Uptake of Microplastics in the Wedge Clam Donax trunculus: First Evidence from the Mediterranean Sea
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sample Collection
2.2.1. Wedge Clams
2.2.2. Water
2.3. Analysis of Microplastics
2.4. Quality Control
2.5. Statistical Analysis
3. Results
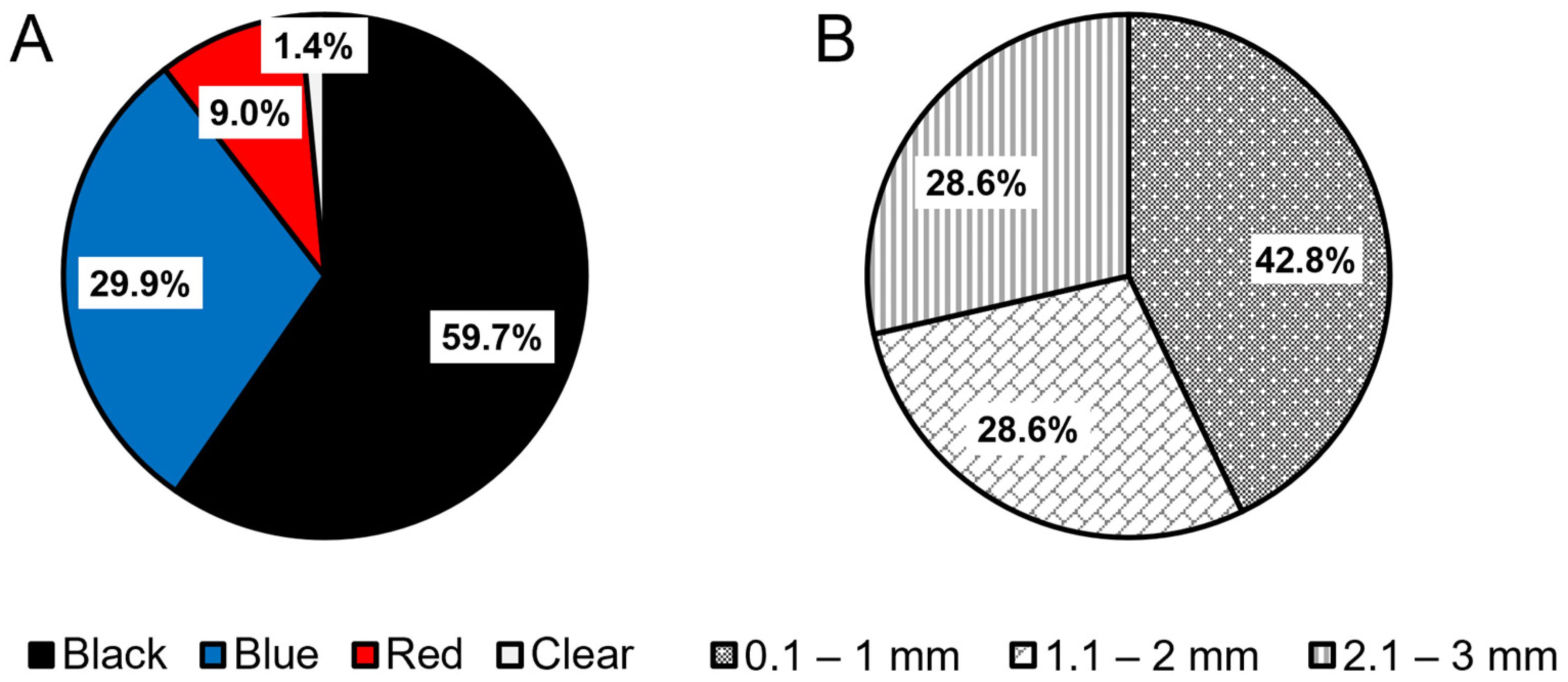
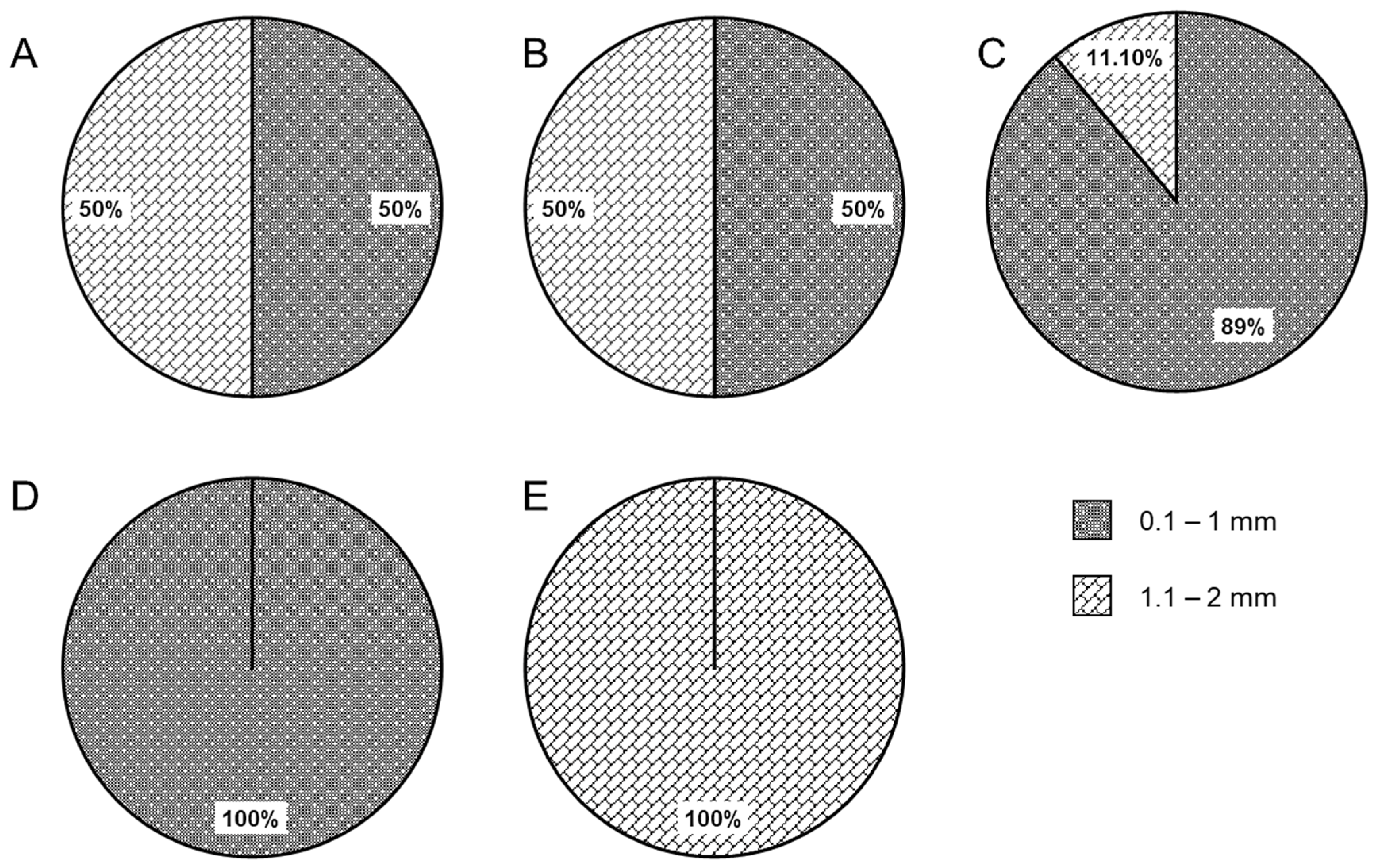
3.1. Microplastics in Wedge Clams
3.2. Microplastics in Water
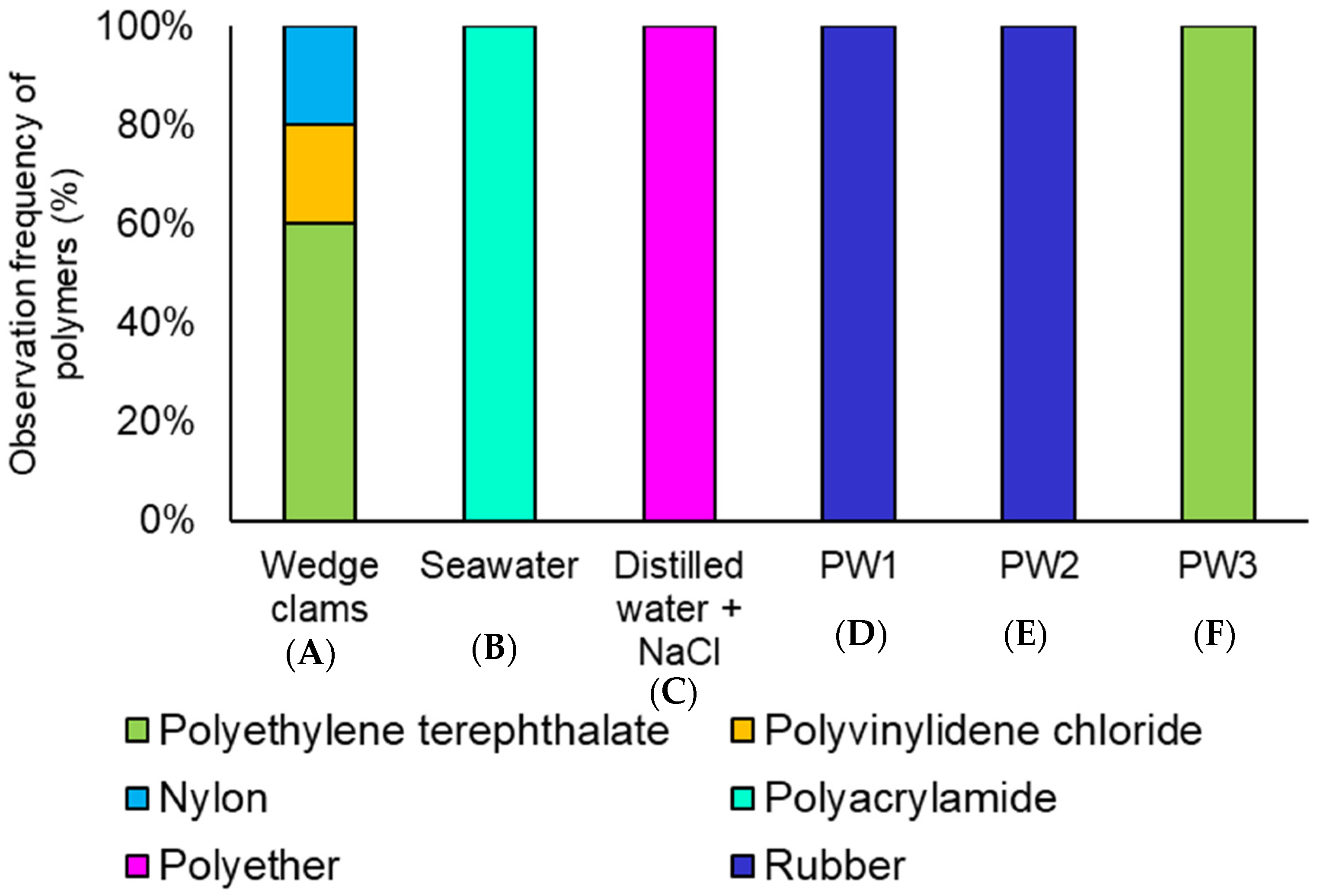
3.3. Micro-FTIR Characterization
3.4. Comparison of Studies about Microplastics in Wedge Clams
4. Discussion
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Corrales, X.; Coll, M.; Ofir, E.; Heymans, J.J.; Steenbeek, J.; Goren, M.; Edelist, D.; Gal, G. Future scenarios of marine resources and ecosystem conditions in the Eastern Mediterranean under the impacts of fishing, alien species and sea warming. Sci. Rep. 2018, 8, 14284. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R. Climate change impacts on the biota and on vulnerable habitats of the deep Mediterranean Sea. Rend. Lincei Sci. Fis. Nat. 2018, 29, 525–541. [Google Scholar] [CrossRef]
- Consoli, P.; Romeo, T.; Angiolillo, M.; Canese, S.; Esposito, V.; Salvati, E.; Scotti, G.; Andaloro, F.; Tunesi, L. Marine litter from fishery activities in the Western Mediterranean sea: The impact of entanglement on marine animal forests. Environ. Pollut. 2019, 249, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Sharma, V.; Chatterjee, S. Microplastics in the Mediterranean Sea: Sources, pollution intensity, sea health, and regulatory policies. Front. Mar. Sci. 2021, 8, 634934. [Google Scholar] [CrossRef]
- Cesarini, G.; Secco, S.; Battisti, C.; Questino, B.; Marcello, L.; Scalici, M. Temporal changes of plastic litter and associated encrusting biota: Evidence from Central Italy (Mediterranean Sea). Mar. Pollut. Bull. 2022, 181, 113890. [Google Scholar] [CrossRef] [PubMed]
- Derraik, J.G. The pollution of the marine environment by plastic debris: A review. Mar. Pollut. Bull. 2022, 44, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Fossi, M.C.; Pedà, C.; Compa, M.; Tsangaris, C.; Alomar, C.; Claro, F.; Ioakeimidis, C.; Galgani, F.; Hema, T.; Deudero, S.; et al. Bioindicators for monitoring marine litter ingestion and its impacts on Mediterranean biodiversity. Environ. Pollut. 2018, 237, 1023–1040. [Google Scholar] [CrossRef] [PubMed]
- Anastasopoulou, A.; Fortibuoni, T. Impact of plastic pollution on marine life in the Mediterranean Sea. In Plastics in the Aquatic Environment-Part I; Springer: Cham, Switzerland, 2019; pp. 135–196. [Google Scholar]
- Compa, M.; Alomar, C.; Wilcox, C.; van Sebille, E.; Lebreton, L.; Hardesty, B.D.; Deudero, S. Risk assessment of plastic pollution on marine diversity in the Mediterranean Sea. Sci. Total. Environ. 2019, 678, 188–196. [Google Scholar] [CrossRef]
- Soto-Navarro, J.; Jordá, G.; Compa, M.; Alomar, C.; Fossi, M.C.; Deudero, S. Impact of the marine litter pollution on the Mediterranean biodiversity: A risk assessment study with focus on the marine protected areas. Mar. Pollut. Bull. 2021, 165, 112169. [Google Scholar] [CrossRef]
- PlasticsEurope. Plastic-the Facts 2020. In An Analysis of European Plastics Production, Demand and Waste Data; PlasticsEurope: Brussels, Belgium, 2020; Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts-2020/ (accessed on 24 November 2022).
- Cho, Y.; Shim, W.J.; Jang, M.; Han, G.M.; Hong, S.H. Abundance and characteristics of microplastics in market bivalves from South Korea. Environ. Pollut. 2019, 245, 1107–1116. [Google Scholar] [CrossRef]
- Windsor, F.; Durance, I.; Horton, A.A.; Thompson, R.C.; Tyler, C.R.; Ormerod, S.J. A catchment-scale perspective of plastic pollution. Glob. Chang. Biol. 2019, 25, 1207–1221. [Google Scholar] [CrossRef] [PubMed]
- Cera, A.; Cesarini, G.; Scalici, M. Microplastics in freshwater: What is the news from the world? Diversity 2020, 12, 276. [Google Scholar] [CrossRef]
- Iroegbu, A.O.C.; Ray, S.S.; Mbarane, V.; Bordado, J.C.; Sardinha, J.P. Plastic pollution: A perspective on matters arising: Challenges and opportunities. ACS Omega 2021, 6, 19343–19355. [Google Scholar] [CrossRef] [PubMed]
- Karbalaei, S.; Hanachi, P.; Walker, T.R.; Cole, M. Occurrence, sources, human health impacts and mitigation of microplastic pollution. Environ. Sci. Pollut. Res. 2018, 25, 36046–36063. [Google Scholar] [CrossRef] [PubMed]
- Umar, K.; Yaqoob, A.A.; Ibrahim, M.N.M.; Parveen, T.; Safian, M.T. Environmental applications of smart polymer composites. In Smart Polymer Nanocomposites; Elsevier: Amsterdam, The Netherlands, 2020; Volume 15, pp. 295–320. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Guzzetti, E.; Sureda, A.; Tejada, S.; Faggio, C. Microplastic in marine organism: Environmental and toxicological effects. Environ. Toxicol. Pharmacol. 2018, 64, 164–171. [Google Scholar] [CrossRef]
- Biamis, C.; O’Driscoll, K.; Hardiman, G. Microplastic toxicity: A review of the role of marine sentinel species in assessing the environmental and public health impacts. Case Stud. Chem. Environ. Eng. 2021, 3, 100073. [Google Scholar] [CrossRef]
- Dar, M.A.; Dhole, N.P.; Pawar, K.D.; Xie, R.; Shahnawaz, M.; Pandit, R.S.; Sun, J. Ecotoxic Effects of the plastic waste on marine fauna: An overview. In Impact of Plastic Waste on the Marine Biota; Shahnawaz, M., Sangale, M.K., Daochen, Z., Ade, A.B., Eds.; Springer: Singapore, 2022; pp. 287–300. [Google Scholar] [CrossRef]
- Safian, M.T.-U.; Umar, K.; Parveen, T.; Yaqoob, A.A.; Ibrahim, M.N.M. Biomedical applications of smart polymer composites. In Smart Polymer Nanocomposites; Elsevier: Amsterdam, The Netherlands, 2021; pp. 183–204. [Google Scholar]
- Miller, M.E.; Hamann, M.; Kroon, F.J. Bioaccumulation and biomagnification of microplastics in marine organisms: A review and meta-analysis of current data. PLoS ONE 2020, 15, e0240792. [Google Scholar] [CrossRef]
- Renzi, M.; Guerranti, C.; Blašković, A. Microplastic contents from maricultured and natural mussels. Mar. Pollut. Bull. 2018, 131, 248–251. [Google Scholar] [CrossRef]
- Ribeiro, F.; Garcia, A.R.; Pereira, B.P.; Fonseca, M.; Mestre, N.C.; Fonseca, T.G.; Ilharco, L.M.; Bebianno, M.J. Microplastics effects in Scrobicularia plana. Mar. Pollut. Bull. 2017, 122, 379–391. [Google Scholar] [CrossRef]
- Li, J.; Qu, X.; Su, L.; Zhang, W.; Yang, D.; Kolandhasamy, P.; Li, D.; Shi, H. Microplastics in mussels along the coastal waters of China. Environ. Pollut. 2016, 214, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Revel, M.; Lagarde, F.; Perrein-Ettajani, H.; Bruneau, M.; Akcha, F.; Sussarellu, R.; Rouxel, J.; Costil, K.; Decottignies, P.; Cognie, B. Tissue-specific biomarker responses in the blue mussel Mytilus spp. exposed to a mixture of microplastics at environmentally relevant concentrations. Front. Environ. Sci. 2019, 7, 33. [Google Scholar] [CrossRef]
- Sendra, M.; Sparaventi, E.; Novoa, B.; Figueras, A. An overview of the internalization and effects of microplastics and nanoplastics as pollutants of emerging concern in bivalves. Sci. Total. Environ. 2021, 753, 142024. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chang, X.; Hu, M.; Fang, J.K.-H.; Sokolova, I.M.; Huang, W.; Xu, E.G.; Wang, Y. Is microplastic an oxidative stressor? Evidence from a meta-analysis on bivalves. J. Hazard. Mater. 2022, 423, 127211. [Google Scholar] [CrossRef]
- Wesch, C.; Bredimus, K.; Paulus, M.; Klein, R. Towards the suitable monitoring of ingestion of microplastics by marine biota: A review. Environ. Pollut. 2016, 218, 1200–1208. [Google Scholar] [CrossRef] [PubMed]
- Bonanno, G.; Orlando-Bonaca, M. Perspective on using marine species as bioindicators of plastic pollution. Mar. Pollut. Bull. 2018, 137, 209–221. [Google Scholar] [CrossRef]
- Li, J.; Lusher, A.; Rotchell, J.M.; Company, S.D.; Turra, A.; Brate, I.L.N.; Sun, C.; Hossain, M.S.; Li, Q.; Kolandhasamy, P.; et al. Using mussel as a global bioindicator of coastal microplastic pollution. Environ. Pollut. 2018, 244, 522–533. [Google Scholar] [CrossRef]
- Wright, S.L.; Kelly, F.J. Plastic and human health: A micro issue? Environ. Sci. Technol. 2017, 51, 6634–6647. [Google Scholar] [CrossRef]
- Zhang, F.; Man, Y.B.; Mo, W.Y.; Man, K.Y.; Wong, M.H. Direct and indirect effects of microplastics in bivalves, with a focus on edible species: A mini-review. Environ. Sci. Technol. 2019, 50, 2109–2143. [Google Scholar] [CrossRef]
- Brown, A.C.; McLachlan, A. Ecology of Sandy Shores; Elsevier: Amsterdam, The Netherlands, 1990; 328p. [Google Scholar]
- Bayed, A.; Guillou, J. Contribution à l’étude des Populations du Genre Donax: La Population de D. trunculus L. (Mollusca, Bivalvia) de Mehdia (Maroc); Annales de l’Institut Océanographique: Paris, France, 1985; pp. 139–147. Available online: https://api.semanticscholar.org/CorpusID:131834201 (accessed on 24 November 2022).
- Tlili, S.; Mouneyrac, C. The wedge clam Donax trunculus as sentinel organism for Mediterranean coastal monitoring in a global change context. Reg. Environ. Change 2019, 19, 995–1007. [Google Scholar] [CrossRef]
- Suepbala, W.; Yeemin, T.; Sutthacheep, M.; Sangiamdee, D.; Phaoduang, S.; Sangsawang, L.; Rangseethampanya, P.; Wongnutpranont, A. Comparing the abundance of microplastics in the wedge shell, Donax semigranosus, from Ta Kuan and Leam Mea Phim beaches, Rayong Province. Ramkhamhaeng Int. J. Sci. Technol. 2018, 1, 25–32. [Google Scholar]
- Corami, F.; Rosso, B.; Roman, M.; Picone, M.; Gambaro, A.; Barbante, C. Evidence of small microplastics (<100 μm) ingestion by Pacific oysters (Crassostrea gigas): A novel method of extraction, purification, and analysis using Micro-FTIR. Mar. Pollut. Bull. 2020, 160, 111606. [Google Scholar] [PubMed]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Ben-Haddad, M.; Abelouah, M.R.; Hajji, S.; De-La-Torre, G.E.; Oualid, H.A.; Rangel-Buitrago, N.; Alla, A.A. The wedge clam Donax trunculus L., 1758 as a bioindicator of microplastic pollution. Mar. Pollut. Bull. 2022, 178, 113607. [Google Scholar] [CrossRef] [PubMed]
- Alexandrova, A.V.; Ignatova-Ivanova, T.V.; Bachvarova, D.G.; Toschkova, S.G.; Doichinov, A.H.; Ibryamova, S.F.; Chipev, N.H. Pilot Screening and Assessment of Microplastic Bioaccumulation in Wedge Clams Donax trunculus Linnaeus, 1758 (Bivalvia) from the Bulgarian Black Sea Coast. Acta Zool. BulG. 2022, in press. Available online: http://www.acta-zoologica-bulgarica.eu/2022/002641 (accessed on 24 November 2022).
- Cho, Y.; Shim, W.J.; Jang, M.; Han, G.M.; Hong, S.H. Nationwide monitoring of microplastics in bivalves from the coastal environment of Korea. Environ. Pollut. 2021, 270, 116175. [Google Scholar] [CrossRef]
- Phuong, N.N.; Poirier, L.; Pham, Q.T.; Lagarde, F.; Zalouk-Vergnoux, A. Factors influencing the microplastic contamination of bivalves from the French Atlantic coast: Location, season and/or mode of life? Mar. Pollut. Bull. 2018, 129, 664–674. [Google Scholar] [CrossRef]
- De Witte, B.; Devriese, L.; Bekaert, K.; Hoffman, S.; Vandermeersch, G.; Cooreman, K.; Robbens, J. Quality assessment of the blue mussel (Mytilus edulis): Comparison between commercial and wild types. Mar. Pollut. Bull. 2014, 85, 146–155. [Google Scholar] [CrossRef]
- Naidu, S.A. Preliminary study and first evidence of presence of microplastics and colorants in green mussel, Perna viridis (Linnaeus, 1758), from southeast coast of India. Mar. Pollut. Bull. 2019, 140, 416–422. [Google Scholar]
- Ding, J.; Sun, C.; He, C.; Li, J.; Ju, P.; Li, F. Microplastics in four bivalve species and basis for using bivalves as bioindicators of microplastic pollution. Sci. Total. Environ. 2021, 782, 146830. [Google Scholar] [CrossRef]
- Truchet, D.M.; Lopez, A.D.F.; Ardusso, M.G.; Rimondino, G.N.; Buzzi, N.S.; Malanca, F.E.; Spetter, C.V.; Severini, M.D.F. Microplastics in bivalves, water and sediments from a touristic sandy beach of Argentina. Mar. Pollut. Bull. 2021, 173, 113023. [Google Scholar] [CrossRef] [PubMed]
- Erni-Cassola, G.; Gibson, M.I.; Thompson, R.C.; Christie-Oleza, J.A. Lost, but found with Nile red: A novel methos for detecting and quantifying small microplastics (1 mm to 20 mm) in environmental samples. Environ. Sci. Technol. 2017, 51, 13641–13648. [Google Scholar] [CrossRef] [PubMed]
- Jâms, I.B.; Windsor, F.M.; Poudevigne-Durance, T.; Ormerod, S.J.; Durance, I. Estimating the size distribution of plastics ingested by animals. Nat. Commun. 2020, 11, 1594. [Google Scholar] [CrossRef] [PubMed]
- Santana, M.F.M.; Ascer, L.G.; Custódio, M.R.; Moreira, F.T.; Turra, A. Microplastic contamination in natural mussel beds from a Brazilian urbanized coastal region: Rapid evaluation through bioassessment. Mar. Pollut. Bull. 2016, 106, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Covernton, G.A.; Collicutt, B.; Gurney-Smith, H.J.; Pearce, C.M.; Dower, J.F.; Ross, P.S.; Dudas, S.E. Microplastics in bivalves and their habitat in relation to shellfish aquaculture proximity in coastal British Columbia, Canada. Aquac. Environ. Interact. 2019, 11, 357–374. [Google Scholar] [CrossRef]
- Rios-Fuster, B.; Alomar, C.; Compa, M.; Guijarro, B.; Deudero, S. Antropogenic particles ingestion in fish species from two areas of the western Mediterranean Sea. Mar. Pollut. Bull. 2019, 144, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Savoca, S.; Capillo, G.; Mancuso, M.; Faggio, C.; Panarello, G.; Crupi, R.; Bonsignore, M.; D’Urso, L.; Compagnini, G.; Neri, F.; et al. Detection of artificial cellulose microfibers in Boops boops from the northern coasts of Sicily (Central Mediterranean). Sci. Total Environ. 2019, 691, 455–465. [Google Scholar] [CrossRef]
- Tsangaris, C.; Digka, N.; Valente, T.; Aguilar, A.; Borrell, A.; de Lucia, G.A.; Gambaiani, D.; Garcia-Garin, O.; Kaberi, H.; Martin, J.; et al. Using Boops boops (osteichthyes) to assess microplastic ingestion of the Mediterranean Sea. Mar. Pollut. Bull. 2020, 158, 111397. [Google Scholar] [CrossRef]
- Ory, N.C.; Sobral, P.; Ferreira, J.L.; Thiel, M. Amberstripe scad Decapterus muroadsi (Carangidae) fish ingest blue microplastics resembling their copepod prey along the coast of Rapa Nui (Easter Island) in the South Pacific subtropical gyre. Sci. Total. Environ. 2017, 586, 430–437. [Google Scholar] [CrossRef]
- de Haan, W.P.; Sanchez-Vidal, A.; Canals, M. Floating microplastics and aggregate formation in the Western Mediterranean Sea. Mar. Pollut. Bull. 2019, 140, 523–535. [Google Scholar] [CrossRef]
- Amira, A.; Sifim, K.; Soltani, N. Measure of environmental stress biomarkers in Donax trunculus (Mollusca, Bivalvia) from the gulf of Annaba (Algeria). Eur. J. Exp. Biol. 2011, 2, 7–16. [Google Scholar]
- Sifi, K.; Amira, A.; Soltani, N. Oxidative stress and biochemical composition in Donax trunculus (Mollusca, Bivalvia) from the gulf of Annaba (Algeria). Adv. Environ. Biol. 2013, 7, 595–604. [Google Scholar]
- Nisticò, R. Polyethylene terephthalate (PET) in the packaging industry. Polym. Test. 2020, 90, 106707. [Google Scholar] [CrossRef]
- Napper, I.E.; Thompson, R.C. Release of synthetic microplastic plastic fibres from domestic washing machines: Effects of fabric type and washing conditions. Mar. Pollut. Bull. 2016, 112, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Narmatha Sathish, M.; Immaculate Jeyasanta, K.; Patterson, J. Monitoring of microplastics in the clam Donax cuneatus and its habitat in Tuticorin coast of Gulf of Mannar (GoM), India. Environ. Pollut. 2020, 266, 115219. [Google Scholar] [CrossRef] [PubMed]
- Delgado, M.; Silva, L.; Gómez, S.; Masferrer, E.; Cojan, M.; Gaspar, M.B. Population and production parameters of the wedge clam Donax trunculus (Linnaeus, 1758) in intertidal areas on the southwest Spanish coast: Considerations in relation to protected areas. Fish. Res. 2017, 193, 232–241. [Google Scholar] [CrossRef]
- Xiong, B.; Loss, R.D.; Shields, D.; Pawlik, T.; Hochreiter, R.; Zydney, A.L.; Kumar, M. Polyacrylamide degradation and its implications in environmental systems. NPJ Clean Water 2018, 1, 17. [Google Scholar] [CrossRef]
- Browne, M.A.; Crump, P.; Niven, S.J.; Teuten, E.; Tonkin, A.; Galloway, T.; Thompson, R.C. Accumulation of microplastic on shoreline worldwide: Sources and sink. Environ. Sci. Technol. 2011, 45, 9175–9179. [Google Scholar] [CrossRef]
- Murphy, F.; Ewins, C.; Carbonnier, F.; Quinn, B. Wastewater Treatment Works (WwTW) as a source of microplastics in the aquatic environment. Environ. Sci. Technol. 2016, 50, 5800–5808. [Google Scholar] [CrossRef]
- Magni, S.; Gagné, F.; André, C.; Della Torre, C.; Auclair, J.; Hanana, H.; Parenti, C.C.; Bonasoro, F.; Binelli, A. Evaluation of uptake and chronic toxicity of virgin polystyrene microbeads in freshwater zebra mussel Dreissena polymorpha (Mollusca: Bivalvia). Sci. Total Environ. 2018, 631, 778–788. [Google Scholar] [CrossRef]
- Abidli, S.; Pinheiro, M.; Lahbib, Y.; Neuparth, T.; Santos, M.M.; Trigui El Menif, N. Effects of environmentally relevant levels of polyethylene microplastic on Mytilus galloprovincialis (Mollusca: Bivalvia): Filtration rate and oxidative stress. Environ. Sci. Pollut. Res. 2021, 28, 26643–26652. [Google Scholar] [CrossRef] [PubMed]
- Mkuye, R.; Gong, S.; Zhao, L.; Masanja, F.; Ndandala, C.; Bubelwa, E.; Yang, C.; Deng, Y. Effects of microplastics on physiological performance of marine bivalves, potential impacts, and enlightening the future based on a comparative study. Sci. Total Environ. 2022, 838, 155933. [Google Scholar] [CrossRef] [PubMed]
- Tlili, S.; Jemai, D.; Brinis, S.; Regaya, I. Microplastics mixture exposure at environmentally relevant conditions induce oxidative stress and neurotoxicity in the wedge clam Donax trunculus. Chemosphere 2020, 258, 127344. [Google Scholar] [CrossRef] [PubMed]
- Beiras, R.; Verdejo, E.; Campoy-Lopez, P.; Vidal-Linan, L. Aquatic toxicity of chemically defined microplastics can be explained by functional additives. J. Hazard. Mater. 2021, 406, 124338. [Google Scholar] [CrossRef] [PubMed]






| χ2/p | September | October | November | February | March | April | May |
|---|---|---|---|---|---|---|---|
| September | 0.7 | 0.0027 | 0.074 | 0.018 | 0.71 | 0.0027 | |
| October | 0.65 | 0.0075 | 0.16 | 0.045 | 0.45 | 0.0075 | |
| November | 0.000014 | 0.000041 | 0.16 | 0.41 | 0.00018 | 0.0019 | |
| February | 0.039 | 0.11 | 0.061 | 0.53 | 0.033 | 0.16 | |
| March | 0.003 | 0.013 | 0.27 | 0.44 | 0.007 | 0.41 | |
| April | 0.67 | 0.42 | 0.00000003 | 0.012 | 0.00042 | 0.012 | |
| May | 0.000014 | 0.000041 | 0.62 | 0.061 | 0.27 | 0.00019 |
| Reference | Study Area | MPs Abundance | Dominant Color | Dominant Shape | Dominant Size | Dominant Polymer |
|---|---|---|---|---|---|---|
| Present study | Tyrrhenian Coast (Mediterranean Sea) | 0–0.56 MPs/individual | Black Blue | Fiber | 0.1–1 mm | PET |
| [41] | Bulgarian Coast (Black Sea) | 0.31–4.46 MPs/individual | NA | Pellet | ≤25 µm | NA |
| [42] | Atlantic Coast of Morocco (Atlantic Ocean) | 1.75–5.93 g⋅ww−1 | Blue | Fiber | 0.1–0.5 mm 0.5–1 mm | PE |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olivieri, Z.; Cesarini, G.; Orsini, M.; De Santis, S.; Scalici, M. Uptake of Microplastics in the Wedge Clam Donax trunculus: First Evidence from the Mediterranean Sea. Water 2022, 14, 4095. https://doi.org/10.3390/w14244095
Olivieri Z, Cesarini G, Orsini M, De Santis S, Scalici M. Uptake of Microplastics in the Wedge Clam Donax trunculus: First Evidence from the Mediterranean Sea. Water. 2022; 14(24):4095. https://doi.org/10.3390/w14244095
Chicago/Turabian StyleOlivieri, Zoe, Giulia Cesarini, Monica Orsini, Serena De Santis, and Massimiliano Scalici. 2022. "Uptake of Microplastics in the Wedge Clam Donax trunculus: First Evidence from the Mediterranean Sea" Water 14, no. 24: 4095. https://doi.org/10.3390/w14244095
APA StyleOlivieri, Z., Cesarini, G., Orsini, M., De Santis, S., & Scalici, M. (2022). Uptake of Microplastics in the Wedge Clam Donax trunculus: First Evidence from the Mediterranean Sea. Water, 14(24), 4095. https://doi.org/10.3390/w14244095









