Efficiency Determination and Mechanism Investigation of Autotrophic Denitrification Strain F1 to Promote Low-Carbon Development
Abstract
1. Introduction
2. Materials and Methods
2.1. Shewanella sp. Strain F1
2.2. Chemicals and Media
2.3. Morphology Observation
2.4. Determination of the Denitrification Activity
2.5. Effects of Initial pH
2.6. Chemical Analysis
3. Results
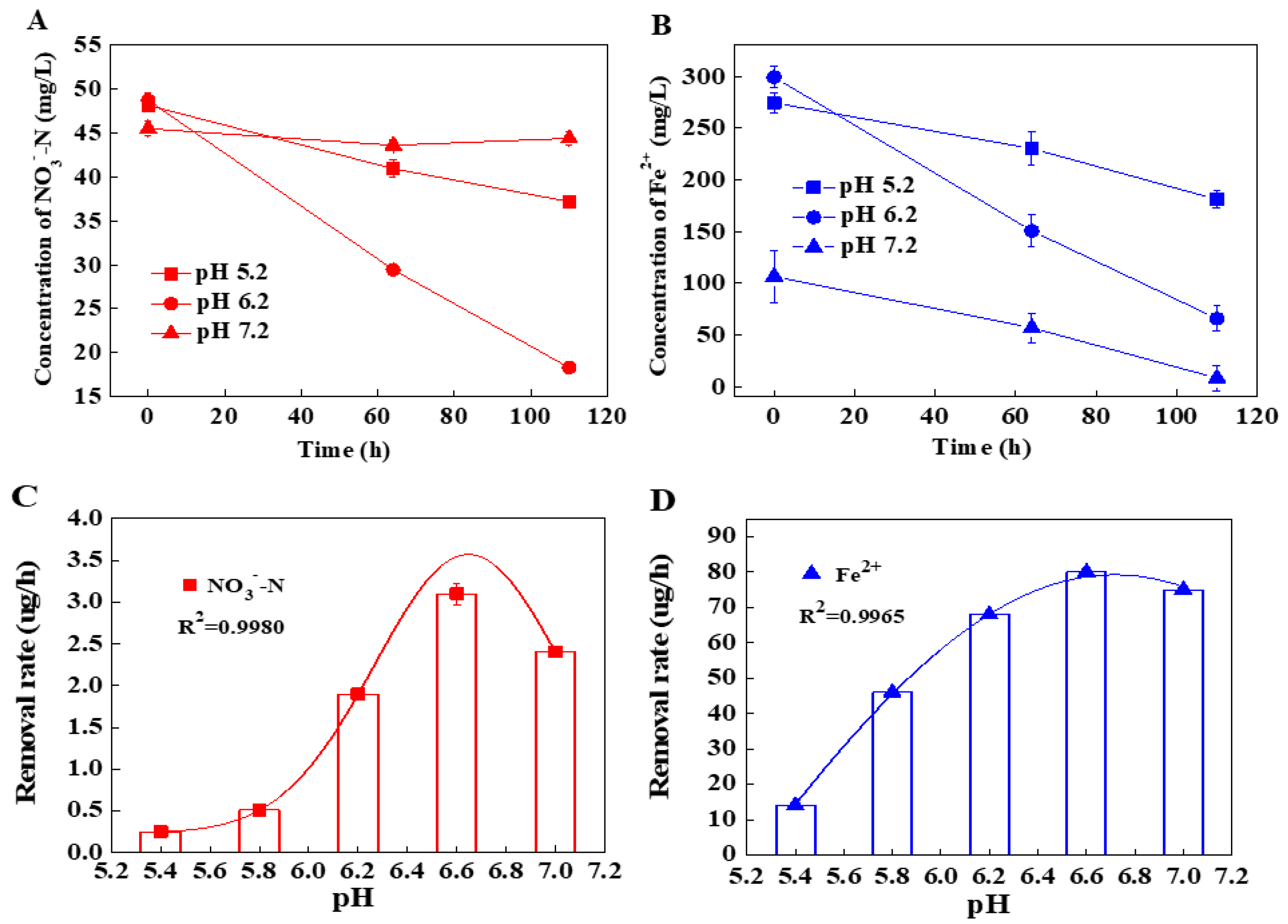
3.1. The Specific Denitrification Activity of Shewanella sp. Strain F1
3.2. The Effects of pH on the Denitrification Activity of Shewanella sp. Strain F1
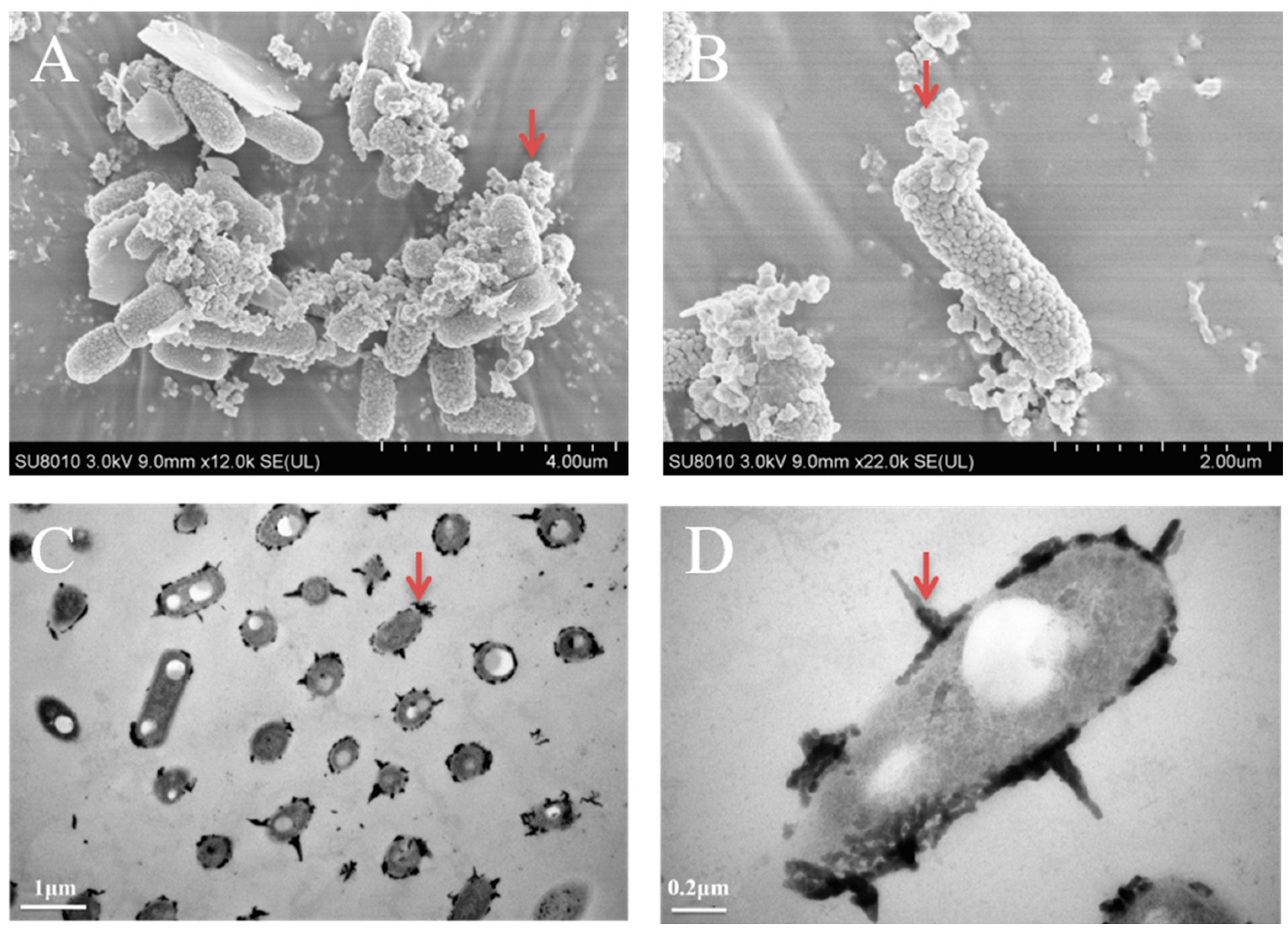
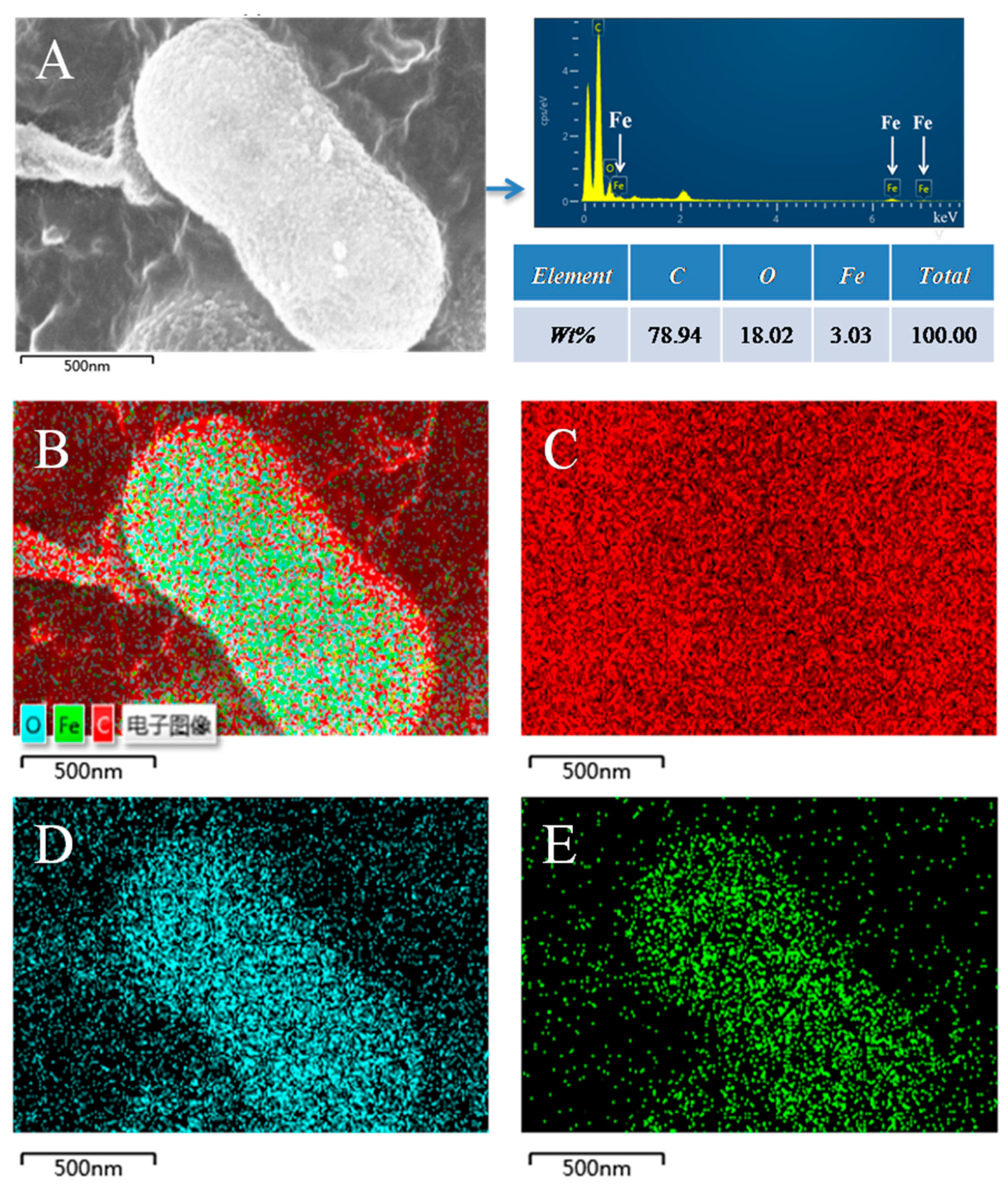
3.3. The Morphology Observation of Shewanella sp. Strain F1
4. Discussion
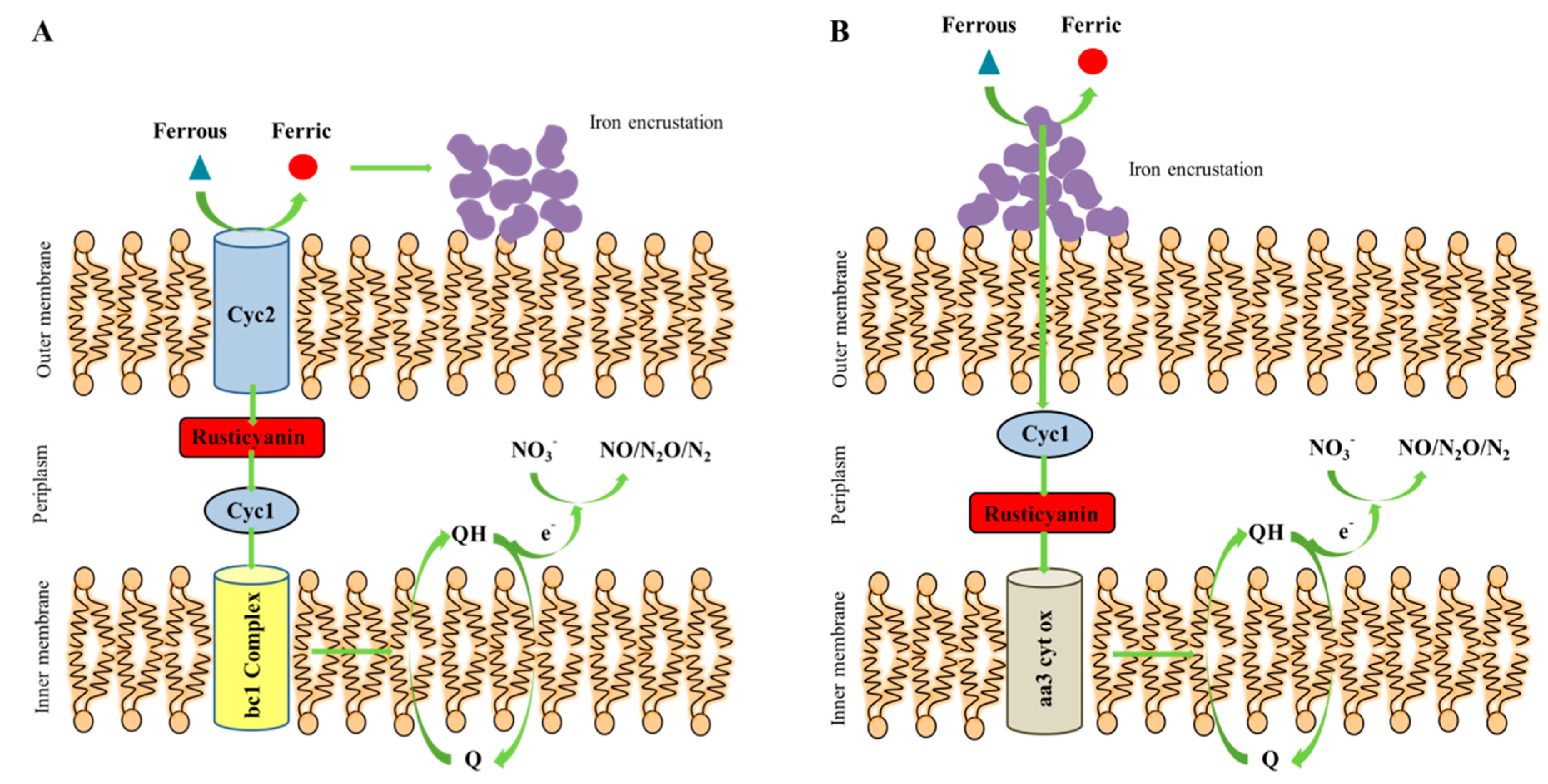
4.1. The Switch for Shewanella sp. Strain F1 to Perform Autotrophic Denitrification
4.2. The Conceptual Mechanism of Shewanella sp. Strain F1 to Perform Autotrophic Denitrification
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kim, T.; Shin, J.; Lee, D.; Kim, Y.; Na, E.; Park, J.; Lim, C.; Cha, Y. Simultaneous feature engineering and interpretation: Forecasting harmful algal blooms using a deep learning approach. Water Res. 2022, 215, 118289. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Zhang, Y.; Wang, Q.; Wei, Y.; Bu, Y.; Ma, B. Achieving advanced nitrogen removal in a novel partial denitrification/anammox-nitrifying (PDA-N) biofilter process treating low C/N ratio municipal wastewater. Bioresour. Technol. 2021, 340, 125661. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Qin, R.; Yang, W.; Su, C.; Luo, Z.; Zhou, Y.; Lin, X.; Lu, Y. Effect of asparagine, corncob biochar and Fe(II) on anaerobic biological treatment under low temperature: Enhanced performance and microbial community dynamic. J. Environ. Manag. 2022, 317, 115348. [Google Scholar] [CrossRef]
- Wang, P.; Li, W.; Ren, S.; Peng, Y.; Wang, Y.; Feng, M.; Guo, K.; Xie, H.; Li, J. Use of sponge iron as an indirect electron donor to provide ferrous iron for nitrate-dependent ferrous oxidation processes: Denitrification performance and mechanism. Bioresour. Technol. 2022, 357, 127318. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zheng, P.; Abbas, G.; Chen, X. Partitionable-space enhanced coagulation (PEC) reactor and its working mechanism: A new prospective chemical technology for phosphorus pollution control. Water Res. 2013, 49, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Xu, S.-Y.; Zhang, M.; Ghulam, A.; Dai, C.-L.; Zheng, P. Iron as electron donor for denitrification: The efficiency, toxicity and mechanism. Ecotoxicol. Environ. Saf. 2020, 194, 110343. [Google Scholar] [CrossRef]
- Wang, R.; Yang, C.; Zhang, M.; Xu, S.-Y.; Dai, C.-L.; Liang, L.-Y.; Zhao, H.-P.; Zheng, P. Chemoautotrophic denitrification based on ferrous iron oxidation: Reactor performance and sludge characteristics. Chem. Eng. J. 2017, 313, 693–701. [Google Scholar] [CrossRef]
- Zhang, M.; Zheng, P.; Li, W.; Wang, R.; Ding, S.; Abbas, G. Performance of nitrate-dependent anaerobic ferrous oxidizing (NAFO) process: A novel prospective technology for autotrophic denitrification. Bioresour. Technol. 2015, 179, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, X.; Liu, T.; Li, F.; Sun, W.; Young, L.Y.; Huang, W. Metagenomic analysis of Fe(II)-oxidizing bacteria for Fe(III) mineral formation and carbon assimilation under microoxic conditions in paddy soil. Sci. Total Environ. 2022, 851, 158068. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Peng, Y.; Gao, H. Enhanced long-term advanced denitrogenation from nitrate wastewater by anammox consortia: Dissimilatory nitrate reduction to ammonium (DNRA) coupling with anammox in an upflow biofilter reactor equipped with EDTA-2Na/Fe(II) ratio and pH control. Bioresour. Technol. 2020, 305, 123083. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Liu, T.; Li, X.; Li, F.; Luo, X.; Wu, Y.; Wang, Y. Biological and chemical processes of microbially mediated nitrate-reducing Fe(II) oxidation by Pseudogulbenkiania sp. strain 2002. Chem. Geol. 2018, 476, 59–69. [Google Scholar] [CrossRef]
- Dong, H.; Zhang, F.; Xu, T.; Liu, Y.L.; Du, Y.; Wang, C.; Liu, T.S.; Gao, J.; He, Y.L.; Wang, X.T.; et al. Culture-dependent and culture-independent methods reveal microbe-clay mineral interactions by dissimilatory iron-reducing bacteria in an integral oilfield. Sci. Total Environ. 2022, 840, 156577. [Google Scholar] [CrossRef]
- Lu, Y.S.; Liu, H.; Huang, X.E.; Xu, L.; Zhou, J.Z.; Qian, G.R.; Shen, J.P.; Chen, X.P. Nitrate removal during Fe(III) bio-reduction in microbial-mediated iron redox cycling systems. Water. Sci. Technol. 2021, 84, 985–994. [Google Scholar] [CrossRef]
- Mateos, G.; Bonilla, A.M.; de Polanco, S.D.; Martinez, J.M.; Escudero, C.; Rodriguez, N.; Sanchez-Andrea, I.; Amils, R. Shewanella sp. T2.3D-1.1 a novel microorganism sustaining the iron cycle in the deep subsurface of the Iberian Pyrite Belt. Microorganisms 2022, 10, 1585. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhao, K.; Liu, X.; Chen, S.; Huang, T.; Guo, H.; Ma, B.; Yang, W.; Yang, Y.; Liu, H. Bacterial community structure and metabolic activity of drinking water pipelines in buildings: A new perspective on dual effects of hydrodynamic stagnation and algal organic matter invasion. Water Res. 2022, 225, 119161. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wan, S.; Lai, L.; Zhang, M.; Zeb, B.S.; Qaisar, M.; Tan, G.; Yuan, L. Recovering phosphate and energy from anaerobic sludge digested wastewater with iron-air fuel cells: Two-chamber cell versus one-chamber cell. Sci. Total Environ. 2022, 825, 154034. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shentu, H.; Chen, H.; Ye, P.; Xu, B.; Zhang, Y.; Zhang, T. Change regularity of water quality parameters in leakage flow conditions and their relationship with iron release. Water Research 2017, 124, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Su, J.F.; Shao, S.C.; Huang, T.L.; Ma, F.; Yang, S.F.; Zhou, Z.M.; Zheng, S.C. Anaerobic nitrate-dependent iron(II) oxidation by a novel autotrophic bacterium, Pseudomonas sp. SZF15. J. Environ. Chem. Eng. 2015, 3, 2187–2193. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, H.; Yang, K.; Sun, Y.; Tian, J. Nitrate removal by nitrate-dependent Fe(II) oxidation in an upflow denitrifying biofilm reactor. Water Sci. Technol. 2015, 72, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.; Yang, K.; Zhu, L.; Jiang, Y.; Wang, H.; Zhou, J.; Zhang, H. Aerobic denitrification: A review of important advances of the last 30 years. Biotechnol. Bioprocess Eng. 2015, 20, 643–651. [Google Scholar] [CrossRef]
- Yuan, Z.; Ma, X.; Wang, S.; Yu, L.; Zhang, P.; Lin, J.; Jia, Y. Effect of co-existent Al(III) in As-rich Acid Mine Drainage (AMD) on As removal during Fe(II) and As(III) abiotic oxidation process. J. Water Process Eng. 2021, 44, 102395. [Google Scholar] [CrossRef]
- Wang, H.; Fan, Y.; Zhou, M.; Wang, W.; Li, X.; Wang, Y. Function of Fe(III)-minerals in the enhancement of anammox performance exploiting integrated network and metagenomics analyses. Water Res. 2022, 210, 117998. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Feng, C.; Sheng, Y.; Dong, S.; Chen, N.; Hao, C. Effect of Fe(II) on reactivity of heterotrophic denitrifiers in the remediation of nitrate- and Fe(II)-contaminated groundwater. Ecotoxicol. Environ. Saf. 2018, 166, 437–445. [Google Scholar] [CrossRef]
- Schmid, G.; Zeitvogel, F.; Hao, L.; Ingino, P.; Flötenmeyer, M.; Stierhof, Y.D.; Schröppel, B.; Burkhardt, C.; Kappler, A.; Obst, M. 3-D analysis of bacterial cell-(iron)mineral aggregates formed during Fe(II) oxidation by the nitrate-reducing Acidovorax sp. strain BoFeN1 using complementary microscopy tomography approaches. Geobiology 2014, 12, 340–361. [Google Scholar] [CrossRef]
- Huang, X.; Yang, X.; Zhu, J.; Yu, J. Microbial interspecific interaction and nitrogen metabolism pathway for the treatment of municipal wastewater by iron carbon based constructed wetland. Bioresour. Technol. 2020, 315, 123814. [Google Scholar] [CrossRef]
- Li, B.; Cheng, Y.; Wu, C.; Li, W.; Yang, Z.; Yu, H. Interaction between ferrihydrite and nitrate respirations by Shewanella oneidensis MR-1. Process Biochem. 2015, 50, 1942–1946. [Google Scholar] [CrossRef]
- Singh, V.K.; Singh, A.L.; Singh, R.; Kumar, A. Iron oxidizing bacteria: Insights on diversity, mechanism of iron oxidation and role in management of metal pollution. Environ. Sustain. 2018, 1, 221–231. [Google Scholar] [CrossRef]
- Zhu, F.; Huang, Y.; Ni, H.; Tang, J.; Zhu, Q.; Long, Z.; Zou, L. Biogenic iron sulfide functioning as electron-mediating interface to accelerate dissimilatory ferrihydrite reduction by Shewanella oneidensis MR-1. Chemosphere 2022, 288, 132661. [Google Scholar] [CrossRef]
- Su, J.F.; Guo, D.X.; Huang, T.L.; Bai, X.C.; Lu, J.S.; Wei, L.; He, L. Microbial Community Analysis and Effect of nZVI on Autotrophic Denitrification in a Biological Reactor. Environ. Eng. Sci. 2019, 36, 564–572. [Google Scholar] [CrossRef]
- Wang, X.-N.; Sun, G.-X.; Li, X.-M.; Clarke, T.A.; Zhu, Y.-G. Electron shuttle-mediated microbial Fe(III) reduction under alkaline conditions. J. Soils Sediments 2017, 18, 159–168. [Google Scholar] [CrossRef]
- Jia, R.; Wang, K.; Li, L.; Qu, Z.; Shen, W.; Qu, D. Abundance and community succession of nitrogen-fixing bacteria in ferrihydrite enriched cultures of paddy soils is closely related to Fe(III)-reduction. Sci. Total Environ. 2020, 720, 137633. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhangzhu, G.C.; Wen, S.X.; Lu, H.F.; Wang, R.; Li, W.; Ding, S.; Ghulam, A.; Zheng, P. Chemolithotrophic denitrification by nitrate-dependent anaerobic iron oxidizing (NAIO) process: Insights into the evaluation of seeding sludge. Chem. Eng. J. 2018, 345, 345–352. [Google Scholar] [CrossRef]
- Zhang, M.; Zheng, P.; Zeng, Z.; Wang, R.; Shan, X.Y.; He, Z.F.; Abbas, G.; Ji, J.Y. Physicochemical characteristics and microbial community of cultivated sludge for nitrate-dependent anaerobic ferrous-oxidizing (NAFO) process. Sep. Purif. Technol. 2016, 169, 296–303. [Google Scholar] [CrossRef]
- Shi, L.; Rosso, K.M.; Zachara, J.M.; Fredrickson, J.K. Mtr extracellular electron-transfer pathways in Fe(III)-reducing or Fe(II)-oxidizing bacteria: A genomic perspective. Biochem. Soc. Trans. 2012, 40, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Pirbadian, S.; Barchinger, S.E.; Leung, K.M.; Byun, H.S.; Jangir, Y.; Bouhenni, R.A.; Reed, S.B.; Romine, M.F.; Saffarini, D.A.; Shi, L.; et al. Shewanella oneidensis MR-1 nanowires are outer membrane and periplasmic extensions of the extracellular electron transport components. Proc. Natl. Acad. Sci. USA 2014, 111, 12883–12888. [Google Scholar] [CrossRef]
- Lin, T.; Ding, W.; Sun, L.; Wang, L.; Liu, C.; Song, H. Engineered Shewanella oneidensis-reduced graphene oxide biohybrid with enhanced biosynthesis and transport of flavins enabled a highest bioelectricity output in microbial fuel cells. Nano Energy 2018, 50, 639–648. [Google Scholar] [CrossRef]
- Beblawy, S.; Bursac, T.; Paquete, C.; Louro, R.; Clarke, T. Extracellular reduction of solid electron acceptors by Shewanella oneidensis. Mol. Microbiol. 2018, 109, 571–583. [Google Scholar] [CrossRef]
- Chen, L.; Wu, Y.; Shen, Q.; Zheng, X.; Chen, Y. Enhancement of hexavalent chromium reduction by Shewanella oneidensis MR-1 in presence of copper nanoparticles via stimulating bacterial extracellular electron transfer and environmental adaptability. Bioresour. Technol. 2022, 361, 127686. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, H.; Zhu, L. Structures of nitroaromatic compounds induce Shewanella oneidensis MR-1 to adopt different electron transport pathways to reduce the contaminants. J. Hazard. Mater. 2020, 384, 121495. [Google Scholar] [CrossRef]

| Component | Concentration | Component | Concentration |
|---|---|---|---|
| NaNO3 | 0.3 g/L | FeSO4∙7H2O | 5 g/L |
| NaHCO3 | 2.5 g/L | MgSO4∙7H2O | 0.5 g/L |
| CaCl2∙2H2O | 0.01 g/L | (NH4)2SO4 | 0.28 g/L |
| KH2PO4 | 0.25 g/L | EDTA | 3.00 mg/L |
| Na2MoO4∙2H2O | 0.036 mg/L | H3BO3 | 0.010 mg/L |
| CoCl2∙6H2O | 0.19 mg/L | NiCl3∙6H2O | 0.024 mg/L |
| MnCl2∙2H2O | 0.50 mg/L | CuCl2∙2H2O | 0.010 mg/L |
| ZnCl2 | 0.07 mg/L |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, F.-K.; Wan, S.-Z.; Liu, B.-Y.; Wang, R.; Zheng, P. Efficiency Determination and Mechanism Investigation of Autotrophic Denitrification Strain F1 to Promote Low-Carbon Development. Water 2022, 14, 3353. https://doi.org/10.3390/w14213353
Qin F-K, Wan S-Z, Liu B-Y, Wang R, Zheng P. Efficiency Determination and Mechanism Investigation of Autotrophic Denitrification Strain F1 to Promote Low-Carbon Development. Water. 2022; 14(21):3353. https://doi.org/10.3390/w14213353
Chicago/Turabian StyleQin, Fang-Kai, Si-Zhuo Wan, Bing-Yin Liu, Ru Wang, and Ping Zheng. 2022. "Efficiency Determination and Mechanism Investigation of Autotrophic Denitrification Strain F1 to Promote Low-Carbon Development" Water 14, no. 21: 3353. https://doi.org/10.3390/w14213353
APA StyleQin, F.-K., Wan, S.-Z., Liu, B.-Y., Wang, R., & Zheng, P. (2022). Efficiency Determination and Mechanism Investigation of Autotrophic Denitrification Strain F1 to Promote Low-Carbon Development. Water, 14(21), 3353. https://doi.org/10.3390/w14213353







