Variability of the Carbon Isotope Composition of Peat-Forming Plants during the Biochemical Transformation
Abstract
:1. Introduction
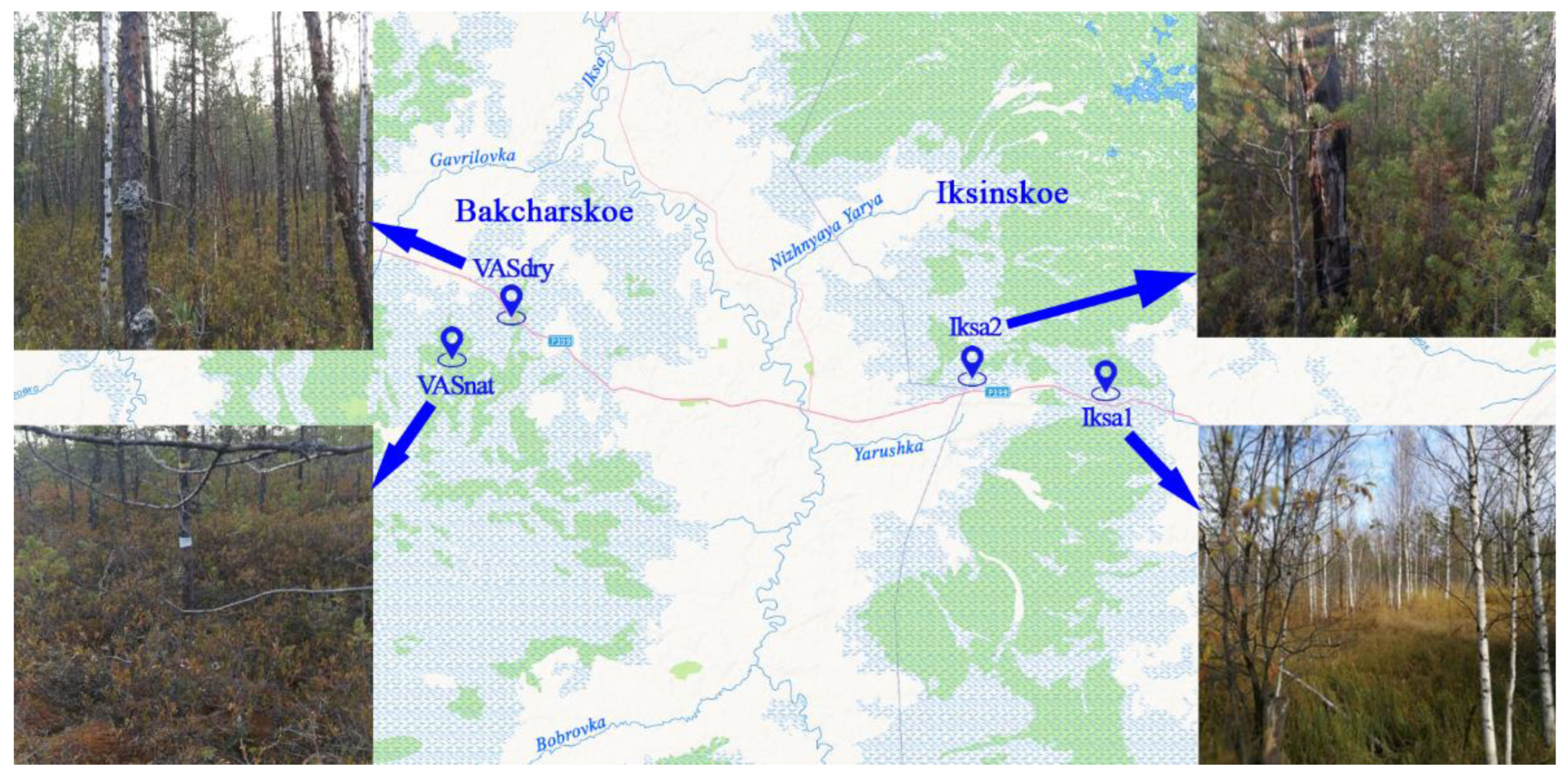
2. Materials and Methods
2.1. Determination of the Decomposition Rate of Peat-Forming Plants
2.2. Statistical Analysis
3. Results
3.1. Weather and Hydrothermal Conditions
3.2. Decomposition Rate
3.3. Variability of the Isotope Composition
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vompersky, S.E. Role in the carbon cycle bogs. In Biogeocenotic Features of Bogs and Their Rational Use; Nauka: Moscow, Russia, 1994; pp. 5–37. [Google Scholar]
- Gorham, E. Northern peatlands: Role in the carbon cycle and probable responses to climatic warming. Ecol. Appl. 1991, 1, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Succow, M.; Joosten, H. Landschaftsökologische Moorkunde; Schweizerbart Science Publishers: Stuttgart, Germany, 2001; p. 622. [Google Scholar]
- Efremov, S.P.; Efremova, T.T.; Melentieva, N.V. Carbon storages in peatland ecosystems. In Carbon in Forest and Peatland Ecosystems of Russia; Alekseev, V.O., Berdsy, R.A., Eds.; Institute of forest SB RAS: Krasnoyarsk, Russia, 1994; pp. 128–139. [Google Scholar]
- Titlyanova, A.A.; Bulavko, G.I.; Kudryashova, S.Y.; Naumov, A.V.; Smirnov, V.V.; Tanasienko, A.A. The Reserves and Losses of Organic Carbon in the Soils of Siberia. Eur. Soil Sci. 1998, 31, 45–53. [Google Scholar]
- Kozlovskaya, L.S.; Medvedeva, V.M.; Piavchenko, N.I. Dynamics of Organic Matter in the Process of Peat Formation; Nauka LO Publ.: Leningrad, Russia, 1978; p. 171. [Google Scholar]
- Boch, M.S.; Masing, V.V. Wetland Ecosystems of the Soviet Union; Nauka: Leningrad, Russia, 1979; p. 188. [Google Scholar]
- Bambalov, N.N.; Horuzhik, A.V.; Lukoshko, E.S.; Strigutsky, V.P. Transformation of dead plants in marsh biogeocenoses. In Experiment and Mathematical Modeling in the Study of Biogeocenoses of Forests and Bog; Nauka Publ.: Moscow, Russia, 1990; pp. 53–63. [Google Scholar]
- Mironycheva-Tokareva, N.P.; Kosykh, N.P.; Vishnyakova, E.K. Carbon balance components of wetlands in the middle taiga and the forest-tundra. West Siberian Peatlands and Carbon Cycle: Past and present. In Proceedings of the Second International Field Symposium, Khanty-Mansiysk, Russia, 24 August–2 September 2007; pp. 117–118. [Google Scholar]
- Parshina, E.K. Destruction of Plant Matter in Bog Ecosystems of the Taiga and Forest-Tundra Zones of Western Siberia. Ph.D. Dissertation, Tomsk State University, Tomsk, Russia, 2009. [Google Scholar]
- Vishnyakova, E.K.; Mironycheva-Tokareva, N.P.; Kosykh, N.P. Decomposition Dynamics of Plants of Bogs (Data: Vasyugan Bog). Tomsk. State Pedagog. Univ. Bull. 2012, 7, 88–93. [Google Scholar]
- Golovatskaya, E.A.; Nikonova, L.G. Decomposition of plant residues in peat soils of oligotrophic peatlands. Vestn. Tomsk. Gos. Univ. Biol. 2013, 3, 137–151. [Google Scholar]
- Verhoeven, J.T.A.; Arts, H.H.M. Decomposition of Carex litter in fens with different water chemistry. Aquat. Bot. 1992, 43, 365–377. [Google Scholar] [CrossRef]
- Bragazza, L.; Siffi, C.; Iacumin, P.; Gerdol, R. Mass loss and nutrient release during litter decay in peatland: The role of microbial adaptability to litter chemistry. Soil Biol. Biochem. 2007, 39, 257–267. [Google Scholar] [CrossRef]
- Bragazza, L.; Buttler, A.; Siegenthaler, A.; Mitchel, E.A. Plant litter decomposition and nutrient release in peatlands. Carbon Cycl. North. Peatl. 2009, 184, 99–100. [Google Scholar]
- Leroy, F.; Gogo, S.; Buttler, A.; Bragazza, L.; Laggoun-Défarge, F. Litter decomposition in peatlands is promoted by mixed plants. J. Soils Sediments 2018, 18, 739–749. [Google Scholar] [CrossRef] [Green Version]
- Denisenkov, V.P. Basics of Swamp Science; St. Petersburg University Publ.: St. Petersburg, Russia, 2000; p. 224. [Google Scholar]
- Peltoniemi, K.; Strakova, P.; Fritze, H.; Iraizoz, P.A.; Pennanen, T.; Laiho, R. How water-level drawdown modified litter-decomposing fungal and actinobacterial communities in boreal peatlands. Soil Biol. Biochem. 2012, 51, 20–34. [Google Scholar] [CrossRef]
- Nikonova, L.G.; Golovatskaya, E.A.; Kur’ina, I.V.; Kurganova, I.N. Decomposition rate of peat-forming plants in oligotrophic bogs of the southern taiga subzone of Western Siberia: Assessment of the effect of water table level and peat deposit temperature. Eurasian Soil Sci. 2019, 52, 1101–1111. [Google Scholar] [CrossRef]
- Nikonova, L.G.; Kurganova, I.N.; Lopes de Gerenyu, V.O.; Zhmurin, V.A.; Golovatskaya, E.A. Impact of abiotic factors on the decomposition of litter of peat-forming plants in the incubation experiment. Tomsk. State Univ. J. Biol. 2019, 46, 148–170. [Google Scholar] [CrossRef] [Green Version]
- Karelin, D.V.; Zamolodchikov, D.G. Carbon Exchange in Cryogenic Ecosystems; Nauka: Moscow, Russia, 2008; p. 344. [Google Scholar]
- Kudeyarov, V.N.; Zavarzin, G.A.; Blagodatskii, S.A.; Borisov, A.V.; Voronin, P.Y.; Demkin, V.A.; Demkina, T.S.; Yevdokimov, I.V.; Zamolodchikov, D.G.; Karelin ; et al. Pools and Fluxes of Carbon in Terrestrial Ecosystems of Russia; Nauka: Moscow, Russia, 2007; p. 315. [Google Scholar]
- Leadley, P.; Pereira, H.M.; Alkemade, R.; Fernandez-Manjarrés, J.F.; Proença, V.; Scharlemann, J.P.W.; Walpole, M.J. Biodiversity Scenarios: Projections of 21st Century Change in Biodiversity and Associated Ecosystem Services; Secretariat of the Convention on Biological Diversity: Montreal, Canada, 2010; p. 132. [Google Scholar]
- Gifford, R.M.; Howden, M. Vegetation thickening in an ecological perspective: Significance to national greenhouse gas inventories. Environ. Sci. Policy 2001, 4, 59–72. [Google Scholar] [CrossRef]
- Bambalov, N.N. Balance of the Organic Matter of Peat Soils and Methods of Its Study; Nauka Publ.: Minsk, Belarus, 1984; p. 175. [Google Scholar]
- Efimov, V.N. Peat Soils and Their Fertility; Agropromizdat Publ.: Leningrad, Russia, 1986; p. 264. [Google Scholar]
- Golovatskaya, E.A. Carbon fluxes in bog ecosystems of Western Siberia. Doctoral Dissertation, Tomsk State University, Tomsk, Russia, 2013. [Google Scholar]
- Parish, F.; Sirin, A.A.; Charman, D.; Joosten, H.; Minaeva, T.Y.; Silvius, M. Assessment on Peatlands, Biodiversity and Climate Change; Wetlands International: Kuala Lumpur, Wageningen, 2008; p. 20. [Google Scholar]
- Titlyanova, A.A.; Sambuu, A.D. Succession in Grasslands; Publishing House SB RAS: Novosibirsk, Russia, 2016; p. 191. [Google Scholar]
- Mironycheva-Tokareva, N.P.; Mikhailova, E.V.; Vishnyakova, E.K. Transformation of the vegetation cover of marsh ecosystems in the construction of the roadway (on the example of the taiga zone of Western Siberia). Vestn. Orenb. Gos. Univ. 2017, 12, 55–59. [Google Scholar]
- Tiunov, A.V. Structure and Functions of Soil Communities of a Monsoon Tropical Forest (Cat Tien National Park, Southern Vietnam); KMK Scientific Press: Moscow, Russia, 2011; p. 277. [Google Scholar]
- Tiunov, A.V. Stable isotopes of carbon and nitrogen in soil ecological studies. Biol Bull. Russ. Acad. Sci. 2007, 34, 395–407. [Google Scholar] [CrossRef]
- Robinson, D. δ15N as an integrator of the nitrogen cycle. Trends Ecol. Evol. 2001, 16, 153–162. [Google Scholar] [CrossRef]
- Fry, B. Stable Isotope Ecology; Springer: New York, NY, USA, 2006; Volume 521, p. 318. [Google Scholar]
- Melillo, J.M.; Aber, J.D.; Linkins, A.E.; Ricca, A.; Fry, B.; Nadelhoffer, K.J. Carbon and nitrogen dynamics along the decay continuum: Plant litter to soil organic matter. Plant. Soil 1989, 115, 189–198. [Google Scholar] [CrossRef]
- Wedin, D.A.; Tieszen, L.L.; Dewey, B.; Pastor, J. Carbon isotope dynamics during grass decomposition and soil organic matter formation. Ecology 1995, 76, 1383–1392. [Google Scholar] [CrossRef]
- Amundson, R.; Baisden, W.T. Stable isotope tracers and mathematical models in soil organic matter studies. In Methods in ecosystem science; Springer: New York, NY, USA, 2000; pp. 117–137. [Google Scholar]
- Connin, S.L.; Feng, X.; Virginia, R.A. Isotopic discrimination during long-term decomposition in an arid land ecosystem. Soil Biol. Biochem. 2001, 33, 41–51. [Google Scholar] [CrossRef]
- Lehmann, M.F.; Bernasconi, S.M.; Barbieri, A.; McKenzie, J.A. Preservation of organic matter and alteration of its carbon and nitrogen isotope composition during simulated and in situ early sedimentary diagenesis. Geochim. Cosmochim. Acta 2002, 66, 3573–3584. [Google Scholar] [CrossRef]
- Fellerhoff, C.; Voss, M.; Wantzen, K.M. Stable carbon and nitrogen isotope signatures of decomposing tropical macrophytes. Aquatic Ecol. 2003, 37, 361–375. [Google Scholar] [CrossRef]
- Fernandez, L.; Mahieu, N.; Cadisch, G. Carbon isotopic fractionation during decomposition of plant materials of different quality. Global Biogeochem. Cycles 2003, 17, 1075. [Google Scholar] [CrossRef]
- Ågren, G.I.; Bosatta, E.; Balesdent, J. Isotope discrimination during decomposition of organic matter: A theoretical analysis. Soil Sci. Soc. Am. J. 1996, 60, 1121–1126. [Google Scholar] [CrossRef]
- Handley, L.L.; Raven, J.A. The use of natural abundance of nitrogen isotopes in plant physiology and ecology. Plant. Cell Environ. 1992, 15, 965–985. [Google Scholar] [CrossRef]
- Högberg, P. Tansley review no. 95 15N natural abundance in soil–plant systems. New Phytol. 1997, 137, 179–203. [Google Scholar] [CrossRef]
- Kendall, C. Tracing nitrogen sources and cycling in catchments. In Isotope Tracers in Catchment Hydrology; Kendall, C., McDonnell, J.J., Eds.; Elsevier Science, B.V.: Amsterdam, The Netherlands, 1998; pp. 519–576. [Google Scholar]
- Bunting, M.J.; Warner, B.G.; Aravena, R. Late Quaternary vegetation dynamics and hydroseral development in a Thuja occidentals swamp in southern Ontario. Can. J. Earth Sci. 1996, 33, 1439–1456. [Google Scholar] [CrossRef]
- Lowe, J.J.; Walker, M.J.C. Reconstructing Quaternary Environments, 2nd ed.; Addison Wesley Longman: Harlow, UK, 1997; p. 446. [Google Scholar]
- Vardy, S.R.; Warner, B.G.; Aravena, R. Holocene Climate Effects on the Development of a Peatland on the Tuktoyaktuk Peninsula, Northwest Territories. Quat Res. 1997, 47, 90–104. [Google Scholar] [CrossRef]
- Jasinski, J.P.P.; Warner, B.G.; Andreev, A.A.; Aravena, R.; Gilbert, S.E.; Zeeb, B.A.; Smol, J.P.; Velichko, A.A. Holocene environmental history of a peatland in the Lena River valley, Siberia. Can. J. Earth Sci. 1998, 35, 637–648. [Google Scholar] [CrossRef]
- Wolfe, B.B.; Edwards, T.W.D.; Aravena, R.; Forman, S.L.; Warner, B.G.; Velichko, A.A.; MacDonald, G.M. Holocene paleohydrology and paleoclimate at treeline, north-central Russia, inferred from oxygen isotope records in lake sediment cellulose. Quat. Res. 2000, 53, 319–329. [Google Scholar] [CrossRef]
- Earle, L.; Warner, B.G.; Aravena, R. Rapid development of an unusual peat-accumulation ecosystem in the Chilean Altiplano. Quat. Res. 2003, 59, 2–11. [Google Scholar] [CrossRef]
- Asada, T.; Warner, B.; Aravena, R. Effects of early stage of decomposition on change in carbon and nitrogen isotopes in Sphagnum litter. J. Plant. Interact. 2005, 1, 229–237. [Google Scholar] [CrossRef] [Green Version]
- Golovatskaya, E.A.; Nikonova, L.G. Influence of the level of bog waters on the processes of transformation of sphagnum mosses in peat soil of oligotrophic bogs. Eurasian Soil Sci. 2017, 5, 580–588. [Google Scholar] [CrossRef]
- Drollinger, S.; Kuzyakov, Y.; Glatzel, S. Effects of peat decomposition on δ13C and δ15N depth profiles of Alpine bogs. Catena 2019, 178, 1–10. [Google Scholar] [CrossRef]
- Drollinger, S.; Knorr, K.H.; Knierzinger, W.; Glatzel, S. Peat decomposition proxies of Alpine bogs along a degradation gradient. Geoderma 2020, 369, 114331. [Google Scholar] [CrossRef]
- Bird, J.A.; Kleber, M.; Torn, M.S. 13C and 15N stabilization dynamics in soil organic matter fractions during needle and fine root decomposition. Org. Geochem. 2008, 39, 465–477. [Google Scholar] [CrossRef]
- Simonova, G.; Volkov, Y.; Markelova, A.; Kalashnikova, D. Isotope monitoring of forest ecosystems. In Proceedings of the International Multidisciplinary Scientific GeoConference: SGEM, Albena, Bulgaria, 29 June–5 July 2017; pp. 885–890. [Google Scholar]
- Nikonova, L.G. Production and Destruction Processes in Oligotrophic Bog Ecosystems of the Southern Taiga Subzone of Western Siberia. Ph.D. Dissertation, Siberian Federal University, Krasnoyarsk, Russia, 2020. [Google Scholar]
- Bazanov, V.A.; Egorov, B.A.; L’gotin, V.A.; Skugarev, A.A. Modern spatial dynamics of the Great Vasyugan Mire (the case of the Iksa-Shegarka interfluve). In The Great Vasyugan Mire Current status and development processes; Kabanov, M.V., Ed.; Institute of Atmospheric Optics SB RAN: Tomsk, Russia, 2002; pp. 190–196. [Google Scholar]
- Kurakov, S.A.; Krutikov, V.A.; Ushakov, V.G. Autonomous meter of the temperature profile of the AIPT. Appar. Exp. Tech. 2008, 5, 166–167. [Google Scholar]
- Ponomareva, V.V.; Plotnikova, T.A. Simultaneous Determination of Overall C and N Content in Peat Soils Using the Anstet Method, Modification Ponomareva and Nikolaeva. Methods of Determination of Humus Content and Composition in Soils (Mineral and Peat) in Russia; Federal Academy of Agricultural Sciences VI Lenin; Central Pedology Museum VV Dokuchaev: Leningrad, Russia, 1975; pp. 79–83. (in Russian) [Google Scholar]
- Vorobyeva, L.A. Theory and Practice of Chemical Analysis of Soils; GEOS: Moscow, Russia, 2006; p. 400. [Google Scholar]
- Lebedev, A.T. Mass Spectrometry for the Analysis of the Environment; Tekhnosfer publ: Moscow, Russia, 2013; p. 632. [Google Scholar]
- Dobrovolskaya, T.G.; Golovchenko, A.V.; Zvyagintsev, D.G. Functions of Microbial Complexes of the High-Moor Peatbogs: Reasons for Slow Peat Destruction; Chernov, I.Y., Ed.; KMK: Moscow, Russia, 2013; p. 131. [Google Scholar]
- Melillo, J.M.; Aber, J.D.; Muratore, J.F. Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 1982, 63, 621–626. [Google Scholar] [CrossRef]
- Sariyildiz, T.; Anderson, J.M. Variation in the chemical composition of green leaves and leaf litters from three deciduous tree species growing on different soil types. For. Ecol. Manag. 2005, 210, 303–319. [Google Scholar] [CrossRef]
- Scheffer, R.A.; van Logtestijn, R.S.P.; Verhoeven, J.T.A. Decomposition of Carex and Sphagnum litter in two mesotrophic fens differing in dominant plant species. Oikos 2001, 92, 44–54. [Google Scholar] [CrossRef]
- Heal, O.W.; French, D.D. Decomposition of organic matter in tundra. In Soil Organisms and Decomposition in Tundra; IBP Tundra Biome: Stockholm, Sweden, 1974; pp. 279–311. [Google Scholar]
- Pereverzev, V.N.; Evdokimova, G.A. Biochemistry of Humus and Nitrogen Soils of the Kola Peninsula; Nauka: Leningrad, 1987; p. 303. [Google Scholar]
- Billett, M.F.; Palmer, S.M.; Hope, D.; Deacon, C.; Storeton-West, R.; Hargreaves, K.J.; Flechard, C.; Fowler, D. Linking land-atmosphere-stream carbon fluxes in a lowland peatland system. Glob. Biogeochem. Cycles 2004, 18, GB1024. [Google Scholar] [CrossRef]
- Rochefort, L.; Vitt, D.H.; Bayley, S.E. Growth, production, and decomposition dynamics of Sphagnum under natural and experimentally acidified conditions. Ecology 1990, 71, 1986–2000. [Google Scholar] [CrossRef]
- Szumigalski, A.R.; Bayley, S.E. Decomposition along a bog to rich fen gradient in central Alberta, Canada. Can. J. Botany 1996, 74, 573–581. [Google Scholar] [CrossRef]
- Hajek, T. Habitat and species controls on Sphagnum production and decomposition in a mountain bog. Boreal Environ. Res. 2009, 14, 947–958. [Google Scholar]
- Maass, W.S.G.; Craigie, J.S. Examinations of some soluble constituents of Sphagnum gametophytes. Can. J. Botany 1964, 42, 805–813. [Google Scholar] [CrossRef]
- Kracht, O.; Gleixner, G. Isotope analysis of pyrolysis products from Sphagnum peat and dissolved organic matter from bog water. Org. Geochem. 2000, 3, 645–654. [Google Scholar] [CrossRef]
- Andrews, J.A.; Matamala, R.; Westover, K.M.; Schlesinger, W.H. Temperature effects on the diversity of soil heterotrophs and the δ13C of soil-respired CO2. Soil Biol. Biochem. 2000, 32, 699–706. [Google Scholar] [CrossRef]
- Benner, R.; Fogel, M.L.; Sprague, E.K.; Hodson, R.E. Depletion of 13C in lignin and its implications for stable carbon isotope studies. Nature 1987, 329, 708–710. [Google Scholar] [CrossRef]
- Adams, M.A.; Grierson, P.F. Stable isotopes at natural abundance in terrestrial plant ecology and ecophysiology: An update. Plant. Biol. 2001, 3, 299–310. [Google Scholar] [CrossRef]
- Thormann, M.N.; Bayley, S.E. Decomposition along a moderate-rich fen-marsh peatland gradient in boreal Alberta, Canada. Wetlands 1997, 17, 123–137. [Google Scholar] [CrossRef]

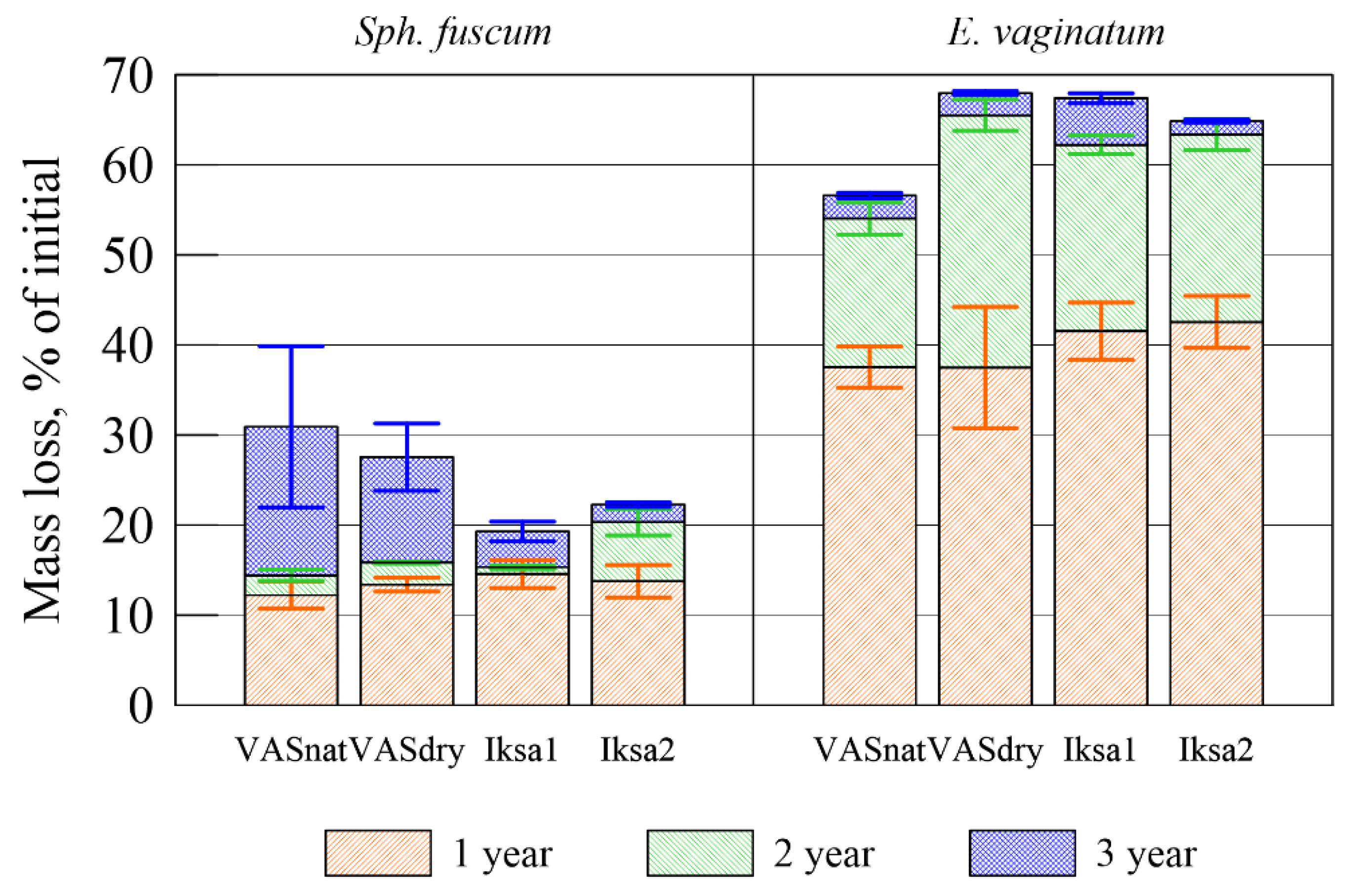
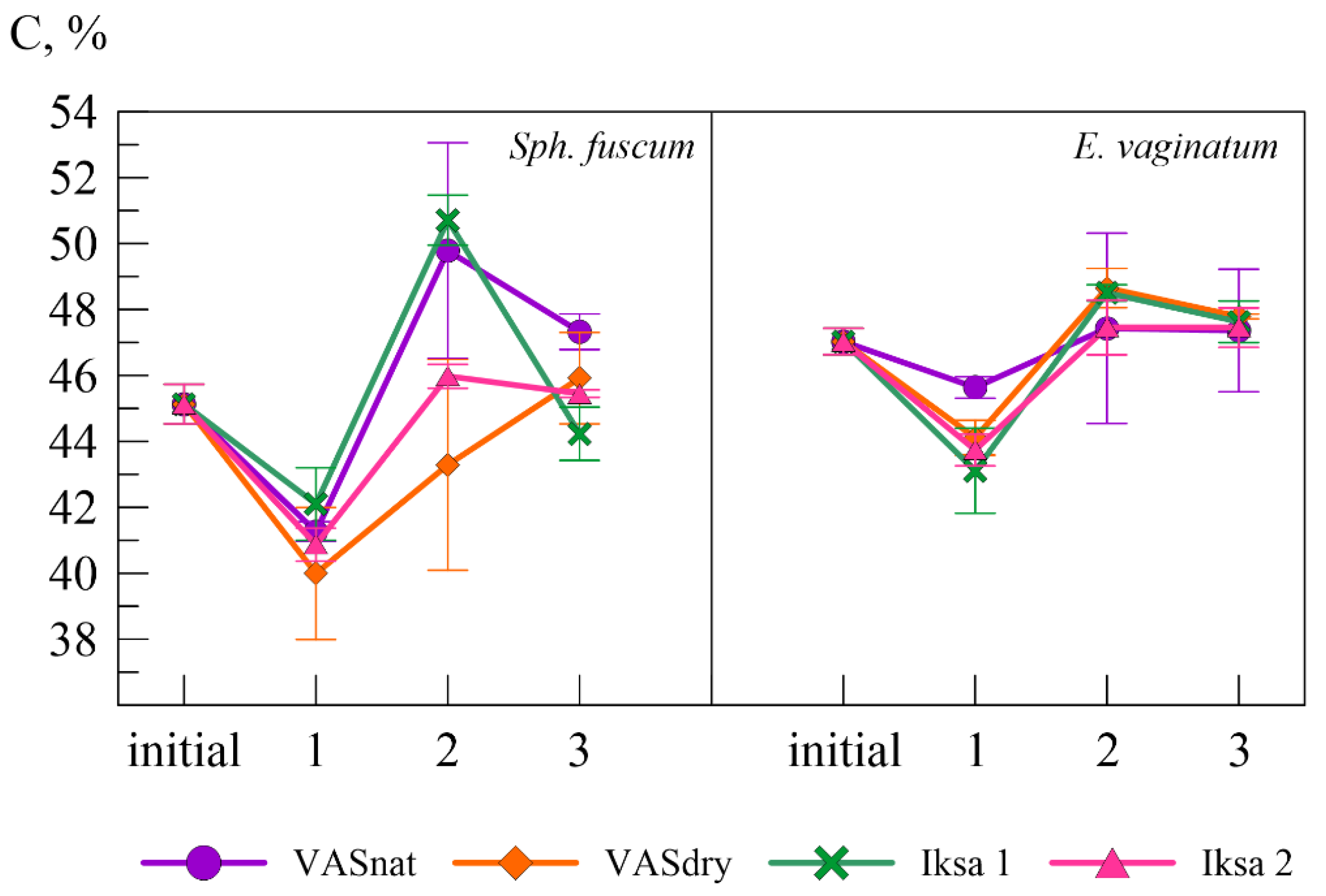


| Plant Species | Site | Mass Loss, % | C, % | δ13C, ‰ | N, % | C/N | Lig | Lig/N |
|---|---|---|---|---|---|---|---|---|
| Sph. fuscum | Initial sample | 45.1 ± 0.6 | −31.2 ± 0.2 | 0.4 ± 0.0 | 118 ± 1 | 12.2 | 32 | |
| VASnat | 30.9 ± 8.9 | 47.3 ± 0.5 | −30.1 ± 0.1 | - | - | - | - | |
| VASdry | 27.6 ± 3.7 | 45.9 ± 1.4 | −29.9 ± 0.2 | - | - | - | - | |
| Iksa1 | 19.3 ± 1.1 | 44.2 ± 0.8 | −30.1 ± 0.2 | - | - | - | - | |
| Iksa2 | 22.3 ± 5.3 | 45.5 ± 0.1 | −30.0 ± 0.2 | - | - | - | - | |
| E. vaginatum | Initial sample | 47.0 ± 0.4 | −26.1 ± 0.2 | 1.0 ± 0.0 | 49 ± 0.1 | 20.3 | 22 | |
| VASnat | 56.6 ± 5.8 | 47.4 ± 1.9 | −27.9 ± 0.1 | - | - | - | - | |
| VASdry | 67.9 ± 1.4 | 47.8 ± 0.1 | −28.3 ± 0.6 | - | - | - | - | |
| Iksa1 | 67.4 ± 3.4 | 47.6 ± 0.6 | −27.9 ± 0.2 | - | - | - | - | |
| Iksa2 | 64.9 ± 5.4 | 47.5 ± 0.6 | −27.4 ± 0.2 | - | - | - | - |
| Factor | Df | F Value | p |
|---|---|---|---|
| Plant species | 1 | 168.238 | <0.05 |
| Sites | 3 | 1.3 | <0.05 |
| Plant species × Sites | 3 | 0.892 | <0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golovatskaya, E.; Nikonova, L.; Simonova, G.; Kalashnikova, D. Variability of the Carbon Isotope Composition of Peat-Forming Plants during the Biochemical Transformation. Water 2022, 14, 4035. https://doi.org/10.3390/w14244035
Golovatskaya E, Nikonova L, Simonova G, Kalashnikova D. Variability of the Carbon Isotope Composition of Peat-Forming Plants during the Biochemical Transformation. Water. 2022; 14(24):4035. https://doi.org/10.3390/w14244035
Chicago/Turabian StyleGolovatskaya, Evgeniya, Liliya Nikonova, Galina Simonova, and Daria Kalashnikova. 2022. "Variability of the Carbon Isotope Composition of Peat-Forming Plants during the Biochemical Transformation" Water 14, no. 24: 4035. https://doi.org/10.3390/w14244035






