Application of Natural Coagulants in Water Treatment: A Sustainable Alternative to Chemicals
Abstract
:1. Introduction
- Physical data: Thes are water that need to be monitored during the treatment process, which include total suspended particles, temperature, conductivity, clarity, total dissolved solids, etc.
- Chemical data: Chemical water-quality metrics of the national comprehensive discharge standard for contaminants in water, such as pH, biochemical oxygen demand, biochemical oxygen consumption, heavy metals, nitrates, etc.
- Biological data: Waterborne microorganisms such as E. coli, mayflies, and other microbes are examples of biomarkers.
1.1. The Concept of Water Treatment
1.2. Factors Affecting WT
2. Strategies of Water Treatments
2.1. Water Treatment Using Chemical Coagulants
2.2. Emerging Use of Natural Coagulants
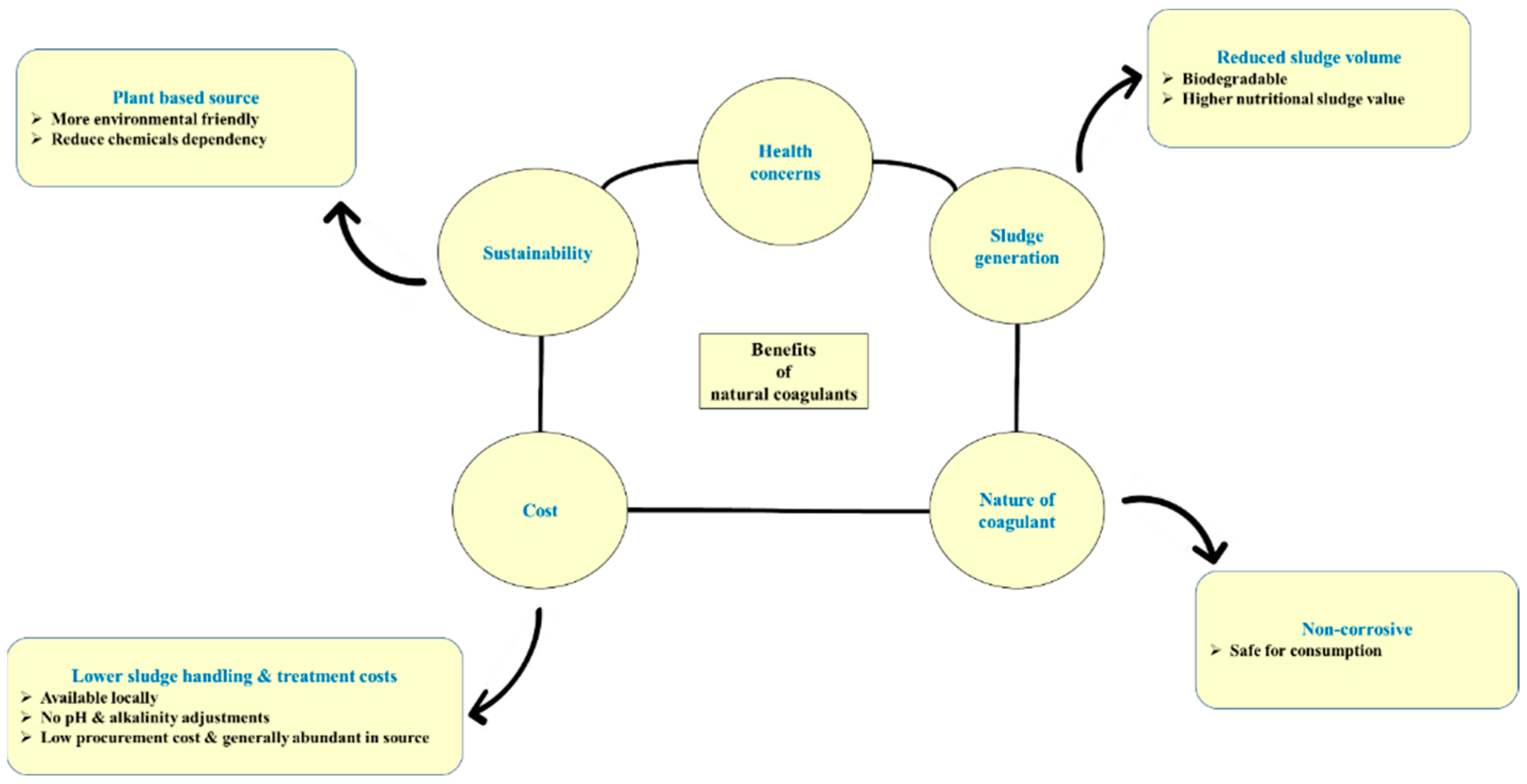
2.2.1. Sustainability of Natural Coagulants
2.2.2. Plant-Based Coagulants
2.2.3. Tannins
2.2.4. Nirmali Seeds
2.2.5. Moringa Seeds
2.2.6. Chickpea Seeds
2.2.7. Peanut Seeds
2.2.8. Soybean Seeds
2.2.9. Cacti Mucilage
2.2.10. Okra Seed Extract
2.3. Animal-Based Coagulants
3. Extraction of Natural Coagulants
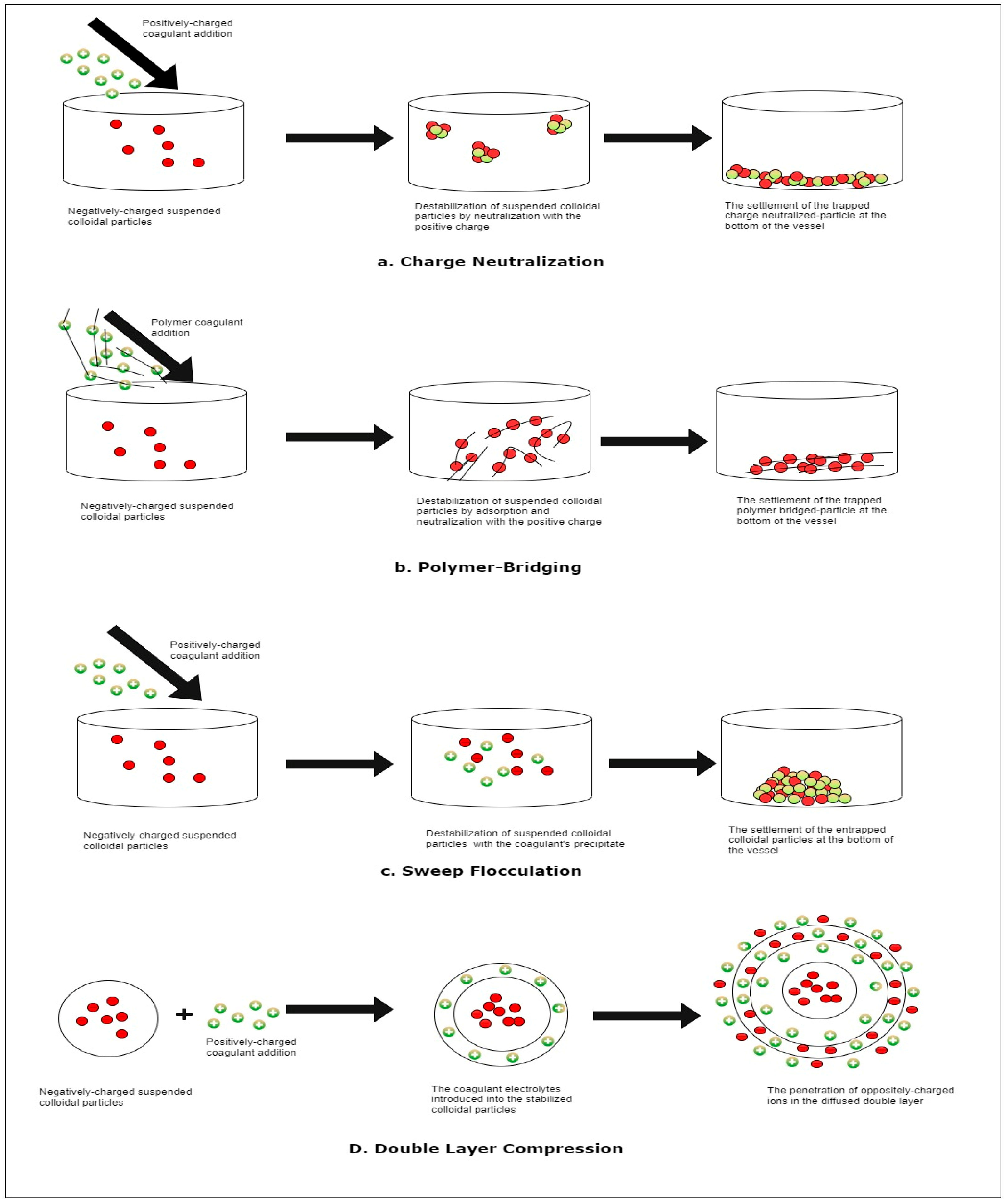
4. Mechanism of Action of Natural Coagulants
5. Natural Coagulants—Constraints in Commercialization
6. Future Prospects of Natural Coagulants
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gleick, P.H. Water in Crisis; Oxford University Press: Oxford, UK; New York, NY, USA, 1993; Volume 100. [Google Scholar]
- Liu, S.X. Food and Agricultural Wastewater Utilization and Treatment; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- World Health Organization. Evaluating Household Water Treatment Options: Health-Based Targets and Microbiological Performance Specifications; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Khatri, N.; Tyagi, S. Influences of natural and anthropogenic factors on surface and groundwater quality in rural and urban areas. Front. Life Sci. 2015, 8, 23–39. [Google Scholar] [CrossRef]
- Murray, J.J.; World Health Organization. Appropriate Use of Fluorides for Human Health; World Health Organization: Geneva, Switzerland, 1986. [Google Scholar]
- Loganathan, P.; Hedley, M.; Grace, N.; Lee, J.; Cronin, S.; Bolan, N.; Zanders, J. Fertiliser contaminants in new zealand grazed pasture with special reference to cadmium and fluorine—A review. Soil Res. 2003, 41, 501–532. [Google Scholar] [CrossRef]
- Jagtap, S.; Yenkie, M.K.; Labhsetwar, N.; Rayalu, S. Fluoride in drinking water and defluoridation of water. Chem. Rev. 2012, 112, 2454–2466. [Google Scholar] [CrossRef] [PubMed]
- Mandinic, Z.; Curcic, M.; Antonijevic, B.; Carevic, M.; Mandic, J.; Djukic-Cosic, D.; Lekic, C.P. Fluoride in drinking water and dental fluorosis. Sci. Total Environ. 2010, 408, 3507–3512. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, N.; Sirisha, D.; Smita, A.; Manjusha, A. Adsorption studies of fluoride on multani matti and red soil. Res. J. Chem. Sci. 2012, 2, 32–37. [Google Scholar]
- Nand, V.; Maata, M.; Koshy, K.; Sotheeswaran, S. Water purification using Moringa oleifera and other locally available seeds in fiji for heavy metal removal. Int. J. Appl. 2012, 2, 125–129. [Google Scholar]
- Ravikumar, K.; Sheeja, A. Heavy metal removal from water using moringa oleifera seed coagulant and double filtration. In Proceedings of the International Conference on Innovations in Civil Engineering Icice-2013, Kochi, India, 9–10 May 2013; p. 9. [Google Scholar]
- Aziz, N.; Jayasuriya, N.; Fan, L. Effectiveness of plant-based indigenous materials for the removal of heavy metals and fluoride from drinking water. In Proceedings of the 5th International Conference on Sustainable Built Environment Proceedings, Kandy, Sri Lanka, 16–18 December 2014; pp. 34–41. [Google Scholar]
- Dubrovsky, N.M.; Burow, K.R.; Clark, G.M.; Gronberg, J.; Hamilton, P.A.; Hitt, K.J.; Mueller, D.K.; Munn, M.D.; Nolan, B.T.; Puckett, L.J. The quality of our nation’s waters—Nutrients in the nation’s streams and groundwater, 1992–2004. US Geol. Surv. Circ. 2010, 1350, 174. [Google Scholar]
- Welch, J.; Simmons, V.; Meléndez, E.; Sees, M.; Gold, Y.; Heider, E.C. Assessment of non-anthropogenic addition of uric acid to a water treatment wetlands. Environments 2020, 7, 60. [Google Scholar] [CrossRef]
- Lovell, S.T.; Sullivan, W.C. Environmental benefits of conservation buffers in the United States: Evidence, promise, and open questions. Agric. Ecosyst. Environ. 2006, 112, 249–260. [Google Scholar] [CrossRef]
- Wallage, Z.E.; Holden, J.; McDonald, A.T. Drain blocking: An effective treatment for reducing dissolved organic carbon loss and water discolouration in a drained peatland. Sci. Total Environ. 2006, 367, 811–821. [Google Scholar] [CrossRef]
- Dorioz, J.-M.; Wang, D.; Poulenard, J.; Trevisan, D. The effect of grass buffer strips on phosphorus dynamics—A critical review and synthesis as a basis for application in agricultural landscapes in France. Agric. Ecosyst. Environ. 2006, 117, 4–21. [Google Scholar] [CrossRef]
- Lloyd, M. Scottish environmental protection agency: Making sense of a fragmenting environment. Scott. Aff. 1999, 29, 28–42. [Google Scholar] [CrossRef]
- Garrod, G.D.; Garratt, J.A.; Kennedy, A.; Willis, K.G. A mixed methodology framework for the assessment of the voluntary initiative. Pest Manag. Sci. Former. Pestic. Sci. 2007, 63, 157–170. [Google Scholar] [CrossRef]
- Smith, V.H.; Tilman, G.D.; Nekola, J.C. Eutrophication: Impacts of excess nutrient inputs on freshwater, marine, and terrestrial ecosystems. Environ. Pollut. 1999, 100, 179–196. [Google Scholar] [CrossRef]
- Bricker, S.; Longstaff, B.; Dennison, W.; Jones, A.; Boicourt, K.; Wicks, C.; Woerner, J. Effects of nutrient enrichment in the nation’s estuaries: A decade of change (national estuarine eutrophication update). NOAA Coast. Ocean Program Decis. Anal. Ser. 2007, 8, 21–23. [Google Scholar]
- Freeman, A.M.; Lamon, E.C., III; Stow, C.A. Nutrient criteria for lakes, ponds, and reservoirs: A bayesian treed model approach. Ecol. Modell. 2009, 220, 630–639. [Google Scholar] [CrossRef]
- Blanchard, P.; Lerch, R. Watershed vulnerability to losses of agricultural chemicals: Interactions of chemistry, hydrology, and land-use. Environ. Sci. Technol. 2000, 34, 3315–3322. [Google Scholar] [CrossRef]
- Avid Hirst, D.; ob Morris, R. Water quality of scottish rivers: Spatial and temporal trends. Sci. Total Environ. 2001, 265, 327–342. [Google Scholar]
- Koul, B.; Taak, P. Biotechnological Strategies for Effective Remediation of Polluted Soils; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Miller, T.H.; Bury, N.R.; Owen, S.F.; MacRae, J.I.; Barron, L.P. A review of the pharmaceutical exposome in aquatic fauna. Environ. Pollut. 2018, 239, 129–146. [Google Scholar] [CrossRef]
- Connor, R. The United Nations World Water Development Report 2015: Water for a Sustainable World; UNESCO Publishing: Paris, France, 2015; Volume 1. [Google Scholar]
- Eman, N.A.; Suleyman, A.M.; Hamzah, M.S.; Md Zahangir, A.; Mohd Ramlan, M.S. Production of natural coagulant from moringa oleifera seed for application in treatment of low turbidity water. J. Water Resour. Prot. 2010, 2010, 259–266. [Google Scholar]
- Abdulredha, M.; Rafid, A.L.; Jordan, D.; Hashim, K. The development of a waste management system in kerbala during major pilgrimage events: Determination of solid waste composition. Procedia Eng. 2017, 196, 779–784. [Google Scholar] [CrossRef]
- Hashim, K.S.; Al Khaddar, R.; Jasim, N.; Shaw, A.; Phipps, D.; Kot, P.; Pedrola, M.O.; Alattabi, A.W.; Abdulredha, M.; Alawsh, R. Electrocoagulation as a green technology for phosphate removal from river water. Sep. Purif. Technol. 2019, 210, 135–144. [Google Scholar] [CrossRef]
- Mohammed, A.-H.; Hussein, A.H.; Yeboah, D.; Al Khaddar, R.; Abdulhadi, B.; Shubbar, A.A.; Hashim, K.S. Electrochemical Removal of Nitrate from Wastewater. IOP Conf. Ser. Mater. Sci. Eng. 2020, 888, 12037. [Google Scholar]
- Idowu, I.A.; Atherton, W.; Hashim, K.; Kot, P.; Alkhaddar, R.; Alo, B.I.; Shaw, A. An analysis of the status of landfill classification systems in developing countries: Sub-saharan africa landfill experiences. Waste Manag. 2019, 87, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Abdulredha, M.; Kot, P.; Al Khaddar, R.; Jordan, D.; Abdulridha, A. Investigating municipal solid waste management system performance during the arba’een event in the city of kerbala, iraq. Environ. Dev. Sustain. 2020, 22, 1431–1454. [Google Scholar] [CrossRef] [Green Version]
- Arora, P. Physical, chemical and biological characteristics of water (e content module). Cent. Univ. Punjab 2017, 2018, 1–16. [Google Scholar]
- Zhou, M.; Zhang, Y.; Wang, J.; Shi, Y.; Puig, V. Water quality indicator interval prediction in wastewater treatment process based on the improved bes-lssvm algorithm. Sensors 2022, 22, 422. [Google Scholar] [CrossRef]
- Alenazi, M.; Hashim, K.S.; Hassan, A.A.; Muradov, M.; Kot, P.; Abdulhadi, B. Turbidity removal using natural coagulants derived from the seeds of Strychnos potatorum: Statistical and experimental approach. IOP Conf. Ser. Mater. Sci. Eng. 2020, 888, 12064. [Google Scholar] [CrossRef]
- Shubbar, A.A.; Sadique, M.; Shanbara, H.K.; Hashim, K. The Development of a New Low Carbon Binder for Construction as an Alternative to Cement. In Advances in Sustainable Construction Materials and Geotechnical Engineering; Springer: Berlin/Heidelberg, Germany, 2020; pp. 205–213. [Google Scholar]
- Abdulraheem, F.S.; Al-Khafaji, Z.S.; Hashim, K.S.; Muradov, M.; Kot, P.; Shubbar, A.A. Natural filtration unit for removal of heavy metals from water. IOP Conf. Ser. Mater. Sci. Eng. 2020, 888, 12034. [Google Scholar] [CrossRef]
- Alhendal, M.; Nasir, M.J.; Hashim, K.S.; Amoako-Attah, J.; Al-Faluji, D.; Muradov, M.; Kot, P.; Abdulhadi, B. Cost-effective hybrid filter for remediation of water from fluoride. IOP Conf. Ser. Mater. Sci. Eng. 2020, 1058, 12012. [Google Scholar] [CrossRef]
- Abdulla, G.; Kareem, M.M.; Hashim, K.S.; Muradov, M.; Kot, P.; Mubarak, H.A.; Abdellatif, M.; Abdulhadi, B. Removal of iron from wastewater using a hybrid filter. IOP Conf. Ser. Mater. Sci. Eng. 2020, 888, 12035. [Google Scholar] [CrossRef]
- Shabaa, G.J.; Al-Jboory, W.S.; Sabre, H.M.; Alazmi, A.; Kareem, M.M.; AlKhayyat, A. Plant-based coagulants for water treatment. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1058, 12001. [Google Scholar] [CrossRef]
- Azizul-Rahman, M.; Mohd-Suhaimi, A.; Othman, N. Biosorption of pb (ii) and zn (ii) in synthetic waste water by watermelon rind (Citrullus lanatus). Appl. Mech. Mater. 2014, 465, 906–910. [Google Scholar] [CrossRef]
- Choudhary, M.; Ray, M.B.; Neogi, S. Evaluation of the potential application of cactus (Opuntia ficus-indica) as a bio-coagulant for pre-treatment of oil sands process-affected water. Sep. Purif. Technol. 2019, 209, 714–724. [Google Scholar] [CrossRef]
- Esa, N.M.; Ling, T.B.; Peng, L.S. By-products of rice processing: An overview of health benefits and applications. Rice Res. Open Access 2013, 1, 107. [Google Scholar]
- Prabhakaran, N.; Jothieswari, M.; Swarnalatha, S.; Sekaran, G. Tannery wastewater treatment process to minimize residual organics and generation of primary chemical sludge. Int. J. Environ. Sci. Technol. 2022, 19, 8857–8870. [Google Scholar] [CrossRef]
- Yin, C.-Y. Emerging usage of plant-based coagulants for water and wastewater treatment. Process Biochem. 2010, 45, 1437–1444. [Google Scholar] [CrossRef] [Green Version]
- Choy, S.Y.; Prasad, K.M.N.; Wu, T.Y.; Raghunandan, M.E.; Ramanan, R.N. Utilization of plant-based natural coagulants as future alternatives towards sustainable water clarification. J. Environ. Sci. 2014, 26, 2178–2189. [Google Scholar] [CrossRef]
- Nimesha, S.; Hewawasam, C.; Jayasanka, D.; Murakami, Y.; Araki, N.; Maharjan, N. Effectiveness of natural coagulants in water and wastewater treatment. Glob. J. Environ. Sci. Manag. 2022, 8, 101–116. [Google Scholar]
- Guo, Y.; Zelekew, O.A.; Sun, H.; Kuo, D.-H.; Lin, J.; Chen, X. Catalytic reduction of organic and hexavalent chromium pollutants with highly active bimetal cubios oxysulfide catalyst under dark. Sep. Purif. Technol. 2020, 242, 116769. [Google Scholar] [CrossRef]
- Alibeigi-Beni, S.; Habibi Zare, M.; Pourafshari Chenar, M.; Sadeghi, M.; Shirazian, S. Design and optimization of a hybrid process based on hollow-fiber membrane/coagulation for wastewater treatment. Environ. Sci. Pollut. Res. 2021, 28, 8235–8245. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Gupta, V. Advances in water treatment by adsorption technology. Nat. Protoc. 2006, 1, 2661–2667. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, E.; O’Brien, J.W.; Verhagen, R.; Mueller, J.F. Annual release of selected uv filters via effluent from wastewater treatment plants in australia. Chemosphere 2020, 247, 125887. [Google Scholar] [CrossRef] [PubMed]
- Ang, W.L.; Mohammad, A.W. State of the art and sustainability of natural coagulants in water and wastewater treatment. J. Clean. Prod. 2020, 262, 121267. [Google Scholar] [CrossRef]
- Hamzah, A.; Manikan, V.; Abd Aziz, N.A.F. Biodegradation of tapis crude oil using consortium of bacteria and fungi: Optimization of crude oil concentration and duration of incubation by response surface methodology. Sains Malays. 2017, 46, 43–50. [Google Scholar] [CrossRef]
- Kumar, K.; Chowdhury, A. Use of Novel Nanostructured Photocatalysts for the Environmental Sustainability of Wastewater Treatments. In Reference Module in Materials Science and Materials Engineering; Elsevier: Dublin, Ireland, 2018. [Google Scholar]
- Haydar, S.; Aziz, J.A. Coagulation–flocculation studies of tannery wastewater using combination of alum with cationic and anionic polymers. J. Hazard. Mater. 2009, 168, 1035–1040. [Google Scholar] [CrossRef]
- Parmar, K.; Prajapati, S.N.; Patel, R.; Dabhi, Y.M. Effective use of ferrous sulfate and alum as a coagulant in treatment of dairy industry wastewater. ARPN J. Eng. Appl. Sci. 2011, 6, 42–45. [Google Scholar]
- Tolkou, A.K.; Meez, E.; Kyzas, G.Z.; Torretta, V.; Collivignarelli, M.C.; Caccamo, F.M.; Deliyanni, E.A.; Katsoyiannis, I.A. A mini review of recent findings in cellulose-, polymer- and graphene-based membranes for fluoride removal from drinking water. C 2021, 7, 74. [Google Scholar] [CrossRef]
- Muhammad Burhanuddin, B. Recent advances on coagulation-based treatment of wastewater: Transition from chemical to natural coagulant. Curr. Pollut. Rep. 2021, 7, 379–391. [Google Scholar] [CrossRef]
- Alwi, H.; Idris, J.; Musa, M.; Ku Hamid, K.H. A preliminary study of banana stem juice as a plant-based coagulant for treatment of spent coolant wastewater. J. Chem. 2013, 2013, 165057. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, M.S.; Alves, B.R.R.; Silva, M.F.; Bergamasco, R.; Coral, L.A.; Bassetti, F.J. Cacl2 applied to the extraction of Moringa oleifera seeds and the use for Microcystis aeruginosa removal. Chem. Eng. J. 2016, 304, 469–475. [Google Scholar] [CrossRef]
- Hamawand, I. Anaerobic digestion process and bio-energy in meat industry: A review and a potential. Renew. Sustain. Energy Rev. 2015, 44, 37–51. [Google Scholar] [CrossRef]
- Madhavi, V.; Vijaya Bhaskar Reddy, A.; Madhavi, G. Synthesis, Characterization, and Properties of Carbon Nanocomposites and Their Application in Wastewater Treatment. In Environmental Remediation through Carbon Based Nano Composites; Springer: Berlin/Heidelberg, Germany, 2021; pp. 61–83. [Google Scholar]
- Jiang, J.-Q.; Lloyd, B. Progress in the development and use of ferrate (vi) salt as an oxidant and coagulant for water and wastewater treatment. Water Res. 2002, 36, 1397–1408. [Google Scholar] [CrossRef]
- Loloei, M.; Rezaee, A.; Roohaghdam, A.S.; Aliofkhazraei, M. Conductive microbial cellulose as a novel biocathode for cr (vi) bioreduction. Carbohydr. Polym. 2017, 162, 56–61. [Google Scholar] [CrossRef]
- Bogacki, J.; Naumczyk, J.; Marcinowski, P.; Kucharska, M. Treatment of cosmetic wastewater using physicochemical and chemical methods. Chemik 2011, 65, 94–97. [Google Scholar]
- Liang, Z.; Wang, Y.; Zhou, Y.; Liu, H. Coagulation removal of melanoidins from biologically treated molasses wastewater using ferric chloride. Chem. Eng. J. 2009, 152, 88–94. [Google Scholar] [CrossRef]
- Panhwar, A.; Bhutto, S. Improved reduction of cod, bod, tss and oil & grease from sugarcane industry effluent by ferric chloride and polyaluminum chloride coupled with polyvinyl alcohol. Ecol. Eng. Environ. Technol. 2021, 22, 8–14. [Google Scholar]
- Panhwar, A.A.; Almani, K.F.; Kandhro, A.A. Environmental degradation by textile industry; performance of chemical coagulants and activated carbon for removal of cod, bod. Tech. J. 2020, 25, 16–20. [Google Scholar]
- Irfan, M.; Butt, T.; Imtiaz, N.; Abbas, N.; Khan, R.A.; Shafique, A. The removal of COD, TSS and colour of black liquor by coagulation–flocculation process at optimized ph, settling and dosing rate. Arab. J. Chem. 2017, 10, S2307–S2318. [Google Scholar] [CrossRef] [Green Version]
- Al-Saati, N.; Hussein, T.; Abbas, M.; Hashim, K.; Al-Saati, Z.; Kot, P.; Sadique, M.; Aljefery, M.; Carnacina, I. Statistical modelling of turbidity removal applied to non-toxic natural coagulants in water treatment: A case study. Desalination Water Treat. 2019, 150, 406–412. [Google Scholar] [CrossRef]
- Zubaidi, S.L.; Al-Bugharbee, H.; Muhsin, Y.R.; Hashim, K.; Alkhaddar, R. Forecasting of monthly stochastic signal of urban water demand: Baghdad as a case study. IOP Conf. Ser. Mater. Sci. Eng. 2020, 888, 12018. [Google Scholar] [CrossRef]
- Liao, L.; Zhang, P. Preparation and characterization of polyaluminum titanium silicate and its performance in the treatment of low-turbidity water. Processes 2018, 6, 125. [Google Scholar] [CrossRef] [Green Version]
- Mustereț, C.P.; Morosanu, I.; Ciobanu, R.; Plavan, O.; Gherghel, A.; Al-Refai, M.; Roman, I.; Teodosiu, C. Assessment of coagulation–flocculation process efficiency for the natural organic matter removal in drinking water treatment. Water 2021, 13, 3073. [Google Scholar] [CrossRef]
- Hashim, K.S.; Ali, S.S.M.; AlRifaie, J.K.; Kot, P.; Shaw, A.; Al Khaddar, R.; Idowu, I.; Gkantou, M. Escherichia coli inactivation using a hybrid ultrasonic-electrocoagulation reactor. Chemosphere 2020, 247, 125868. [Google Scholar] [CrossRef] [PubMed]
- Al-Jumeily, D.; Hashim, K.; Alkaddar, R.; Al-Tufaily, M.; Lunn, J. Sustainable and environmental friendly ancient reed houses (inspired by the past to motivate the future). In Proceedings of the 2018 11th International Conference on Developments in eSystems Engineering (DeSE), Cambridge, UK, 2–5 September 2018; pp. 214–219. [Google Scholar]
- Zouboulis, A.I.; Tzoupanos, N. Alternative cost-effective preparation method of polyaluminium chloride (pac) coagulant agent: Characterization and comparative application for water/wastewater treatment. Desalination 2010, 250, 339–344. [Google Scholar] [CrossRef]
- Walton, M.E.; Samonte-Tan, G.P.; Primavera, J.H.; Edwards-Jones, G.; Le Vay, L. Are mangroves worth replanting? The direct economic benefits of a community-based reforestation project. Environ. Conserv. 2006, 33, 335–343. [Google Scholar] [CrossRef]
- Flaten, T.P. Aluminium as a risk factor in alzheimer’s disease, with emphasis on drinking water. Brain Res. Bull. 2001, 55, 187–196. [Google Scholar] [CrossRef]
- Kandimalla, R.; Vallamkondu, J.; Corgiat, E.B.; Gill, K.D. Understanding aspects of aluminum exposure in alzheimer’s disease development. Brain Pathol. 2016, 26, 139–154. [Google Scholar] [CrossRef]
- Krupińska, I. Aluminium drinking water treatment residuals and their toxic impact on human health. Molecules 2020, 25, 641. [Google Scholar] [CrossRef]
- Bondy, S.C. Low levels of aluminum can lead to behavioral and morphological changes associated with alzheimer’s disease and age-related neurodegeneration. Neurotoxicology 2016, 52, 222–229. [Google Scholar] [CrossRef] [Green Version]
- Lukiw, W.J.; Kruck, T.P.A.; Percy, M.E.; Pogue, A.I.; Alexandrov, P.N.; Walsh, W.J.; Sharfman, N.M.; Jaber, V.R.; Zhao, Y.; Li, W.; et al. Aluminum in neurological disease—A 36 year multicenter study. J. Alzheimers Dis. Park. 2019, 8, 6–10. [Google Scholar] [CrossRef]
- Iwuozor, K.O. Prospects and challenges of using coagulation-flocculation method in the treatment of effluents. Adv. J. Chem.-Sect. A 2019, 2, 105–127. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Jeon, J.H.; Shin, J.; Jang, H.M.; Kim, S.; Song, M.S.; Kim, Y.M. Quantitative and qualitative changes in antibiotic resistance genes after passing through treatment processes in municipal wastewater treatment plants. Sci. Total Environ. 2017, 605, 906–914. [Google Scholar] [CrossRef]
- Abdulhadi, B.; Kot, P.; Hashim, K.; Shaw, A.; Al Khaddar, R. In Influence of current density and electrodes spacing on reactive red 120 dye removal from dyed water using electrocoagulation/electroflotation (ec/ef) process. IOP Conf. Ser. Mater. Sci. Eng. 2019, 584, 12035. [Google Scholar] [CrossRef]
- Zubaidi, S.L.; Ortega-Martorell, S.; Al-Bugharbee, H.; Olier, I.; Hashim, K.S.; Gharghan, S.K.; Kot, P.; Al-Khaddar, R. Urban water demand prediction for a city that suffers from climate change and population growth: Gauteng province case study. Water 2020, 12, 1885. [Google Scholar] [CrossRef]
- Grmasha, R.A.; Al-sareji, O.J.; Salman, J.M.; Hashim, K.S. Polycyclic aromatic hydrocarbons (pahs) in urban street dust within three land-uses of babylon governorate, iraq: Distribution, sources, and health risk assessment. J. King Saud Univ.—Eng. Sci. 2022, 34, 231–239. [Google Scholar] [CrossRef]
- Owodunni, A.A.; Ismail, S. Revolutionary technique for sustainable plant-based green coagulants in industrial wastewater treatment—A review. J. Water Process Eng. 2021, 42, 102096. [Google Scholar] [CrossRef]
- Alazaiza, M.Y.D.; Albahnasawi, A.; Ali, G.A.M.; Bashir, M.J.K.; Nassani, D.E.; Al Maskari, T.; Amr, S.S.A.; Abujazar, M.S.S. Application of natural coagulants for pharmaceutical removal from water and wastewater: A review. Water 2022, 14, 140. [Google Scholar] [CrossRef]
- Renault, F.; Sancey, B.; Badot, P.-M.; Crini, G. Chitosan for coagulation/flocculation processes–an eco-friendly approach. Eur. Polym. J. 2009, 45, 1337–1348. [Google Scholar] [CrossRef]
- Choy, S.; Prasad, K.; Wu, T.; Ramanan, R. A review on common vegetables and legumes as promising plant-based natural coagulants in water clarification. Int. J. Environ. Sci. Technol. 2015, 12, 367–390. [Google Scholar] [CrossRef] [Green Version]
- Saleem, M.; Bachmann, R.T. A contemporary review on plant-based coagulants for applications in water treatment. J. Ind. Eng. Chem. 2019, 72, 281–297. [Google Scholar] [CrossRef]
- Mohd-Salleh, S.N.A.; Mohd-Zin, N.S.; Othman, N. A review of wastewater treatment using natural material and its potential as aid and composite coagulant. Sains Malays. 2019, 48, 155–164. [Google Scholar] [CrossRef]
- Brundtland, G.H. Report of the World Commission on Environment and Development: Our Common Future; UN: New York, NY, USA, 1987. [Google Scholar]
- Kamali, M.; Suhas, D.; Costa, M.E.; Capela, I.; Aminabhavi, T.M. Sustainability considerations in membrane-based technologies for industrial effluents treatment. Chem. Eng. J. 2019, 368, 474–494. [Google Scholar] [CrossRef]
- Kanmani, P.; Aravind, J.; Kamaraj, M.; Sureshbabu, P.; Karthikeyan, S. Environmental applications of chitosan and cellulosic biopolymers: A comprehensive outlook. Bioresour. Technol. 2017, 242, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Do, M.; Ngo, H.; Guo, W.; Liu, Y.; Chang, S.; Nguyen, D.; Nghiem, L.; Ni, B. Challenges in the application of microbial fuel cells to wastewater treatment and energy production: A mini review. Sci. Total Environ. 2018, 639, 910–920. [Google Scholar] [CrossRef]
- Ho, Y.; Norli, I.; FM, A.; Morad, N. New vegetal biopolymeric flocculant: A degradation and flocculation study. Iran. J. Energy Environ. 2014, 5, 1. [Google Scholar] [CrossRef] [Green Version]
- Abidin, Z.; Norhafizah, M.; Robiah, Y.; Aishah, D. Effect of storage conditions on jatropha curcas performance as biocoagulant for treating palm oil mill effluent. J. Environ. Sci. Technol. 2019, 12, 92–101. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos, J.D.; Veit, M.T.; Juchen, P.T.; da Cunha Gonçalves, G.; Palacio, S.M.; Fagundes-Klen, M. Use of different coagulants for cassava processing wastewater treatment. J. Environ. Chem. Eng. 2018, 6, 1821–1827. [Google Scholar] [CrossRef]
- Dezfooli, S.M.; Uversky, V.N.; Saleem, M.; Baharudin, F.S.; Hitam, S.M.S.; Bachmann, R.T. A simplified method for the purification of an intrinsically disordered coagulant protein from defatted Moringa oleifera seeds. Process Biochem. 2016, 51, 1085–1091. [Google Scholar] [CrossRef]
- Barbosa, A.D.; da Silva, L.F.; de Paula, H.M.; Romualdo, L.L.; Sadoyama, G.; Andrade, L.S. Combined use of coagulation (m. Oleifera) and electrochemical techniques in the treatment of industrial paint wastewater for reuse and/or disposal. Water Res. 2018, 145, 153–161. [Google Scholar] [CrossRef]
- Chitra, D.; Muruganandam, L. Performance of natural coagulants on greywater treatment. Recent Innov. Chem. Eng. Former. Recent Pat. Chem. Eng. 2020, 13, 81–92. [Google Scholar] [CrossRef]
- Maurya, A.; Reddy, B.; Theerthagiri, J.; Narayana, P.; Park, C.; Hong, J.; Yeom, J.-T.; Cho, K.; Reddy, N. Modeling and optimization of process parameters of biofilm reactor for wastewater treatment. Sci. Total Environ. 2021, 787, 147624. [Google Scholar] [CrossRef]
- Zaidi, N.; Muda, K.; Rahman, M.A.; Sgawi, M.; Amran, A. Effectiveness of local waste materials as organic-based coagulant in treating water. IOP Conf. Ser. Mater. Sci. Eng. 2019, 636, 12007. [Google Scholar] [CrossRef]
- Sibartie, S.; Ismail, N. Potential of Hibiscus sabdariffa and jatropha curcas as natural coagulants in the treatment of pharmaceutical wastewater. MATEC Web Conf. 2018, 152, 1009. [Google Scholar] [CrossRef]
- Kurniawan, S.B.; Abdullah, S.R.S.; Imron, M.F.; Said, N.S.M.; Ismail, N.; Hasan, H.A.; Othman, A.R.; Purwanti, I.F. Challenges and opportunities of biocoagulant/bioflocculant application for drinking water and wastewater treatment and its potential for sludge recovery. Int. J. Environ. Res. Public Health 2020, 17, 9312. [Google Scholar] [CrossRef]
- Pattnaik, P.; Dangayach, G.S. Sustainability of wastewater management in textile sectors: A conceptual framework. Environ. Eng. Manag. J. 2019, 18, 9. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, F.; Luo, Y.; Yang, H. A preliminary study on cactus as coagulant in water treatment. Process Biochem. 2006, 41, 730–733. [Google Scholar] [CrossRef]
- Choubey, S.; Rajput, S.; Bapat, K. Comparison of efficiency of some natural coagulants-bioremediation. Int. J. Emerg. Technol. Adv. Eng. 2012, 2, 429–434. [Google Scholar]
- Shakir, L.; Ejaz, S.; Ashraf, M.; Qureshi, N.A.; Anjum, A.A.; Iltaf, I.; Javeed, A. Ecotoxicological risks associated with tannery effluent wastewater. Environ. Toxicol. Pharmacol. 2012, 34, 180–191. [Google Scholar] [CrossRef]
- Mahmud, H.N.M.E.; Huq, A.O.; binti Yahya, R. The removal of heavy metal ions from wastewater/aqueous solution using polypyrrole-based adsorbents: A review. RSC Adv. 2016, 6, 14778–14791. [Google Scholar] [CrossRef]
- Kiso, Y.; Jung, Y.-J.; Park, M.-S.; Wang, W.; Shimase, M.; Yamada, T.; Min, K.-S. Coupling of sequencing batch reactor and mesh filtration: Operational parameters and wastewater treatment performance. Water Res. 2005, 39, 4887–4898. [Google Scholar] [CrossRef]
- Jahn, S.A.A. Drinking water from chinese rivers: Challenges of clarification. J. Water Supply: Res. Technol.—AQUA 2001, 50, 15–27. [Google Scholar] [CrossRef]
- Thakre, V.; Bhole, A. Relative evaluation of a few natural coagulants. J. Inst. Eng. Environ. Eng. 1985, 504, 89–92. [Google Scholar]
- Govindan, V. Coagulation studies on natural seed extracts. J. Indian Waterworks Assoc. 2005, 37, 145. [Google Scholar]
- Arukwe, U.; Amadi, B.; Duru, M.; Agomuo, E.; Adindu, E.; Odika, P.; Lele, K.; Egejuru, L.; Anudike, J. Chemical composition of Persea americana leaf, fruit and seed. Int. J. Recent Res. Appl. Stud. 2012, 11, 346–349. [Google Scholar]
- Ramesa, S.; Sooad, A.-d. Antibacterial properties of different cultivars of Phoenix dactylifera l. And their corresponding protein content. Ann. Biol. Res. 2012, 3, 4751–4757. [Google Scholar]
- Som, A.; Idris, J.; Hamid, K. Dragon fruit foliage as a low cost plant-based coagulant in latex concentrate wastewater treatment. Malays. J. Chem. Eng. 2007, 1, 47–59. [Google Scholar]
- Idris, J.; Md Som, A.; Musa, M.; Ku Hamid, K.H.; Husen, R.; Muhd Rodhi, M.N. Dragon fruit foliage plant-based coagulant for treatment of concentrated latex effluent: Comparison of treatment with ferric sulfate. J. Chem. 2013, 2013, 230860. [Google Scholar] [CrossRef] [Green Version]
- Patil, C.; Hugar, M. Treatment of dairy wastewater by natural coagulants. Int. Res. J. Eng. Technol. 2015, 2, 1120–1124. [Google Scholar]
- Camacho, F.P.; Sousa, V.S.; Bergamasco, R.; Teixeira, M.R. The use of moringa oleifera as a natural coagulant in surface water treatment. Chem. Eng. J. 2017, 313, 226–237. [Google Scholar] [CrossRef]
- Pallavi, N.; Mahesh, S. Feasibility study of Moringa oleifera as a natural coagulant for the treatment of dairy wastewater. Int. J. Eng. Res. 2013, 2, 201–203. [Google Scholar]
- Shan, T.C.; Matar, M.A.; Makky, E.A.; Ali, E.N. The use of Moringa oleifera seed as a natural coagulant for wastewater treatment and heavy metals removal. Appl. Water Sci. 2017, 7, 1369–1376. [Google Scholar] [CrossRef]
- Dehghani, M.; Alizadeh, M.H. The effects of the natural coagulant Moringa oleifera and alum in wastewater treatment at the bandar abbas oil refinery. Environ. Health Eng. Manag. J. 2016, 3, 225–230. [Google Scholar] [CrossRef]
- Muralimohan, N.; Palanisamy, T. Treatment of textile effluent by natural coagulants in erode district. Asian J. Chem. 2014, 26, 911. [Google Scholar] [CrossRef]
- Kazi, T.; Virupakshi, A.; Scholar, M. Treatment of tannery wastewater using natural coagulants. Development 2013, 2, 4061–4068. [Google Scholar]
- Anastasakis, K.; Kalderis, D.; Diamadopoulos, E. Flocculation behavior of mallow and okra mucilage in treating wastewater. Desalination 2009, 249, 786–791. [Google Scholar] [CrossRef]
- Carpinteyro-Urban, S.; Vaca, M.; Torres, L. Can vegetal biopolymers work as coagulant–flocculant aids in the treatment of high-load cosmetic industrial wastewaters? Water Air Soil Pollut. 2012, 223, 4925–4936. [Google Scholar] [CrossRef]
- Al-Hamadani, Y.A.; Yusoff, M.S.; Umar, M.; Bashir, M.J.; Adlan, M.N. Application of psyllium husk as coagulant and coagulant aid in semi-aerobic landfill leachate treatment. J. Hazard. Mater. 2011, 190, 582–587. [Google Scholar] [CrossRef]
- Shamsnejati, S.; Chaibakhsh, N.; Pendashteh, A.R.; Hayeripour, S. Mucilaginous seed of Ocimum basilicum as a natural coagulant for textile wastewater treatment. Ind. Crops Prod. 2015, 69, 40–47. [Google Scholar] [CrossRef]
- Chaibakhsh, N.; Ahmadi, N.; Zanjanchi, M.A. Use of Plantago major l. As a natural coagulant for optimized decolorization of dye-containing wastewater. Ind. Crops Prod. 2014, 61, 169–175. [Google Scholar] [CrossRef]
- Awang, N.A.; Aziz, H.A. Hibiscus rosa-sinensis leaf extract as coagulant aid in leachate treatment. Appl. Water Sci. 2012, 2, 293–298. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.-P.; Chen, Y.-Z.; Wang, Y.; Yuan, S.-J.; Yu, H.-Q. Optimization of the coagulation-flocculation process for pulp mill wastewater treatment using a combination of uniform design and response surface methodology. Water Res. 2011, 45, 5633–5640. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.I.; Sheikh Abdullah, S.R.; Idris, M.; Abu Hasan, H.; Halmi, M.I.E.; Hussin AL Sbani, N.; Hamed Jehawi, O.; Sanusi, S.N.A.; Hashim, M.H. Accumulation of fecal by Scirpus grossus grown in synthetic bauxite mining wastewater and identification of resistant rhizobacteria. Environ. Eng. Sci. 2017, 34, 367–375. [Google Scholar] [CrossRef]
- Ronke, R.A.; Saidat, O.G.; Abdulwahab, G. Coagulation-flocculation treatment of industrial wastewater using tamarind seed powder. Int. J. ChemTech Res. 2016, 9, 771–780. [Google Scholar]
- Saharudin, N.; Nithyanandam, R. Wastewater treatment by using natural coagulant. 2nd Eureca. Engineering 2014, 2–3, 202720485. [Google Scholar]
- Thawari, D.; Verma, S. Coal washery wastewater treatment using natural coagulants and chemical precipitation. Int. J. Sci. Res. 2015, 4, 1877–1881. [Google Scholar]
- Awasthi, M.K.; Li, J.; Kumar, S.; Awasthi, S.K.; Wang, Q.; Chen, H.; Wang, M.; Ren, X.; Zhang, Z. Effects of biochar amendment on bacterial and fungal diversity for co-composting of gelatin industry sludge mixed with organic fraction of municipal solid waste. Bioresour. Technol. 2017, 246, 214–223. [Google Scholar] [CrossRef]
- Patel, H.; Vashi, R. Comparison of naturally prepared coagulants for removal of cod and color from textile wastewater. Glob. NEST J. 2013, 15, 522–528. [Google Scholar]
- Ramavandi, B.; Farjadfard, S. Removal of chemical oxygen demand from textile wastewater using a natural coagulant. Korean J. Chem. Eng. 2014, 31, 81–87. [Google Scholar] [CrossRef]
- Lekshmi, B.; Joseph, R.S.; Jose, A.; Abinandan, S.; Shanthakumar, S. Studies on reduction of inorganic pollutants from wastewater by Chlorella pyrenoidosa and Scenedesmus abundans. Alex. Eng. J. 2015, 54, 1291–1296. [Google Scholar] [CrossRef] [Green Version]
- Hanif, M.A.; Nadeem, R.; Zafar, M.N.; Bhatti, H.N.; Nawaz, R. Physico-chemical treatment of textile wastewater using natural coagulant cassia. J. Chem. Soc. Pak. 2008, 30, 385–393. [Google Scholar]
- Jeon, E.-C.; Son, H.-K.; Sa, J.-H. Emission characteristics and factors of selected odorous compounds at a wastewater treatment plant. Sensors 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anju, S.; Mophin-Kani, K. Exploring the use of orange peel and neem leaf powder as alternative coagulant in treatment of dairy wastewater. Int. J. Sci. Eng. Res. 2016, 7, 238–244. [Google Scholar]
- Choy, S.Y.; Prasad, K.M.N.; Wu, T.Y.; Raghunandan, M.E.; Yang, B.; Phang, S.-M.; Ramanan, R.N. Isolation, characterization and the potential use of starch from jackfruit seed wastes as a coagulant aid for treatment of turbid water. Environ. Sci. Pollut. Res. 2017, 24, 2876–2889. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, M.; Mkandawire, T.; Edmondson, A.; O’neill, J.; Kululanga, G. Potential of using plant extracts for purification of shallow well water in malawi. Phys. Chem. Earth Parts A/B/C 2009, 34, 799–805. [Google Scholar] [CrossRef]
- Babu, R.; Chaudhuri, M. Home water treatment by direct filtration with natural coagulant. J. Water Health 2005, 3, 27–30. [Google Scholar] [CrossRef] [Green Version]
- Diaz, A.; Rincon, N.; Escorihuela, A.; Fernandez, N.; Chacin, E.; Forster, C. A preliminary evaluation of turbidity removal by natural coagulants indigenous to venezuela. Process Biochem. 1999, 35, 391–395. [Google Scholar] [CrossRef]
- Bunce, J.T.; Ndam, E.; Ofiteru, I.D.; Moore, A.; Graham, D.W. A review of phosphorus removal technologies and their applicability to small-scale domestic wastewater treatment systems. Front. Environ. Sci. 2018, 6, 8. [Google Scholar] [CrossRef] [Green Version]
- Beltrán-Heredia, J.; Sánchez-Martín, J. Municipal wastewater treatment by modified tannin flocculant agent. Desalination 2009, 249, 353–358. [Google Scholar] [CrossRef]
- Bongiovani, M.C.; Camacho, F.P.; Coldebella, P.F.; Valverde, K.C.; Nishi, L.; Bergamasco, R. Removal of natural organic matter and trihalomethane minimization by coagulation/flocculation/filtration using a natural tannin. Desalination Water Treat. 2016, 57, 5406–5415. [Google Scholar] [CrossRef]
- Özacar, M.; Şengil, İ.A. The use of tannins from turkish acorns (valonia) in water treatment as a coagulant and coagulant aid. Turk. J. Eng. Environ. Sci. 2002, 26, 255–264. [Google Scholar]
- Bacelo, H.A.M. Tannin Resins from Maritime Pine Bark as Adsorbents for Water Treatment and Recovery of Substances. Ph.D. Thesis, University of Porto, Porto, Portugal, 2021. [Google Scholar]
- Beltrán-Heredia, J.; Sánchez-Martín, J.; Solera-Hernández, C. Anionic surfactants removal by natural coagulant/flocculant products. Ind. Eng. Chem. Res. 2009, 48, 5085–5092. [Google Scholar] [CrossRef]
- Jayaram, K.; Murthy, I.; Lalhruaitluanga, H.; Prasad, M. Biosorption of lead from aqueous solution by seed powder of Strychnos potatorum L. Colloids Surf. B Biointerfaces 2009, 71, 248–254. [Google Scholar] [CrossRef]
- Raghuwanshi, P.K.; Mandloi, M.; Sharma, A.J.; Malviya, H.S.; Chaudhari, S. Improving filtrate quality using agrobased materials as coagulant aid. Water Qual. Res. J. 2002, 37, 745–756. [Google Scholar] [CrossRef]
- Schulz, C.R.; Okun, D.A.; Donaldson, D.; Austin, J. Surface Water Treatment for Communities in Developing Countries; U.S. Agency for International Development: Washington, DC, USA, 1992. [Google Scholar]
- Selvaraj, K.; Sevugaperumal, R.; Ramasubramanian, V. Impact of match industry effluent on growth and biochemical characteristics of Cyamopsis tetragonoloba taub and amelioration of the stress by seaweed treatment. Indian J. Fund. Appl. Life Sci 2013, 3, 192–197. [Google Scholar]
- Sarawgi, G.; Kamra, A.; Suri, N.; Kaur, A.; Sarethy, I.P. Effect of Strychnos potatorum linn. Seed extracts on water samples from different sources and with diverse properties. Asian J. Water Environ. Pollut. 2009, 6, 13–17. [Google Scholar]
- Tripathi, P.; Chaudhuri, M.; Bokil, S. Nirmali seed—A naturally occurring coagulant. Indian J. Environ. Health 1976, 18, 72–81. [Google Scholar]
- Adinolfi, M.; Corsaro, M.M.; Lanzetta, R.; Parrilli, M.; Folkard, G.; Grant, W.; Sutherland, J. Composition of the coagulant polysaccharide fraction from Strychnos potatorum seeds. Carbohydr. Res. 1994, 263, 103–110. [Google Scholar] [CrossRef]
- Muthuraman, G.; Sasikala, S. Removal of turbidity from drinking water using natural coagulants. J. Ind. Eng. Chem. 2014, 20, 1727–1731. [Google Scholar] [CrossRef]
- Bhuptawat, H.; Folkard, G.; Chaudhari, S. Innovative physico-chemical treatment of wastewater incorporating Moringa oleifera seed coagulant. J. Hazard. Mater. 2007, 142, 477–482. [Google Scholar] [CrossRef]
- Anwar, F.; Bhanger, M. Analytical characterization of Moringa oleifera seed oil grown in temperate regions of pakistan. J. Agric. Food Chem. 2003, 51, 6558–6563. [Google Scholar] [CrossRef] [PubMed]
- Koul, B.; Chase, N. Moringa oleifera lam.: Panacea to several maladies. J. Chem. Pharm. Res. 2015, 7, 687–707. [Google Scholar]
- Farooq, B.; Koul, B. Comparative analysis of the antioxidant, antibacterial and plant growth promoting potential of five indian varieties of Moringa oleifera L. South Afr. J. Bot. 2020, 129, 47–55. [Google Scholar] [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S.; Talbot, B.G. Active agents and mechanism of coagulation of turbid waters using Moringa oleifera. Water Res. 1995, 29, 703–710. [Google Scholar] [CrossRef]
- Muller, S. Wirkstoffe zur Trinkwasseraufbereitung aus Samen von Moringa Oleifera. Master’s Thesis, Universitat Heidelberg, Heidelberg, Germany, 1980. [Google Scholar]
- Jahn, S.A.A. Using moringa seeds as coagulants in developing countries. J. Am. Water Work. Assoc. 1988, 80, 43–50. [Google Scholar] [CrossRef]
- Muyibi, S.A.; Evison, L.M. Optimizing physical parameters affecting coagulation of turbid water with Morninga oleifera seeds. Water Res. 1995, 29, 2689–2695. [Google Scholar] [CrossRef]
- Gassenschmidt, U.; Jany, K.D.; Bernhard, T.; Niebergall, H. Isolation and characterization of a flocculating protein from Moringa oleifera lam. Biochim. Et Biophys. Acta Gen. Subj. 1995, 1243, 477–481. [Google Scholar] [CrossRef]
- Ghebremichael, K.A.; Gunaratna, K.; Henriksson, H.; Brumer, H.; Dalhammar, G. A simple purification and activity assay of the coagulant protein from Moringa oleifera seed. Water Res. 2005, 39, 2338–2344. [Google Scholar] [CrossRef]
- Okuda, T.; Baes, A.U.; Nishijima, W.; Okada, M. Isolation and characterization of coagulant extracted from Moringa oleifera seed by salt solution. Water Res. 2001, 35, 405–410. [Google Scholar] [CrossRef] [Green Version]
- Bolto, B.; Gregory, J. Organic polyelectrolytes in water treatment. Water Res. 2007, 41, 2301–2324. [Google Scholar] [CrossRef]
- Sulaiman, M.; Zhigila, D.A.; Mohammed, K.; Umar, D.M.; Aliyu, B.; Abd Manan, F. Moringa oleifera seed as alternative natural coagulant for potential application in water treatment: A review. J. Adv. Rev. Sci. Res 2017, 30, 1–11. [Google Scholar]
- Dotto, J.; Fagundes-Klen, M.R.; Veit, M.T.; Palacio, S.M.; Bergamasco, R. Performance of different coagulants in the coagulation/flocculation process of textile wastewater. J. Clean. Prod. 2019, 208, 656–665. [Google Scholar] [CrossRef]
- Ashmawy, M.; Moussa, M.; Ghoneim, A.; Tammam, A. Enhancingthe efficiency of primary sedimentation in wastewater treatment plants with the application of Moringa oliefera seeds and quicklime. J. Am. Sci. 2012, 8, 494–502. [Google Scholar]
- Hiremath, P.J.; Farmer, A.; Cannon, S.B.; Woodward, J.; Kudapa, H.; Tuteja, R.; Kumar, A.; BhanuPrakash, A.; Mulaosmanovic, B.; Gujaria, N. Large-scale transcriptome analysis in chickpea (Cicer arietinum L.), an orphan legume crop of the semi-arid tropics of asia and africa. Plant Biotechnol. J. 2011, 9, 922–931. [Google Scholar] [CrossRef] [Green Version]
- Jaseela, L.; Chadaga, M. Treatment of dairy effluent using Cicer arietinum. Int. J. Innov. Res. Sci. Engg. Technol 2015, 4, 4881–4885. [Google Scholar]
- Asrafuzzaman, M.; Fakhruddin, A.; Hossain, M. Reduction of turbidity of water using locally available natural coagulants. Int. Sch. Res. Not. 2011, 2011, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Lim, T.K. Edible Medicinal and Non-Medicinal Plants; Springer: Berlin/Heidelberg, Germany, 2012; Volume 1. [Google Scholar]
- Mataka, L.; Henry, E.; Masamba, W.; Sajidu, S. Lead remediation of contaminated water using Moringa stenopetala and Moringa oleifera seed powder. Int. J. Environ. Sci. Technol. 2006, 3, 131–139. [Google Scholar] [CrossRef]
- Bhole, A. Relative evaluation of a few natural coagulants. Aqua J. Water Supply: Res. Technol. 1995, 44, 284–290. [Google Scholar]
- Mbogo, S.A. A novel technology to improve drinking water quality using natural treatment methods in rural tanzania. J. Environ. Health 2008, 70, 46–50. [Google Scholar]
- Gupta, V.K.; Mittal, A.; Malviya, A.; Mittal, J. Adsorption of carmoisine a from wastewater using waste materials—Bottom ash and deoiled soya. J. Colloid Interface Sci. 2009, 335, 24–33. [Google Scholar] [CrossRef] [Green Version]
- Mittal, A.; Mittal, J.; Malviya, A.; Kaur, D.; Gupta, V. Adsorption of hazardous dye crystal violet from wastewater by waste materials. J. Colloid Interface Sci. 2010, 343, 463–473. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, C.C.; Cruz, P.A.; da Fonseca, M.M.R.; Xavier-Filho, L. Antibacterial properties of the extract of Abelmoschus esculentus. Biotechnol. Bioprocess Eng. 2011, 16, 971–977. [Google Scholar] [CrossRef]
- Sáenz, C.; Sepúlveda, E.; Matsuhiro, B. Opuntia spp mucilage’s: A functional component with industrial perspectives. J. Arid Environ. 2004, 57, 275–290. [Google Scholar] [CrossRef]
- Nharingo, T.; Moyo, M. Application of opuntia Ficus-indica in bioremediation of wastewaters. A critical review. J. Environ. Manage. 2016, 166, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Rebah, F.B.; Siddeeg, S. Cactus an eco-friendly material for wastewater treatment: A review. J. Mater. Environ. Sci. 2017, 8, 1770–1782. [Google Scholar]
- Miller, S.M.; Fugate, E.J.; Craver, V.O.; Smith, J.A.; Zimmerman, J.B. Toward understanding the efficacy and mechanism of Opuntia spp. As a natural coagulant for potential application in water treatment. Environ. Sci. Technol. 2008, 42, 4274–4279. [Google Scholar] [CrossRef]
- Manunza, B.; Deiana, S.; Pintore, M.; Gessa, C. Molecular dynamics study of polygalacturonic acid chains in aqueous solution. Carbohydr. Res. 1997, 300, 85–88. [Google Scholar] [CrossRef]
- Fahmi, M.R.; Hamidin, N.; Abidin, C.Z.A.; Fazara, U.; Ali, M.; Hatim, M. Performance evaluation of okra (Abelmoschus esculentus) as coagulant for turbidity removal in water treatment. Trans. Tech. Publ. 2014, 594, 226–230. [Google Scholar] [CrossRef]
- Raji, Y.O.; Abubakar, L.; Giwa, S.O.; Giwa, A. Assessment of coagulation efficiency of okra seed extract for surface water treatment. Int. J. Sci. Eng. Res. 2016, 6, 1–7. [Google Scholar]
- Thakur, S.S.; Choubey, S. Assessment of coagulation efficiency of Moringa oleifera and okra for treatment of turbid water. Arch. Appl. Sci. Res. 2014, 6, 24–30. [Google Scholar]
- Mishra, S.; Singh, S.; Srivastava, R. Okra seeds: An efficient coagulant. Int. J. Res. Appl. Sci. Eng. 2017, 5, 1–5. [Google Scholar]
- Nath, A.; Mishra, A.; Pande, P.P. A review natural polymeric coagulants in wastewater treatment. Mater. Today: Proc. 2021, 46, 6113–6117. [Google Scholar] [CrossRef]
- Wang, J.; Chen, X. Removal of antibiotic resistance genes (args) in various wastewater treatment processes: An overview. Crit. Rev. Environ. Sci. Technol. 2022, 52, 571–630. [Google Scholar] [CrossRef]
- Shewa, W.A.; Dagnew, M. Revisiting chemically enhanced primary treatment of wastewater: A review. Sustainability 2020, 12, 5928. [Google Scholar] [CrossRef]
- Zainal, S.F.F.S.; Abdul Aziz, H.; Mohd Omar, F.; Alazaiza, M.Y. Sludge performance in coagulation-flocculation treatment for suspended solids removal from landfill leachate using tin (iv) chloride and Jatropha curcas. Int. J. Environ. Anal. Chem. 2021, 1–15. [Google Scholar] [CrossRef]
- Oladoja, N.A. Headway on natural polymeric coagulants in water and wastewater treatment operations. J. Water Process Eng. 2015, 6, 174–192. [Google Scholar] [CrossRef]
- Dwarapureddi, B.K.; Saritha, V. Evaluation of factors affecting coagulation of water with natural polymers. Int. J. Adv. Res. Biol. Sci. 2015, 2, 98–113. [Google Scholar]
- Jiao, Y.-N.; Chen, H.; Gao, R.-X.; Zhu, Y.-G.; Rensing, C. Organic compounds stimulate horizontal transfer of antibiotic resistance genes in mixed wastewater treatment systems. Chemosphere 2017, 184, 53–61. [Google Scholar] [CrossRef]
- Li, Y.S.; Yan, L.; Xiang, C.B.; Hong, L.J. Treatment of oily wastewater by organic–inorganic composite tubular ultrafiltration (uf) membranes. Desalination 2006, 196, 76–83. [Google Scholar] [CrossRef]
- Chong, J.W.R.; Khoo, K.S.; Yew, G.Y.; Leong, W.H.; Lim, J.W.; Lam, M.K.; Ho, Y.-C.; Ng, H.S.; Munawaroh, H.S.H.; Show, P.L. Advances in production of bioplastics by microalgae using food waste hydrolysate and wastewater: A review. Bioresour. Technol. 2021, 342, 125947. [Google Scholar] [CrossRef]
- Ghernaout, D.; Elboughdiri, N.; Ghareba, S. Fenton technology for wastewater treatment: Dares and trends. Open Access Libr. J. 2020, 7, e6045. [Google Scholar] [CrossRef]
- Wu, T.Y.; Mohammad, A.W.; Jahim, J.M.; Anuar, N. Pollution control technologies for the treatment of palm oil mill effluent (pome) through end-of-pipe processes. J. Environ. Manag. 2010, 91, 1467–1490. [Google Scholar] [CrossRef] [PubMed]
- Nouri, J.; Nouri, N.; Moeeni, M. Development of industrial waste disposal scenarios using life-cycle assessment approach. Int. J. Environ. Sci. Technol. 2012, 9, 417–424. [Google Scholar] [CrossRef] [Green Version]
- FAO. Food Outlook: Global Market Analysis; FAO: Rome, Italy, 2012. [Google Scholar]
- Sutherland, J.; Folkard, G.; Poirier, Y. Moringa Oleifera. The Constraints to Commercialisation; CIRAD: Montpellier, France, 2002. [Google Scholar]
- Sahu, O.; Chaudhari, P. Review on chemical treatment of industrial waste water. J. Appl. Sci. Environ. Manag. 2013, 17, 241–257. [Google Scholar] [CrossRef]
- Rodrigues, A.C.; Boroski, M.; Shimada, N.S.; Garcia, J.C.; Nozaki, J.; Hioka, N. Treatment of paper pulp and paper mill wastewater by coagulation–flocculation followed by heterogeneous photocatalysis. J. Photochem. Photobiol. A Chem. 2008, 194, 1–10. [Google Scholar] [CrossRef]
- Santo, C.E.; Vilar, V.J.; Botelho, C.M.; Bhatnagar, A.; Kumar, E.; Boaventura, R.A. Optimization of coagulation–flocculation and flotation parameters for the treatment of a petroleum refinery effluent from a portuguese plant. Chem. Eng. J. 2012, 183, 117–123. [Google Scholar] [CrossRef]
- Verma, A.K.; Dash, R.R.; Bhunia, P. A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J. Environ. Manage. 2012, 93, 154–168. [Google Scholar] [CrossRef]
- Colantoni, A.; Evic, N.; Lord, R.; Retschitzegger, S.; Proto, A.; Gallucci, F.; Monarca, D. Characterization of biochars produced from pyrolysis of pelletized agricultural residues. Renew. Sustain. Energy Rev. 2016, 64, 187–194. [Google Scholar] [CrossRef] [Green Version]
- Xiong, X.; Iris, K.; Tsang, D.C.; Bolan, N.S.; Ok, Y.S.; Igalavithana, A.D.; Kirkham, M.; Kim, K.-H.; Vikrant, K. Value-added chemicals from food supply chain wastes: State-of-the-art review and future prospects. Chem. Eng. J. 2019, 375, 121983. [Google Scholar] [CrossRef]
- Xiang, W.; Zhang, X.; Chen, J.; Zou, W.; He, F.; Hu, X.; Tsang, D.C.; Ok, Y.S.; Gao, B. Biochar technology in wastewater treatment: A critical review. Chemosphere 2020, 252, 126539. [Google Scholar] [CrossRef]
- Creamer, A.E.; Gao, B. Carbon-based adsorbents for postcombustion CO2 capture: A critical review. Environ. Sci. Technol. 2016, 50, 7276–7289. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, H.; Wang, J.; Hou, H.; Du, C.; Ma, P.-C.; Kadier, A. Application of Biochar for Wastewater Treatment. In Biochar and Its Application in Bioremediation; Springer: Berlin/Heidelberg, Germany, 2021; pp. 67–90. [Google Scholar]
- Omran, B.A.; Baek, K.-H. Valorization of agro-industrial biowaste to green nanomaterials for wastewater treatment: Approaching green chemistry and circular economy principles. J. Environ. Manag. 2022, 311, 114806. [Google Scholar] [CrossRef]
- Yang, F.; Xu, Z.; Yu, L.; Gao, B.; Xu, X.; Zhao, L.; Cao, X. Kaolinite enhances the stability of the dissolvable and undissolvable fractions of biochar via different mechanisms. Environ. Sci. Technol. 2018, 52, 8321–8329. [Google Scholar] [CrossRef]
- Inyang, M.I.; Gao, B.; Yao, Y.; Xue, Y.; Zimmerman, A.; Mosa, A.; Pullammanappallil, P.; Ok, Y.S.; Cao, X. A review of biochar as a low-cost adsorbent for aqueous heavy metal removal. Crit. Rev. Environ. Sci. Technol. 2016, 46, 406–433. [Google Scholar] [CrossRef]
- Wang, B.; Gao, B.; Fang, J. Recent advances in engineered biochar productions and applications. Crit. Rev. Environ. Sci. Technol. 2017, 47, 2158–2207. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, P.; Liu, S.; Peng, P.; Min, M.; Cheng, Y.; Anderson, E.; Zhou, N.; Fan, L.; Liu, C. Effects of feedstock characteristics on microwave-assisted pyrolysis–a review. Bioresour. Technol. 2017, 230, 143–151. [Google Scholar] [CrossRef]
- Van Vinh, N.; Zafar, M.; Behera, S.; Park, H.-S. Arsenic (iii) removal from aqueous solution by raw and zinc-loaded pine cone biochar: Equilibrium, kinetics, and thermodynamics studies. Int. J. Environ. Sci. Technol. 2015, 12, 1283–1294. [Google Scholar] [CrossRef] [Green Version]
- Palansooriya, K.N.; Yang, Y.; Tsang, Y.F.; Sarkar, B.; Hou, D.; Cao, X.; Meers, E.; Rinklebe, J.; Kim, K.-H.; Ok, Y.S. Occurrence of contaminants in drinking water sources and the potential of biochar for water quality improvement: A review. Crit. Rev. Environ. Sci. Technol. 2020, 50, 549–611. [Google Scholar] [CrossRef]
- Cao, L.; Iris, K.; Cho, D.-W.; Wang, D.; Tsang, D.C.; Zhang, S.; Ding, S.; Wang, L.; Ok, Y.S. Microwave-assisted low-temperature hydrothermal treatment of red seaweed (Gracilaria lemaneiformis) for production of levulinic acid and algae hydrochar. Bioresour. Technol. 2019, 273, 251–258. [Google Scholar] [CrossRef]
- Ahmad, M.; Lee, S.S.; Dou, X.; Mohan, D.; Sung, J.-K.; Yang, J.E.; Ok, Y.S. Effects of pyrolysis temperature on soybean stover-and peanut shell-derived biochar properties and tce adsorption in water. Bioresour. Technol. 2012, 118, 536–544. [Google Scholar] [CrossRef]
- Shimabuku, K.K.; Kearns, J.P.; Martinez, J.E.; Mahoney, R.B.; Moreno-Vasquez, L.; Summers, R.S. Biochar sorbents for sulfamethoxazole removal from surface water, stormwater, and wastewater effluent. Water Res. 2016, 96, 236–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajapaksha, A.U.; Vithanage, M.; Ahmad, M.; Seo, D.C.; Cho, J.S.; Lee, S.E.; Lee, S.S.; Ok, Y.S. Enhanced sulfamethazine removal by steam-activated invasive plant-derived biochar. J. Hazard. Mater. 2015, 290, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Choy, S.Y.; Prasad, K.N.; Wu, T.Y.; Raghunandan, M.E.; Ramanan, R.N. Performance of conventional starches as natural coagulants for turbidity removal. Ecol. Eng. 2016, 94, 352–364. [Google Scholar] [CrossRef]
- Zhou, N.; Chen, H.; Xi, J.; Yao, D.; Zhou, Z.; Tian, Y.; Lu, X. Biochars with excellent pb (ii) adsorption property produced from fresh and dehydrated banana peels via hydrothermal carbonization. Bioresour. Technol. 2017, 232, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Son, E.-B.; Poo, K.-M.; Chang, J.-S.; Chae, K.-J. Heavy metal removal from aqueous solutions using engineered magnetic biochars derived from waste marine macro-algal biomass. Sci. Total Environ. 2018, 615, 161–168. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, L.; Gorring, M.; Deng, Y. Aluminum-impregnated biochar for adsorption of arsenic (v) in urban stormwater runoff. J. Environ. Eng. 2019, 145, 4019008. [Google Scholar] [CrossRef]
- Gray, M. Black is green: Biochar for stormwater management. In Proceedings of the WEFTEC 2016, New Orleans, LO, USA, 24–28 September 2016. [Google Scholar]



| Parameter | Instrument Used | Units of Measurement | Importance |
|---|---|---|---|
| Turbidity | Turbidimeter | NTU | Measure of relative clarity of water. |
| pH | pH meter | H+ conc. | Indicator of water quality& measure of acidity/basicity. |
| Temperature | Thermometer | °C | Impacts both biological & chemical characteristics of water. |
| Conductivity | Conductivity meter | Sm−1 | Measure of water capability to pass electrical flow. |
| Alkalinity | Potentiometer | V m−1 | Measure of capacity of water to neutralize acids. |
| Acidity | pH meter | H+ conc. | Indicator of industrial pollution. |
| Total dissolved solids | Conductivity meter | Sm−1 | Measure of combined organic & inorganic substances dissolved in water. |
| Total suspended solids | Suspended solids density meter | mgL−1 | Includes all particles suspended in water which won’t pass through a filter. |
| Total hardness | Potentiometer | mgL−1 | Measure of amount of dissolved Ca & Mg. |
| Ca hardness | Potentiometer | mgL−1 | Measure of amount of dissolved Ca. |
| Mg hardness | Potentiometer | mgL−1 | Measure of amount of dissolved Mg. |
| Chlorides | Potentiometer | mgL−1 | Indicator of pollution in water. |
| Sulphate | Spectrophotometer | mgL−1 | Indicator of algal growth in water. |
| Ammonia | Spectrophotometer | mgL−1 | Indicator of fecal pollution. |
| Nitrate | Spectrophotometer | mgL−1 | Indicator of sewage pollution. |
| Nitrite | Spectrophotometer | mgL−1 | Indicator of contamination from fertilizer run off. |
| Calcium | Spectrophotometer | mgL−1 | Measure of water hardness. |
| Magnesium | Spectrophotometer | mgL−1 | Measure of water hardness. |
| Iron | Spectrophotometer | mgL−1 | Excess level indicates presence of contaminants. |
| Manganese | Spectrophotometer | mgL−1 | Excess level results in undesirable taste. |
| Fluoride | Spectrophotometer | mgL−1 | Reduces tooth decay. |
| Sodium | Spectrophotometer | mgL−1 | Maintains blood pressure & osmotic pressure. |
| Potassium | Spectrophotometer | mgL−1 | Maintains osmotic pressure. |
| Dissolved oxygen | Colorimeter | mgL−1 | Indicator of water quality. |
| Biological oxygen demand (BOD) | BOD meter | mgL−1 | Measure of water quality. Higher BOD signifies lower water quality. |
| Chemical oxygen demand (COD) | COD meter | mgL−1 | Measure of water & waste water quality. |
| Pesticides | Spectrophotometer | mgL−1 | Indicator of water toxicity. |
| E. coli | Paper strip method | per 100 mL | Indicator of fecal contamination. |
| Fecal coliform | Paper strip method | per 100 mL | Indicator of fecal contamination. |
| Velocity | Current meter | ms−1 | The speed at which water flows. |
| Detergent | pH meter | H+ conc. | Results in algal blooms &depletes oxygen in water leading to water pollution. |
| Coagulant | Part Used | State of Coagulant | Opt. Dosage | Removal Efficiency (%) | References |
|---|---|---|---|---|---|
| M.oleifera Lam. (Drumstick) | Seeds | Powder | 0.2 gL−1 | Turbidity removal (61.60%); COD removal (65.00%) | [122] |
| M.oleifera Lam. (Drumstick) | Seeds | Deoiled powder | 6000 mgL−1 | TSS removal (95%) | [123] |
| M.oleifera Lam. (Drumstick) | Seeds | Powder | 500 mgL−1 | Turbidity removal (96.80%) | [124] |
| M.oleifera Lam. (Drumstick) | Seeds | Powder | 0.6 gL−1 | Turbidity removal (82%), COD removal (83%) | [125] |
| M. oleifera Lam. (Drumstick) | Seeds | Stock solution | 0.3 mgL−1 | Cu and Cd removal (98%); Pb removal (78%) | [126] |
| M.oleifera Lam. (Drumstick) and alum | Seeds | Solution | 70 and 80 mgL−1 | COD removal (50.41%); Turbidity removal (86.14%); TSS removal (81.52%) | [127] |
| M.oleifera Lam. (Drumstick) and alum | Seeds | Stock solution | 50 mgL−1 each | BOD removal (80.67%); COD removal (66.73%) | [128] |
| M.oleifera Lam. (Drumstick) Cicer arietinum L. (Chickpea) | Seeds | Powder | 50 mgL−1 | Turbidity removal M. oleifera (82.2%); Chickpea (81.2%); Cactus (78.54%) | [129] |
| M. oleifera Lam. (Drumstick) and Musa acuminate L. (Banana peel) | Seeds | Powder | 200 and 400 mgL−1 | Pb removal (81%); Ni removal (74%); Cd removal (97%) | [12] |
| Malus sylvestris L. (European crab apple) and aluminum sulphate | Seeds | Stock solution | 62.5 mgL−1 | Turbidity removal (66%) | [130] |
| Cyamopsis tetragonoloba L. (Taub.) (Guar) | Gum | Powder | 300 mgL−1 | Turbidity removal (67.82%) | [130] |
| P.ovata (Psyllium) and PAC | Husk | Powder | 0.4 and 7.2 gL−1 | Color removal (90%); COD removal (96%) | [131] |
| Parkia biglobosa Jacq. (Locust bean) | Gum | Powder | 300 mgL−1 | Turbidity removal (67.82%) | [130] |
| Ocimum basilicum L. (Basil plant) | Seeds | Stock solution | 1.6 mgL−1 | Color removal (68.50%); COD removal (61.60%) | [132] |
| Opuntia ficus Linn. (Miller) (Cactus) | Mucilage | Powder | 150 mgL−1 | Turbidity removal (49.56%) | [130] |
| Plantago major Linn. (Carls) (Broadleaf plantain) | Mucilage | Powder | 297.6 mgL−1 | Color removal (92.4%); COD removal (81.60%) | [133] |
| Hibiscus rosasinensis Linn.(Carls) (China rose) and alum | Seeds | Powder | 500 and 400 mgL−1 | Turbidity removal; Hibiscus (60%); Alum (100%) | [134] |
| Trigonella foenum L. (Fenugreek) | Seeds | Powder | 0.1 gL−1 | Turbidity removal (58%); COD removal (63%) | [122] |
| Abelmoschus esculentus L. (Okra) | Mucilage | Powder | 3.2 mgL−1 | Turbidity removal (97.24%); COD removal (85.69%) | [135] |
| Abelmoschus esculentus L. (Okra) and Iron (III) chloride hexa hydrate | Mucilage | Stock solution | 2.5 mgL−1 | Turbidity removal (74%) | [130] |
| Cicer arietinum L. (Chickpea) | Seeds | Powder | 2 gL−1 | TDS removal (82%); COD removal (84%); BOD removal (83%) | [136] |
| Cicer arietinum L. (Chickpea) | Seeds | Powder | 0.1 gL−1 | Turbidity removal (78.33%); COD removal (83%) | [122] |
| Dolichos lablab Linn (Hyacinth bean) | Fruits | Powder | 0.2 gL−1 | Turbidity removal (71.74%); COD removal (75%) | [122] |
| Tamarindus indica L. (Tamarind) | Seed | Powder | 400 mgL−1 | Turbidity removal (97,72%) | [137] |
| Hibiscus sabdariffa L. (Roselle) | Seed | Powder | 60 mgL−1 | Turbidity removal (87.18%) | [138] |
| Opuntia indica L. (Mill.) (Cactus) | Mucilage | Powder | 0.4 gL−1 | Turbidity removal (78.54%); COD removal (75%) | [128] |
| Momordica charantia L. (Bitter gourd) | Seed | Solution | 400 ppm | Turbidity removal (61.03%) | [139] |
| Gossypium barbadense L. (Cotton) | Seed | Oil | 30 mLL−1 | TSS removal (66.27%) | [140] |
| Ricinus communis L. (Castor) | Seed | Oil | 40 mLL−1 | TSS removal (66.67%) | [136] |
| S.potatorum L. f.(Nirmali) | Seed | Stock solution | 60 mLL−1 | BOD removal (65.23%), COD removal (72.71%), Turbidity removal (75.20%) | [140] |
| Musa acuminate L. (Banana) | Peel | Juice | 90 mLL−1 | Turbidity removal (98.50%) | [60] |
| Zea mays L. (Maize) | Seed | Powder | 30 gL−1 | COD removal (68.82%); Color removal (47.03%) | [141] |
| Plantago ovata Forssk(Isabgol) | Seed | Powder | 1.5 mgL−1 | COD removal (89.30%) | [142] |
| P.vulgaris Gustav Hauser (French bean) | Seed | Powder | 1.5 mgL−1 | Color removal (73%) | [143] |
| Cassia fistula L. (Golden shower) | Seed | Powder | 0.5 gL−1 | Color removal (71.3%) | [144] |
| Vitis vinifera L. (Grape vine) | Seed | Powder | 1.5 mgL−1 | Color removal (80%) | [145] |
| Citrus sinensis L. (Osbeck) (Orange) | Peel | Powder | 0.2 gL−1 | Turbidity removal (97%) | [146] |
| Artocarpus heterophyllus Lam. (Jackfruit) | Seed | Powder | 60 mgL−1 | Turbidity removal (43%) | [147] |
| Jatropha curcas L. Britton and Mills.(Barbados nut) | Seed | Powder | 14 mgL−1 | Turbidity removal (93%) | [148] |
| Strychnos potatorum L. f. (Nirmali) | Seed | Powder | 1.5 mgL−1 | Turbidity removal (90%) | [149] |
| Carica papaya Linn. (Papaw) | Seed | Powder | 0.4 g per 200 mL | Turbidity removal (90%) | [150] |
| Julifora Prosopis var. juliflora (Sw.) DC (Mesquite bean) | Seed | Powder | 1.5 mgL−1 | Turbidity removal (96%) | [151] |
| Acacia mearnsii De Wild. (Black wattle) | Bark | Powder | 14 mgL−1 | Turbidity removal (75%) | [151] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koul, B.; Bhat, N.; Abubakar, M.; Mishra, M.; Arukha, A.P.; Yadav, D. Application of Natural Coagulants in Water Treatment: A Sustainable Alternative to Chemicals. Water 2022, 14, 3751. https://doi.org/10.3390/w14223751
Koul B, Bhat N, Abubakar M, Mishra M, Arukha AP, Yadav D. Application of Natural Coagulants in Water Treatment: A Sustainable Alternative to Chemicals. Water. 2022; 14(22):3751. https://doi.org/10.3390/w14223751
Chicago/Turabian StyleKoul, Bhupendra, Nargis Bhat, Mustapha Abubakar, Meerambika Mishra, Ananta Prasad Arukha, and Dhananjay Yadav. 2022. "Application of Natural Coagulants in Water Treatment: A Sustainable Alternative to Chemicals" Water 14, no. 22: 3751. https://doi.org/10.3390/w14223751





