Removal of Heavy Metals from Acid Mine Drainage with Lab-Scale Constructed Wetlands Filled with Oyster Shells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Real and Synthetic AMD
2.2. Substrates
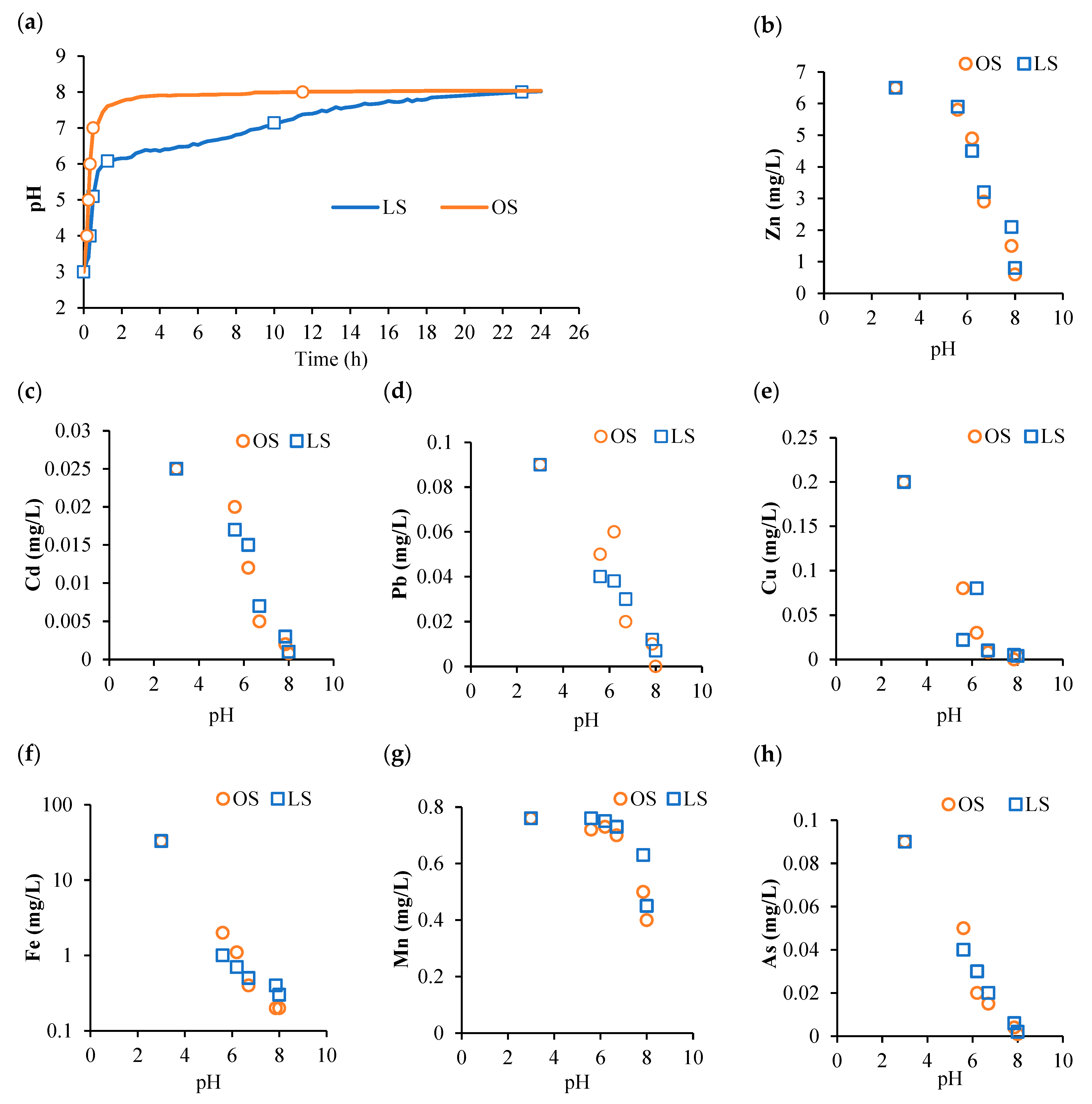
2.3. Batch Experiments for Neutralizing AMD Using OSs and Limestone
2.4. Setting up and Operating Lab-Scale CWs
2.5. Sampling and Analysis
2.6. Calculation
3. Results
3.1. pH Neutralization of AMD in the Batch Experiment
3.2. Environmental Conditions in the Greenhouse and Water Parameters
3.3. Overall Performance of CWs for Heavy Metal Removal
3.4. Heavy Metal Accumulation in Substrates
3.5. Heavy Metal Accumulation in Cattails
3.6. Bacteria in CWs
3.7. Mass Balance of Metals in CWs
4. Discussion
4.1. Relation between pH Changes and Metal Removal Efficiencies
4.2. Heavy Metal Removal Mechanism in Constructed Wetlands
4.3. Recommendations for Further Studies in Using Alternative Filter Materials in CWs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Joshua, O.; Setyo, B.; Kingsley, O.; Chukwunonso, O.; Oluwaseun, J.; Stephen, N.; Felicitas, U.; Shabnam, A.; Chinenye, A. A review of treatment technologies for the mitigation of the toxic environmental effects of acid mine drainage (AMD). Process Saf. Environ. Prot. 2022, 157, 37–58. [Google Scholar] [CrossRef]
- Naidu, G.; Ryu, S.; Thiruvenkatachari, R.; Choi, Y.; Jeong, S.; Vigneswaran, S. A critical review on remediation, reuse, and resource recovery from acid mine drainage. Environ. Pollut. 2019, 247, 1110–1124. [Google Scholar] [CrossRef] [PubMed]
- Skousen, J.G.; Ziemkiewicz, P.F.; McDonald, L.M. Acid mine drainage formation, control and treatment: Approaches and strategies. Extr. Ind. Soc. 2019, 6, 241–249. [Google Scholar] [CrossRef]
- Ueda, H.; Masuda, N. An analysis of mine drainage treatment cost and the technical development to prevent mine pollution. Shigen-to-Sozai 2005, 121, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Koide, R.; Tokoro, C.; Murakami, S.; Adachi, T.; Takahashi, A. A model for prediction of neutralizer usage and sludge generation in the treatment of acid mine drainage from abandoned mines: Case studies in Japan. Mine Water Environ. 2012, 31, 287–296. [Google Scholar] [CrossRef]
- Chen, Q.; Yao, Y.; Li, X.; Lu, J.; Zhou, J.; Huang, Z. Comparison of heavy metal removals from aqueous solutions by chemical precipitation and characteristics of precipitates. J. Water Process. Eng. 2018, 26, 289–300. [Google Scholar] [CrossRef]
- Rodríguez-Galán, M.; Moreno, F.M.B.; Vázquez, S.; Torralvo, F.A.; Vilches, L.F.; Zhang, Z. Remediation of acid mine drainage. Environ. Chem. Lett. 2019, 17, 1529–1538. [Google Scholar] [CrossRef]
- Acheampong, M.A.; Ansa, E.D.O. Low-cost technologies for mining wastewater treatment. J. Environ. Sci. Eng. 2017, 6, 391–405. [Google Scholar] [CrossRef]
- Pat-Espadas, A.; Loredo Portales, R.; Amabilis-Sosa, L.E.; Gómez, G.; Vidal, G. Review of constructed wetlands for acid mine drainage treatment. Water 2018, 10, 1685. [Google Scholar] [CrossRef] [Green Version]
- Yu, G.; Wang, G.; Chi, T.; Du, C.; Wang, J.; Li, P.; Chen, H. Enhanced removal of heavy metals and metalloids by constructed wetlands: A review of approaches and mechanisms. Sci. Total Environ. 2022, 821, 153516. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, J.; Ngo, H.H.; Guo, W.; Hu, Z.; Liang, S.; Liu, H. A review on the sustainability of constructed wetlands for wastewater treatment: Design and operation. Bioresour. Technol. 2015, 175, 594–601. [Google Scholar] [CrossRef] [PubMed]
- de Alvarenga, R.A.F.; Galindro, B.M.; de Fátima Helpa, C.; Soares, S.R. The recycling of oyster shells: An environmental analysis using Life Cycle Assessment. J. Environ. Manag. 2012, 106, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Xia, M.; Li, H.; Chen, T.; Ye, Y.; Zheng, H. Bivalve shell: Not an abundant useless waste but a functional and versatile biomaterial. Crit. Rev. Env. Sci. Tech. 2014, 44, 2502–2530. [Google Scholar] [CrossRef]
- Yoon, G.L.; Yoon, Y.W.; Chae, K.S. Shear strength and compressibility of oyster shell–sand mixtures. Environ. Earth Sci. 2009, 60, 1701–1709. [Google Scholar] [CrossRef]
- Miyaji, Y.; Okamura, T. Geo-material properties of wasted oyster shell sand mixture and its application as material for sand compaction pile. In Coastal Geotechnical Engineering in Practice, Proceedings of the International Symposium IS-Yokohama 2000, Yokohama, Japan, 20–22 September 2000; Balkema: Rotterdam, The Netherlands, 2000; pp. 675–680. [Google Scholar]
- Yang, E.I.; Yi, S.T.; Leem, Y.M. Effect of oyster shell substituted for fine aggregate on concrete characteristics: Part I. Fundamental properties. Cem. Concr. Res. 2005, 35, 2175–2182. [Google Scholar] [CrossRef]
- Yoon, H.; Park, S.; Lee, K.; Park, J. Oyster shell as substitute for aggregate in mortar. Waste Manag. Res. 2004, 22, 158–170. [Google Scholar] [CrossRef]
- Nakatani, N.; Takamori, H.; Takeda, K.; Sakugawa, H. Transesterification of soybean oil using combusted oyster shell waste as a catalyst. Bioresour. Technol. 2009, 100, 1510–1513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, J.H.; Yoo, K.S.; Kim, H.G.; Lee, H.K.; Shon, B.H. Reuse of waste oyster shells as a SO2/NOx removal absorbent. J. Ind. Eng. Chem. 2007, 13, 512–517. [Google Scholar]
- Asaoka, S.; Yamamoto, T.; Kondo, S.; Hayakawa, S. Removal of hydrogen sulfide using crushed oyster shell from pore water to remediate organically enriched coastal marine sediments. Bioresour. Technol. 2009, 100, 4127–4132. [Google Scholar] [CrossRef] [Green Version]
- Choi, Y.M.; Whang, J.H.; Kim, J.M.; Suh, H.J. The effect of oyster shell powder on the extension of the shelf-life of Kimchi. Food Control. 2006, 17, 695–699. [Google Scholar] [CrossRef]
- Yuangsawad, R.; Na-Ranong, D. Recycling oyster shell as adsorbent for phosphate removal. In Proceedings of the 21st Thai Institute of Chemical Engineering and Applied Chemistry, Hat Yai, Thailand, 10–11 November 2011; pp. 1–5. [Google Scholar]
- Masukume, M.; Onyango, M.O.; Maree, J.P. Seashell derived adsorbent and its potential for treating acid mine drainage. Int. J. Miner. Process. 2014, 133, 52–59. [Google Scholar] [CrossRef]
- Park, W.H.; Polprasert, C. Roles of oyster shells in an integrated constructed wetland system designed for P removal. Ecol. Eng. 2008, 34, 50–56. [Google Scholar] [CrossRef]
- Bavandpour, F.; Zou, Y.; He, Y.; Saeed, T.; Sun, Y.; Sun, G. Removal of dissolved metals in wetland columns filled with shell grits and plant biomass. Chem. Eng. J. 2018, 331, 234–241. [Google Scholar] [CrossRef]
- Habert, G.; Bouzidi, Y.; Chen, C.; Jullien, A. Development of a depletion indicator for natural resources used in concrete. Resour. Conserv. Recycl. 2010, 54, 364–376. [Google Scholar] [CrossRef]
- Hara, T.; Kawamoto, K.; Fukuda, K.; Nakagawa, I.; Nguyen, T.T.; Soda, S. Trials on neutralization and metal removal for acid mine drainage utilizing lab-scale constructed wetlands packed with limestone and charcoal. J. Water Waste 2021, 63, 437–443. (In Japanese) [Google Scholar]
- APHA/AWWA/WEF. Standard Methods for the Examination of Water and Wastewater. Standard Methods 541; APHA: Washington, DC, USA, 2012; ISBN 9780875532356. [Google Scholar]
- Ha, N.T.H.; Ha, N.T.; Nga, T.T.H.; Minh, N.N.; Anh, B.T.K.; Hang, N.T.A.; Duc, N.A.; Nhuan, M.T.; Kim, K.W. Uptake of arsenic and heavy metals by native plants growing near Nui Phao multi-metal mine, northern Vietnam. Appl. Geochem. 2019, 108, 104368. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Soda, S.; Kanayama, A.; Hamai, T. Effects of cattails and hydraulic loading on heavy metal removal from closed mine drainage by pilot-scale constructed wetlands. Water 2021, 13, 1937. [Google Scholar] [CrossRef]
- Yeh, T.Y. Removal of metals in constructed wetlands: Review. J. Hazard. Toxic Radioact. Waste 2008, 12, 96–101. [Google Scholar] [CrossRef]
- Allende, K.L.; McCarthy, D.T.; Fletcher, T.D. The influence of media type on removal of arsenic, iron and boron from acidic wastewater in horizontal flow wetland microcosms planted with Phragmites australis. Chem. Eng. J. 2014, 246, 217–228. [Google Scholar] [CrossRef]
- Saeed, T.; Alam, M.K.; Miah, M.J.; Majed, N. Removal of heavy metals in subsurface flow constructed wetlands: Application of effluent recirculation. Environ. Sustain. Indic. 2021, 12, 100146. [Google Scholar] [CrossRef]
- Machemer, S.D.; Wildeman, T.R. Adsorption compared with sulfide precipitation as metal removal processes from acid mine drainage in a constructed wetland. J. Contam. Hydrol. 1992, 9, 115–131. [Google Scholar] [CrossRef]
- White, C.; Gadd, G.M. An internal sedimentation bioreactor for laboratory-scale removal of toxic metals from soil leachates using biogenic sulphide precipitation. J. Ind. Microbiol. Biotechnol. 1997, 18, 414–421. [Google Scholar] [CrossRef]
- Wu, S.; Kuschk, P.; Wiessner, A.; Müller, J.; Saad, R.A.; Dong, R. Sulphur transformations in constructed wetlands for wastewater treatment: A review. Ecol. Eng. 2013, 52, 278–289. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Huang, H.; Nguyen, T.A.H.; Soda, S. Recycling clamshell as substrate in lab-scale constructed wetlands for heavy metal removal from simulated acid mine drainage. Process Saf. Environ. Prot. 2022, 165, 950–958. [Google Scholar] [CrossRef]
- Hasegawa, N.; Onitsuka, T.; Takeyama, S.; Maekawa, K. Oyster Culture in Hokkaido, Japan. Bull. Fish. Res. Agen. 2015, 40, 173. [Google Scholar]
- Ventilla, R.F. Recent developments in the Japanese oyster culture industry. Adv. Mar. Biol. 1984, 21, 1–57. [Google Scholar] [CrossRef]





| Synthetic AMD | Real AMD (mg/L) | Effluent Standard (mg/L) | ||
|---|---|---|---|---|
| Reagents | Concentration (mg/L) | |||
| Zn | ZnSO4•7H2O | 7.22 ± 0.20 | 7.63 ± 1.64 | <2 |
| Cu | CuSO4•5H2O | 0.20 ± 0.07 | 0.20 ± 0.09 | <3 |
| Cd | CdCl2 | 0.04 ± 0.02 | 0.03 ± 0.02 | <0.03 |
| Mn | MnSO4•5H2O | 0.90 ± 0.29 | 1.40 ± 0.29 | <10 |
| Pb | PbCl2 | 0.17 ± 0.10 | 0.11 ± 0.11 | <0.1 |
| Fe | FeSO4•7H2O | 37.00 ± 2.53 | 35.40 ± 6.40 | <10 |
| As | AsNaO2 | 0.06 ± 0.04 | 0.05 ± 0.05 | <0.1 |
| Ca | CaSO4•2H2O | 30.00 ± 3.00 | 30.00 ± 4.50 | |
| Na | NaCl | 6.70 ± 0.60 | 6.80 ± 1.20 | |
| Mg | MgSO4•7H2O | 10.00 ± 1.00 | 10.10 ± 2.00 | |
| K | KCl | 1.50 ± 0.15 | 1.60 ± 0.20 | |
| Al | AlNa(SO4)2•12H2O | 2.01 ± 0.21 | 2.60 ± 0.30 | |
| N | KNO3 | 0.40 ± 0.06 | 0.30 ± 0.10 | <100 |
| pH | 3.70 ± 0.40 | 3.30 ± 0.16 | 5.8–8.6 | |
| Parameters | Simulated AMD (n = 21) | Real AMD (n = 6) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Influent | Effluent | Influent | Effluent | |||||||
| OS-P | OS-UP | LS-P | LS-UP | OS-P | OS-UP | LS-P | LS-UP | |||
| Water (L/cycle) | 1 | 0.8 ± 0.0 | 0.9 ± 0.0 | 0.8 ± 0.0 | 0.9 ± 0.0 | 1 | 0.8 ± 0.1 | 0.8 ± 0.1 | 0.9 ± 0.0 | 0.9 ± 0.0 |
| Evapot (%) | N/A | (14.1 ± 5.5) | (8.4 ± 3.4) | (12.5 ± 2.6) | (9.3 ± 1.8) | N/A | (20.4 ± 15.2) | (11.5 ± 6.6) | (16.7 ± 7.8) | (11.3 ± 4.7) |
| pH | 3.7 ± 0.4 | 7.2 ± 0.3 | 7.3 ± 0.3 | 7.2 ± 0.2 | 7.3 ± 0.35 | 3.2 ± 0.2 | 7.0 ± 0.2 | 7.1 ± 0.2 | 7.0 ± 0.3 | 7.0 ± 0.5 |
| ORP (mV) | 185.0 ± 9.8 | 75.5 ± 38.2 | 79.0 ± 31.8 | 90.3 ± 34.2 | 88.6 ± 34.0 | 184.1 ± 6.6 | 69.6 ± 50.9 | 61.5 ± 50.0 | 65.4 ± 51.5 | 67.0 ± 52.0 |
| DO (mg/L) | 9.2 ± 0.3 | 8.2 ± 2.7 | 9.8 ± 2.9 | 7.6 ± 1.4 | 9.5 ± 0.9 | 0.5 ± 0.1 * | 9.3 ± 3.4 | 13.1 ± 4.2 | 8.9 ± 2.9 | 10.9 ± 2.4 |
| SS (mg/L) | N/A | 29.1 ± 19.6 | 28.1 ± 17.7 | 30.4 ± 17.6 | 24.9 ± 118.2 | N/A | 15.1 ± 1.8 | 15.4 ± 4.8 | 17.9 ± 2.2 | 15.5 ± 0.9 |
| TDS (mg/L) | 228.8 ± 14.7 | 327.7 ± 69.7 | 320.4 ± 56.5 | 238.6 ± 8.5 | 232.3 ± 8.8 | 291.6 ± 1.5 | 308.6 ± 4.9 | 287.8 ± 1.4 | 255.2 ± 1.6 | 246.5 ± 7.7 |
| SO42-(mg/L) | 236.1 ± 22.1 | 216.0 ± 29.9 | 206.4 ± 32.5 | 200.4 ± 22.9 | 218.6 ± 18.7 | 236.5 ± 9.5 | 190.5 ± 24.4 | 193.5 ± 42.2 | 205.0 ± 40.0 | 228.0 ± 36.0 |
| Metal Removal | Simulated AMD (n = 21) | Real AMD (n = 6) | ||||||
|---|---|---|---|---|---|---|---|---|
| OS-P | OS-UP | LS-P | LS-UP | OS-P | OS-UP | LS-P | LS-UP | |
| Zn (%) | 98.7 ± 1.0 | 98.9 ± 1.0 | 93.1 ± 3.5 | 93.2 ± 4.9 | 99.2 ± 0.7 | 99.0 ± 1.2 | 88.6 ± 7.2 | 91.6 ± 5.9 |
| Cd (%) | 98.9 ± 1.3 | 98.5 ± 1.4 | 96.9 ± 1.6 | 95.8 ± 4.8 | 91.5 ± 5.9 | 91.5 ± 5.3 | 89.9 ± 6.1 | 87.8 ± 7.8 |
| Cu (%) | 93.4 ± 16.5 | 94.8 ± 4.9 | 98.7 ± 0.8 | 98.4 ± 1.0 | 95.3 ± 5.9 | 95.6 ± 4.9 | 98.9 ± 0.9 | 98.7 ± 1.0 |
| Pb (%) | 89.4 ± 4.5 | 88.7 ± 8.8 | 89.1 ± 6.8 | 88.8 ± 6.1 | 91.0 ± 2.8 | 90.7 ± 2.5 | 88.3 ± 12.4 | 88.3 ± 10.3 |
| As (%) | 95.6 ± 4.5 | 95.9 ± 3.9 | 95.2 ± 2.2 | 92.6 ± 8.9 | 91.3 ± 6.3 | 92.7 ± 6.4 | 63.6 ± 40.5 | 48.2 ± 52.8 |
| Mn (%) | 93.8 ± 8.3 | 93.8 ± 5.3 | 67.9 ± 19.3 | 54.3 ± 24.4 | 92.6 ± 6.2 | 90.9 ± 4.4 | 65.8 ± 19.1 | 58.8 ± 25.9 |
| Fe (%) | 99.7 ± 0.1 | 99.7 ± 0.1 | 99.7 ± 0.1 | 99.7 ± 0.1 | 99.0 ± 2.1 | 98.9 ± 1.8 | 98.8 ± 0.2 | 98.7 ± 0.3 |
| CWs | Metal | TF | BCF | |
|---|---|---|---|---|
| Before Use | After Use | |||
| OS-based CW | As | 0.56 | 0.55 | 11.49 |
| Zn | 0.65 | 0.53 | 3.22 | |
| Cd | 0.49 | 0.36 | 19.73 | |
| Cu | 0.82 | 0.82 | 12.83 | |
| Pb | 0.25 | 0.38 | 4.20 | |
| Mn | 1.12 | 1.11 | 4.68 | |
| Fe | 0.13 | 0.12 | 8.19 | |
| LS-based CW | As | 0.56 | 0.59 | 17.30 |
| Zn | 0.65 | 0.39 | 11.09 | |
| Cd | 0.49 | 0.35 | 40.60 | |
| Cu | 0.82 | 0.82 | 69.57 | |
| Pb | 0.25 | 0.30 | 4.81 | |
| Mn | 1.12 | 1.10 | 15.85 | |
| Fe | 0.13 | 0.11 | 15.58 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.T.; Soda, S.; Horiuchi, K. Removal of Heavy Metals from Acid Mine Drainage with Lab-Scale Constructed Wetlands Filled with Oyster Shells. Water 2022, 14, 3325. https://doi.org/10.3390/w14203325
Nguyen TT, Soda S, Horiuchi K. Removal of Heavy Metals from Acid Mine Drainage with Lab-Scale Constructed Wetlands Filled with Oyster Shells. Water. 2022; 14(20):3325. https://doi.org/10.3390/w14203325
Chicago/Turabian StyleNguyen, Thuong Thi, Satoshi Soda, and Kengo Horiuchi. 2022. "Removal of Heavy Metals from Acid Mine Drainage with Lab-Scale Constructed Wetlands Filled with Oyster Shells" Water 14, no. 20: 3325. https://doi.org/10.3390/w14203325




