The Nitrogen Removal Performance and Functional Bacteria in Heterotrophic Denitrification and Mixotrophic Denitrification Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. System Setup and Operation Conditions
2.2. Analytical Methods
2.3. EPS Extraction and Analysis Methods
2.4. Microbial Communities and Denitrifying Genes
3. Results and Discussion
3.1. The Performance of the Biological Denitrification System
3.2. The Contents and Characteristics of EPS
3.3. Microbial Diversity and Communities
3.3.1. Alpha Diversity Index
3.3.2. Microbial Communities
3.4. Abundances of Denitrifying Genes in Different Stages
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, W.; Lin, J.G.; Zhang, L.B.; Zhu, K.P.; Liu, X.Y.; Wang, Z.; Cao, B.X.; Guo, P. Mixotrophic Denitrification Desulfurization Wastewater Treatment Process: Bioreactor Performance and Analysis of the Microbial Community. Pol. J. Environ. Stud. 2016, 25, 2491–2497. [Google Scholar] [CrossRef] [Green Version]
- Moussavi, G.; Jafari, S.J.; Yaghmaeian, K. Enhanced biological denitrification in the cyclic rotating bed reactor with catechol as carbon source. Bioresour. Technol. 2015, 189, 266–272. [Google Scholar] [CrossRef]
- Wang, D.B.; Wang, Y.L.; Liu, Y.W.; Ngo, H.H.; Lian, Y.; Zhao, J.W.; Chen, F.; Yang, Q.; Zeng, G.M.; Li, X.M. Is denitrifying anaerobic methane oxidation-centered technologies a solution for the sustainable operation of wastewater treatment Plants? Bioresour. Technol. 2017, 234, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.R.; Hou, R.R.; Yang, P.; Qian, S.T.; Feng, Z.Q.; Chen, Z.B.; Wang, F.; Yuan, R.F.; Chen, H.L.; Zhou, B.H. Application of external carbon source in heterotrophic denitrification of domestic sewage: A review. Sci. Total Environ. 2022, 817, 153061. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.H.; Peng, J.J.; Feng, C.J.; Fang, F.; Chen, S.H.; Xu, Y.J.; Wang, X.Z. Evaluation of simultaneous autotrophic and heterotrophic denitrification processes and bacterial community structure analysis. Appl. Microbiol. Biotechnol. 2015, 99, 6527–6536. [Google Scholar] [CrossRef]
- Pang, Y.M.; Wang, J.L. Various electron donors for biological nitrate removal: A review. Sci. Total Environ. 2021, 794, 148699. [Google Scholar] [CrossRef]
- Sun, S.P.; Nacher, C.P.I.; Merkey, B.; Zhou, Q.; Xia, S.Q.; Yang, D.H.; Sun, J.H.; Smets, B.F. Effective Biological Nitrogen Removal Treatment Processes for Domestic Wastewaters with Low C/N Ratios: A Review. Environ. Eng. Sci. 2010, 27, 111–126. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Duan, C.S.; Yu, J.H.; Dong, W.Y. Transforming heterotrophic to autotrophic denitrification process: Insights into microbial community, interspecific interaction and nitrogen metabolism. Bioresour. Technol. 2022, 345, 126471. [Google Scholar] [CrossRef]
- Hellman, M.; Bonilla-Rosso, G.; Widerlund, A.; Juhanson, J.; Hallin, S. External carbon addition for enhancing denitrification modifies bacterial community composition and affects CH4 and N2O production in sub-arctic mining pond sediments. Water Res. 2019, 158, 22–33. [Google Scholar] [CrossRef]
- Chen, C.; Ren, N.Q.; Wang, A.J.; Yu, Z.G.; Lee, D.J. Simultaneous biological removal of sulfur, nitrogen and carbon using EGSB reactor. Appl. Microbiol. Biotechnol. 2008, 78, 1057–1063. [Google Scholar] [CrossRef]
- Tang, K.; An, S.J.; Nemati, M. Evaluation of autotrophic and heterotrophic processes in biofilm reactors used for removal of sulphide, nitrate and COD. Bioresour. Technol. 2010, 101, 8109–8118. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Yu, D.S.; Chen, G.H.; Wang, Y.Y.; Tang, P.; Liu, C.C.; Tian, Y.; Zhang, M. Realization of nitrite accumulation in a sulfide-driven autotrophic denitrification process: Simultaneous nitrate and sulfur removal. Chemosphere 2021, 278, 130413. [Google Scholar] [CrossRef] [PubMed]
- Show, K.Y.; Lee, D.J.; Pan, X.L. Simultaneous biological removal of nitrogen-sulfur-carbon: Recent advances and challenges. Biotechnol. Adv. 2013, 31, 409–420. [Google Scholar] [CrossRef]
- Wang, B.; Wu, D.; Dai, J.; Ekama, G.A.; Hao, X.D.; Chen, G.H. Elucidating the effects of starvation and reactivation on anaerobic sulfidogenic granular sludge: Reactor performance and granular sludge transformation. Water Res. 2019, 151, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Goorissen, H.P.; Stams, A.J.M.; Hansen, T.A. Methanol utilization in defined mixed cultures of thermophilic anaerobes in the presence of sulfate. FEMS Microbiol. Ecol. 2004, 49, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.M.; Gutierrez, O.; Sharma, K.R.; Keller, J.; Yuan, Z.G. Optimization of intermittent, simultaneous dosage of nitrite and hydrochloric acid to control sulfide and methane productions in sewers. Water Res. 2011, 45, 6163–6172. [Google Scholar] [CrossRef]
- Mohanakrishnan, J.; Gutierrez, O.; Sharma, K.R.; Guisasola, A.; Werner, U.; Meyer, R.L.; Keller, J.; Yuan, Z. Impact of nitrate addition on biofilm properties and activities in rising main sewers. Water Res. 2009, 43, 4225–4237. [Google Scholar] [CrossRef]
- Rathnayake, D.; Sathasivan, A.; Kastl, G.; Krishna, K.C.B. Hydrogen sulphide control in sewers by catalysing the reaction with oxygen. Sci. Total Environ. 2019, 689, 1192–1200. [Google Scholar] [CrossRef]
- Yan, X.F.; Sun, J.; Kenjiahan, A.; Dai, X.H.; Ni, B.J.; Yuan, Z.G. Rapid and strong biocidal effect of ferrate on sulfidogenic and methanogenic sewer biofilms. Water Res. 2020, 169, 115208. [Google Scholar] [CrossRef]
- Zhang, L.; De Schryver, P.; De Gusseme, B.; De Muynck, W.; Boon, N.; Verstraete, W. Chemical and biological technologies for hydrogen sulfide emission control in sewer systems: A review. Water Res. 2008, 42, 1–12. [Google Scholar] [CrossRef]
- Nielsen, A.H.; Lens, P.; Vollertsen, J.; Hvitved-Jacobsen, T. Sulfide-iron interactions in domestic wastewater from a gravity sewer. Water Res. 2005, 39, 2747–2755. [Google Scholar]
- Park, K.; Lee, H.; Phelan, S.; Liyanaarachchi, S.; Marleni, N.; Navaratna, D.; Jegatheesan, V.; Shu, L. Mitigation strategies of hydrogen sulphide emission in sewer networks—A review. Int. Biodeterior. Biodegrad. 2014, 95, 251–261. [Google Scholar] [CrossRef]
- Wisniewski, K.; di Biase, A.; Munz, G.; Oleszkiewicz, J.A.; Makinia, J. Kinetic characterization of hydrogen sulfide inhibition of suspended anammox biomass from a membrane bioreactor. Biochem. Eng. J. 2019, 143, 48–57. [Google Scholar] [CrossRef]
- Joo, H.S.; Hirai, M.; Shoda, M. Characteristics of ammonium removal by heterotrophic nitrification-aerobic denitrification by Alcaligenes faecalis no. 4. J. Biosci. Bioeng. 2005, 100, 184–191. [Google Scholar] [CrossRef]
- Huang, C.; Liu, Q.; Li, Z.L.; Ma, X.D.; Hou, Y.N.; Ren, N.Q.; Wang, A.J. Relationship between functional bacteria in a denitrification desulfurization system under autotrophic, heterotrophic, and mixotrophic conditions. Water Res. 2021, 188, 116526. [Google Scholar] [CrossRef]
- Huang, C.; Liu, Q.; Chen, C.; Chen, F.; Zhao, Y.K.; Gao, L.F.; Liu, W.Z.; Zhou, J.Z.; Li, Z.L.; Wang, A.J. Elemental sulfur recovery and spatial distribution of functional bacteria and expressed genes under different carbon/nitrate/sulfide loadings in up-flow anaerobic sludge blanket reactors. J. Hazard. Mater. 2017, 324, 48–53. [Google Scholar] [CrossRef] [Green Version]
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- Adav, S.S.; Lee, D.J. Characterization of extracellular polymeric substances (EPS) from phenol degrading aerobic granules. J. Taiwan Inst. Chem. Eng. 2011, 42, 645–651. [Google Scholar] [CrossRef]
- Adav, S.S.; Lee, D.J. Extraction of extracellular polymeric substances from aerobic granule with compact interior structure. J. Hazard. Mater. 2008, 154, 1120–1126. [Google Scholar] [CrossRef]
- Luo, J.H.; Hao, T.W.; Wei, L.; Mackey, H.R.; Lin, Z.Q.; Chen, G.H. Impact of influent COD/N ratio on disintegration of aerobic granular sludge. Water Res. 2014, 62, 127–135. [Google Scholar] [CrossRef]
- US Environmental Protection Agency (US EPA). Drinking Water Advisory: Consumer Acceptability Advice and Health Effects Analysis on Sulfate; US Environmental Protection Agency: Washington, DC, USA, 2003.
- Bichai, F.; Barbeau, B.; Dullemont, Y.; Hijnen, W. Role of predation by zooplankton in transport and fate of protozoan (oo)cysts in granular activated carbon filtration. Water Res. 2010, 44, 1072–1081. [Google Scholar] [CrossRef]
- More, T.T.; Yadav, J.S.S.; Yan, S.; Tyagi, R.D.; Surampalli, R.Y. Extracellular polymeric substances of bacteria and their potential environmental applications. J. Environ. Manag. 2014, 144, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Han, F.; Ye, W.; Wei, D.; Xu, W.Y.; Du, B.; Wei, Q. Simultaneous nitrification-denitrification and membrane fouling alleviation in a submerged biofilm membrane bioreactor with coupling of sponge and biodegradable PBS carrier. Bioresour. Technol. 2018, 270, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Wilen, B.M.; Jin, B.; Lant, P. The influence of key chemical constituents in activated sludge on surface and flocculating properties. Water Res. 2003, 37, 2127–2139. [Google Scholar] [CrossRef]
- Laspidou, C.S.; Rittmann, B.E. A unified theory for extracellular polymeric substances, soluble microbial products, and active and inert biomass. Water Res. 2002, 36, 2711–2720. [Google Scholar] [CrossRef]
- Chu, L.B.; Wang, J.L. Denitrification of groundwater using PHBV blends in packed bed reactors and the microbial diversity. Chemosphere 2016, 155, 463–470. [Google Scholar] [CrossRef]
- Liao, R.H.; Shen, K.; Li, A.M.; Shi, P.; Li, Y.; Shin, Q.Q.; Wang, Z. High-nitrate wastewater treatment in an expanded granular sludge bed reactor and microbial diversity using 454 pyrosequencing analysis. Bioresour. Technol. 2013, 134, 190–197. [Google Scholar] [CrossRef]
- Yamada, T.; Sekiguchi, Y.; Imachi, H.; Kamagata, Y.; Ohashi, A.; Harada, H. Diversity, localization, and physiological properties of filamentous microbes belonging to Chloroflexi subphylum I in mesophilic and thermophilic methanogenic sludge granules. Appl. Environ. Microbiol. 2005, 71, 7493–7503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, Y.; Zheng, Y.; Wu, S.; Yang, Z.H.; Zhao, F. Bacterial Community Structure of Autotrophic Denitrification Biocathode by 454 Pyrosequencing of the 16S rRNA Gene. Microb. Ecol. 2015, 69, 492–499. [Google Scholar] [CrossRef]
- Hille, A.; He, M.; Ochmann, C.; Neu, T.; Horn, H. Application of two component biodegradable carriers in a particle-fixed biofilm airlift suspension reactor: Development and structure of biofilms. Bioprocess Biosyst. Eng. 2009, 32, 31–39. [Google Scholar] [CrossRef]
- Kim, K.H.; Ten, L.N.; Liu, Q.M.; Im, W.T.; Lee, S.T. Comamonas granuli sp nov., isolated from granules used in a wastewater treatment plant. J. Microbiol. 2008, 46, 390–395. [Google Scholar] [CrossRef]
- Liao, R.H.; Li, Y.; Yu, X.M.; Shi, P.; Wang, Z.; Shen, K.; Shi, Q.Q.; Miao, Y.; Li, W.T.; Li, A.M. Performance and microbial diversity of an expanded granular sludge bed reactor for high sulfate and nitrate waste brine treatment. J. Environ. Sci. 2014, 26, 717–725. [Google Scholar] [CrossRef]
- Lu, H.J.; Chandran, K.; Stensel, D. Microbial ecology of denitrification in biological wastewater treatment. Water Res. 2014, 64, 237–254. [Google Scholar] [CrossRef]
- Coates, J.D.; Chakraborty, R.; Lack, J.G.; O’Connor, S.M.; Cole, K.A.; Bender, K.S.; Achenbach, L.A. Anaerobic benzene oxidation coupled to nitrate reduction in pure culture by two strains of Dechloromonas. Nature 2001, 411, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.L.; Li, Y.; Liu, X.; He, S.B.; Huang, J.C. Comparison of microbial communities in different sulfur-based autotrophic denitrification reactors. Appl. Microbiol. Biotechnol. 2017, 101, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gerrity, S.; Collins, G.; Chen, T.H.; Li, R.H.; Xie, S.H.; Zhan, X.M. Enrichment and characterization of autotrophic Thiobacillus denitrifiers from anaerobic sludge for nitrate removal. Process Biochem. 2018, 68, 165–170. [Google Scholar] [CrossRef]
- Zhang, R.C.; Xu, X.J.; Chen, C.; Xing, D.F.; Shao, B.; Liu, W.Z.; Wang, A.J.; Lee, D.J.; Ren, N.Q. Interactions of functional bacteria and their contributions to the performance in integrated autotrophic and heterotrophic denitrification. Water Res. 2018, 143, 355–366. [Google Scholar] [CrossRef]
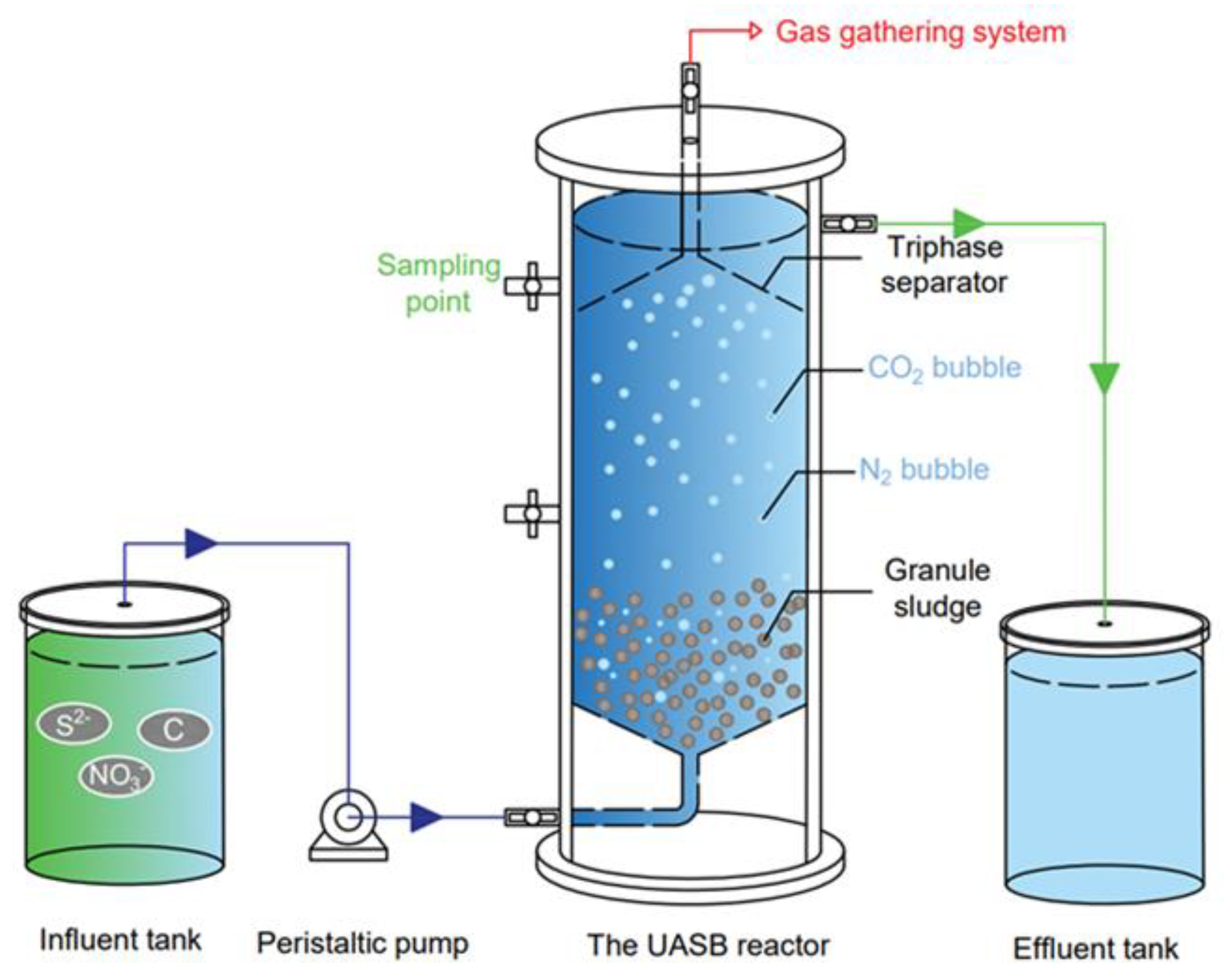



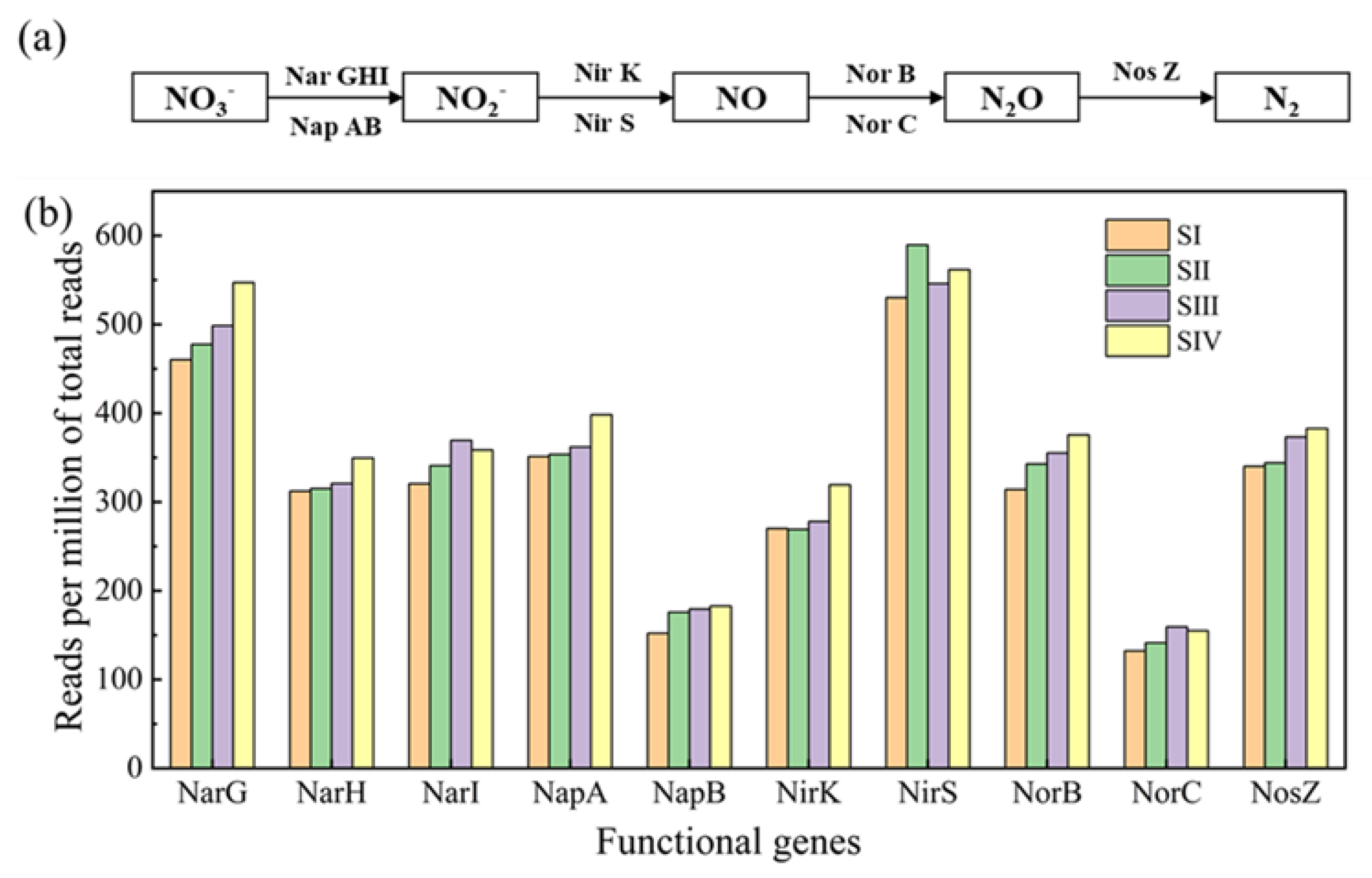
| Stage | Nitrate-N (mg/L) | COD (mg/L) | S2− (mg/L) | TP (mg/L) | HRT (h) | C/N | S/N |
|---|---|---|---|---|---|---|---|
| I (1–30 d) | 30 | 100 | 0 | 5 | 4 | 3.33 | - |
| II (31–60 d) | 30 | 100 | 5 | 5 | 4 | 3.33 | 0.17 |
| III (61–90 d) | 30 | 100 | 10 | 5 | 4 | 3.33 | 0.33 |
| IV (91–120 d) | 30 | 100 | 20 | 5 | 4 | 3.33 | 0.67 |
| Sample | Ace | Chao1 | Shannon | Simpson | Coverage |
|---|---|---|---|---|---|
| SI | 276 | 276 | 4.847 | 0.938 | 1.000 |
| SII | 260 | 260 | 4.291 | 0.950 | 1.000 |
| SIII | 230 | 229 | 3.558 | 0.948 | 0.999 |
| SIV | 198 | 198 | 3.218 | 0.964 | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, X.; Wang, Y.; Wan, J.; Yan, Z.; Ma, Y.; Zhang, G.; Zhu, B. The Nitrogen Removal Performance and Functional Bacteria in Heterotrophic Denitrification and Mixotrophic Denitrification Process. Water 2022, 14, 3603. https://doi.org/10.3390/w14223603
Ren X, Wang Y, Wan J, Yan Z, Ma Y, Zhang G, Zhu B. The Nitrogen Removal Performance and Functional Bacteria in Heterotrophic Denitrification and Mixotrophic Denitrification Process. Water. 2022; 14(22):3603. https://doi.org/10.3390/w14223603
Chicago/Turabian StyleRen, Xinghao, Yan Wang, Jinquan Wan, Zhicheng Yan, Yongwen Ma, Guihua Zhang, and Bin Zhu. 2022. "The Nitrogen Removal Performance and Functional Bacteria in Heterotrophic Denitrification and Mixotrophic Denitrification Process" Water 14, no. 22: 3603. https://doi.org/10.3390/w14223603




