Investigating Landfill Leachate Influence on Soil Microbial Biodiversity and Its Cytotoxicity
Abstract
:1. Introduction
2. Materials and Methods
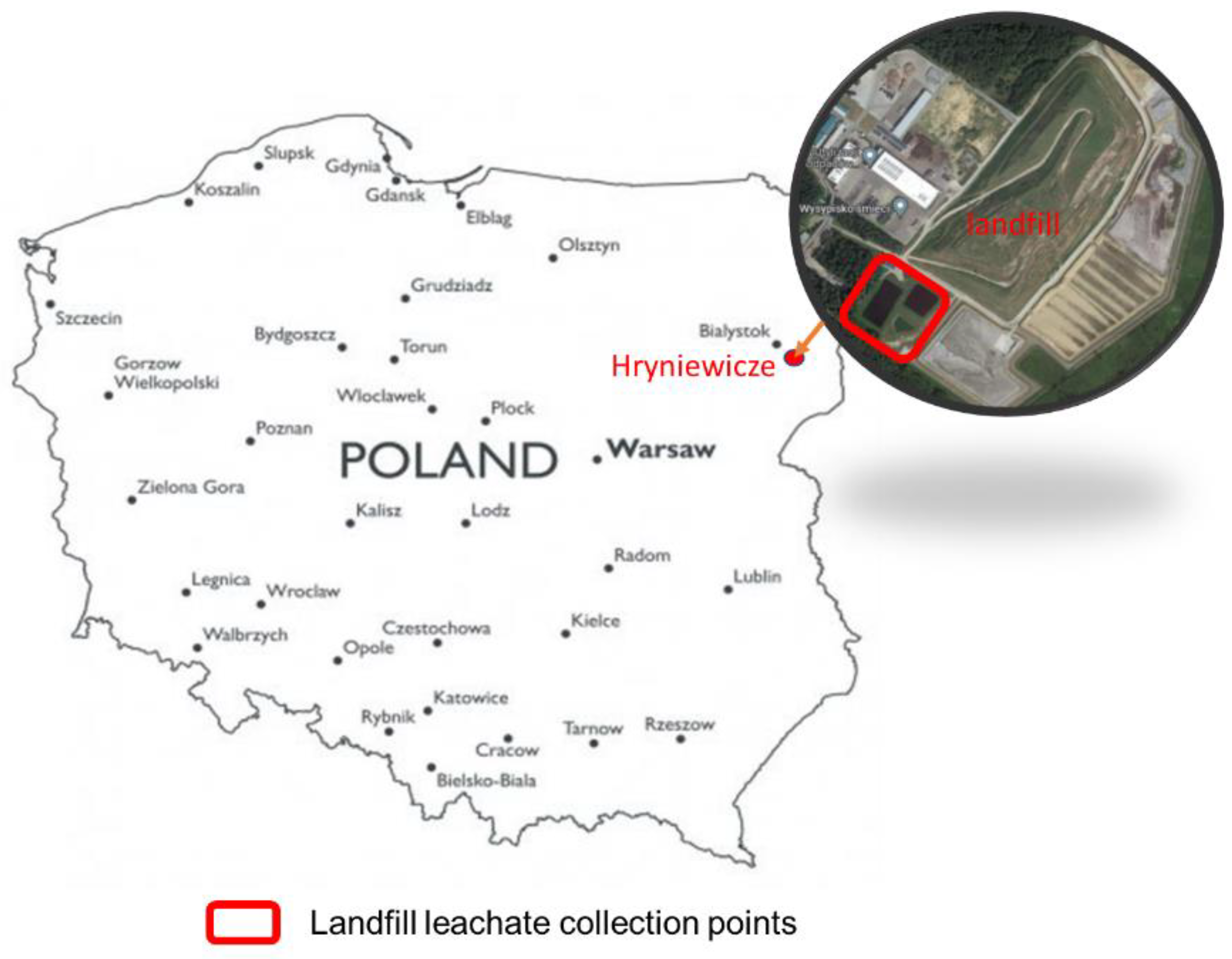
2.1. Landfill Leachate and Soil for Pot Experiment Characteristic
2.2. Pot Experiment and Sampling Preparation
2.3. Physicochemical Analyses of Soil and SF Samples
2.4. Bacteria Abundance and Soil Enzymatic Activity
2.5. DNA Extraction and Microbial Communities Fingerprinting by T-RFLP
2.6. Analysis of FL Cytotoxicity in Eukaryotic Cells
2.7. Study of the Level of Apoptosis
2.8. Statistical Analysis
3. Results
3.1. Physicochemical Properties of Soil and SF Samples
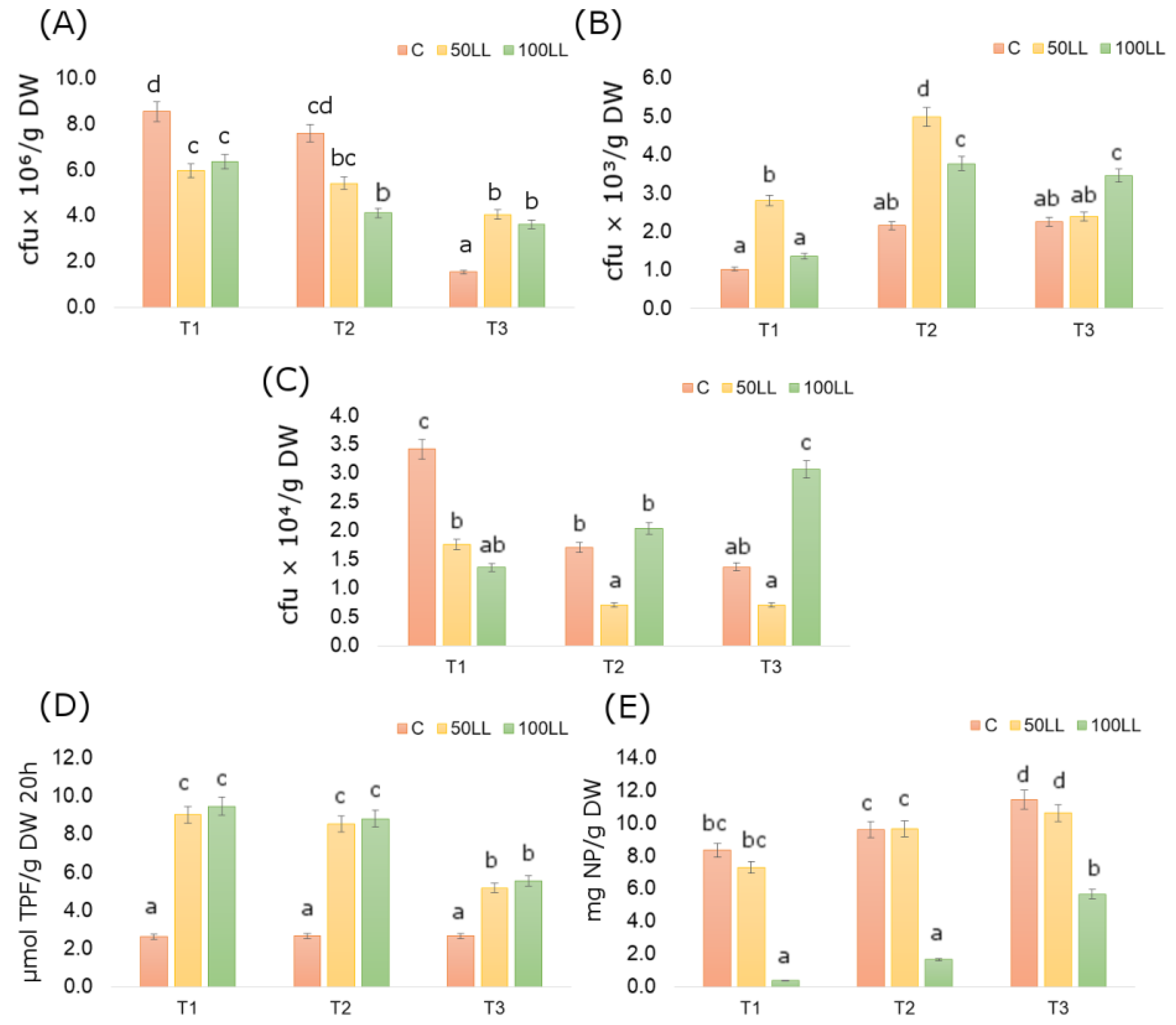
3.2. Bacteria Abundance and Soil Enzymatic Activity
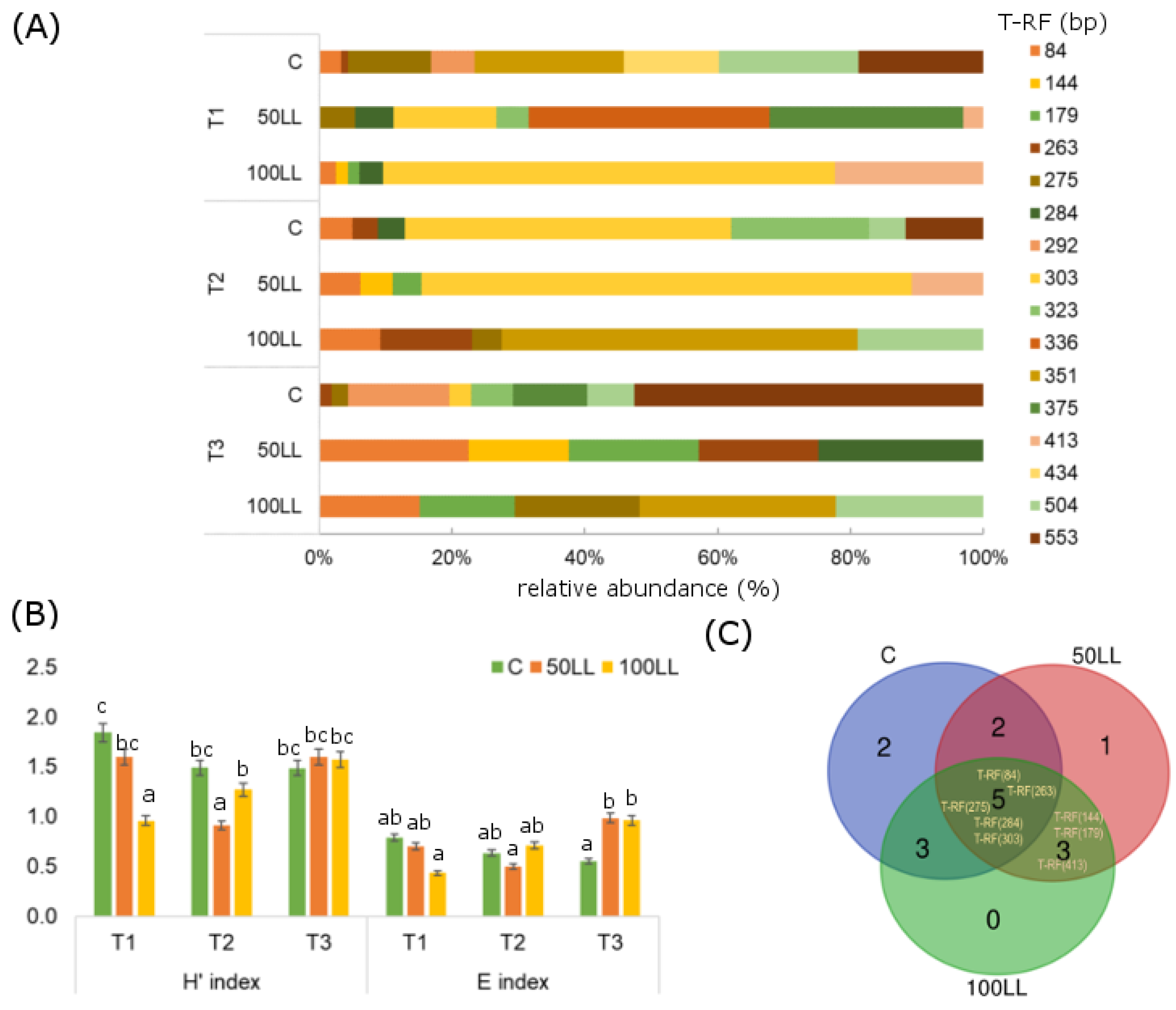
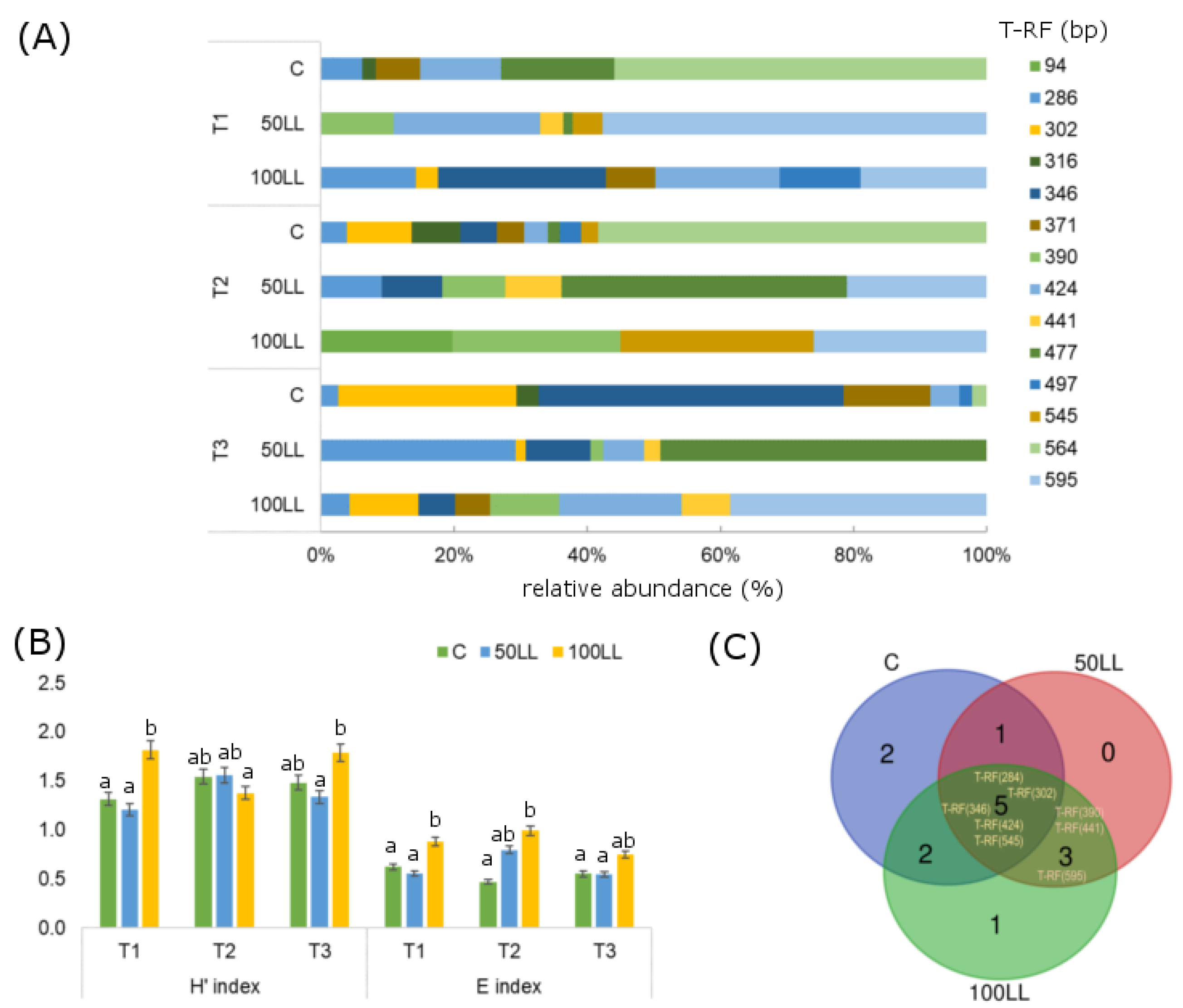
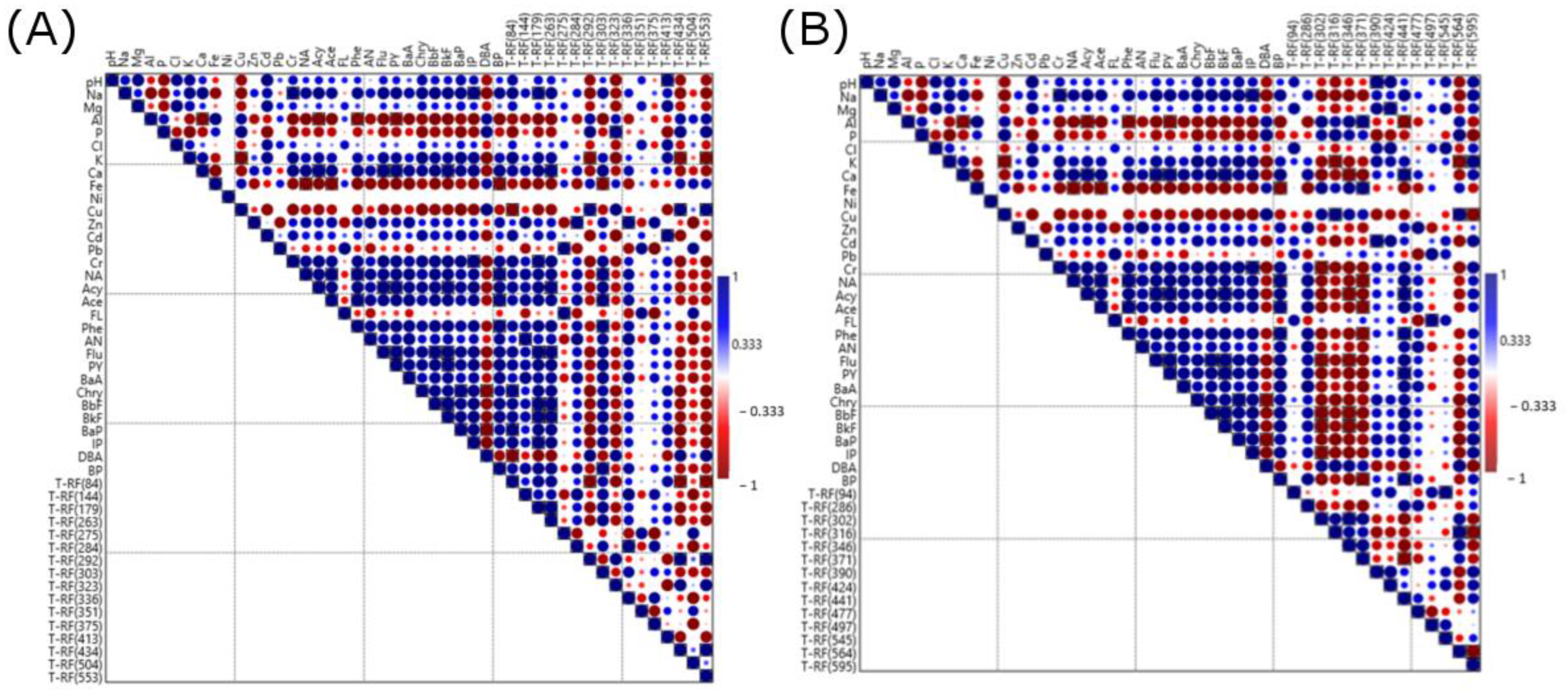
3.3. Microbial Community Structure Shifts
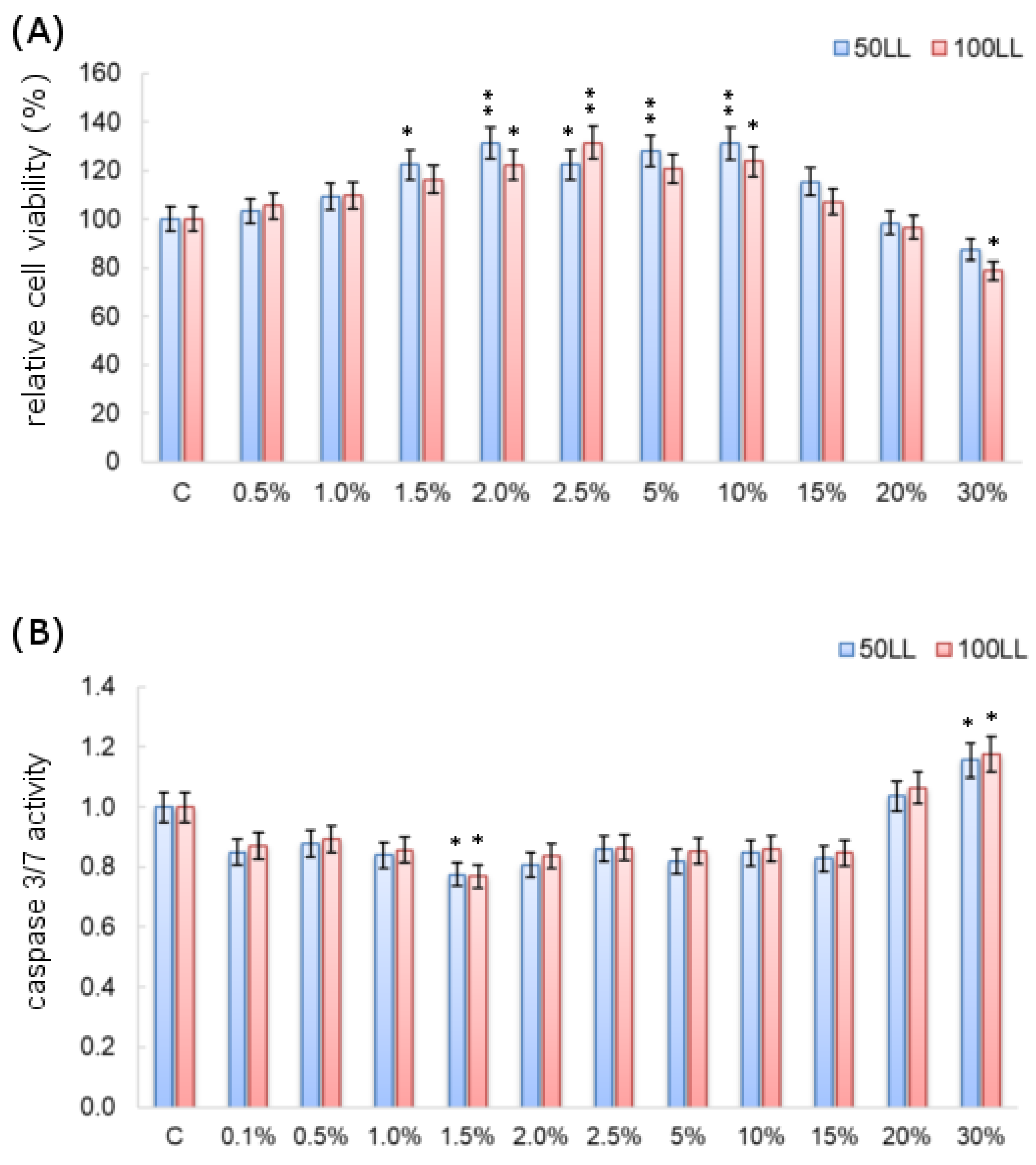
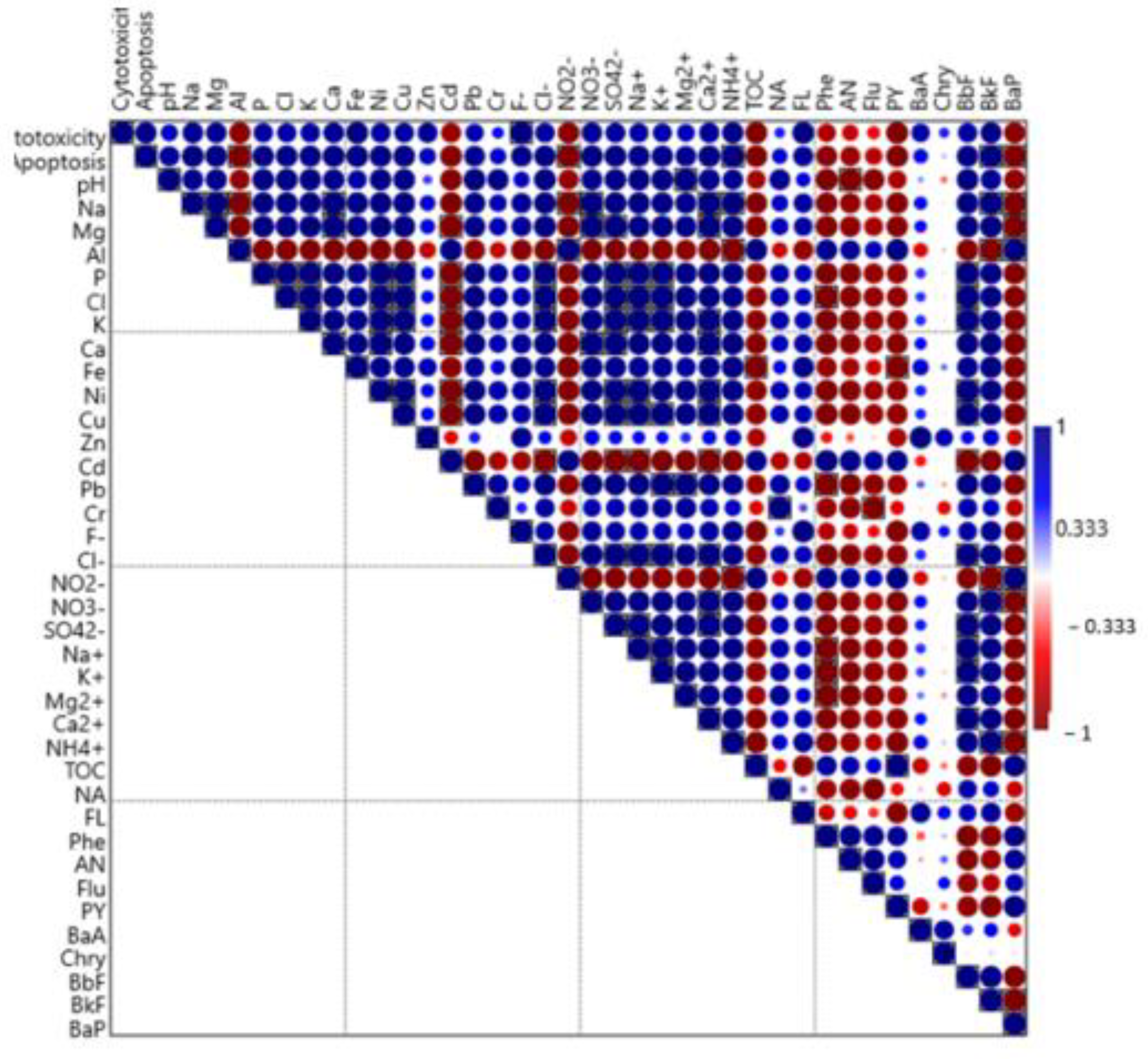
3.4. Effect of FL on Cytotoxicity and Apoptosis Level
4. Discussion
4.1. Effect of LL on Microbial Activity and Community Structure
4.2. Effect of SF on Cytotoxicity and Apoptosis Level
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Salehi, N.; Azhdarpoor, A.; Shirdarreh, M. The Effect of Different Levels of Leachate on Phytoremediation of Pyrene-Contaminated Soil and Simultaneous Extraction of Lead and Cadmium. Chemosphere 2020, 246, 125845. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.; Zhou, K.; Peng, C.; Chen, W. Characterization and Treatment of Landfill Leachate: A Review. Water Res. 2021, 203, 117525. [Google Scholar] [CrossRef] [PubMed]
- Omar, H.; Rohani, S. Treatment of Landfill Waste, Leachate and Landfill Gas: A Review. Front. Chem. Sci. Eng. 2015, 9, 15–32. [Google Scholar] [CrossRef]
- Torretta, V.; Ferronato, N.; Katsoyiannis, I.A.; Tolkou, A.K.; Airoldi, M. Novel and Conventional Technologies for Landfill Leachates Treatment: A Review. Sustainability 2017, 9, 9. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y. Perspectives on Technology for Landfill Leachate Treatment. Arab. J. Chem. 2017, 10, S2567–S2574. [Google Scholar] [CrossRef] [Green Version]
- Christensen, T.H.; Kjeldsen, P.; Bjerg, P.L.; Jensen, D.L.; Christensen, J.B.; Baun, A.; Albrechtsen, H.-J.; Heron, G. Biogeochemistry of Landfill Leachate Plumes. Appl. Geochem. 2001, 16, 659–718. [Google Scholar] [CrossRef]
- Gu, Z.; Feng, K.; Li, Y.; Li, Q. Microbial Characteristics of the Leachate Contaminated Soil of an Informal Landfill Site. Chemosphere 2022, 287 Pt 2, 132155. [Google Scholar] [CrossRef]
- Khalil, C.; Al Hageh, C.; Korfali, S.; Khnayzer, R.S. Municipal Leachates Health Risks: Chemical and Cytotoxicity Assessment from Regulated and Unregulated Municipal Dumpsites in Lebanon. Chemosphere 2018, 208, 1–13. [Google Scholar] [CrossRef]
- Sang, N.; Li, G. Genotoxicity of Municipal Landfill Leachate on Root Tips of Vicia Faba. Mutat. Res. 2004, 560, 159–165. [Google Scholar] [CrossRef]
- Clarke, B.O.; Anumol, T.; Barlaz, M.; Snyder, S.A. Investigating Landfill Leachate as a Source of Trace Organic Pollutants. Chemosphere 2015, 127, 269–275. [Google Scholar] [CrossRef]
- Tiwari, R.; Dwivedi, B.S.; Sharma, Y.M.; Sharma, A.; Dwivedi, A.K. Activities of β-Glucosidase, Phosphatase and Dehydrogenase as Soil Quality Indicators: A Review. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 834–846. [Google Scholar] [CrossRef]
- De Vrieze, J.; Ijaz, U.Z.; Saunders, A.M.; Theuerl, S. Terminal Restriction Fragment Length Polymorphism Is an “Old School” Reliable Technique for Swift Microbial Community Screening in Anaerobic Digestion. Sci. Rep. 2018, 8, 16818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baderna, D.; Caloni, F.; Benfenati, E. Investigating Landfill Leachate Toxicity in Vitro: A Review of Cell Models and Endpoints. Environ. Int. 2019, 122, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Glatt, H.; Gemperlein, I.; Setiabudi, F.; Platt, K.L.; Oesch, F. Expression of Xenobiotic-Metabolizing Enzymes in Propagatable Cell Cultures and Induction of Micronuclei by 13 Compounds. Mutagenesis 1990, 5, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Chow, E.C.; Liu, S.; Du, Y.; Pang, K.S. The Caco-2 Cell Monolayer: Usefulness and Limitations. Expert Opin. Drug Metab. Toxicol. 2008, 4, 395–411. [Google Scholar] [CrossRef]
- Sambuy, Y.; De Angelis, I.; Ranaldi, G.; Scarino, M.L.; Stammati, A.; Zucco, F. The Caco-2 Cell Line as a Model of the Intestinal Barrier: Influence of Cell and Culture-Related Factors on Caco-2 Cell Functional Characteristics. Cell Biol. Toxicol. 2005, 21, 1–26. [Google Scholar] [CrossRef]
- Jabłońska-Trypuć, A.; Wydro, U.; Wołejko, E.; Pietryczuk, A.; Cudowski, A.; Leszczyński, J.; Rodziewicz, J.; Janczukowicz, W.; Butarewicz, A. Potential Toxicity of Leachate from the Municipal Landfill in View of the Possibility of Their Migration to the Environment through Infiltration into Groundwater. Environ. Geochem. Health 2021, 43, 3683–3698. [Google Scholar] [CrossRef]
- Panchoni, L.C.; Santos, C.A.; Kuwano, B.H.; Carmo, K.B.; Cely, M.V.T.; Oliveira-Júnior, A.G.; Fagotti, D.S.L.; Cervantes, V.N.M.; Zangaro, W.; Andrade, D.S.; et al. Effect of Landfill Leachate on Cereal Nutrition and Productivity and on Soil Properties. J. Environ. Qual. 2016, 45, 1080–1086. [Google Scholar] [CrossRef]
- Ming, L.; Li, L.; Zhang, Y.; Lin, W.; Wang, G.; Zhang, S.; Guo, P. Effects of Dissolved Organic Matter on the Desorption of Cd in Freeze–Thaw Treated Cd-Contaminated Soils. Chem. Ecol. 2014, 30, 76–86. [Google Scholar] [CrossRef]
- Kaczynski, P.; Lozowicka, B.; Wolejko, E.; Iwaniuk, P.; Konecki, R.; Dragowski, W.; Lozowicki, J.; Amanbek, N.; Rusilowska, J.; Pietraszko, A. Complex Study of Glyphosate and Metabolites Influence on Enzymatic Activity and Microorganisms Association in Soil Enriched with Pseudomonas Fluorescens and Sewage Sludge. J. Hazard. Mater. 2020, 393, 122443. [Google Scholar] [CrossRef]
- Wołejko, E.; Wydro, U.; Jabłońska-Trypuć, A.; Butarewicz, A.; Łoboda, T. Pseudomonas fluoresces occurrence in soil after fertilization with sewage sludge. Econ. Environ. 2018, 65, 10. [Google Scholar]
- Kramer, C.L.; Pady, S.M. Inhibition of Growth of Fungi on Rose Bengal Media by Light. Trans. Kans. Acad. Sci. 1961, 64, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Tabatabai, M.A. Soil enzymes. In Methods of Soil Analysis: Microbiological and Biochemical Properties; Weaver, R.W., Angle, J.S., Botttomley, P.S., Eds.; Soil Science Society of America: Madison, WI, USA, 1994. [Google Scholar]
- Alef, K.; Nannipieri, P. Β-Glucosidase Activity. In Methods in Applied Soil Microbiology and Biochemistry; Academic Press: Cambridge, MA, USA; Harcourt Brace & Coy Publishers: London, UK, 1995; pp. 350–352. [Google Scholar]
- Mattana, S.; Chelinho, S.; Sousa, J.P.; Alcañiz, J.M.; Domene, X. Nonylphenol Causes Shifts in Microbial Communities and Nitrogen Mineralization in Soil Microcosms. Ecotoxicol. Environ. Saf. 2019, 181, 395–403. [Google Scholar] [CrossRef]
- Wydro, U.; Wołejko, E.; Łozowicka, B.; Jabłońska-Trypuć, A. Microbial Diversity and P Content Changes after the Application of Sewage Sludge and Glyphosate to Soil. Minerals 2021, 11, 1423. [Google Scholar] [CrossRef]
- Ondreičková, K.; Gubišová, M.; Piliarová, M.; Horník, M.; Matušinský, P.; Gubiš, J.; Klčová, L.; Hudcovicová, M.; Kraic, J. Responses of Rhizosphere Fungal Communities to the Sewage Sludge Application into the Soil. Microorganisms 2019, 7, 505. [Google Scholar] [CrossRef] [Green Version]
- Kandziora-Ciupa, M.; Nadgórska-Socha, A.; Barczyk, G. The Influence of Heavy Metals on Biological Soil Quality Assessments in the Vaccinium Myrtillus, L. Rhizosphere under Different Field Conditions. Ecotoxicology 2021, 30, 292–310. [Google Scholar] [CrossRef]
- Chakravarty, P.; Chowdhury, D.; Deka, H. Ecological Risk Assessment of Priority PAHs Pollutants in Crude Oil Contaminated Soil and Its Impacts on Soil Biological Properties. J. Hazard. Mater. 2022, 437, 129325. [Google Scholar] [CrossRef] [PubMed]
- Baran, S.; Bielińska, J.E.; Oleszczuk, P. Enzymatic Activity in an Airfield Soil Polluted with Polycyclic Aromatic Hydrocarbons. Geoderma 2004, 118, 221–232. [Google Scholar] [CrossRef]
- Zhu, N.; Ku, T.; Li, G.; Sang, N. Evaluating Biotoxicity Variations of Landfill Leachate as Penetrating through the Soil Column. Waste Manag. 2013, 33, 1750–1757. [Google Scholar] [CrossRef]
- Yeboah, J.O.; Shi, G.; Shi, W. Effect of Heavy Metal Contamination on Soil Enzymes Activities. J. Geosci. Environ. Prot. 2021, 09, 135–154. [Google Scholar] [CrossRef]
- Li, X.; Meng, D.; Li, J.; Yin, H.; Liu, H.; Liu, X.; Cheng, C.; Xiao, Y.; Liu, Z.; Yan, M. Response of Soil Microbial Communities and Microbial Interactions to Long-Term Heavy Metal Contamination. Environ. Pollut. 2017, 231, 908–917. [Google Scholar] [CrossRef] [PubMed]
- Suhadolc, M.; Schroll, R.; Hagn, A.; Dörfler, U.; Schloter, M.; Lobnik, F. Single Application of Sewage Sludge–Impact on the Quality of an Alluvial Agricultural Soil. Chemosphere 2010, 81, 1536–1543. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, N.W.; Rediske, R.R.; Scull, B.T.; Wierzbicki, D. Landfill Cover Soil, Soil Solution, and Vegetation Responses to Municipal Landfill Leachate Applications. J. Environ. Qual. 2008, 37, 1974–1985. [Google Scholar] [CrossRef] [PubMed]
- Gryta, A.; Frąc, M.; Oszust, K. Community Shift in Structure and Functions across Soil Profile in Response to Organic Waste and Mineral Fertilization Strategies. Appl. Soil Ecol. 2019, 143, 55–60. [Google Scholar] [CrossRef]
- Frąc, M.; Hannula, S.E.; Bełka, M.; Jędryczka, M. Fungal Biodiversity and Their Role in Soil Health. Front. Microbiol. 2018, 9, 707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rashid, M.I.; Mujawar, L.H.; Shahzad, T.; Almeelbi, T.; Ismail, I.M.I.; Oves, M. Bacteria and Fungi Can Contribute to Nutrients Bioavailability and Aggregate Formation in Degraded Soils. Microbiol. Res. 2016, 183, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Hashem, A.; Abd Allah, E.F.; Alqarawi, A.A.; Egamberdieva, D. Arbuscular Mycorrhizal Fungi and Plant Stress Tolerance. In Plant Microbiome: Stress Response; Springer: Singapore, 2018; pp. 81–103. [Google Scholar] [CrossRef]
- Talbot, J.M.; Martin, F.; Kohler, A.; Henrissat, B.; Peay, K.G. Functional Guild Classification Predicts the Enzymatic Role of Fungi in Litter and Soil Biogeochemistry. Soil Biol. Biochem. 2015, 88, 441–456. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, P.; Gupta, A.; Thakur, I.S. Combined Chemical and Toxicological Evaluation of Leachate from Municipal Solid Waste Landfill Sites of Delhi, India. Environ. Sci. Pollut. Res. Int. 2015, 22, 9148–9158. [Google Scholar] [CrossRef]
- Eckers, A.; Reimann, K.; Klotz, L.-O. Nickel and Copper Ion-Induced Stress Signaling in Human Hepatoma Cells: Analysis of Phosphoinositide 3′-Kinase/Akt Signaling. Biometals 2009, 22, 307–316. [Google Scholar] [CrossRef]
| Chemical Properties | C | 50LL | 100LL | C | 50LL | 100LL |
|---|---|---|---|---|---|---|
| Soil (g/kg DW) | SF (mg/L) | |||||
| pH | 6.82 | 7.28 | 7.63 | 6.81 | 7.21 | 7.51 |
| Na | 0.075 | 0.557 | 0.431 | 3.047 | 827.58 | 903.99 |
| Mg | 1.536 | 1.668 | 1.860 | 14.26 | 73.02 | 81.75 |
| Al | 11.26 | 10.62 | 10.92 | 2.512 | nd | nd |
| p | 0.671 | 0.616 | 0.607 | 2.395 | 14.38 | 18.34 |
| Cl | 0.648 | 2.166 | 5.583 | 2.894 | 2628.80 | 3523.09 |
| K | 2.108 | 3.317 | 3.339 | 6.899 | 447.23 | 586.54 |
| Ca | 0.164 | 0.667 | 0.450 | 116.70 | 597.12 | 675.03 |
| Fe | 7.334 | 7.005 | 7.198 | 0.110 | 0.321 | 0.293 |
| Ni | 0.008 | 0.008 | 0.008 | 0.005 | 0.031 | 0.038 |
| Cu | 0.009 | 0.008 | 0.008 | 0.034 | 0.055 | 0.061 |
| Zn | 0.057 | 0.203 | 0.045 | 0.078 | 0.187 | 0.111 |
| Cd | 0.131 | 0.213 | 0.251 | 0.031 | 0.023 | 0.021 |
| Pb | 11.47 | 11.41 | 11.52 | 3.312 | 4.141 | 4.578 |
| Cr | 0.021 | 0.028 | 0.026 | <0.001 | 0.017 | 0.058 |
| F− | - | - | - | nd | 22.688 | 14.83 |
| Cl− | - | - | - | 58.37 | 2446.22 | 3178.97 |
| NO2− | - | - | - | 16.705 | nd | nd |
| NO3− | - | - | - | 270.90 | 948.50 | 1039.05 |
| SO42− | - | - | - | 4.432 | 6.481 | 7.001 |
| Na+ | - | - | - | 15.112 | 1036.26 | 1381.82 |
| K+ | - | - | - | 2.829 | 326.78 | 457.48 |
| Mg2+ | - | - | - | 20.427 | 39.061 | 50.35 |
| Ca2+ | - | - | - | 130.31 | 547.52 | 629.89 |
| NH4+ | - | - | - | 0.895 | 7.978 | 8.044 |
| TOC | 20.960 | 13.026 | 17.249 | 5.452 | 3.517 | 3.794 |
| PAHs in soil (µg/kg DW) | PAHs in SF (ng/L) | |||||
| NA | 1.989 | 7.237 | 14.218 | 4.726 | 6.151 | 10.852 |
| Acy | 0.0143 | 0.0286 | 0.0396 | nd | nd | nd |
| Ace | 0.0051 | 0.0119 | 0.0208 | nd | nd | nd |
| FL | 0.0182 | 0.0251 | 0.0120 | 1.4511 | 5.2751 | 3.5854 |
| Phe | 7.4624 | 9.234 | 11.123 | 0.4936 | 0.1603 | nd |
| AN | 3.8322 | 3.9724 | 5.1698 | 0.1960 | 0.0911 | nd |
| Flu | 15.035 | 23.263 | 28.333 | 1.2519 | 0.8811 | 0.2079 |
| PY | 14.008 | 20.528 | 25.804 | 0.2451 | 0.0360 | 0.0711 |
| BaA | 12.976 | 15.227 | 20.471 | 1.1508 | 2.1020 | 1.3003 |
| Chry | 12.897 | 21.529 | 22.881 | 0.4276 | 0.9215 | 0.1417 |
| BbF | 32.714 | 43.574 | 49.683 | 0.0926 | 0.2474 | 0.2958 |
| BkF | 0.0430 | 0.0615 | 0.0733 | 0.0397 | 0.0945 | 0.0965 |
| BaP | 0.0409 | 0.0852 | 0.0917 | 0.1883 | 0.0553 | 0.0476 |
| IP | 0.0034 | 0.0059 | 0.0069 | nd | nd | nd |
| DBA | 0.0088 | 0.0016 | 0.0009 | nd | nd | nd |
| BP | 0.0018 | 0.0029 | 0.0042 | nd | nd | nd |
| Σ PAHs | 101.05 | 144.79 | 177.93 | 10.262 | 16.015 | 16.598 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wydro, U.; Wołejko, E.; Sokołowska, G.; Leszczyński, J.; Jabłońska-Trypuć, A. Investigating Landfill Leachate Influence on Soil Microbial Biodiversity and Its Cytotoxicity. Water 2022, 14, 3634. https://doi.org/10.3390/w14223634
Wydro U, Wołejko E, Sokołowska G, Leszczyński J, Jabłońska-Trypuć A. Investigating Landfill Leachate Influence on Soil Microbial Biodiversity and Its Cytotoxicity. Water. 2022; 14(22):3634. https://doi.org/10.3390/w14223634
Chicago/Turabian StyleWydro, Urszula, Elżbieta Wołejko, Gabriela Sokołowska, Jacek Leszczyński, and Agata Jabłońska-Trypuć. 2022. "Investigating Landfill Leachate Influence on Soil Microbial Biodiversity and Its Cytotoxicity" Water 14, no. 22: 3634. https://doi.org/10.3390/w14223634
APA StyleWydro, U., Wołejko, E., Sokołowska, G., Leszczyński, J., & Jabłońska-Trypuć, A. (2022). Investigating Landfill Leachate Influence on Soil Microbial Biodiversity and Its Cytotoxicity. Water, 14(22), 3634. https://doi.org/10.3390/w14223634









