Assessment of Levonorgestrel Leaching in a Landfill and Its Effects on Placental Cell Lines and Sperm Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Soil Layer Sampling and Monitoring of LNG in the Effluent of the UASB Reactor from the Buenavista Landfill
2.3. Leaching Analysis of LNG from the Soil Layers under Laboratory Conditions
2.4. Validation Parameters for LNG Analysis by uHPLC–DAD
2.5. Assessment of Leached Fractions in Human Sperm Cells
2.6. Effects of Leachate Sample from the UASB Reactor on BeWo Cells and Their Production of β-hCG
2.7. Statistical Analysis and Graphs
2.8. Ethical Statements
3. Results
3.1. Validation Parameters for LNG Analysis by uHPLC–DAD
3.2. Analysis of LNG in the Effluent of the UASB Reactor from the Buenavista Landfill
3.3. Assessment of Leaching Processes for LNG in Soil Layers Used as Cover Materials in the Buenavista Landfill
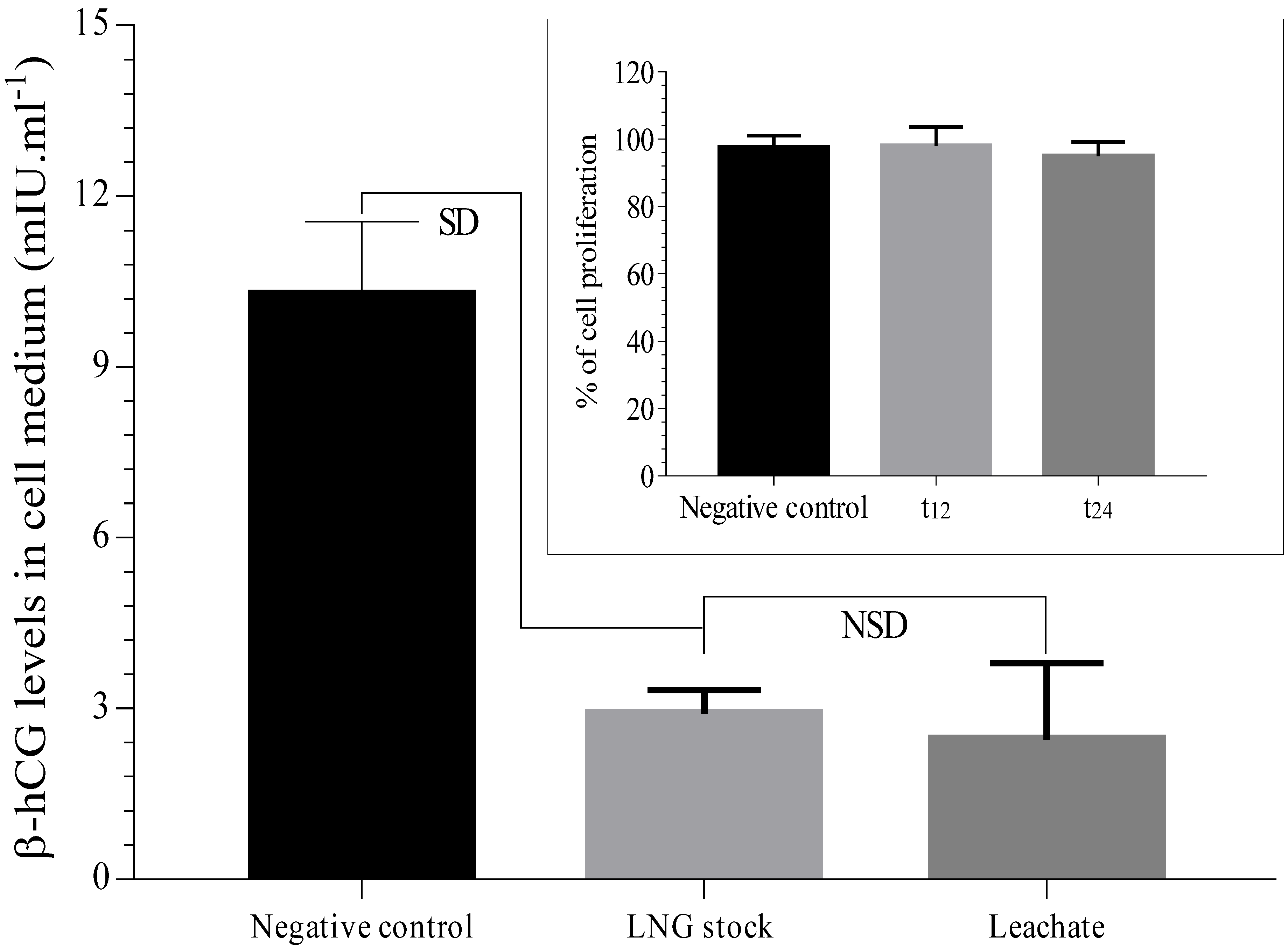
3.4. Assessment of Leachate Sample in Placental Cell Lines (BeWo) and Sperm Cells
3.5. A Possible Implication of Leached LNG in Medellín, Colombia
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Narvaez, J.F.; Jimenez, C. Pharmaceutical Products in the Environment: Sources, Effects and Risks. Vitae 2012, 19, 93–108. [Google Scholar]
- Tang, T.; Shi, T.; Li, D.; Xia, J.; Hu, Q.; Cao, Y. Adsorption properties and degradation dynamics of endocrine-disrupting chemical levonorgestrel in soils. J. Agric. Food Chem. 2012, 60, 3999–4004. [Google Scholar] [CrossRef] [PubMed]
- Padhye, L.P.; Yao, H.; Kung’U, F.T.; Huang, C.-H. Year-long evaluation on the occurrence and fate of pharmaceuticals, personal care products, and endocrine disrupting chemicals in an urban drinking water treatment plant. Water Res. 2014, 51, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Christen, V.; Hickmann, S.; Rechenberg, B.; Fent, K. Highly active human pharmaceuticals in aquatic systems: A concept for their identification based on their mode of action. Aquat. Toxicol. 2010, 96, 167–181. [Google Scholar] [CrossRef]
- Bonneuil, N.; Medina, M. Between Tradition and Modernity: The Transition of Contraception Use in Colombia. Rev. Desarro. Soc. 2009, 2, 119–151. [Google Scholar] [CrossRef]
- Africander, D.; Verhoog, N.; Hapgood, J.P. Molecular mechanisms of steroid receptor-mediated actions by synthetic progestins used in HRT and contraception. Steroids 2011, 76, 636–652. [Google Scholar] [CrossRef]
- Kroupova, H.K.; Trubiroha, A.; Lorenz, C.; Contardo-Jara, V.; Lutz, I.; Grabic, R.; Kocour, M.; Kloas, W. The progestin levonorgestrel disrupts gonadotropin expression and sex steroid levels in pubertal roach (Rutilus rutilus). Aquat. Toxicol. 2014, 154, 154–162. [Google Scholar] [CrossRef]
- Olivera, E.R.; Luengo, J.M. Steroids as Environmental Compounds Recalcitrant to Degradation: Genetic Mechanisms of Bacterial Biodegradation Pathways. Genes 2019, 10, 512. [Google Scholar] [CrossRef] [Green Version]
- Narvaez, J.F.; Grant, H.; Gil, V.C.; Porras, J.; Sanchez, J.C.B.; Duque, L.F.O.; Sossa, R.R.; Quintana-Castillo, J.C. Assessment of endocrine disruptor effects of levonorgestrel and its photoproducts: Environmental implications of released fractions after their photocatalytic removal. J. Hazard. Mater. 2019, 371, 273–279. [Google Scholar] [CrossRef]
- Yu, X.; Sui, Q.; Lyu, S.; Zhao, W.; Wu, D.; Yu, G.; Barcelo, D. Rainfall Influences Occurrence of Pharmaceutical and Personal Care Products in Landfill Leachates: Evidence from Seasonal Variations and Extreme Rainfall Episodes. Environ. Sci. Technol. 2021, 55, 4822–4830. [Google Scholar] [CrossRef]
- Torres-González, O.R.; Sánchez-Hernández, I.M.; Flores-Soto, M.E.; Chaparro-Huerta, V.; Soria-Fregozo, C.; Hernández-García, L.; Padilla-Camberos, E.; Flores-Fernández, J.M. Landfill Leachate from an Urban Solid Waste Storage System Produces Genotoxicity and Cytotoxicity in Pre-Adolescent and Young Adults Rats. Int. J. Environ. Res. Public Health 2021, 18, 11029. [Google Scholar] [CrossRef]
- Yang, X.; Lin, H.; Zhang, Y.; He, Z.; Dai, X.; Zhang, Z.; Li, Y. Sorption and desorption of seven steroidal synthetic progestins in five agricultural soil-water systems. Ecotoxicol. Environ. Saf. 2020, 196, 110586. [Google Scholar] [CrossRef] [PubMed]
- Fent, K. Progestins as endocrine disrupters in aquatic ecosystems: Concentrations, effects and risk assessment. Environ. Int. 2015, 84, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Klavarioti, M.; Mantzavinos, D.; Kassinos, D. Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ. Int. 2009, 35, 402–417. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Laboratory Manual for the Examination and Processing of Human Semen, 6th ed.; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Lomonte, B.; Gutiérrez, J.; Romero, M.; Núñez, J.; Tarkowski, A.; Hanson, L. An MTT-based method for the in vivo quantification of myotoxic activity of snake venoms and its neutralization by antibodies. J. Immunol. Methods 1993, 161, 231–237. [Google Scholar] [CrossRef]
- Oropesa, A.L.; Guimaraes, L. Occurrence of Levonorgestrel in Water Systems and Its Effects on Aquatic Organisms: A Review. Rev. Environ. Contam. Toxicol. 2021, 254, 57–84. [Google Scholar]
- Huang, Y.; Lin, H.; Song, R.; Tian, Y.; Zhang, Z. Optimization of dispersive liquid-liquid microextraction for analysis of levonorgestrel in water samples using uniform design. Anal. Methods 2011, 3, 857–864. [Google Scholar] [CrossRef]
- Kot-Wasik, A.; Jakimska, A.; Sliwka-Kaszynska, M. Occurrence and seasonal variations of 25 pharmaceutical residues in wastewater and drinking water treatment plants. Environ. Monit. Assess. 2016, 188, 661. [Google Scholar] [CrossRef]
- Yu, Q.; Geng, J.; Zong, X.; Zhang, Y.; Xu, K.; Hu, H.; Deng, Y.; Zhao, F.; Ren, H. Occurrence and removal of progestagens in municipal wastewater treatment plants from different regions in China. Sci. Total Environ. 2019, 668, 1191–1199. [Google Scholar] [CrossRef]
- Fick, J.; Lindberg, R.H.; Parkkonen, J.; Arvidsson, B.; Tysklind, M.; Larsson, D.G.J. Therapeutic levels of levonorgestrel detected in blood plasma of fish: Results from screening rainbow trout exposed to treated sewage effluents. Environ. Sci. Technol. 2010, 44, 2661–2666. [Google Scholar] [CrossRef]
- Hua, J.; Han, J.; Guo, Y.; Zhou, B. The progestin levonorgestrel affects sex differentiation in zebrafish at environmentally relevant concentrations. Aquat. Toxicol. 2015, 166, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Liu, X.; Ding, S.; Su, S.; Gan, Z. Sorption and leaching behavior of bithionol and levamisole in soils. Chemosphere 2019, 224, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Grathwohl, P. Influence of Organic Matter from Soils and Sediments from Various Origins on the Sorption of Some Chlorinated Aliphatic Hydrocarbons: Implications on Koc Correlations. Environ. Sci. Technol. 1990, 24, 1687–1693. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Thiele-Bruhn, S.; Aziz, S.G.; Hilal, R.H.; Elroby, S.A.; Al-Youbi, A.O.; Leinweber, P.; Kühn, O. Interaction of polar and nonpolar organic pollutants with soil organic matter: Sorption experiments and molecular dynamics simulation. Sci. Total Environ. 2015, 508, 276–287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Dai, X.; Zhang, Y.; Lin, H.; Wang, J.; He, Z.; Li, Y. Sorption, desorption, and transformation of synthetic progestins in soil and sediment systems. Geoderma 2020, 362, 114141. [Google Scholar] [CrossRef]
- Lambert, J.-F. 7-Organic pollutant adsorption on clay minerals. In Developments in Clay Science; Elsevier: Amsterdam, The Netherlands, 2018; Volume 9. [Google Scholar]
- Jianghuan, H.J.H.; Yongyong, G.; Bingsheng, Z. Reproductive Toxicity in Male Zebrafish after Long-term Exposure to Low Concentrations of Progestin Levonorgestrel. Asian J. Ecotoxicol. 2015, 10, 100–107. [Google Scholar]
- Alfradique, V.A.P.; Batista, R.I.T.P.; Souza-Fabjan, J.M.G.; Côrtes, L.R.; Bragança, G.M.; de Souza, C.V.; da Costa, L.C.; Brandão, F.Z. Supplementation of 17beta-estradiol and progesterone in the co-culture medium of bovine oviductal epithelial cells and ovine spermatozoa reduces the sperm kinematics and capacitation. Reprod. Biol. 2018, 18, 368–379. [Google Scholar] [CrossRef]
- Frankel, T.; Yonkos, L.; Ampy, F.; Frankel, J. Exposure to levonorgestrel increases nest acquisition success and decreases sperm motility in the male fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 2018, 37, 1131–1137. [Google Scholar] [CrossRef]
- Lorenz, C.; Contardo-Jara, V.; Trubiroha, A.; Krüger, A.; Viehmann, V.; Wiegand, C.; Pflugmacher, S.; Nützmann, G.; Lutz, I.; Kloas, W. The synthetic gestagen Levonorgestrel disrupts sexual development in Xenopus laevis by affecting gene expression of pituitary gonadotropins and gonadal steroidogenic enzymes. Toxicol. Sci. 2011, 124, 311–319. [Google Scholar] [CrossRef]
- Overturf, M.D.; Huggett, D.B. Responses to various exposure durations of levonorgestrel during early-life stages of fathead minnows (Pimephales promelas). Aquat. Toxicol. 2015, 161, 33–40. [Google Scholar] [CrossRef]
- Stanczyk, F.Z.; Hapgood, J.; Winer, S.; Mishell, D.R. Progestogens used in postmenopausal hormone therapy: Differences in their pharmacological properties, intracellular actions, and clinical effects. Endocr. Rev. 2013, 34, 171–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Botero-Coy, A.M.; Martínez-Pachón, D.; Boix, C.; Rincón, R.J.; Castillo, N.; Arias-Marín, L.; Manrique-Losada, R.; Torres-Palma, R.; Moncayo-Lasso, A.; Hernández, F. ‘An investigation into the occurrence and removal of pharmaceuticals in Colombian wastewater’. Sci. Total Environ. 2018, 642, 842–853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, C.; Liu, W.; Qu, J.; Liu, M.; Zhang, Y.; Zhao, M. Degradation of the potential rodent contraceptive quinestrol and elimination of its estrogenic activity in soil and water. Environ. Sci. Pollut. Res. Int. 2014, 21, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, E.; Giwercman, A.; Keiding, N.; Skakkebaek, N.E. Evidence for decreasing quality of semen during past 50 years. BMJ 1992, 305, 609–613. [Google Scholar] [CrossRef] [Green Version]
- Crisp, T.M.; Clegg, E.D.; Cooper, R.L. Special Report on Environmental Endocrine Disruption An Effects Assessment and Analysis; U.S. Environmental Protection Agency: Washington, DC, USA, 1997; p. 11.
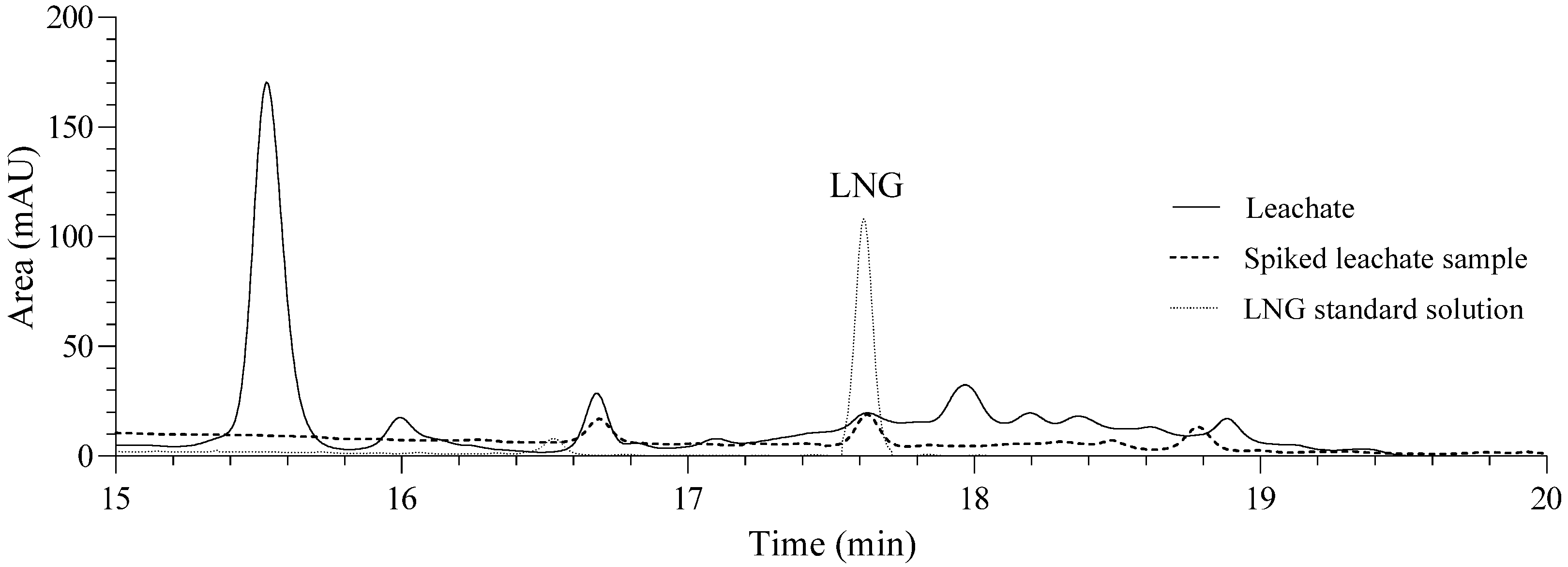


| LNG | Rt (min) | r2 | LOD (ppm) | LOQ (ppm) | Linearity range (ppm) | Linear equation | Log Kow [17] |
| 17.63 | 0.998 | 0.09 | 0.30 | 0.3–10.5 | y = 0.7271x + 0.6011 | 3.48 |
| Property | Layer 1 (L1) | Layer 2 (L2) | Layer 3 (L3) |
|---|---|---|---|
| Soil texture | Clay sandy loam | Sandy loam | Loam |
| % Clay | 50 | 36 | 44 |
| % Silt | 18 | 26 | 44 |
| % Sand | 32 | 38 | 12 |
| pH | 5.6 | 5.1 | 5.1 |
| Org. matter (%) | 4.1 | 0.15 | 0.12 |
| Al (cmol kg−1) | – | 1.4 | 2 |
| Ca (cmol kg−1) | 0.2 | 0.1 | 0.1 |
| p | 1.0 | 2.0 | 8 |
| CEC (cmol kg−1) | 0.2 | 1.5 | 2.1 |
| Soil water infiltration (%) | 75.6 | 42.6 | 31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ríos-Sossa, R.; García-Londoño, J.J.; Gil-Ramírez, D.; Patiño, A.C.; Cardona-Maya, W.D.; Quintana-Castillo, J.C.; Narváez-Valderrama, J.F. Assessment of Levonorgestrel Leaching in a Landfill and Its Effects on Placental Cell Lines and Sperm Cells. Water 2022, 14, 871. https://doi.org/10.3390/w14060871
Ríos-Sossa R, García-Londoño JJ, Gil-Ramírez D, Patiño AC, Cardona-Maya WD, Quintana-Castillo JC, Narváez-Valderrama JF. Assessment of Levonorgestrel Leaching in a Landfill and Its Effects on Placental Cell Lines and Sperm Cells. Water. 2022; 14(6):871. https://doi.org/10.3390/w14060871
Chicago/Turabian StyleRíos-Sossa, Ramiro, Juan José García-Londoño, Daniel Gil-Ramírez, Arley Camilo Patiño, Walter D. Cardona-Maya, Juan Carlos Quintana-Castillo, and Jhon Fredy Narváez-Valderrama. 2022. "Assessment of Levonorgestrel Leaching in a Landfill and Its Effects on Placental Cell Lines and Sperm Cells" Water 14, no. 6: 871. https://doi.org/10.3390/w14060871







