Assessment of Seasonal Changes on the Carbon Cycle in the Critical Zone of a Surface Water (SW)–Groundwater (GW) System
Abstract
:1. Introduction
2. Materials and Methods
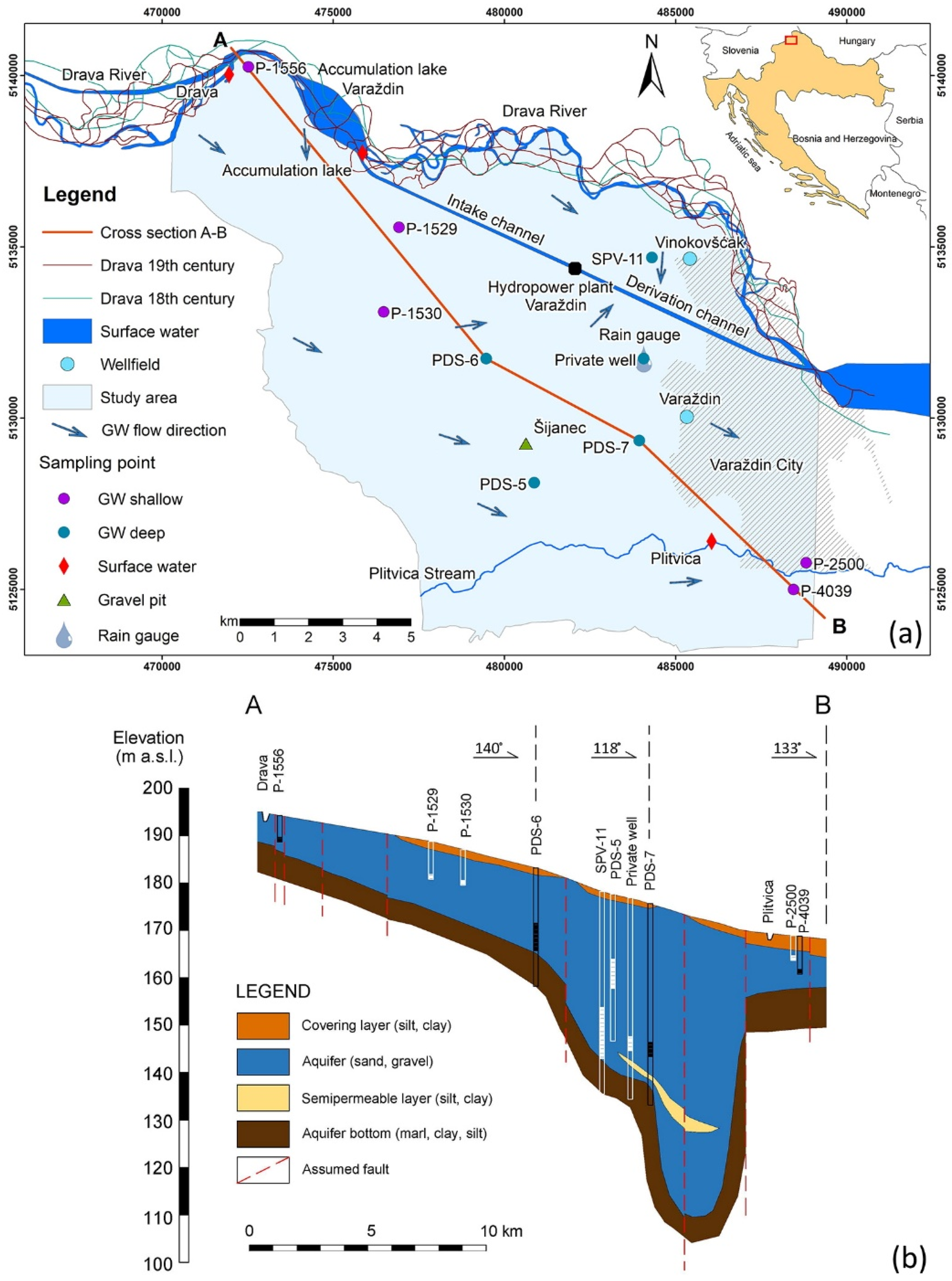
2.1. Study Area
2.2. Sampling Procedure
2.3. Analytical Methods
2.3.1. Determination of δ13CDIC
2.3.2. Determination of δ13C in Plant and Manure Samples
2.3.3. Determination of δ13Ccarb
2.3.4. Determination of δ13Cbulk and δ13Corg in Soil
2.4. Data Processing
3. Results and Discussion
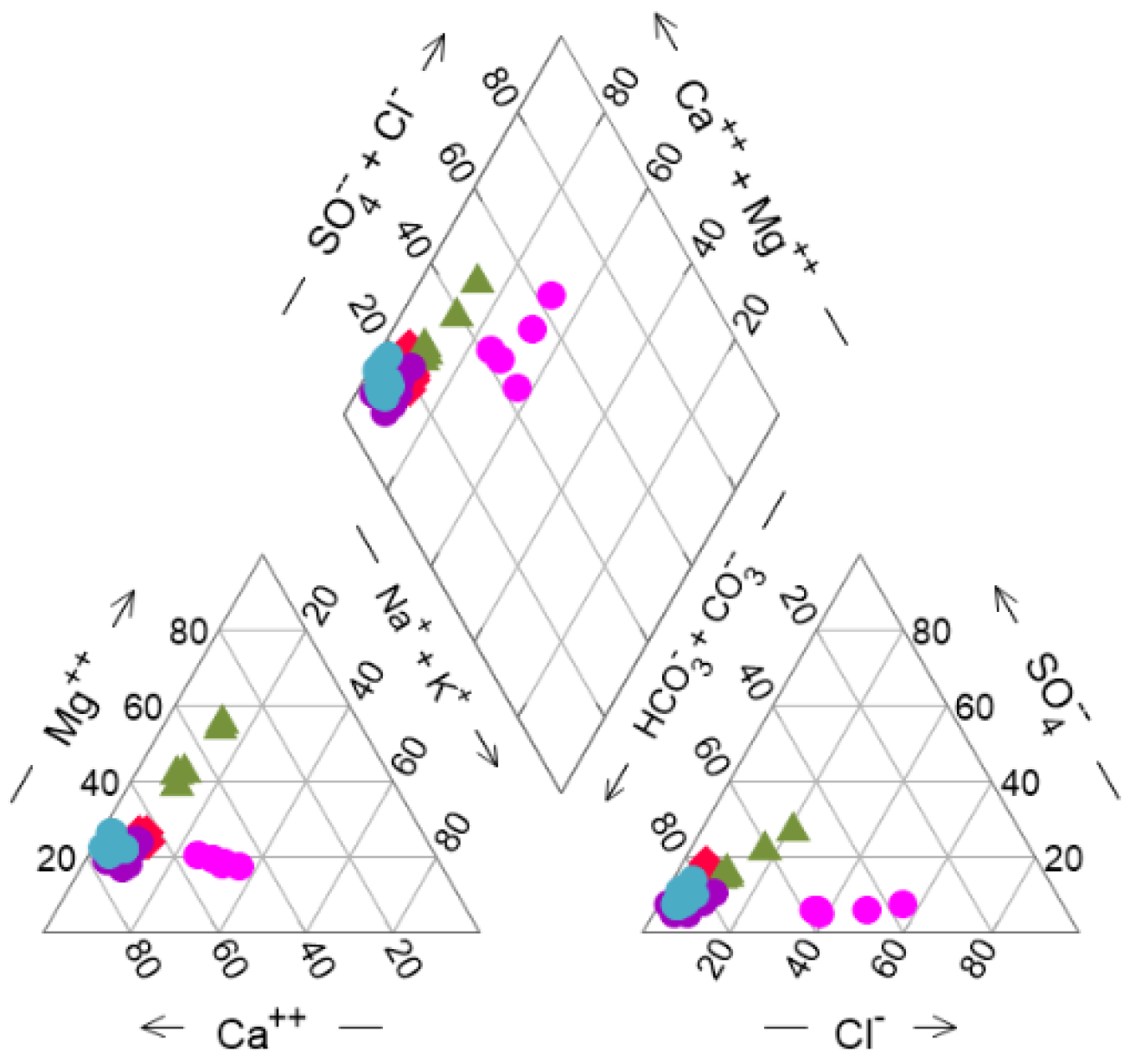
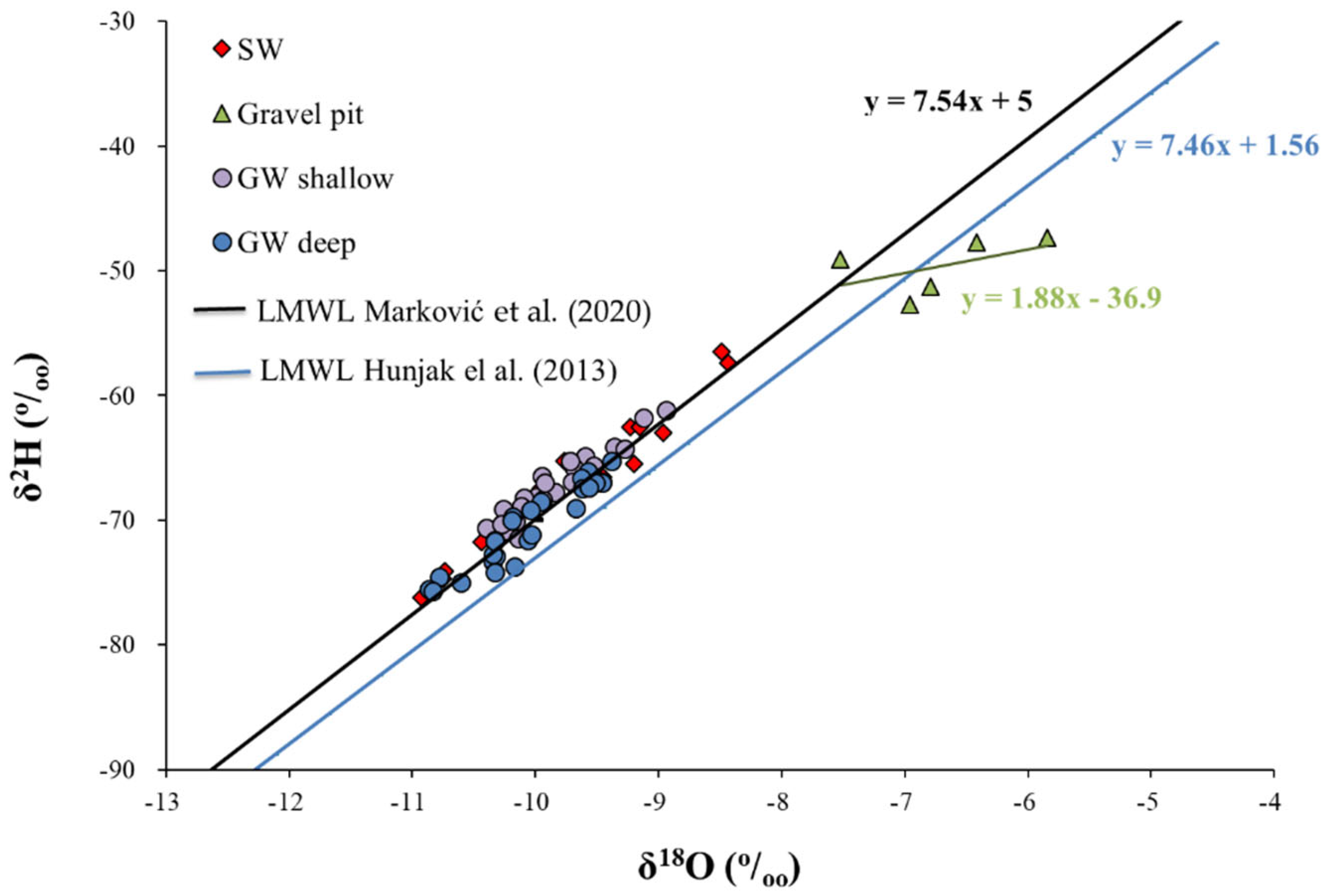
3.1. Hydrochemical and Isotopic Composition of GW and SW
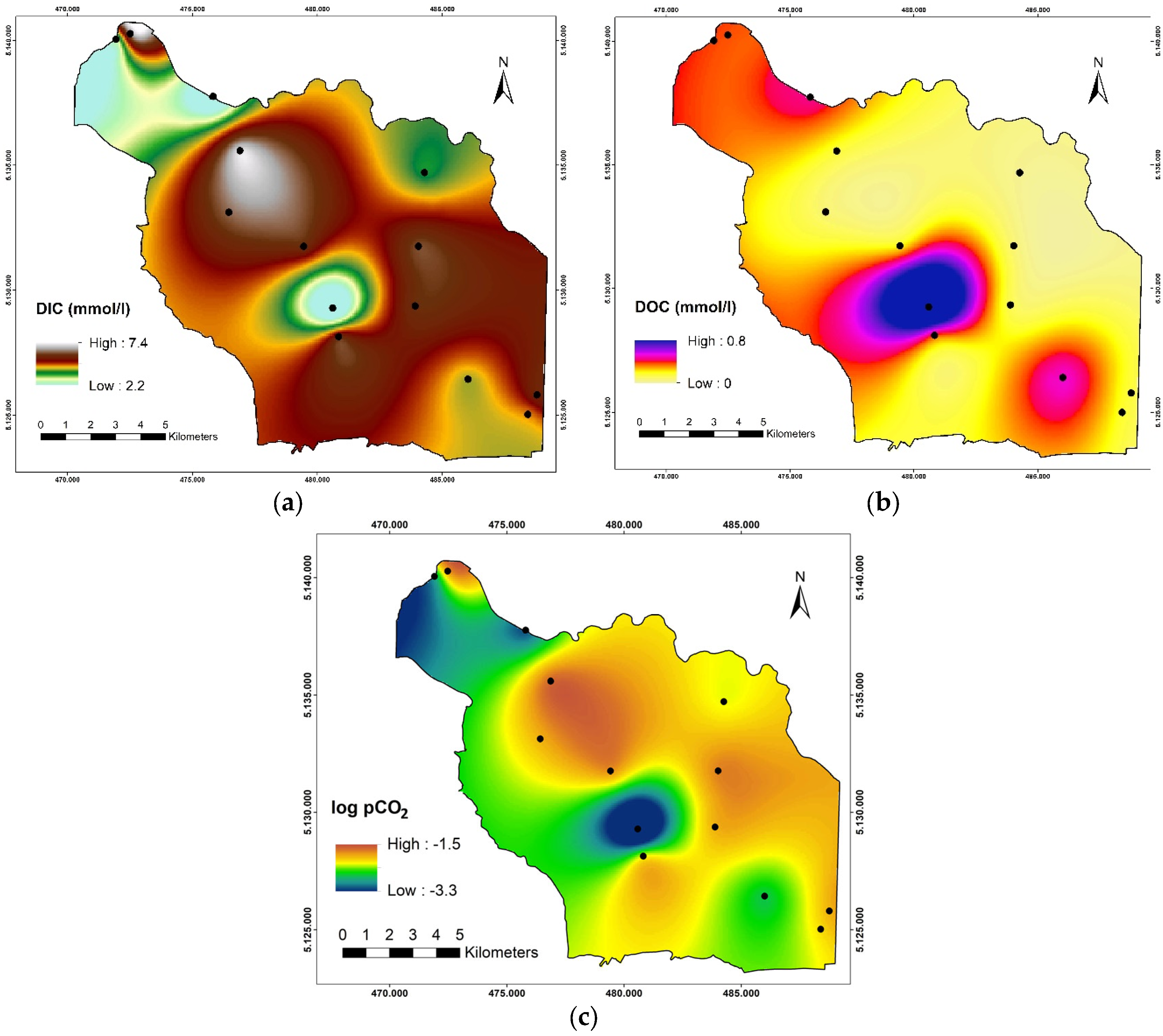
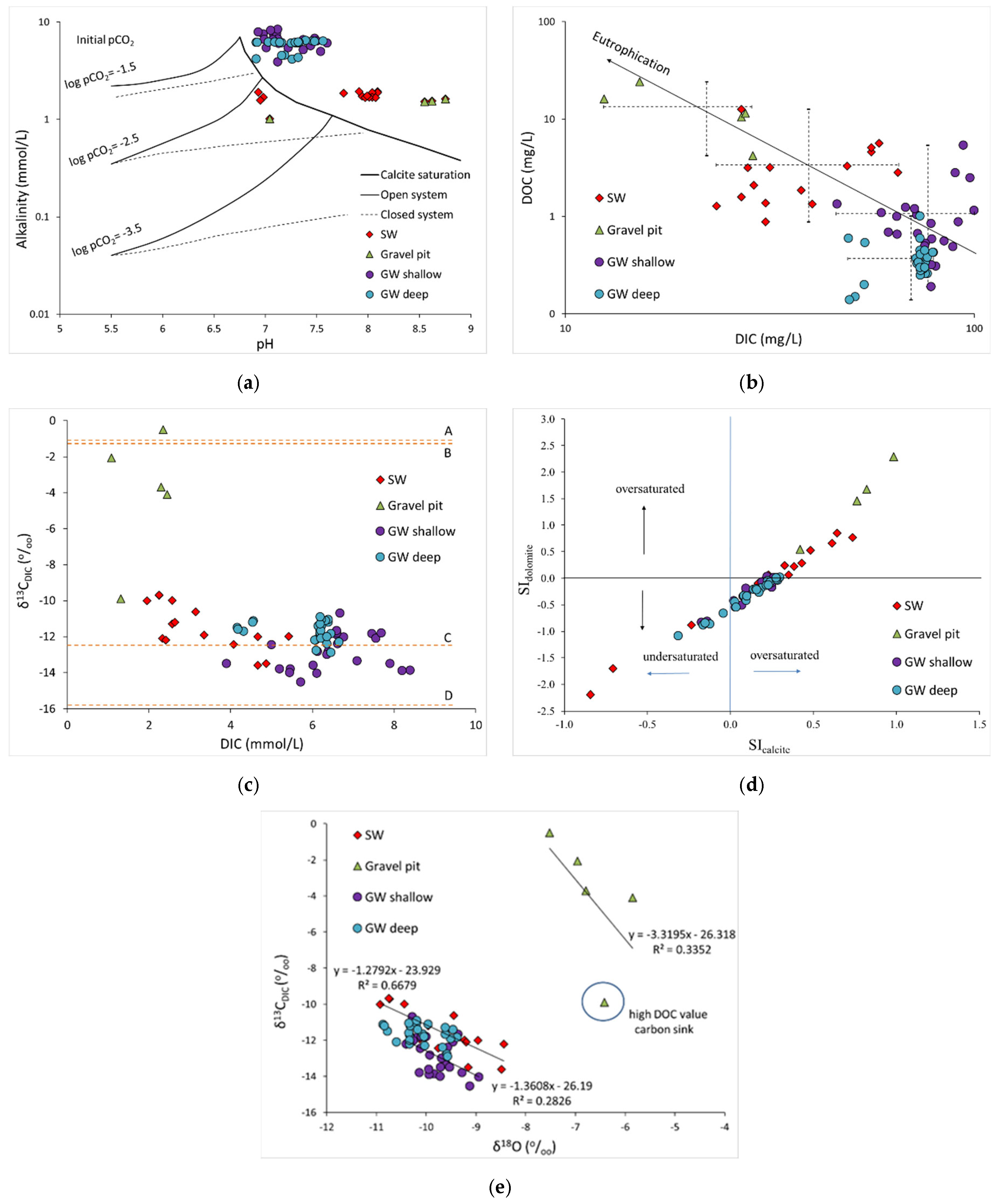
3.2. Spatial and Seasonal Variation of Carbon Species and Evaluation of Biogeochemical Processes
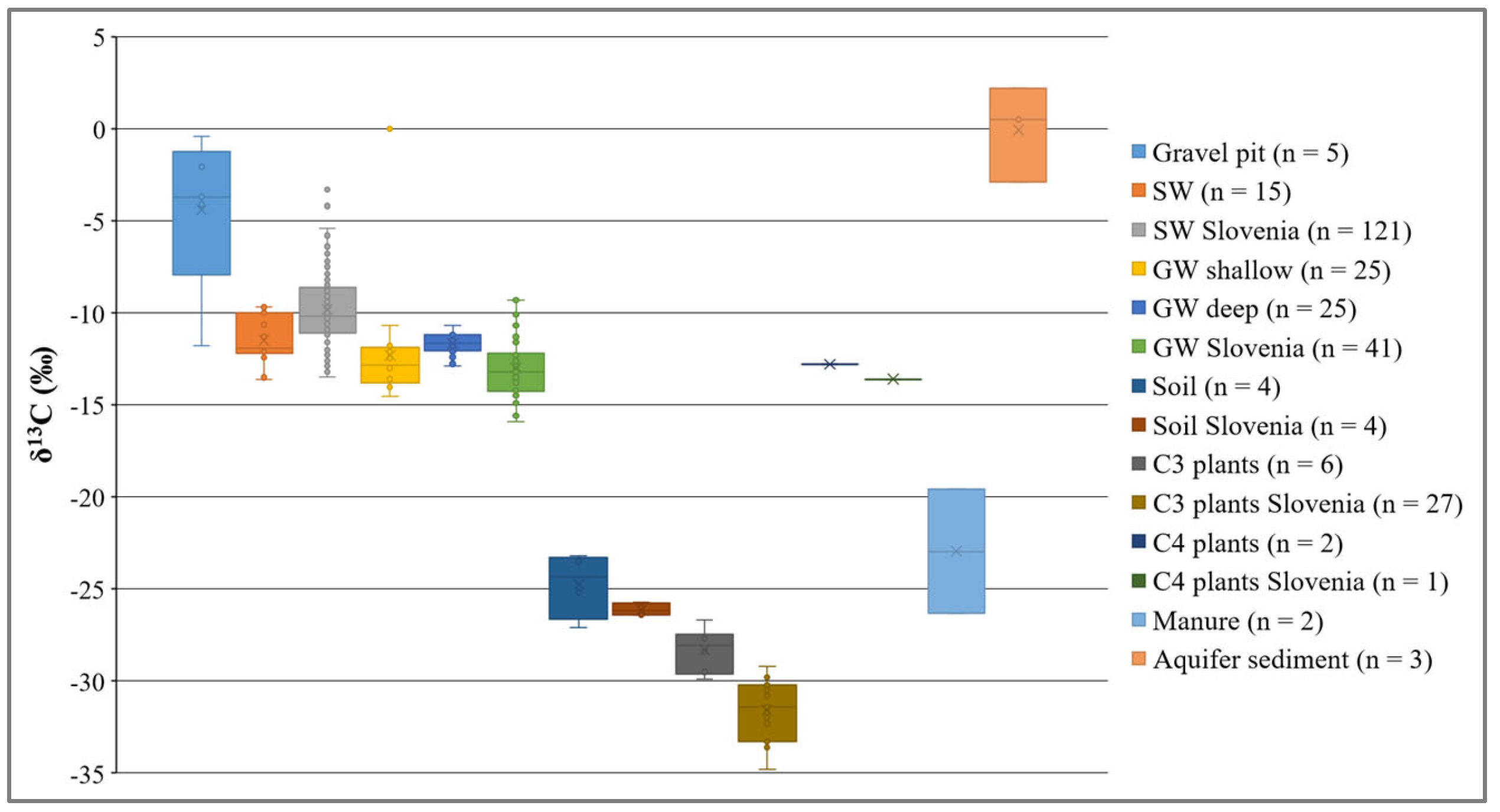
3.3. Carbon Sources and Sinks in SW–GW System
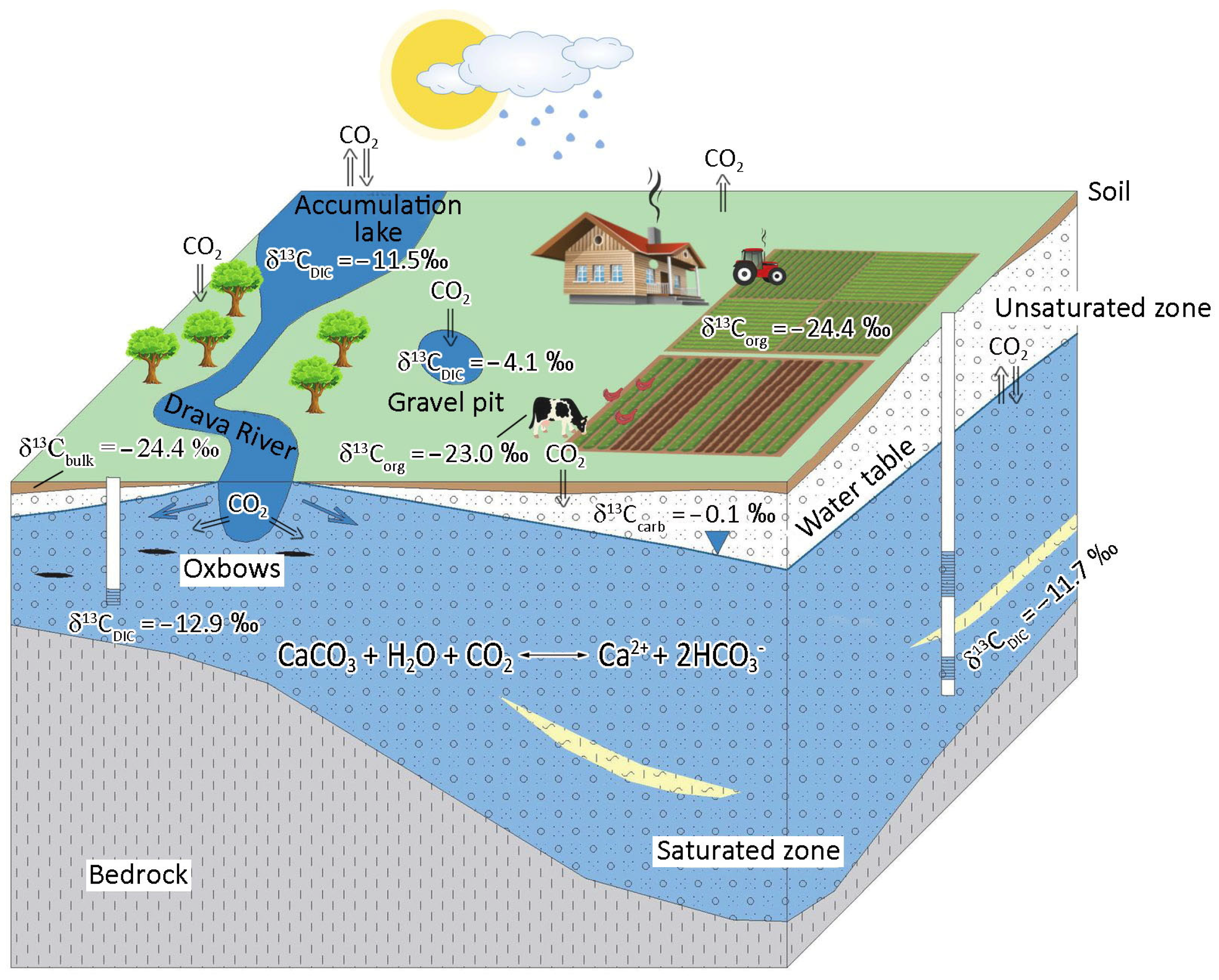
3.4. Conceptual Model for Catchment Area Based on Hydrological and Carbon Isotopic Data
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Sprenger, M.; Stumpp, C.; Weiler, M.; Aeschbach, W.; Allen, S.T.; Benettin, P.; Dubbert, M.; Hartmann, A.; Hrachowitz, M.; Kirchner, J.W.; et al. The demographics of water: A review of water ages in the critical zone. Rev. Geophys. 2019, 57, 800–834. [Google Scholar] [CrossRef] [Green Version]
- National Research Council. Basic Research Opportunities in Earth Science; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Grant, G.E.; Dietrich, W.E. The frontier beneath our feet. Water Resour. Res. 2017, 53, 2605–2609. [Google Scholar] [CrossRef]
- Mayorga, E.; Aufdenkampe, A.K.; Masiello, C.A.; Krusche, A.V.; Hedges, J.I.; Quay, P.D.; Richey, J.E.; Brown, T.A. Young organic matter as a source of carbon dioxide outgassing from Amazonian rivers. Nature 2005, 436, 538–541. [Google Scholar] [CrossRef] [Green Version]
- Kanduč, T.; Szramek, K.; Ogrinc, N.; Walter, L.M. Origin and cycling of riverine inorganic carbon in the Sava River watershed (Slovenia) inferred from major solutes and stable carbon isotopes. Biogeochemistry 2007, 86, 137–154. [Google Scholar] [CrossRef]
- Kanduč, T.; Ogrinc, N.; Mrak, T. Characteristics of suspended matter in the River Sava watershed, Slovenia. Isot. Environ. Health Stud. 2007, 43, 369–385. [Google Scholar] [CrossRef]
- Porowska, D. Determination of the origin of dissolved inorganic carbon in groundwater and reclaimed landfill in Otwock using stable carbon isotopes. Waste Manag. 2015, 39, 216–225. [Google Scholar] [CrossRef]
- Monger, H.C.; Kraimer, R.A.; Khresat, S.; Cole, D.R.; Wang, X.; Wang, J. Sequestration of inorganic carbon in soil and groundwater. Geology 2015, 43, 375–378. [Google Scholar] [CrossRef]
- Druhan, J.L.; Lawrence, C.R.; Coney, A.K.; Giometta, M.G.; Oster, J.L. A reactive transport approach to modelling cave seepage water chemistry I: Carbon isotope transformations. Geochim. Cosmochim. Acta 2021, 311, 374–400. [Google Scholar] [CrossRef]
- Li, C.; Gao, X.; Wang, W.; Zhang, X.; Jiang, C.; Wang, Y. Hydro-biogeochemical processes of surface water: Leakage into groundwater in large scale karst water system: A case study at Jinci, northern China. J. Hydrol. 2021, 596, 125691. [Google Scholar] [CrossRef]
- Zuliani, T.; Kanduč, T.; Novak, R.; Vreča, P. Characterization of bottled waters by multielemental analysis, stable and radiogenic isotopes. Water 2020, 12, 2454. [Google Scholar] [CrossRef]
- Nagode, K.; Kanduč, T.; Zuliani, T.; Bračič-Železnik, B.; Jamnik, B.; Vreča, P. Daily fluctuations in the isotope and elemental composition of tap water in Ljubljana, Slovenia. Water 2021, 13, 1451. [Google Scholar] [CrossRef]
- Atekwana, E.A.; Krishnamurthy, R.V. Seasonal variations of dissolved inorganic carbon and δ13C of surface waters: Application of a modified gas evolution technique. J. Hydrol. 1998, 205, 265–278. [Google Scholar] [CrossRef]
- Colombani, N.; Mostracicco, M.; Costaldelli, G.; Aravena, R. Contrasting biogeochemical processes revealed by stable isotopes of H2O, N, C, S in shallow aquifers underlying agricultural lowlands. Sci. Total Environ. 2019, 691, 1283–1296. [Google Scholar] [CrossRef]
- Berner, R.A. Biological aspects of the long-term carbon cycle. Bull. Inst. Océanogr. 1996, 14, 11–22. [Google Scholar]
- Scholes, R.J.; Monteiro, P.M.; Sabine, C.L.; Canadell, J.G. Systematic long-term observations of the global carbon cycle. Trends Ecol. Evol. 2009, 24, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Kanduč, T.; Šlejkovec, Z.; Vreča, P.; Samardžija, Z.; Verbovšek, T.; Božič, D.; Jamnikar, S.; Solomon, D.K.; Fernandez, D.P.; Eastoe, C.; et al. The effect of geochemical processes on groundwater in the Velenje coal basin, Slovenia: Insights from mineralogy, trace elements and isotope signatures. SN Appl. Sci. 2018, 1, 1518. [Google Scholar] [CrossRef] [Green Version]
- Mahlangu, S.; Lorentz, S.; Diamond, R.; Dippenaar, M. Surface water—Groundwater interaction using tritium and stable water isotopes: A case study of Middelburg, South Africa. J. Afr. Earth Sci. 2020, 171, 103886. [Google Scholar] [CrossRef]
- Marković, T.; Karlović, I.; Perčec Tadić, M.; Larva, O. Application of Stable Water Isotopes to Improve Conceptual Model of Alluvial Aquifer in the Varaždin Area. Water 2020, 12, 379. [Google Scholar] [CrossRef] [Green Version]
- Karlović, I.; Marković, T.; Vujnović, T. Groundwater Recharge Assessment Using Multi Component Analysis: Case Study at the NW Edge of the Varaždin Alluvial Aquifer, Croatia. Water 2022, 14, 42. [Google Scholar] [CrossRef]
- Rozanski, K.; Araguas-Araguas, L.; Gonfiantini, R. Isotopic patterns in modern global precipitation. Continental Isotopic Indicators of Climate. Am. Geophys. Union Monogr. 1993, 78, 1–36. [Google Scholar]
- Vreča, P.; Krajcar Bronić, I.; Horvatinčić, N.; Barešić, J. Isotopic characteristics of precipitation in Slovenia and Croatia: Comparison of continental and maritime stations. J. Hydrol. 2006, 330, 457–469. [Google Scholar] [CrossRef]
- Dotsika, E.; Lykoudis, S.; Poutoukis, D. Spatial distribution of the isotopic composition of precipitation and spring water in Greece. Glob. Planet. Change 2010, 71, 141–149. [Google Scholar] [CrossRef]
- Kanduč, T.; Mori, N.; Kocman, D.; Stibilj, V.; Grassa, F. Hydrogeochemistry of Alpine springs from North Slovenia: Insights from stable isotopes. Chem. Geol. 2012, 300–301, 40–54. [Google Scholar] [CrossRef]
- Lukač Reberski, J.; Marković, T.; Nakić, Z. Definition of the river Gacka springs subcatchment areas on the basis of hydrogeological parameters. Geol. Croat. 2013, 66, 39–53. [Google Scholar] [CrossRef]
- Marković, T.; Brkić, Ž.; Larva, O. Using hydrochemical data and modelling to enhance the knowledge of groundwater flow and quality in an alluvial aquifer of Zagreb, Croatia. Sci. Total Environ. 2013, 458–460, 508–516. [Google Scholar] [CrossRef]
- Kanduč, T.; Grassa, F.; McIntosh, J.; Stibilj, V.; Ulrich-Supovec, M.; Supovec, I.; Jamnikar, S. A geochemical and stable isotope investigation of groundwater/surface water interactions in the Velenje Basin, Slovenia. Hydrogeol. J. 2014, 22, 971–984. [Google Scholar] [CrossRef]
- Cervi, F.; Dadomo, A.; Martinelli, G. The Analysis of Short-Term Dataset of Water Stable Isotopes Provides Information on Hydrological Processes Occurring in Large Catchments from the Northern Italian Apennines. Water 2019, 11, 1360. [Google Scholar] [CrossRef] [Green Version]
- Gross, E.; Andrews, S.; Bergamaschi, B.; Downing, B.; Holleman, R.; Burdick, S.; Durand, D. The Use of Stable Isotope-Based Water Age to Evaluate a Hydrodynamic Model. Water 2019, 11, 2207. [Google Scholar] [CrossRef] [Green Version]
- Hepburn, E.; Cendón, D.I.; Bekele, D.; Currel, M. Environmental isotopes as indications of groundwater recharge, residence times and salinity in a coastal urban redevelopment precinct in Australia. Hydrogeol. J. 2020, 28, 503–520. [Google Scholar] [CrossRef]
- Karlović, I.; Marković, T.; Šparica Miko, M.; Maldini, K. Geochemical Characteristics of Alluvial Aquifer in the Varaždin Region. Water 2021, 13, 1508. [Google Scholar] [CrossRef]
- Karlović, I.; Marković, T.; Vujnović, T.; Larva, O. Development of a Hydrogeological Conceptual Model of the Varaždin Alluvial Aquifer. Hydrology 2021, 8, 19. [Google Scholar] [CrossRef]
- Corine Land Cover. Available online: https://land.copernicus.eu/pan-european/corine-land-cover/clc2018 (accessed on 22 September 2022).
- Prelogović, E.; Velić, I. Quaternary tectonic activity in western part of Drava basin. Geol. Vjesn. 1988, 41, 237–253. [Google Scholar]
- Babić, Ž.; Čakarun, I.; Sokač, A.; Mraz, V. On geological features of quaternary sediments of Drava basin on Croatian territory. Geol. Vjesn. 1978, 30, 43–61. [Google Scholar]
- Urumović, K.; Hlevnjak, B.; Prelogović, E.; Mayer, D. Hydrogeological conditions of Varaždin aquifer. Geol. Vjesn. 1990, 43, 149–158. [Google Scholar]
- Crnički, J. Mineralne sirovine varaždinske regije i njihovo privredno značenje. In Varaždinski Zbornik 1181-1981. Varaždin, Croatia, 1–3 October 1981; Mohorovičić, A., Ed.; Jugoslavenska Akademija Znanosti i Umjetnosti: Zagreb, Croatia, 1983; pp. 75–85. [Google Scholar]
- Mioč, P.; Marković, S. Osnovna Geološka Karta SFRJ 1:100.000. Tumač za List Čakovec L33–57 (Basic Geological Map of SFRY, Scale 1:100.000. The Map Book for Sheet Čakovec L33–57); Geological Survey: Zagreb, Croatia; Geological, Geotechnical and Geophysical Survey: Ljubljana, Slovenia, 1998. (In Croatian) [Google Scholar]
- Karlović, I.; Pavlić, K.; Posavec, K.; Marković, T. Analysis of the hydraulic connection of the Plitvica stream and the groundwater of the Varaždin alluvial aquifer. Geofizika 2021, 38, 15–35. [Google Scholar] [CrossRef]
- Nimac, I.; Perčec Tadić, M. New 1981–2010 Climatological Normals for Croatia and Comparison to Previous 1961–1990 and 1971–2000 Normals, Proceedings from GeoMLA Conference; University of Belgrade—Faculty of Civil Engineering: Belgrade, Serbia, 2016; pp. 79–85. [Google Scholar]
- Miyajima, T.; Yamada, Y.; Hanba, Y.T. Determining the stable isotope ratio of total dissolved inorganic carbon in lake water by GC/C/IRMS. Limnol. Oceanogr. 1995, 40, 994–1000. [Google Scholar] [CrossRef]
- Spötl, C. A robust and fast method of sampling and analysis of δ13C of dissolved inorganic carbon in groundwaters. Isot. Environ. Health Stud. 2005, 41, 217–221. [Google Scholar] [CrossRef]
- Coplen, T.B. New guidances for reporting stable hydrogen, carbon and oxygen isotopes ratio data. Geochim. Cosmochim. Acta 1996, 60, 390–3360. [Google Scholar] [CrossRef]
- Brand, W.A.; Coplen, T.B.; Vogl, J.; Rosner, M.; Prohaska, T. Assessment of international reference materials for isotope-ratio analysis (IUPAC technical report). Pure Appl. Chem. 2014, 86, 425–467. [Google Scholar] [CrossRef] [Green Version]
- McCrea, J.M. On the Isotopic chemistry of carbonates and a paleo temperature Scale. J. Chem. Phys. 1950, 18, 849–857. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; U.S. Geological Survey Techniques and Methods, Book 6; USGS: Denver, CO, USA, 2013; Chapter A43; p. 497. Available online: https://pubs.usgs.gov/tm/06/a43/ (accessed on 22 September 2022).
- Zhong, J.; Li, S.; Tao, F.; Yue, F.; Liu, C.-Q. Sensitivity of chemical weathering and dissolved carbon dynamics to hydrological conditions in a typical karst river. Sci. Rep. 2017, 7, 42944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunjak, T.; Lutz, H.O.; Roller-Lutz, Z. Stable isotope composition of the meteoric precipitation in Croatia. Isot. Environ. Health Stud. 2013, 49, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Karlović, I.; Posavec, K.; Larva, O.; Marković, T. Numerical groundwater flow and nitrate transport assessment in alluvial aquifer of Varaždin region, NW Croatia. J. Hydrol. Reg. Stud. 2022, 41, 101084. [Google Scholar] [CrossRef]
- Kanduč, T. Hidrogeokemične Značilnosti in Kroženje Ogljika v Porečju Reke Save v Sloveniji. PhD Thesis, University of Ljubljana, Faculty of Natural Sciences and Engineering, Department of geology, Ljubljana, Slovenia, 2006; p. 141, appendixes. (In Slovenian). [Google Scholar]
- Šturm, M.; Vreča, P.; Krajcar Bronić, I. Carbon isotopic composition (δ13C and 14C activity) of plant samples in the vicinity of the Slovene nuclear power plant. J. Environ. Radioact. 2012, 110, 24–29. [Google Scholar] [CrossRef]
- Kulaš, A.; Marković, T.; Žutinić, P.; Kajan, K.; Karlović, I.; Orlić, S.; Keskin, E.; Filipović, V.; Gligora Udovič, M. Succession of Microbial Community in a Small Water Body within the Alluvial Aquifer of a Large River. Water 2021, 13, 115. [Google Scholar] [CrossRef]
- Barth, J.A.C.; Veizer, J. Carbon cycle in St. Lawrence aquatic ecosystems at Cornwall (Ontario), Canada: Seasonal and spatial variations. Chem. Geol. 1999, 159, 107–128. [Google Scholar] [CrossRef]
- Kulaš, A.; Gligora Udovič, M.; Žutinić, P. Biodervisity Report for Surface Waters Drava River, Accumulation Lake Varaždin, Gravel Pit in Šijanec and Plitvica Stream for the TRANITAL Project; Croatian Geological Survey: Zagreb, Croatia, 2019; 50p. (In Croatian) [Google Scholar]
- Wollast, R.; Chou, L. Rate control of weathering of silicate minerals at room temperature and pressure. In Physical and Chemical Weathering in Geochemical Cycles; Lerman, A., Meybeck, M., Eds.; NATO ASI Series; Springer: Dordrecht, The Netherlands, 1988; Volume 251. [Google Scholar] [CrossRef]
- Kessler, T.J.; Harvey, C.F. The global flux of carbon dioxide into groundwater. Geophys. Res. Lett. 2001, 28, 279–282. [Google Scholar] [CrossRef] [Green Version]
- Cai, W.-J.; Guo, X.; Chen, C.-T.A.; Dai, M.; Zhang, L.; Zhai, W.; Lohrenz, S.E.; Yin, K.; Harrison, P.J.; Wang, Y. A comparative overview of weathering intensity and HCO3− flux in the world’s major rivers with emphasis on the Changjiang, Huanghe, Zhujiang (Pearl) and Mississippi Rivers. Cont. Shelf Res. 2008, 28, 1538–1549. [Google Scholar] [CrossRef]
- Hartmann, J.; Jansen, N.; Dürr, H.H.; Kempe, S.; Köhler, P. Global CO2-consumption by chemical weathering: What is the contribution of highly active weathering regions? Glob. Planet. Change 2009, 69, 185–194. [Google Scholar] [CrossRef]
| Observation Well | Latitude (° N) | Longitude (° E) | Elevation (m a.s.l.) | Well Depth (m) | Type of Well | Depth Interval of the Screen (m) | Min Water Level (m a.s.l.) | Max Water Level (m a.s.l.) |
|---|---|---|---|---|---|---|---|---|
| Private well | 46.325158 | 16.293106 | 176.00 | 15.0 | Deep | 5–15 | 169.28 | 169.90 |
| PDS-5 | 46.292462 | 16.25173 | 178.36 | 31.0 | Deep | 13.7–19.7 | 174.25 | 174.86 |
| PDS-6 | 46.324993 | 16.233373 | 184.07 | 25.0 | Deep | 11.7–17.7 | 176.05 | 177.20 |
| PDS-7 | 46.303596 | 16.291492 | 175.71 | 42.5 | Deep | 29.3–32.3 | 170.26 | 170.84 |
| P-1529 | 46.359419 | 16.200068 | 187.32 | 8.0 | Shallow | n.a. 1 | 181.98 | 182.40 |
| P-1530 | 46.337192 | 16.194381 | 183.72 | 7.5 | Shallow | n.a. 1 | 177.46 | 178.31 |
| P-1556 | 46.401421 | 16.142607 | 193.03 | 5.6 | Shallow | n.a. 1 | 190.11 | 190.77 |
| P-2500 | 46.271647 | 16.354878 | 167.81 | 5.2 | Shallow | n.a. 1 | 164.61 | 165.02 |
| P-4039 | 46.264594 | 16.350134 | 167.76 | 8.0 | Shallow | n.a. 1 | 164.62 | 165.29 |
| SPV-11 | 46.351711 | 16.296098 | 177.69 | 40.0 | Deep | 24.5–35.8 | 170.96 | 171.51 |
| Sampling Point | SW | Gravel Pit | GW Shallow | GW Deep | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Values | Min | Max | Average | Min | Max | Average | Min | Max | Average | Min | Max | Average |
| EC (µS/cm) | 232 | 584 | 403 | 229 | 332 | 283 | 660 | 1091 | 807 | 434 | 769 | 669 |
| T (°C) | 3.3 | 22.7 | 13.9 | 3.0 | 32.1 | 18.3 | 8.8 | 15.6 | 13.3 | 12.1 | 13.6 | 12.8 |
| pH | 6.93 | 8.09 | 7.87 | 7.04 | 10.09 | 8.61 | 6.93 | 7.6 | 7.22 | 6.91 | 7.56 | 7.22 |
| DO (mg/l) | 6.4 | 12.7 | 9.4 | 10.5 | 19.5 | 14.6 | 0.2 | 11.8 | 5.6 | 1.5 | 11 | 7.3 |
| Ca2+ (mg/L) | 38.9 | 85.6 | 55.0 | 13.3 | 35.4 | 25.3 | 87.5 | 136.2 | 111.3 | 74.0 | 120.8 | 104.5 |
| Mg2+ (mg/L) | 6.6 | 17.1 | 11.7 | 14.5 | 17.0 | 16.3 | 16.5 | 25.3 | 20.5 | 16.3 | 21.5 | 19.5 |
| Na+ (mg/L) | 3.5 | 11.4 | 7.6 | 6.0 | 8.9 | 7.3 | 5.3 | 80.0 | 24.2 | 2.4 | 10.4 | 6.1 |
| K+ (mg/L) | 0.7 | 3.3 | 1.9 | 0.6 | 1.1 | 0.9 | 1.1 | 6.7 | 3.4 | 0.4 | 12.6 | 4.1 |
| DIC (mg/L) | 23.4 | 65.0 | 39.3 | 12.4 | 28.7 | 22.2 | 46.2 | 99.9 | 77.0 | 49.3 | 79.1 | 70.0 |
| DOC (mg/L) | 0.9 | 12.6 | 3.4 | 4.2 | 24.3 | 13.3 | 0.2 | 5.4 | 1.0 | 0.1 | 1.0 | 0.4 |
| HCO3− (mg/L) | 122 | 334 | 203 | 66 | 149 | 116 | 238 | 512 | 395 | 254 | 406 | 360 |
| δ13CDIC (‰) | −13.6 | −9.7 | −11.5 | −9.9 | −0.5 | −4.1 | −14.5 | −10.7 | −12.9 | −12.9 | −10.9 | −11.7 |
| δ18O (o/oo) | −10.93 | −8.44 | −9.73 | −7.52 | −5.85 | −6.71 | −10.40 | −8.94 | −9.80 | −10.87 | −9.38 | −10.09 |
| δ2H (o/oo) | −76.2 | −56.6 | −66.9 | −52.7 | −47.4 | −49.6 | −71.5 | −61.2 | −67.1 | −75.6 | −65.3 | −70.7 |
| Cl− (mg/L) | 5.5 | 13.8 | 9.5 | 12.4 | 15.9 | 13.8 | 7.1 | 212.7 | 46.8 | 6.7 | 19.5 | 12.7 |
| SO42− (mg/L) | 18.4 | 27.2 | 22.3 | 22.2 | 27.4 | 24.4 | 20.1 | 35.4 | 26.0 | 22.0 | 33.6 | 26.9 |
| PO43−-P (mg/L) | <0.01 | 0.07 | 0.03 | 0.01 | 0.06 | 0.03 | <0.01 | 0.16 | 0.04 | <0.01 | 0.04 | 0.02 |
| NH4+ (mg/L) | <0.01 | 0.18 | 0.03 | <0.01 | 0.35 | 0.12 | <0.01 | 0.06 | 0.03 | <0.01 | 0.02 | 0.02 |
| NO2− (mg/L) | 0.006 | 0.101 | 0.03 | 0.014 | 0.199 | 0.07 | <0.001 | 0.026 | 0.007 | <0.001 | 0.033 | 0.006 |
| NO3− (mg/L) | 5.3 | 23.2 | 11.3 | 5.1 | 13.7 | 11.2 | <0.1 | 58.9 | 35.5 | <0.1 | 95.6 | 49.0 |
| SiO2 (mg/L) | 1.0 | 18.9 | 10.3 | 3.2 | 25.0 | 12.2 | 10.1 | 24.7 | 14.5 | 11.5 | 18.9 | 14.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karlović, I.; Marković, T.; Kanduč, T.; Vreča, P. Assessment of Seasonal Changes on the Carbon Cycle in the Critical Zone of a Surface Water (SW)–Groundwater (GW) System. Water 2022, 14, 3372. https://doi.org/10.3390/w14213372
Karlović I, Marković T, Kanduč T, Vreča P. Assessment of Seasonal Changes on the Carbon Cycle in the Critical Zone of a Surface Water (SW)–Groundwater (GW) System. Water. 2022; 14(21):3372. https://doi.org/10.3390/w14213372
Chicago/Turabian StyleKarlović, Igor, Tamara Marković, Tjaša Kanduč, and Polona Vreča. 2022. "Assessment of Seasonal Changes on the Carbon Cycle in the Critical Zone of a Surface Water (SW)–Groundwater (GW) System" Water 14, no. 21: 3372. https://doi.org/10.3390/w14213372
APA StyleKarlović, I., Marković, T., Kanduč, T., & Vreča, P. (2022). Assessment of Seasonal Changes on the Carbon Cycle in the Critical Zone of a Surface Water (SW)–Groundwater (GW) System. Water, 14(21), 3372. https://doi.org/10.3390/w14213372










