Synthesis of Fumed-Pr-Pi-TCT as a Fluorescent Chemosensor for the Detection of Cyanide Ions in Aqueous Media
Abstract
:1. Introduction
2. Materials and Methods
2.1. Instruments and Materials
2.2. Methods
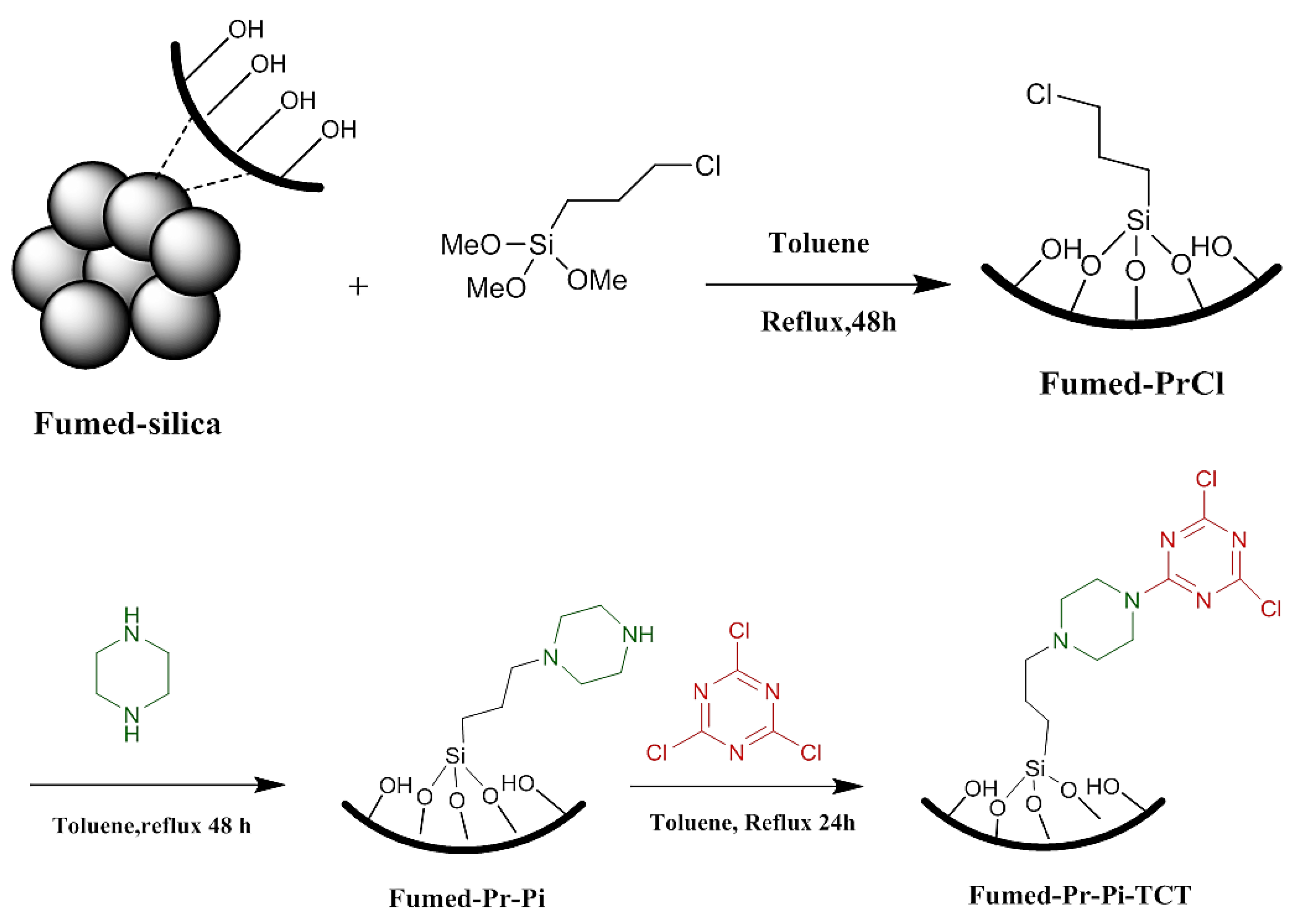
2.2.1. General Synthesis of Fumed-Pr–Pi-TCT
2.2.2. Preparation of 10−3 M Aqueous Solution of Fumed-Pr-Pi-TCT in H2O
2.2.3. Synthesis of Fumed-Pr-Pi-TCT
3. Results and Discussion
3.1. Characterization Data
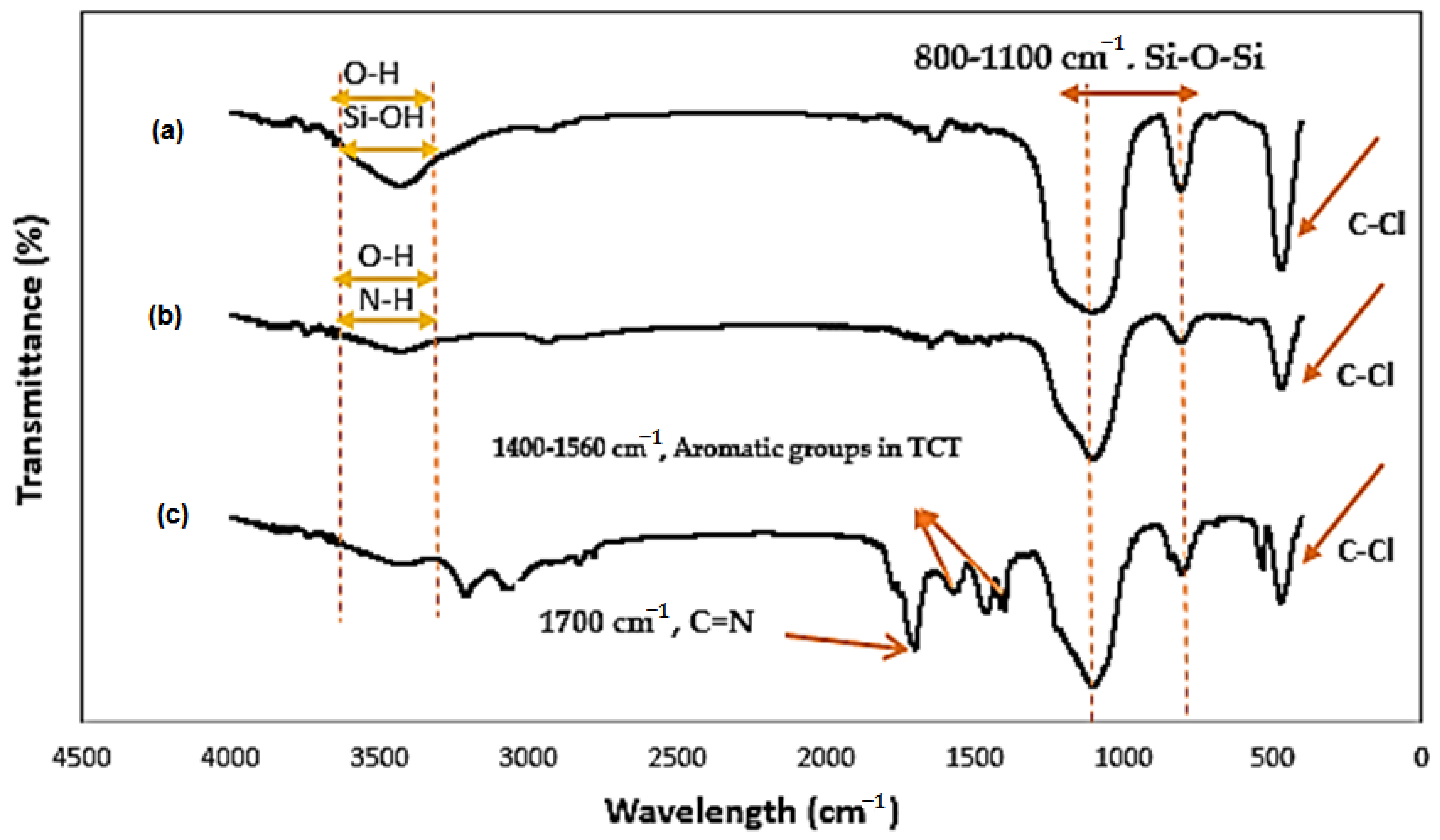
3.1.1. FT-IR Studies
3.1.2. Thermogravimetric Analysis (TGA) Studies
3.1.3. SEM Studies
3.2. Fluorescence Response of Fumed-Pr–Pi-TCT for CNˉ
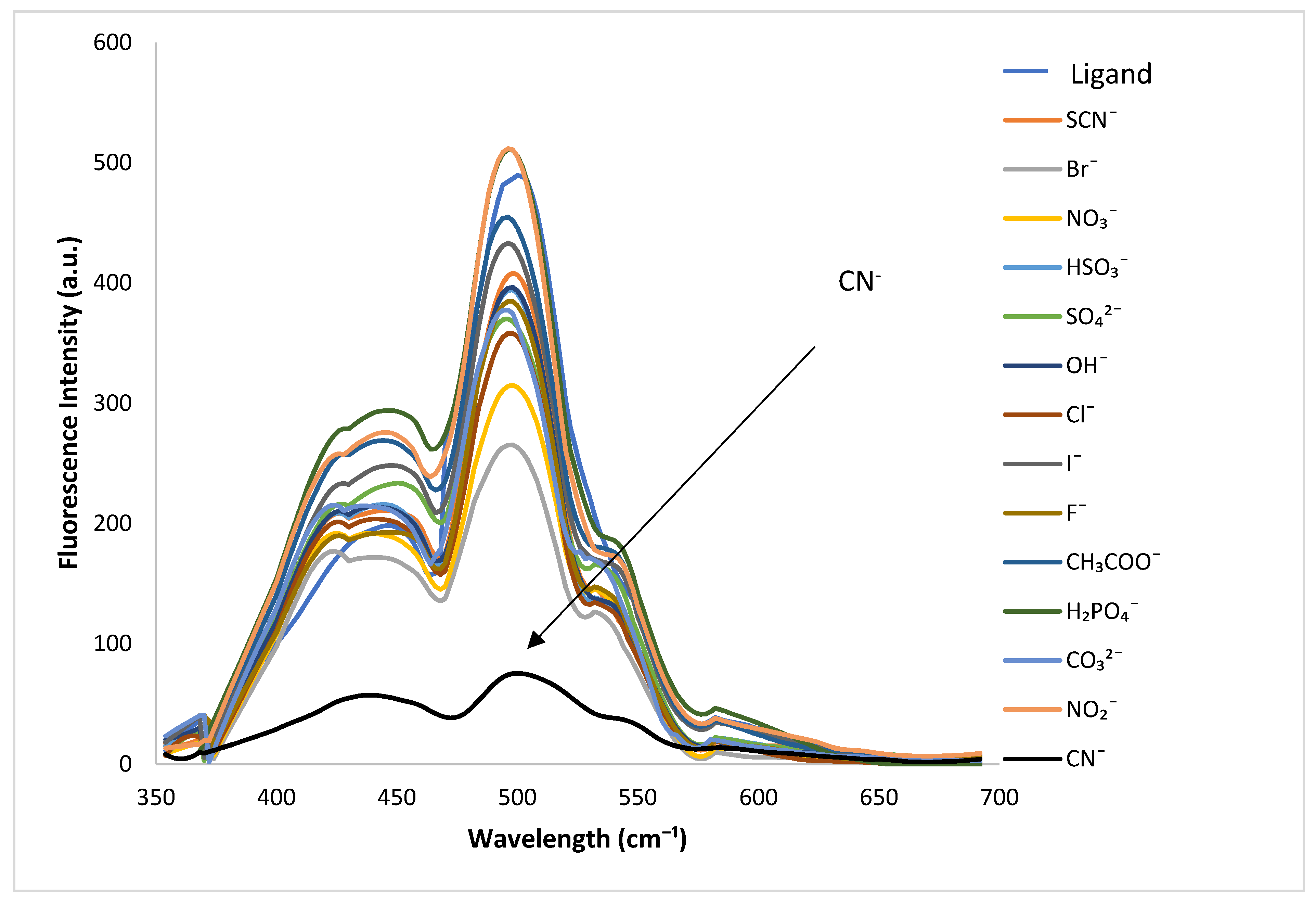
3.2.1. Selectivity Studies
3.2.2. Titration Studies
3.2.3. Competition Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ishii, A.; Seno, H.; Watanabe-Suzuki, K.; Suzuki, O.; Kumazawa, T. Determination of cyanide in whole blood by capillary gas chromatography with cryogenic oven trapping. Anal. Chem. 1998, 70, 4873–4876. [Google Scholar] [CrossRef] [PubMed]
- Nelson, L. Acute cyanide toxicity: Mechanisms and manifestations. J. Emerg. Nurs. 2006, 32, S8–S11. [Google Scholar] [CrossRef] [PubMed]
- Borron, S.W. Recognition and treatment of acute cyanide poisoning. J. Emerg. Nurs. 2006, 32, S12–S18. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, D.; Xiao, X.; Su, C.; Li, Z.; Xue, J.; Hu, N.; Peng, P.; Liao, L.; Wang, H. A highly sensitive and selective sensor for trace uranyl (VI) ion based on a graphene-coated carbon paste electrode modified with ion imprinted polymer. Microchem. J. 2020, 155, 104767. [Google Scholar] [CrossRef]
- Bai, C.-B.; Zhang, J.; Qiao, R.; Zhang, Q.-Y.; Mei, M.-Y.; Chen, M.-Y.; Wei, B.; Wang, C.; Qu, C.-Q. Reversible and Selective Turn-on Fluorescent and Naked-Eye Colorimetric Sensor to Detect Cyanide in Tap Water, Food Samples, and Living Systems. Ind. Eng. Chem. Res. 2020, 59, 8125–8135. [Google Scholar] [CrossRef]
- László, F. Lessons learned from the cyanide and heavy metal accidental water pollution in the Tisa River basin in the year 2000. In JSTOR; Springer: Berlin/Heidelberg, Germany, 2006; pp. 43–50. [Google Scholar]
- Vasudevan, S.; Oturan, M.A. Electrochemistry: As cause and cure in water pollution. Environ. Chem. Lett. 2014, 12, 97–108. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, Y.; Tang, J.; Tang, W. Surface ligands engineering of semiconductor quantum dots for chemosensory and biological applications. Mater. Today Commun. 2017, 20, 360–376. [Google Scholar] [CrossRef]
- Yung, K.Y.; Schadock-Hewitt, A.J.; Hunter, N.P.; Bright, F.V.; Baker, G.A. ‘Liquid litmus’: Chemosensory pH-responsive photonic ionic liquids. Commun. Chem. 2011, 47, 4775–4777. [Google Scholar] [CrossRef]
- Karimi, M.; Badiei, A.; Mohammadi Ziarani, G. A single hybrid optical sensor based on nanoporous silica type SBA-15 for detection of Pb 2+ and I− in aqueous media. RSC Adv. 2015, 5, 36530–36539. [Google Scholar] [CrossRef]
- Arumugam, N.; Kim, J. Synthesis of carbon quantum dots from Broccoli and their ability to detect silver ions. Mater. Lett. 2018, 219, 37–40. [Google Scholar] [CrossRef]
- Wei, T.-B.; Li, W.-T.; Li, Q.; Su, J.-X.; Qu, W.-J.; Lin, Q.; Yao, H.; Zhang, Y.-M. A turn-on fluorescent chemosensor selectively detects cyanide in pure water and food sample. Tetrahedron Lett. 2016, 57, 2767–2771. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, Z.; Yang, J.; Yi, L.; Liao, L.; Xiao, X. Development of a method for the detection of Cu2+ in the environment and live cells using a synthesized spider web-like fluorescent probe. Biosens. Bioelectron. 2021, 182, 113174. [Google Scholar] [CrossRef] [PubMed]
- Turro, N.J. Modern Molecular Photochemistry; University science books: Sausalito, CA, USA, 1991. [Google Scholar]
- Prodi, L.; Montalti, M.; Zaccheroni, N. Luminescence Applied in Sensor Science; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Bonacchi, S.; Genovese, D.; Juris, R.; Marzocchi, E.; Montalti, M.; Prodi, L.; Rampazzo, E.; Zaccheroni, N. Energy transfer in silica nanoparticles: An essential tool for the amplification of the fluorescence signal. In Reviews in Fluorescence 2008; Springer: Berlin/Heidelberg, Germany, 2010; pp. 119–137. [Google Scholar]
- Hajiaghababaei, L.; Mardvar, A.; Allahgholi Ghasri, M.R.; Dehghan Abkenar, S.; Badiei, A.; Ganjali, M.; Mohammadi Ziarani, G. Simultaneous Removal of Pb2+ and Cu2+ by SBA-15/di-urea as a Nano Adsorbent. Iran. J. Chem. Chem. Eng. 2022, 41, 163–173. [Google Scholar]
- Mohammadi Ziarani, G.; Moradi, R.; Mohajer, F.; Badiei, A. Synthesis of SBA-Pr-NHC as a selective fluorescent sensor for the detection of Ag+ ion in aqueous media. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 267, 120580. [Google Scholar] [CrossRef] [PubMed]
- Jamasbi, N.; Mohammadi Ziarani, G.; Mohajer, F.; Badiei, A. A new Hg2+ colorimetric chemosensor: The synthesis of chromeno[d]pyrimidine-2,5-dione/thione derivatives using Fe3O4@SiO2@(BuSO3H)3. Res. Chem. Intermed. 2022, 48, 899–909. [Google Scholar] [CrossRef]
- Mohammadi Ziarani, G.; Khademi, M.; Mohajer, F.; Badiei, A. The application of modified SBA-15 as a chemosensor. Curr. Nanomater. 2021, 7, 4–24. [Google Scholar] [CrossRef]
- Murray, K.; Cao, Y.-C.; Ali, S.; Hanley, Q. Lanthanide doped silica nanoparticles applied to multiplexed immunoassays. Analyst 2010, 135, 2132–2138. [Google Scholar] [CrossRef]
- Liu, W.; Ma, C.; Yang, H.; Zhang, Y.; Yan, M.; Ge, S.; Yu, J.; Song, X. Electrochemiluminescence immunoassay using a paper electrode incorporating porous silver and modified with mesoporous silica nanoparticles functionalized with blue-luminescent carbon dots. Microchim. Acta 2014, 181, 1415–1422. [Google Scholar] [CrossRef]
- Di Fusco, M.; Quintavalla, A.; Lombardo, M.; Guardigli, M.; Mirasoli, M.; Trombini, C.; Roda, A. Organically modified silica nanoparticles doped with new acridine-1, 2-dioxetane analogues as thermochemiluminescence reagentless labels for ultrasensitive immunoassays. Anal. Bioanal. Chem. 2015, 407, 1567–1576. [Google Scholar] [CrossRef]
- Wang, Z.; Zong, S.; Chen, H.; Wu, H.; Cui, Y. Silica coated gold nanoaggregates prepared by reverse microemulsion method: Dual mode probes for multiplex immunoassay using SERS and fluorescence. Talanta 2011, 86, 170–177. [Google Scholar] [CrossRef]
- Moon, J.H.; McDaniel, W.; Hancock, L.F. Facile fabrication of poly (p-phenylene ethynylene)/colloidal silica composite for nucleic acid detection. J. Colloid Interface Sci. 2006, 300, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Quan, Z.; Lu, L.; Huang, S.; Lin, J. Luminescence functionalization of mesoporous silica with different morphologies and applications as drug delivery systems. Biomaterials 2008, 29, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Gai, S.; Lin, J. Functionalized mesoporous silica materials for controlled drug delivery. Chem. Soc. Rev. 2012, 41, 3679–3698. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Li, C.; Ma, P.; Li, G.; Cheng, Z.; Peng, C.; Yang, D.; Yang, P.; Lin, J. Electrospinning Preparation and Drug-Delivery Properties of an Up-conversion Luminescent Porous NaYF4: Yb3+, Er3+@ Silica Fiber Nanocomposite. Adv. Funct. Mater. 2011, 21, 2356–2365. [Google Scholar] [CrossRef]
- Su, C.; Li, Z.; Zhang, D.; Wang, Z.; Zhou, X.; Liao, L.; Xiao, X. A highly sensitive sensor based on a computer-designed magnetic molecularly imprinted membrane for the determination of acetaminophen. Biosens. Bioelectron. 2020, 148, 111819. [Google Scholar] [CrossRef]
- Ow, H.; Larson, D.R.; Srivastava, M.; Baird, B.A.; Webb, W.W.; Wiesner, U. Bright and stable core− shell fluorescent silica nanoparticles. Nano Lett. 2005, 5, 113–117. [Google Scholar] [CrossRef]
- Karimi, M.; Badiei, A.; Mohammadi Ziarani, G. Fluorescence-enhanced optical sensor for detection of Al3+ in water based on functionalised nanoporous silica type SBA-15. Chem. Pap. 2016, 70, 1431–1438. [Google Scholar] [CrossRef]
- Ahmadi, T.; Bahar, S.; Mohammadi Ziarani, G.; Badiei, A. Formation of functionalized silica-based nanoparticles and their application for extraction and determination of Hg (II) ion in fish samples. Food Chem. 2019, 300, 125180. [Google Scholar] [CrossRef]
- Jin, Y.; Kannan, S.; Wu, M.; Zhao, J.X. Toxicity of Luminescent Silica Nanoparticles to Living Cells. Chem. Res. Toxicol. 2007, 20, 1126–1133. [Google Scholar] [CrossRef]
- Zu, G.; Shen, J.; Wang, W.; Zou, L.; Lian, Y.; Zhang, Z.; Interfaces. Silica–titania composite aerogel photocatalysts by chemical liquid deposition of titania onto nanoporous silica scaffolds. ACS Appl. Mater. Interfaces 2015, 7, 5400–5409. [Google Scholar] [CrossRef]
- Carn, F.; Colin, A.; Achard, M.F.; Deleuze, H.; Saadi, Z.; Backov, R. Rational design of macrocellular silica scaffolds obtained by a tunable sol–gel foaming process. J. Adv. Mater. 2004, 16, 140–144. [Google Scholar] [CrossRef]
- Shin, J.H.; Schoenfisch, M.H. Inorganic/organic hybrid silica nanoparticles as a nitric oxide delivery scaffold. Chem. Mater. 2008, 20, 239–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burns, A.; Sengupta, P.; Zedayko, T.; Baird, B.; Wiesner, U. Core/shell fluorescent silica nanoparticles for chemical sensing: Towards single-particle laboratories. Nanomicro. Lett. 2006, 2, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Jalageri, M.D.; Nagaraja, A.; Puttaiahgowda, Y.M. Piperazine based antimicrobial polymers: A review. RSC Adv. 2021, 11, 15213–15230. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Rajput, S.S.; Das, M.; Laha, S.; Choudhuri, I.; Bhattacharyya, N.; Das, A.; Samanta, B.C.; Alam, M.M.; Maity, T. Easy, selective and colorimetric detection of Zn (II), Cu (II), F− ions by a new piperazine based Schiff base chemosensor along with molecular logic gate formation and live cell images study. J. Photochem. Photobiol. 2022, 427, 113817. [Google Scholar] [CrossRef]
- Goswami, S.; Maity, S.; Maity, A.C.; Das, A.K.; Khanra, K.; Mandal, T.K.; Bhattacharyya, N. A macrocyclic piperazine linked extremely Zn2+ selective fluorescent chemosensor with bio-imaging and for H2PO4− sensing. Tetrahedron Lett. 2014, 55, 5993–5997. [Google Scholar] [CrossRef]
- Melde, B.J.; Johnson, B.J. Mesoporous materials in sensing: Morphology and functionality at the meso-interface. Anal. Bioanal. Chem. 2010, 398, 1565–1573. [Google Scholar] [CrossRef]
- Martín Várguez, P.E.; Raimundo, J.-M. Naked-Eye Chromogenic Test Strip for Cyanide Sensing Based on Novel Phenothiazine Push– Pull Derivatives. Biosensors 2022, 12, 407. [Google Scholar]
- Shi, Q.; Wu, S.T.; Shen, L.; Zhou, T.; Xu, H.; Wang, Z.Y.; Yang, X.J.; Huang, Y.L.; Zhang, Q.L. A Turn-On Fluorescent Chemosensor for Cyanide Ion Detection in Real Water Samples. Front. Chem. 2022, 10, 923149. [Google Scholar] [CrossRef]
- Mousavi, Z.; Ghasemi, J.B.; Mohammadi Ziarani, G.; Saidi, M.; Badiei, A. Dihydropyrano quinoline derivatives functionalized nanoporous silica as novel fluorescence sensor for Fe3+ in aqueous solutions(aq). J. Mol. Struct. 2022, 1265, 133408. [Google Scholar] [CrossRef]
- Gholamzadeh, P.; Mohammadi Ziarani, G.; Zandi, F.; Abolhasani Soorki, A.; Badiei, A.; Yazdian, F. Modification of fumed silica surface with different sulfonamides via a postsynthesis method and their application as antibacterial agents. Comptes Rendus Chim. 2017, 20, 833–840. [Google Scholar] [CrossRef]
- Afshani, J.; Badiei, A.; Lashgari, N.; Mohammadi Ziarani, G. A simple nanoporous silica-based dual mode optical sensor for detection of multiple analytes (Fe3+, Al3+ and CN−) in water mimicking XOR logic gate. RSC Adv. 2016, 6, 5957–5964. [Google Scholar] [CrossRef]
- Karimi, M.; Badiei, A.; Mohammadi Ziarani, G. A click-derived dual organic-inorganic hybrid optical sensor based on SBA-15 for selective recognition of Zn2+ and CN− in water. Inorg. Chim. Acta 2016, 450, 346–352. [Google Scholar] [CrossRef]





| Sensors | Solvent System | Sensing Ions | LOD | Reference |
|---|---|---|---|---|
| SSA1 | H2O | CN- | 2 × 10−4 M | [46] |
| SBA-II | H2O | CN- | 3.9 × 10−4 M | [47] |
| Fumed-Pr-Pi-TCT | H2O | CN- | 0.82 × 10−4 | Present work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afshar, S.S.; Ziarani, G.M.; Mohajer, F.; Badiei, A.; Iravani, S.; Varma, R.S. Synthesis of Fumed-Pr-Pi-TCT as a Fluorescent Chemosensor for the Detection of Cyanide Ions in Aqueous Media. Water 2022, 14, 4137. https://doi.org/10.3390/w14244137
Afshar SS, Ziarani GM, Mohajer F, Badiei A, Iravani S, Varma RS. Synthesis of Fumed-Pr-Pi-TCT as a Fluorescent Chemosensor for the Detection of Cyanide Ions in Aqueous Media. Water. 2022; 14(24):4137. https://doi.org/10.3390/w14244137
Chicago/Turabian StyleAfshar, Sepideh Saberi, Ghodsi Mohammadi Ziarani, Fatemeh Mohajer, Alireza Badiei, Siavash Iravani, and Rajender S. Varma. 2022. "Synthesis of Fumed-Pr-Pi-TCT as a Fluorescent Chemosensor for the Detection of Cyanide Ions in Aqueous Media" Water 14, no. 24: 4137. https://doi.org/10.3390/w14244137








