Biowaste Valorization to Produce Advance Carbon Material-Hydrochar for Potential Application of Cr (VI) and Cd (II) Adsorption in Wastewater: A Review
Abstract
:1. Introduction
2. Data Sources and Methodology
3. Modification of Hydrochar
3.1. Physical Modification
3.2. Chemical Modification
4. Potentials Application of Hydrochar for Heavy Metal Adsorption
4.1. Application of Hydrochar in the Adsorption of Cr (VI)
| Metal | Region | Standard | Harm | References |
|---|---|---|---|---|
| Cr | China | 50 ug/L | Disruption of cell membrane; Protein degradation; Lung cancer; Liver cancer; nausea; Gastric bleeding; gastric ulcer. | GB 5749-2022 |
| Canada | 50 ug/L | [86] | ||
| WHO | 50 ug/L | [87] | ||
| NZ | 50 ug/L | [88] | ||
| AUS | 50 ug/L | [89] | ||
| USEPA | 100 ug/L | [90] | ||
| UK | 50 ug/L | [91] |

4.1.1. Application of Hydrochar with Low Adsorption Capacity of Cr (VI)
4.1.2. Application of Hydrochar with High Adsorption Capacity of Cr (VI)
| Raw Materials | Synthesis Conditions | Material Usage | pH | Kinetics | Isotherm | Absorption Time | Adsorption Capacity | References |
|---|---|---|---|---|---|---|---|---|
| Pine wood chips | 240 °C, 4 h | 10 g/L | 1–3 | Pseudo-second-order | Langmuir | 24 h | —— | [99] |
| Chitosan powder | 180 °C, 12 h | 1.34 g/L | 1–3 | Pseudo-second-order | Freundlich | 0.5 h | 42.50 mg/g | [93] |
| Chitosan | 180 °C, 10 h | 0.5 g/L | 4 | Pseudo-first-order | Langmuir | 12 h | 388.6 mg/g | [105] |
| Blue algae | 240 °C, 1 h | 5 g/L | 7 | Pseudo-second-order | —— | 4 h | 1.982 mg/g | [97] |
| Bagasse | 190 °C, 12 h | 14 g/L | 2 | Pseudo-second-order | Langmuir | 1.5 h | 3.871 mg/g | [98] |
| Pomelo peel | 190 °C, 2 h | 5 g/L | 6 | Pseudo-second-order | Freundlich | 1.5 h | 6.19 mg/g | [95] |
| Lignin | 180 °C | 1 g/L | 2 | Pseudo-second-order | Freundlich | 72 h | 859.43 mg/g | [104] |
| Bamboo powder | 200 °C, 24 h | 1 g/L | 2 | Pseudo-second-order | Freundlich | 7 h | 195.76 mg/g | [100] |
| Hemicellulose | 220 °C, 12 h | 4 g/L | 2 | Pseudo-second-order | Langmuir | 12 h | 349.6 mg/g | [101] |
4.2. Application of Hydrochar in the Adsorption of Cd (II)
4.2.1. Application of Hydrochar with Low Adsorption Capacity of Cd (II)
4.2.2. Application of Hydrochar with High Adsorption Capacity of Cd (II)
| Raw Materials | Material Usage | pH | Kinetics | Isotherm | Absorption Time | Adsorption Amount | References |
|---|---|---|---|---|---|---|---|
| Strain P. chrysogenum | 1 g/L | 5 | Pseudo-second-order | Langmuir | 5 h | 203 mg/g | [120] |
| Hydrotalcite | 50 g/L | —— | Pseudo-second-order | —— | 1 h | 1035.4 mg/g | [117] |
| Titanate nanotubes | 0.2 g/L | 5–6 | Pseudo-second-order | Langmuir | 3 h | 238.61 mg/g | [116] |
| MgAl-LDH/chitosan | 1 g/L | 7 | Pseudo-second-order | Langmuir | 1 h | 39.7 mg/g | [125] |
| Hydrotalcite, glucose | 0.4 g/L | 5 | —— | Langmuir | 6 h | 475 mg/g | [124] |
| Sawdust | 1.65 g/L | 8 | Pseudo-second-order | Langmuir | 24 h | 19.99 mg/g | [111] |
| Graphene Oxide | 3.5 g/L | 6 | Pseudo-second-order | Langmuir | 4 h | 45.1 mg/g | [108] |
| Glucose | 0.67 g/L | 6 | Pseudo-second-order | Langmuir | 24 h | 72.63 m/g | [112] |
| Sawdust | 2 g/L | 4–8 | Pseudo-second-order | Langmuir | 24 h | 40.78 mg/g | [114] |
5. Conclusions
6. Further Prospects
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Babu, S.; Rathore, S.S.; Singh, R.; Kumar, S.; Singh, V.K.; Yadav, S.K.; Yadav, V.; Raj, R.; Yadav, D.; Shekhawat, K.; et al. Exploring agricultural waste biomass for energy, food and feed production and pollution mitigation: A review. Biores. Technol. 2022, 360, 127566. [Google Scholar] [CrossRef] [PubMed]
- Dimitriadis, A.; Bezergianni, S. Hydrothermal liquefaction of various biomass and waste feedstocks for biocrude production: A state of the art review. Renew. Sustain. Energy Rev. 2017, 68, 113–125. [Google Scholar] [CrossRef]
- Wu, F.; Li, F.; Zhao, X.; Bolan, N.S.; Fu, P.; Lam, S.S.; Wang, H. Meet the challenges in the “Carbon Age”. Carbon Res. 2022, 1, 1. [Google Scholar] [CrossRef]
- Hasan, M.M.; Rasul, M.G.; Khan, M.M.K.; Ashwath, N.; Jahirula, M.I. Energy recovery from municipal solid waste using pyrolysis technology: A review on current status and developments. Renew. Sustain. Energy Rev. 2021, 145, 111073. [Google Scholar] [CrossRef]
- Ge, D.; Yuan, H.; Xiao, J.; Zhu, N. Insight into the enhanced sludge dewaterability by tannic acid conditioning and pH regulation. Sci. Total Environ. 2019, 679, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Guan, Q.; Zeng, G.; Song, J.; Liu, C.; Wang, Z.; Wu, S. Ultrasonic power combined with seed materials for recovery of phosphorus from swine wastewater via struvite crystallization process. J. Environ. Manag. 2021, 293, 112961. [Google Scholar] [CrossRef]
- Gössling, S. Carbon neutral destinations: A conceptual analysis. J. Sustain. Tour. 2009, 17, 17–37. [Google Scholar] [CrossRef]
- Zou, C.N.; Xiong, B.; Xue, H.Q.; Zheng, D.W.; Ge, Z.X.; Wang, Y.; Jiang, L.Y.; Pan, S.Q.; Wu, S.T. The role of new energy in carbon neutral. Pet. Explor. Dev. 2021, 48, 480–491. [Google Scholar] [CrossRef]
- Liu, K.; Ran, Q.; Li, F. Carbon-based strategy enables sustainable remediation of paddy soils in harmony with carbon neutrality. Carbon Res. 2022, 1, 12. [Google Scholar] [CrossRef]
- Mallapaty, S. How China could be carbon neutral by mid-century. Nature 2020, 586, 482–484. [Google Scholar] [CrossRef]
- Gunarathne, V.; Ashiq, A.; Ramanayaka, S.; Wijekoon, P.; Vithanage, M. Biochar from municipal solid waste for resource recovery and pollution remediation. Environ. Chem. Lett. 2019, 17, 1225–1235. [Google Scholar] [CrossRef]
- Sansaniwal, S.K.; Rosen, M.A.; Tyagi, S.K. Global challenges in the sustainable development of biomass gasification: An overview. Renew. Sustain. Energy Rev. 2017, 80, 23–43. [Google Scholar] [CrossRef]
- Harder, R.; Wielemaker, R.; Larsen, T.A.; Zeeman, G.; Öberg, G. Recycling nutrients contained in human excreta to agriculture: Pathways, processes, and products. Crit. Rev. Environ. Sci. Technol. 2019, 49, 695–743. [Google Scholar] [CrossRef] [Green Version]
- Ali, A.; Zhao, C. Direct liquefaction techniques on lignite coal: A review. Chin. J. Catal. 2020, 41, 375–389. [Google Scholar] [CrossRef]
- Li, H.; Zhang, T.; Tsang, D.C.; Li, G. Effects of external additives Biochar, bentonite, phosphate, on co-composting for swine manure and corn straw. Chemosphere 2020, 248, 125927. [Google Scholar] [CrossRef]
- Li, H.; Zhang, T.; Shaheen, S.M.; Abdelrahman, H.; Ali, E.F.; Bolan, N.S.; Rinklebe, J. Microbial inoculants and struvite improved organic matter humification and stabilized phosphorus during swine manure composting: Multivariate and multiscale investigations. Biores. Technol. 2022, 351, 126976. [Google Scholar] [CrossRef]
- Tan, Z.; Dong, B.; Xing, M.; Sun, X.; Xi, B.; Dai, W.; He, C.; Luo, Y.; Huang, Y. Electric field applications enhance the electron transfer capacity of dissolved organic matter in sludge compost. Environ. Technol. 2022, in press. [Google Scholar] [CrossRef]
- Tan, Z.; Zhu, H.; He, X.; Xi, B.; Tian, Y.; Sun, X.; Zhang, H.; Ouche, Q. Effect of ventilation quantity on electron transfer capacity and spectral characteristics of humic substances during sludge composting. Environ. Sci. Pollut. Res. 2022, 29, 70269–70284. [Google Scholar] [CrossRef]
- Nogueira, C.C.; de Araújo Padilha, C.E.; de Souza Filho, P.F.; dos Santos, E.S. Effects of the addition of poly (ethylene glycol) and non-ionic surfactants on pretreatment, enzymatic hydrolysis, and ethanol fermentation. BioEnergy Res. 2022, 15, 889–904. [Google Scholar] [CrossRef]
- Kasinath, A.; Fudala-Ksiazek, S.; Szopinska, M.; Bylinski, H.; Artichowicz, W.; Remiszewska-Skwarek, A.; Luczkiewicz, A. Biomass in biogas production: Pretreatment and codigestion. Renew. Sustain. Energy Rev. 2021, 150, 111509. [Google Scholar] [CrossRef]
- Wang, Q.; Sha, H.; Cao, S.; Zhao, B.; Wang, G.; Zheng, P. Tourmaline enhanced methane yield via regulating microbial metabolic balance during anaerobic co-digestion of corn stover and cow manure. Biores. Technol. 2022, 359, 127470. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, X.; Zhang, H.; Chu, C.; Zheng, K.; Ju, M.; Liu, L. Liquefaction of biomass and upgrading of bio-oil: A review. Molecules 2019, 24, 2250. [Google Scholar] [CrossRef] [Green Version]
- Esposito, R.; Melchiorre, M.; Annunziata, A.; Cucciolito, M.E.; Ruffo, F. Emerging catalysis in biomass valorisation: Simple Zn (II) catalysts for fatty acids esterification and transesterification. ChemCatChem 2020, 12, 5858–5879. [Google Scholar] [CrossRef]
- Bollakayala, V.L.; Etakula, N.; Vuba, K.K.; Kasala, P.; Naidu, A. Sustainable Use of Recycled Polymer and Biomass Materials in the Preparation of Composites for Possible Applications. IJMTST 2022, 8, 445–451. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, E.; Mishra, R.; Kumar, S. Biochar as environmental armour and its diverse role towards protecting soil, water and air. Sci. Total Environ. 2022, 806, 150444. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, Z.; Xu, L.; Buyong, F.; Chay, T.C.; Li, Z.; Wang, X. Modified biochar: Synthesis and mechanism for removal of environmental heavy metals. Carbon Res. 2022, 1, 8. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, modification and environmental application of biochar: A review. J. Clean Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, Q.; Deng, Y.; Jiang, R. Recovery of phosphorus from swine manure by ultrasound/H2O2 digestion, struvite crystallization, and ferric oxide hydrate/biochar adsorption. Front. Chem. 2018, 6, 464. [Google Scholar] [CrossRef] [Green Version]
- Qin, Y.; Xi, B.; Sun, X.; Zhang, H.; Xue, C.; Wu, B. Methane emission reduction and biological characteristics of landfill cover soil amended with hydrophobic biochar. Front. Bioeng. Biotechnol. 2022, 10, 905466. [Google Scholar] [CrossRef] [PubMed]
- Puziy, A.M.; Poddubnaya, O.I.; Sevastyanova, O. Carbon materials from technical lignins: Recent advances. Lignin Chem. 2018, 376, 95–128. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Zhang, T.; Li, P.; Jiang, R.F.; Wang, Y.C. Application of magnesium modified corn biochar for phosphorus removal and recovery from swine wastewater. Int. J. Environ. Res. Public Health 2014, 11, 9217–9237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.; Li, L.; Tu, H.; Zhang, H.; Silvester, D.S.; Banks, C.E.; Ji, X. The development of carbon dots: From the perspective of materials chemistry. Mater. Today 2021, 51, 188–207. [Google Scholar] [CrossRef]
- Fang, C.; Zhang, T.; Li, P.; Jiang, R.; Wu, S.; Nie, H.; Wang, Y. Phosphorus recovery from biogas fermentation liquid by Ca-Mg loaded biochar. J. Environ. Sci. 2015, 29, 106–114. [Google Scholar] [CrossRef]
- He, X.; Zhang, T.; Ren, H.; Li, G.; Ding, L.; Pawlowski, L. Phosphorus recovery from biogas slurry by ultrasound/H2O2 digestion coupled with HFO/biochar adsorption process. Waste Manag. 2017, 60, 219–229. [Google Scholar] [CrossRef]
- Khitab, A.; Ahmad, S.; Khan, R.A.; Arshad, M.T.; Anwar, W.; Tariq, J.; Tariq, Z. Production of Biochar and Its Potential Application in Cementitious Composites. Crystals 2021, 11, 527. [Google Scholar] [CrossRef]
- Khan, T.A.; Saud, A.S.; Jamari, S.S.; Ab Rahim, M.H.; Park, J.W.; Kim, H.J. Hydrocharization of lignocellulosic biomass for carbon rich material preparation: A review. Biomass Bioenergy 2019, 130, 105384. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, S.; Cui, D.; Zhou, H.; Wu, D.; Pan, S.; Xu, F.; Wang, Z. Co-hydrothermal carbonization of organic solid wastes to hydrochar as potential fuel: A review. Sci. Total Environ. 2022, 850, 158034. [Google Scholar] [CrossRef]
- Funke, A.; Ziegler, F. Hydrothermal carbonization of biomass: A summary and discussion of chemical mechanisms for process engineering. Biofuel Bioprod. Biores. 2010, 4, 160–177. [Google Scholar] [CrossRef]
- He, X.; Zheng, N.; Hu, R.; Hu, Z.; Yu, J.C. Hydrothermal and pyrolytic conversion of biomasses into catalysts for advanced oxidation treatments. Adv. Funct. Mater. 2021, 31, 2006505. [Google Scholar] [CrossRef]
- Kambo, H.S.; Dutta, A. Strength, storage, and combustion characteristics of densified lignocellulosic biomass produced via torrefaction and hydrocharization. Appl. Energy 2014, 135, 182–191. [Google Scholar] [CrossRef]
- Liu, Z.; Balasubramanian, R. Upgrading of waste biomass by hydrocharization (HTC) and low temperature pyrolysis (LTP): A comparative evaluation. Appl. Energy 2013, 114, 857–864. [Google Scholar] [CrossRef]
- Zhang, T.; Wu, X.; Shaheen, S.M.; Zhao, Q.; Liu, X.; Rinklebe, J.; Ren, H. Ammonium nitrogen recovery from digestate by hydrothermal pretreatment followed by activated hydrochar sorption. Chem. Eng. J. 2020, 379, 122254. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, T.; Sharma, B.K.; Nie, H. Optimization and mechanism studies on cell disruption and phosphorus recovery from microalgae with magnesium modified hydrochar in assisted hydrothermal system. Sci. Total Environ. 2019, 646, 1140–1154. [Google Scholar] [CrossRef]
- Shi, Y.; Zhang, T.; Ren, H.; Kruse, A.; Cui, R. Polyethylene imine modified hydrochar adsorption for chromium (VI) and nickel (II) removal from aqueous solution. Biores. Technol. 2018, 247, 370–379. [Google Scholar] [CrossRef]
- Titirici, M.M.; Antonietti, M. Chemistry and materials options of sustainable carbon materials made by hydrocharization. Chem. Soc. Rev. 2010, 39, 103–116. [Google Scholar] [CrossRef]
- Jain, A.; Balasubramanian, R.; Srinivasan, M.P. Hydrothermal conversion of biomass waste to activated carbon with high porosity: A review. Chem. Eng. J. 2016, 283, 789–805. [Google Scholar] [CrossRef]
- Lan, Y.; Du, Q.; Tang, C.; Cheng, K.; Yang, F. Application of typical artificial carbon materials from biomass in environmental remediation and improvement: A review. J. Environ. Manag. 2021, 296, 113340. [Google Scholar] [CrossRef]
- Xiao, Y.; Raheem, A.; Ding, L.; Chen, W.H.; Chen, X.; Wang, F.; Lin, S.L. Pretreatment, modification and applications of sewage sludge-derived biochar for resource recovery—A review. Chemosphere 2022, 287, 131969. [Google Scholar] [CrossRef]
- Roman, S.; Nabais, J.V.; Ledesma, B.; González, J.F.; Laginhas, C.; Titirici, M.M. Production of low-cost adsorbents with tunable surface chemistry by conjunction of hydrocharization and activation processes. Micropor. Mesopor. Mat. 2013, 165, 127–133. [Google Scholar] [CrossRef]
- de Castro, A.R.; Saraiva, G.D.; Oliveira, A.C.; Neto, V.S.; Paula, A.J.; Souza Filho, A.G.; Ferreira, O.P. Ordered porous carbons from hydrothermally treated biomass: Effects of the thermal treatments on the structure and porosity. Vib. Spectrosc. 2020, 111, 103175. [Google Scholar] [CrossRef]
- Jain, A.; Balasubramanian, R.; Srinivasan, M.P. Production of high surface area mesoporous activated carbons from waste biomass using hydrogen peroxide-mediated hydrothermal treatment for adsorption applications. Chem. Eng. J. 2015, 273, 622–629. [Google Scholar] [CrossRef]
- Deng, Q.; Li, M.; Wang, J.; Jiang, K.; Hu, Z.; Chu, J. Carbonized polydopamine wrapping layered KNb3O8 nanoflakes based on alkaline hydrothermal for enhanced and discrepant lithium storage. J. Alloy. Compd. 2018, 749, 803–810. [Google Scholar] [CrossRef]
- Zhang, S.; Sheng, K.; Yan, W.; Liu, J.; Shuang, E.; Yang, M.; Zhang, X. Bamboo derived hydrochar microspheres fabricated by acid-assisted hydrothermal carbonization. Chemosphere 2021, 263, 128093. [Google Scholar] [CrossRef]
- Tang, Z.; Deng, Y.; Luo, T.; Xu, Y.S.; Zhu, N.M. Enhanced removal of Pb (II) by supported nanoscale Ni/Fe on hydrochar derived from biogas residues. Chem. Eng. J. 2016, 292, 224–232. [Google Scholar] [CrossRef]
- Jimenez-Cordero, D.; Heras, F.; Alonso-Morales, N.; Gilarranz, M.A.; Rodriguez, J.J. Ozone as oxidation agent in cyclic activation of biochar. Fuel Process. Technol. 2015, 139, 42–48. [Google Scholar] [CrossRef] [Green Version]
- Saeed, A.A.H.; Harun, N.Y.; Sufian, S.; Bilad, M.R.; Nufida, B.A.; Ismail, N.M.; Al-Dhawi, B.N.S. Modeling and optimization of biochar based adsorbent derived from Kenaf using response surface methodology on adsorption of Cd2+. Water 2021, 13, 999. [Google Scholar] [CrossRef]
- Saeed, A.A.H.; Harun, N.Y.; Sufian, S.; Ghaleb, A.A.S.; Jagaba, A.H.; Mohammed, H.G.; Abdulrab, H.Q.A. Removal of Cadmium (II) from Aqueous Solution by Rice Husk Waste. Int. Congr. Adv. Tech. Eng. 2021, 1–6. [Google Scholar] [CrossRef]
- Ababneh, H.; Hameed, B.H. Chitosan-derived hydrothermally carbonized materials and its applications: A review of recent literature. Int. J. Biol. Macromol. 2021, 186, 314–327. [Google Scholar] [CrossRef]
- Jiang, F.; Cao, D.; Hu, S.; Wang, Y.; Zhang, Y.; Huang, X.; Liu, K. High-pressure carbon dioxide-hydrothermal enhance yield and methylene blue adsorption performance of banana pseudo-stem activated carbon. Biores. Technol. 2022, 354, 127137. [Google Scholar] [CrossRef]
- Lv, B.W.; Xu, H.; Guo, J.Z.; Bai, L.Q.; Li, B. Efficient adsorption of methylene blue on carboxylate-rich hydrochar prepared by one-step hydrothermal carbonization of bamboo and acrylic acid with ammonium persulphate. J. Hazard Mater. 2022, 421, 126741. [Google Scholar] [CrossRef]
- Pan, C.; Fan, C.; Wang, W.; Long, T.; Huang, B.; Zhang, D.; Sun, Y. Micro-sized hydrothermal carbon supporting metal oxide nanoparticles as efficient catalyst for mono-dehydration of sugar alcohol. Front. Energy 2020, 2020, 1–18. [Google Scholar] [CrossRef]
- Wen, Z.; Ma, Z.; Mai, F.; Yan, F.; Yu, L.; Jin, M.; Li, Y. Catalytic ethanolysis of microcrystalline cellulose over a sulfonated hydrothermal carbon catalyst. Catal. Today 2020, 355, 272–279. [Google Scholar] [CrossRef]
- Hou, J.; Guo, Z.; Meng, F.; Li, M.; Hou, L.A. Restoration of organic-matter-impoverished arable soils through the application of soil conditioner prepared via short-time hydrothermal fermentation. Environ. Res. 2022, 204, 112088. [Google Scholar] [CrossRef]
- Scheifele, M.; Hobi, A.; Buegger, F.; Gattinger, A.; Schulin, R.; Boller, T.; Mäder, P. Impact of pyrochar and hydrochar on soybean (Glycine max L.) root nodulation and biological nitrogen fixation. J. Plant Nutr. Soil Sci. 2017, 180, 199–211. [Google Scholar] [CrossRef]
- Jagaba, A.H.; Kutty, S.R.M.; Hayder, G.; Baloo, L.; Ghaleb, A.A.S.; Lawal, I.M.; Umaru, I. Degradation of Cd, Cu, Fe, Mn, Pb and Zn by Moringa-oleifera, zeolite, ferric-chloride, chitosan and alum in an industrial effluent. Ain Shams Eng. J. 2021, 12, 57–64. [Google Scholar] [CrossRef]
- Al-mahbashi, N.; Kutty, S.R.M.; Jagaba, A.H.; Al-Nini, A.; Ali, M.; Saeed, A.A.H.; Rathnayake, U. Column Study for Adsorption of Copper and Cadmium Using Activated Carbon Derived from Sewage Sludge. Adv. Civ. Eng. 2022, 2022, 3590462. [Google Scholar] [CrossRef]
- Poomsawat, S.; Poomsawat, W. Analysis of hydrochar fuel characterization and combustion behavior derived from aquatic biomass via hydrothermal carbonization process. Case Stud. Therm. Eng. 2021, 27, 101255. [Google Scholar] [CrossRef]
- Parshetti, G.K.; Hoekman, S.K.; Balasubramanian, R. Chemical, structural and combustion characteristics of carbonaceous products obtained by hydrothermal carbonization of palm empty fruit bunches. Biores. Technol. 2013, 135, 683–689. [Google Scholar] [CrossRef]
- Hussin, F.; Aroua, M.K.; Szlachta, M. Biochar derived from fruit by-products using pyrolysis process for the elimination of Pb (II) ion, An updated review. Chemosphere 2022, 287, 132250. [Google Scholar] [CrossRef]
- Wei, C.; Xiang, C.; Ren, E.; Cui, C.; Zhou, M.; Xiao, H.; Guo, R. Synthesis of 3D lotus biochar/reduced graphene oxide aerogel as a green adsorbent for Cr (VI). Mater. Chem. Phys. 2020, 253, 123271. [Google Scholar] [CrossRef]
- Li, S.; Yang, F.; Li, J.; Cheng, K. Porous biochar-nanoscale zero-valent iron composites: Synthesis, characterization and application for lead ion removal. Sci. Total Environ. 2020, 746, 141037. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.C.; Lee, C.K.; Han, Y.L.; Chao, W.C.; Chao, H.P. Preparation of activated carbon using micro-nano carbon spheres through chemical activation. J. Taiwan Inst. Chem. E 2014, 45, 2805–2812. [Google Scholar] [CrossRef]
- Zhang, N.; Pan, J.; Cheng, F. Preparation of carbon spheres/lignin materials and adsorption of toxic metal ions. Desalination Water Treat. 2021, 237, 214–225. [Google Scholar] [CrossRef]
- Lu, X.; Jiang, J.; Sun, K.; Zhu, G.; Lin, G. Enhancement of Pb2+ removal by activating carbon spheres/activated carbon composite material with H2O vapor. Colloid Surf. A 2016, 506, 637–645. [Google Scholar] [CrossRef]
- Xie, Y.; Yuan, X.; Wu, Z.; Zeng, G.; Jiang, L.; Peng, X.; Li, H. Adsorption behavior and mechanism of Mg/Fe layered double hydroxide with Fe3O4-carbon spheres on the removal of Pb (II) and Cu (II). J. Colloid Interf. Sci. 2019, 536, 440–455. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, J.; Wen, T.; Liu, X.; Wang, Y.; Yang, H.; Sun, J. Highly effective remediation of Pb (II) and Hg (II) contaminated wastewater and soil by flower-like magnetic MoS2 nanohybrid. Sci. Total Environ. 2020, 699, 134341. [Google Scholar] [CrossRef]
- Song, X.; Gunawan, P.; Jiang, R.; Leong, S.S.J.; Wang, K.; Xu, R. Surface activated carbon nanospheres for fast adsorption of silver ions from aqueous solutions. J. Hazard Mater. 2011, 194, 162–168. [Google Scholar] [CrossRef]
- Xue, Y.; Teng, W.; Chen, Y.; Ma, Q.; Chen, X.; Sun, Y.; Fu, R. Amorphous Mn-La oxides immobilized on carbon sphere for efficient removal of As (V), Cd (II), and Pb (II): Co-adsorption and roles of Mn species. Chem. Eng. J. 2022, 429, 132262. [Google Scholar] [CrossRef]
- Hu, Z.; Shen, Z.; Yu, J.C. Converting carbohydrates to carbon-based photocatalysts for environmental treatment. Environ. Sci. Technol. 2017, 51, 7076–7083. [Google Scholar] [CrossRef]
- Zhang, Z.B.; Zhou, Z.W.; Cao, X.H.; Liu, Y.H.; Xiong, G.X.; Liang, P. Removal of uranium (VI) from aqueous solutions by new phosphorus-containing carbon spheres synthesized via one-step hydrothermal carbonization of glucose in the presence of phosphoric acid. J. Radioanal. Nucl. Chem. 2014, 299, 1479–1487. [Google Scholar] [CrossRef]
- Feng, J.; Gao, M.; Zhang, Z.; Liu, S.; Zhao, X.; Ren, Y.; Fan, Z. Fabrication of mesoporous magnesium oxide nanosheets using magnesium powder and their excellent adsorption of Ni (II). J. Colloid Interf. Sci. 2018, 510, 69–76. [Google Scholar] [CrossRef]
- Zhou, Z.W.; Xiong, G.X.; Liu, Y.H.; Cao, X.H.; Zhang, Z.B. Removal of thorium (IV) from aqueous solutions by carboxyl-rich hydrothermal carbon spheres through low-temperature heat treatment in air. Desalination Water Treat. 2015, 54, 2516–2529. [Google Scholar] [CrossRef]
- He, X.; Zhang, T.; Xue, Q.; Zhou, Y.; Wang, H.; Bolan, N.S.; Tsang, D.C. Enhanced adsorption of Cu (II) and Zn (II) from aqueous solution by polyethyleneimine modified straw hydrochar. Sci. Total Environ. 2021, 778, 146116. [Google Scholar] [CrossRef]
- Liu, W.; Huang, F.; Liao, Y.; Zhang, J.; Ren, G.; Zhuang, Z.; Zhen, J.; Lin, Z.; Wang, C. Treatment of CrVI-containing Mg(OH)2 nanowaste. Angew. Chem. Int. Ed. 2008, 47, 5619–5622. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, J.; Ou, X.; Liu, X.; Song, Y.; Tian, C.; Rong, W.; Shi, Z.; Dang, Z.; Lin, Z. Effective extraction of Cr(VI) from hazardous gypsum sludge via controlling the phase transformation and chromium species. Environ. Sci. Technol. 2018, 52, 13336–13342. [Google Scholar] [CrossRef]
- Available online: https://cdn.dwi.gov.uk/wp-content/uploads/2020/11/26112406/uksi_20160614_en.pdf (accessed on 1 October 2022).
- Available online: https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/water-quality/guidelines-canadian-drinking-water-quality-summary-table.html (accessed on 1 October 2022).
- Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations#Inorganic (accessed on 1 October 2022).
- Available online: https://www.moh.govt.nz/notebook/nbbooks.nsf/0/B9917ABBB22BE387CC2583B2007928FE/$file/dwsnz-2005-revised-mar2019.pdf (accessed on 1 October 2022).
- Available online: https://www.nhmrc.gov.au/about-us/publications/australian-drinking-water-guidelines (accessed on 1 October 2022).
- Available online: https://www.who.int/publications/i/item/9789241549950 (accessed on 1 October 2022).
- Sinha, V.; Pakshirajan, K.; Chaturvedi, R. Chromium tolerance, bioaccumulation and localization in plants: An overview. J. Environ. Manag. 2018, 206, 715–730. [Google Scholar] [CrossRef]
- Yi, R.; Cai, W.; Dang, C.; Han, B.; Liu, L.; Fan, J. Mild hydrothermal preparation of millimeter-sized carbon beads from chitosan with significantly improved adsorption stability for Cr (VI). Chem. Eng. Res. Des. 2020, 156, 43–53. [Google Scholar] [CrossRef]
- Liu, M.X.; Zeng, R.Y. Study on adsorption properties of carbonate hydroxyapatite for Cr(VI)in simulative wastewater. Environ. Pollut. Prev. 2009, 31, 55–58. [Google Scholar] [CrossRef]
- Zhang, S.J.; Xing, B.L.; Huang, G.X.; Liu, Q.R.; Jia, J.B.; Duan, Y.L.; Zhang, C.X. Adsorption of Cr(VI) by hydrothermal carbon from shaddock peel. J. Environ. Eng. 2017, 11, 2731–2737. [Google Scholar] [CrossRef]
- Gong, H.; Li, H.; Tan, L.; Liu, M. In situ growth of layered double hydroxides on sawdust for Pb (II) adsorption. Chem. Sel. 2019, 4, 5386–5393. [Google Scholar] [CrossRef]
- Yao, B.Y.; Liu, Y.Z.; Zhao, M.H.; Gao, T.Y.; Zhao, Y.X. Preparation of cyanobacteria-based hydrochar and its adsorption of hexavalent chromium. Environ. Prot. Circ. Econ. 2019, 39, 19–23. [Google Scholar] [CrossRef]
- Liu, X.M.; Feng, C.; Wu, F.; Zhao, B. Adsorption of hexavalent chromium in electroplating wastewater on bagasse-based hydrothermally synthesized carbon. Electroplat. Finish. 2018, 37, 738–744. [Google Scholar] [CrossRef]
- Wang, X.; He, Y.J.; Xu, W.; Duan, J.N.; Li, D.X. Adsorption of Cr(VI) by amino hydrothermal carbon. Appl. Chem. Ind. 2021, 50, 290–294+323. [Google Scholar] [CrossRef]
- Li, S.Y.; Teng, H.J.; Guo, J.Z.; Wang, Y.X.; Li, B. Enhanced removal of Cr (VI) by nitrogen-doped hydrochar prepared from bamboo and ammonium chloride. Biores. Technol. 2021, 342, 126028. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Wang, H.; Zhang, X.; Liu, C. Ammonia-assisted hydrochar material with schiff base structures synthesized from factory waste hemicelluloses for Cr (VI) adsorption. J. Environ. Chem. Eng. 2021, 9, 106187. [Google Scholar] [CrossRef]
- Poudel, M.B.; Awasthi, G.P.; Kim, H.J. Novel insight into the adsorption of Cr (VI) and Pb (II) ions by MOF derived Co-Al layered double hydroxide@ hematite nanorods on 3D porous carbon nanofiber network. Chem. Eng. J. 2021, 417, 129312. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, C.; Zan, Y.; Miao, G.; Wang, H.; Kong, L. Hydrocharization of microalgae (Chlorococcum sp.) for porous carbons with high Cr (VI) adsorption performance. Appl. Biochem. Biotech. 2018, 186, 414–424. [Google Scholar] [CrossRef]
- Tian, Y.; Yin, Y.; Liu, H.; Zhou, H. One-step hydrocharization of amine modified black liquor and lignin for efficient Cr (VI) adsorption. J. Water Process. Eng. 2022, 46, 102583. [Google Scholar] [CrossRef]
- Shen, F.; Su, J.; Zhang, X.; Zhang, K.; Qi, X. Chitosan-derived carbonaceous material for highly efficient adsorption of Cr (VI) from aqueous solution. Int. J. Biol. Macromol. 2016, 91, 443–449. [Google Scholar] [CrossRef]
- Mahajan, P.; Kaushal, J. Role of phytoremediation in reducing cadmium toxicity in soil and water. J. Toxicol. 2018, 2018, 4864365. [Google Scholar] [CrossRef]
- Jagaba, A.H.; Kutty, S.R.M.; Hayder, G.; Baloo, L.; Abubakar, S.; Ghaleb, A.A.S.; Almahbashi, N.M.Y. Water quality hazard assessment for hand dug wells in Rafin Zurfi, Bauchi State, Nigeria. Ain Shams Eng. J. 2020, 11, 983–999. [Google Scholar] [CrossRef]
- Huang, Q.; Chen, Y.; Yu, H.; Yan, L.; Zhang, J.; Wang, B.; Xing, L. Magnetic graphene oxide/MgAl-layered double hydroxide nanocomposite: One-pot solvothermal synthesis, adsorption performance and mechanisms for Pb2+, Cd2+, and Cu2+. Chem. Eng. J. 2018, 341, 1–9. [Google Scholar] [CrossRef]
- Liu, Y.; Fu, R.; Lou, Z.; Fang, W.; Wang, Z.; Xu, X. Preparation of functional carbon-based materials for removal of heavy metals from aqueous solution. Prog Chem. 2015, 27, 1665. [Google Scholar] [CrossRef]
- Saeed, A.A.H.; Harun, N.Y.; Sufian, S.; Bilad, M.R.; Zakaria, Z.Y.; Jagaba, A.H.; Mohammed, H.G. Pristine and magnetic kenaf fiber biochar for Cd2+ adsorption from aqueous solution. Int. J. Environ. Res. Public Health 2021, 18, 7949. [Google Scholar] [CrossRef]
- Li, D.; Cui, H.; Cheng, Y.; Xue, L.; Wang, B.; He, H.; Yang, L. Chemical aging of hydrochar improves the Cd2+ adsorption capacity from aqueous solution. Environ. Pollut. 2021, 287, 117562. [Google Scholar] [CrossRef]
- Zhou, X.; You, S.J.; Wang, X.H.; Gan, Y.; Zhong, Y.J.; Ren, N.Q. Hydrothermal synthesis of magnetic carbon microspheres for effective adsorption of Cd (II) in water. J. Chem. Technol. Biotechnol. 2014, 89, 1051–1059. [Google Scholar] [CrossRef]
- Xiong, T.; Yuan, X.; Chen, X.; Wu, Z.; Wang, H.; Leng, L.; Zeng, G. Insight into highly efficient removal of cadmium and methylene blue by eco-friendly magnesium silicate-hydrochar composite. Appl. Surf. Sci. 2018, 427, 1107–1117. [Google Scholar] [CrossRef]
- Sun, K.; Tang, J.; Gong, Y.; Zhang, H. Characterization of potassium hydroxide (KOH) modified hydrochars from different feedstocks for enhanced removal of heavy metals from water. Environ. Sci. Pollut. Res. 2015, 22, 16640–16651. [Google Scholar] [CrossRef]
- Gusain, R.; Kumar, N.; Fosso-Kankeu, E.; Ray, S.S. Efficient removal of Pb (II) and Cd (II) from industrial mine water by a hierarchical MoS2/SH-MWCNT nanocomposite. ACS Omega 2019, 4, 13922–13935. [Google Scholar] [CrossRef] [Green Version]
- Xiong, L.; Chen, C.; Chen, Q.; Ni, J. Adsorption of Pb (II) and Cd (II) from aqueous solutions using titanate nanotubes prepared via hydrothermal method. J. Hazard. Mater. 2011, 189, 741–748. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, J.; Yu, J.; Yu, Q.; Qiu, X. Remediation of Cd-contaminated soil through different layered double hydroxides: The weakness of delamination and mechanism. J. Environ. Chem. Eng. 2022, 10, 107815. [Google Scholar] [CrossRef]
- Nworie, F.S.; Frank, N.I.; Oti, W.; Nduka, O.; Happiness, I. Hydrothermal synthesis of multifunctional Biochar-supported SALEN nanocomposite for adsorption of Cd (II) ions: Function, mechanism, equilibrium and kinetic studies. Anal. Bioanal. Chem. Res. 2021, 8, 91–112. [Google Scholar] [CrossRef]
- Liu, T.; Chen, Z.; Li, Z.; Fu, H.; Chen, G.; Feng, T.; Chen, Z. Preparation of magnetic hydrochar derived from iron-rich Phytolacca acinosa Roxb. for Cd removal. Sci. Total Environ. 2021, 769, 145159. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Ting, Y.P. Fungal biomass with grafted poly (acrylic acid) for enhancement of Cu (II) and Cd(II) biosorption. Langmuir 2005, 21, 5940–5948. [Google Scholar] [CrossRef] [PubMed]
- Laipan, M.; Zhu, J.; Xu, Y.; Sun, L.; Zhu, R. Fabrication of layered double hydroxide/carbon nanomaterial for heavy metals removal. Appl. Clay Sci. 2020, 199, 105867. [Google Scholar] [CrossRef]
- Zhang, Y.; Qu, J.; Yuan, Y.; Song, H.; Liu, Y.; Wang, S.; Li, Z. Simultaneous scavenging of Cd (II) and Pb (II) from water by sulfide-modified magnetic pinecone-derived hydrochar. J. Clean Prod. 2022, 341, 130758. [Google Scholar] [CrossRef]
- Chen, L.; Msigwa, G.; Yang, M.; Osman, A.I.; Fawzy, S.; Rooney, D.W.; Yap, P.S. Strategies to achieve a carbon neutral society: A review. Environ. Chem. Lett. 2022, 20, 2277–2310. [Google Scholar] [CrossRef]
- Zhang, J.W.; Nur’aini, A.D.; Wang, Y.C.; Hai, N.D.; Van Minh, D.; Chao, H.P. Multiple pollutants removal by carbon sphere and layered double hydroxide composites: Adsorption behavior and mechanisms. J. Environ. Chem. Eng. 2022, 10, 108014. [Google Scholar] [CrossRef]
- Lyu, F.; Yu, H.; Hou, T.; Yan, L.; Zhang, X.; Du, B. Efficient and fast removal of Pb2+ and Cd2+ from an aqueous solution using a chitosan/Mg-Al-layered double hydroxide nanocomposite. J. Colloid Interf. Sci. 2019, 539, 184–193. [Google Scholar] [CrossRef]
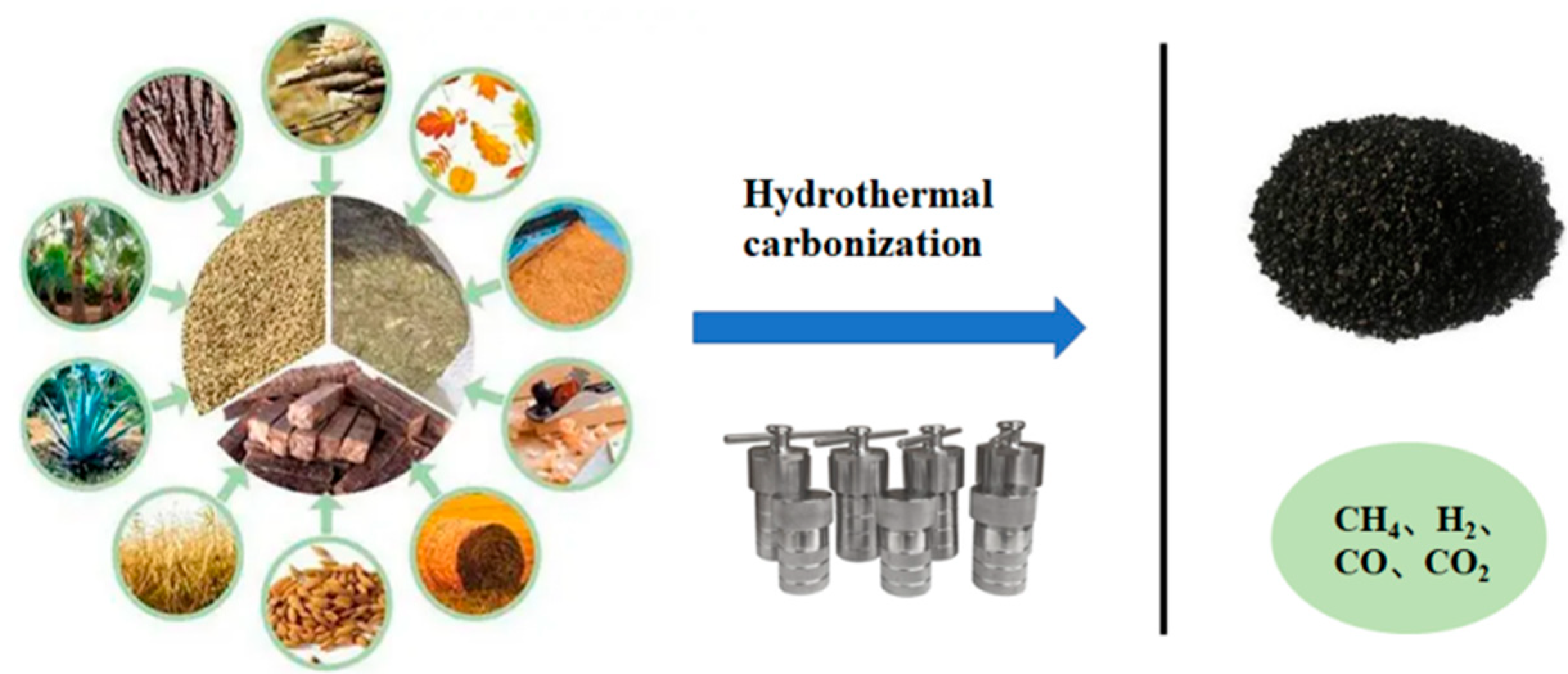
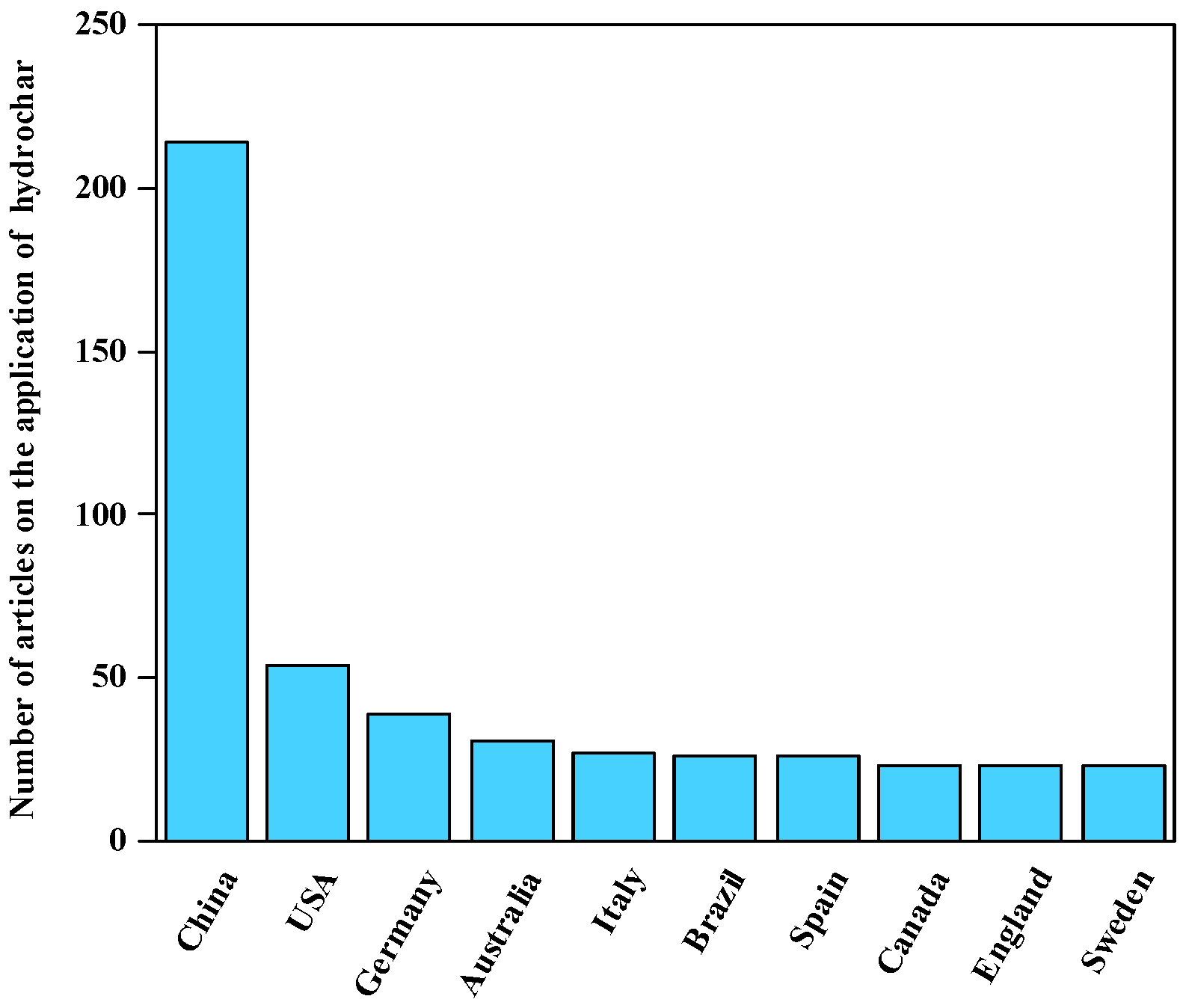
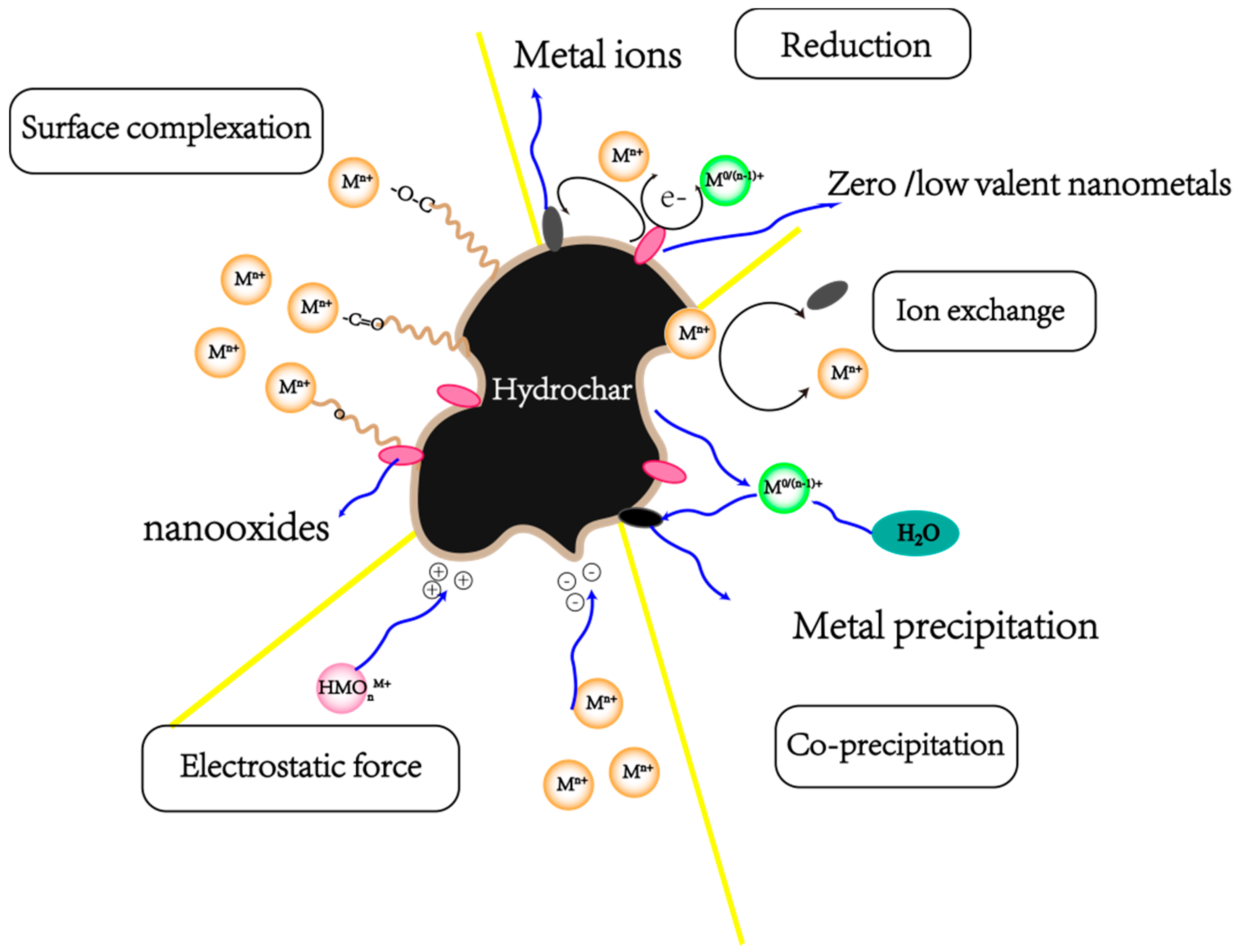
| Resource Utilization Methods | Specific Techniques | Final Products | References |
|---|---|---|---|
| Thermochemical method | Gasification | Biomass gas | [12] |
| Pyrolysis | Charcoal/Biocrude | [13] | |
| Direct liquefying | Liquefied oils | [14] | |
| Biochemical methods | Compost | Organic fertilizer | [15,16,17,18] |
| Hydrolysis; fermentation | Ethyl alcohol | [19] | |
| Biogas technology | Biogas | [20,21] | |
| Chemical method | Indirect liquefaction | Methanol and ethers | [22] |
| Esterification | Biodiesel | [23] | |
| Physical and chemical methods | Compression moulding | Fuel | [24] |
| Modification Methods | Specific Methods | Application Cases | References | |
|---|---|---|---|---|
| Physical modification | Gas activation | Treatment of hydrochar at temperatures exceeding 700 °C with a variety of gases such as steam, CO2, ozone, and so on. | When activated using CO2 and O2, the CO2 increases the specific surface area of the hydrochar, while the pore volume of the hydrochar is also increased. | [49] |
| Steam activation | Following the hydrothermal treatment, the pyrolysis steam activation was performed again. | A greater post-treatment temperature would raise the carbonaceous materials’ graphitization degree. | [50] | |
| Chemical modification | Add oxidant | Adding oxidants can enhance in the synthesis of oxygen functional groups on the surface of hydrochar, increasing its oxygen level. | Preparation of mesoporous activated carbon with a high specific surface area by using H2O2 as an oxidant during the raw material’s hydrothermal preparation. | [51] |
| Add alkaline substances | The groups in the base contribute to chelate the reaction with the metal ions. | After modification with alkaline substances, the surface area of the hydrochar increases and the volume of the total pores also increases. | [52] | |
| Add dilute acid | Increases the quantity of carboxylate and phosphate on the surface of hydrochar, resulting in increased adsorption capability. | Hydrothermal carbonization assisted by various acids such as hydrochloric acid, sulphuric acid, or nitric acid was used to produce successful carbon microspheres obtained from bamboo residues, where the addition of acid enhanced the hydrolysis process. | [53] | |
| Load metal salts or transition metal nanoparticles | Modification of hydrochar by immersion in a metal salt solution or by surface loading with a metal substance. | The synthesis of functionalized carbonaceous materials from biogas leftovers, as well as the loading of Ni/Fe onto hydrochar, resulted in a final product. | [54] | |
| Heavy Metal | Source of Hydrochar | Adsorption Capacity | Adsorption Kinetic | Adsorption Isotherm | References |
|---|---|---|---|---|---|
| Cr (VI) | Lotus stems | 232.56 mg/g | Pseudo-second-order | Langmuir | [70] |
| Pb (II) | Corn stalk | 480.9 mg/g | Pseudo-second-order | Langmuir | [71] |
| Cu (II) | Xylose | 42 mg/g | Langmuir | [72] | |
| Cu (II) | 338.976 mg/g | Pseudo-second-order | Langmuir | [75] | |
| Pb (II) | Lignin | Pseudo-second-order | Langmuir | [73] | |
| Hg (II) | Polrvidone | 428.9 mg/g | Pseudo-second-order | Langmuir | [74] |
| Ag (I) | 152 mg/g | Pseudo-second-order | Langmuir | [77] | |
| As (V) | 41.91 mg/g | Pseudo-second-order | Langmuir | [78] | |
| U (VI) | 285.70 mg/g | Pseudo-second-order | Langmuir | [80] | |
| Ni (II) | Mg(OH)2 | 2217 mg/g | Pseudo-second-order | Langmuir | [81] |
| Th (IV) | 69.93 mg/g | Pseudo-second-order | Langmuir | [82] | |
| Zn (II) | Corn stalk | 207.6 mg/g | Pseudo-second-order | Freundlich | [83] |
| Metal | Region | Standard | Harm | References |
|---|---|---|---|---|
| Cd | China | 5 ug/L | Pneumonitis; Mucous membrane destruction; Proteinuria; Glomerular and Tubular damage; Itai-Itai disease; Progesterone and Testosterone disturbance; Testicular necrosis. | GB 5749-2022 |
| Canada | 7 ug/L | [86] | ||
| WHO | 3 ug/L | [87] | ||
| NZ | 4 ug/L | [88] | ||
| AUS | 2 ug/L | [89] | ||
| USEPA | 5 ug/L | [90] | ||
| UK | 5 ug/L | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhang, T. Biowaste Valorization to Produce Advance Carbon Material-Hydrochar for Potential Application of Cr (VI) and Cd (II) Adsorption in Wastewater: A Review. Water 2022, 14, 3675. https://doi.org/10.3390/w14223675
Zhang Y, Zhang T. Biowaste Valorization to Produce Advance Carbon Material-Hydrochar for Potential Application of Cr (VI) and Cd (II) Adsorption in Wastewater: A Review. Water. 2022; 14(22):3675. https://doi.org/10.3390/w14223675
Chicago/Turabian StyleZhang, Yingyu, and Tao Zhang. 2022. "Biowaste Valorization to Produce Advance Carbon Material-Hydrochar for Potential Application of Cr (VI) and Cd (II) Adsorption in Wastewater: A Review" Water 14, no. 22: 3675. https://doi.org/10.3390/w14223675







