New Interpretations of the Adsorption Process of Tetracycline on Biochar via Experimental and Theoretical Studies
Abstract
:1. Introduction
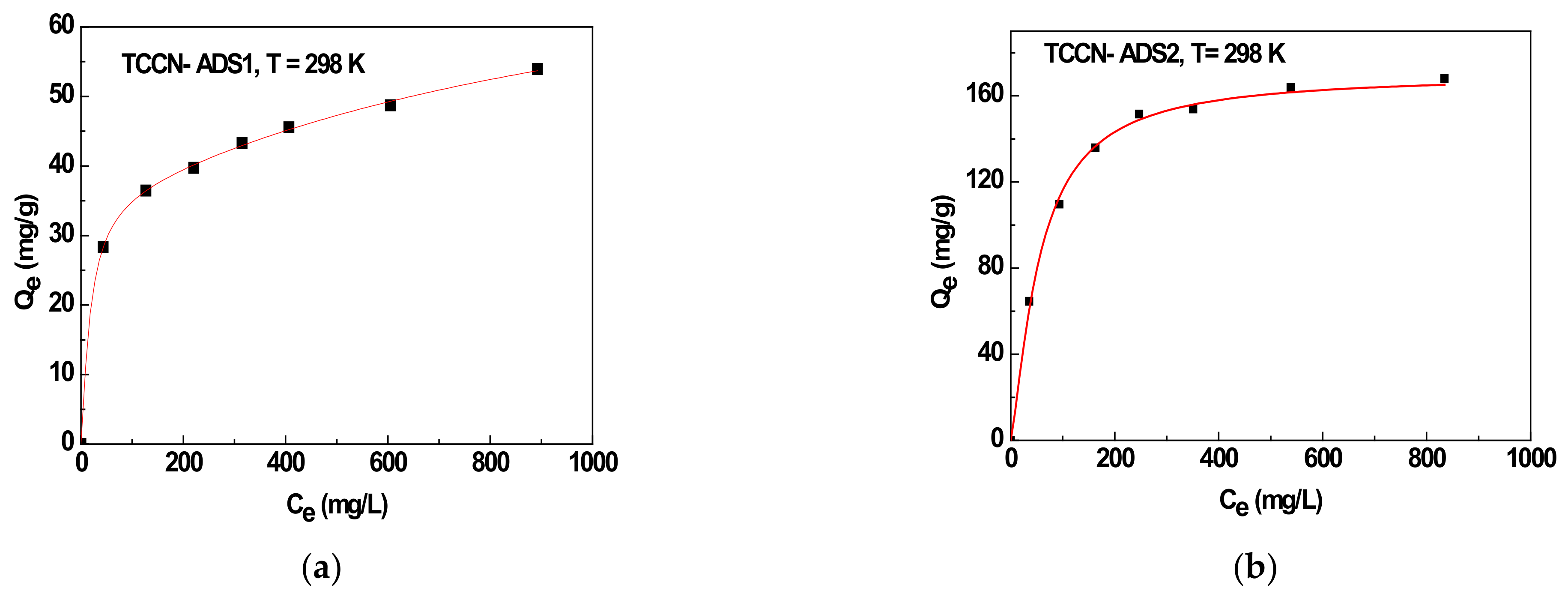
2. Experimental Adsorption Data: Batch Experiments
3. Theoretical Study
3.1. Mono-Layer Adsorption Process: M1
3.2. Double-Layer Adsorption Process: M2
4. Assessment of TCCN Adsorption Mechanism
4.1. Interpretation of ACS
4.2. Estimated Number of TCCN That is Detected by Biochar Adsorption Site (n)
4.3. Parameter DRS
5. Adsorption Energy and General Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Le-Minh, N.; Khan, S.J.; Drewes, J.E.; Stuetz, R.M. Fate of antibiotics during municipal water recycling treatment processes. Water Res. 2010, 44, 4295–4323. [Google Scholar] [CrossRef]
- Fuoco, D. Classification Framework and Chemical Biology of Tetracycline-Structure-Based Drugs. Antibiotics 2012, 1, 1–13. [Google Scholar] [CrossRef]
- Omidvar, M.; Mousavi, S.M.; Soltanieh, M.; Safekordi, A.A. Preparation and characterization of poly (ethersulfone) nanofiltration membranes for amoxicillin removal from contaminated water. J. Environ. Health Sci. Eng. 2014, 12, 18. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Zhang, D.; Zhang, H.; Ghosh, S.; Pan, B. Fast and slow adsorption of carbamazepine on biochar as affected by carbon structure and mineral composition. Sci. Total Environ. 2017, 579, 598–605. [Google Scholar] [CrossRef]
- Norvill, Z.N.; Toledo-Cervantes, A.; Blanco, S.; Shilton, A.; Guieysse, B.; Muñoz, R. Photodegradation and sorption govern tetracycline removal during wastewater treatment in algal ponds. Bioresour. Technol. 2017, 232, 35–43. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.-Q.; Lam, J.C.; Li, W.-W.; Yu, H.-Q.; Lam, P.K. Spatial distribution and removal performance of pharmaceuticals in municipal wastewater treatment plants in China. Sci. Total Environ. 2017, 586, 1162–1169. [Google Scholar] [CrossRef]
- Baumgarten, B.; Jährig, J.; Reemtsma, T.; Jekel, M. Long term laboratory column experiments to simulate bank filtration: Factors controlling removal of sulfamethoxazole. Water Res. 2011, 45, 211–220. [Google Scholar] [CrossRef]
- Homem, V.; Santos, L. Degradation and removal methods of antibiotics from aqueous matrices—A review. J. Environ. Manag. 2011, 92, 2304–2347. [Google Scholar] [CrossRef]
- Gao, L.; Sun, Y.; He, L.; Zhao, W.; Xiang, G.; Jiang, X.; Zhang, S. A polyhedral oligomeric silsesquioxanes/dual ligands-based magnetic adsorbent for effective extraction of aflatoxins in cereals via multiple interactions. Microchem. J. 2021, 160, 105626. [Google Scholar] [CrossRef]
- Leung, H.; Minh, T.; Murphy, M.; Lam, J.C.; So, M.; Martin, M.; Lam, P.K.; Richardson, B. Distribution, fate and risk assessment of antibiotics in sewage treatment plants in Hong Kong, South China. Environ. Int. 2012, 42, 1–9. [Google Scholar] [CrossRef]
- Xu, J.; Xu, Y.; Wang, H.; Guo, C.; Qiu, H.; He, Y.; Zhang, Y.; Li, X.; Meng, W. Occurrence of antibiotics and antibiotic resistance genes in a sewage treatment plant and its effluent-receiving river. Chemosphere 2015, 119, 1379–1385. [Google Scholar] [CrossRef]
- Lu, T.; Xu, X.; Liu, X.; Sun, T. Super hydrophilic PVDF based composite membrane for efficient separation of tetracycline. Chem. Eng. J. 2017, 308, 151–159. [Google Scholar] [CrossRef]
- Yang, S.; Feng, Y.; Gao, D.; Wang, X.; Suo, N.; Yu, Y.; Zhang, S. Electrocatalysis degradation of tetracycline in a three-dimensional aeration electrocatalysis reactor (3D-AER) with a flotation-tailings particle electrode (FPE): Physicochemical properties, influencing factors and the degradation mechanism. J. Hazard. Mater. 2021, 407, 124361. [Google Scholar] [CrossRef]
- Chen, B.; Li, Y.; Li, M.; Cui, M.; Xu, W.; Li, L.; Sun, Y.; Wang, M.; Zhang, Y.; Chen, K. Rapid adsorption of tetracycline in aqueous solution by using MOF-525/graphene oxide composite. Microporous Mesoporous Mater. 2021, 328, 111457. [Google Scholar] [CrossRef]
- Pandi, K.; Choi, J. Selective removal of anionic ions from aqueous environment using iron-based metal-organic frameworks and their mechanistic investigations. J. Mol. Liq. 2021, 329, 115367. [Google Scholar] [CrossRef]
- Huang, D.; Wu, J.; Wang, L.; Liu, X.; Meng, J.; Tang, X.; Tang, C.; Xu, J. Novel insight into adsorption and co-adsorption of heavy metal ions and an organic pollutant by magnetic graphene nanomaterials in water. Chem. Eng. J. 2019, 358, 1399–1409. [Google Scholar] [CrossRef]
- Bai, Y.; Su, J.; Wen, Q.; Li, G.; Xue, L.; Huang, T. Removal of tetracycline by denitrifying Mn (II)-oxidizing bacterium Pseudomonas sp. H117 and biomaterials (BMO and MBMO): Efficiency and mechanisms. Bioresour. Technol. 2020, 312, 123565. [Google Scholar] [CrossRef]
- Duan, Z.; Zhang, W.; Lu, M.; Shao, Z.; Huang, W.; Li, J.; Li, Y.; Mo, J.; Li, Y.; Chen, C. Magnetic Fe3O4/activated carbon for combined adsorption and Fenton oxidation of 4-chlorophenol. Carbon 2020, 167, 351–363. [Google Scholar] [CrossRef]
- Zaher, A.; Taha, M.; Mahmoud, R.K. Possible adsorption mechanisms of the removal of tetracycline from water by La-doped Zn-Fe-layered double hydroxide. J. Mol. Liq. 2021, 322, 114546. [Google Scholar] [CrossRef]
- Zhao, W.; Tian, Y.; Chu, X.; Cui, L.; Zhang, H.; Li, M.; Zhao, P. Preparation and characteristics of a magnetic carbon nanotube adsorbent: Its efficient adsorption and recoverable performances. Sep. Purif. Technol. 2021, 257, 117917. [Google Scholar] [CrossRef]
- Xiang, W.; Zhang, X.; Chen, J.; Zou, W.; He, F.; Hu, X.; Tsang, D.C.W.; Ok, Y.S.; Gao, B. Biochar technology in wastewater treatment: A critical review. Chemosphere 2020, 252, 126539. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, B.; Zhang, H.; Ma, J.; Mu, B.; Zhang, W. A novel Biochar modified by Chitosan-Fe/S for tetracycline adsorption and studies on site energy distribution. Bioresour. Technol. 2019, 294, 122152. [Google Scholar] [CrossRef]
- Sellaoui, L.; Guedidi, H.; Knani, S.; Reinert, L.; Duclaux, L.; Ben Lamine, A. Application of statistical physics formalism to the modeling of adsorption isotherms of ibuprofen on activated carbon. Fluid Phase Equilibria 2015, 387, 103–110. [Google Scholar] [CrossRef]


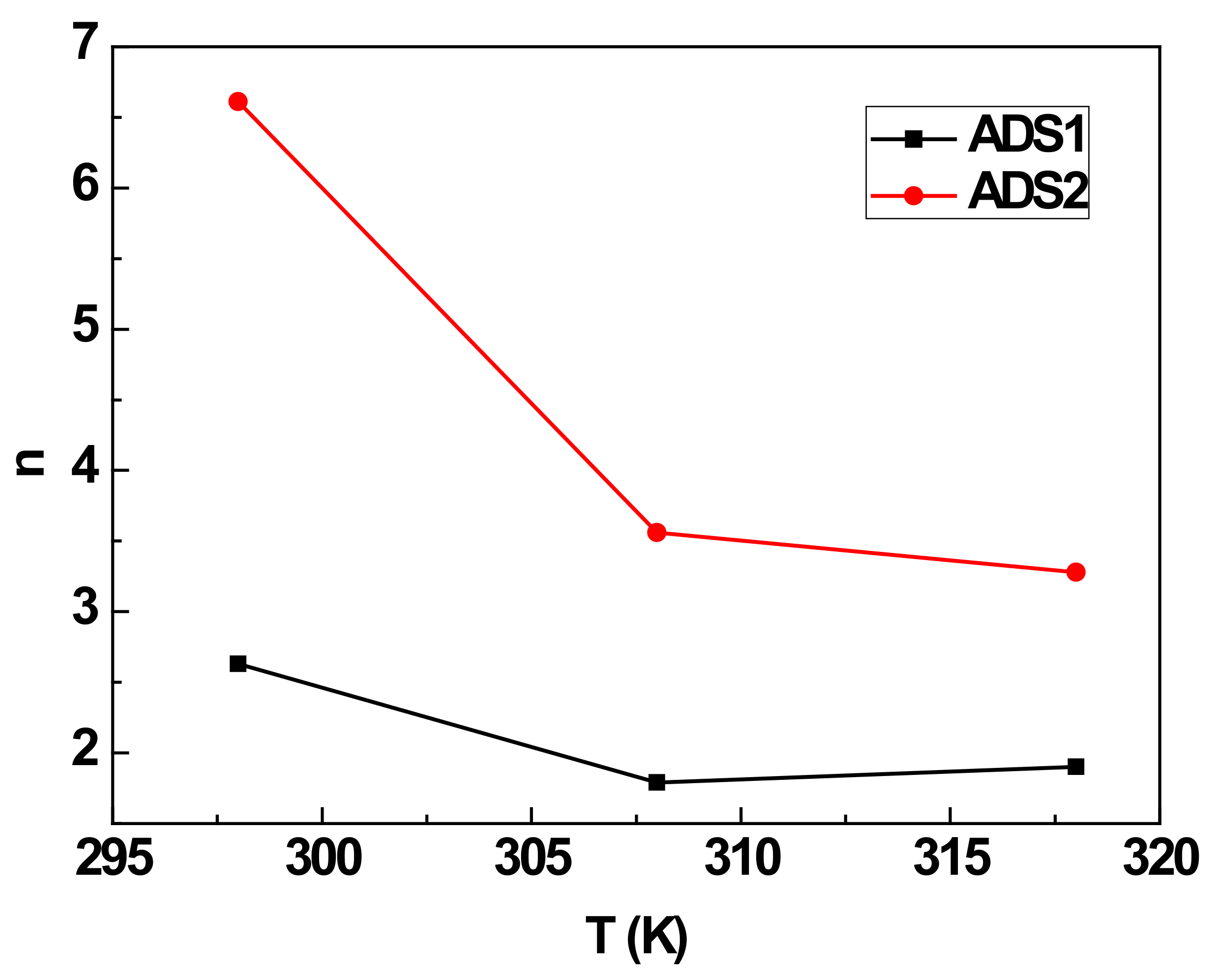
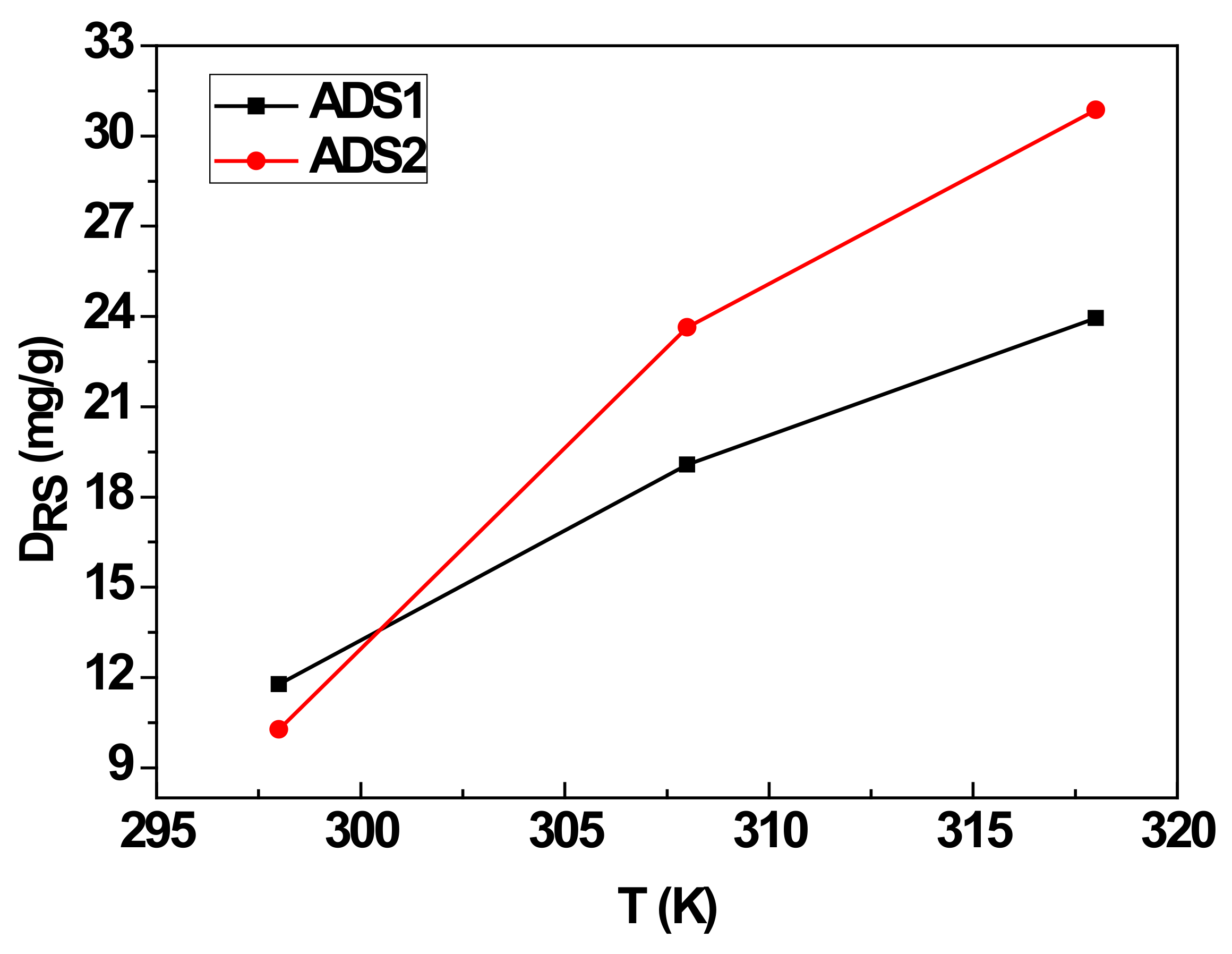
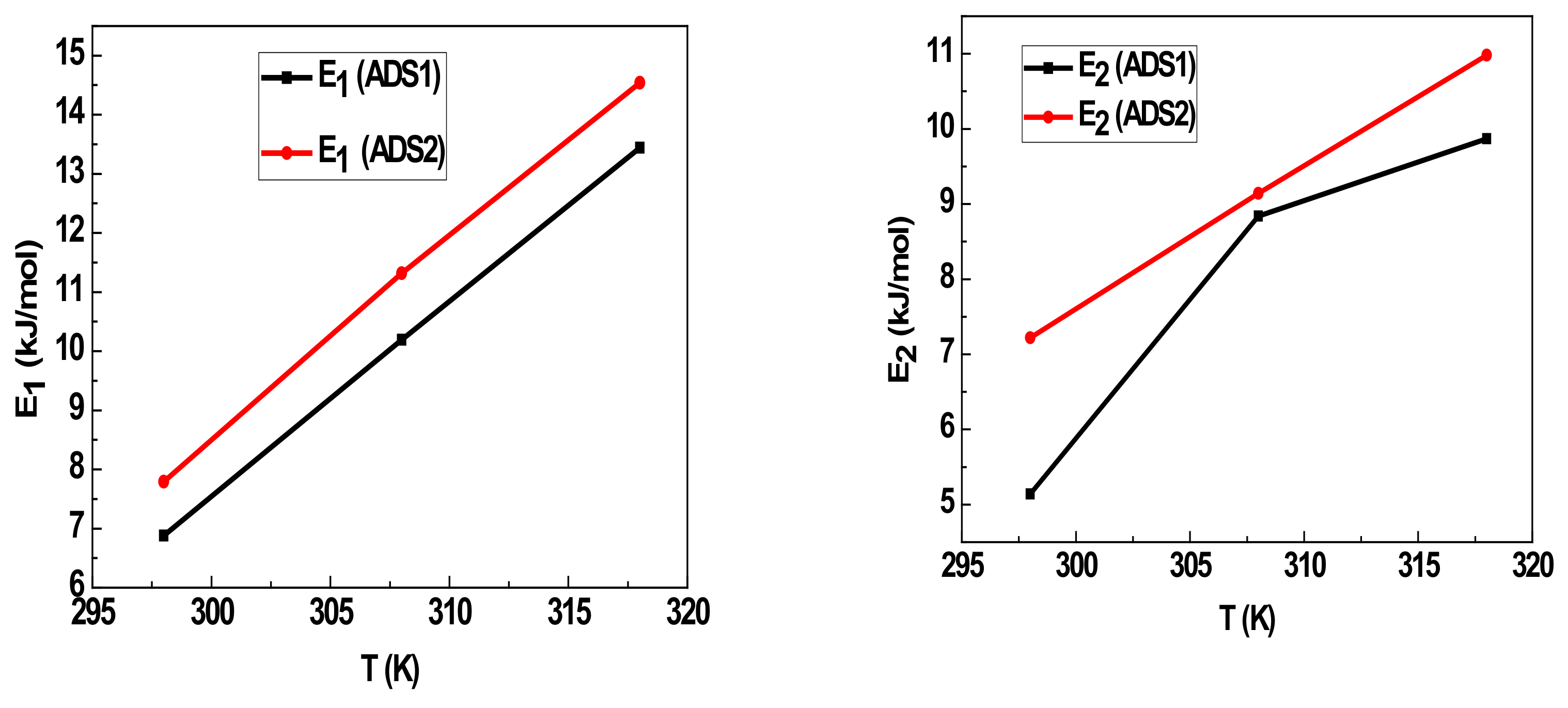
| n | DRS (mg/g) | QACS (mg/g) | E1 (kJ/mol) | E2 (kJ/mol) | |
|---|---|---|---|---|---|
| TCCN-ADS1 | |||||
| 298 K | 2.63 | 11.77 | 61.91 | 6.88 | 5.14 |
| 308 K | 1.79 | 19.08 | 68.30 | 10.19 | 8.84 |
| 318 K | 1.90 | 23.95 | 91 | 13.44 | 9.87 |
| TCCN-ADS2 | |||||
| 298 K | 6.61 | 10.27 | 135.76 | 7.79 | 7.22 |
| 308 K | 3.56 | 23.64 | 168.31 | 11.32 | 9.14 |
| 318 K | 3.28 | 30.87 | 202.5 | 14.54 | 10.98 |
| Possible Cases | Type of Orientation |
|---|---|
| If n ≤ 0.5 | Parallel orientation: the TCCN can be removed via an interaction with two sites |
| If n < 0.5 < 1 | Parallel and non-parallel orientation at the same time |
| n > 1 | Non-parallel orientation: the TCCN can be removed via an interaction with one site |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yahia, M.B.; Sellaoui, L. New Interpretations of the Adsorption Process of Tetracycline on Biochar via Experimental and Theoretical Studies. Water 2022, 14, 3821. https://doi.org/10.3390/w14233821
Yahia MB, Sellaoui L. New Interpretations of the Adsorption Process of Tetracycline on Biochar via Experimental and Theoretical Studies. Water. 2022; 14(23):3821. https://doi.org/10.3390/w14233821
Chicago/Turabian StyleYahia, Manel Ben, and Lotfi Sellaoui. 2022. "New Interpretations of the Adsorption Process of Tetracycline on Biochar via Experimental and Theoretical Studies" Water 14, no. 23: 3821. https://doi.org/10.3390/w14233821
APA StyleYahia, M. B., & Sellaoui, L. (2022). New Interpretations of the Adsorption Process of Tetracycline on Biochar via Experimental and Theoretical Studies. Water, 14(23), 3821. https://doi.org/10.3390/w14233821





