Kinetics of Chromium Reduction Associated with Varying Characteristics of Agricultural Soils
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection and Characterization of Soil Samples
2.2. Incubation Experiments
2.3. Calculation and Statistical Analysis
3. Results and Discussion
3.1. Soil Characteristics
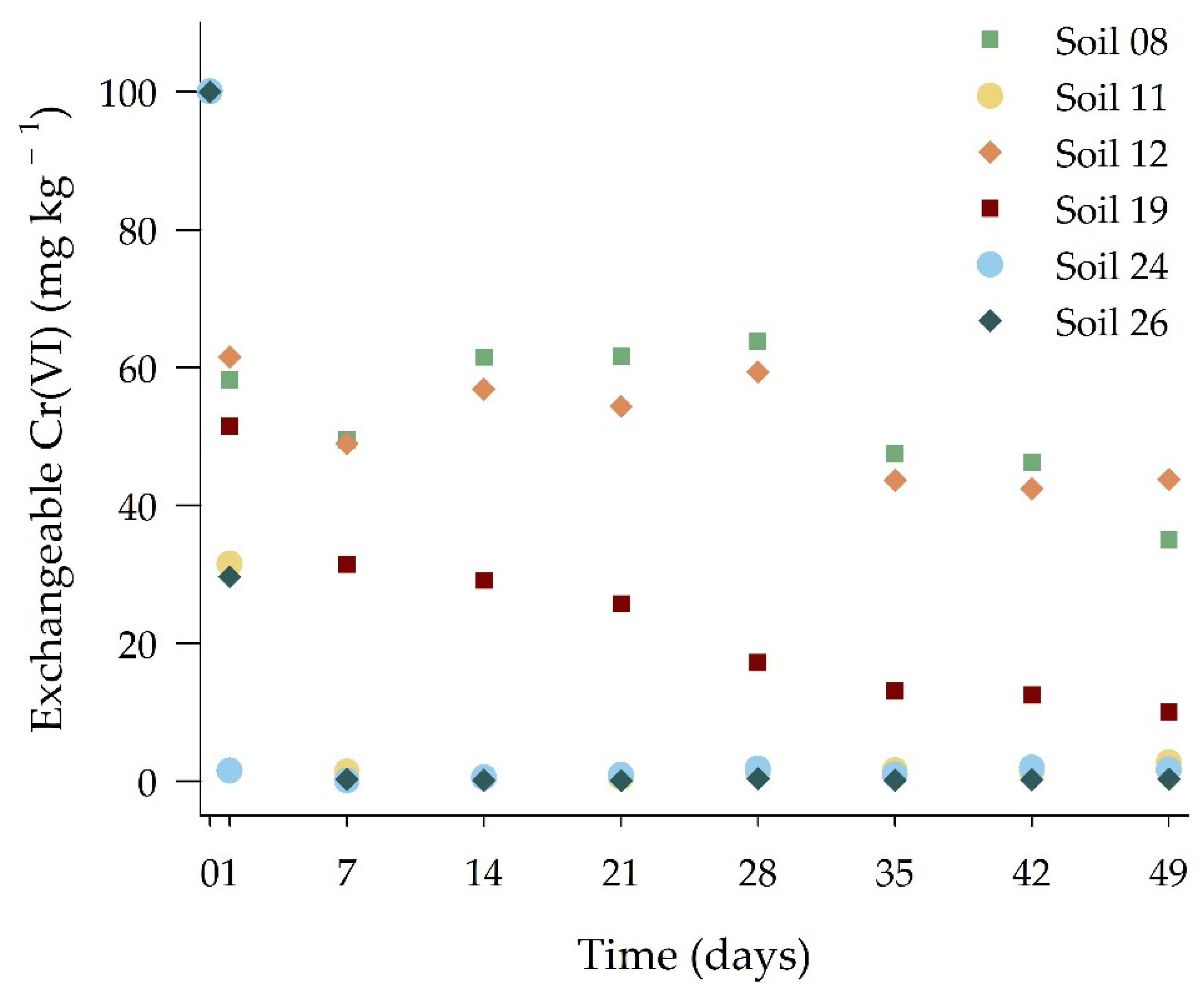
3.2. Reduction Processes of Chromium (VI) in the Studied Soils
3.3. The Relationship between Hexavalent Chromium Reduction and Soil Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taylor, S.R.; McLennan, S. The geochemical evolution of the continental crust. Rev. Geophys. 1995, 33, 241–265. [Google Scholar] [CrossRef]
- Prasad, S.; Yadav, K.K.; Kumar, S.; Gupta, N.; Cabral-Pinto, M.M.; Rezania, S.; Radwan, N.; Alam, J. Chromium contamination and effect on environmental health and its remediation: A sustainable approaches. J. Environ. Manag. 2021, 285, 112174. [Google Scholar] [CrossRef]
- Zayed, A.M.; Terry, N. Chromium in the environment: Factors affecting biological remediation. Plant Soil 2003, 249, 139–156. [Google Scholar] [CrossRef]
- Yoshinaga, M.; Ninomiya, H.; Al Hossain, M.A.; Sudo, M.; Akhand, A.A.; Ahsan, N.; Alim, A.; Khalequzzaman; Iida, M.; Yajima, I.; et al. A comprehensive study including monitoring, assessment of health effects and development of a remediation method for chromium pollution. Chemosphere 2018, 201, 667–675. [Google Scholar] [CrossRef]
- Vincent, J.B. The biochemistry of chromium. J. Nutr. 2000, 130, 715–718. [Google Scholar] [CrossRef]
- Whitacre, D.M. Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2008; Volume 202. [Google Scholar]
- Costa, M.; Klein, C. Toxicity and carcinogenicity of chromium compounds in humans. Crit. Rev. Toxicol. 2006, 36, 155–163. [Google Scholar] [CrossRef]
- Shanker, A.K.; Cervantes, C.; Loza-Tavera, H.; Avudainayagam, S. Chromium toxicity in plants. Environ. Int. 2005, 31, 739–753. [Google Scholar] [CrossRef]
- Xu, T.; Nan, F.; Jiang, X.; Tang, Y.; Zeng, Y.; Zhang, W.; Shi, B. Effect of soil pH on the transport, fractionation, and oxidation of chromium(III). Ecotoxicol. Environ. Saf. 2020, 195, 110459. [Google Scholar] [CrossRef]
- Lv, D.; Zhou, J.; Cao, Z.; Xu, J.; Liu, Y.; Li, Y.; Yang, K.; Lou, Z.; Lou, L.; Xu, X. Mechanism and influence factors of chromium(VI) removal by sulfide-modified nanoscale zerovalent iron. Chemosphere 2019, 224, 306–315. [Google Scholar] [CrossRef]
- Bartlett, R.J.; James, B. Mobility and bioavailability of chromium in soils. In Chromium in the Natural and Human Environments; Nriagu, J.O., Nieboer, E., Eds.; Wiley Interscience: New York, NY, USA, 1988; Volume 20, p. 571. [Google Scholar]
- Bartlett, R.J. Chromium cycling in soils and water: Links, gaps, and methods. Environ. Health Perspect. 1991, 92, 17–24. [Google Scholar] [CrossRef]
- Richard, F.C.; Bourg, A.C. Aqueous geochemistry of chromium: A review. Water Res. 1991, 25, 807–816. [Google Scholar] [CrossRef]
- Dong, X.; Ma, L.Q.; Gress, J.; Harris, W.; Li, Y. Enhanced Cr(VI) reduction and As(III) oxidation in ice phase: Important role of dissolved organic matter from biochar. J. Hazard. Mater. 2014, 267, 62–70. [Google Scholar] [CrossRef]
- Eary, L.E.; Rai, D. Kinetics of chromium(III) oxidation to chromium(VI) by reaction with manganese dioxide. Environ. Sci. Technol. 1987, 21, 1187–1193. [Google Scholar] [CrossRef]
- Rai, D.; Eary, L.E.; Zachara, J.M. Environmental chemistry of chromium. Sci. Total Environ. 1989, 86, 15–23. [Google Scholar] [CrossRef]
- Reijonen, I.; Hartikainen, H. Oxidation mechanisms and chemical bioavailability of chromium in agricultural soil–pH as the master variable. Appl. Geochem. 2016, 74, 84–93. [Google Scholar] [CrossRef]
- Evans, L.J. Chemistry of metal retention by soils. Environ. Sci. Technol. 1989, 23, 1046–1056. [Google Scholar] [CrossRef]
- Losi, M.E.; Amrhein, C.; Frankenberger, W.T., Jr. Factors affecting chemical and biological reduction of hexavalent chromium in soil. Environ. Toxicol. Chem. 1994, 13, 1727–1735. [Google Scholar] [CrossRef]
- Xiao, W.; Zhang, Y.; Li, T.; Chen, B.; Wang, H.; He, Z.; Yang, X. Reduction kinetics of hexavalent chromium in soils and its correlation with soil properties. J. Environ. Qual. 2012, 41, 1452–1458. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Peng, Y.; Yang, Z.; Cheng, P.; Li, F.; Wang, M.; Liu, T. The kinetics of aging and reducing processes of Cr(VI) in two soils. Bull. Environ. Contam. Toxicol. 2019, 103, 82–89. [Google Scholar] [CrossRef]
- Staff, S.S. Keys to Soil Taxonomy, 12th ed.; USDA Soil Conservation Service: Washington, DC, USA, 2014.
- Chen, Z.-S.; Hseu, Z.-Y.; Tsai, C.-C. The Soils of Taiwan; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar]
- Gee, G.W.; Bauder, J.W. Particle size analysis by hydrometer: A simplified method for routine textural analysis and a sensitivity test of measurement parameters. Soil Sci. Soc. Am. J. 1979, 43, 1004–1007. [Google Scholar] [CrossRef]
- McLean, E.O. Soil pH and lime requirement. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Ed.; ASA, Inc.; SSSA, Inc.: Madison, WI, USA, 1983; Volume 9, pp. 199–224. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis: Part 3 Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Eds.; ASA, Inc.; SSSA, Inc.: Madison, WI, USA, 1996; Volume 5, pp. 961–1010. [Google Scholar]
- Rhoades, J. Cation exchange capacity. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Ed.; ASA, Inc.; SSSA, Inc.: Madison, WI, USA, 1983; Volume 9, pp. 149–157. [Google Scholar]
- Mehra, O.P.; Jackson, M.L. Iron oxide removal from soils and clays by a dithionite-citrate system buffered with bicarbonate. In Proceedings of the 7th National Conference on Clays and Clay Minerals, Washington, DC, USA, 20–23 October 1958; Swineford, A., Ed.; Pergamon: London, UK, 2013; pp. 317–327. [Google Scholar]
- Sherman, G.D.; McHARGUE, J.S.; Hodgkiss, W.S. Determination of active manganese in soil. Soil Sci. 1942, 54, 253–258. [Google Scholar] [CrossRef]
- Vitale, R.J.; Mussoline, G.R.; Petura, J.C.; James, B.R. Cr(vi) soil analytical method: A reliable analytical method for extracting and quantifying Cr(vi) in soils. Soil Sediment Contam. Int. J. 1997, 6, 581–593. [Google Scholar] [CrossRef]
- Jones, D.L.; Willett, V.B. Experimental evaluation of methods to quantify dissolved organic nitrogen (DON) and dissolved organic carbon (DOC) in soil. Soil Biol. Biochem. 2006, 38, 991–999. [Google Scholar] [CrossRef]
- Brookes, P.; Landman, A.; Pruden, G.; Jenkinson, D. Chloroform fumigation and the release of soil nitrogen: A rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 1985, 17, 837–842. [Google Scholar] [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Guertin, J.; Jacobs, J.A.; Avakian, C.P. Chromium (VI) Handbook; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Wittbrodt, P.R.; Palmer, C.D. Reduction of Cr(VI) in the presence of excessive soil fulvic acid. Environ. Sci. Technol. 1995, 29, 255–263. [Google Scholar] [CrossRef]
| Item | Mean | SD | Min. | Max. |
|---|---|---|---|---|
| Total Cr (mg kg−1) | 124 | 293 | 15.8 | 1520 |
| Total Cr(VI) (mg kg−1) | 8.66 | 8.48 | 1.34 | 29.4 |
| pH | 6.22 | 1.24 | 4.00 | 8.10 |
| OC (g kg−1) | 18.9 | 26.2 | 2.64 | 150 |
| DOC (mg kg−1) | 10.6 | 9.36 | 3.61 | 47.4 |
| CEC (cmol(+) kg−1) | 14.4 | 10.6 | 3.62 | 48.2 |
| Fed (g kg−1) | 16.4 | 9.72 | 1.22 | 43.8 |
| Mnd (g kg−1) | 0.30 | 0.17 | 0.03 | 0.87 |
| Mnr (mg kg−1) | 87.3 | 80.4 | 2.00 | 355 |
| Sand (%) | 23.7 | 19.5 | 2.00 | 79.0 |
| Silt (%) | 34.1 | 14.0 | 6.00 | 59.0 |
| Clay (%) | 42.2 | 15.3 | 15.0 | 80.0 |
| MBC (g C kg−1) | 1.02 | 0.87 | 0.02 | 3.64 |
| MBN (g N kg−1) | 0.65 | 0.36 | 0.08 | 1.39 |
| Soil Code | Reduction Rate Constant, k | R2 | p Value |
|---|---|---|---|
| 01 | 1.02 | 0.99 | <0.0001 |
| 02 | 0.03 | 0.63 | 0.0215 |
| 03 | 0.04 | 0.61 | 0.0289 |
| 04 | 1.30 | 1.00 | <0.0001 |
| 05 | 1.04 | 0.99 | <0.0001 |
| 06 | 0.02 | 0.50 | 0.0404 |
| 07 | 0.80 | 0.98 | 0.0005 |
| 08 | 0.01 | 0.47 | 0.0497 |
| 09 | 1.07 | 0.99 | <0.0001 |
| 10 | 0.78 | 1.00 | <0.0001 |
| 11 | 1.15 | 1.00 | <0.0001 |
| 12 | 0.01 | 0.49 | 0.0380 |
| 13 | 0.02 | 0.51 | 0.0343 |
| 14 | 0.03 | 0.60 | 0.0222 |
| 15 | 0.71 | 0.92 | 0.0167 |
| 16 | 1.24 | 1.00 | <0.0001 |
| 17 | 0.13 | 0.83 | 0.0203 |
| 18 | 0.37 | 0.97 | <0.0001 |
| 19 | 0.06 | 0.78 | 0.0119 |
| 20 | 0.62 | 1.00 | <0.0001 |
| 21 | 0.73 | 1.00 | <0.0001 |
| 22 | 0.07 | 0.85 | 0.0080 |
| 23 | 0.01 | 0.47 | 0.0436 |
| 24 | 4.21 | 1.00 | 0.0018 |
| 25 | 0.82 | 0.95 | 0.0042 |
| 26 | 1.22 | 1.00 | <0.0001 |
| 27 | 0.79 | 0.97 | 0.0008 |
| 28 | 0.02 | 0.68 | 0.0091 |
| Item | pH | OM | DOC | CEC | Fed | Mnd | Mnr | Sand | Silt | Clay | MBC | MBN |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OM | −0.27 | |||||||||||
| DOC | −0.13 | −0.05 | ||||||||||
| CEC | −0.38 * | 0.67 *** | 0.16 | |||||||||
| Fed | −0.61 *** | 0.01 | −0.07 | 0.46 * | ||||||||
| Mnd | −0.01 | −0.25 | −0.19 | 0.17 | 0.38 * | |||||||
| Mnr | 0.32 | −0.14 | −0.05 | 0.29 | 0.12 | 0.82 *** | ||||||
| Sand | 0.10 | −0.07 | 0.15 | −0.27 | −0.58 ** | −0.31 | −0.19 | |||||
| Silt | 0.41 * | 0.25 | −0.14 | 0.11 | −0.08 | 0.23 | 0.38 * | −0.63 *** | ||||
| Clay | −0.50 ** | −0.14 | −0.06 | 0.23 | 0.81 *** | 0.18 | −0.10 | −0.70 *** | −0.11 | |||
| MBC | −0.12 | −0.19 | −0.05 | −0.21 | 0.16 | −0.17 | −0.14 | 0.17 | −0.04 | −0.18 | ||
| MBN | −0.24 | −0.21 | 0.37 | −0.04 | 0.65 *** | −0.35 | −0.42 * | −0.25 | −0.14 | 0.47 * | 0.21 | |
| k | −0.38 * | 0.85 *** | 0.29 | 0.58 ** | −0.04 | −0.44 * | −0.29 | 0.08 | 0.10 | −0.19 | 0.12 | 0.07 |
| Parameter | Parameter Estimate | p Value | Partial R2 | Model R2 |
|---|---|---|---|---|
| Intercept | 0.158 | 0.382 | ||
| OC | 0.015 | 0.000 | 0.73 | 0.73 |
| DOC | 0.027 | 0.000 | 0.11 | 0.84 |
| Mnd | −0.939 | 0.024 | 0.03 | 0.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.-Y.; Tseng, Y.-L.; Hseu, Z.-Y. Kinetics of Chromium Reduction Associated with Varying Characteristics of Agricultural Soils. Water 2022, 14, 570. https://doi.org/10.3390/w14040570
Yang C-Y, Tseng Y-L, Hseu Z-Y. Kinetics of Chromium Reduction Associated with Varying Characteristics of Agricultural Soils. Water. 2022; 14(4):570. https://doi.org/10.3390/w14040570
Chicago/Turabian StyleYang, Chia-Yu, Yan-Lin Tseng, and Zeng-Yei Hseu. 2022. "Kinetics of Chromium Reduction Associated with Varying Characteristics of Agricultural Soils" Water 14, no. 4: 570. https://doi.org/10.3390/w14040570






