Organic Matter and Heavy Metal Ions Removal from Surface Water in Processes of Oxidation with Ozone, UV Irradiation, Coagulation and Adsorption
Abstract
:1. Introduction
2. Research Materials, Course, and Methodology
2.1. Materials
2.2. Course of Research and Analysis Methodology
3. Results and Discussion
3.1. Water Ozonation
3.2. Coagulation
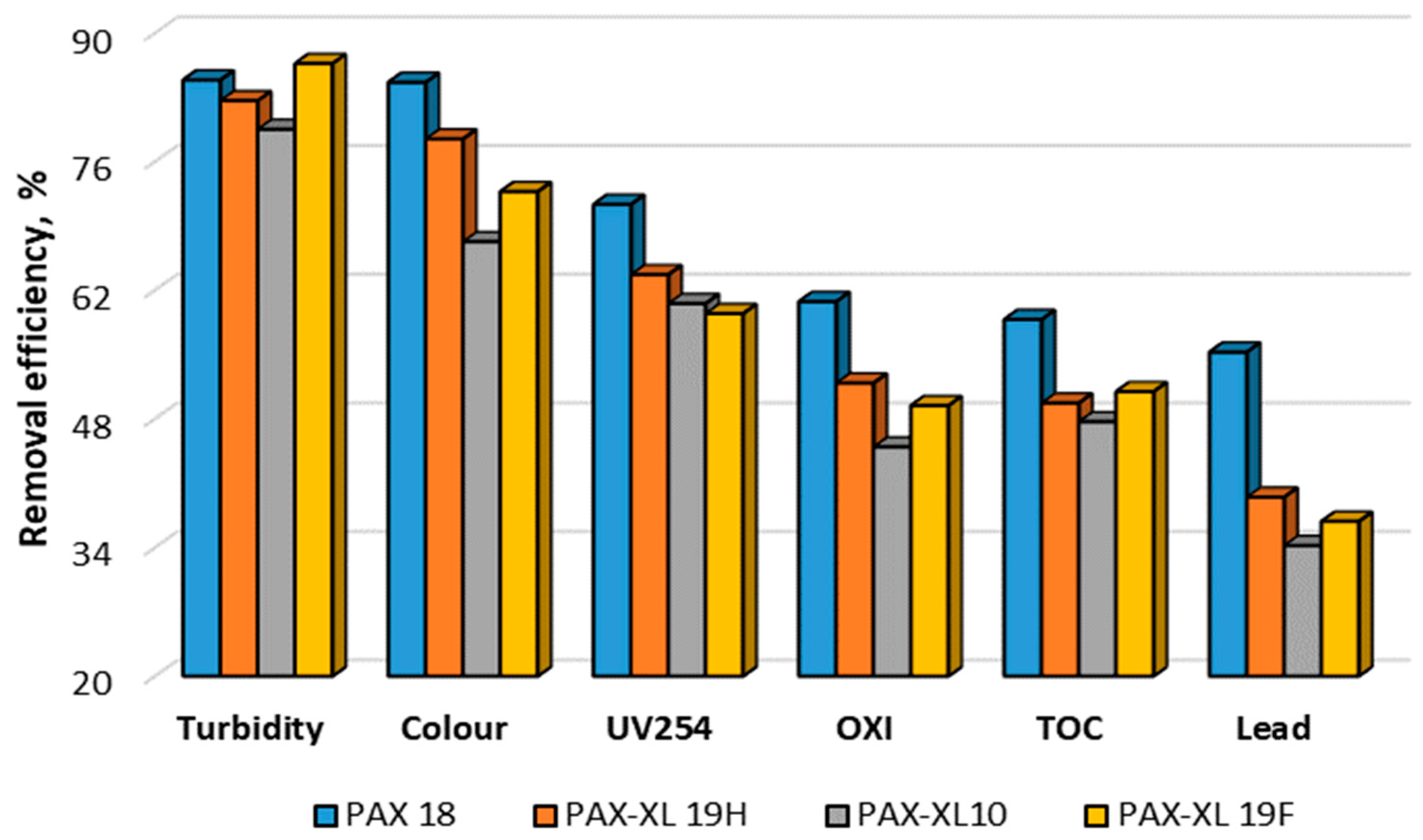
3.3. Adsorption
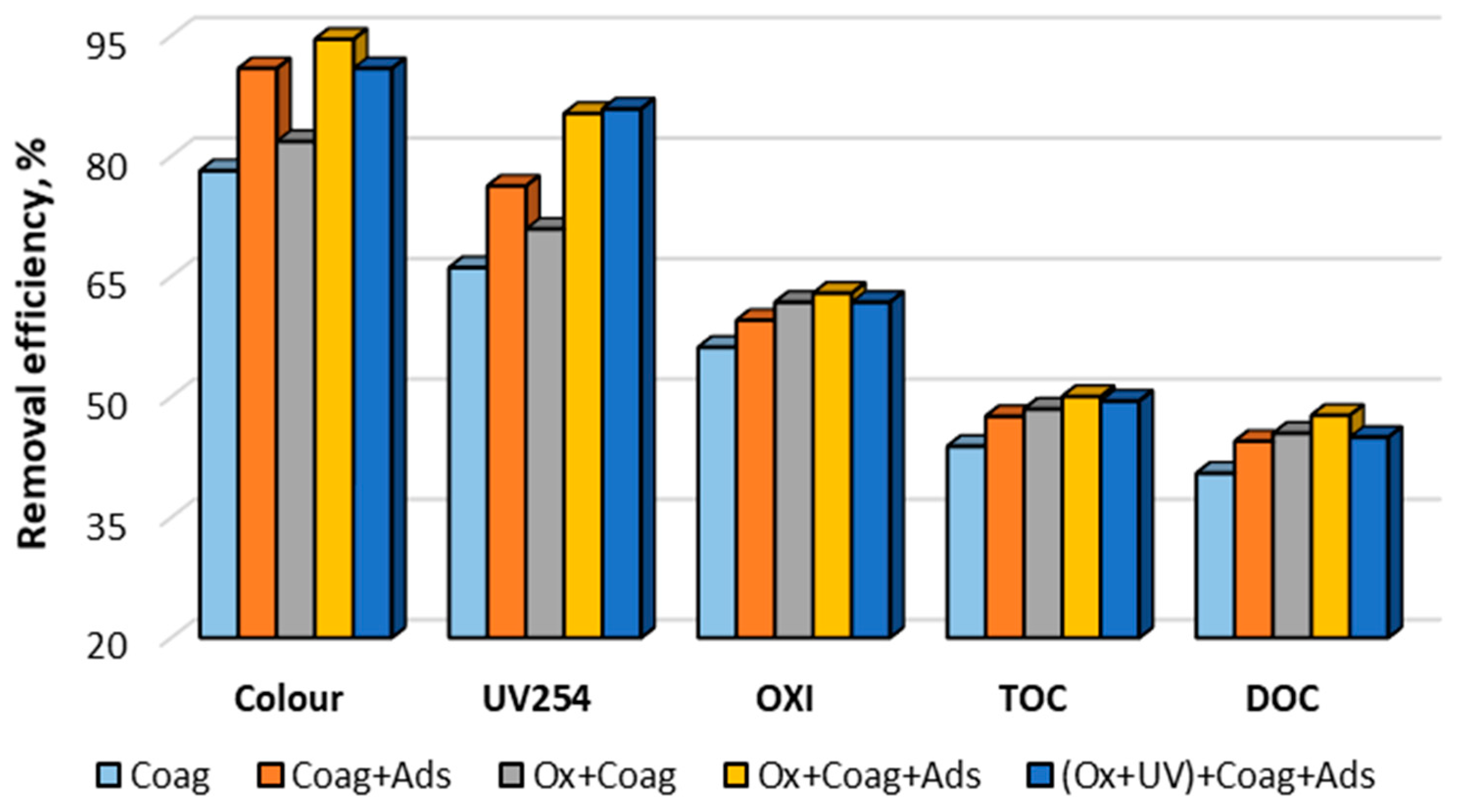
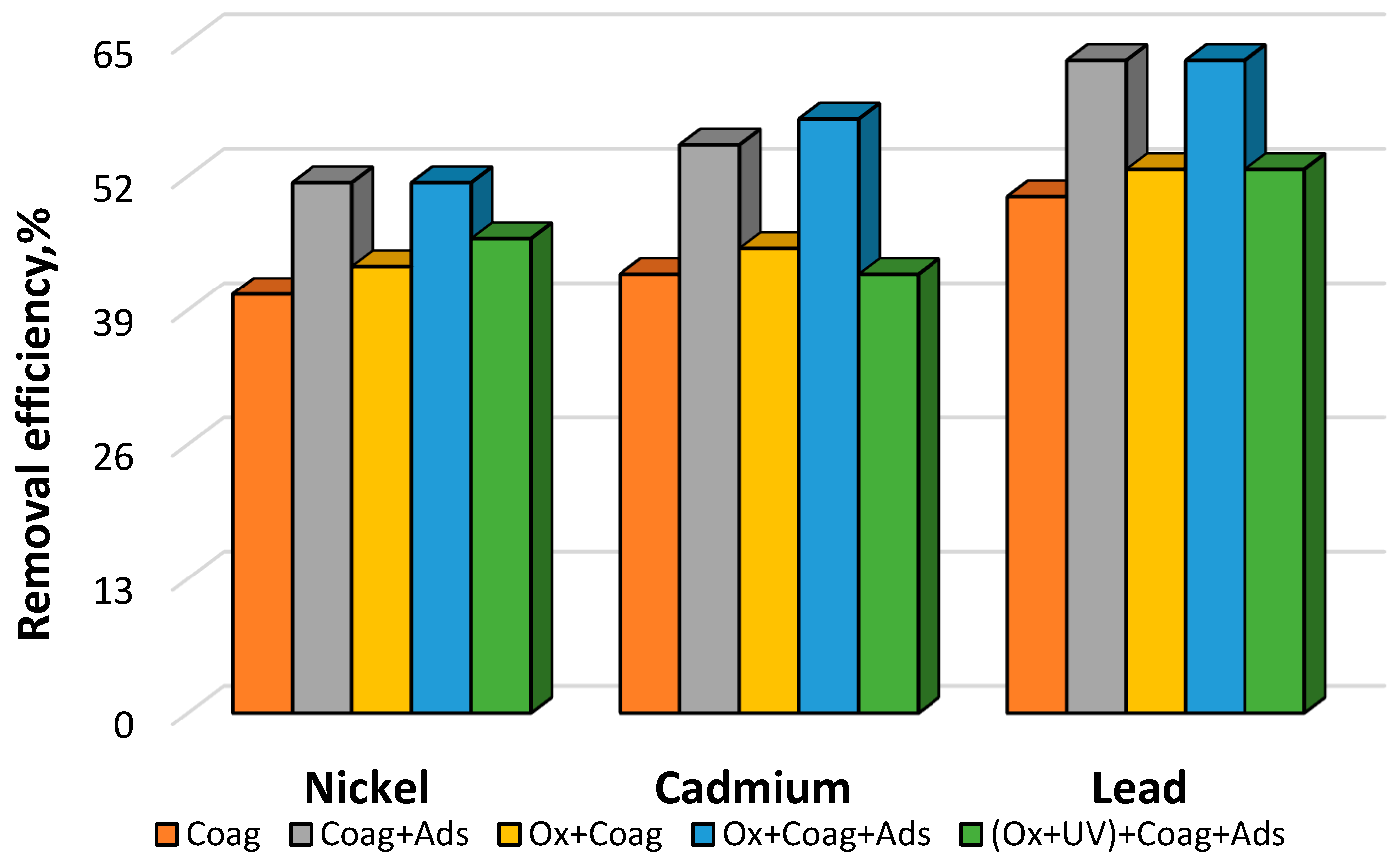
3.4. Ozonation, Coagulation, Adsorption
4. Summary
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Matilainen, A.; Vepsäläinen, M.; Sillanpää, M. Natural organic matter removal by coagulation during drinking water treatment: A review. Adv. Colloid Interface 2010, 159, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Champagne, P.; Mclellan, P.J. Models for 468 predicting disinfection byproduct (DBP) formation in drinking 469 waters: A chronological review. Sci. Total Environ. 2009, 407, 4189. [Google Scholar] [CrossRef]
- Tang, Y.; Long, X.; Wu, M.; Yang, S.; Gao, N.; Xu, B.; Dutta, S. Bibliometric review of research trends on disinfection by-products in drinking water during 1975–2018. Sep. Purif. Technol. 2020, 241, 116741. [Google Scholar] [CrossRef]
- Carrèire, A.; Vachon, M.; Bélisle, J.-L.; Barbeau, B. Supplementing coagulation with powdered activated carbon as a control strategy for trihalomethanes: Application to an existing utility. J. Water Supply Res. Technol. AQUA 2009, 58, 363–371. [Google Scholar] [CrossRef]
- Wang, F.; Gao, B.; Yue, Q.; Bu, F.; Shen, X. Effects of ozonation, powdered activated carbon adsorption, and coagulation on the removal of disinfection by-product precursors in reservoir water. Environ. Sci. Pollut. Res. 2017, 24, 17945–17954. [Google Scholar] [CrossRef] [PubMed]
- Newcombe, G. Removal of organic material and algal metabolites using activated carbon. In Interface Science in Drinking Water Treatment, 1st ed.; Newcombe, G., Dixon, D., Eds.; Elsevier: Oxford, UK, 2006; Volume 10, pp. 133–153. [Google Scholar] [CrossRef]
- Jin, X.; Jin, P.; Hou, R.; Yang, L.; Wang, X.C. Enhanced WWTP effluent organic matter removal in hybrid ozonation-coagulation (HOC) process catalyzed by Al-based coagulant. J. Hazard. Mater. 2017, 327, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Im, D.; Nakada, N.; Fukuma, Y.; Kato, Y.; Tanaka, H. Performance of combined ozonation, coagulation and ceramic membrane process for water reclamation: Effects and mechanism of ozonation on virus coagulation. Sep. Purif. Technol. 2018, 192, 429–434. [Google Scholar] [CrossRef]
- Chiang, P.-C.; Chang, E.-E.; Chang, P.-C.; Huang, C.-P. Effects of pre-ozonation on the removal of THM precursors by coagulation. Sci. Total Environ. 2009, 407, 5735–5742. [Google Scholar] [CrossRef]
- Lim, S.; Shi, J.L.; von Guten, U.; McCurry, D.L. Ozonation of organic compounds and water and wastewater: A critical review. Water Res. 2022, 213, 118053. [Google Scholar] [CrossRef]
- Directive (EU) 2020/2184 of the European Parliament and of the Council of 16 December 2020 on the Quality of Water Intended for Human Consumption. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32020L2184 (accessed on 25 September 2022).
- Regulation of the Minister of Health of 7 December 2017 on the Quality of Water Intended for Human Consumption Dz.U. 2017 poz. 2294. (In Polish). Available online: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU20170002294 (accessed on 25 September 2022).
- Hussain, R.; Wei, C.; Luo, K. Hydrogeochemical characteristics, source identification and health risks of surface water and groundwater in mining and non-mining areas of Handan, China. Environ. Earth Sci. 2019, 78, 402. [Google Scholar] [CrossRef]
- Çiner, F.; Sunkari, E.D.; Şenbaş, B.A. Geochemical and multivariate statistical evaluation of trace elements in groundwater of Niğde municipality, south-central Turkey: Implications for arsenic contamination and human health risks assessment. Arch. Environ. Contam. Toxicol. 2021, 80, 164–182. [Google Scholar] [CrossRef]
- Rakotondrabe, F.; Ngoupayou, J.R.N.; Mfonka, Z.; Rasolomanana, E.H.; Abolo, A.J.N.; Ako, A.A. Water quality assessment Rasolomanana in the Bétaré-Oya gold mining area (East-Cameroon): Multivariate statistical analysis approach. Sci. Total Environ. 2018, 610, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Ren, B.; Hursthouse, A.S.; Jiang, F.; Deng, R.J. Potentially toxic elements (PTEs) in crops, soil, and water near Xiangtan manganese mine, China: Potential risk to health in the food chain. Environ. Geochem. Health 2019, 42, 1965–1976. [Google Scholar] [CrossRef]
- Ewusi, A.; Sunkari, E.D.; Seidu, J.; Coffie-Anum, E. Hydrogeochemical characteristics, sources and human health risk assessment of heavy metal dispersion in the mine pit water–surface water–groundwater system in the largest manganese mine in Ghana. Environ. Technol. Innov. 2022, 26, 102312. [Google Scholar] [CrossRef]
- Wang, L.; Tao, Y.; Su, B.; Wang, L.; Liu, P. Environmental and health risks posed by heavy metal contamination of groundwater in the Sunan coal mine, China. Toxics 2022, 10, 390. [Google Scholar] [CrossRef] [PubMed]
- Bonilla-Petricolet, A.; Mendoza-Castillo, D.I.; Reynel-Ávila, H.E. (Eds.) Adsorption Processes for Water Treatment and Purification; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Zainuddin, N.A.; Mamat, T.A.R.; Imam Maarof, H.; Puasa, S.W.; Mohd Yatim, S.R. Removal of nickel, zinc and copper from plating processes industrial raw effluent via hydroxide precipitation versus sulphide precipitation. IOP Conf. Ser. Mater. Sci. Eng. 2019, 551, 012122. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Xu, Q.; Li, Z.; Pan, B.; Ndagijimana, P.; Wang, Y. Simultaneous removal of cationic heavy metals and arsenic from drinking water by an activated carbon supported nanoscale zero-valent iron and nanosilver composite. Colloids Surf. A Physicochem. Eng. Asp. 2022, 650, 129581. [Google Scholar] [CrossRef]
- Bilal, M.; Ihsanullah, I.; Younas, M.; Ul Hassan Shah, M. Recent advances in applications of low-cost adsorbents for the removal of heavy metals from water: A critical review. Sep. Purif. Technol. 2022, 278, 119510. [Google Scholar] [CrossRef]
- Soon, W.L.; Peydayesh, M.; Mezzenga, R.; Miserez, A. Plant-based amyloids from food waste for removal of heavy metals from contaminated water. Chem. Eng. J. 2022, 445, 136513. [Google Scholar] [CrossRef]
- APHA. AWWA Standard Methods for Examination of Water and Wastewater, 20th ed.; Clesceri, L.S., Greenberg, A.E., Eaton, A.D., Eds.; APHA Publishing House: Washington, DC, USA, 1998. [Google Scholar]
- Zainudin, F.M.; Hasan, H.A.; Abdullah, S.R.S. An overview of the technology used to remove trihalomethane (THM), trihalomethane precursors, and trihalomethane formation potential (THMFP) from water and wastewater. J. Ind. Eng. Chem. 2018, 57, 1–14. [Google Scholar] [CrossRef]
- Sadrnourmohammadi, M.; Brezinski, K.; Gorczyca, B. Ozonation of natural organic matter and aquatic humic substances: The effects of ozone on the structural characteristics and subsequent trihalomethane formation potential. Water Qual. Res. J. 2020, 55, 155–166. [Google Scholar] [CrossRef] [Green Version]
- Szerzyna, S.; Mołczan, M.; Wolska, M.; Adamski, W.; Wiśniewski, J. Absorbance based water quality indicators as parameters for treatment process control with respect to organic substance removal. E3S Web. Conf. 2017, 17, 00091. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Jing, R.; He, S.; Qian, J.; Zhang, K.; Ma, G.; Chang, X.; Zhang, M.; Li, Y. Coagulation of low temperature and low turbidity water: Adjusting basicity of polyaluminium chloride (PAC) and using chitosan as coagulant aid. Sep. Purif. Technol. 2018, 206, 131–139. [Google Scholar] [CrossRef]
- Tubić, A.; Agbaba, J.; Jazić, J.M.; Watson, M.; Dalmacija, B.; Tubi, A.; Jazi, J.M. Pilot scale investigation of coagulation combined with ozonation and pH adjustment in treatment of NOM rich water. Water Supply 2016, 16, 837–844. [Google Scholar] [CrossRef]
- Świetlik, J.; Dąbrowska, A.; Raczyk-Stanisławiak, U.; Nawrocki, J. Reactivity of natural organic matter fractions with chlorine dioxide and ozone. Water Res. 2004, 38, 547–558. [Google Scholar] [CrossRef]
- Matilainen, A.; Gjessing, E.T.; Lahtinen, T.; Hed, L.; Bhatnagar, A.; Sillanpää, M. An overview of the methods used in the characterisation of natural organic matter (NOM) in relation to drinking water treatment. Chemosphere 2011, 83, 1431–1442. [Google Scholar] [CrossRef]
- Jena, P.S.; Pradhan, A.; Nanda, S.P.; Aditya Kishore Dash, A.K.; Naik, B. Biosorption of Heavy Metals from Wastewater Using Saccharomyces Cerevisiae as a Biosorbent: A. mini Review. Mater. Today 2022, in press. [Google Scholar] [CrossRef]

| Parameter | Unit | Coagulant | |||
|---|---|---|---|---|---|
| PAX18 | PAX-XL10 | PAX-XL19H | PAX-XL19F | ||
| Basicity | % | 41 ± 3 | 65 ± 5 | 85 ± 5 | 85 ± 5 |
| OH−/Al3+ | - | 1.23 | 1.95 | 2.55 | 2.55 |
| Aluminum | % | 9.0 ± 0.3 | 5 ± 0.2 | 12.5 ± 0.5 | 8.5 ± 0.3 |
| Chlorides | % | 21.0 ± 1.0 | 11.5 ± 1.0 | 8.5 ± 1 | 5.5 ± 0.5 |
| pH | - | <1.0 | 3.0 ± 0.5 | 3.5 ± 0.4 | 4.0 ± 0.5 |
| Properties | Unit | Powdered Activated Carbon | |
|---|---|---|---|
| CWZ-22 | AKPA-22 | ||
| Specific surface area | m2/g | 960 | 914 |
| Iodine number | mg/g | 1032 | 949 |
| Methylene number | cm3 | 29 | 28 |
| Granulation < 0.06 mm | % | 93 | 98 |
| Parameter | Unit | Modified Surface Water | Treated Water | |||
|---|---|---|---|---|---|---|
| Ozonation–O3 | O3 + UV Irradiation | |||||
| 5 min | 10 min | 15 min | 10 min | |||
| pH | - | 7.95 ± 0.01 | 8.12 ± 0.02 | 8.07 ± 0.01 | 8.03 ± 0.01 | 8.01 ± 0.02 |
| Turbidity | NTU | 6.89 ± 0.03 | 5.91 ± 0.02 | 5.36 ± 0.02 | 5.13 ± 0.02 | 5.11 ± 0.02 |
| Colour | mg Pt/L | 60 ± 3 | 32 ± 3 | 25 ± 2 | 20 ± 2 | 22 ± 2 |
| Absorbance 254 nm 272 nm | 1/cm | 0.298 ± 0.011 0.245 ± 0.012 | 0.188 ± 0.009 0.140 ± 0.010 | 0.116 ± 0.004 0.078 ± 0.010 | 0.071 ± 0.009 0.055 ± 0.007 | 0.049 ± 0.006 0.035 ± 0.008 |
| OXI * | mg O2/L | 7.3 ± 0.2 | 6.2 ± 0.1 | 5.8 ± 0.1 | 5.3 ± 0.1 | 5.4 ± 0.1 |
| TOC * | mg C/L | 7.24 ± 0.05 | 7.08 ± 0.05 | 6.71 ± 0.03 | 6.52 ± 0.04 | 5.96 ± 0.02 |
| DOC * | mg C/L | 7.04 ± 0.04 | 6.96 ± 0.04 | 6.58 ± 0.04 | 6.46 ± 0.02 | 5.70 ± 0.02 |
| Nickel | mg/L | 0.38 ± 0.01 | 0.34 ± 0.02 | 0.34 ± 0.02 | 0.34 ± 0.01 | 0.34 ± 0.01 |
| Cadmium | mg/L | 0.39 ± 0.02 | 0.36 ± 0.01 | 0.35 ± 0.01 | 0.35 ± 0.02 | 0.34 ± 0.01 |
| Lead | mg/L | 0.38 ± 0.03 | 0.36 ± 0.03 | 0.34 ± 0.02 | 0.34 ± 0.02 | 0.33 ± 0.01 |
| Parameter | Unit | Modified Surface Water | Treated Water | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| PAX18 Dose, mg/L | PAX-XL19H Dose, mg/L | |||||||||
| 1.5 | 2.5 | 3.5 | 4.5 | 1.5 | 2.5 | 3.5 | 4.5 | |||
| pH | - | 7.64 ± 0.02 | 7.28 ± 0.01 | 7.14 ± 0.01 | 7.02 ± 0.01 | 6.94 ± 0.02 | 7.54 ± 0.01 | 7.65 ± 0.03 | 7.68 ± 0.01 | 7.66 ± 0.01 |
| Turbidity | NTU | 8.46 ± 0.03 | 6.05 ± 0.02 | 3.04 ± 0.02 | 1.39 ± 0.01 | 1.28 ± 0.01 | 4.49 ± 0.02 | 2.92 ± 0.02 | 2.16 ± 0.02 | 1.47 ± 0.01 |
| Colour | mg Pt/L | 65 ± 3 | 40 ± 3 | 20 ± 2 | 13 ± 1 | 10 ± 1 | 35 ± 2 | 27 ± 2 | 18 ± 1 | 14 ± 1 |
| Absorbance 254 nm 272 nm | 1/cm | 0.314 ± 0.020 0.260 ± 0.020 | 0.173 ± 0.002 0.139 ± 0.020 | 0.140 ± 0.009 0.113 ± 0.009 | 0.106 ± 0.010 0.089 ± 0.010 | 0.090 ± 0.009 0.073 ± 0.011 | 0.201 ± 0.020 0.171 ± 0.008 | 0.150 ± 0.012 0.124 ± 0.008 | 0.133 ± 0.010 0.106 ± 0.008 | 0.114 ± 0.009 0.091 ± 0.008 |
| OXI | mg O2/L | 7.9 ± 0.2 | 6.5 ± 0.1 | 4.8 ± 0.1 | 3.9 ± 0.1 | 3.1 ± 0.1 | 6.1 ± 0.2 | 5.6 ± 0.1 | 4.3 ± 0.1 | 3.8 ± 0.1 |
| TOC | mg C/L | 7.38 ± 0.05 | 6.04 ± 0.04 | 4.66 ± 0.02 | 3.77 ± 0.03 | 3.04 ± 0.02 | 5.69 ± 0.04 | 4.78 ± 0.05 | 4.19 ± 0.03 | 3.71 ± 0.04 |
| DOC | mg /L | 6.96 ± 0.04 | 5.76 ± 0.02 | 4.37 ± 0.04 | 3.53 ± 0.05 | 2.87 ± 0.03 | 5.57 ± 0.03 | 4.74 ± 0.02 | 4.14 ± 0.04 | 3.63 ± 0.03 |
| Aluminum | mg Al/L | 0.01 ± 0.01 | 0.14 ± 0.02 | 0.08 ± 0.01 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.02 ± 0.01 | 0.02 ± 0.01 | <0.01 | <0.01 |
| Nickel | mg/L | 0.38 ± 0.01 | 0.29 ± 0.01 | 0.26 ± 0.01 | 0.24 ± 0.01 | 0.23 ± 0.01 | 0.31 ± 0.02 | 0.29 ± 0.02 | 0.26 ± 0.01 | 0.25 ± 0.01 |
| Cadmium | mg/L | 0.39 ± 0.02 | 0.29 ± 0.01 | 0.26 ± 0.01 | 0.22 ± 0.01 | 0.21 ± 0.01 | 0.30 ± 0.02 | 0.27 ± 0.02 | 0.24 ± 0.01 | 0.24 ± 0.01 |
| Lead | mg/L | 0.38 ± 0.03 | 0.21 ± 0.01 | 0.19 ± 0.01 | 0.17 ± 0.01 | 0.17 ± 0.02 | 0.26 ± 0.01 | 0.25 ± 0.01 | 0.23 ± 0.01 | 0.23 ± 0.02 |
| Parameter | Unit | Modified Surface Water | Treated Water | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| PAX-XL10 Dose, mg/L | PAX-XL19F Dose, mg/L | |||||||||
| 1.5 | 2.5 | 3.5 | 4.5 | 1.5 | 2.5 | 3.5 | 4.5 | |||
| pH | - | 7.26 ± 0.01 | 7.25 ± 0.01 | 7.22 ± 0.02 | 7.19 ± 0.01 | 7.13 ± 0.01 | 7.39 ± 0.02 | 7.43 ± 0.01 | 7.42 ± 0.01 | 7.41 ± 0.02 |
| Turbidity | NTU | 12.30 ± 0.08 | 8.22 ± 0.04 | 5.78 ± 0.04 | 3.62 ± 0.02 | 2.52 ± 0.04 | 6.45 ± 0.04 | 4.82 ± 0.02 | 1.93 ± 0.02 | 1.64 ± 0.01 |
| Colour | mg Pt/L | 55 ± 3 | 40 ± 3 | 35 ± 3 | 25 ± 2 | 18 ± 1 | 40 ± 3 | 30 ± 2 | 20 ± 2 | 15 ± 1 |
| Absorbance 254 nm 272 nm | 1/cm | 0.365 ± 0.005 0.296 ± 0.008 | 0.253 ± 0.009 0.198 ± 0.007 | 0.191 ± 0.010 0.155 ± 0.011 | 0.166 ± 0.007 0.131 ± 0.010 | 0.144 ± 0.006 0.113 ± 0.008 | 0.225 ± 0.012 0.182 ± 0.006 | 0.207 ± 0.008 0.165 ± 0.009 | 0.161 ± 0.007 0.127 ± 0.006 | 0.148 ± 0.006 0.118 ± 0.008 |
| OXI | mg O2/L | 8.9 ± 0.2 | 7.8 ± 0.1 | 7.3 ± 0.1 | 6.0 ± 0.2 | 4.9 ± 0.1 | 7.0 ± 0.1 | 6.5 ± 0.2 | 5.3 ± 0.1 | 4.5 ± 0.1 |
| TOC | mg C/L | 8.85 ± 0.05 | 6.65 ± 0.04 | 6.04 ± 0.04 | 5.10 ± 0.07 | 4.63 ± 0.06 | 6.23 ± 0.03 | 5.62 ± 0.08 | 4.87 ± 0.08 | 4.34 ± 0.04 |
| DOC | mg C/L | 8.05 ± 0.04 | 6.41 ± 0.06 | 5.78 ± 0.08 | 4.88 ± 0.08 | 4.37 ± 0.05 | 6.08 ± 0.06 | 5.35 ± 0.06 | 4.57 ± 0.07 | 4.20 ± 0.02 |
| Aluminum | mg Al/L | 0.01 ± 0.01 | 0.16 ± 0.2 | 0.15 ± 0.02 | 0.12 ± 0.2 | 0.10 ± 0.01 | 0.08 ± 0.01 | 0.06 ± 0.01 | <0.01 | <0.01 |
| Nickel | mg/L | 0.37 ± 0.01 | 0.35 ± 0.01 | 0.34 ± 0.01 | 0.32 ± 0.01 | 0.28 ± 0.01 | 0.28 ± 0.02 | 0.27 ± 0.01 | 0.25 ± 0.01 | 0.25 ± 0.01 |
| Cadmium | mg/L | 0.40 ± 0.01 | 0.33 ± 0.01 | 0.30 ± 0.01 | 0.29 ± 0.02 | 0.27 ± 0.01 | 0.30 ± 0.01 | 0.30 ± 0.02 | 0.27 ± 0.01 | 0.27 ± 0.01 |
| Lead | mg/L | 0.38 ± 0.03 | 0.33 ± 0.01 | 0.30 ± 0.02 | 0.27 ± 0.01 | 0.25 ± 0.02 | 0.27 ± 0.01 | 0.27 ± 0.01 | 0.27 ± 0.01 | 0.24 ± 0.03 |
| Parameter | Unit | Modified Surface Water | Treated Water | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AKPA-22 Dose, mg/L | CWZ-22 Dose, mg/L | |||||||||
| 30 | 60 | 90 | 120 | 30 | 60 | 90 | 120 | |||
| pH | - | 7.74 ± 0.02 | 7.28 ± 0.01 | 7.14 ± 0.02 | 7.02 ± 0.02 | 6.94 ± 0.01 | 7.54 ± 0.02 | 7.65 ± 0.01 | 7.68 ± 0.01 | 7.66 ± 0.01 |
| Colour | mg Pt/L | 65 ± 3 | 40 ± 3 | 40 ± 3 | 37 ± 3 | 30 ± 2 | 37 ± 3 | 35 ± 3 | 27 ± 2 | 22 ± 2 |
| Absorbance 254 nm 272 nm | 1/cm | 0.318 ± 0.011 0.254 ± 0.011 | 0.316 ± 0.011 0.252 ± 0.011 | 0.296 ± 0.008 0.240 ± 0.006 | 0.293 ± 0.006 0.236 ± 0.008 | 0.286 ± 0.011 0.227 ± 0.010 | 0.277 ± 0.008 0.223 ± 0.006 | 0.270 ± 0.010 0.219 ± 0.011 | 0.232 ± 0.009 0.186 ± 0.006 | 0.212 ± 0.004 0.171 ± 0.006 |
| OXI | mg O2/L | 7.9 ± 0.05 | 7.7 ± 0.2 | 7.6 ± 0.1 | 7.4 ± 0.2 | 6.9 ± 0.1 | 7.4 ± 0.2 | 7.3 ± 0.1 | 6.5 ± 0.1 | 5.9 ± 0.1 |
| TOC | mg C/L | 7.91 ± 0.06 | 7.12 ± 0.10 | 6.88 ± 0.04 | 6.64 ± 0.06 | 6.44 ± 0.06 | 6.68 ± 0.04 | 6.42 ± 0.08 | 6.17 ± 0.04 | 5.87 ± 0.06 |
| DOC | mg C/L | 7.22 ± 0.09 | 6.48 ± 0.10 | 6.21 ± 0.08 | 6.11 ± 0.07 | 5.94 ± 0.08 | 6.14 ± 0.02 | 5.87 ± 0.07 | 5.63 ± 0.02 | 5.42 ± 0.09 |
| Nickel | mg/L | 0.38 ± 0.01 | 0.22 ± 0.01 | 0.19 ± 0.01 | 0.18 ± 0.02 | 0.18 ± 0.01 | 0.24 ± 0.01 | 0.23 ± 0.01 | 0.21 ± 0.02 | 0.21 ± 0.01 |
| Cadmium | mg/L | 0.39 ± 0.02 | 0.21 ± 0.02 | 0.19 ± 0.01 | 0.18 ± 0.01 | 0.16 ± 0.02 | 0.22 ± 0.02 | 0.21 ± 0.01 | 0.18 ± 0.01 | 0.17 ± 0.01 |
| Lead | mg/L | 0.38 ± 0.01 | 0.21 ± 0.01 | 0.18 ± 0.01 | 0.17 ± 0.02 | 0.16 ± 0.01 | 0.20 ± 0.01 | 0.20 ± 0.03 | 0.18 ± 0.01 | 0.17 ± 0.01 |
| Parameter | Unit | Modified Surface Water | Treated Water | ||||
|---|---|---|---|---|---|---|---|
| Coag | Coag + Ads | Ox + Coag | Ox + Coag + Ads | (Ox + UV) + Coag + Ads | |||
| pH | - | 7.08 ± 0.01 | 6.75 ± 0.01 | 6.80 ± 0.03 | 6.84 ± 0.01 | 6.84 ± 0.01 | 6.82 ± 0.01 |
| Turbidity | NTU | 12.80 ± 0.03 | 1.39 ± 0.02 | 2.08 ± 0.02 | 1.28 ± 0.02 | 2.62 ± 0.03 | 1.33 ± 0.02 |
| Colour | mg Pt/L | 55 ± 3 | 12 ± 1 | 5 ± 1 | 10 ± 1 | 3 ± 1 | 5 ± 1 |
| Absorbance 254 nm 272 nm | 1/cm | 0.354 ± 0.020 0.289 ± 0.020 | 0.120 ± 0.010 0.099 ± 0.009 | 0.084 ± 0.010 0.070 ± 0.009 | 0.103 ± 0.010 0.083 ± 0.010 | 0.052 ± 0.010 0.041 ± 0.009 | 0.050 ± 0.010 0.039 ± 0.008 |
| OXI | mg O2/L | 8.9 ± 0.2 | 3.9 ± 0.1 | 3.6 ± 0.1 | 3.4 ± 0.1 | 3.3 ± 0.1 | 3.4 ± 0.1 |
| TOC | mg C/L | 8.85 ± 0.05 | 4.97 ± 0.04 | 4.64 ± 0.05 | 4.56 ± 0.05 | 4.42 ± 0.06 | 4.47 ± 0.04 |
| DOC | mg C/L | 8.05 ± 0.05 | 4.79 ± 0.06 | 4.47 ± 0.05 | 4.39 ± 0.04 | 4.21 ± 0.05 | 4.43 ± 0.04 |
| Aluminum | mg Al/L | <0.01 | 0.05 ± 0.01 | 0.02 ± 0.01 | 0.02 ± 0.01 | <0.01 | <0.01 |
| Nickel | mg/L | 0.37 ± 0.02 | 0.22 ± 0.02 | 0.18 ± 0.01 | 0.21 ± 0.01 | 0.18 ± 0.01 | 0.20 ± 0.01 |
| Cadmium | mg/L | 0.40 ± 0.01 | 0.23 ± 0.01 | 0.18 ± 0.02 | 0.22 ± 0.02 | 0.17 ± 0.01 | 0.23 ± 0.01 |
| Lead | mg/L | 0.38 ± 0.02 | 0.19 ± 0.01 | 0.14 ± 0.02 | 0.18 ± 0.01 | 0.14 ± 0.02 | 0.18 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karwowska, B.; Sperczyńska, E. Organic Matter and Heavy Metal Ions Removal from Surface Water in Processes of Oxidation with Ozone, UV Irradiation, Coagulation and Adsorption. Water 2022, 14, 3763. https://doi.org/10.3390/w14223763
Karwowska B, Sperczyńska E. Organic Matter and Heavy Metal Ions Removal from Surface Water in Processes of Oxidation with Ozone, UV Irradiation, Coagulation and Adsorption. Water. 2022; 14(22):3763. https://doi.org/10.3390/w14223763
Chicago/Turabian StyleKarwowska, Beata, and Elżbieta Sperczyńska. 2022. "Organic Matter and Heavy Metal Ions Removal from Surface Water in Processes of Oxidation with Ozone, UV Irradiation, Coagulation and Adsorption" Water 14, no. 22: 3763. https://doi.org/10.3390/w14223763






