Novel Vertical Flow Wetland Filtration Combined with Co-Zeotype Material Based Catalytic Ozonation Process for the Treatment of Municipal Wastewater
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Methods
2.2. Synthesis of Catalyst
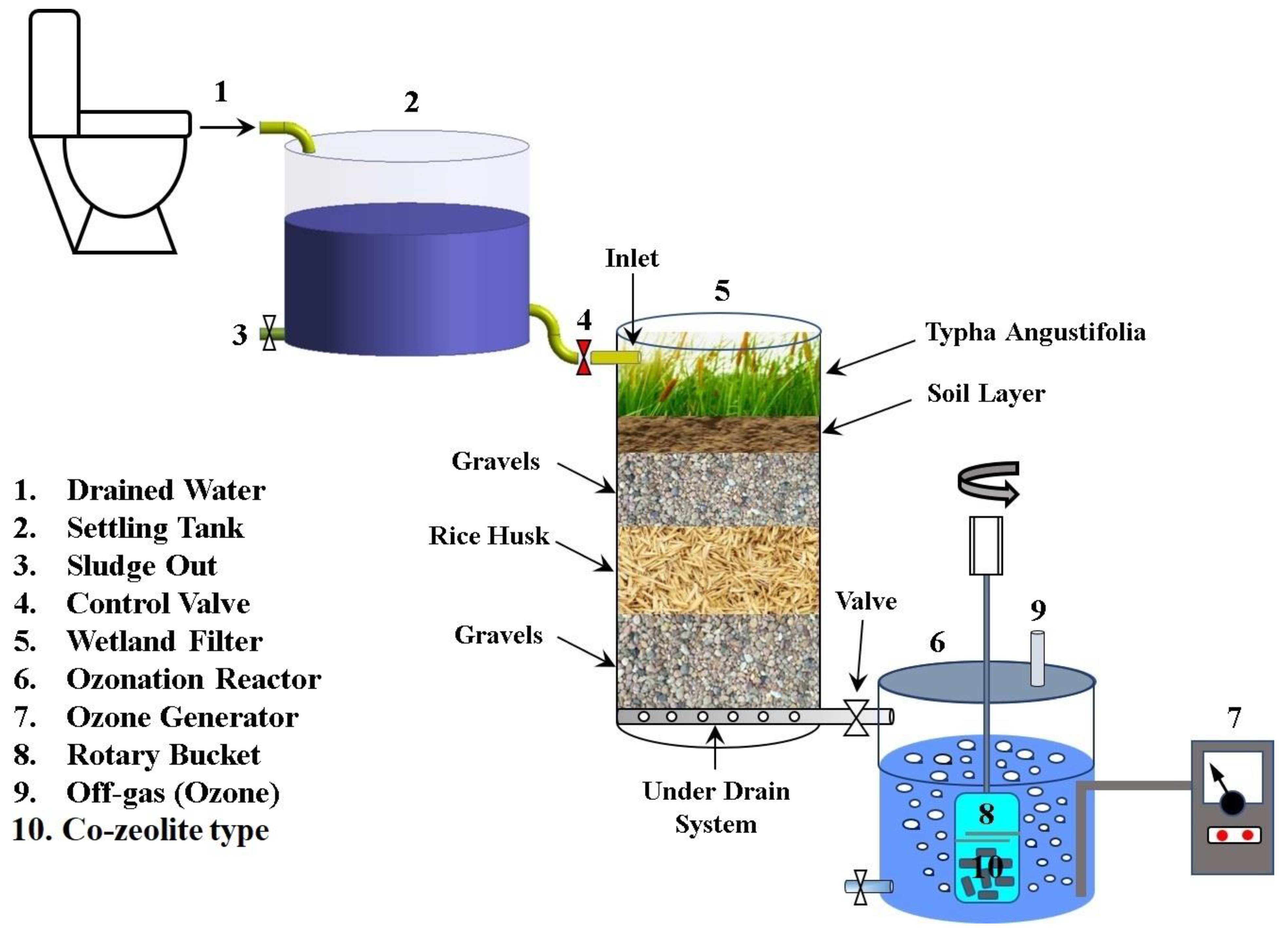
2.3. Experimental Setup
2.4. Experimental Procedure
2.5. Characterization of Co-Zeotype Catalyst
2.6. Ozone Dose Analysis
3. Results and Discussion
3.1. Wastewater Characterization
3.2. Characterization of Catalyst
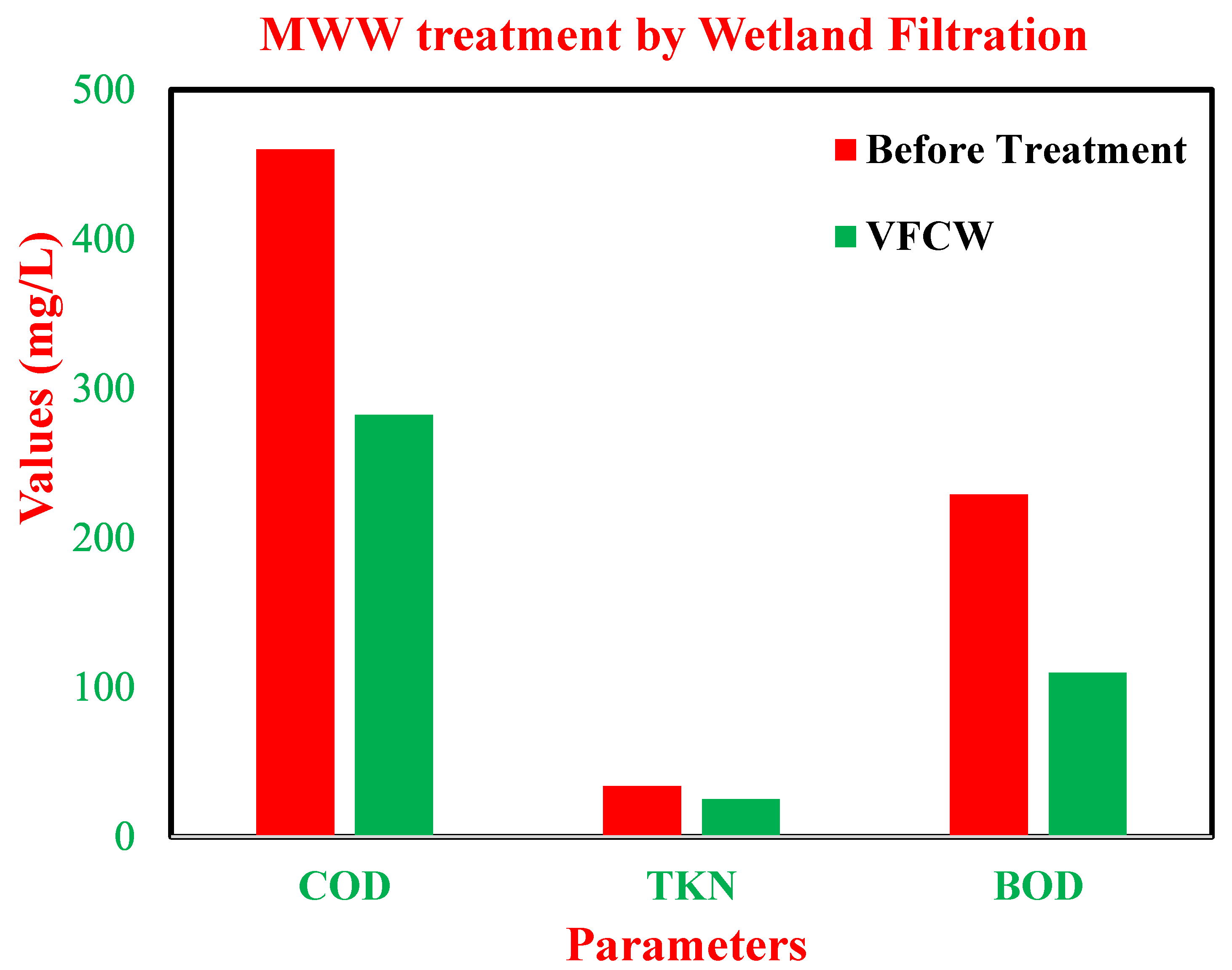
3.3. Wastewater Treatment by VFCW
3.4. Contact Time Optimization for Simple Ozonation
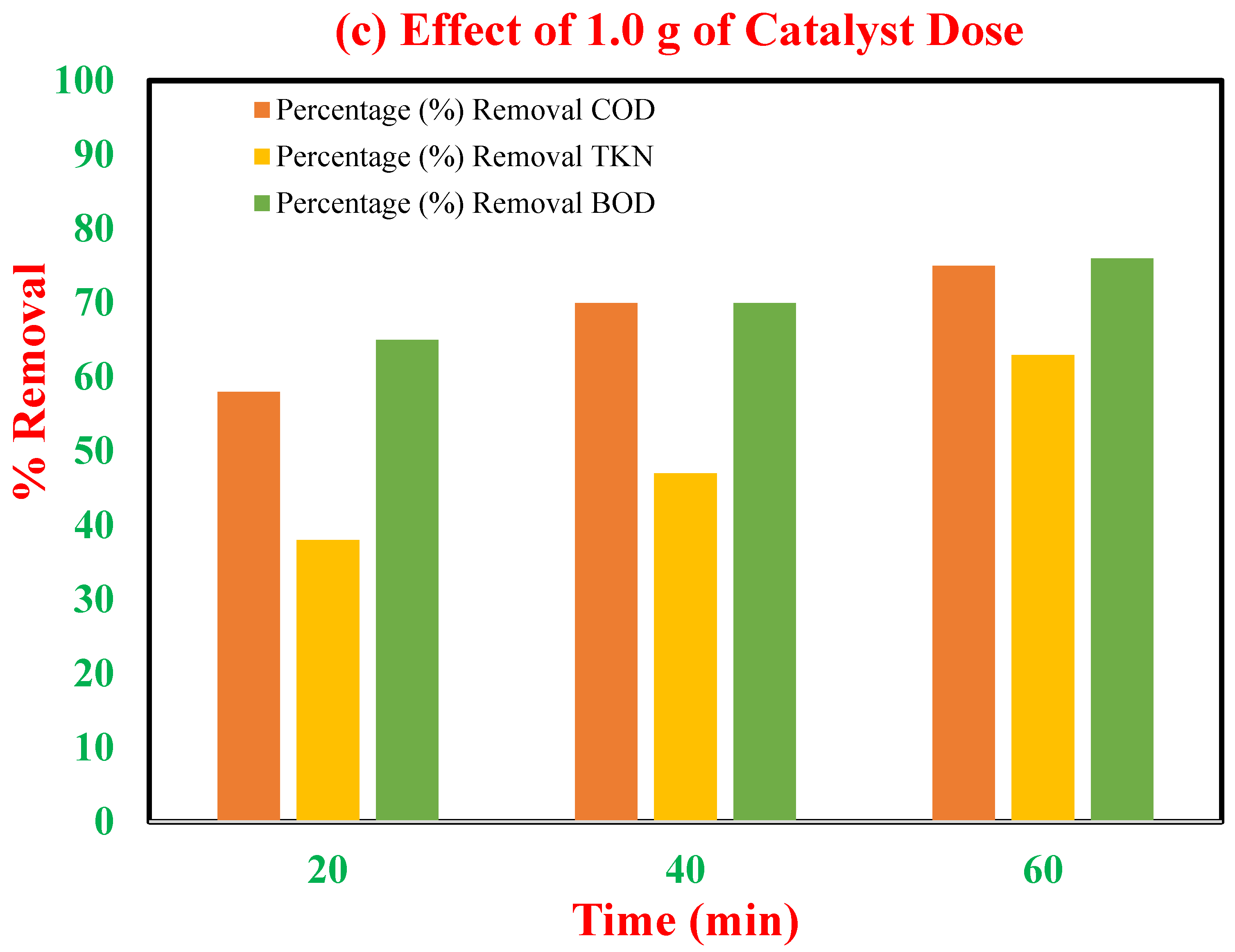
3.5. Catalyst Dose Optimization for Catalytic Ozonation
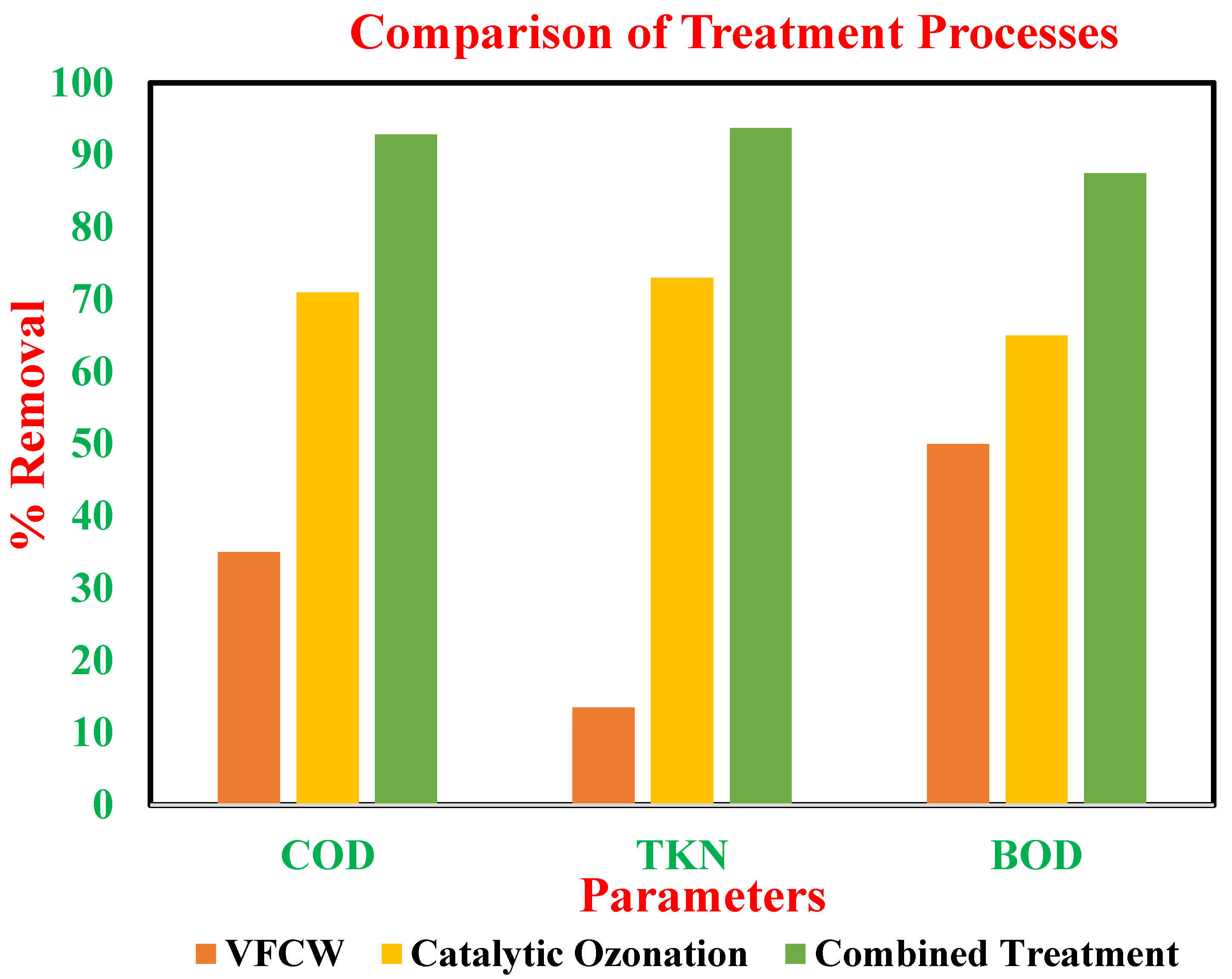
3.6. Comparison of Treatment Processes
3.7. Removal of Heavy Metals
3.8. Proposed Process Mechanism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kivaisi, A.K. The potential for constructed wetlands for wastewater treatment and reuse in developing countries: A review. Ecol. Eng. 2001, 16, 545–560. [Google Scholar] [CrossRef]
- Jia, X.; O’Connor, D.; Hou, D.; Jin, Y.; Li, G.; Zheng, C.; Ok, Y.S.; Tsang, D.C.; Luo, J. Groundwater depletion and contamination: Spatial distribution of groundwater resources sustainability in China. Sci. Total Environ. 2019, 672, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Basharat, M.; Rizvi, S.A. Groundwater extraction and waste water disposal regulation. Is Lahore Aquifer at stake with as usual approach. Pakistan Engineering Congress, World Water Day. 2011, pp. 112–134. Available online: https://www.pecongress.org.pk/images/upload/books/Ground%20Water%20Extraction%20and%20Waste%20Water%20Disposal%20Regulation%20.pdf (accessed on 15 October 2022).
- Murtaza, G.; Zia, M.H. Wastewater production, treatment and use in Pakistan. Second Regional Workshop of the Project ‘Safe Use of Wastewater in Agriculture. 2012, pp. 16–18. Available online: https://www.ais.unwater.org/ais/pluginfile.php/232/mod_page/content/128/pakistan_murtaza_finalcountryreport2012.pdf (accessed on 15 October 2022).
- Göbel, A.; McArdell, C.S.; Joss, A.; Siegrist, H.; Giger, W. Fate of sulfonamides, macrolides, and trimethoprim in different wastewater treatment technologies. Sci. Total Environ. 2007, 372, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Cesaro, A.; Naddeo, V.; Belgiorno, V. Wastewater treatment by combination of advanced oxidation processes and conventional biological systems. J. Bioremediation Biodegrad. 2013, 4, 1–8. [Google Scholar]
- Garrido-Cardenas, J.A.; Esteban-García, B.; Agüera, A.; Sánchez-Pérez, J.A.; Manzano-Agugliaro, F. Wastewater treatment by advanced oxidation process and their worldwide research trends. Int. J. Environ. Res. Public Health 2020, 17, 170. [Google Scholar] [CrossRef] [Green Version]
- Chiu, J.M.; Degger, N.; Leung, J.Y.; Po, B.H.; Zheng, G.J.; Richardson, B.J.; Lau, T.C.; Wu, R.S. A novel approach for estimating the removal efficiencies of endocrine disrupting chemicals and heavy metals in wastewater treatment processes. Mar. Pollut. Bull. 2016, 112, 53–57. [Google Scholar] [CrossRef]
- Cowardin, L.M.; Carter, V.; Golet, F.C.; Laroe, E.T. Classification of wetlands and deepwater habitats of the United States. Water Encycl. 2005, 3, 496–498. [Google Scholar]
- Ghatak, H.R. Advanced oxidation processes for the treatment of biorecalcitrant organics in wastewater. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1167–1219. [Google Scholar] [CrossRef]
- Mishra, N.S.; Reddy, R.; Kuila, A.; Rani, A.; Mukherjee, P.; Nawaz, A.; Pichiah, S. A review on advanced oxidation processes for effective water treatment. Curr. World Environ. 2017, 12, 470. [Google Scholar] [CrossRef]
- Rizvi, O.S.; Ikhlaq, A.; Ashar, U.U.; Qazi, U.Y.; Akram, A.; Kalim, I.; Alazmi, A.; Shamsah, S.M.I.; Al-Sodani, K.A.A.; Javaid, R. Application of poly aluminum chloride and alum as catalyst in catalytic ozonation process after coagulation for the treatment of textile wastewater. J. Environ. Manag. 2022, 323, 115977. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Wallace, S. Treatment Wetlands; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Kansiime, F.; Maimuna, N. Wastewater Treatment by a Natural Wetland: The Nakivubo Swamp, Uganda; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Brix, H. Do macrophytes play a role in constructed treatment wetlands? Water Sci. Technol. 1997, 35, 11–17. [Google Scholar] [CrossRef]
- Rosal, R.; Gonzalo, M.S.; Rodríguez, A.; García-Calvo, E. Catalytic ozonation of fenofibric acid over alumina-supported manganese oxide. J. Hazard. Mater. 2010, 183, 271–278. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Kasprzyk-Hordern, B. Catalytic ozonation of chlorinated VOCs on ZSM-5 zeolites and alumina: Formation of chlorides. Appl. Catal. B Environ. 2017, 200, 274–282. [Google Scholar] [CrossRef] [Green Version]
- Ikhlaq, A.; Javed, F.; Akram, A.; Rehman, A.; Qi, F.; Javed, M.; Mehdi, M.J.; Waheed, F.; Naveed, S.; Aziz, H.A. Synergic catalytic ozonation and electroflocculation process for the treatment of veterinary pharmaceutical wastewater in a hybrid reactor. J. Water Process Eng. 2020, 38, 101597. [Google Scholar] [CrossRef]
- Legube, B.; Leitner, N.K.V. Catalytic ozonation: A promising advanced oxidation technology for water treatment. Catal. Today 1999, 53, 61–72. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Brown, D.R.; Kasprzyk-Hordern, B. Mechanisms of catalytic ozonation on alumina and zeolites in water: Formation of hydroxyl radicals. Appl. Catal. B Environ. 2012, 123, 94–106. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Waheed, S.; Joya, K.S.; Kazmi, M. Catalytic ozonation of paracetamol on zeolite A: Non-radical mechanism. Catal. Commun. 2018, 112, 15–20. [Google Scholar] [CrossRef]
- Guo, Y.; Yang, L.; Cheng, X.; Wang, X. The application and reaction mechanism of catalytic ozonation in water treatment. J. Environ. Anal. Toxicol. 2012, 2, 2161-0525. [Google Scholar] [CrossRef] [Green Version]
- Ikhlaq, A.; Fatima, R.; Qazi, U.Y.; Javaid, R.; Akram, A.; Shamsah, S.I.; Qi, F. Combined iron-loaded zeolites and ozone-based process for the purification of drinking water in a novel hybrid reactor: Removal of faecal coliforms and arsenic. Catalysts 2021, 11, 373. [Google Scholar] [CrossRef]
- Zhang, B.-L.; Deng, L.-F.; Liu, B.; Luo, C.-Y.; Liebau, M.; Zhang, S.-G.; Gläser, R. Synergistic effect of cobalt and niobium in Co3-Nb-Ox on performance of selective catalytic reduction of NO with NH3. Rare Metals 2022, 41, 166–178. [Google Scholar] [CrossRef]
- Jin, Z.; Li, Y.; Hao, X. Ni, Co-based selenide anchored g-C3N4 for boosting photocatalytic hydrogen evolution. Acta Phys.-Chim. Sin. 2021, 37, 1912033. [Google Scholar]
- Hu, H.; Cai, S.; Li, H.; Huang, L.; Shi, L.; Zhang, D. Mechanistic aspects of deNO x processing over TiO2 supported Co–Mn oxide catalysts: Structure–activity relationships and in situ DRIFTs analysis. ACS Catal. 2015, 5, 6069–6077. [Google Scholar] [CrossRef]
- Xie, X.; Li, Y.; Liu, Z.-Q.; Haruta, M.; Shen, W. Low-temperature oxidation of CO catalysed by Co3O4 nanorods. Nature 2009, 458, 746–749. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Peng, Q.; Li, Y. Selective synthesis of Co3O4 nanocrystal with different shape and crystal plane effect on catalytic property for methane combustion. J. Am. Chem. Soc. 2008, 130, 16136–16137. [Google Scholar] [CrossRef] [PubMed]
- Arden, S.; Ma, X. Constructed wetlands for greywater recycle and reuse: A review. Sci. Total Environ. 2018, 630, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Ikhlaq, A.; Javed, F.; Akram, A.; Qazi, U.Y.; Masood, Z.; Ahmed, T.; Arshad, Z.; Khalid, S.; Qi, F. Treatment of leachate through constructed wetlands using Typha angustifolia in combination with catalytic ozonation on Fe-zeolite A. Int. J. Phytoremediation 2021, 23, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.; Hench, K.; Garbutt, K.; Sexstone, A.; Bissonnette, G.; Skousen, J. Treatment of domestic wastewater by three plant species in constructed wetlands. Water Air Soil Pollut. 2001, 128, 283–295. [Google Scholar] [CrossRef]
- Rice, A.; Baird, E.; Eaton, R. Standard Methods for Examination of Water and Wastewater; American Water Works Association, and Water Env. Federation ISBN; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- Adams, V.D. Water and Wastewater Examination Manual; Routledge: New York, NY, USA, 2017; ISBN 978-0-203-73413-1. [Google Scholar]
- Sajjad, S.; Ikhlaq, A.; Javed, F.; Ahmad, S.W.; Qi, F. A study on the influence of pH changes during catalytic ozonation process on alumina, zeolites and activated carbons for the decolorization of Reactive Red-241. Water Sci. Technol. 2021, 83, 727–738. [Google Scholar] [CrossRef]
- Amir, I.; Mehwish, A.; Farhan, J.; Hafsa, G.; Munir, H.M.S.; Kashif, I. Catalytic ozonation for the treatment of municipal wastewater by iron loaded zeolite A. Desalination Water Treat. 2019, 152, 108–115. [Google Scholar]
- Raashid, M.; Kazmi, M.; Ikhlaq, A.; Iqbal, T.; Sulaiman, M.; Shakeel, A. Degradation of Aqueous CONFIDOR® Pesticide by Simultaneous TiO2 Photocatalysis and Fe-Zeolite Catalytic Ozonation. Water 2021, 13, 3327. [Google Scholar] [CrossRef]
- Cheng, Q.; Li, H.; Xu, Y.; Chen, S.; Liao, Y.; Deng, F.; Li, J. Study on the adsorption of nitrogen and phosphorus from biogas slurry by NaCl-modified zeolite. PLoS ONE 2017, 12, 176109. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Fletcher, T.D.; Sun, G. Nitrogen removal in constructed wetland systems. Eng. Life Sci. 2009, 9, 11–22. [Google Scholar] [CrossRef]
- Truu, M.; Juhanson, J.; Truu, J. Microbial biomass, activity and community composition in constructed wetlands. Sci. Total Environ. 2009, 407, 3958–3971. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-J.; Fang, G.-C.; Wang, C.-C. A nanometer-ZnO catalyst to enhance the ozonation of 2, 4, 6-trichlorophenol in water. Colloids Surf. A Physicochem. Eng. Asp. 2005, 260, 45–51. [Google Scholar] [CrossRef]
- Marion, G.M.; Catling, D.C.; Kargel, J.S. Modeling aqueous ferrous iron chemistry at low temperatures with application to Mars. Geochim. et Cosmochim. Acta 2003, 67, 4251–4266. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Qazi, U.Y.; Akram, A.; Rizvi, O.S.; Sultan, A.; Javaid, R.; Al-Sodani, K.A.A.; Ibn Shamsah, S.M. Potable Water Treatment in a Batch Reactor Benefited by Combined Filtration and Catalytic Ozonation. Water 2022, 14, 2357. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.E.; Lee, H.W.; Jeon, J.-K.; Song, J.; Jung, S.-C.; Tsang, Y.F.; Park, Y.-K. Catalytic ozonation of toluene using Mn–M bimetallic HZSM-5 (M: Fe, Cu, Ru, Ag) catalysts at room temperature. J. Hazard. Mater. 2020, 397, 122577. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.-H.; Ma, Y.-L.; Lin, F.; Ge, S.-Y.; Zhu, L. Refractory organic compounds in coal chemical wastewater treatment by catalytic ozonation using Mn-Cu-Ce/Al2O3. Environ. Sci. Pollut. Res. 2021, 28, 41504–41515. [Google Scholar] [CrossRef]
- Ikhlaqa, A.; Aslama, T.; Zafara, A.M.; Javedb, F.; Munirc, H.M.S. Combined ozonation and adsorption system for the removal of heavy metals from municipal wastewater: Effect of COD removal. Desalination Water Treat. 2019, 159, 304–309. [Google Scholar] [CrossRef] [Green Version]
- Qazi, U.Y.; Javaid, R.; Ikhlaq, A.; Al-Sodani, K.A.A.; Rizvi, O.S.; Alazmi, A.; Asiri, A.M.; Ibn Shamsah, S.M. Synergistically Improved Catalytic Ozonation Process Using Iron-Loaded Activated Carbons for the Removal of Arsenic in Drinking Water. Water 2022, 14, 2406. [Google Scholar] [CrossRef]
- Khosravi, M.; Mehrdadi, N.; Baghdadi, M. Optimization of ozonation/adsorption combined method for the removal of toxic metals and COD using sewage sludge based carbon/TiO2/ZnO nanocomposite. Mater. Res. Express 2019, 6, 125531. [Google Scholar] [CrossRef]
- Pillai, P.; Kakadiya, N.; Timaniya, Z.; Dharaskar, S.; Sillanpaa, M. Removal of arsenic using iron oxide amended with rice husk nanoparticles from aqueous solution. Mater. Today Proc. 2020, 28, 830–835. [Google Scholar] [CrossRef]
- Javaid, R.; Qazi, U.Y. Catalytic Oxidation Process for the Degradation of Synthetic Dyes: An Overview. Int. J. Environ. Res. Public Health 2019, 16, 2066. [Google Scholar] [CrossRef] [PubMed]





| Parameters | Units | Saggiyan Drain | NEQS |
|---|---|---|---|
| Temperature | (°C) | 18 ± 2 | 40 |
| EC | (dS/m) | 90 ± 15 | 0 |
| pH | - | 7 ± 0.2 | 6–9 |
| BOD5 | (mg/L) | 177 ± 18 | 80 |
| COD | 450 ± 25 | 150 | |
| TKN | 95.6 ± 19 | 40 | |
| TDS | 517 ± 28 | 3500 | |
| TSS | 175 ± 13 | 150 | |
| Fe | 3.1 ± 0.02 | 2.0 | |
| Cd | 2 ± 0.03 | 0.1 | |
| Zn | 0.3 ± 0.01 | 5.0 | |
| Cr | 0.45 ± 0.03 | 1.0 | |
| Ni | 0.69 ± 0.02 | 1.0 | |
| Cu | 0.17 ± 0.02 | 1.0 |
| Material | Surface Area BET (m2/g) | Pore Size (nm) | Pore Volume (cc/g) | Cobalt Content [%] EDX | Point of Zero Charge (pHpzc) |
|---|---|---|---|---|---|
| Co-Zeotype 3A | 37.1 | 3.21 | 12.45 | 7.03 | 6.3 ± 0.2 |
| Metals (mg/L) | NEQS | After Treatment | % Removal |
|---|---|---|---|
| Fe | 2.0 | 0.01 | 90 |
| Zn | 5.0 | BDL * | - |
| Cd | 0.1 | 1 | 50 |
| Cr | 1.0 | 0.05 | 88 |
| Ni | 1.0 | BDL | - |
| Cu | 1.0 | 0.09 | 47 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qazi, U.Y.; Ikhlaq, A.; Akram, A.; Rizvi, O.S.; Javed, F.; Ul-Hasan, I.; Alazmi, A.; Ibn Shamsah, S.M.; Javaid, R. Novel Vertical Flow Wetland Filtration Combined with Co-Zeotype Material Based Catalytic Ozonation Process for the Treatment of Municipal Wastewater. Water 2022, 14, 3361. https://doi.org/10.3390/w14213361
Qazi UY, Ikhlaq A, Akram A, Rizvi OS, Javed F, Ul-Hasan I, Alazmi A, Ibn Shamsah SM, Javaid R. Novel Vertical Flow Wetland Filtration Combined with Co-Zeotype Material Based Catalytic Ozonation Process for the Treatment of Municipal Wastewater. Water. 2022; 14(21):3361. https://doi.org/10.3390/w14213361
Chicago/Turabian StyleQazi, Umair Yaqub, Amir Ikhlaq, Asia Akram, Osama Shaheen Rizvi, Farhan Javed, Iftikhar Ul-Hasan, Amira Alazmi, Sami M. Ibn Shamsah, and Rahat Javaid. 2022. "Novel Vertical Flow Wetland Filtration Combined with Co-Zeotype Material Based Catalytic Ozonation Process for the Treatment of Municipal Wastewater" Water 14, no. 21: 3361. https://doi.org/10.3390/w14213361









