Spatiotemporal Distribution of Nitrous Oxide on the Northeastern Bering Sea Shelf
Abstract
:1. Introduction
2. Materials and Methods
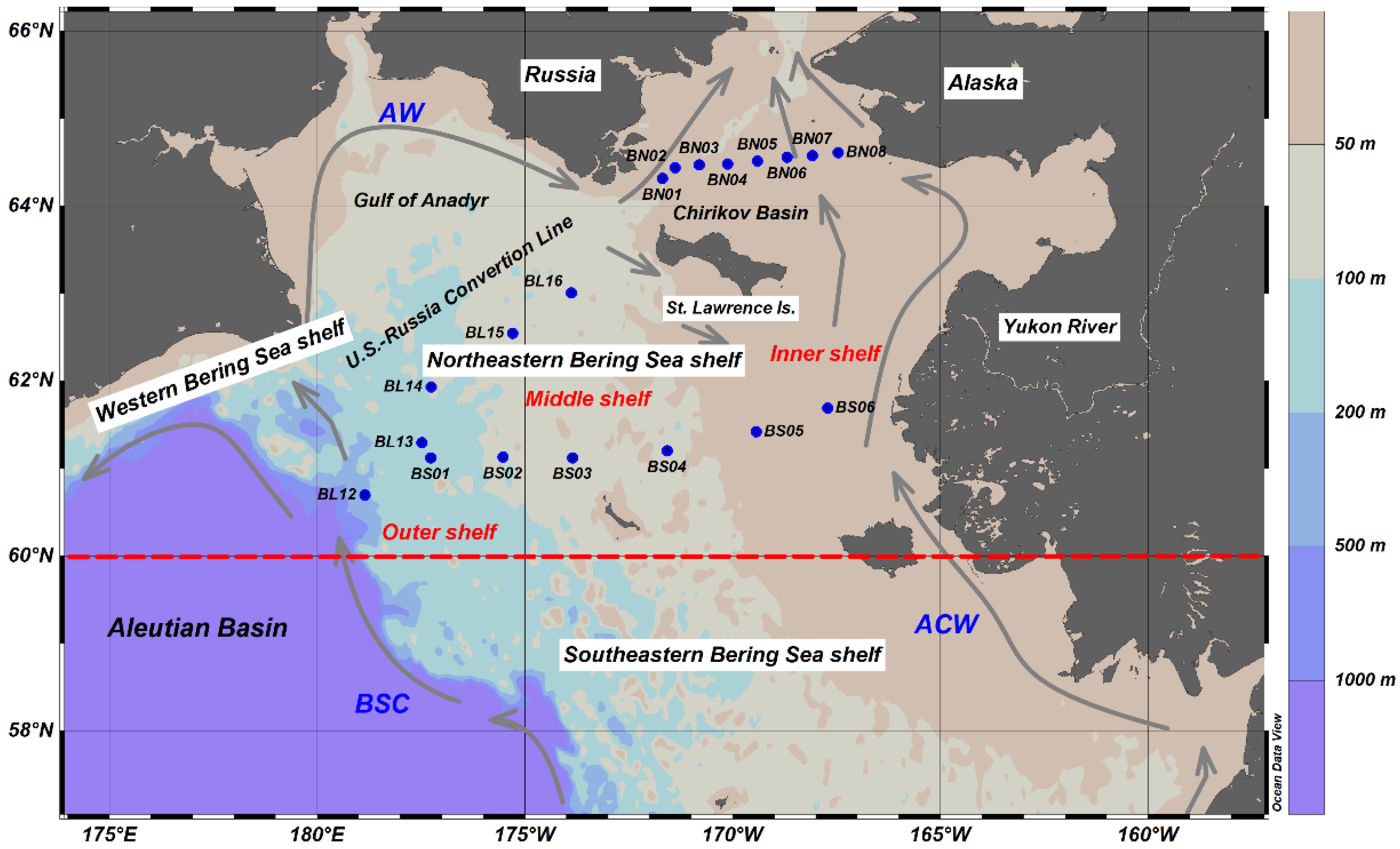
2.1. Study Area
2.2. Seawater Sampling
2.3. Analysis
2.4. Calculation
2.4.1. Saturation and Excess N2O (ΔN2O)
2.4.2. Apparent Oxygen Utilization (AOU)
2.4.3. N**
2.4.4. Sea–Air Flux
3. Results
3.1. N2O in the Surface Water
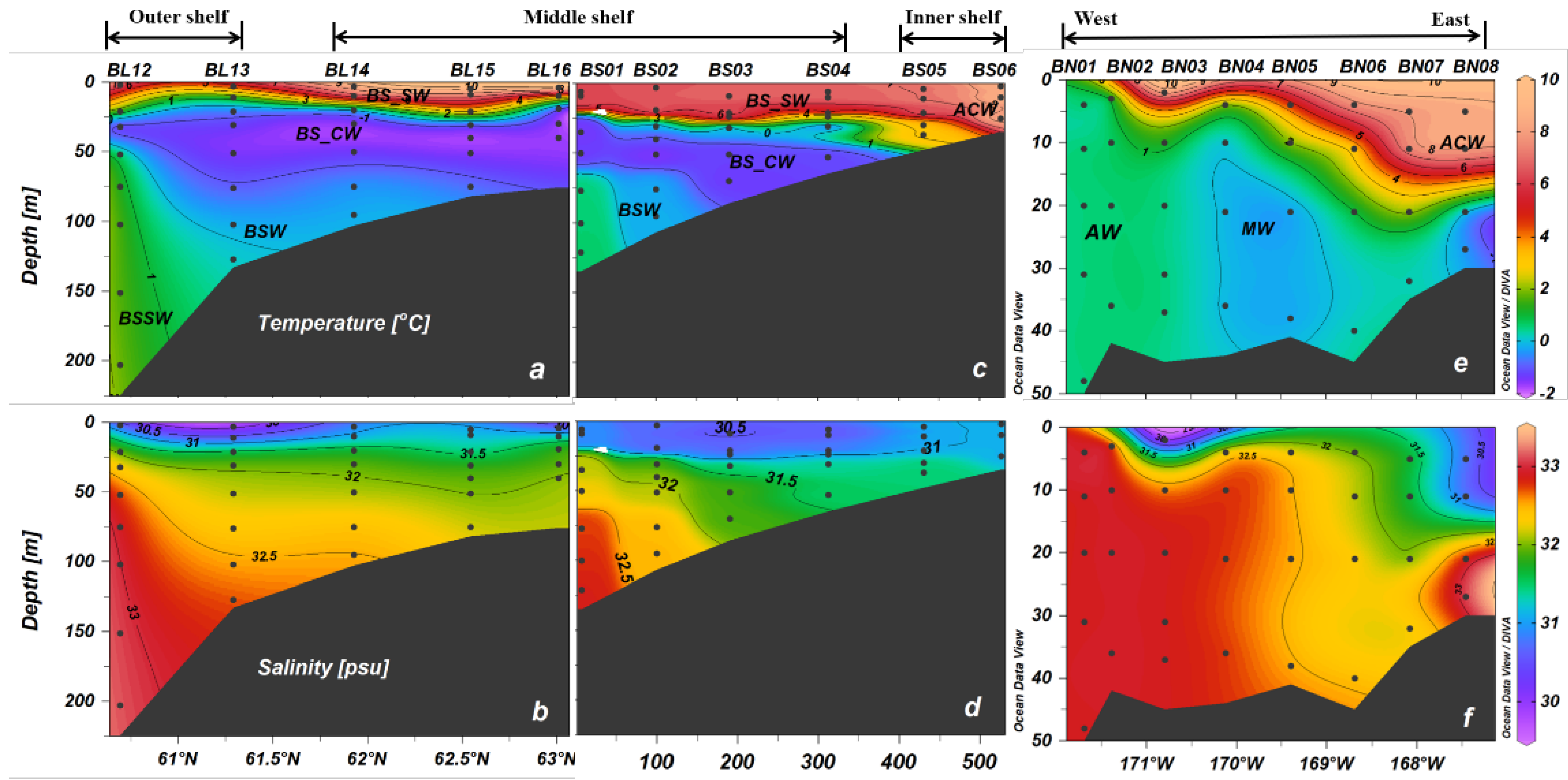
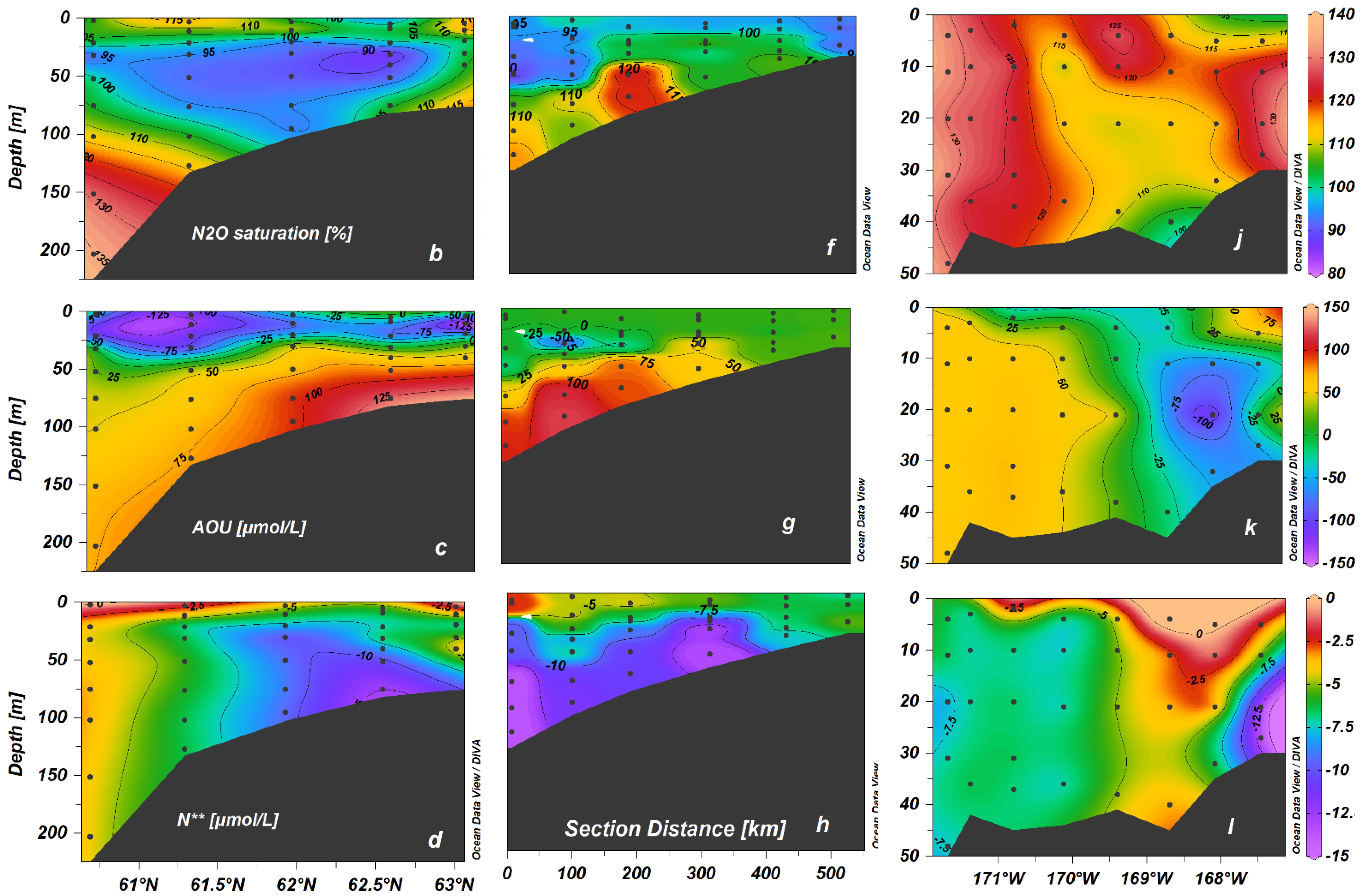
3.2. The Vertical Distribution of N2O
4. Discussion
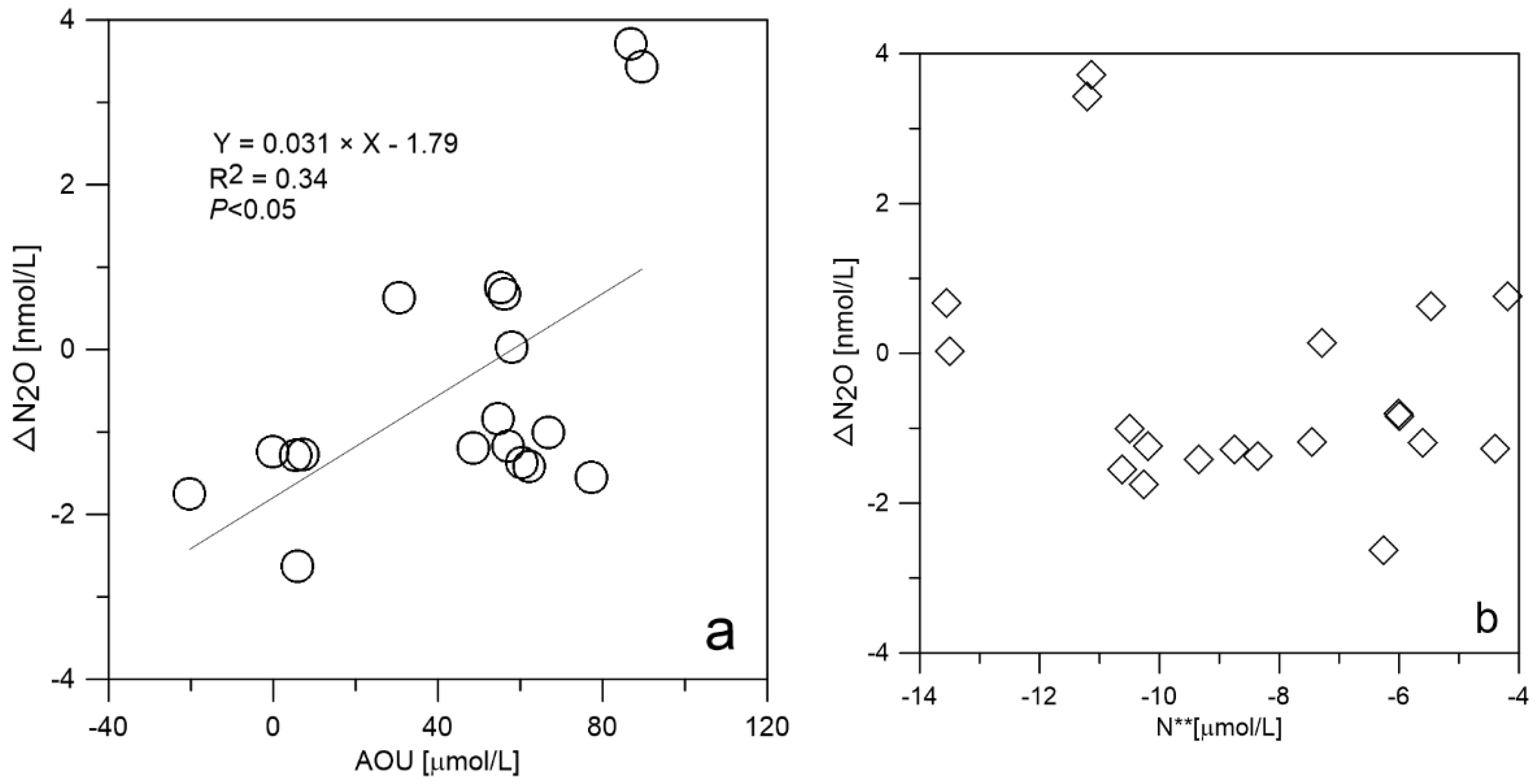
4.1. N2O Flux Variations in Surface Water
4.2. Temporal and Spatial Variations in N2O in the Northern Bering Sea Shelf
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yung, Y.L.; Wang, W.C.; Lacis, A.A. Greenhouse effect due to atmospheric nitrous oxide. Geophys. Res. Lett. 1976, 3, 619–621. [Google Scholar] [CrossRef]
- Ravishankara, A.R.; Daniel, J.S.; Portmann, R.W. Nitrous Oxide (N2O): The Dominant Ozone-Depleting Substance Emitted in the 21st Century. Science 2009, 326, 123–125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canadell, J.G.; Monteiro, P.; Costa, M.H.; Cotrim da Cunha, L.; Cox, P.M.; Eliseev, A.V.; Henson, S.; Ishii, M.; Jaccard, S.; Koven, C.; et al. Global Carbon and other Biogeochemical Cycles and Feedbacks. In Climate Change 2021: The Physical Science Basis; Masson-Delmotte, V., Ed.; Cambridge University Press: Cambridge/New York, NY, USA, 2021. [Google Scholar]
- Nevison, C.D.; Keeling, R.F.; Weiss, R.F.; Popp, B.N.; Jin, X.; Fraser, P.J.; Porter, L.W.; Hess, P.G. Southern Ocean ventilation inferred from seasonal cycles of atmospheric N2O and O2/N2at Cape Grim, Tasmania. Tellus B Chem. Phys. Meteorol. 2005, 57, 218–229. [Google Scholar] [CrossRef]
- Nevison, C.D.; Weiss, R.F.; Erickson, D.J., III. Global oceanic emissions of nitrous oxide. J. Geophys. Res. Space Phys. 1995, 100, 15809–15820. [Google Scholar] [CrossRef]
- Bange, H.W.; Freing, A.; Kock, A.; Löscher, C. Marine pathways to nitrous oxide. In Nitrous Oxide and Climate Change; Smith, K., Ed.; Routledge: London, UK, 2010; pp. 36–62. [Google Scholar]
- Dore, J.E.; Karl, D. Nitrification in the euphotic zone as a source for nitrite, nitrate, and nitrous oxide at Station ALOHA. Limnol. Oceanogr. 1996, 41, 1619–1628. [Google Scholar] [CrossRef]
- Ostrom, N.E.; Russ, M.E.; Popp, B.; Rust, T.M.; Karl, D. Mechanisms of nitrous oxide production in the subtropical North Pacific based on determinations of the isotopic abundances of nitrous oxide and di-oxygen. Chem. Glob. Change Sci. 2000, 2, 281–290. [Google Scholar] [CrossRef]
- Walter, S.; Bange, H.W.; Breitenbach, U.; Wallace, D.W.R. Nitrous oxide in the North Atlantic Ocean. Biogeosciences 2006, 3, 607–619. [Google Scholar] [CrossRef] [Green Version]
- Nevison, C.; Butler, J.H.; Elkins, J.W. Global distribution of N2O and the ΔN2O-AOU yield in the subsurface ocean. Glob. Biogeochem. Cycles 2003, 17, 1–30. [Google Scholar] [CrossRef]
- Toyoda, S.; Yoshida, N. Determination of Nitrogen Isotopomers of Nitrous Oxide on a Modified Isotope Ratio Mass Spectrometer. Anal. Chem. 1999, 71, 4711–4718. [Google Scholar] [CrossRef]
- Yamagishi, H.; Westley, M.B.; Popp, B.N.; Toyoda, S.; Yoshida, N.; Watanabe, S.; Koba, K.; Yamanaka, Y. Role of nitrification and denitrification on the nitrous oxide cycle in the eastern tropical North Pacific and Gulf of California. J. Geophys. Res. Earth Surf. 2007, 112, 139–158. [Google Scholar] [CrossRef]
- Steele, M.; Ermold, W.; Zhang, J. Arctic Ocean surface warming trends over the past 100 years. Geophys. Res. Lett. 2008, 35, 2614. [Google Scholar] [CrossRef] [Green Version]
- Giles, K.A.; Laxon, S.W.; Ridout, A.L.; Wingham, D.J.; Bacon, S. Western Arctic Ocean freshwater storage increased by wind-driven spin-up of the Beaufort Gyre. Nat. Geosci. 2012, 5, 194–197. [Google Scholar] [CrossRef]
- Perovich, D.K.; Richter-Menge, J.A. Loss of Sea Ice in the Arctic. Annu. Rev. Mar. Sci. 2009, 1, 417–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, D.; Chen, L.; Chen, B.; Gao, Z.; Zhong, W.; Feely, R.A.; Anderson, L.G.; Sun, H.; Chen, J.; Chen, M.; et al. Increase in acidifying water in the western Arctic Ocean. Nat. Clim. Change 2017, 7, 195–199. [Google Scholar] [CrossRef]
- Li, C.; Wang, B.; Wang, Z.; Li, J.; Yang, G.; Chen, J.; Lin, L.; Lyu, Y.; Guo, F. Spatial and Interannual Variability in Distributions and Cycling of Summer Biogenic Sulfur in the Bering Sea. Geophys. Res. Lett. 2019, 46, 4816–4825. [Google Scholar] [CrossRef]
- Sun, H.; Gao, Z.-Y.; Zhao, D.-R.; Sun, X.-W.; Chen, L.-Q. Spatial variability of summertime aragonite saturation states and its influencing factor in the Bering Sea. Adv. Clim. Change Res. 2021, 12, 508–516. [Google Scholar] [CrossRef]
- Wu, M.; Chen, L.; Zhan, L.; Zhang, J.; Li, Y.; Liu, J. Spatial Variability and Factors Influencing the Air-Sea N2O Flux in the Bering Sea, Chukchi Sea and Chukchi Abyssal Plain. Atmosphere 2017, 8, 65. [Google Scholar] [CrossRef] [Green Version]
- Mathis, J.T.; Bates, N.R.; Hansell, D.A.; Babila, T. Net community production in the northeastern Chukchi Sea. Deep Sea Res. Part II Top. Stud. Oceanogr. 2009, 56, 1213–1222. [Google Scholar] [CrossRef]
- Springer, A.M.; McROY, C.P.; Flint, M.V. The Bering Sea Green Belt: Shelf-edge processes and ecosystem production. Fish. Oceanogr. 1996, 5, 205–223. [Google Scholar] [CrossRef]
- Andreev, A.G.; Budyansky, M.V.; Khen, G.V.; Uleysky, M.Y. Water dynamics in the western Bering Sea and its impact on chlorophyll a concentration. Ocean Dyn. 2020, 70, 593–602. [Google Scholar] [CrossRef]
- Devol, A.H.; A Codispoti, L.; Christensen, J.P. Summer and winter denitrification rates in western Arctic shelf sediments. Cont. Shelf Res. 1997, 17, 1029–1050. [Google Scholar] [CrossRef]
- Jones, E.P.; Swift, J.H.; Anderson, L.G.; Lipizer, M.; Civitarese, G.; Falkner, K.K.; Kattner, G.; McLaughlin, F. Tracing Pacific water in the North Atlantic Ocean. J. Geophys. Res. Earth Surf. 2003, 108, 1–13. [Google Scholar] [CrossRef]
- Coachman, L.K.; Coachman, L.K.; Aagaard, K.; Tripp, R.B. Bering Strait: The Regional Physical Oceanography; University of Washington Press: Washington, DC, USA, 1975; Volume 172, pp. 502–503. [Google Scholar]
- Liu, N.; Lin, L.; Wang, Y.; Chen, H.; He, Y. The distribution and inter-annual variation of water masses on the Bering Sea shelf in summer. Acta Oceanol. Sin. 2016, 35, 59–67. [Google Scholar] [CrossRef]
- Danielson, S.; Aagaard, K.; Weingartner, T.; Martin, S.; Winsor, P.; Gawarkiewicz, G.; Quadfasel, D. The St. Lawrence polynya and the Bering shelf circulation: New observations and a model comparison. J. Geophys. Res. Earth Surf. 2006, 111, 1–18. [Google Scholar] [CrossRef]
- Muench, R.D.; Schumacher, J.D.; Salo, S.A. Winter currents and hydrographic conditions on the northern central Bering Sea shelf. J. Geophys. Res. Earth Surf. 1988, 93, 516. [Google Scholar] [CrossRef]
- Stabeno, P.J.; Schumacher, J.D.; Ohtani, K. The physical oceanography of the Bering Sea. In Dynamics of the Bering Sea: A Summary of Physical, Chemical, and Biological Characteristics, and a Synopsis of Research on the Bering Sea, North Pacific Marine Science Organization (PICES); Loughlin, T.R., Ohtani, K., Eds.; University of Alaska: Fairbanks, AK, USA, 1999; pp. 1–28. [Google Scholar]
- Zhan, L.; Zhang, J.; Ouyang, Z.; Lei, R.; Xu, S.; Qi, D.; Gao, Z.; Sun, H.; Li, Y.; Wu, M.; et al. High-resolution distribution pattern of surface water nitrous oxide along a cruise track from the Okhotsk Sea to the western Arctic Ocean. Limnol. Oceanogr. 2020, 66, S401–S410. [Google Scholar] [CrossRef]
- Fenwick, L.; Capelle, D.; Damm, E.; Zimmermann, S.; Williams, W.J.; Vagle, S.; Tortell, P.D. Methane and nitrous oxide distributions across the North American Arctic Ocean during summer, 2015. J. Geophys. Res. Oceans 2017, 122, 390–412. [Google Scholar] [CrossRef] [Green Version]
- Hirota, A.; Ijiri, A.; Komatsu, D.; Ohkubo, S.; Nakagawa, F.; Tsunogai, U. Enrichment of nitrous oxide in the water columns in the area of the Bering and Chukchi Seas. Mar. Chem. 2009, 116, 47–53. [Google Scholar] [CrossRef]
- Kitidis, V.; Upstill-Goddard, R.; Anderson, L.G. Methane and nitrous oxide in surface water along the North-West Passage, Arctic Ocean. Mar. Chem. 2010, 121, 80–86. [Google Scholar] [CrossRef]
- Toyoda, S.; Kakimoto, T.; Kudo, K.; Yoshida, N.; Sasano, D.; Kosugi, N.; Ishii, M.; Kameyama, S.; Inagawa, M.; Yoshikawa-Inoue, H.; et al. Distribution and Production Mechanisms of N2O in the Western Arctic Ocean. Glob. Biogeochem. Cycles 2021, 35, e2020GB006881. [Google Scholar] [CrossRef]
- Zhang, J.; Zhan, L.; Chen, L.; Li, Y.; Chen, J. Coexistence of nitrous oxide undersaturation and oversaturation in the surface and subsurface of the western Arctic Ocean. J. Geophys. Res. Oceans 2015, 120, 8392–8401. [Google Scholar] [CrossRef] [Green Version]
- Stabeno, P.J.; Farley, E.V., Jr.; Kachel, N.B.; Moore, S.; Mordy, C.W.; Napp, J.M.; Overland, J.E.; Pinchuk, A.I.; Sigler, M.F. A comparison of the physics of the northern and southern shelves of the eastern Bering Sea and some implications for the ecosystem. Deep Sea Res. Part II Top. Stud. Oceanogr. 2012, 65–70, 14–30. [Google Scholar] [CrossRef]
- Zhan, L.-Y.; Chen, L.-Q.; Zhang, J.-X.; Lin, Q. A system for the automated static headspace analysis of dissolved N2O in seawater. Int. J. Environ. Anal. Chem. 2013, 93, 828–842. [Google Scholar] [CrossRef]
- Dickson, A.G. Determination of Dissolved Oxygen in Sea Water by Winkler Titration. WHP Op. Methods 1996, 3. [Google Scholar]
- Zhuang, Y.; Jin, H.; Li, H.; Chen, J.; Lin, L.; Bai, Y.; Ji, Z.; Zhang, Y.; Gu, F. Pacific inflow control on phytoplankton community in the Eastern Chukchi Shelf during summer. Cont. Shelf Res. 2016, 129, 23–32. [Google Scholar] [CrossRef]
- Weiss, R.F.; Price, B.A. Nitrous oxide solubility in water and seawater. Mar. Chem. 1980, 8, 347–359. [Google Scholar] [CrossRef]
- Weiss, R.F. The solubility of nitrogen, oxygen and argon in water and seawater. Deep. Sea Res. Oceanogr. Abstr. 1970, 17, 721–735. [Google Scholar] [CrossRef]
- Gruber, N.; Sarmiento, J.L. Global patterns of marine nitrogen fixation and denitrification. Glob. Biogeochem. Cycles 1997, 11, 235–266. [Google Scholar] [CrossRef]
- Nishino, S.; Shimada, K.; Itoh, M. Use of ammonium and other nitrogen tracers to investigate the spreading of shelf waters in the western Arctic halocline. J. Geophys. Res. Earth Surf. 2005, 110, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Wanninkhof, R. Relationship between wind speed and gas exchange over the ocean revisited. Limnol. Oceanogr. Methods 2014, 12, 351–362. [Google Scholar] [CrossRef]
- Zhan, L.; Wu, M.; Chen, L.; Zhang, J.; Li, Y.; Liu, J. The Air-Sea Nitrous Oxide Flux along Cruise Tracks to the Arctic Ocean and Southern Ocean. Atmosphere 2017, 8, 216. [Google Scholar] [CrossRef]
- Randall, K.; Scarratt, M.; Levasseur, M.; Michaud, S.; Xie, H.; Gosselin, M. First measurements of nitrous oxide in Arctic sea ice. J. Geophys. Res. Earth Surf. 2012, 117, 247–253. [Google Scholar] [CrossRef]
- Nihoul, J.C.J. Aspects of the Northern Bering Sea Ecohydrodynamics. Elsevier Oceanogr. Ser. 1986, 42, 385–399. [Google Scholar] [CrossRef]
- Tanaka, T.; Guo, L.; Deal, C.; Tanaka, N.; Whitledge, T.; Murata, A. N deficiency in a well-oxygenated cold bottom water over the Bering Sea shelf: Influence of sedimentary denitrification. Cont. Shelf Res. 2004, 24, 1271–1283. [Google Scholar] [CrossRef]
- Zhuang, Y.; Jin, H.; Cai, W.; Li, H.; Qi, D.; Chen, J. Extreme Nitrate Deficits in the Western Arctic Ocean: Origin, Decadal Changes, and Implications for Denitrification on a Polar Marginal Shelf. Glob. Biogeochem. Cycles 2022, 36, e2022GB007304. [Google Scholar] [CrossRef]
- Bange, H.W.; Andreae, M. Nitrous oxide in the deep waters of the world’s oceans. Glob. Biogeochem. Cycles 1999, 13, 1127–1135. [Google Scholar] [CrossRef]


| Region | Station | Date | Longitude [°E] | Latitude [°N] | Bot. Depth [m] | Temperature [°C] | Salinity | N2O [nmol/L] | N2O Saturation [%] | N2O Flux Mean ± Std [μmol/(m2·d)] |
|---|---|---|---|---|---|---|---|---|---|---|
| South of St. Lawrence Island | BL12 | 14 July 2012 | −178.85 | 60.69 | 233 | 6.61 | 30.68 | 13.0 | 106 | 0.3 ± 2.1 |
| BL13 | 15 July 2012 | −177.48 | 61.29 | 133 | 4.57 | 29.88 | 15.3 | 115 | ||
| BL14 | 15 July 2012 | −177.25 | 61.93 | 103 | 8.43 | 30.945 | 11.9 | 104 | ||
| BL15 | 16 July 2012 | −175.30 | 62.54 | 82 | 8.74 | 31.129 | 11.0 | 97 | ||
| BL16 | 15 July 2012 | −173.89 | 63.00 | 76 | 9.92 | 30.62 | 12.5 | 114 | ||
| BS01 | 11 September 2012 | −177.26 | 61.12 | 135 | 6.19 | 30.80 | 11.8 | 95 | ||
| BS02 | 11 September 2012 | −175.53 | 61.12 | 107 | 6.51 | 30.60 | 12.2 | 100 | ||
| BS03 | 11 September 2012 | −173.85 | 61.12 | 86 | 6.67 | 30.49 | 11.8 | 97 | ||
| BS04 | 10 September 2012 | −171.58 | 61.20 | 65 | 6.60 | 30.49 | 11.7 | 96 | ||
| BS05 | 10 September 2012 | −169.43 | 61.40 | 48 | 7.04 | 31.01 | 11.5 | 96 | ||
| BS06 | 10 September 2012 | −167.72 | 61.70 | 35 | 8.50 | 31.02 | 10.7 | 93 | ||
| Chirikov Basin | BN01 | 16 July 2012 | −171.69 | 64.30 | 53 | 0.77 | 32.87 | 20.1 | 132 | 14.7 ± 0.8 |
| BN02 | 16 July 2012 | −171.38 | 64.40 | 42 | 0.64 | 32.94 | 19.6 | 128 | ||
| BN03 | 17 July 2012 | −170.80 | 64.50 | 45 | 8.85 | 29.79 | 13.7 | 120 | ||
| BN04 | 17 July 2012 | −170.12 | 64.50 | 44 | 1.55 | 32.39 | 17.2 | 116 | ||
| BN05 | 17 July 2012 | −169.40 | 64.50 | 41 | 4.73 | 32.21 | 16.9 | 129 | ||
| BN06 | 17 July 2012 | −168.70 | 64.50 | 45 | 7.91 | 31.95 | 13.3 | 114 | ||
| BN07 | 17 July 2012 | −168.08 | 64.60 | 35 | 7.96 | 31.71 | 13.1 | 112 | ||
| BN08 | 17 July 2012 | −167.46 | 64.60 | 30 | 8.58 | 30.753 | 12.9 | 112 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Zhan, L.; Chen, L.; Jin, H.; Wu, M.; Ye, W.; Liu, J. Spatiotemporal Distribution of Nitrous Oxide on the Northeastern Bering Sea Shelf. Water 2022, 14, 3738. https://doi.org/10.3390/w14223738
Zhang J, Zhan L, Chen L, Jin H, Wu M, Ye W, Liu J. Spatiotemporal Distribution of Nitrous Oxide on the Northeastern Bering Sea Shelf. Water. 2022; 14(22):3738. https://doi.org/10.3390/w14223738
Chicago/Turabian StyleZhang, Jiexia, Liyang Zhan, Liqi Chen, Haiyan Jin, Man Wu, Wangwang Ye, and Jian Liu. 2022. "Spatiotemporal Distribution of Nitrous Oxide on the Northeastern Bering Sea Shelf" Water 14, no. 22: 3738. https://doi.org/10.3390/w14223738






