Three-Dimensional Biofilm Electrode Reactors with Polyurethane Sponge Carrier for Highly Efficient Treatment of Pharmaceuticals Wastewater Containing Tetrahydrofuran
Abstract
:1. Introduction
2. Materials and Methods
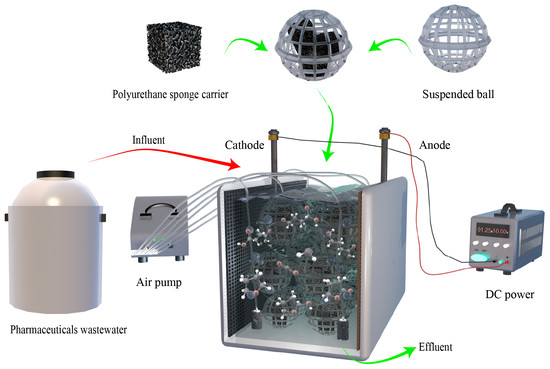
2.1. Experimental Setup
2.2. Wastewater Source
2.3. Experimental Procedure
3. Results and Discussion
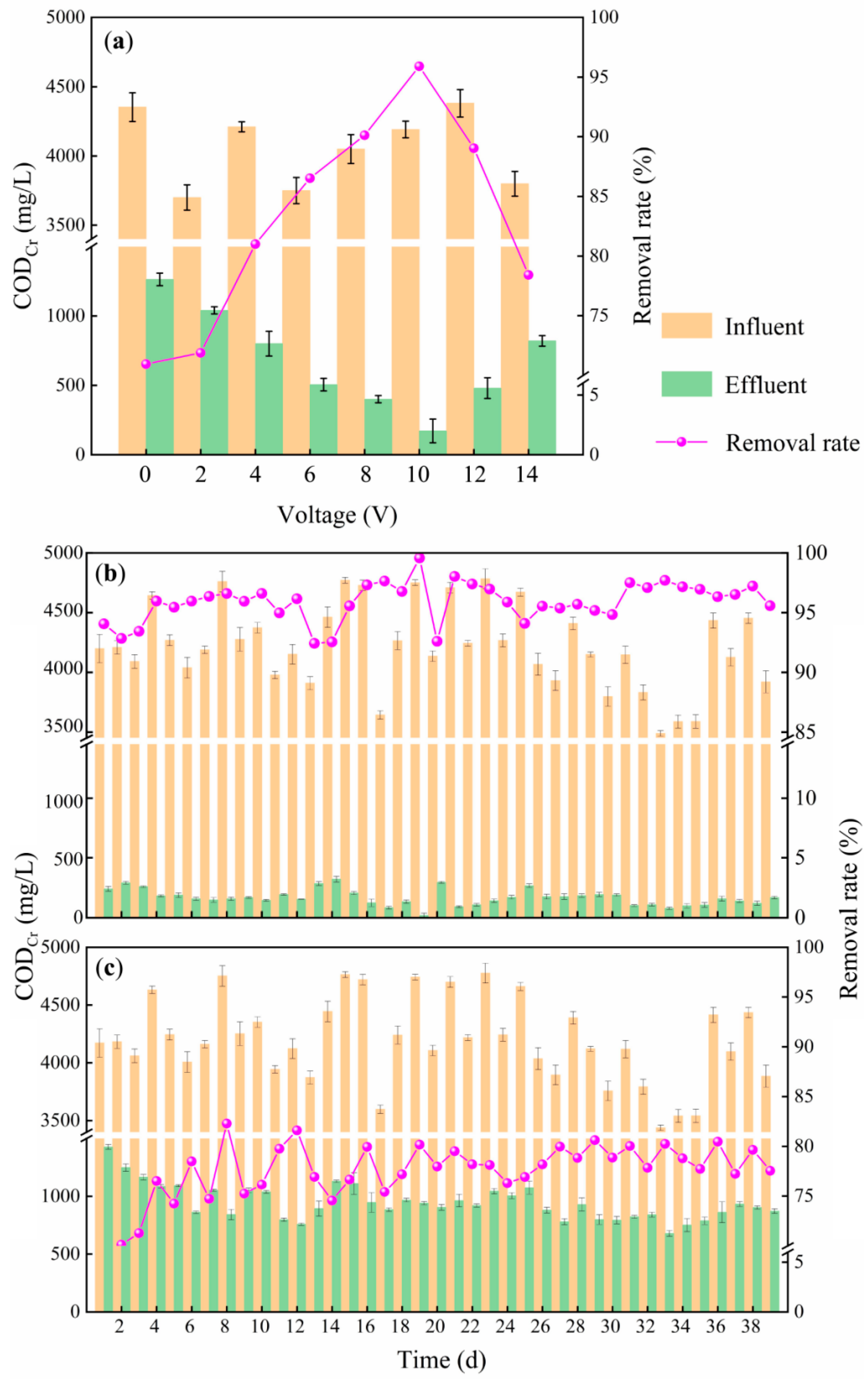
3.1. CODCr Degradation
3.2. Variations in the Organic Components
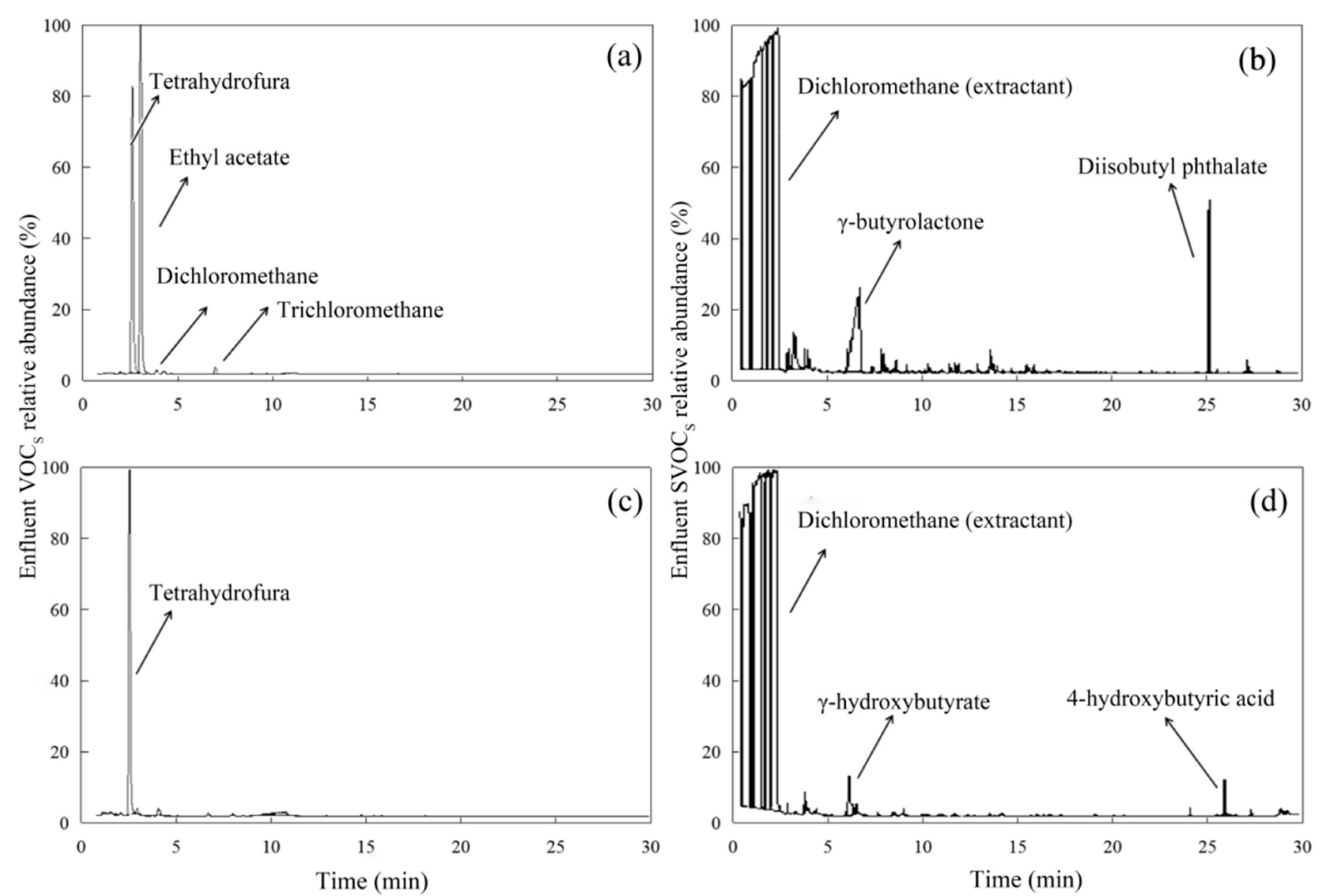
3.2.1. Identification of Intermediates Using GC-MS
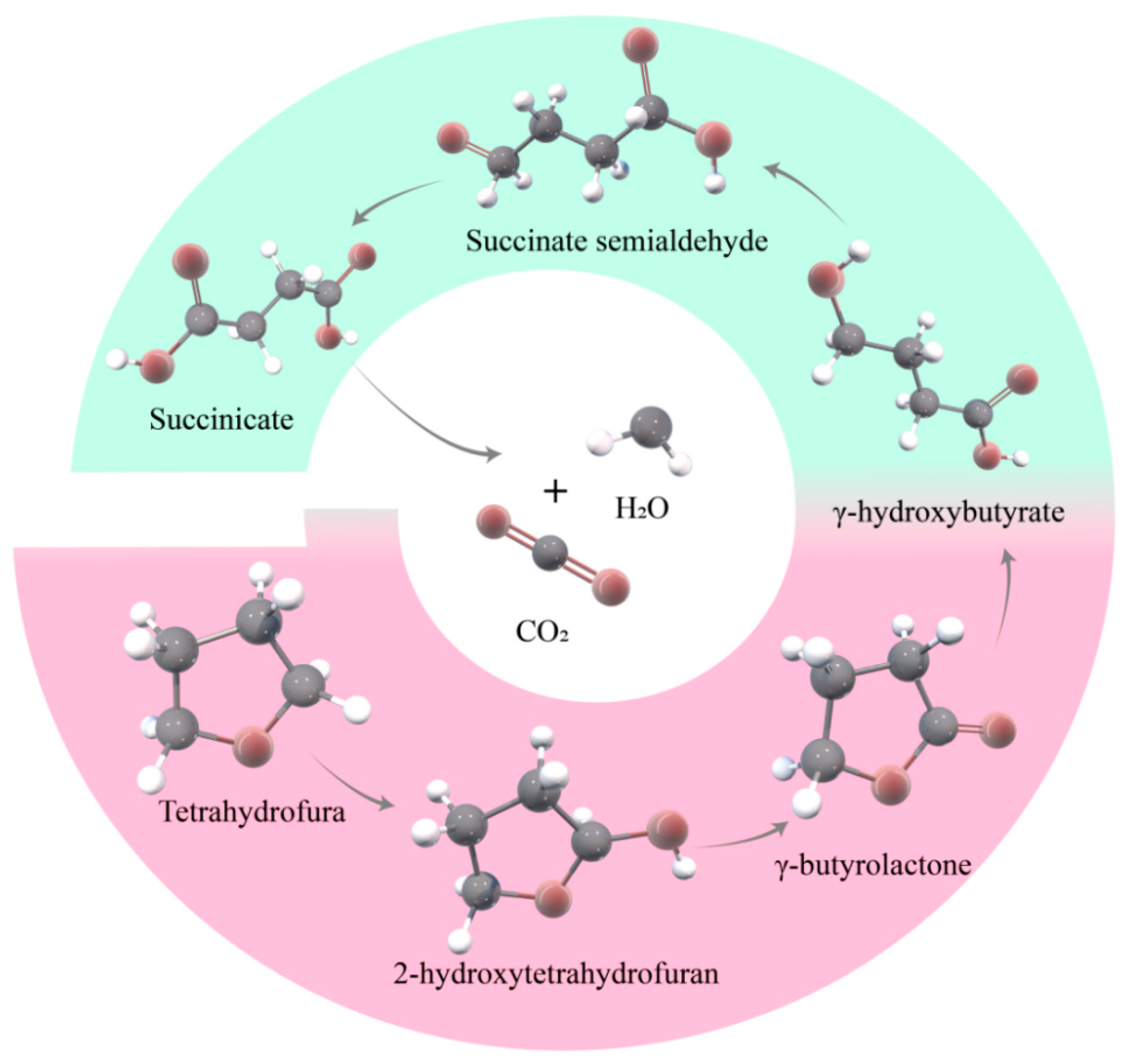
3.2.2. Analysis of Degradation Process of THF in 3D-BERs
3.3. Microbial Community in 3D-BERs
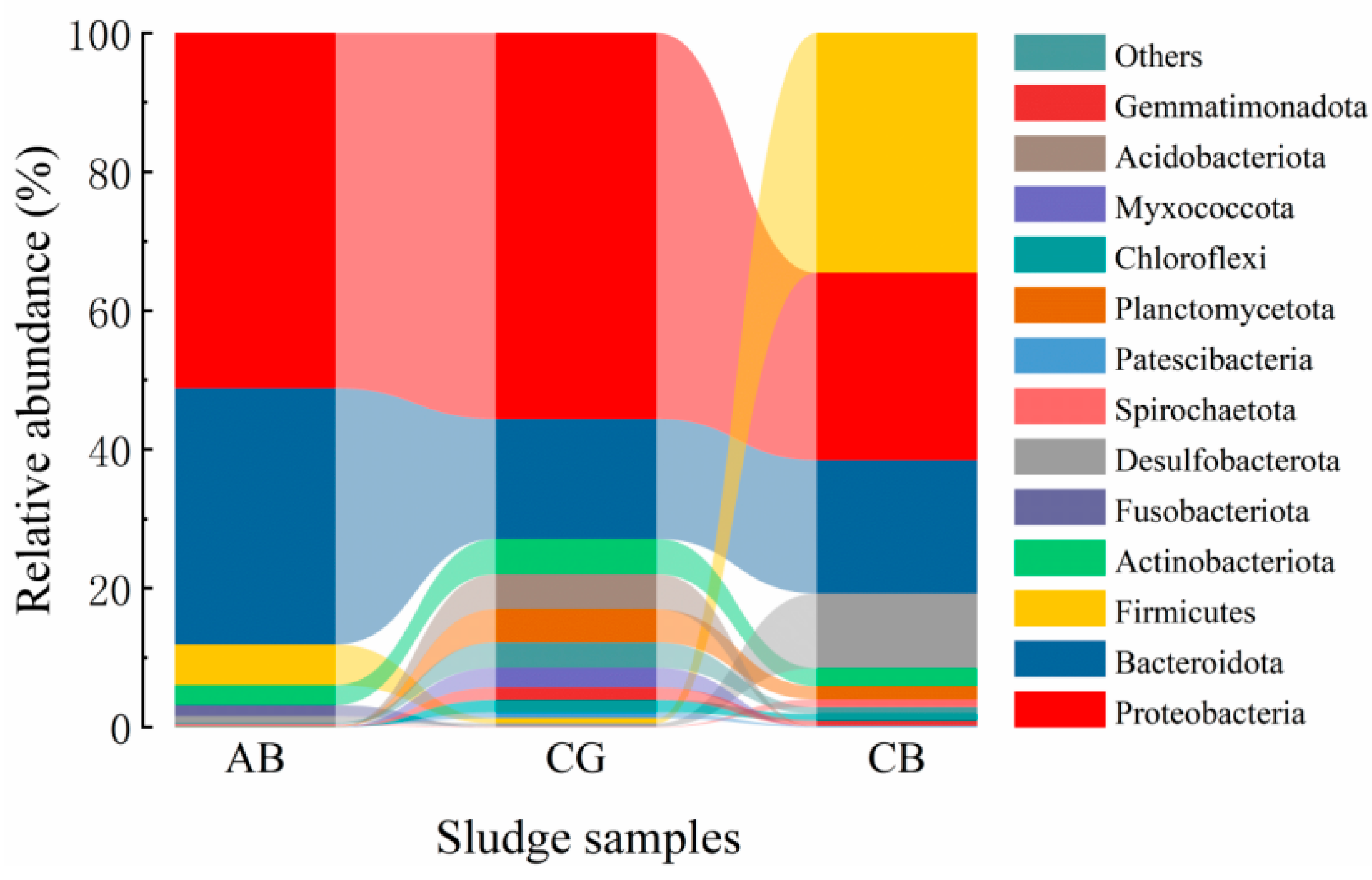
3.3.1. Microbial Community Analysis at the Phylum Level
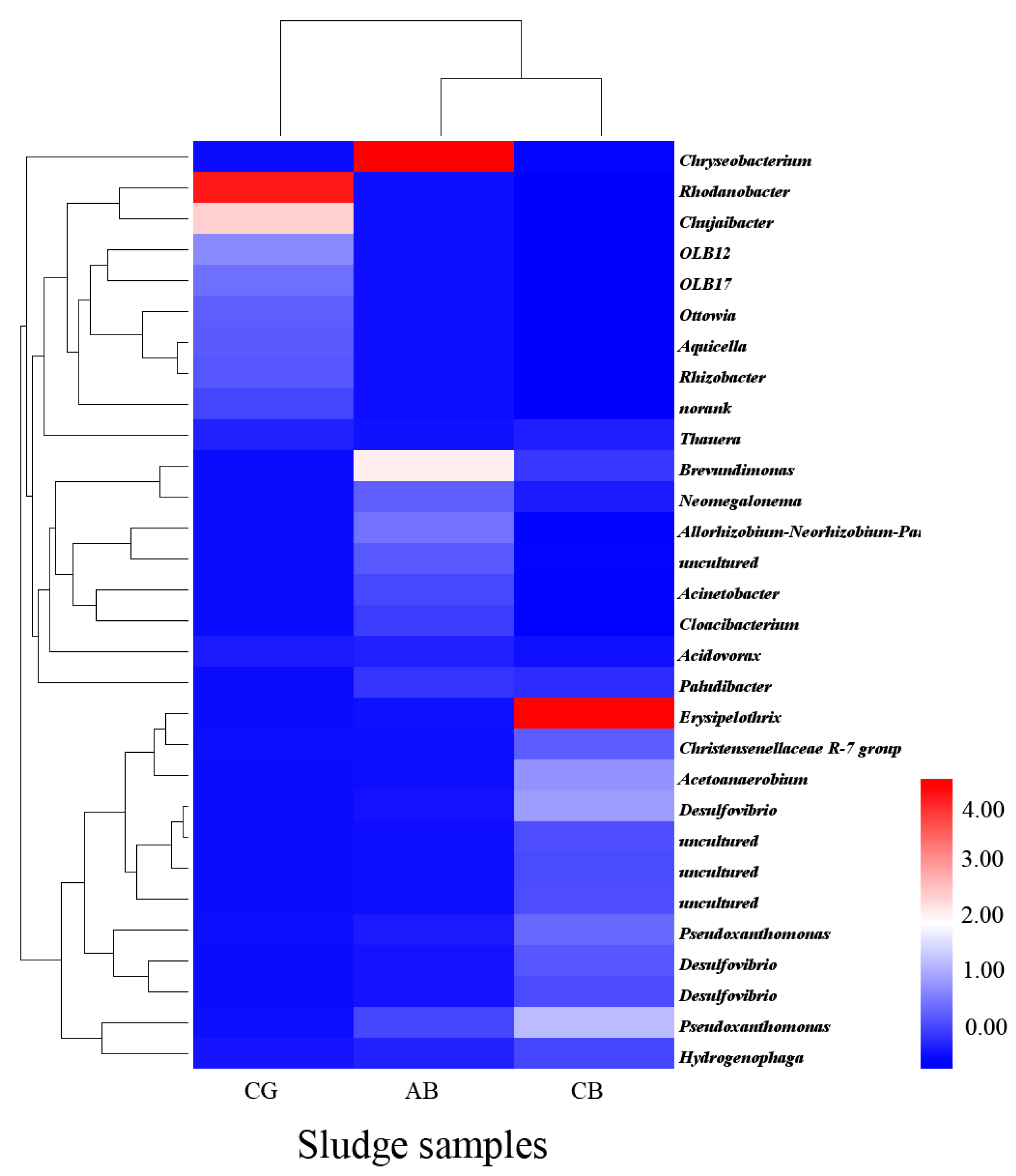
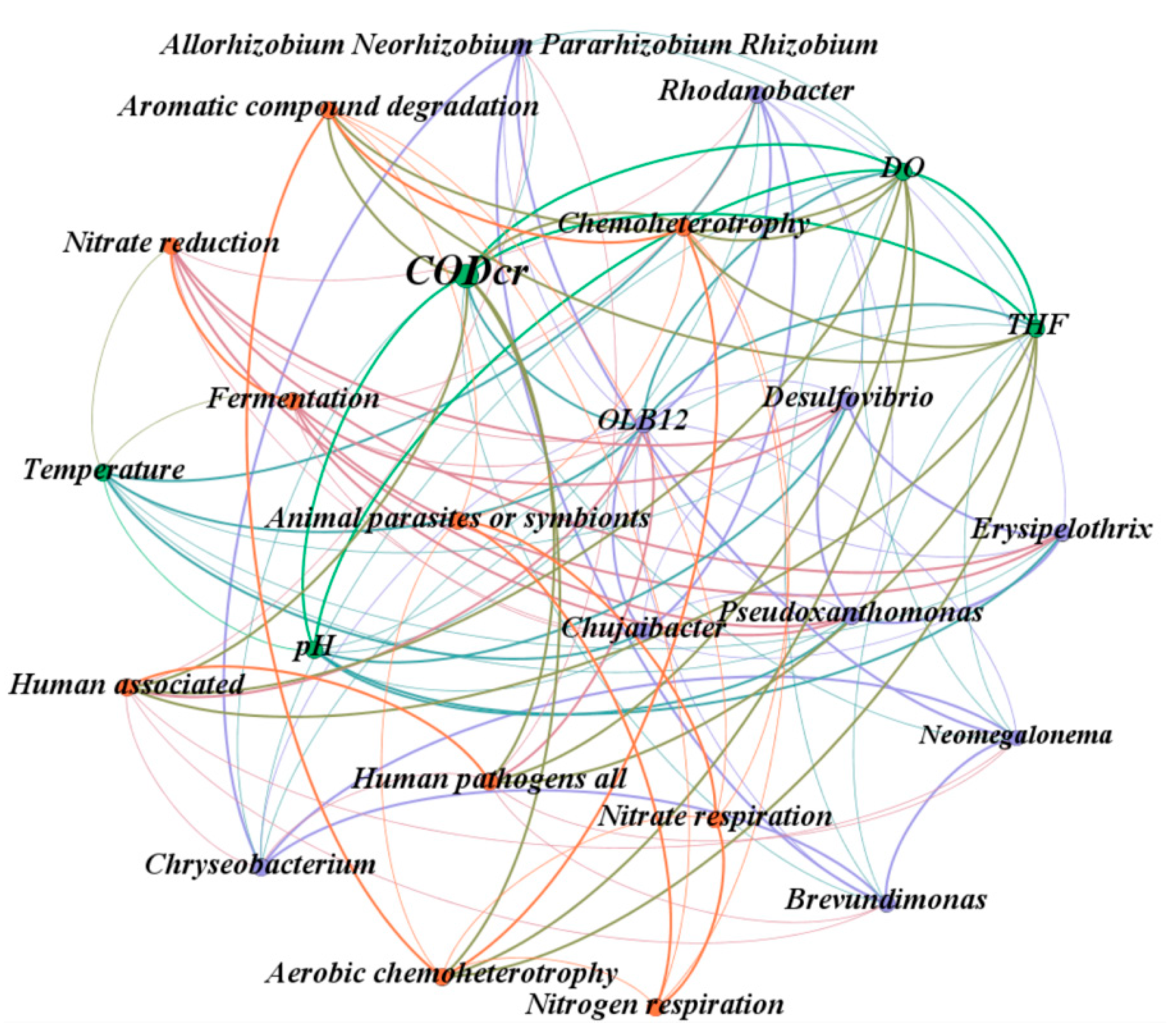
3.3.2. Analysis at the Bacterial Genus Level
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ghazal, H.; Koumaki, E.; Hoslett, J.; Malamis, S.; Katsou, E.; Barcelo, D.; Jouhara, H. Insights into current physical, chemical and hybrid technologies used for the treatment of wastewater contaminated with pharmaceuticals. J. Clean. Prod. 2022, 361, 132079. [Google Scholar] [CrossRef]
- Hu, D.; Li, X.; Chen, Z.; Cui, Y.; Gu, F.; Jia, F.; Xiao, T.; Su, H.; Xu, J.; Wang, H.; et al. Performance and extracellular polymers substance analysis of a pilot scale anaerobic membrane bioreactor for treating tetrahydrofuran pharmaceutical wastewater at different HRTs. J. Hazard. Mater. 2017, 342, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Asad, S.; Amoozegar, M.A.; Pourbabaee, A.A.; Sarbolouki, M.N.; Dastgheib, S.M.M. Decolorization of textile azo dyes by newly isolated halophilic and halotolerant bacteria. Bioresour. Technol. 2007, 98, 2082–2088. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, S.; Ezhil Priya, M.; Boopathy, R.; Velan, M.; Mandal, A.B.; Sekaran, G. Heterocatalytic Fenton oxidation process for the treatment of tannery effluent: Kinetic and thermodynamic studies. Environ. Sci. Pollut. Res. 2012, 19, 1828–1840. [Google Scholar] [CrossRef] [PubMed]
- Klavarioti, M.; Mantzavinos, D.; Kassinos, D. Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ. Int. 2009, 35, 402–417. [Google Scholar] [CrossRef]
- Brown, A.K.; Wong, C.S. Distribution and fate of pharmaceuticals and their metabolite conjugates in a municipal wastewater treatment plant. Water Res. 2018, 144, 774–783. [Google Scholar] [CrossRef]
- Adityosulindro, S.; Barthe, L.; González-Labrada, K.; Jáuregui Haza, U.J.; Delmas, H.; Julcour, C. Sonolysis and sono-Fenton oxidation for removal of ibuprofen in (waste) water. Ultrason Sonochem. 2017, 39, 889–896. [Google Scholar] [CrossRef] [Green Version]
- Shen, B.; Wen, X.; Huang, X. Enhanced removal performance of estriol by a three-dimensional electrode reactor. Chem. Eng. J. 2017, 327, 597–607. [Google Scholar] [CrossRef]
- Zhang, S.; Song, H.L.; Yang, X.L.; Yang, K.Y.; Wang, X.Y. Effect of electrical stimulation on the fate of sulfamethoxazole and tetracycline with their corresponding resistance genes in three-dimensional biofilm-electrode reactors. Chemosphere. 2016, 164, 113–119. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Xie, G.; Pan, Z.; Zhou, X.; Lai, W.; Zheng, L.; Xu, Y. A novel Pb/PbO2 electrodes prepared by the method of thermal oxidation-electrochemical oxidation: Characteristic and electrocatalytic oxidation performance. J. Alloys Compd. 2020, 851, 156834. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Xu, J.; Wu, L.; Ni, B.J. Three-dimensional biofilm electrode reactors (3D-BERs) for wastewater treatment. Bioresour. Technol. 2021, 344, 126274. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Li, X.; Gan, L.; Xu, J. Synergistic effects of electricity and biofilm on Rhodamine B (RhB) degradation in three-dimensional biofilm electrode reactors (3D-BERs). Electrochim. Acta 2018, 290, 165–175. [Google Scholar] [CrossRef]
- Zeyoudi, M.; Altenaiji, E.; Ozer, L.Y.; Ahmed, I.; Yousef, A.F.; Hasan, S.W. Impact of continuous and intermittent supply of electric field on the function and microbial community of wastewater treatment electro-bioreactors. Electrochim. Acta 2015, 181, 271–279. [Google Scholar] [CrossRef]
- Liu, F.; Luo, S.; Wang, H.; Zuo, K.; Wang, L.; Zhang, X.; Liang, P.; Huang, X. Improving wastewater treatment capacity by optimizing hydraulic retention time of dual-anode assembled microbial desalination cell system. Sep. Purif. Technol. 2019, 226, 39–47. [Google Scholar] [CrossRef]
- Feng, Y.; Long, Y.; Wang, Z.; Wang, X.; Shi, N.; Suo, N.; Shi, Y.; Yu, Y. Performance and microbial community of an electric biological integration reactor (EBIR) for treatment of wastewater containing ibuprofen. Bioresour. Technol. 2018, 274, 447–458. [Google Scholar] [CrossRef]
- Chen, M.; Xu, J.; Dai, R.; Wu, Z.; Liu, M.; Wang, Z. Development of a moving-bed electrochemical membrane bioreactor to enhance removal of low-concentration antibiotic from wastewater. Bioresour. Technol. 2019, 293, 122022. [Google Scholar] [CrossRef]
- Tang, Q.; Sheng, Y.; Li, C.; Wang, W.; Liu, X. Simultaneous removal of nitrate and sulfate using an up-flow three-dimensional biofilm electrode reactor: Performance and microbial response. Bioresour. Technol. 2020, 318, 124096. [Google Scholar] [CrossRef]
- Wei, V.; Oleszkiewicz, J.A.; Elektorowicz, M. Nutrient removal in an electrically enhanced membrane bioreactor. Water Sci. Technol. 2009, 60, 3159–3163. [Google Scholar] [CrossRef]
- Liu, S.; Feng, X.; Gu, F.; Li, X.; Wang, Y. Sequential reduction/oxidation of azo dyes in a three-dimensional biofilm electrode reactor. Chemosphere 2017, 186, 287–294. [Google Scholar] [CrossRef]
- Zhao, J.; Li, F.; Cao, Y.; Zhang, X.; Chen, T.; Song, H.; Wang, Z. Microbial extracellular electron transfer and strategies for engineering electroactive microorganisms. Biotechnol. Adv. 2021, 53, 107682. [Google Scholar] [CrossRef]
- Koch, C.; Harnisch, F. What Is the Essence of Microbial Electroactivity? Front. Microbiol. 2016, 7, 1890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glaven, S.M. Bioelectrochemical systems and synthetic biology: More power, more products. Microb. Biotechnol. 2019, 12, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Wie, Y.M.; Lee, Y.-S. Characterization of 1,4-Dioxane Biodegradation by a Microbial Community. Water 2020, 12, 3372. [Google Scholar] [CrossRef]
- Zhang, Z.X.; Wang, B.S.; Xu, Y.B.; Wang, G.Z.; Wang, Y.; Guo, H.J. Degradation of wastewater from traditional Chinese medicine extraction by electro-biological technology and analysis of microbial community. Fine Chem. 2021, 38, 387–394. [Google Scholar] [CrossRef]
- Xu, P.; Hao, R.; Zhang, Y. Feasibility of 3BER-S Process for the Deep Denitrification in Synch with the Removal of PAEs from Reclaimed Water. China Environ. Sci. 2016, 37, 662–667. [Google Scholar] [CrossRef]
- Feng, Y.; Long, Y.Y.; Wang, Z. Construction of three dimensional electrocatalytic biological aerated filter (TDE-BAF) and its degradation efficiency of ibuprofen. New Ser. 2019, 51, 447–458. [Google Scholar] [CrossRef]
- Zhou, Y.Y. Characteristics Mechanism and Removal Process of THE Degradation by Pseudomonas Oleovorans DT4. Ph.D. Dissertation, Zhejiang University of Technology, Hangzhou, China, 2011. [Google Scholar]
- Bernhardt, D.; Diekmann, H. Degradation of dioxane, tetrahydrofuran and other cyclic ethers by an environmental Rhodococcus strain. Appl. Microbiol. Biotechnol. 1991, 36, 120–123. [Google Scholar] [CrossRef]
- Wen, G.; Wang, T.; Huang, T.L.; Wan, Q.Q. Nitrogen Removal Performance and Nitrogen/Carbon Balance of Oligotrophic Aerobic Denitrifiers. Water Sci. Technol. 2020, 41, 2339–2348. [Google Scholar] [CrossRef]
- Wang, K.; Ke, S.Z.; Yuan, H.Z.; Zhu, J.; Li, J.W. Effect of ammonia-nitrogen concentration on microbial community structure in a MBBR process. Environ. Eng. 2020, 38, 119–125. [Google Scholar] [CrossRef]
- Li, H.; Wang, B.; Deng, S.; Dai, J.; Shao, S. Oxygen-containing functional groups on bioelectrode surface enhance expression of c-type cytochromes in biofilm and boost extracellular electron transfer. Bioresour. Technol. 2019, 292, 121995. [Google Scholar] [CrossRef]
- Tang, L.Q.; Su, C.Y.; Zhao, L.J.; Wang, P.F.; Wang, A.L.; Li, F.Q.; Fan, C.P.; Huang, X.Y. Effects of different substrate biochar on the performance and microecology of anaerobic biological treating food waste. China Environ. Sci. 2020, 40, 4831–4840. [Google Scholar] [CrossRef]
- Wang, S.B. The Screening of Electrigens for Microbial Fuel Cell and the Study of Microbial Community Structure. Master’s Thesis, Zhejiang Ocean University, Zhoushan, China, 2017. [Google Scholar]
- Li, W.; Niu, Q.; Zhang, H.; Tian, Z.; Zhang, Y.; Gao, Y.Y.; Li, Y.; Nishimura, O.; Yang, M. UASB treatment of chemical synthesis-based pharmaceutical wastewater containing rich organic sulfur compounds and sulfate and associated microbial characteristics. Chem. Eng. J. 2015, 260, 55–63. [Google Scholar] [CrossRef]
- Zhang, W.; Li, J.; Zhang, Y.; Wu, X.; Zhou, Z.; Huang, Y.; Zhao, Y.; Mishra, S.; Bhatt, P.; Chen, S. Characterization of a novel glyphosate-degrading bacterial species, Chryseobacterium sp. Y16C, and evaluation of its effects on microbial communities in glyphosate-contaminated soil. J. Hazard. Mater. 2022, 432, 128689. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.; Souza, A.C.O.; Constancio, M.T.L.; Alvarenga, D.O.; Pylro, V.S.; Alves, L.M.C.; Varani, A.M. Unraveling a Lignocellulose-Decomposing Bacterial Consortium from Soil Associated with Dry Sugarcane Straw by Genomic-Centered Metagenomics. Microorganisms 2021, 9, 995. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Deng, Y.; Chen, H.; Ye, J.; Zhang, S.; Zhao, J. Enhancement of gaseous o-xylene degradation in a microbial fuel cell by adding Shewanella oneidensis MR-1. Chemosphere 2020, 252, 126571. [Google Scholar] [CrossRef]
- Rashid, R.; Morohoshi, T.; Someya, N.; Ikeda, T. Degradation of N-Acylhomoserine Lactone Quorum Sensing Signaling Molecules by Potato Root Surface-Associated Chryseobacterium Strains. Microbes Environ. 2011, 26, 144–148. [Google Scholar] [CrossRef] [Green Version]
- Wang, P. Cometabolic degradation of 1,4-dioxane by a tetrahydrofuran-growing Arthrobacter sp. WN18. Ecotoxicol. Environ. Saf. 2021, 217, 112206. [Google Scholar] [CrossRef]
- Xu, X.; Wang, J.; Yu, T.; Nian, H.; Zhang, H.; Wang, G.; Li, F. Characterization of a novel aryloxyphenoxypropionate herbicide-hydrolyzing carboxylesterase with R-enantiomer preference from Brevundimonas sp. QPT-2. Process Biochem. 2019, 82, 102–109. [Google Scholar] [CrossRef]
- Wang, J.; Liu, X.; Jiang, X.; Zhang, L.; Hou, C.; Su, G.; Wang, L.; Mu, Y.; Shen, J. Facilitated bio-mineralization of N, N-dimethylformamide in anoxic denitrification system: Long-term performance and biological mechanism. Water Res. 2020, 186, 116306. [Google Scholar] [CrossRef]
- Zhang, R.C.; Chen, C.; Shao, B.; Wang, W.; Xu, X.J.; Ren, N.Q. Electron equilibrium analysis of integrated autotrophic and heterotrophic denitrification process under micro-aerobic conditions. Environ. Sci. 2019, 40, 4128–4135. [Google Scholar] [CrossRef]
- Zhou, X.T. Characteristics and Mechanisms of Microbial Fuel Cell Removing Azide and Ammonia. Ph.D. Dissertation, Harbin Institute of Technology, Harbin, China, 2016. [Google Scholar]
- Guo, N. The fate and Transfer Mechanisms of Antibiotic Resistance Genes During Bioelectrochemical Treatment of Chloramphenicol Wastewater. Ph.D. Dissertation, Shandong University, Jinan, China, 2018. [Google Scholar]
- Xu, H. Study on Treatment of Acid Mine Drainage by Sulfate Reducing Bacteria Strengthened by Microelectrolysis. Master’s Thesis, Liaoning Technical University, Fuxin, China, 2020. [Google Scholar]
- Yang, M.Q. Research on New Technique of Microb Electrochemical Coupling with Anaerobi Aerobic Degrading the Wastewater from Cotton Dyeing Pretreatment. Master’s Thesis, Zhejiang University, Hangzhou, China, 2017. [Google Scholar]
- Paredes, D.; Kuschk, P.; Mbwette, T.S.A. New Aspects of Microbial Nitrogen Transformations in the Context of Wastewater Treatment—A Review. Eng. Life Sci. 2007, 1, 13–25. [Google Scholar] [CrossRef]
- Selvaraj, G.K.; Wang, H.; Zhang, Y.; Tian, Z.; Chai, W.; Lu, H. Class 1 In-Tn5393c array contributed to antibiotic resistance of non-pathogenic Pseudoxanthomonas mexicana isolated from a wastewater bioreactor treating streptomycin. Sci. Total Environ. 2022, 821, 153537. [Google Scholar] [CrossRef] [PubMed]
- Xi, S.; Gao, L.; Liu, W.; Wang, A. Domestic sewage enhancing azo dye wastewater treatment in anaerobic digestionbioelectrochemical system and functional microbial community analysis. Acta Sci. Circumstantiae 2019, 39, 290–300. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, X.; Fan, J.; Zhang, Z.; Ma, Q.; Peng, X. Indigoids Biosynthesis from Indole by Two Phenol-Degrading Strains, Pseudomonas sp. PI1 and Acinetobacter sp. PI2. Appl. Biochem. Biotechnol. 2015, 176, 1263–1276. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Du, F.; Xue, G. Microbial community analysis of anaerobic activated sludge in the process of alcohol wastewater treatment using high throughput sequencing. Acta Sci. Circumstantiae 2016, 36, 4112–4119. [Google Scholar] [CrossRef]
- Cao, S.J.; Deng, C.P.; Li, B.Z.; Dong, X.Q.; Yuan, H.L. Cloacibacterium rupense sp. nov, isolated from freshwater lake sediment. Int. J. Syst. Evol. Microbiol. 2010, 60, 2023–2026. [Google Scholar] [CrossRef] [PubMed]

| Parameter | pH | Temperature (℃) | NH4+-N(mg/L) | CODCr (mg/L) | THF (mg/L) |
|---|---|---|---|---|---|
| Raw wastewater | 5.5~7.0 | 25 ± 5 | 9 ± 1 | 5750 ± 750 | 304 ± 39 |
| Electro-Fenton effluent | 3.0~4.0 | 20 ± 5 | 8 ± 1 | 4190 ± 585 | 195 ± 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Chen, X.; Xu, Y.; Zhang, Z.; Zhang, Y. Three-Dimensional Biofilm Electrode Reactors with Polyurethane Sponge Carrier for Highly Efficient Treatment of Pharmaceuticals Wastewater Containing Tetrahydrofuran. Water 2022, 14, 3792. https://doi.org/10.3390/w14223792
Wang B, Chen X, Xu Y, Zhang Z, Zhang Y. Three-Dimensional Biofilm Electrode Reactors with Polyurethane Sponge Carrier for Highly Efficient Treatment of Pharmaceuticals Wastewater Containing Tetrahydrofuran. Water. 2022; 14(22):3792. https://doi.org/10.3390/w14223792
Chicago/Turabian StyleWang, Baoshan, Xiaojie Chen, Yabing Xu, Zexi Zhang, and Yang Zhang. 2022. "Three-Dimensional Biofilm Electrode Reactors with Polyurethane Sponge Carrier for Highly Efficient Treatment of Pharmaceuticals Wastewater Containing Tetrahydrofuran" Water 14, no. 22: 3792. https://doi.org/10.3390/w14223792
APA StyleWang, B., Chen, X., Xu, Y., Zhang, Z., & Zhang, Y. (2022). Three-Dimensional Biofilm Electrode Reactors with Polyurethane Sponge Carrier for Highly Efficient Treatment of Pharmaceuticals Wastewater Containing Tetrahydrofuran. Water, 14(22), 3792. https://doi.org/10.3390/w14223792