Efficient Arsenate Decontamination from Water Using MgO-Itsit Biochar Composite: An Equilibrium, Kinetics and Thermodynamic Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Itsit Biochar (IBC)
2.2. Synthesis of IBC Composite with Magnesium Oxide Nanoparticles
2.3. Characterization of Materials
2.4. Adsorption Studies of Arsenic onto MgO-IBC
2.5. Reusability and Stability Study of MgO-IBC
3. Results
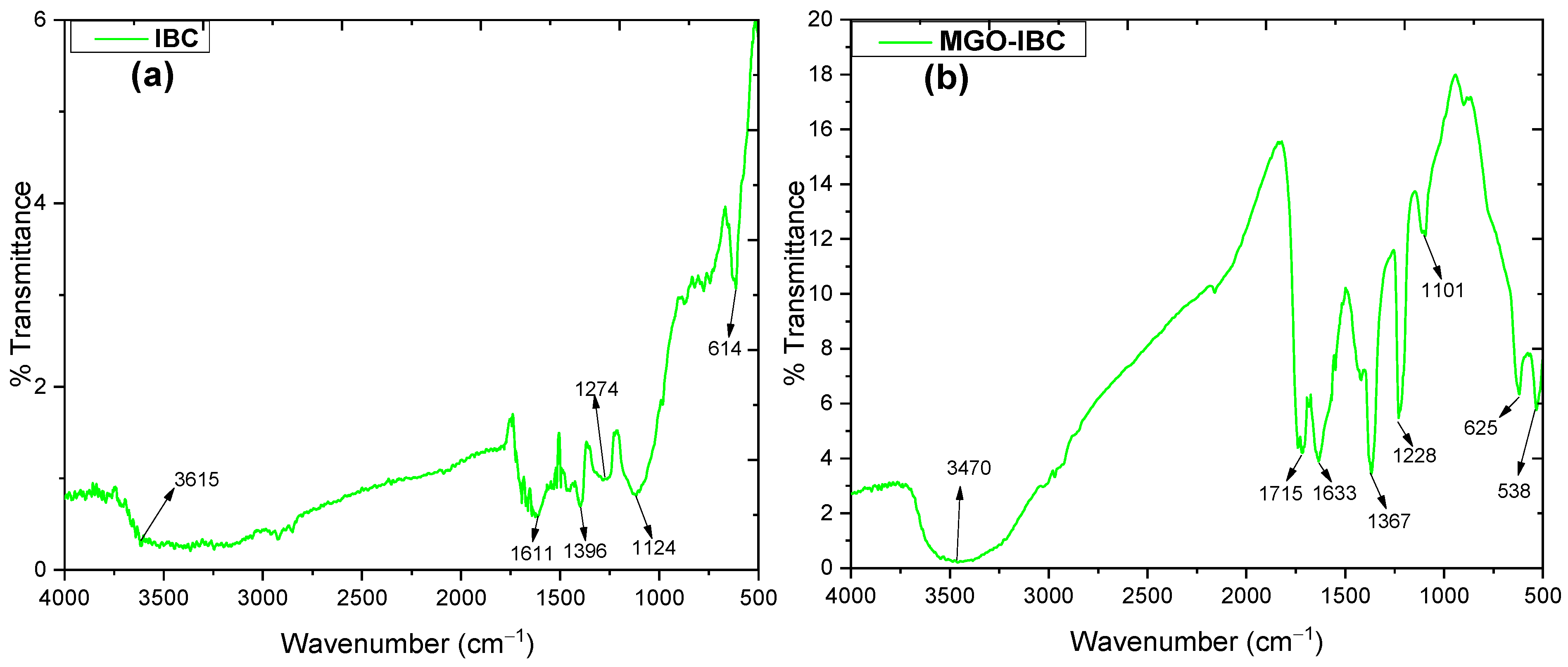
3.1. FTIR Analysis
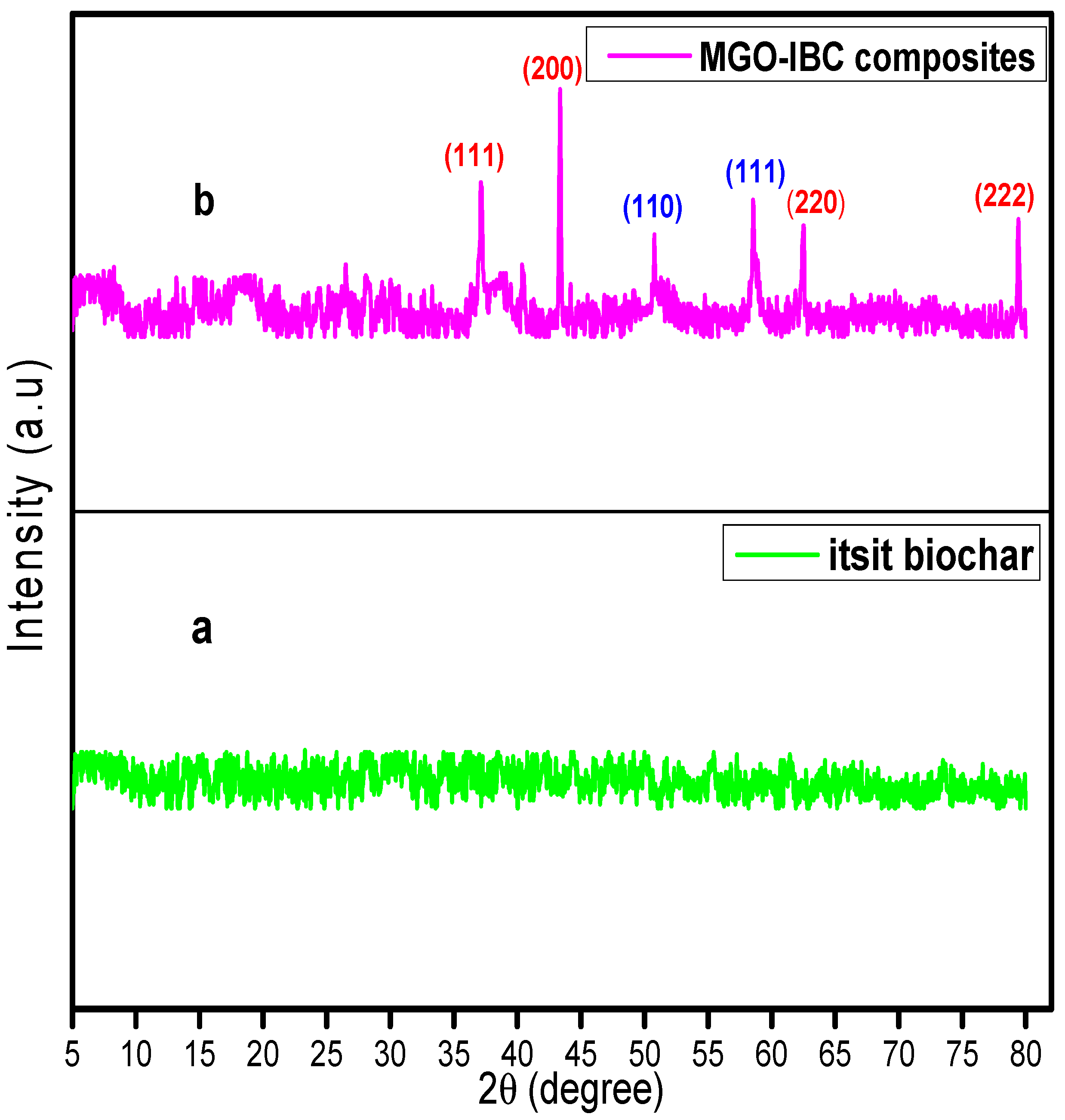
3.2. XRD Analysis
3.3. SEM Analysis
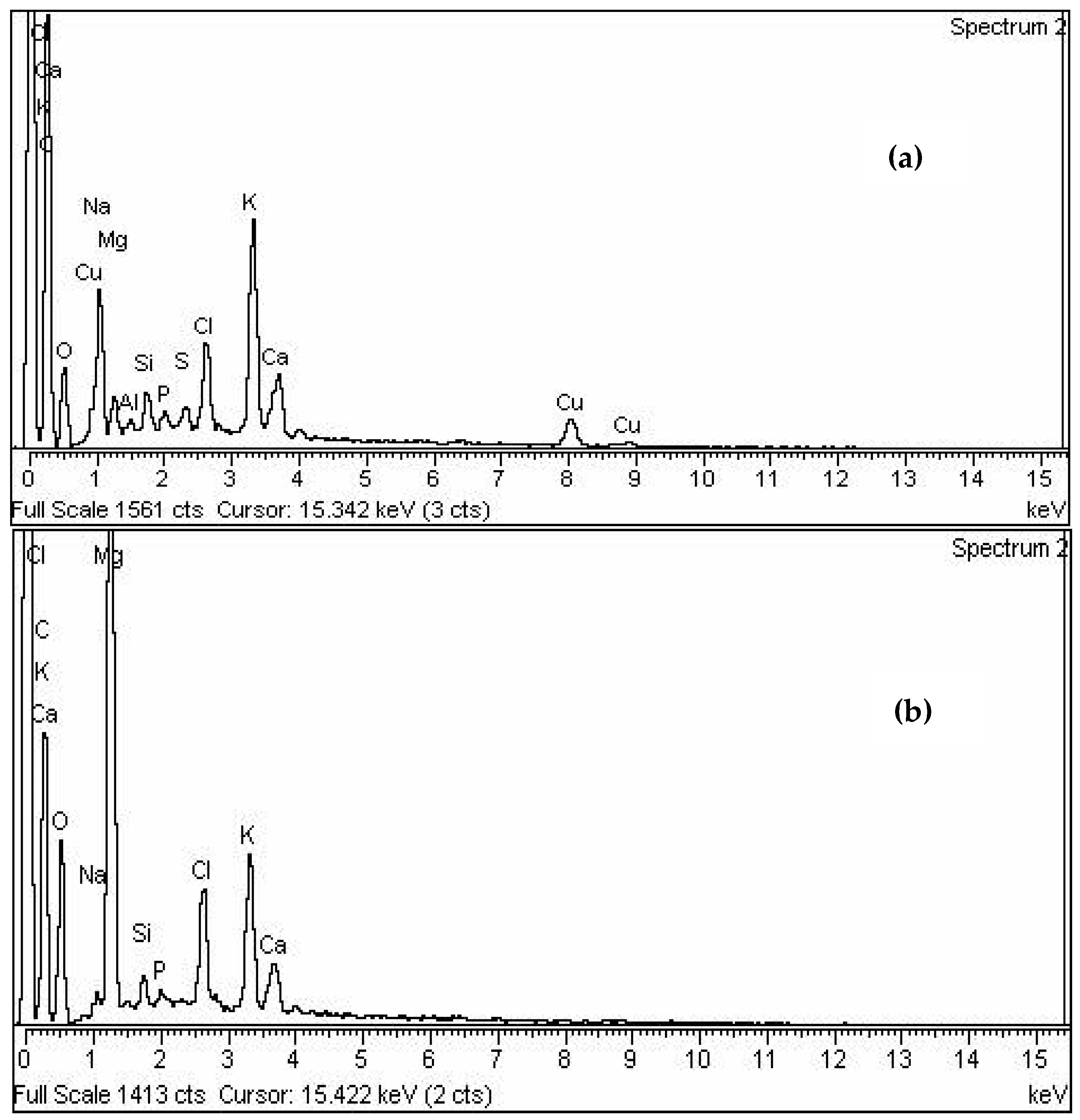
3.4. EDX Analysis
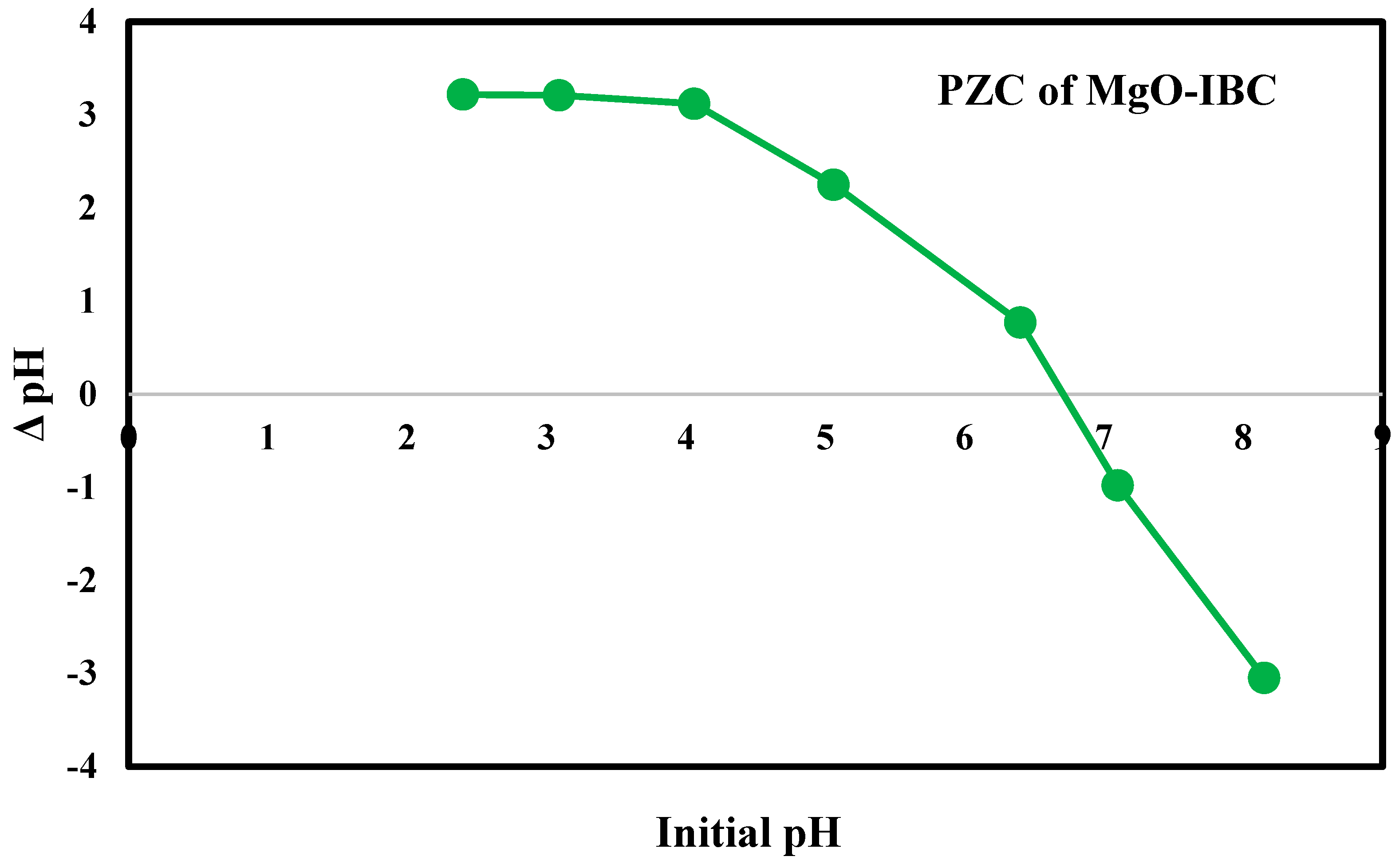
3.5. Point of Zero Charge (PZC)
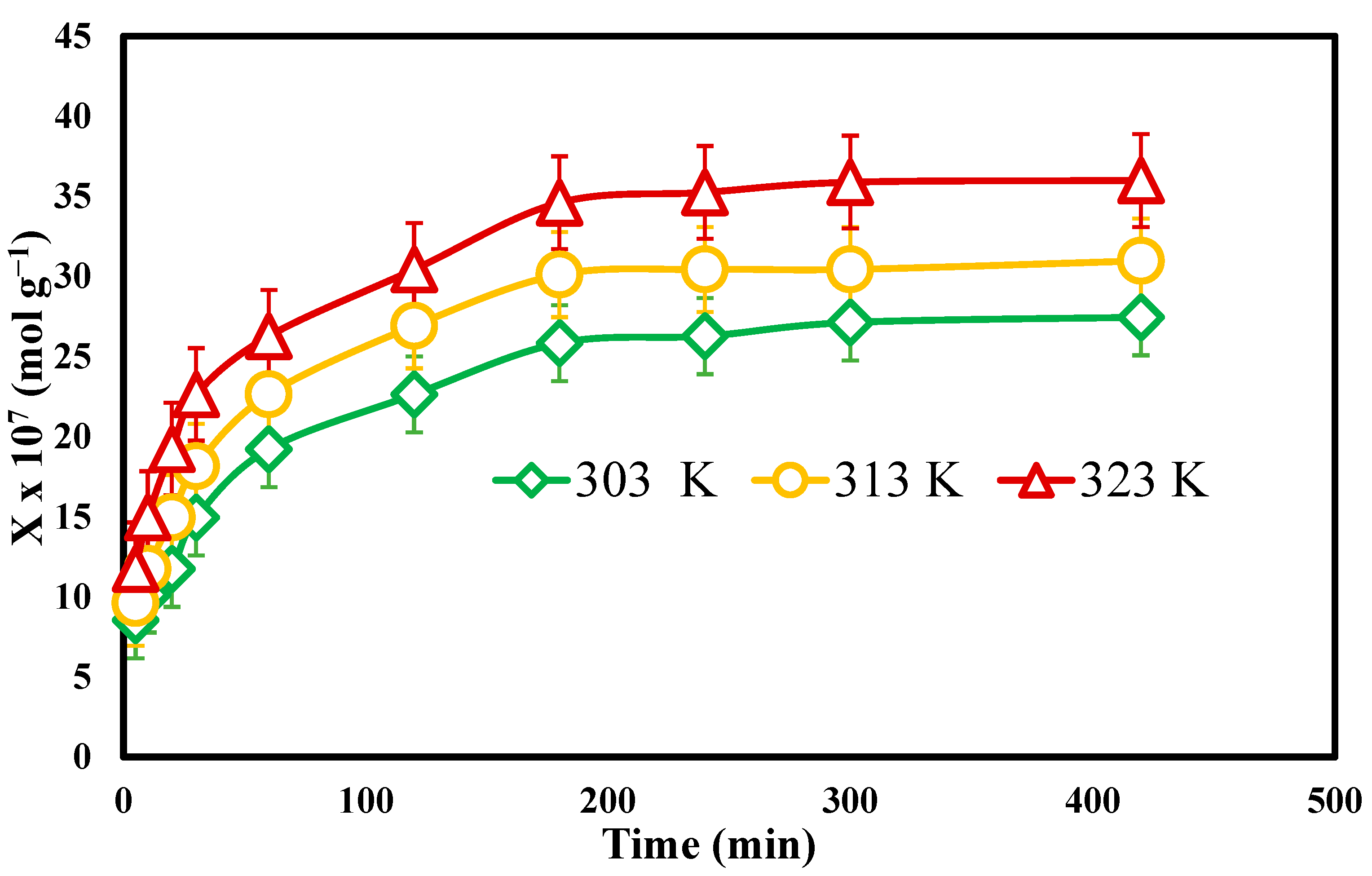
3.6. Effect of Contact Time
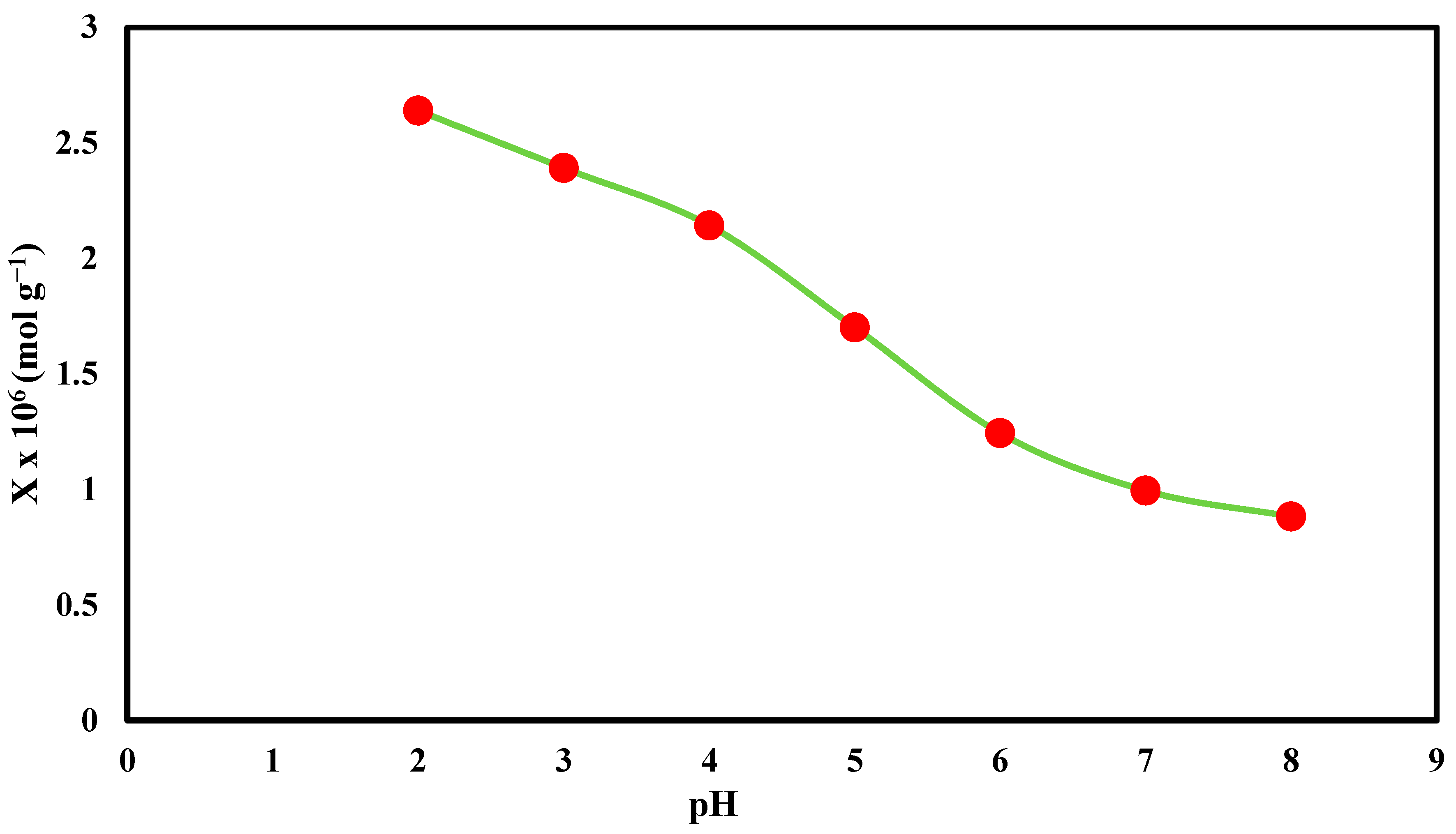
3.7. Effect of pH
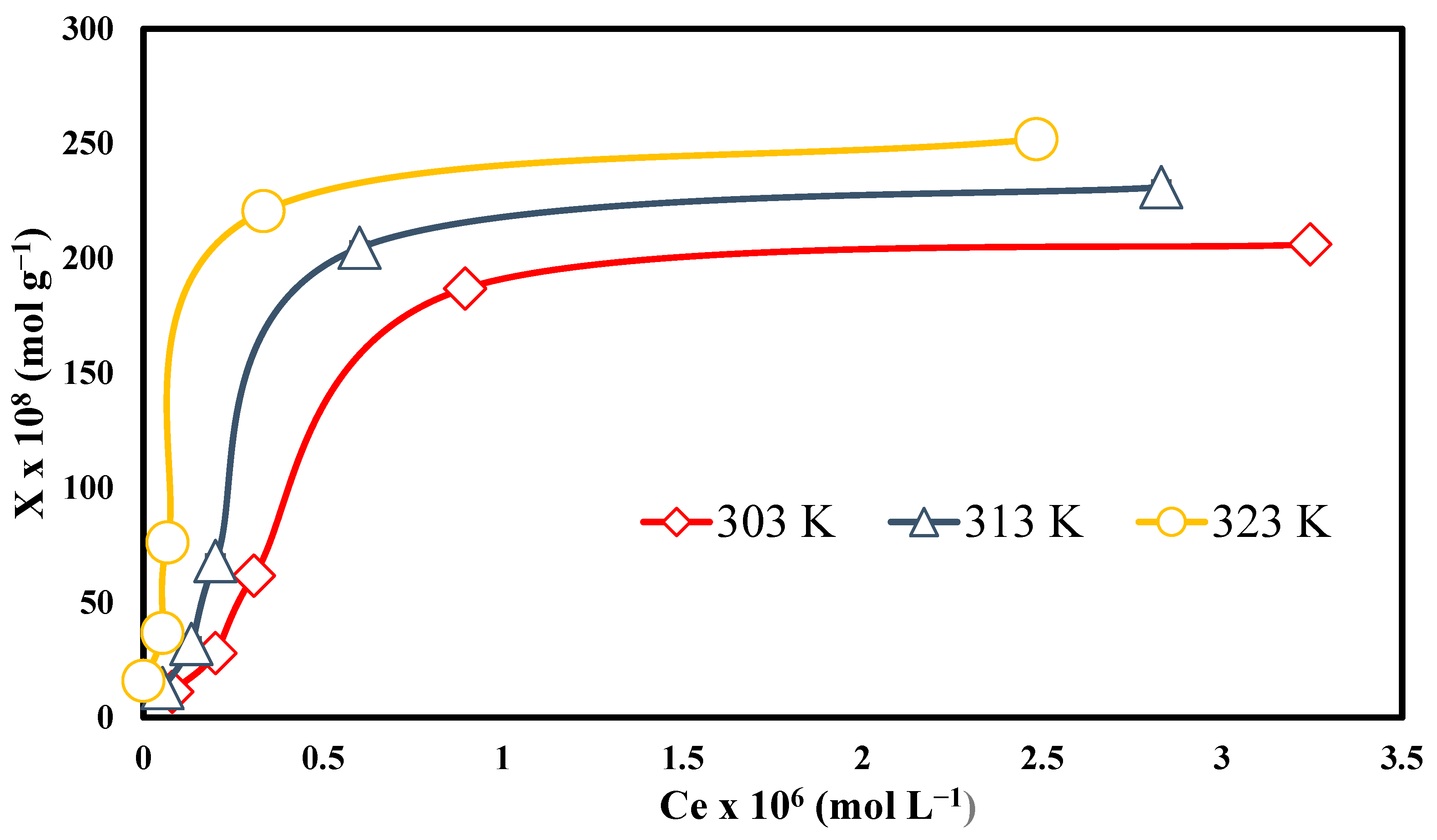
3.8. Effect of Temperature and Concentration
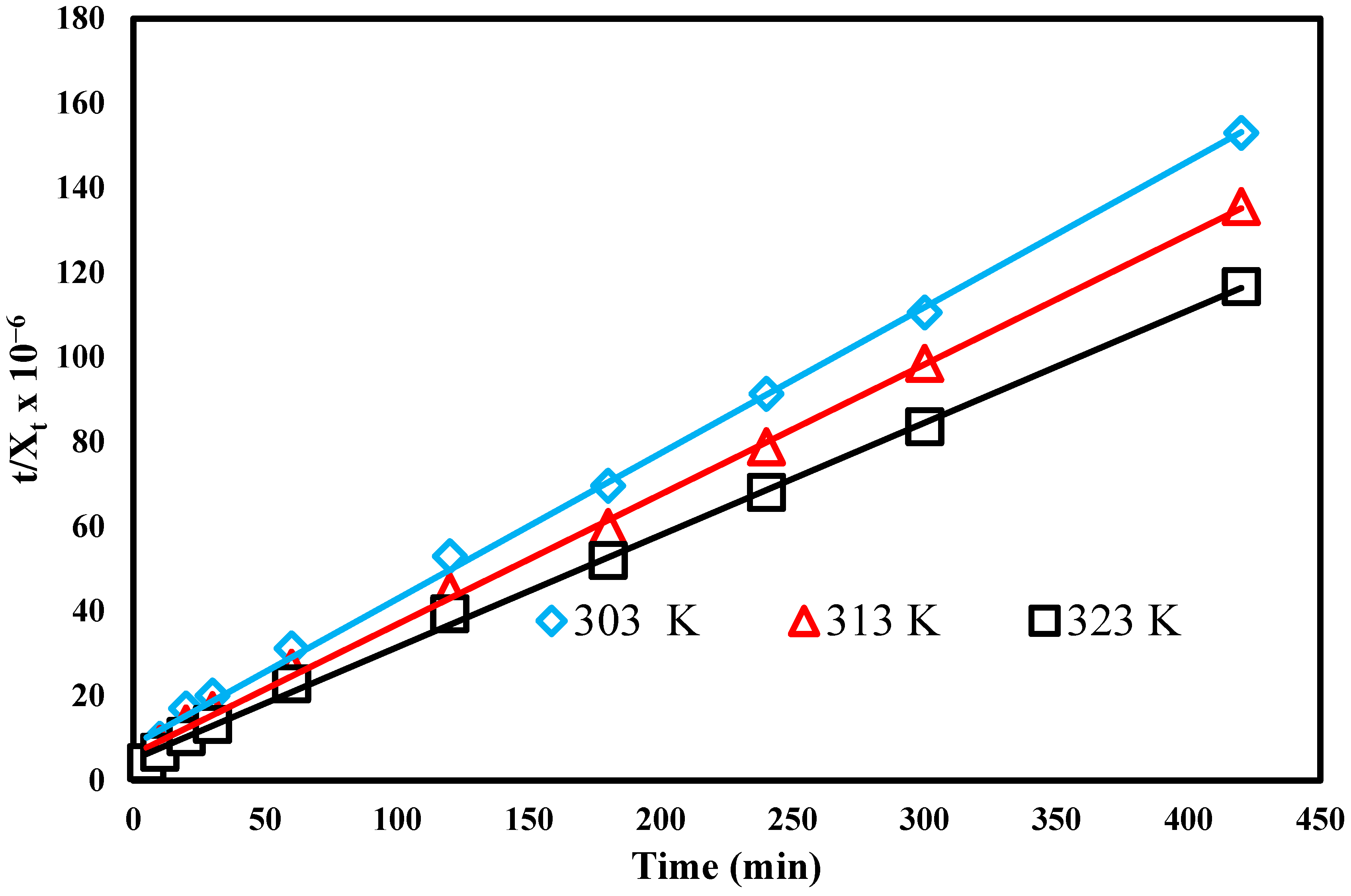
3.9. Ho and Mckay’s- Plot
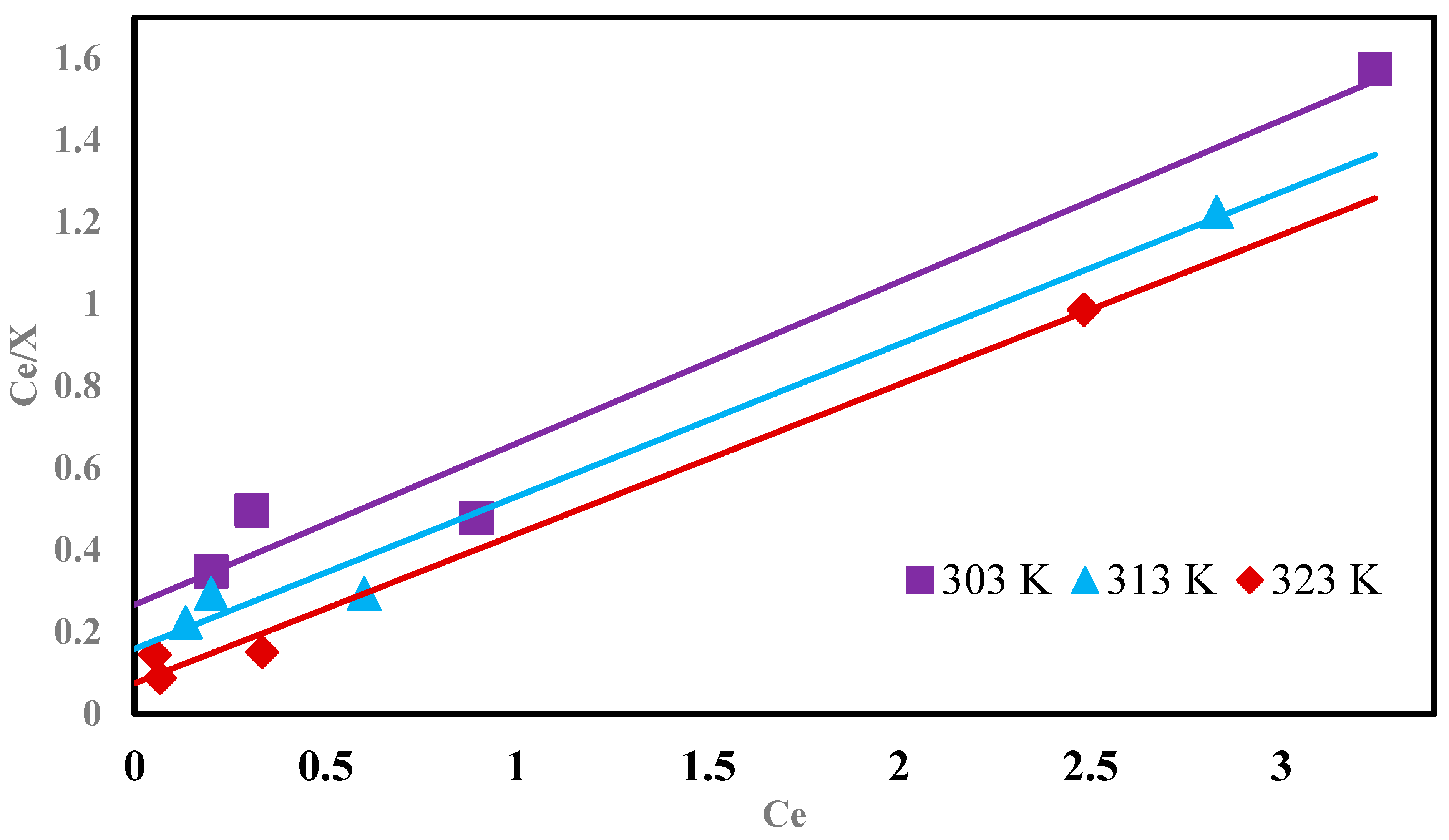
3.10. Langmuir Model
3.11. Estimation of Activation Energy
3.12. Thermodynamic Parameters
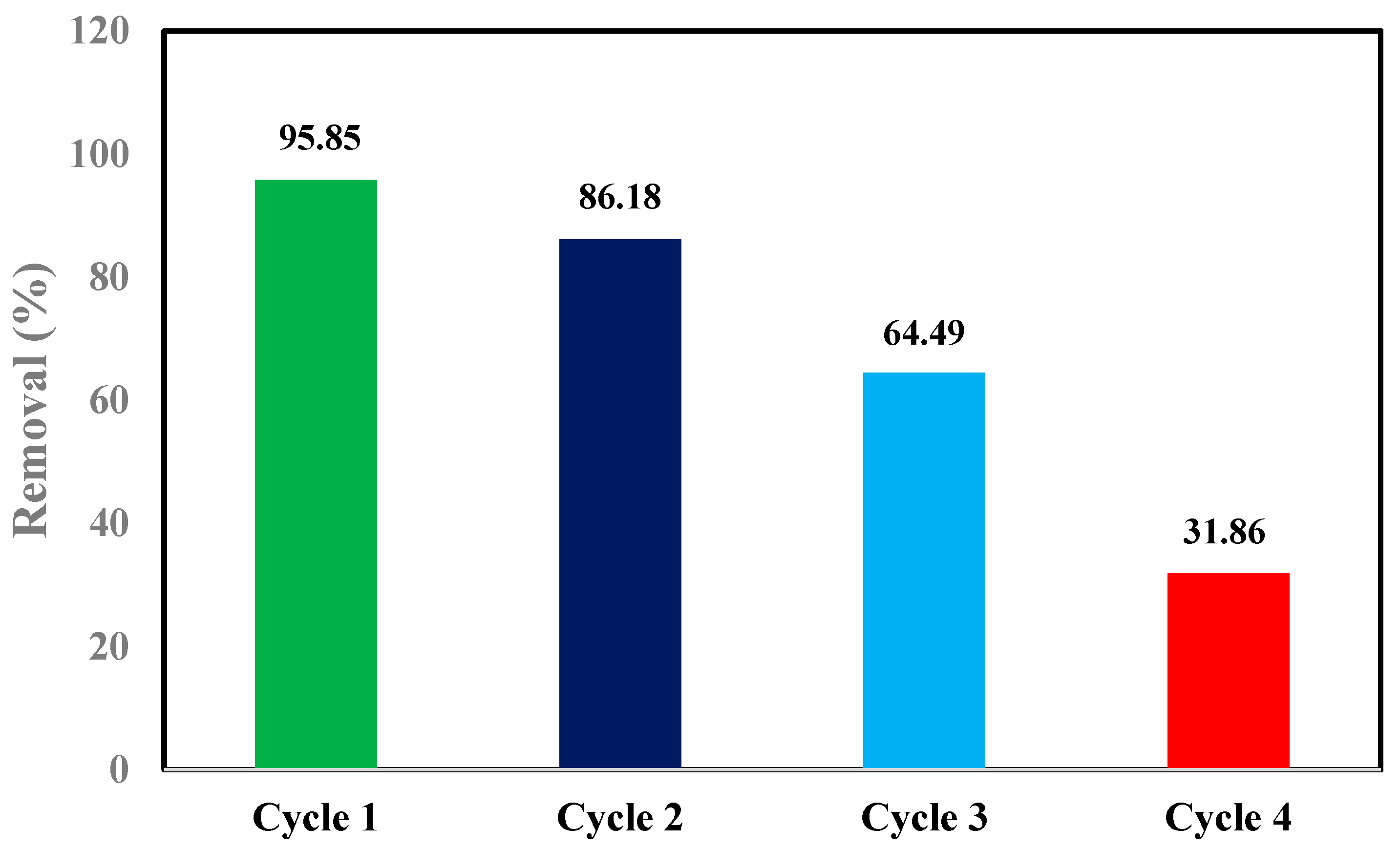
3.13. Reusability and Stability Study of MgO-IBC
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sanjrani, M.A.; Zhou, B.; Zhao, H.; Bhutto, S.A.; Muneer, A.S.; Xia, S.B. Arsenic contaminated groundwater in China and its treatment options, a review. Appl. Ecol. Environ. Res. 2019, 17, 1655–1683. [Google Scholar] [CrossRef]
- Raza, M.; Mahjabeen, I.; Fahim, M.; Malik, W.A.; Khan, A.U.; Kayani, M.A.; Akram, Z. Redox balance and DNA fragmentation in arsenic-exposed occupational workers from different industries of Pakistan. Environ. Sci. Pollut. Res. 2018, 25, 33381–33390. [Google Scholar] [CrossRef] [PubMed]
- Rathi, B.S.; Kumar, P.S. A review on sources, identification and treatment strategies for the removal of toxic Arsenic from water system. J. Hazard. Mater. 2021, 418, 126299. [Google Scholar] [CrossRef]
- Dos Santos, H.H.; Demarchi, C.A.; Rodrigues, C.A.; Greneche, J.M.; Nedelko, N.; Ślawska-Waniewska, A. Adsorption of As (III) on chitosan-Fe-crosslinked complex (Ch-Fe). Chemosphere 2011, 82, 278–283. [Google Scholar] [CrossRef]
- Das, D.; Chatterjee, A.; Samanta, G.; Mandal, B.; Chowdhury, T.R.; Chowdhury, P.; Lodh, D. Arsenic contamination in groundwater in six districts of West Bengal, India: The biggest arsenic calamity in the world. Analyst 1994, 119, 168N–170N. [Google Scholar] [CrossRef]
- Berg, M.; Tran, H.C.; Nguyen, T.C.; Pham, H.V.; Schertenleib, R.; Giger, W. Arsenic contamination of groundwater and drinking water in Vietnam: A human health threat. Environ. Sci. Technol. 2001, 35, 2621–2626. [Google Scholar] [CrossRef]
- Hug, S.J.; Leupin, O.X.; Berg, M. Bangladesh and Vietnam: Different groundwater compositions require different approaches to arsenic mitigation. Environ. Sci. Technol. 2008, 42, 6318–6323. [Google Scholar] [CrossRef]
- Bundschuh, J.; Farias, B.; Martin, R.; Storniolo, A.; Bhattacharya, P.; Cortes, J.; Albouy, R. Groundwater arsenic in the Chaco-Pampean plain, Argentina: Case study from Robles county, Santiago del Estero province. Appl. Geochem. 2004, 19, 231–243. [Google Scholar] [CrossRef]
- Harvey, C.F.; Ashfaque, K.N.; Yu, W.; Badruzzaman, A.; Ali, M.A.; Oates, P.M.; Islam, S. Groundwater dynamics and arsenic contamination in Bangladesh. Chem. Geol. 2006, 228, 112–136. [Google Scholar] [CrossRef]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behaviour and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- World Health Organization. The World Health Report 2004: Changing History; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Ning, R.Y. Arsenic removal by reverse osmosis. Desalination 2002, 143, 237–241. [Google Scholar] [CrossRef]
- Leupin, O.X.; Hug, S.J. Oxidation and removal of arsenic (III) from aerated groundwater by filtration through sand and zero-valent iron. Water Res. 2005, 39, 1729–1740. [Google Scholar] [CrossRef]
- Jegadeesan, G.; Mondal, K.; Lalvani, S.B. Arsenate remediation using nanosized modified zerovalent iron particles. Environ. Prog. 2005, 24, 289–296. [Google Scholar] [CrossRef]
- Kim, T.U.; Drewes, J.E.; Summers, R.S.; Amy, G.L. Solute transport model for trace organic neutral and charged compounds through nanofiltration and reverse osmosis membranes. Water Res. 2007, 41, 3977–3988. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.K.; Núñez, P.; Grandon, R. Electrocoagulation as a remediation tool for wastewaters containing arsenic. Miner. Eng. 2006, 19, 521–524. [Google Scholar] [CrossRef]
- Owlad, M.; Aroua, M.K.; Daud, W.A.W.; Baroutian, S. Removal of hexavalent chromium-contaminated water and wastewater: A review. Water Air Soil Pollut. 2009, 200, 59–77. [Google Scholar] [CrossRef]
- Imran, M.; Iqbal, M.M.; Iqbal, J.; Shah, N.S.; Khan, Z.U.H.; Murtaza, B.; Rizwan, M. Synthesis, characterization and application of novel MnO and CuO impregnated biochar composites to sequester arsenic (As) from water: Modeling, thermodynamics and reusability. J. Hazard. Mater. 2021, 401, 123338. [Google Scholar] [CrossRef]
- Iqbal, M.M.; Imran, M.; Ali, B.; Nawaz, M.; Siddique, M.H.; Al-Kahtani, A.A.; Hussain, K.; Murtaza, B.; Shah, N.S.; Khan, Z.U.H. Nanocomposites of sedimentary material with ZnO and magnetite for the effective sequestration of arsenic from aqueous systems: Reusability, modeling and kinetics. Environ. Technol. Innov. 2021, 21, 101298. [Google Scholar] [CrossRef]
- Muedi, K.L.; Brink, H.G.; Masindi, V.; Maree, J.P. Effective removal of arsenate from wastewater using aluminium enriched ferric oxide-hydroxide recovered from authentic acid mine drainage. J. Hazard. Mater. 2021, 414, 125491. [Google Scholar] [CrossRef]
- Rahman, H.L.; Erdem, H.; Sahin, M.; Erdem, M. Iron-incorporated activated carbon synthesis from biomass mixture for enhanced arsenic adsorption. Water Air Soil Pollut. 2020, 231, 6. [Google Scholar] [CrossRef]
- Hussain, M.; Imran, M.; Abbas, G.; Shahid, M.; Iqbal, M.; Naeem, M.A.; Murtaza, B.; Amjad, M.; Shah, N.S.; Ul Haq Khan, Z. A new biochar from cotton stalks for As (V) removal from aqueous solutions: Its improvement with H3PO4 and KOH. Environ. Geochem. Health 2020, 42, 2519–2534. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, K.; Kobayashi, T.J. Arsenate and arsenite adsorbents composed of nano-sized cerium oxide deposited on activated alumina. Sep. Sci. Technol. 2019, 54, 523–534. [Google Scholar] [CrossRef]
- Tresintsi, S.; Simeonidis, K.; Katsikini, M.; Paloura, E.C.; Bantsis, G.; Mitrakas, M. A novel approach for arsenic adsorbents regeneration using MgO. J. Hazard. Mater. 2014, 265, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Salameh, Y.; Albadarin, A.B.; Allen, S.; Walker, G.; Ahmad, M.N.M. Arsenic (III, V) adsorption onto charred dolomite: Charring optimization and batch studies. Chem. Eng. J. 2015, 259, 663–671. [Google Scholar] [CrossRef]
- Chen, T.; Wei, Y.; Yang, W.; Liu, C. Highly efficient As (III) removal in water using millimeter-sized porous granular MgO-biochar with high adsorption capacity. J. Hazard. Mater. 2021, 416, 125822. [Google Scholar] [CrossRef]
- Xu, X.; Cao, X.; Zhao, L. Comparison of rice husk-and dairy manure-derived biochars for simultaneously removing heavy metals from aqueous solutions: Role of mineral components in biochars. Chemosphere 2013, 92, 955–961. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.; Pullammanappallil, P.; Yang, L. Biochar derived from anaerobically digested sugar beet tailings: Characterization and phosphate removal potential. Bioresour. Technol. 2011, 102, 6273–6278. [Google Scholar] [CrossRef]
- Tauber, M.J.; Mathies, R.A. Structure of the aqueous solvated electron from resonance Raman spectroscopy: Lessons from isotopic mixtures. J. Am. Chem. Soc. 2003, 125, 1394–1402. [Google Scholar] [CrossRef]
- Yadanaparthi, S.K.R.; Graybill, D.; von Wandruszka, R. Adsorbents for the removal of arsenic, cadmium, and lead from contaminated waters. J. Hazard. Mater. 2009, 171, 1–15. [Google Scholar] [CrossRef]
- Deng, Y.; Li, X.; Ni, F.; Liu, Q.; Yang, Y.; Wang, M.; Chen, W. Synthesis of magnesium modified biochar for removing copper, lead and cadmium in single and binary systems from aqueous solutions: Adsorption mechanism. Water 2021, 13, 599. [Google Scholar] [CrossRef]
- Wetteland, C.L.; de Jesus Sanchez, J.; Silken, C.A.; Nguyen, N.-Y.T.; Mahmood, O.; Liu, H. Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids. J. Nanoparticle Res. 2018, 20, 215. [Google Scholar] [CrossRef]
- Biçer, M.; Şişman, İ. Controlled synthesis of copper nano/microstructures using ascorbic acid in aqueous CTAB solution. Powder Technol. 2010, 198, 279–284. [Google Scholar] [CrossRef]
- Zhu, D.; Chen, Y.; Yang, H.; Wang, S.; Wang, X.; Zhang, S.; Chen, H. Synthesis and characterization of magnesium oxide nanoparticle-containing biochar composites for efficient phosphorus removal from aqueous solution. Chemosphere 2020, 247, 125847. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Ali, S.; Adrees, M.; Rizvi, H.; Zia-ur-Rehman, M.; Hannan, F.; Ok, Y.S. Cadmium stress in rice: Toxic effects, tolerance mechanisms, and management: A critical review. Environ. Sci. Pollut. Res. 2016, 23, 17859–17879. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, B.; Li, Y. Enhanced arsenic removal by biochar modified with nickel (Ni) and manganese (Mn) oxyhydroxides. J. Ind. Eng. Chem. 2016, 37, 361–365. [Google Scholar] [CrossRef]
- Din, S.U.; Mahmood, T.; Naeem, A.; Hamayun, M.; Shah, N.S. Detailed kinetics study of arsenate adsorption by a sequentially precipitated binary oxide of iron and silicon. Environ. Technol. 2019, 40, 261–269. [Google Scholar] [CrossRef]
- Banerjee, K.; Amy, G.L.; Prevost, M.; Nour, S.; Jekel, M.; Gallagher, P.M.; Blumenschein, C.D. Kinetic and thermodynamic aspects of adsorption of arsenic onto granular ferric hydroxide (GFH). Water Res. 2008, 42, 3371–3378. [Google Scholar] [CrossRef]
- Zhu, D.; Yang, H.; Chen, X.; Chen, W.; Cai, N.; Chen, Y.; Chen, H. Temperature-dependent magnesium citrate modified formation of MgO nanoparticles biochar composites with efficient phosphate removal. Chemosphere 2021, 274, 129904. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Niazi, N.K.; Hassan, N.E.; Bibi, I.; Wang, H.; Tsang, D.C.; Rinklebe, J. Wood-based biochar for the removal of potentially toxic elements in water and wastewater: A critical review. Int. Mater. Rev. 2019, 64, 216–247. [Google Scholar] [CrossRef]
- Van Vinh, N.; Zafar, M.; Behera, S.K.; Park, H.S. Arsenic (III) removal from aqueous solution by raw and zinc-loaded pine cone biochar: Equilibrium, kinetics, and thermodynamics studies. Environ. Sci. Technol. 2015, 12, 1283–1294. [Google Scholar] [CrossRef]
- Lin, L.; Zhou, S.; Huang, Q.; Huang, Y.; Qiu, W.; Song, Z. Capacity and mechanism of arsenic adsorption on red soil supplemented with ferromanganese oxide–biochar composites. Environ. Sci. Pollut. Res. 2018, 25, 20116–20124. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Ohana, N.; HM, L.F.; Luis, N.G. Adsorption of arsenic anions in water using modified lignocellulosic adsorbents. Results Eng. 2022, 13, 100340. [Google Scholar]
- Shaikh, W.A.; Alam, M.A.; Alam, M.O.; Chakraborty, S.; Owens, G.; Bhattacharya, T. Enhanced aqueous phase arsenic removal by a biochar based iron nanocomposite. Environ. Technol. Innov. 2020, 19, 100936. [Google Scholar] [CrossRef]
- Cuong, D.V.; Wu, P.C.; Liou, S.Y.H.; Hou, C.H. An integrated active biochar filter and capacitive deionization system for high-performance removal of arsenic from groundwater. J. Hazard. Mater. 2022, 423, 127084. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Pei, P.; Sun, Y.; Xu, Y.; Jia, H. Performance and mechanism of As (III/Ⅴ) removal from aqueous solution by novel positively charged animal-derived biochar. Sep. Purif. Technol. 2022, 290, 120836. Available online: https://www.sciencedirect.com/science/article/abs/pii/S1383586622003938 (accessed on 19 September 2022). [CrossRef]
- Lin, L.; Gao, M.; Song, Z.; Mu, H. Mitigating arsenic accumulation in rice (Oryza sativa L.) using Fe-Mn-La-impregnated biochar composites in arsenic-contaminated paddy soil. Environ. Sci. Pollut. Res. 2020, 27, 41446–41457. [Google Scholar] [CrossRef]
- Niazi, N.K.; Bibi, I.; Shahid, M.; Ok, Y.S.; Shaheen, S.M.; Rinklebe, J.; Lüttge, A. Arsenic removal by Japanese oak wood biochar in aqueous solutions and well water: Investigating arsenic fate using integrated spectroscopic and microscopic techniques. Sci. Total Environ. 2018, 621, 1642–1651. [Google Scholar] [CrossRef]
- Lin, M.F.; Nguyen, N.T.; Chen, C.K.; Le, T.T.; Chen, S.S.; Chen, P.H. Preparation of Metal Modified onto Biochar from Hazardous Waste for Arsenic Removal. J. Nanosci. Nanotechnol. 2021, 21, 3227–3236. [Google Scholar] [CrossRef]
- Shen, C.; Gu, L.; Chen, S.; Jiang, Y.; Huang, P.; Li, H.; Xia, D. Sewage sludge derived FeCl3-activated biochars as efficient adsorbents for the treatment of toxic As (III) and Cr (VI) wastewater. Environ. Chem. Eng. 2022, 10, 108575. [Google Scholar] [CrossRef]
- Balarak, D.; Abasizadeh, H.; Yan, J.-K.; Shim, M.J.; Lee, S.-M. Biosorption of Acid Orange 7 (AO7) dye by canola waste: Equilibrium, kinetic and thermodynamics studies. Desalin. Water Treat. 2020, 190, 331–339. [Google Scholar] [CrossRef]
- Linh, N.L.M.; Van Hoang, D.; Duong, T.; Tinh, M.X.; Quang Khieu, D. Adsorption of arsenate from aqueous solution onto modified Vietnamese bentonite. Adv. Mater. Sci. Eng. 2019, 2019, 2710926. [Google Scholar]
- Sanou, Y.; Samuel, P.J. The Comparative study of adsorption capacity of two mixed materials for arsenic remediation in aqueous solutions. J. Environ. Treat. Tech. 2021, 9, 559–565. [Google Scholar]
- Drweesh, S.A.; Fathy, N.A.; Wahba, M.A.; Hanna, A.A.; Akarish, A.I.; Elzahany, E.A.; Abou-El-Sherbini, K.S. Equilibrium, kinetic and thermodynamic studies of Pb (II) adsorption from aqueous solutions on HCl-treated Egyptian kaolin. J. Environ. Chem. Eng. 2016, 4, 1674–1684. [Google Scholar] [CrossRef]
- Manzoor, Q.; Nadeem, R.; Iqbal, M.; Saeed, R.; Ansari, T.M. Organic acids pretreatment effect on Rosa bourbonia phyto-biomass for removal of Pb (II) and Cu (II) from aqueous media. Bioresour. Technol. 2013, 132, 446–452. [Google Scholar] [CrossRef] [PubMed]

| Temperature (K) | k2 (g m−1 mol−1) | Xe × 107 (mol g−1) | Ea (kJ mol−1) |
|---|---|---|---|
| 303 | 1.41 | 29.010 | 20.61 |
| 313 | 1.49 | 32.562 | |
| 323 | 1.57 | 37.707 |
| Temperature (K) | Xmax × 102 (mg g−1) | Kb × 10−4 (L mol−1) |
|---|---|---|
| 303 | 19.7 | 2.018 |
| 313 | 23.1 | 9.103 |
| 323 | 24.3 | 15.43 |
| Adsorbent | Xm (mg g−1) | References |
|---|---|---|
| Zn-loaded biochar | 0.28 | [41] |
| Ferromanganese oxide–biochar | 0.712 | [42] |
| Lignocelluloic | 0.94 | [43] |
| Nano-zerovalent iron biochar | 1.40 | [44] |
| Integrated active biochar filter | 2.12 | [45] |
| Animal-derived biochar | 2.76 | [46] |
| Fe-Mn-La-impregnated biochar | 3.18 | [47] |
| Japanese-oak-wood biochar | 3.89 | [48] |
| Metal-modified biochar | 4.12 | [49] |
| FeCl3-activated biochar | 4.4 | [50] |
| MgO-IBC composite | 4.86 | Current study |
| ∆H (kJ mol−1) | ∆S (J mol−1) | ∆G (kJ mol−1) 303 K | ∆G (kJ mol−1) 313 K | ∆G (kJ mol−1) 318 K |
|---|---|---|---|---|
| 83.28 | 0.3584 | −24.97 | −29.71 | −32.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Din, S.U.; Hussain, B.; Haq, S.; Imran, M.; Ahmad, P.; Khandaker, M.U.; Rehman, F.U.; Eldin, S.M.; Mousa, A.A.A.; Khan, I.; et al. Efficient Arsenate Decontamination from Water Using MgO-Itsit Biochar Composite: An Equilibrium, Kinetics and Thermodynamic Study. Water 2022, 14, 3559. https://doi.org/10.3390/w14213559
Din SU, Hussain B, Haq S, Imran M, Ahmad P, Khandaker MU, Rehman FU, Eldin SM, Mousa AAA, Khan I, et al. Efficient Arsenate Decontamination from Water Using MgO-Itsit Biochar Composite: An Equilibrium, Kinetics and Thermodynamic Study. Water. 2022; 14(21):3559. https://doi.org/10.3390/w14213559
Chicago/Turabian StyleDin, Salah Ud, Babar Hussain, Sirajul Haq, Muhammad Imran, Pervaiz Ahmad, Mayeen Uddin Khandaker, Fazal Ur Rehman, Sayed M. Eldin, Abd Allah A. Mousa, Ilyas Khan, and et al. 2022. "Efficient Arsenate Decontamination from Water Using MgO-Itsit Biochar Composite: An Equilibrium, Kinetics and Thermodynamic Study" Water 14, no. 21: 3559. https://doi.org/10.3390/w14213559
APA StyleDin, S. U., Hussain, B., Haq, S., Imran, M., Ahmad, P., Khandaker, M. U., Rehman, F. U., Eldin, S. M., Mousa, A. A. A., Khan, I., & Emran, T. B. (2022). Efficient Arsenate Decontamination from Water Using MgO-Itsit Biochar Composite: An Equilibrium, Kinetics and Thermodynamic Study. Water, 14(21), 3559. https://doi.org/10.3390/w14213559














