar] [CrossRef]
- Sha, J.; Xiong, H.; Li, C.; Lu, Z.; Zhang, J.; Zhong, H.; Zhang, W.; Yan, B. Harmful algal blooms and their eco-environmental indication. Chemosphere 2021, 274, 129912. [Google Scholar] [CrossRef]
- Crain, C.M.; Kroeker, K.; Halpern, B.S. Interactive and cumulative effects of multiple human stressors in marine systems. Ecol. Lett. 2008, 11, 1304–1315. [Google Scholar] [CrossRef]
- Ban, S.S.; Graham, N.A.; Connolly, S.R. Evidence for multiple stressor interactions and effects on coral reefs. Glob. Change Biol. 2014, 20, 681–697. [Google Scholar] [CrossRef]
- Orr, J.A.; Luijckx, P.; Arnoldi, J.F.; Jackson, A.L.; Piggott, J.J. Rapid evolution generates synergism between multiple stressors: Linking theory and an evolution experiment. Glob. Change Biol. 2022, 28, 1740–1752. [Google Scholar] [CrossRef]
- Boyd, P.W.; Collins, S.; Dupont, S.; Fabricius, K.; Gattuso, J.P.; Havenhand, J.; Hutchins, D.A.; Riebesell, U.; Rintoul, M.S.; Vichi, M. Experimental strategies to assess the biological ramifications of multiple drivers of global ocean change—A review. Glob. Change Biol. 2018, 24, 2239–2261. [Google Scholar] [CrossRef] [PubMed]
- Strain, E.M.; Thomson, R.J.; Micheli, F.; Mancuso, F.P.; Airoldi, L. Identifying the interacting roles of stressors in driving the global loss of canopy-forming to mat-forming algae in marine ecosystems. Glob. Change Biol. 2014, 20, 3300–3312. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.-P. Effects of two copper compounds on Microcystis aeruginosa cell density, membrane integrity, and microcystin release. Ecotoxicol. Environ. Safe 2015, 120, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Anjum, M.F.; Tasadduq, I.; Al-Sultan, K. Response surface methodology: A neural network approach. Eur. J. Oper. Res. 1997, 101, 65–73. [Google Scholar] [CrossRef]
- Hu, Y.; Meng, F.L.; Hu, Y.Y.; Habibul, N.; Sheng, G.P. Concentration- and nutrient-dependent cellular responses of microalgae Chlorella pyrenoidosa to perfluorooctanoic acid. Water Res. 2020, 185, 116248. [Google Scholar] [CrossRef]
- Long, T.Y.; Wu, L.; Meng, G.H.; Guo, W.H. Numerical simulation for impacts of hydrodynamic conditions on algae growth in Chongqing Section of Jialing River, China. Ecol. Model 2011, 222, 112–119. [Google Scholar] [CrossRef]
- Levy, J.L.; Angel, B.M.; Stauber, J.L.; Poon, W.L.; Simpson, S.L.; Cheng, S.H.; Jolley, D.F. Uptake and internalisation of copper by three marine microalgae: Comparison of copper-sensitive and copper-tolerant species. Aquat. Toxicol. 2008, 89, 82–93. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Son, J.; Mo, H.H.; Lee, Y.S.; Cho, K. Modeling the influence of initial density and copper exposure on the interspecific competition of two algal species. Ecol. Model 2018, 383, 160–170. [Google Scholar] [CrossRef]
- Aslan, N.; Cebeci, Y. Application of Box–Behnken design and response surface methodology for modeling of some Turkish coals. Fuel 2007, 86, 90–97. [Google Scholar] [CrossRef]
- Liu, J. Optimisation of biomass and lipid production by adjusting the interspecific competition mode of Dunaliella salina and Nannochloropsis gaditana in mixed culture. J. Appl. Phycol. 2014, 26, 163–171. [Google Scholar] [CrossRef]
- Rezaee, R.; Maleki, A.; Jafari, A.; Mazloomi, S.; Zandsalimi, Y.; Mahvi, A.H. Application of response surface methodology for optimization of natural organic matter degradation by UV/H2O2 advanced oxidation process. J. Environ. Health Sci. 2014, 12, 67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, M.; Gupta, S. Modeling of trihalomethanes (THMs) in drinking water supplies: A case study of eastern part of India. Environ. Sci. Pollut. R 2015, 22, 12615–12623. [Google Scholar] [CrossRef] [PubMed]
- Danbaba, N.; Nkama, I.; Badau, M.H. Application of response surface methodology (RSM) and central composite design (CCD) to optimize minerals composition of rice-cowpea composite blends during extrusion cooking. Int. J. Food Sci. Tech. 2015, 5, 40–52. [Google Scholar]
- Hill, W.R.; Bednarek, A.T.; Larsen, I.L. Cadmium sorption and toxicity in autotrophic biofilms. Can. J. Fish Aquat. Sci. 2000, 57, 530–537. [Google Scholar] [CrossRef]
- Wang, C.; Lan, C.Q. Effects of shear stress on microalgae—A review. Biotechnol. Adv. 2018, 36, 986–1002. [Google Scholar] [CrossRef]
- Nagajyoti, P.C.; Lee, K.D.; Sreekanth, T. Heavy metals, occurrence and toxicity for plants: A review. Environ. Chem. Lett. 2010, 8, 199–216. [Google Scholar] [CrossRef]
- Xue, B.; Wang, G.; Xiao, J.; Helman, D.; Sun, W.; Wang, J.; Liu, T. Global convergence but regional disparity in the hydrological resilience of ecosystems and watersheds to drought. J. Hydrol. 2020, 591, 125589. [Google Scholar] [CrossRef]
- Xue, B.; Helman, D.; Wang, G.; Xu, C.Y.; Xiao, J.; Liu, T.; Wang, L.; Li, X.; Duan, L.; Lei, H. The low hydrologic resilience of Asian Water Tower basins to adverse climatic changes. Adv. Water Resour. 2021, 155, 103996. [Google Scholar] [CrossRef]
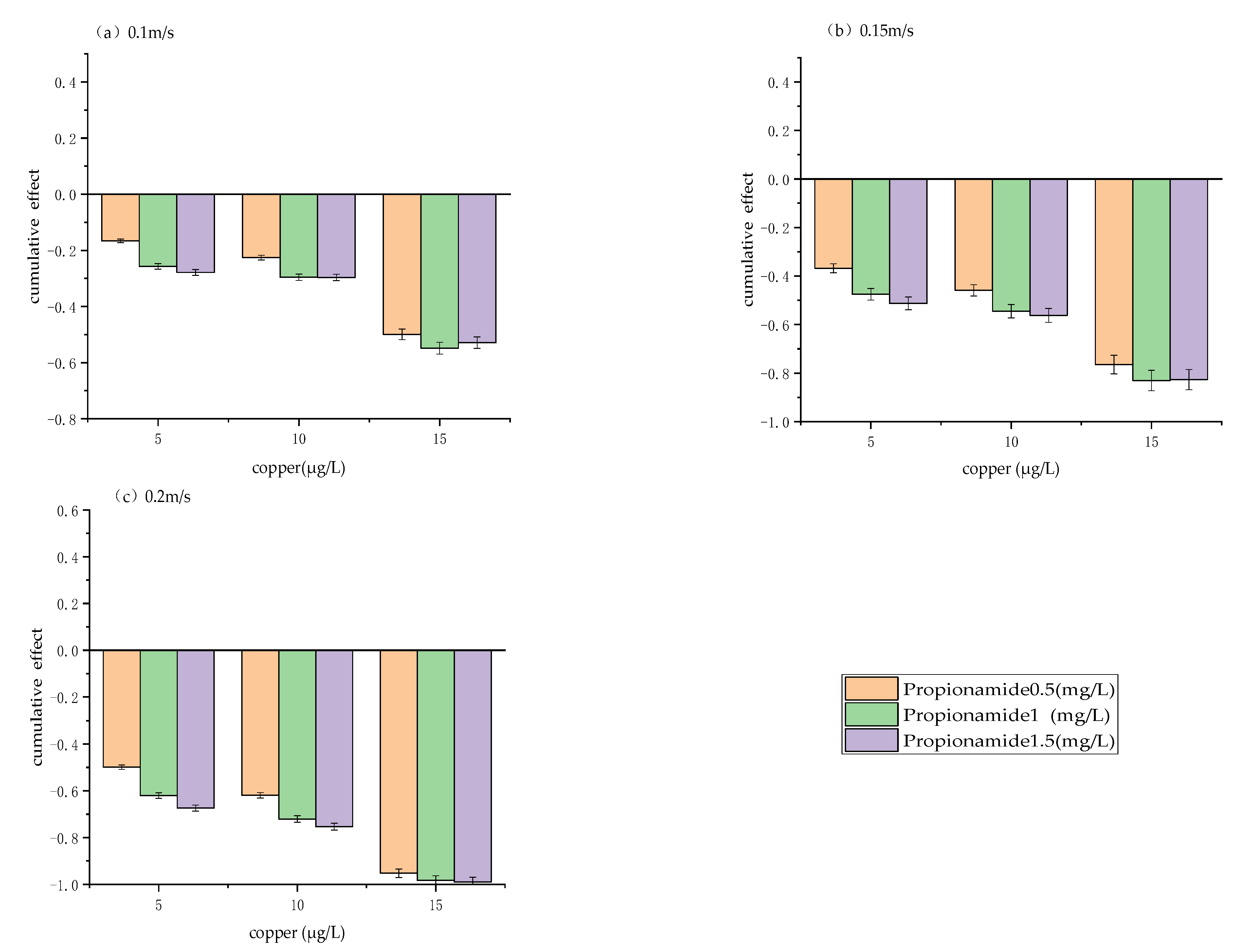
| Variable | Level | ||
|---|---|---|---|
| Low | Medium | High | |
| Flow velocity (m/s) | 0.1 | 0.15 | 0.2 |
| Propionamide (mg/L) | 0.5 | 1 | 1.5 |
| Copper (μg/L) | 5 | 10 | 15 |
| Source | Sum of Squares | Df | Mean Square | F-Value | p-Value Prob > F |
|---|---|---|---|---|---|
| Model | 0.2153 | 9 | 0.0239 | 90.12 | <0.0001 (significant) |
| A-Velocity | 0.1100 | 1 | 0.1100 | 414.28 | <0.0001 |
| B-Propionamide | 0.0164 | 1 | 0.0164 | 61.85 | 0.0001 |
| C-copper | 0.0680 | 1 | 0.0680 | 256.17 | <0.0001 |
| AB | 0.0015 | 1 | 0.0015 | 5.62 | 0.0495 |
| AC | 0.0000 | 1 | 0.0000 | 0.0470 | 0.8345 |
| BC | 0.0018 | 1 | 0.0018 | 6.78 | 0.0352 |
| A² | 0.0071 | 1 | 0.0071 | 26.82 | 0.0013 |
| B² | 0.0030 | 1 | 0.0030 | 11.25 | 0.0122 |
| C² | 0.0083 | 1 | 0.0083 | 31.23 | 0.0008 |
| Residual | 0.0019 | 7 | 0.0003 | ||
| Lack of Fit | 0.0015 | 3 | 0.0005 | 5.60 | 0.0648 (not significant) |
| Pure Error | 0.0004 | 4 | 0.0001 |
| Statistical Parameters | Values of Model |
|---|---|
| Std. Dev. | 0.02 |
| R² | 0.99 |
| Adjusted R² | 0.98 |
| Adeq Precision | 33.76 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Wang, J.; Zhang, E.; Shao, Y.; Yang, L.; Yang, B.; Tan, Y.; Gao, T. Cumulative Effects of Physical, Chemical, and Biological Measures on Algae Growth Inhibition. Water 2022, 14, 877. https://doi.org/10.3390/w14060877
Li H, Wang J, Zhang E, Shao Y, Yang L, Yang B, Tan Y, Gao T. Cumulative Effects of Physical, Chemical, and Biological Measures on Algae Growth Inhibition. Water. 2022; 14(6):877. https://doi.org/10.3390/w14060877
Chicago/Turabian StyleLi, Hao, Jiaqi Wang, Enze Zhang, Yanan Shao, Lin Yang, Baiheng Yang, Yi Tan, and Ting Gao. 2022. "Cumulative Effects of Physical, Chemical, and Biological Measures on Algae Growth Inhibition" Water 14, no. 6: 877. https://doi.org/10.3390/w14060877





