Sugarcane Bagasse as Carbon Source and Filler to Enhance the Treatment of Low C/N Wastewater by Aerobic Denitrification Flora
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Activated Sludge
2.1.2. Culture Media
2.1.3. Sugarcane Bagasse
2.1.4. Reactors
2.2. Methods
2.2.1. AD Flora Culture
2.2.2. Carbon-Release Performance Examination of Sugarcane Bagasse
- (1)
- Shake-flask experiment on soaking carbon release
- (2)
- Shake-flask experiment of batch water replacement
2.2.3. Enhanced Treatment of Low C/N Wastewater with Sugarcane Bagasse by AD Flora
- (1)
- Decarbonization of AD flora
- (2)
- Enhancement of denitrification by AD flora in sugarcane bagasse
2.2.4. Small-Scale Treatment of Low C/N Wastewater by Bagasse-Loaded SBBR
2.3. Parameter Analysis
2.4. Data Analysis
3. Results
3.1. Culture of AD Flora
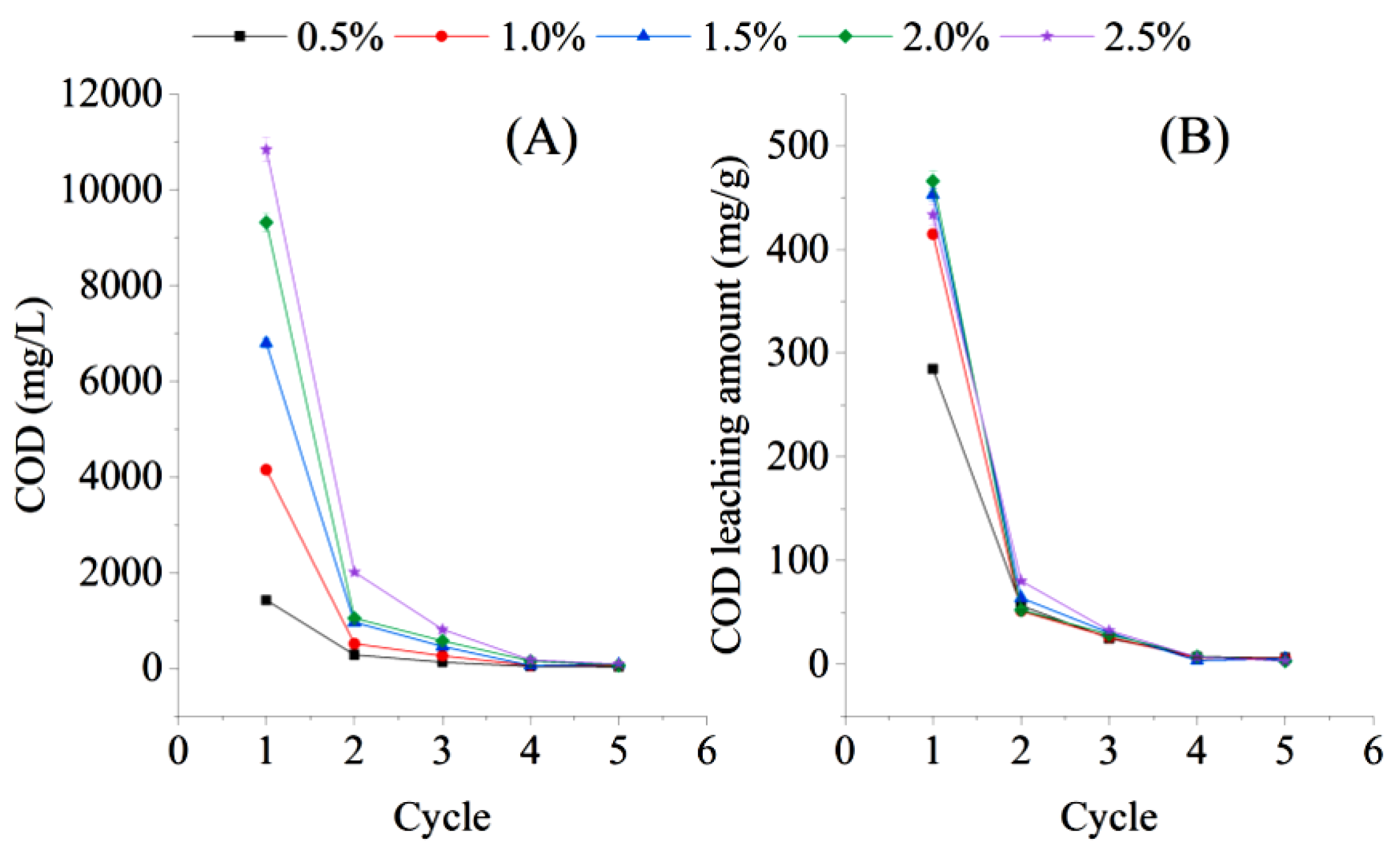
3.2. Carbon Source Release from Sugarcane Bagasse
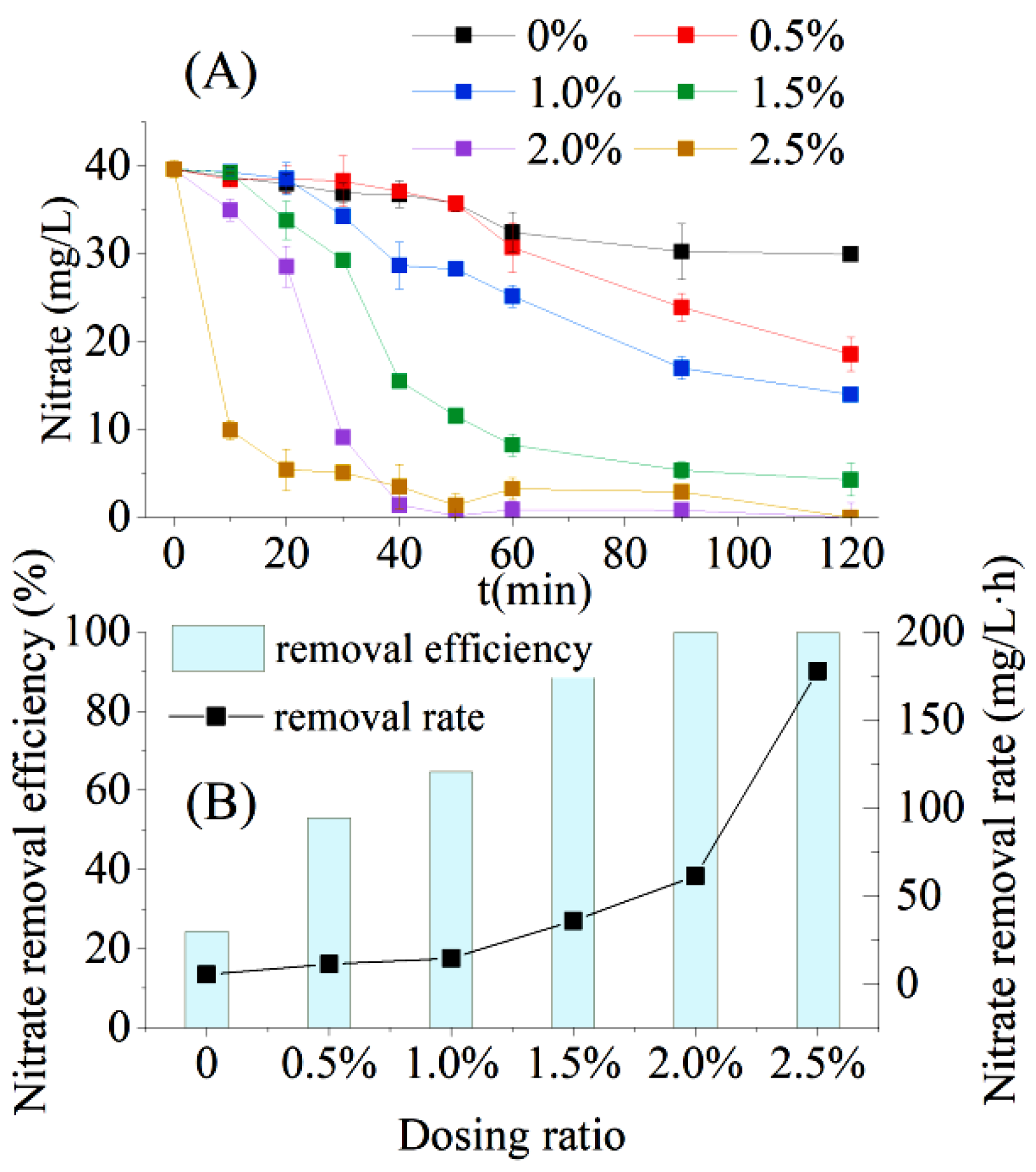
3.3. Enhanced Treatment of Low C/N Wastewater with Sugarcane Bagasse by AD Flora
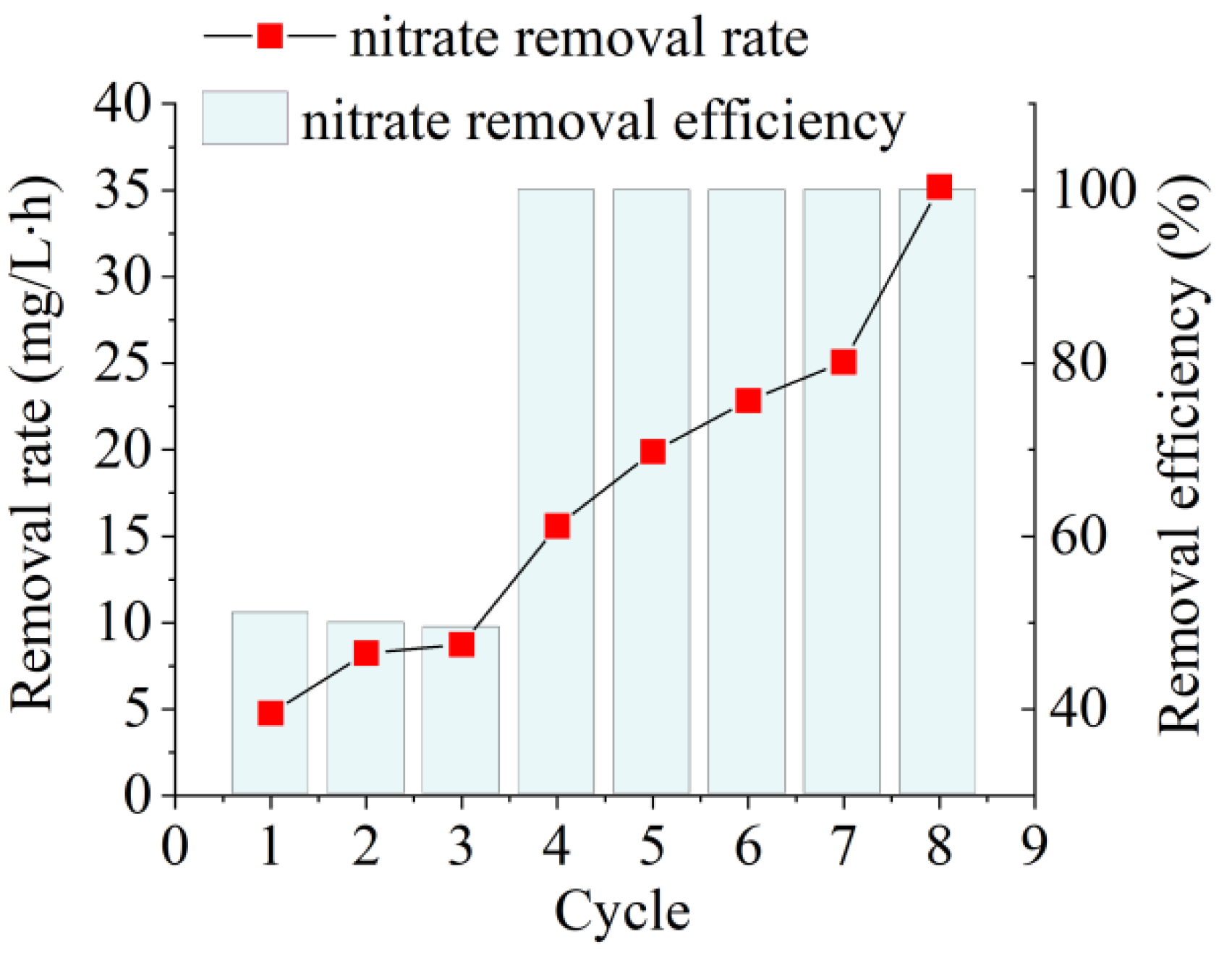
3.4. Treatment of Low C/N Wastewater by AD Flora in Bagasse-Loaded SBBR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, J.; Feng, L.; Pi, S.; Cui, D.; Ma, F.; Zhao, H.P.; Li, A. A critical review of aerobic denitrification: Insights into the intracellular electron transfer. Sci. Total Environ. 2022, 731, 139080. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.L.; Ali, A.; Ren, Y.; Su, J.F.; Wang, Z. A mechanistic review on aerobic denitrification for nitrogen removal in water treatment. Sci. Total Environ. 2022, 847, 157452. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Guo, W.; Liang, D.; Zhang, J.; Li, J.; Li, P.; Wu, Y.; Bian, X.; Ding, F. Rapid start-up and advanced nutrient removal of simultaneous nitrification, endogenous denitrification and phosphorus removal aerobic granular sequence batch reactor for treating low C/N domestic wastewater. Environ. Res. 2022, 212, 113464. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Quan, J.; Huang, K.; Zhao, J.; Xing, G.; Wu, P.; Chen, Y.; Ding, X.; Hu, Y. Effects of C/N ratio and dissolved oxygen on aerobic denitrification process: A mathematical modeling study. Chemosphere 2021, 272, 129521. [Google Scholar] [CrossRef]
- Wang, X.J.; Wang, D.S.; Li, J.; Du, S.D.; Zhou, T.; Wu, G.D. Separation identification and the characteristics research of simultaneous removal of nitrogen and carbon about Marine heterotrophic nitrification and aerobic denitrification strain y6. China Environ. Sci. 2017, 37, 686–695. [Google Scholar] [CrossRef]
- Wiboonluk, P.; Cholticha, P.; Sorawit, P. Optimization and evaluation of a bottom substrate denitrification tank for nitrate removal from a recirculating aquaculture system. J. Environ. Sci. 2013, 25, 1557–1564. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, Y.; Fan, Y.; Liu, Y.; Yu, M.; He, C.; Wu, J. Bioaugmentation of low C/N ratio wastewater: Effect of acetate and propionate on nutrient removal, substrate transformation, and microbial community behavior. Bioresour. Technol. 2019, 306, 122465. [Google Scholar] [CrossRef]
- Wang, J.; Chu, L. Biological nitrate removal from water and wastewater by solid-phase denitrification process. Biotechnol. Adv. 2016, 34, 1103–1112. [Google Scholar] [CrossRef]
- Fu, X.; Hou, R.; Yang, P.; Qian, S.; Feng, Z.; Chen, Z.; Wang, F.; Yuan, R.; Chen, H.; Zhou, B. Application of external carbon source in heterotrophic denitrification of domestic sewage: A review. Sci. Total Environ. 2022, 817, 153061. [Google Scholar] [CrossRef]
- Shi, Y.; Hu, Y.; Liang, D.; Wang, G.; Xie, J.; Zhu, X. Enhanced denitrification of sewage via bio-microcapsules embedding heterotrophic nitrification-aerobic denitrification bacteria Acinetobacter pittii SY9 and corn cob. Bioresour. Technol. 2022, 358, 127260. [Google Scholar] [CrossRef]
- Feng, L.; Pi, S.; Zhu, W.; Wang, X.; Xu, X. Nitrification and aerobic denitrification in solid phase denitrification systems with various biodegradable carriers for ammonium-contaminated water purification. J. Chem. Technol. Biotechnol. 2019, 94, 3569–3577. [Google Scholar] [CrossRef]
- Bilba, K.; Arsene, M.A. Silane treatment of bagasse fiber for reinforcement of cementitious composites. Compos. Part A Appl. Sci. Manuf. 2008, 39, 1488–1495. [Google Scholar] [CrossRef]
- Bao, G.Y.; Lan, Y.H. Sugarcane biorefinery—New development on utilization of bagasse. Sugarcane Canesugar. 2011, 5, 59–65. [Google Scholar] [CrossRef]
- Ajala, E.O.; Ighalo, J.O.; Ajala, M.A.; Adeniyi, A.G.; Ayanshola, A.M. Sugarcane bagasse: A biomass sufficiently applied for improving global energy, environment and economic sustainability. Bioresour. Bioprocess. 2021, 8, 87. [Google Scholar] [CrossRef]
- Hu, B.; Wang, T.; Ye, J.; Zhao, J.; Yang, L.; Wu, P.; Duan, J.; Ye, G. Effects of carbon sources and operation modes on the performances of aerobic denitrification process and its microbial community shifts. J. Environ. Manag. 2019, 239, 299–305. [Google Scholar] [CrossRef]
- Yang, X.R. Determination of COD by Doal-wavelength Equivalent Absorbance Spectrophotometry. J. Wuhan Univ. Technol. 2010, 32, 177–180. [Google Scholar] [CrossRef]
- Wang, J.; Rong, H.; Zhang, C. Evaluation of the impact of dissolved oxygen concentration on biofilm microbial community in sequencing batch biofilm reactor. J. Biosci. Bioeng. 2018, 125, 532–542. [Google Scholar] [CrossRef]
- Di, J.Z.; Li, T.D.; Zhao, W. Release law of sugarcane slag carbon source and orthogonal testing of sulfate-reducing bacteria. J. Agro-Environ. Sci. 2019, 38, 1151–1157. [Google Scholar] [CrossRef]
- Cao, X.; Li, Y.; Jiang, X.; Zhou, P.; Zhang, J.; Zheng, Z. Treatment of artificial secondary effluent for effective nitrogen removal using a combination of corncob carbon source and bamboo charcoal filter. Int. Biodeterior. Biodegrad. 2016, 115, 164–170. [Google Scholar] [CrossRef] [Green Version]
- Pan, Z.; Zhou, J.; Lin, Z.; Wang, Y.; Zhao, P.; Zhou, J.; Liu, S.; He, X. Effects of COD/TN ratio on nitrogen removal efficiency, microbial community for high saline wastewater treatment based on heterotrophic nitrification-aerobic denitrification process. Bioresour. Technol. 2020, 301, 122726. [Google Scholar] [CrossRef]
- Wang, T.; Chen, M.; Liang, X.; Chen, F.; He, T.; Li, Z. Corrigendum: The alkali-tolerant bacterium of Bacillus thuringiensis EM-A1 can effectively perform heterotrophic nitrification and aerobic denitrification. Front. Environ. Sci. 2022, 19, 818316. [Google Scholar] [CrossRef]
- Su, J.F.; Ma, M.; Ma, F.; Lu, J.S. Kinetic analysis of heterotrophic nitrification -aerobic denitrification by an oligotrophic Acinetobacter sp. SYF26. Environ. Eng. Sci. 2017, 34, 844–851. [Google Scholar] [CrossRef]
- Prangya, R.R.; Puspendu, B.; Rajesh, R.D. Simultaneous removal of nitrogen and phosphorous from domestic wastewater using Bacillus cereus GS-5 strain exhibiting heterotrophic nitrification, aerobic denitrification and denitrifying phosphorous removal. Bioresour. Technol. 2017, 244, 484–495. [Google Scholar] [CrossRef]
- Zeng, M.; Zeng, Y.; Zhang, B.; Cheng, Y.; Long, B.; Wu, J.; Ren, S.; Liu, Y. Coupling of endogenous/exogenous nitrification and denitrification in an aerobic granular sequencing batch reactor. Environ. Technol. 2022, 20, 68380. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Ma, B.; Li, X.; Zhang, Q.; Peng, Y. Advanced nitrogen removal without addition of external carbon source in an anaerobic/aerobic/anoxic sequencing batch reactor. Bioprocess Biosyst. Eng. 2019, 42, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Xiao, L.Y.; Hai, L.S.; Jia, J.W.; Jia, Y.X. Alkali-treated cellulose carrier enhancing denitrification in membrane bioreactor. Int. Biodeter. Biodegr. 2019, 145, 104813. [Google Scholar] [CrossRef]
- Cui, Y.; Zhao, B.; Xie, F.; Zhang, X.; Zhou, A.; Wang, S.; Yue, X. Study on the preparation and feasibility of a novel adding-type biological slow-release carbon source. J. Environ. Manag. 2022, 316, 115236. [Google Scholar] [CrossRef]
- Simon, A.; Luca, Q.; Bert, G.; Inez, D.; Simon, B.; Monica, A.; Tomas, A.; Fernando, M.; Anton, K.; Markus, H.; et al. Integration of biopolymer production with process water treatment at a sugar factory. New Biotechnol. 2014, 31, 308–323. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, J.; Liu, Z.; Yang, W.; Hu, J.; Jia, Z.; Hu, B. Biofilm: A strategy for the dominance of comammox Nitrospira. J. Clean. Prod. 2022, 363, 132316. [Google Scholar] [CrossRef]
- Chang, J.; Ma, L.; Zhou, Y.; Zhang, S.; Wang, W. Remediation of nitrate-contaminated wastewater using denitrification biofilters with straws of ornamental flowers added as carbon source. Water Sci. Technol. 2016, 74, 416–423. [Google Scholar] [CrossRef]
- Yang, Y.; Lin, E.; Sun, S.; Tao, X.; Zhong, L.; Hu, K. Piggery wastewater treatment by Acinetobacter sp. TX5 immobilized with spent mushroom substrate in a fixed-bed reactor. Sci. Total Environ. 2018, 644, 1460–1468. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W. The Research and Application of Sustainable Long-release Carbon Material with Agricultural Waste. Ph.D. Thesis, Nanjing University, Nanjing, China, January 2017. [Google Scholar] [CrossRef]
- Zhong, S.Q.; Yang, Y.; Tao, R.; Li, L.; Zhang, M.; Zhao, J.C. Carbon releasing characteristics and denitrification effects of five plant materials. Chin. J. Environ. Eng. 2014, 8, 1817–1824. Available online: http://www.cjee.ac.cn/article/id/20140520?viewType=HTML (accessed on 1 September 2022).
- Wang, P.C. Performance of Nitrogen and Antibiotics Removal from the Effluent of Wastewater Treatment Plants by Biomass-iron Mixture Denitrification Biological Filter. Master’s Thesis, East China Normal University, Shanghai, China, May 2021. [Google Scholar] [CrossRef]
- Sun, F. The Study on the Improving Technology of Nitrogen and Phosphorus Removal Efficiency in Biofiltration System by Different’ Electron Donors. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, June 2021. [Google Scholar] [CrossRef]
- Sareh, T.; Hadi, M.; Abd, A.N.; Seyyed, E.H.G.; Saeed, B.; Amit, B. Investigation on the performance of sugarcane bagasse as a new carbon source in two hydraulic dimensions of denitrification beds. J. Clean. Prod. 2017, 140, 1176–1181. [Google Scholar] [CrossRef]
- Cui, H. A Tubular Bio-reactor Device Used for Enhancing Denitrification of the Effluent from Rural Wastewater Treatment Facilities and the Demonstration Project. Ph.D. Thesis, East China Normal University, Shanghai, China, May 2019. [Google Scholar]



| Item | Length (mm) | Width (mm) | Thickness (mm) | Mass (g) |
|---|---|---|---|---|
| Average value (mean) | 26.60 | 17.48 | 6.32 | 0.27 |
| Standard deviation (SD) | 7.76 | 4.43 | 2.47 | 0.15 |
| Dosing Ratio | 0% | 0.5% | 1.0% | 1.5% | 2.0% | 2.5% | |
|---|---|---|---|---|---|---|---|
| First-order | k1 (min−1) | 0.0371 | 0.0416 | 0.0761 | 0.0780 | 0.0418 | 0.0040 |
| Intercept | −0.3340 | −0.6204 | −1.3069 | −0.5377 | 1.5911 | 3.4074 | |
| R2 | 0.9518 | 0.8860 | 0.8634 | 0.7805 | 0.7742 | 0.7338 | |
| Second-order | k2 (L·mg−1·s−1) | −0.0169 | −0.0172 | −0.0494 | −0.0362 | −0.0033 | −0.0001 |
| Intercept | 1.0356 | 1.1221 | 2.4313 | 1.7149 | 0.1590 | 0.0078 | |
| R2 | 0.8246 | 0.9102 | 0.6327 | 0.4772 | 0.6553 | 0.7217 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, M.; Tang, Q.; Zou, J.; Lv, X.; Deng, Y.; Ma, X.; Ma, S. Sugarcane Bagasse as Carbon Source and Filler to Enhance the Treatment of Low C/N Wastewater by Aerobic Denitrification Flora. Water 2022, 14, 3355. https://doi.org/10.3390/w14213355
Chen M, Tang Q, Zou J, Lv X, Deng Y, Ma X, Ma S. Sugarcane Bagasse as Carbon Source and Filler to Enhance the Treatment of Low C/N Wastewater by Aerobic Denitrification Flora. Water. 2022; 14(21):3355. https://doi.org/10.3390/w14213355
Chicago/Turabian StyleChen, Maoxia, Qiong Tang, Jiawei Zou, Xiaoyu Lv, Yu Deng, Xuguang Ma, and Shichun Ma. 2022. "Sugarcane Bagasse as Carbon Source and Filler to Enhance the Treatment of Low C/N Wastewater by Aerobic Denitrification Flora" Water 14, no. 21: 3355. https://doi.org/10.3390/w14213355






