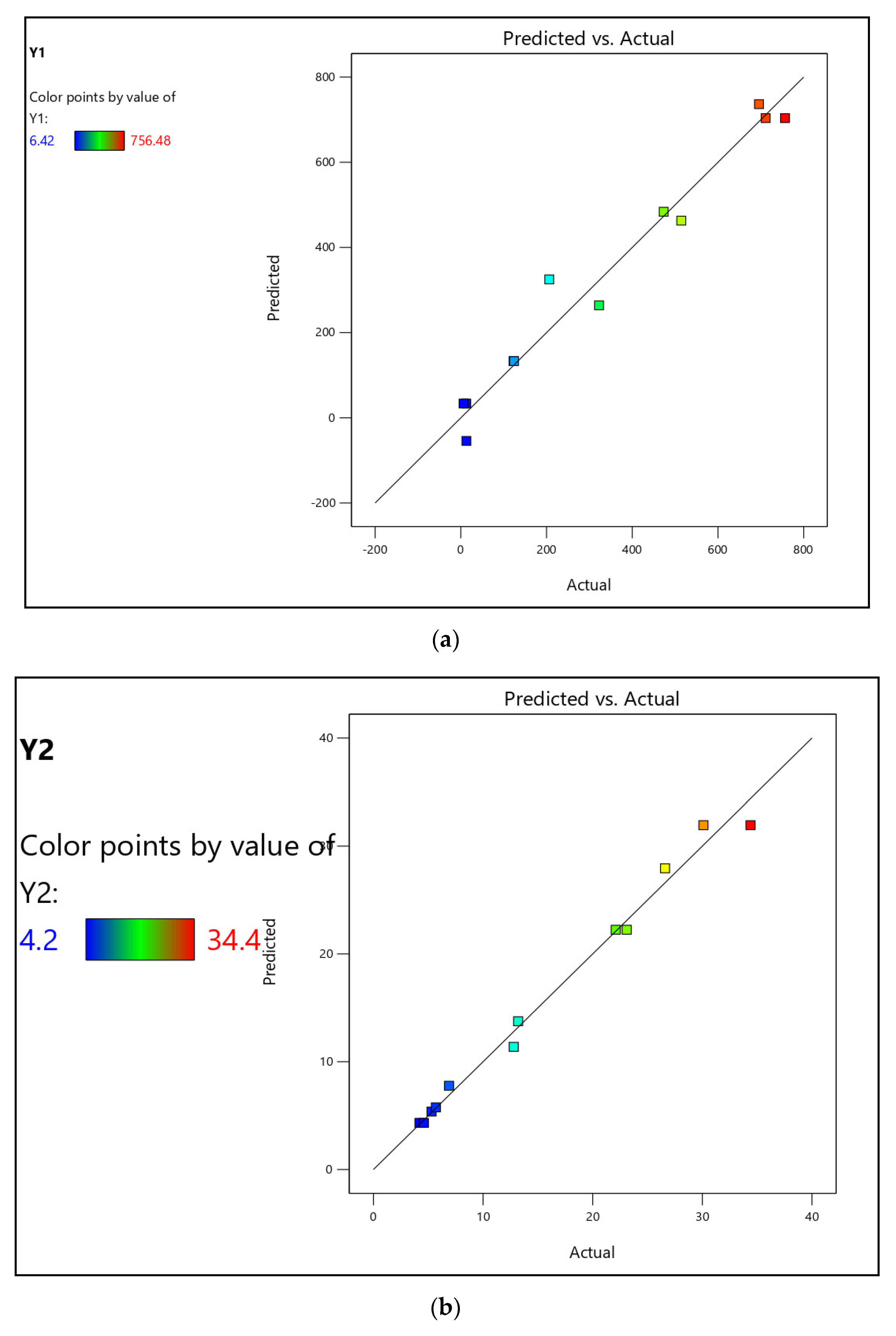
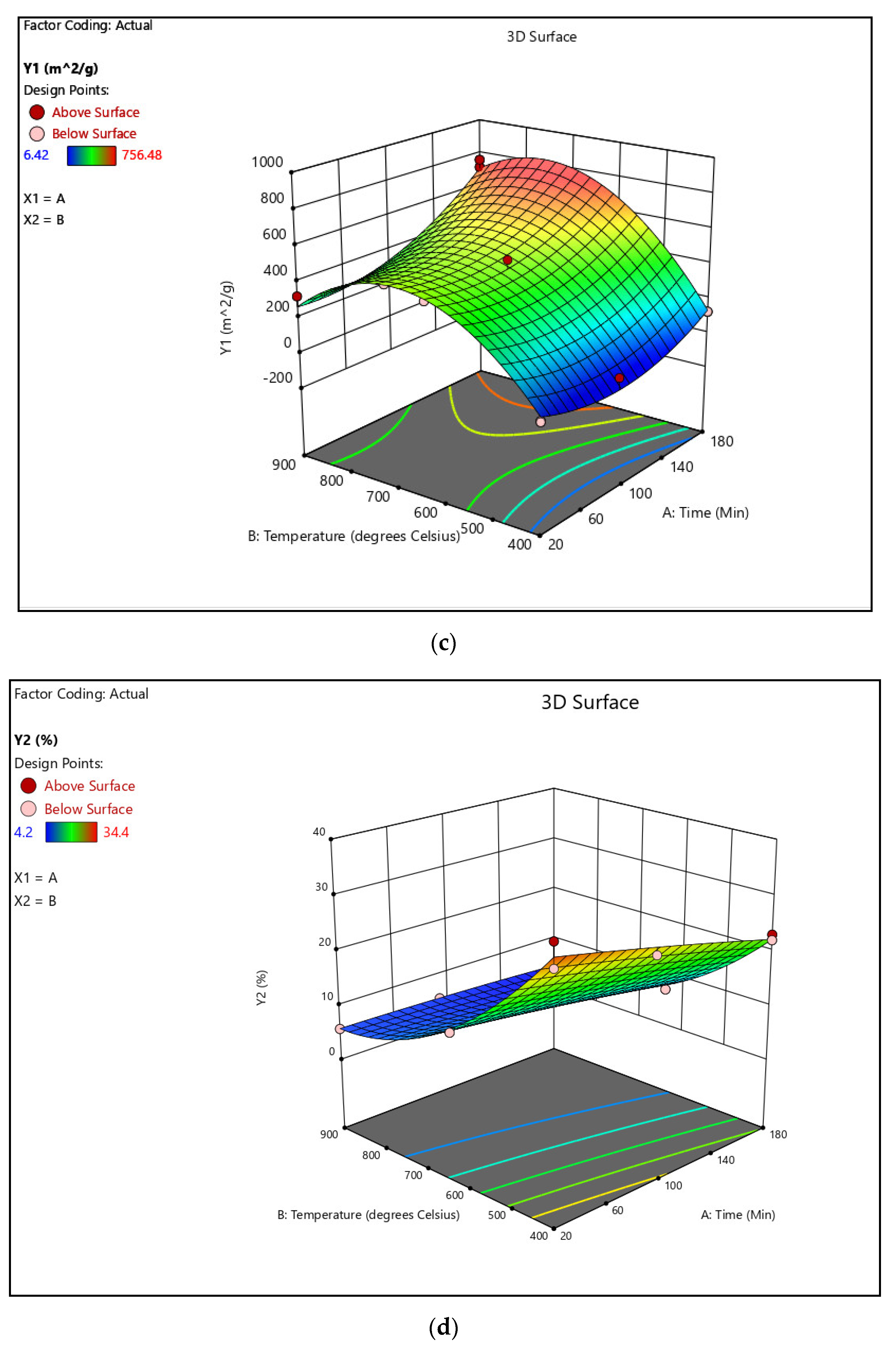
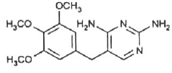
3.2. Competitive Removal of APIs by CPAC
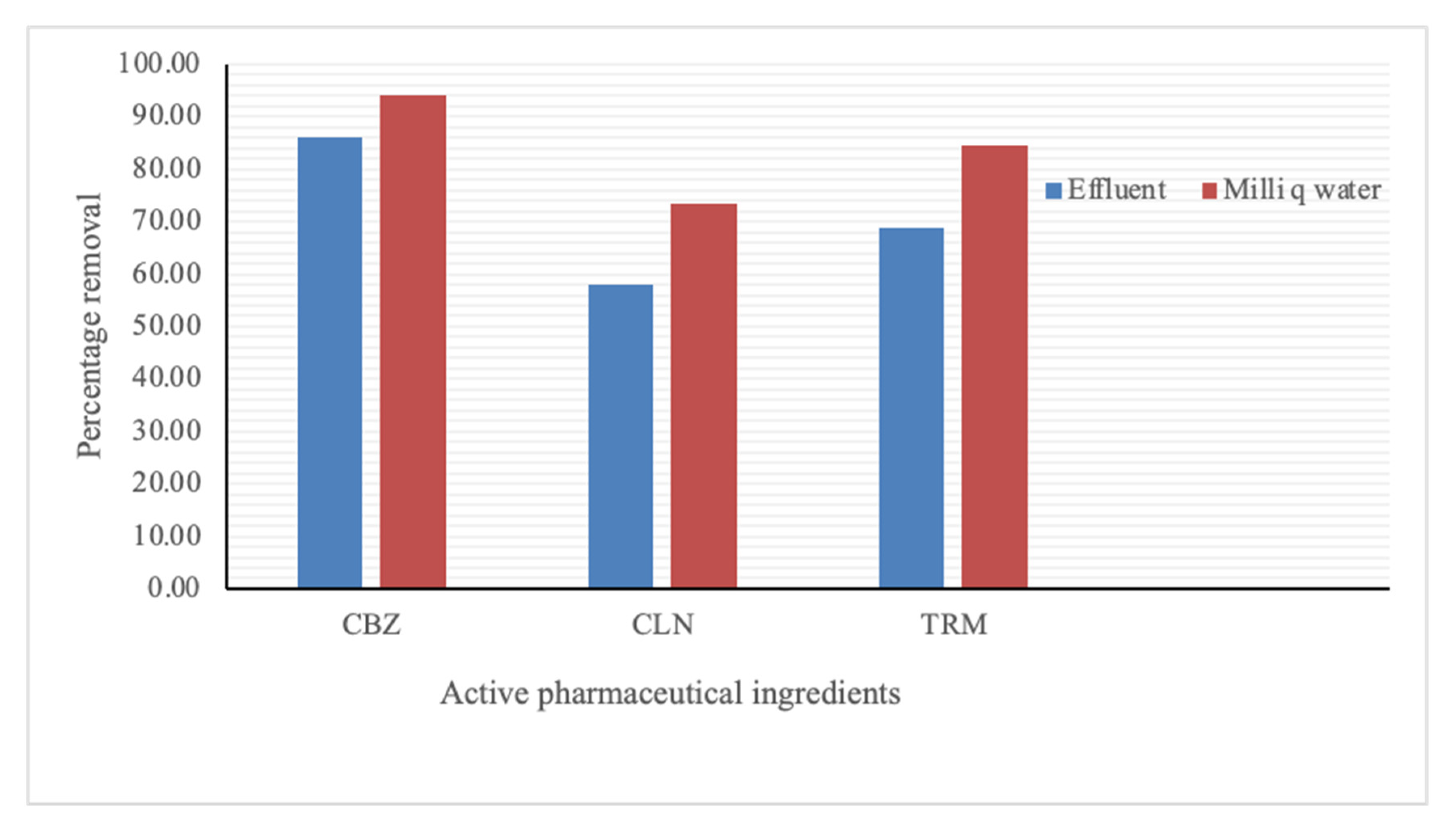
The maximum removal percentages of CBZ, CLN, and TRM from the effluent water were 86.00, 58.00, and 68.75%, respectively. From the Milli-Q water, a similar pattern was observed at 94.25, 73.50, and 84.50% for CBZ, CLN, and TRM, respectively, as shown in
Figure 2. The adsorption could have been both chemical (through
n-π bonding between the CPAC surface groups and the APIs) and physical (through diffusion into the CPAC sites). The dominant functional groups in the cassava peel activated carbon are hydroxyl and carboxyl groups [
37,
38,
39]. The deprotonated functional groups could have provided vacant pairs of electrons that are favorable for divalent bonding with more protonated APIs. This in turn may have increased the adsorption sites and consequently the electrostatic bonding forces. As shown in
Table 2, the hydrogen bond acceptor counts for the studied APIs are in the order CLN > TRM > CBZ and are greater than the hydrogen bond donor counts for both TRM and CLN but equal for CBZ. The implication is that electrostatic interactions occur between APIs and CPAC functional groups with strength values in the order of CBZ > TRM > CLN. These interactions partly explain the adsorption of the APIs in the same order. Moreover, pharmaceuticals with higher proton donor counts have been found to be better removed from solutions compared to those with neutral and lower proton donor counts [
12].
Organic hydrophilic micropollutants have in general a lower affinity for AC than hydrophobic micropollutants [
40]. The hydrophobic APIs are highly insoluble in water and are better removed from the solutions since they have more affinity for the adsorbents. The high insolubility in the water partly explains why the three-API CBZ was the most sequestrated. Moreover, Kumar and Siril [
41] reported CBZ as one of the practically insoluble drugs in water, with an improvement in its solubility being only possible at an ultra-fine nanoparticle size. Trimethoprim, being hydrophilic and highly soluble in water, would be expected to be the least adsorbed API, yet it was sequestrated more than CLN from both the effluent and Milli-Q water. The molecular weight of the CLN outweighed its hydrophobicity and insolubility in water and could not be accommodated effectively in the CPAC pores. Additionally, the steric hindrance due to its large molecules could have weakened the electrostatic interactions with the CPAC molecules [
42]. Pore diffusion, therefore, was the dominant mass transfer mechanism [
43].
3.2.1. Effect of CPAC Dosage
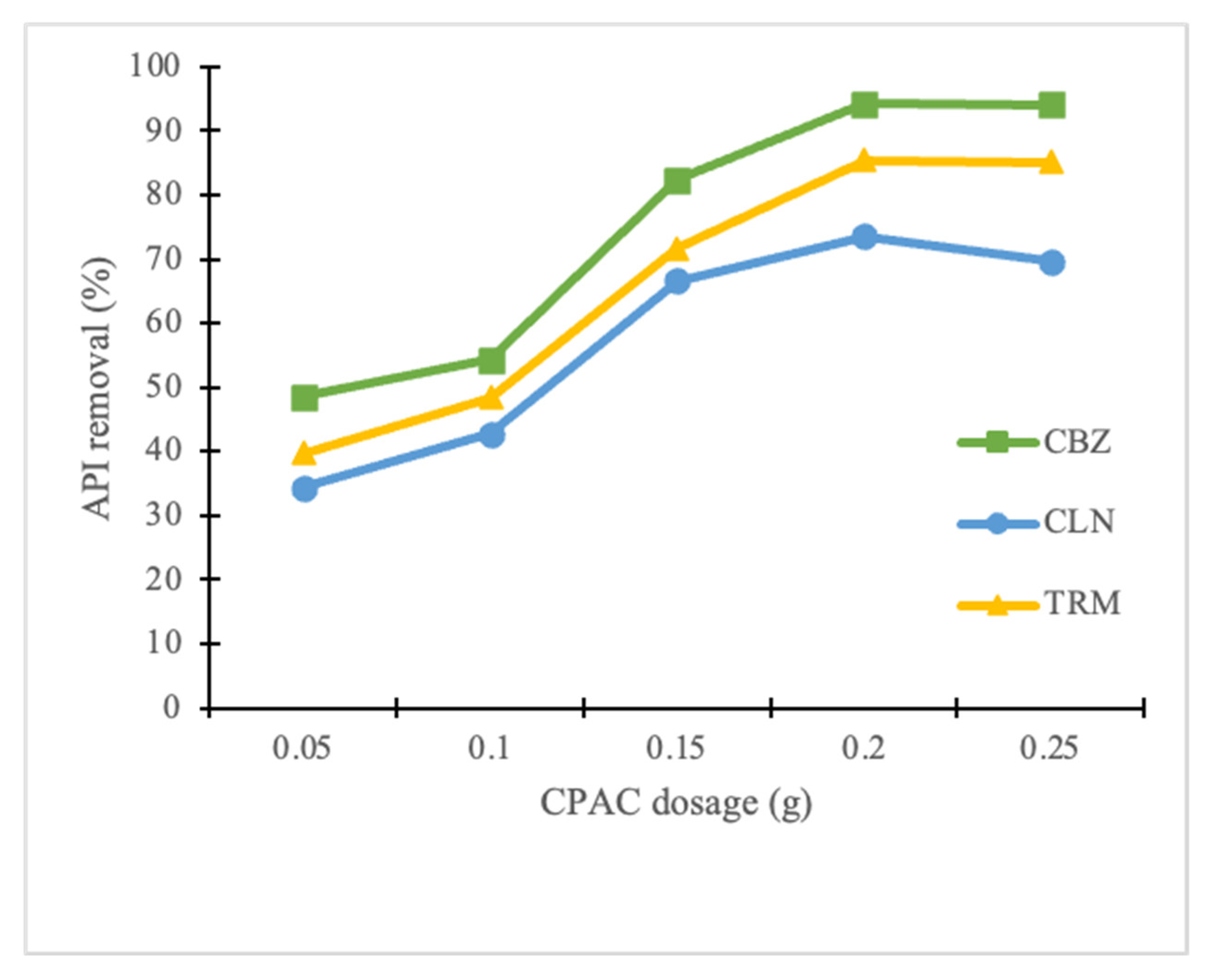
The results showed that when the CPAC dosage increased from 0.05 to 0.25 g, the API removal also increased gradually for all the APIs from 48.5% to 94.3%, 34.4 to 73.6%, and 39.7 to 85.5% for CBZ, CLN, and TRM, respectively, as shown in
Figure 3. The increase in CPAC dosage provided a larger surface area and an increase in the number of adsorbing sites on the CPAC [
44]. The results from this experiment showed that 0.2 g of CAPC when added to a solution containing 20 mg/L of CBZ, CLN, or TRM solution produces the highest removal efficiency rates for the respective APIs. At the 0.25 g dosage, the removal rates for CBZ and TRM were almost maintained at the same level as for the 0.20 g dosage at 94.1% and 85.2%, respectively, while the CLN removal was remarkably reduced to 69.6%. This implies that increasing the CPAC dosage beyond 0.2 g could not correspondingly increase the percentage removal of the APIs. A similar scenario was observed by Gorzin and Bahri [
45] in their study on the adsorption of Cr (VI) from an aqueous solution by an adsorbent prepared from paper mill sludge. This could have been due to the increase in the number of unsaturated CPAC adsorption sites reducing the CPAC adsorption density. This experiment confirmed that the CPAC dosage influences the removal efficiency of APIs from water.
3.2.2. Effect of Contact Time
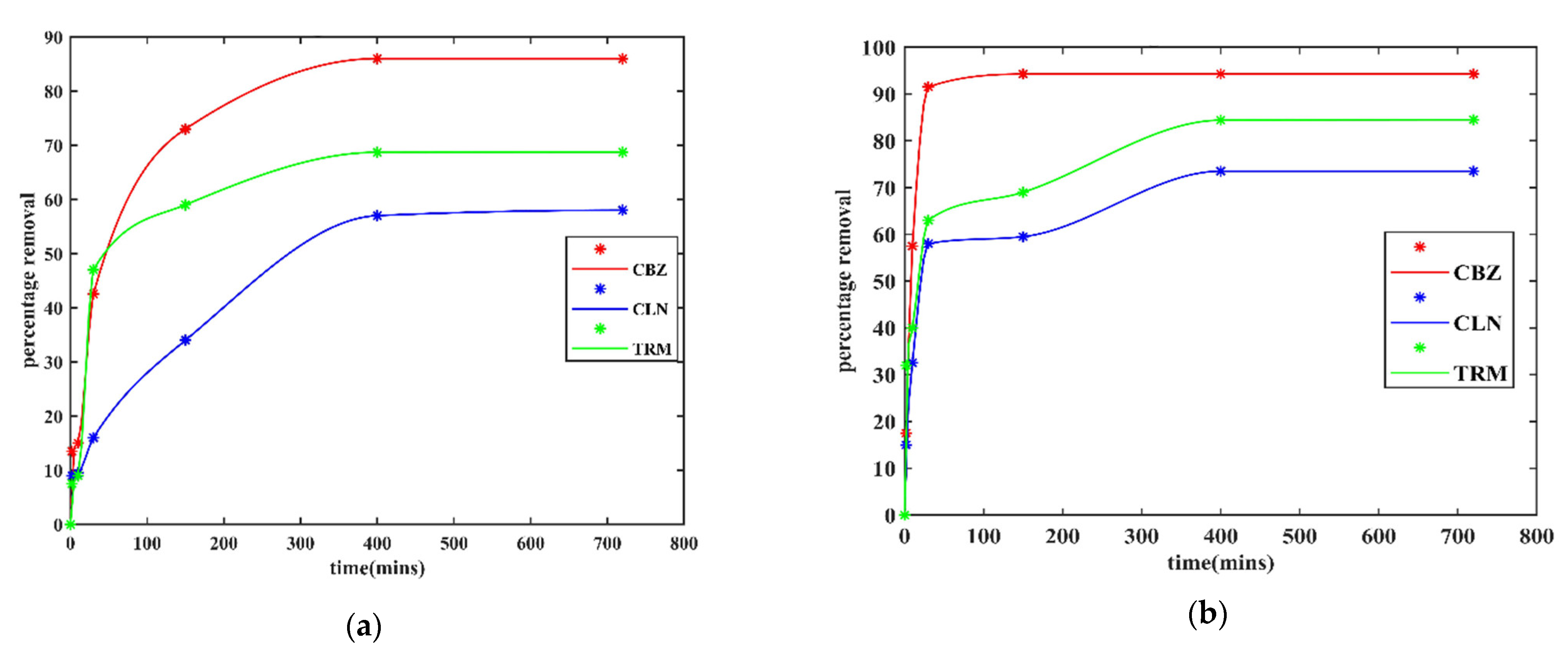
The adsorption rates were fast at the start of both experiments (effluent and Milli-Q water) and decreased as the contact time increased, as shown in
Figure 4. This could have been due to the reduction in active sites with time [
46]. At the start, all sites were available, the adsorption was fast, and it slowed down due to the intense competition for the remaining active sites. The percentage removal rates for all APIs increased with the contact time. The longer the contact time, the higher the probability of the API molecules reaching a free adsorption site. A longer contact time enables the adsorption of system-suppressed adsorbates. This was evident in the effluent water since the organic matter could have blocked some of the surface gateway sites and necessitated more time to diffuse to the inner CPAC surfaces. Hence, reaching equilibrium in the case of the effluent water took approximately 400 min for all APIs compared to 30 min for the CBZ in Milli-Q water.
3.2.3. Effect of Background Organic Matter on API Adsorption by CPAC
There was a delay in reaching equilibrium for all 3 APIs with effluent water compared to the Milli-Q water solution. This could be due to the adsorption competition and adsorption site obstruction by the organic matter [
1,
40].
Figure 4b depicts that the removal of CLN was more affected compared to the others. Its adsorption peak was reached far later than for CBZ and TRM. This was probably due to the larger molecules of CLN, which could have limited its adsorption relatively more than for CBZ and TRM. The smaller and fewer pores left due to organic matter clogging could not effectively allow for faster diffusion of the relatively larger molecules of CLN. There is also the possibility of the organic matter having masked the AC surface charge as reported by de Ridder et al. [
47]. This could have reduced the AC charge capacity, causing a reduction in the electrostatic attraction between the AC surface and the relatively more positive API molecules.
3.2.4. Effect of Initial API Concentration
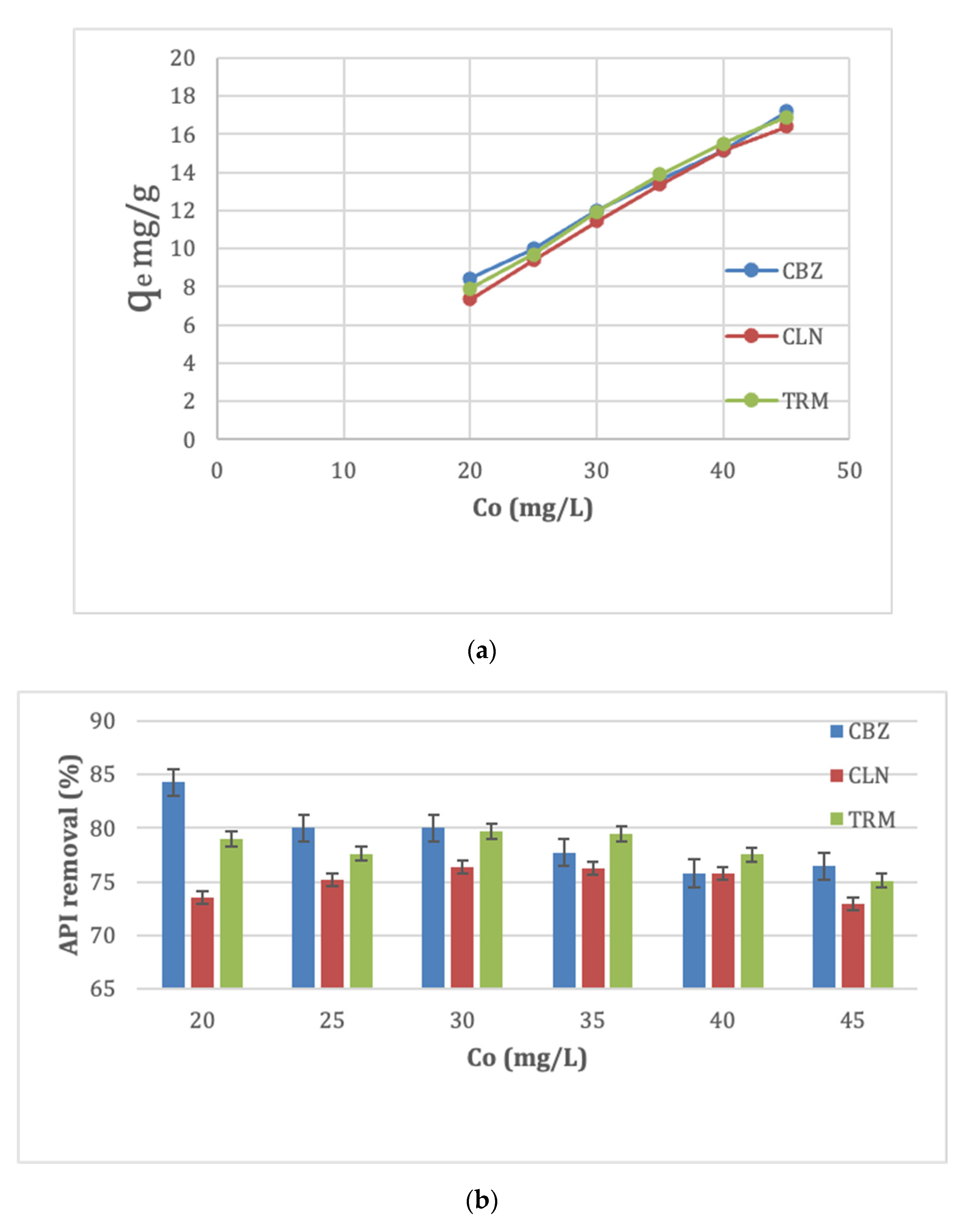
The equilibrium adsorption capacity for all APIs increased with the API concentration, as shown in
Figure 5a. This was due to the increased availability of API molecules surrounding the CPAC adsorption sites at higher initial API concentrations, which enhanced the adsorption process. The removal efficiency of the CBZ reduced with the increase in its initial concentration. This is expected of most of the adsorbates, owing to the low ratios of adsorbates to active adsorbent sites at low initial adsorbate concentrations [
45]. At low initial adsorbate concentrations, more sites are available for relatively fewer adsorbate molecules, leading to higher removal efficiencies. At higher initial adsorbate concentrations, there are residual adsorbate molecules in the solution due to the limited active sites, thereby lowering the removal efficiency [
48].
Figure 5b, however, shows a disagreement to this trend for CLN and TRM in the lower half of the respective initial API concentrations. The removal efficiency of the CLN increased from 73.50 to 76.33 as its initial concentration increased from 20 to 30 mg/L, as that of TRM almost stagnated at 79.66 from 79.00 at 20 and 30 mg/L initial concentrations, respectively. The discrepancy could have been due to the interactive forces between the API and the CPAC sites that outweighed the molecular size effects at low concentrations for CLN and TRM. CLN and TRM have 4 and 2 hydrogen bond donors, respectively, compared to CBZ’s 1. In this regard, a higher tendency to form more bonds with the anions from the CPAC sites could have led to an increase in removal efficiency with the increase in their initial concentrations. However, at concentrations >30 mg/L, the adsorption sites could have been limited, with most of them being occupied by the relatively smaller molecules of CBZ. This limited the intraparticle diffusion of CLN and TRM, in addition to the steric hindrance of the large molecules of CLN and TRM increasing at higher concentrations [
49].
3.2.5. Effect of pH Variations on Adsorption Capacity of APIs
The adsorption capacites of the APIs with different pH values are shown in
Figure 6. Generally, the adsorption capacity of the APIs decreases with a decrease in pH. The CPAC used in this study was prepared via KOH activation and its pH
zpc most probably could have been in the range of 7.0–8.0, as reported by Alongamo et al. [
50]. Reducing the pH below the point of zero charge (pH
zpc) could have rendered the CPAC surfaces more positively charged and reduced the electrostatic interaction with the APIs, whereas the increase in pH increased the electrostatic interaction between the CPAC surface and the API molecules due to the increase in the anionic tendency of the CPAC surface groups [
51]. The other possible explanation for this trend could have been the dissociation of the API molecules at pH > pH
zpc into more hydrophilic species that are negatively charged, thereby initiating electrostatic repulsions with the CPAC surfaces, which may have reduced the adsorption capacities [
52].
3.3. Adsorption Isotherm Models
In this study, two isotherm models, the Freundlich and Langmuir models, were explored to characterize the CPAC adsorption on the APIs. The values of q
e and C
e were determined. The corresponding K
L, q
m, and R
L values and the K
F and 1/n values for the Langmuir and Freundlich isotherm models, respectively, are shown in
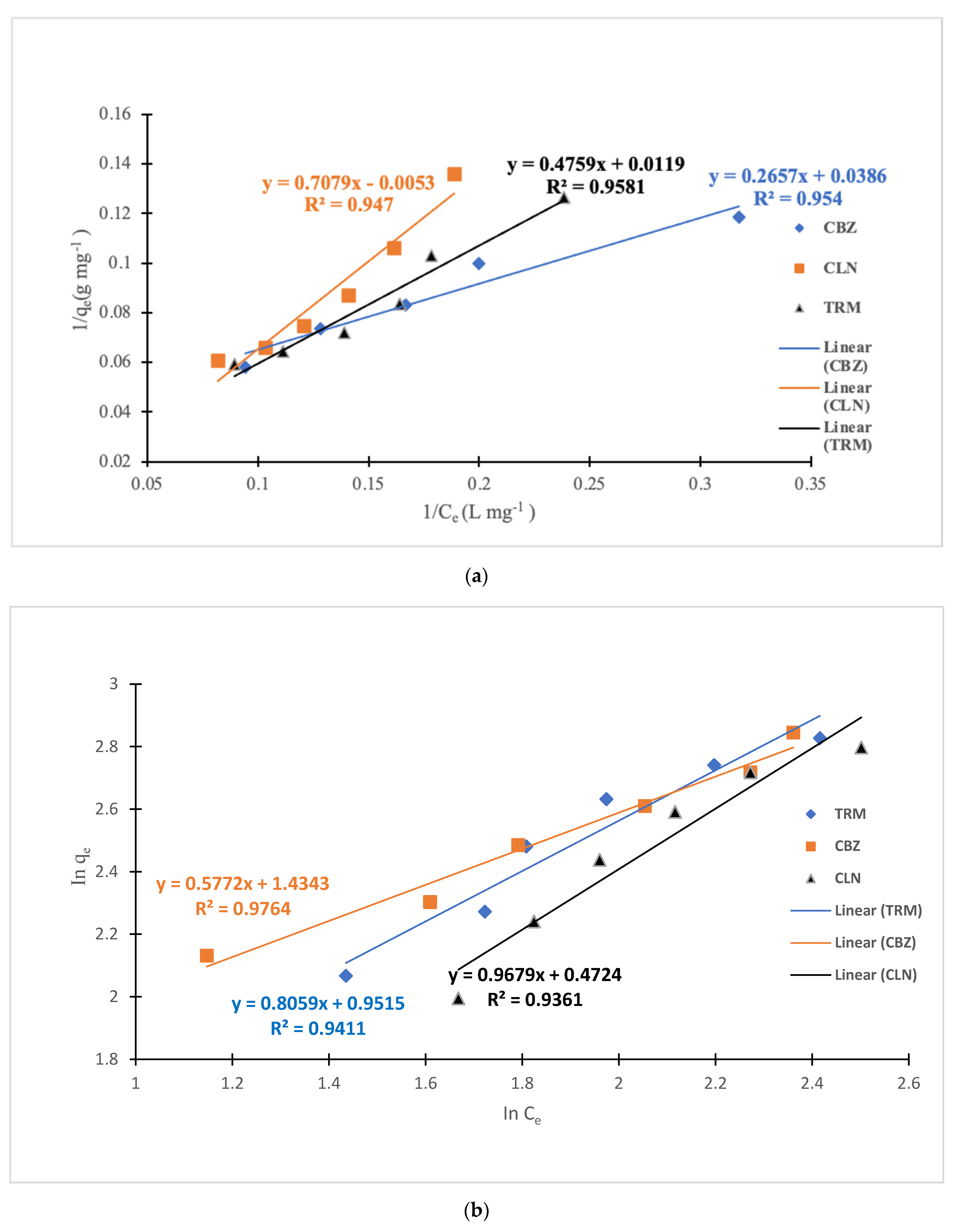
Table 5. The sorption process for CLN was better explained by the Freundlich model, whereas the CBZ and TRM adsorption processes were explained by both models.
The plots of I/q
e as a function of 1/C
e and logq
e vs. logC
e in
Figure 7 show appreciable linearity for both the CBZ and TRM based on the R
2 values for both models, as shown in
Table 5. For the CBZ, the R
2 value was 0.954 for the Langmuir model as compared to 0.976 for the Freundlich model, whereas for the TRM the R
2 values were 0.958 and 0.9411 for the Langmuir and Freundlich models, respectively. The maximum adsorption capacities (q
max) for the CBZ and TRM were, therefore, chosen based on the Freundlich and Langmuir models, respectively, due to the relatively higher R
2 values for the respective models. The adsorption rates for both APIs were also further confirmed as being favorable under Langmuir conditions by the R
L value of 0 < R
L < 1. The linearity for the I/q
e vs. 1/C
e plot for the CLN was more appreciable at R
2 of 0.947 compared to the Freundlich model at the R
2 of 0.936. However, the negative K
L value implied that the adsorption of the CPAC on the CLN could not be described by the Langmuir model. The implication, therefore, is the dominance of chemisorption in the sequestration process, with a possibility of active sites occurring in a monolayer and being uniformly distributed on the CPAC as per the Langmuir model assumptions. There could also be multilayers of the CPAC with heterogenous sites accruing to the Freundlich model. This is partly ascribable to the nature of the CPAC, with extrinsic micro-, macro-, and mesopores, as presented in our earlier work [
37].
The affinity rates for the CPAC of the 3 APIs was in the order of CBZ > TRM > CLN based on their K
L values. The order of hydrophilicity of the APIs represented by their logD
OW shown in
Table 2 was TRM > CBZ > CLN. Margot et al. (2013) studied the removal of over 70 APIs using ozone and AC, with the findings showing the most hydrophilic APIs being eliminated to a lesser extent by the AC. Therefore, in line with Margot et al.’s findings, the removal of APIs would be in the order CLN > TRM > CBZ. This, however, was not the case, as per the K
L and K
F values and the removal percentages shown in
Figure 1.
This discrepancy could have been due to the differences in molecular masses of the three APIs. As shown in
Table 2, the molecular masses of the 3 APIs in this study were in the order of CLN > TRM > CBZ. The smaller the molecular mass, the higher the diffusion rate and the probability of being accommodated in the adsorbent pores. This further confirms the dominance of chemisorption over physisorption in the sequestration of APIs by CPAC. Another functional characteristic that could have contributed to this adsorption behavior was the functional group structures in the pharmaceuticals. Both TRM and CBZ are planar, with aromatic groups throughout. CLN is bulky, non-planar, and aliphatic. Molecular sieving could have contributed to it having the lowest CPAC adsorption capacity. Aromatic compounds have been reported to be removed more efficiently from wastewater compared to those that have a relatively smaller number of aromatic rings [
1]. Overall, the adsorption of CBZ and TRM was favorable owing to the 1/n values < 1. The 1/n value for CLN was >1, implying unfavorable adsorption. This was further evident from the K
L values of CBZ and TRM of between 0 and 1, whereas that of CLN was <0. The maximum adsorption capacities for the APIs based on the Langmuir model and Freundlich model were 25.907, 84.034, and 1.487 mgg
−1 for CBZ, TRM, and CLN, respectively. This is a remarkable step towards harnessing CPAC for API sequestration. These adsorption capacities differed from those from other studies due to the differences in the process conditions and adsorbent nature, as shown in
Table 6. For example, Wang et al. [
42] attained a remarkable adsorption capacity of activated carbon fiber for CLN of 70.90 mgg
−1 through electrolysis. The greater CBZ adsorption capacity (25.907 mgg
−1) in this study compared to that reported by Sekulic et al. [
53] at 17.69 mgg
−1 was probably due to the lower adsorption time.