Contaminant Removal from Wastewater by Microalgal Photobioreactors and Modeling by Artificial Neural Network
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Proparation of Synthetic Wastewater
2.2. Photobioreactors
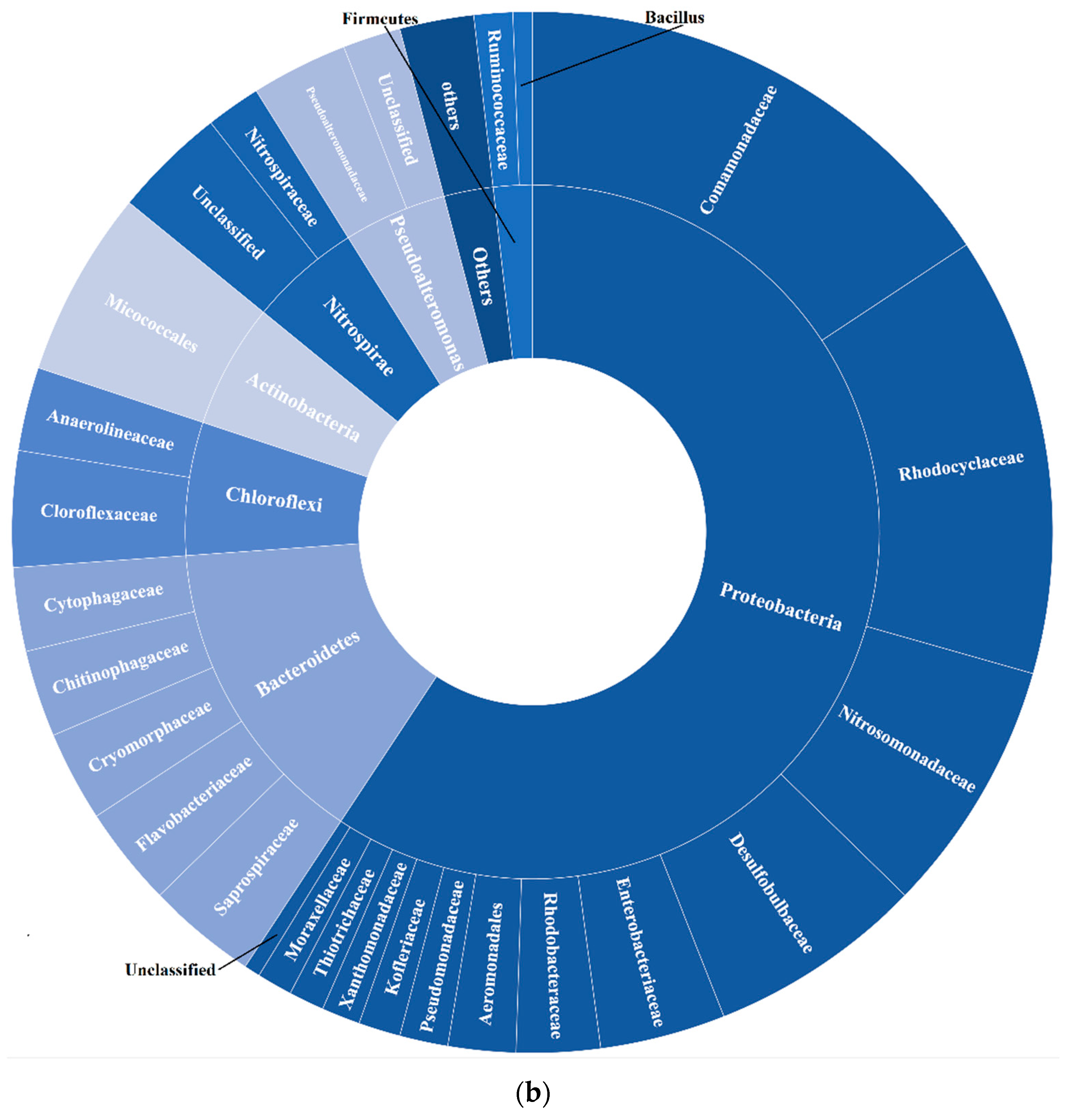
2.3. DNA Extraction and Microbial Community in Activated Sludge
2.4. Photolysis of Contaminants
2.5. Analyses of Contaminant Concentrations in Water
2.6. Optimization of Photobioreactors by Artificial Neural Network
2.7. Effects of CAF and DEET on Chlorella vulgaris
3. Results
3.1. Photodegradation of PPCPs
3.2. Contaminant Removal in Photobioreactors
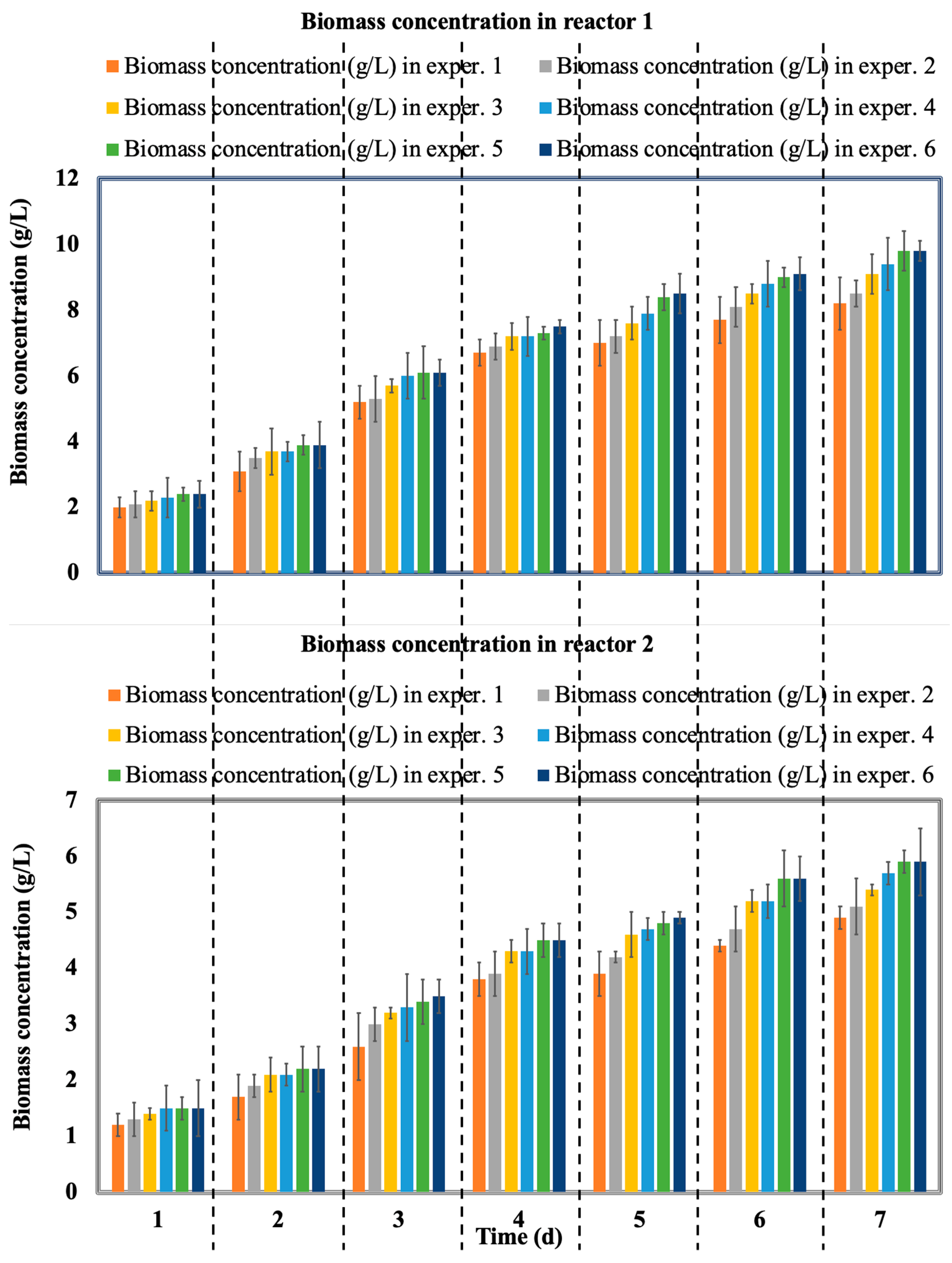
3.3. Evolution of Microbial Biomass
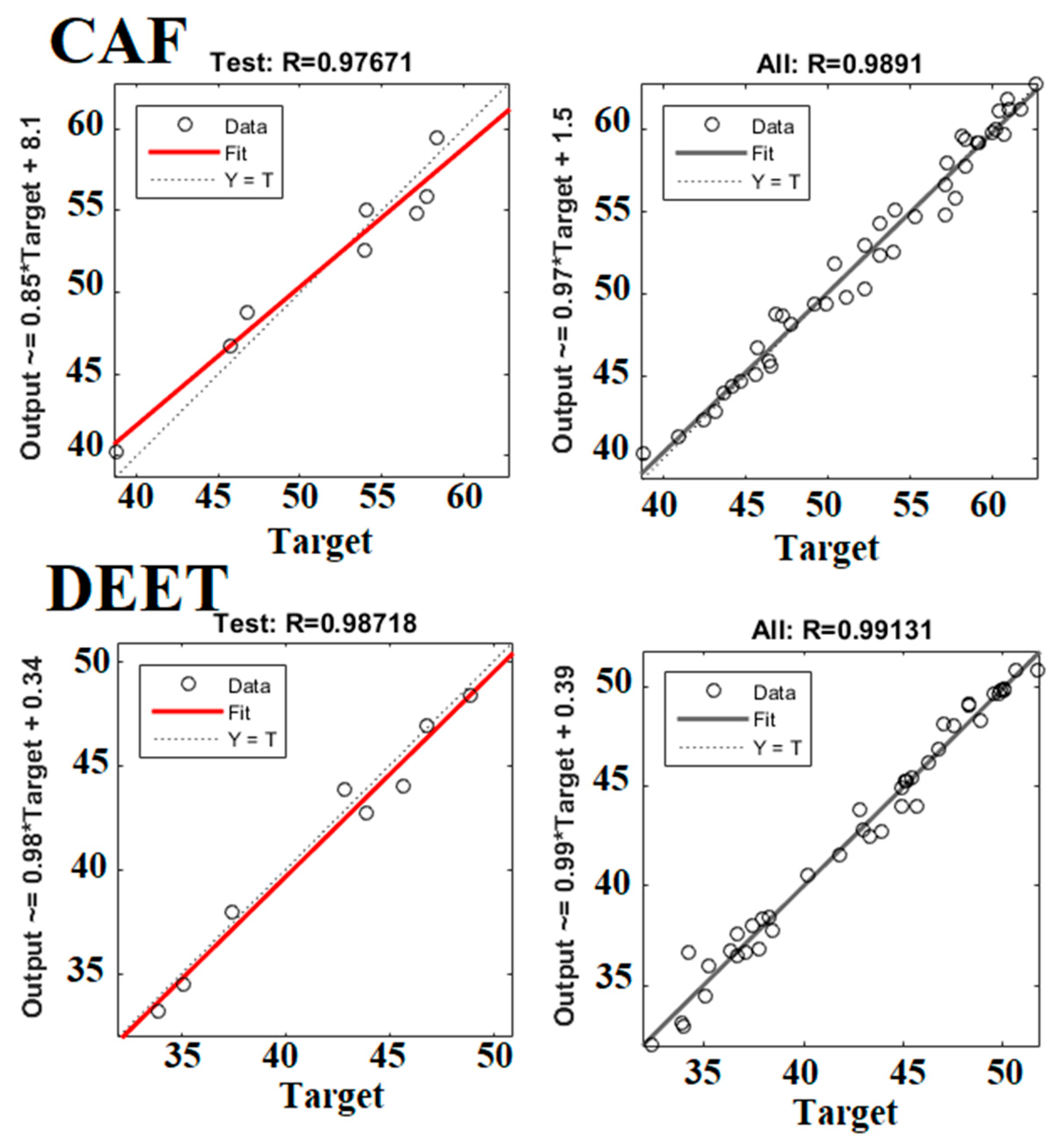
3.4. Optimization of Removal Performance by the ANN
3.5. Effects of Emerging Contaminants on Microalgae
4. Conclusions
- The biological consortium in photobioreactor 1 removed a maximum of 82.3% TAN and 67.7% COD, higher than 62.2% TAN and 52.8% COD by microalgae alone.
- The consortium achieved a maximum removal of 85.7% for CAF and 73.3% for DEET, which was higher than 62.7% and 51.8% by microalgae alone.
- ANN was able to optimize photobioreactor performance, as demonstrated by the high R2 (>0.99) and low MSE (<0.1) values.
- High concentrations of PPCPs and long contact time reduced the content of chlorophyll and protein in microalgae.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kargol, A.K.; Cao, C.; James, C.A.; Gough, H.L. Wastewater reuse for tree irrigation: Influence on rhizosphere microbial communities. Resour. Environ. Sustain. 2022, 9, 100063. [Google Scholar] [CrossRef]
- Kassahun, S.K.; Kiflie, Z.; Kim, H.; Baye, A.F. Process optimization and kinetics analysis for photocatalytic degradation of emerging contaminant using N-doped TiO2-SiO2 nanoparticle: Artificial Neural Network and Surface Response Methodology approach. Environ. Technol. Innov. 2021, 23, 101761. [Google Scholar] [CrossRef]
- Zhao, H.; Zhou, J.L.; Zhang, J. Tidal impact on the dynamic behavior of dissolved pharmaceuticals in the Yangtze Estuary, China. Sci. Total Environ. 2015, 536, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Mojiri, A.; Baharlooeian, M.; Zahed, M.A. The Potential of Chaetoceros muelleri in Bioremediation of Antibiotics: Performance and Optimization. Int. J. Environ. Res. Public Health 2021, 18, 977. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.-R.; Fan, X.-Y.; Li, X.; Gao, Y.-X.; Zhang, Z.-X. Impact of ciprofloxacin and copper combined pollution on activated sludge: Abundant-rare taxa and antibiotic resistance genes. Bioresour. Technol. 2022, 349, 126882. [Google Scholar] [CrossRef]
- Mojiri, A.; Zhou, J.L.; Ratnaweera, H.; Rezania, S.; Nazari, V.M. Pharmaceuticals and personal care products in aquatic environments and their removal by algae-based systems. Chemosphere 2022, 288, 132580. [Google Scholar] [CrossRef]
- Tran, N.H.; Hu, J.; Urase, T. Removal of the insect repellent N,N-diethyl-m-toluamide (DEET) by laccase-mediated systems. Bioresour. Technol. 2013, 147, 667–671. [Google Scholar] [CrossRef]
- Bachmann, S.A.L.; Calvete, T.; Féris, L.A. Caffeine removal from aqueous media by adsorption: An overview of adsorbents evolution and the kinetic, equilibrium and thermodynamic studies. Sci. Total Environ. 2021, 767, 144229. [Google Scholar] [CrossRef]
- García, J.; García-Galán, M.J.; Day, J.W.; Boopathy, R.; White, J.R.; Wallace, S.; Hunter, R.G. A review of emerging organic contaminants (EOCs), antibiotic resistant bacteria (ARB), and antibiotic resistance genes (ARGs) in the environment: Increasing removal with wetlands and reducing environmental impacts. Bioresour. Technol. 2020, 307, 123228. [Google Scholar] [CrossRef]
- Al Qarni, H.; Collier, P.; O’Keeffe, J.; Akunna, J. Investigating the removal of some pharmaceutical compounds in hospital wastewater treatment plants operating in Saudi Arabia. Environ. Sci. Pollut. Res. 2016, 23, 13003–13014. [Google Scholar] [CrossRef]
- Leng, L.; Li, W.; Chen, J.; Leng, S.; Chen, J.; Wei, L.; Peng, H.; Li, J.; Zhou, W.; Huang, H. Co-culture of fungi-microalgae consortium for wastewater treatment: A review. Bioresour. Technol. 2021, 330, 125008. [Google Scholar] [CrossRef] [PubMed]
- Marchão, L.; Fernandes, J.R.; Sampaio, A.; Peres, J.A.; Tavares, P.B.; Lucas, M.S. Microalgae and immobilized TiO2/UV-A LEDs as a sustainable alternative for winery wastewater treatment. Water Res. 2021, 203, 117464. [Google Scholar] [CrossRef]
- Terán Hilares, R.; Garcia Bustos, K.A.; Sanchez Vera, F.P.; Colina Andrade, G.J.; Pacheco Tanaka, D.A. Acid precipitation followed by microalgae (Chlorella vulgaris) cultivation as a new approach for poultry slaughterhouse wastewater treatment. Bioresour. Technol. 2021, 335, 125284. [Google Scholar] [CrossRef] [PubMed]
- Soleimanikhorramdashti, M.; Samipoorgiri, M.; Majidian, N. Extraction lipids from chlorella vulgaris by supercritical CO2 for biodiesel production. S. Afr. J. Chem. Eng. 2021, 38, 121–131. [Google Scholar] [CrossRef]
- Romdhane, L.; Panozzo, A.; Radhouane, L.; Dal Cortivo, C.; Barion, G.; Vamerali, T. Root Characteristics and Metal Uptake of Maize (Zea mays L.) under Extreme Soil Contamination. Agronomy 2021, 11, 178. [Google Scholar] [CrossRef]
- Rossi, S.; Díez-Montero, R.; Rueda, E.; Castillo Cascino, F.; Parati, K.; García, J.; Ficara, E. Free ammonia inhibition in microalgae and cyanobacteria grown in wastewaters: Photo-respirometric evaluation and modelling. Bioresour. Technol. 2020, 305, 123046. [Google Scholar] [CrossRef] [PubMed]
- Shahsavari, E.; Rouch, D.; Khudur, L.S.; Thomas, D.; Aburto-Medina, A.; Ball, A.S. Challenges and Current Status of the Biological Treatment of PFAS-Contaminated Soils. Front. Bioeng. Biotechnol. 2021, 8, 602040. [Google Scholar] [CrossRef]
- Ebrahimzade, I.; Ebrahimi-Nik, M.; Rohani, A.; Tedesco, S. Towards monitoring biodegradation of starch-based bioplastic in anaerobic condition: Finding a proper kinetic model. Bioresour. Technol. 2022, 347, 126661. [Google Scholar] [CrossRef]
- Makut, B.B.; Das, D.; Goswami, G. Production of microbial biomass feedstock via co-cultivation of microalgae-bacteria consortium coupled with effective wastewater treatment: A sustainable approach. Algal Res. 2019, 37, 228–239. [Google Scholar] [CrossRef]
- Yang, J.; Gou, Y.; Fang, F.; Guo, J.; Lu, L.; Zhou, Y.; Ma, H. Potential of wastewater treatment using a concentrated and suspended algal-bacterial consortium in a photo membrane bioreactor. Chem. Eng. J. 2018, 335, 154–160. [Google Scholar] [CrossRef]
- Cao, J.; Zhu, Q.; Zhang, T.; Wu, Y.; Zhang, Q.; Fu, B.; Fang, F.; Feng, Q.; Luo, J. Distribution patterns of microbial community and functional characteristics in full-scale wastewater treatment plants: Focusing on the influent types. Chemosphere 2021, 281, 130899. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Chen, Y.; Zhang, T.C.; Ji, B.; Cao, L. Performance of Chlorella sorokiniana-activated sludge consortium treating wastewater under light-limited heterotrophic condition. Chem. Eng. J. 2020, 382, 122799. [Google Scholar] [CrossRef]
- Ludwig, W. ARB: A software environment for sequence data. Nucleic Acids Res. 2004, 32, 1363–1371. [Google Scholar] [CrossRef] [Green Version]
- Pirmoradi, N.; Ghaneian, M.T.; Ehrampoush, M.H.; Salmani, M.H.; Hatami, B. The conversion of poultry slaughterhouse wastewater sludge into biodiesel: Process modeling and optimization. Renew. Energy 2021, 178, 1236–1249. [Google Scholar] [CrossRef]
- Xiong, J.-Q.; Kurade, M.B.; Kim, J.R.; Roh, H.-S.; Jeon, B.-H. Ciprofloxacin toxicity and its co-metabolic removal by a freshwater microalga Chlamydomonas mexicana. J. Hazard. Mater. 2017, 323, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Damergi, E.; Schwitzguébel, J.-P.; Refardt, D.; Sharma, S.; Holliger, C.; Ludwig, C. Extraction of carotenoids from Chlorella vulgaris using green solvents and syngas production from residual biomass. Algal Res. 2017, 25, 488–495. [Google Scholar] [CrossRef]
- Jabir, T.F.; Noor Abbood, H.A.; Salman, F.S.; Hafit, A.Y. Influence of pH, pesticide and radiation interactions on the chemical composition of Chlorella vulgaris algae. IOP Conf. Ser. Earth Environ. Sci. 2021, 722, 012046. [Google Scholar] [CrossRef]
- Drouin, G.; Droz, B.; Leresche, F.; Payraudeau, S.; Masbou, J.; Imfeld, G. Direct and indirect photodegradation of atrazine and S -metolachlor in agriculturally impacted surface water and associated C and N isotope fractionation. Environ. Sci. Process. Impacts 2021, 23, 1791–1802. [Google Scholar] [CrossRef]
- de Wilt, A.; Butkovskyi, A.; Tuantet, K.; Leal, L.H.; Fernandes, T.V.; Langenhoff, A.; Zeeman, G. Micropollutant removal in an algal treatment system fed with source separated wastewater streams. J. Hazard. Mater. 2016, 304, 84–92. [Google Scholar] [CrossRef]
- Presentato, A.; Lampis, S.; Vantini, A.; Manea, F.; Daprà, F.; Zuccoli, S.; Vallini, G. On the Ability of Perfluorohexane Sulfonate (PFHxS) Bioaccumulation by Two Pseudomonas sp. Strains Isolated from PFAS-Contaminated Environmental Matrices. Microorganisms 2020, 8, 92. [Google Scholar] [CrossRef]
- García-Galán, M.J.; Matamoros, V.; Uggetti, E.; Díez-Montero, R.; García, J. Removal and environmental risk assessment of contaminants of emerging concern from irrigation waters in a semi-closed microalgae photobioreactor. Environ. Res. 2021, 194, 110278. [Google Scholar] [CrossRef]
- Prosenc, F.; Piechocka, J.; Škufca, D.; Heath, E.; Griessler Bulc, T.; Istenič, D.; Buttiglieri, G. Microalgae-based removal of contaminants of emerging concern: Mechanisms in Chlorella vulgaris and mixed algal-bacterial cultures. J. Hazard. Mater. 2021, 418, 126284. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Wang, J.; Li, X.; Yu, Y.; Zhu, Y. Microbial community and molecular ecological network in the EGSB reactor treating antibiotic wastewater: Response to environmental factors. Ecotoxicol. Environ. Saf. 2021, 208, 111669. [Google Scholar] [CrossRef] [PubMed]
- Matamoros, V.; Uggetti, E.; García, J.; Bayona, J.M. Assessment of the mechanisms involved in the removal of emerging contaminants by microalgae from wastewater: A laboratory scale study. J. Hazard. Mater. 2016, 301, 197–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- da Silva Ribeiro, T.; Grossi, C.D.; Merma, A.G.; dos Santos, B.F.; Torem, M.L. Removal of boron from mining wastewaters by electrocoagulation method: Modelling experimental data using artificial neural networks. Miner. Eng. 2019, 131, 8–13. [Google Scholar] [CrossRef]
- Sharma, R.; Mishra, A.; Pant, D.; Malaviya, P. Recent adva nces in microalgae-based remediation of industrial and non-industrial wastewaters with simultaneous recovery of value-added products. Bioresour. Technol. 2021, 344, 126129. [Google Scholar] [CrossRef]
- Akizuki, S.; Cuevas-Rodríguez, G.; Toda, T. Microalgal-nitrifying bacterial consortium for energy-saving ammonia removal from anaerobic digestate of slaughterhouse wastewater. J. Water Process Eng. 2019, 31, 100753. [Google Scholar] [CrossRef]
- Wang, J.; Song, A.; Huang, Y.; Liao, Q.; Xia, A.; Zhu, X.; Zhu, X. Domesticating Chlorella vulgaris with gradually increased the concentration of digested piggery wastewater to bio-remove ammonia nitrogen. Algal Res. 2021, 60, 102526. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, J.; Shinde, S.; Wang, X.; Li, Y.; Dai, Y.; Ren, J.; Zhang, P.; Liu, X. Simultaneous wastewater treatment and energy harvesting in microbial fuel cells: An update on the biocatalysts. RSC Adv. 2020, 10, 25874–25887. [Google Scholar] [CrossRef]
- Nguyen, T.-T.-D.; Bui, X.-T.; Nguyen, T.-T.; Hao Ngo, H.; Yi Andrew Lin, K.; Lin, C.; Le, L.-T.; Dang, B.-T.; Bui, M.-H.; Varjani, S. Co-culture of microalgae-activated sludge in sequencing batch photobioreactor systems: Effects of natural and artificial lighting on wastewater treatment. Bioresour. Technol. 2022, 343, 126091. [Google Scholar] [CrossRef]
- Zhu, S.; Qin, L.; Feng, P.; Shang, C.; Wang, Z.; Yuan, Z. Treatment of low C/N ratio wastewater and biomass production using co-culture of Chlorella vulgaris and activated sludge in a batch photobioreactor. Bioresour. Technol. 2019, 274, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Shen, Z.; Fang, W.; Gao, G. Composition of bacterial communities in municipal wastewater treatment plant. Sci. Total Environ. 2019, 689, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.-T.-D.; Nguyen, T.-T.; An Binh, Q.; Bui, X.-T.; Ngo, H.H.; Vo, H.N.P.; Andrew Lin, K.-Y.; Vo, T.-D.-H.; Guo, W.; Lin, C.; et al. Co-culture of microalgae-activated sludge for wastewater treatment and biomass production: Exploring their role under different inoculation ratios. Bioresour. Technol. 2020, 314, 123754. [Google Scholar] [CrossRef] [PubMed]
- Elmaadawy, K.; Liu, B.; Hassan, G.; Wang, X.; Wang, Q.; Hu, J.; Hou, H.; Yang, J.; Wu, X. Microalgae-assisted fixed-film activated sludge MFC for landfill leachate treatment and energy recovery. Process Saf. Environ. Prot. 2022, 160, 221–231. [Google Scholar] [CrossRef]
- Maresca, J.A.; Keffer, J.L.; Hempel, P.P.; Polson, S.W.; Shevchenko, O.; Bhavsar, J.; Powell, D.; Miller, K.J.; Singh, A.; Hahn, M.W. Light Modulates the Physiology of Nonphototrophic Actinobacteria. J. Bacteriol. 2019, 201, e00740-18. [Google Scholar] [CrossRef] [Green Version]
- Berthold, D.E.; Shetty, K.G.; Jayachandran, K.; Laughinghouse, H.D.; Gantar, M. Enhancing algal biomass and lipid production through bacterial co-culture. Biomass Bioenergy 2019, 122, 280–289. [Google Scholar] [CrossRef]
- Ray, A.; Nayak, M.; Ghosh, A. A review on co-culturing of microalgae: A greener strategy towards sustainable biofuels production. Sci. Total Environ. 2022, 802, 149765. [Google Scholar] [CrossRef]
- Anyanwu, R.C.; Rodriguez, C.; Durrant, A.; Olabi, A.G. Evaluation of Growth Rate and Biomass Productivity of Scenedesmus quadricauda and Chlorella vulgaris under Different LED Wavelengths and Photoperiods. Sustainability 2022, 14, 6108. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.; Zheng, X.; Yang, M.; Gao, X.; Huang, J.; Zhang, L.; Fan, Z. Toxic effects and mechanisms of PFOA and its substitute GenX on the photosynthesis of Chlorella pyrenoidosa. Sci. Total Environ. 2021, 765, 144431. [Google Scholar] [CrossRef]





| Experiment | Contact Time (d) | Initial Concentration of PPCPs (mg/L) | Initial Concentration of TAN and COD (mg/L) |
|---|---|---|---|
| 1 | 1–7 | 0.2 | 50 |
| 2 | 1–7 | 0.6 | 80 |
| 3 | 1–7 | 1.0 | 110 |
| 4 | 1–7 | 1.4 | 140 |
| 5 | 1–7 | 1.8 | 170 |
| 6 | 1–7 | 2.2 | 200 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mojiri, A.; Ozaki, N.; Kazeroon, R.A.; Rezania, S.; Baharlooeian, M.; Vakili, M.; Farraji, H.; Ohashi, A.; Kindaichi, T.; Zhou, J.L. Contaminant Removal from Wastewater by Microalgal Photobioreactors and Modeling by Artificial Neural Network. Water 2022, 14, 4046. https://doi.org/10.3390/w14244046
Mojiri A, Ozaki N, Kazeroon RA, Rezania S, Baharlooeian M, Vakili M, Farraji H, Ohashi A, Kindaichi T, Zhou JL. Contaminant Removal from Wastewater by Microalgal Photobioreactors and Modeling by Artificial Neural Network. Water. 2022; 14(24):4046. https://doi.org/10.3390/w14244046
Chicago/Turabian StyleMojiri, Amin, Noriatsu Ozaki, Reza Andasht Kazeroon, Shahabaldin Rezania, Maedeh Baharlooeian, Mohammadtaghi Vakili, Hossein Farraji, Akiyoshi Ohashi, Tomonori Kindaichi, and John L. Zhou. 2022. "Contaminant Removal from Wastewater by Microalgal Photobioreactors and Modeling by Artificial Neural Network" Water 14, no. 24: 4046. https://doi.org/10.3390/w14244046
APA StyleMojiri, A., Ozaki, N., Kazeroon, R. A., Rezania, S., Baharlooeian, M., Vakili, M., Farraji, H., Ohashi, A., Kindaichi, T., & Zhou, J. L. (2022). Contaminant Removal from Wastewater by Microalgal Photobioreactors and Modeling by Artificial Neural Network. Water, 14(24), 4046. https://doi.org/10.3390/w14244046












