Controls on Groundwater Fluoride Contamination in Eastern Parts of India: Insights from Unsaturated Zone Fluoride Profiles and AI-Based Modeling
Abstract
:1. Introduction
2. Materials and Methods
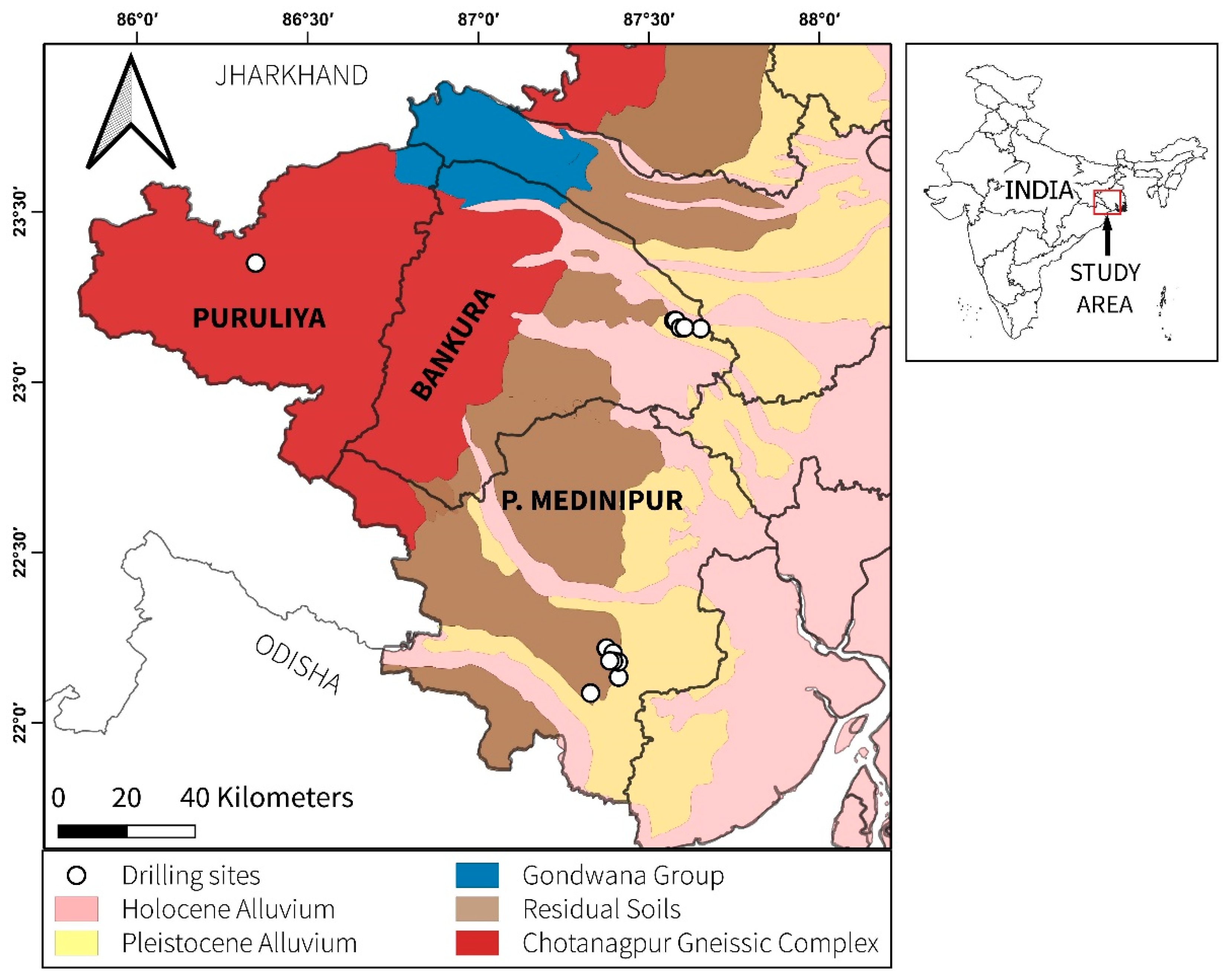
2.1. Study Area
2.2. Groundwater Sampling and Analysis
2.3. Groundwater F Prediction Model
2.4. Sediment Sampling and Analysis
3. Results
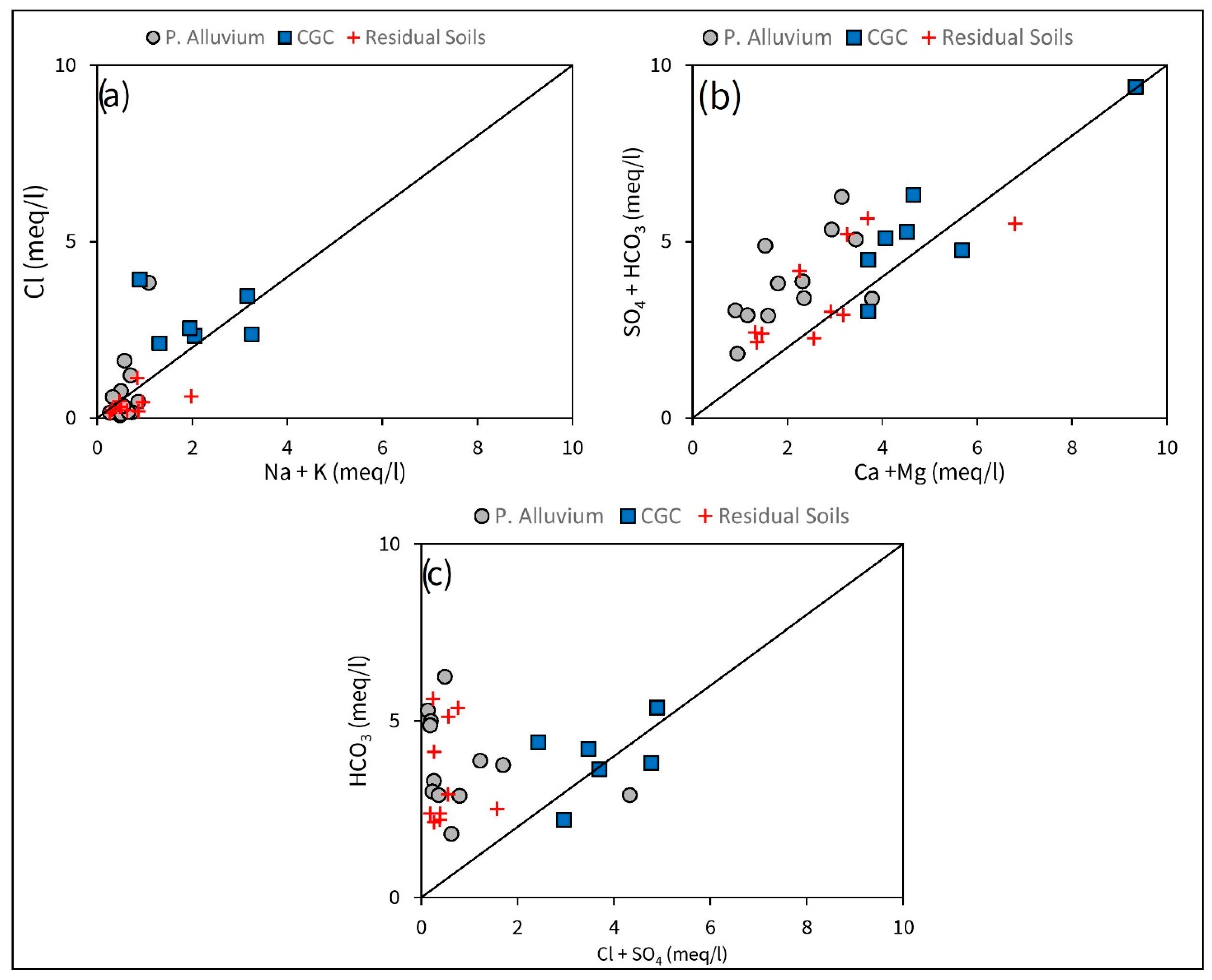
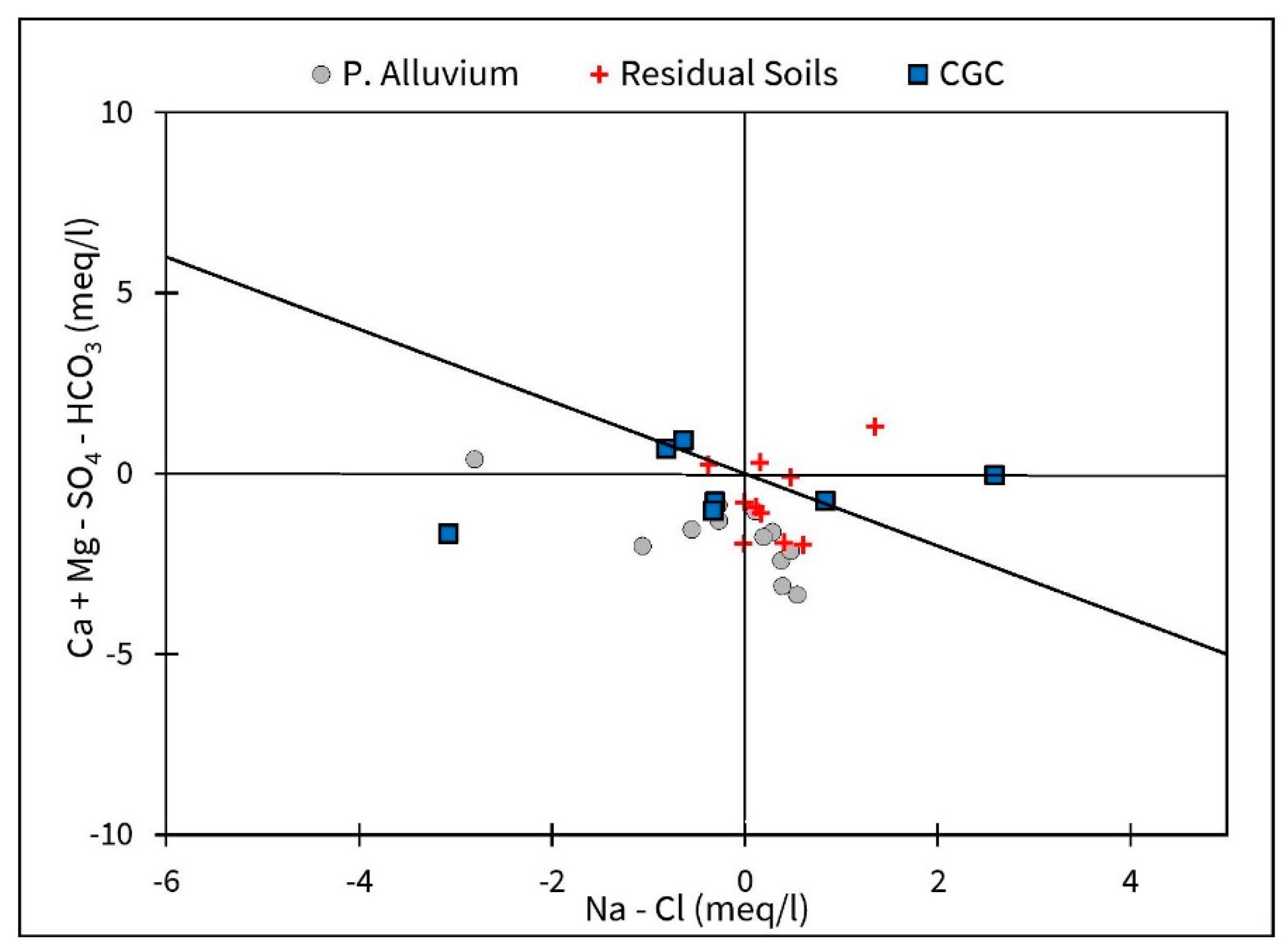
3.1. Groundwater Chemistry and F Distribution
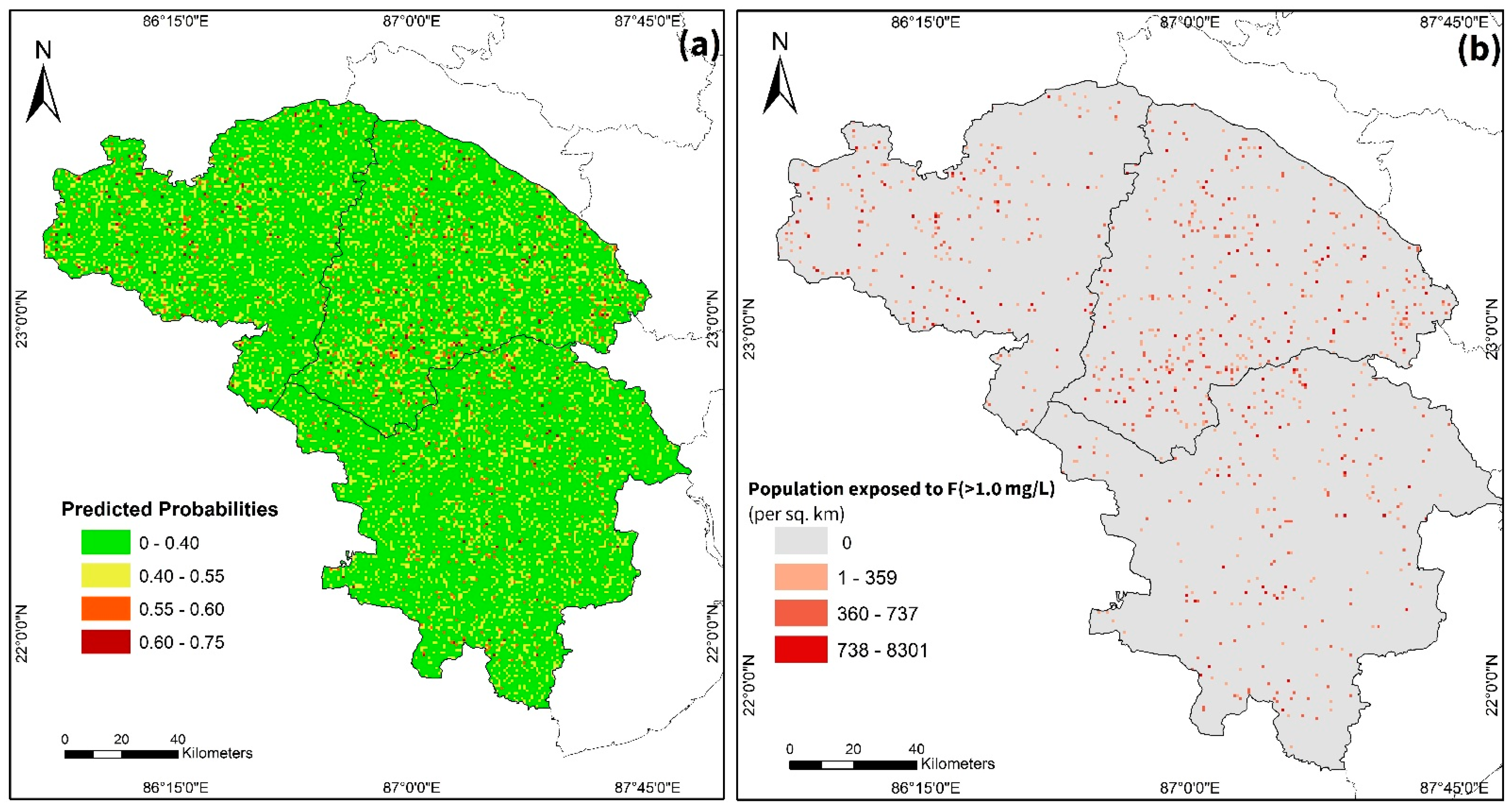
3.2. Model Prediction of the F Distribution
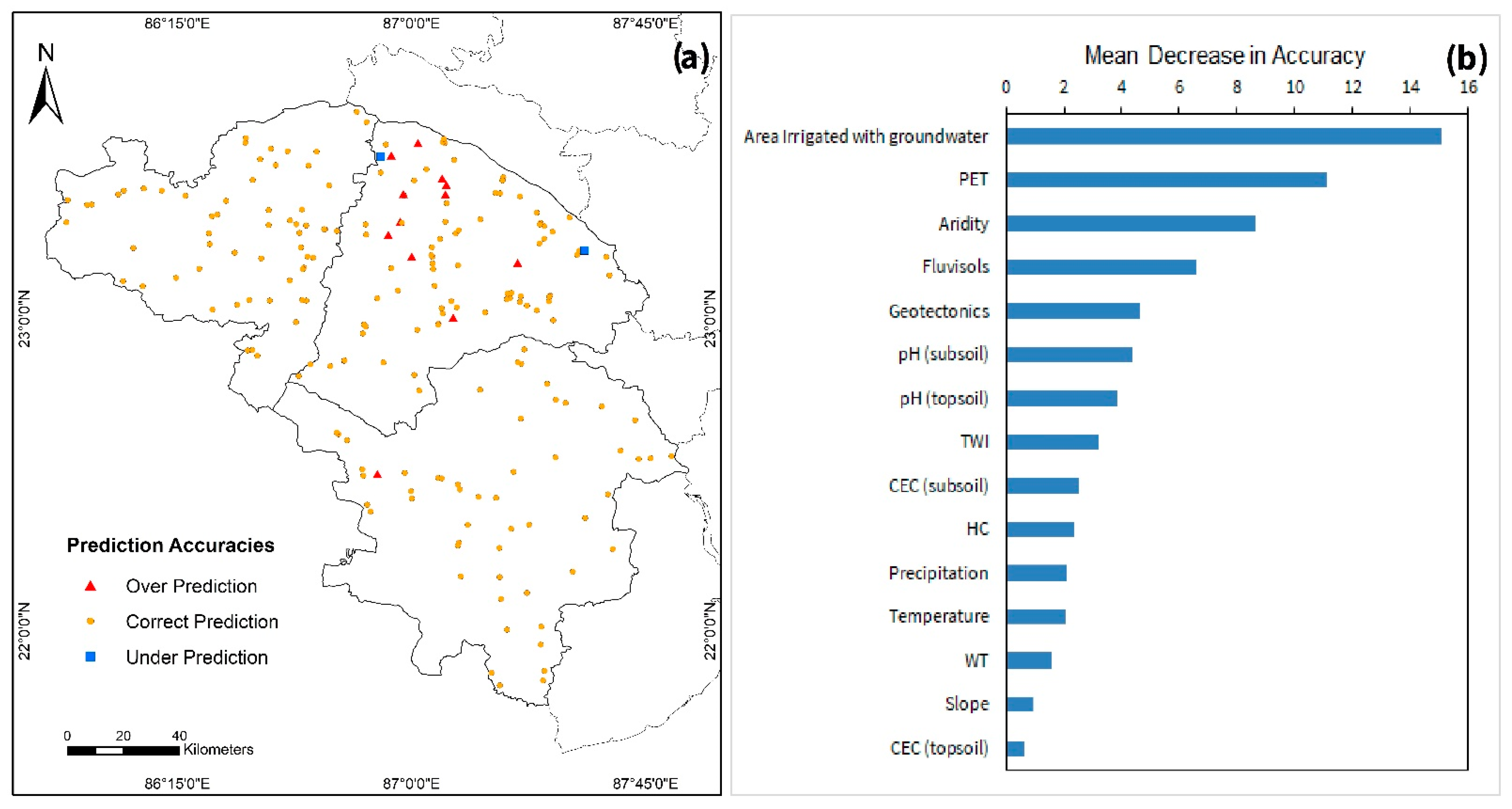
3.3. Model Limitations
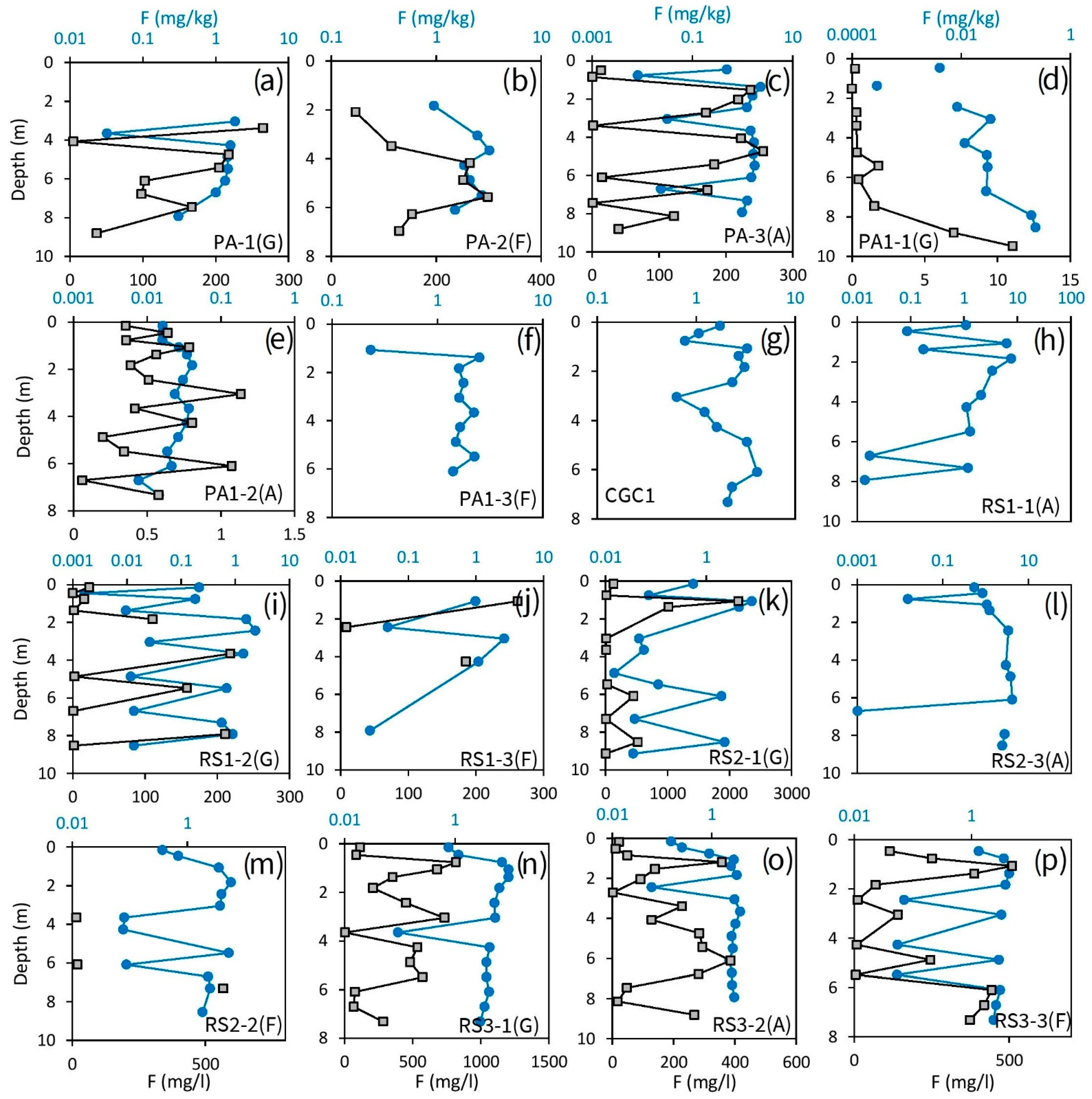
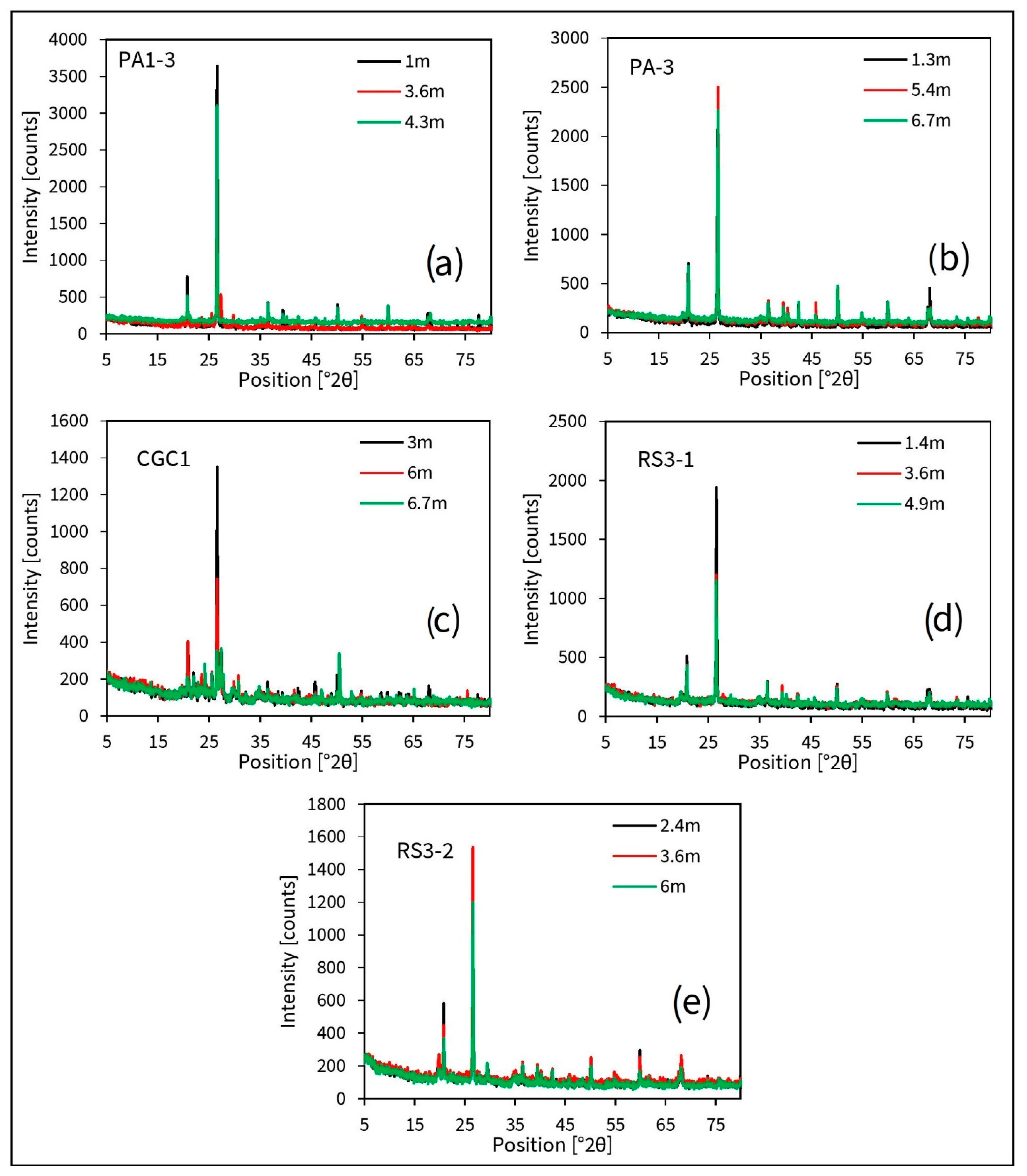
3.4. Unsaturated Zone F profile
4. Discussion
4.1. Hydrogeochemical Controls of F Mobilization
4.2. Significance of Predictor Variables
4.3. Fate of F and Impact of Unsaturated Zone Matrix
4.4. Land-Use Patterns and F Distribution
4.5. Implications for Groundwater F Pollution
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Subba Rao, N.; John Devadas, D. Fluoride Incidence in Groundwater in an Area of Peninsular India. Environ. Geol. 2003, 45, 243–251. [Google Scholar] [CrossRef]
- Arnesen, A.K.M. Availability of Fluoride to Plants Grown in Contaminated Soils. Plant Soil 1997, 191, 13–25. [Google Scholar] [CrossRef]
- Edmunds, W.M.; Smedley, P.L. Fluoride in Natural Waters. In Essentials of Medical Geology: Revised Edition; Springer: Dordrecht, The Netherlands, 2013; pp. 311–336. ISBN 9789400743755. [Google Scholar]
- Dabeka, R.W.; Mckenzie, A.D. Survey of Lead, Cadmium, Fluoride, Nickel, and Cobalt in Food Composites and Estimation of Dietary Intakes of These Elements by Canadians in 1986–1988. J. AOAC Int. 1995, 78, 897–909. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.I.; Hasson, H. Fluoride Supplements, Dental Caries and Fluorosis: A Systematic Review. J. Am. Dent. Assoc. 2008, 139, 1457–1468. [Google Scholar] [CrossRef]
- Oliveira, M.J.L.; Paiva, S.M.; Martins, L.H.P.M.; Ramos-Jorge, M.L.; Lima, Y.B.O.; Cury, J.A. Fluoride Intake by Children at Risk for the Development of Dental Fluorosis: Comparison of Regular Dentifrices and Flavoured Dentifrices for Children. Caries Res. 2007, 41, 460–466. [Google Scholar] [CrossRef]
- Symonds, R.B.; Rose, W.I.; Reed, M.H. Contribution of C1- and F-Bearing Gases to the Atmosphere by Volcanoes. Nature 1988, 334, 415–418. [Google Scholar] [CrossRef]
- Fejerskov, O.; Larsen, M.J.; Richards, A.; Baelum, V. Dental Tissue Effects of Fluoride. Adv. Dent. Res. 1994, 8, 15–31. [Google Scholar] [CrossRef]
- Izuora, K.; Twombly, J.G.; Whitford, G.M.; Demertzis, J.; Pacifici, R.; Whyte, M.P. Skeletal Fluorosis from Brewed Tea. J. Clin. Endocrinol. Metab. 2011, 96, 2318–2324. [Google Scholar] [CrossRef] [Green Version]
- Freni, S.C. Exposure to High Fluoride Concentrations in Drinking Water Is Associated with Decreased Birth Rates. J. Toxicol. Environ. Health 1994, 42, 109–121. [Google Scholar] [CrossRef]
- Xiong, X.Z.; Liu, J.L.; He, W.H.; Xia, T.; He, P.; Chen, X.M.; Yang, K.D.; Wang, A.G. Dose-Effect Relationship between Drinking Water Fluoride Levels and Damage to Liver and Kidney Functions in Children. Environ. Res. 2007, 103, 112–116. [Google Scholar] [CrossRef]
- Viswanathan, G.; Jaswanth, A.; Gopalakrishnan, S.; Siva ilango, S.; Aditya, G. Determining the Optimal Fluoride Concentration in Drinking Water for Fluoride Endemic Regions in South India. Sci. Total Environ. 2009, 407, 5298–5307. [Google Scholar] [CrossRef] [PubMed]
- Heller, K.E.; Eklund, S.A.; Burt, B.A. Dental Caries and Dental Fluorosis at Varying Water Fluoride Concentrations. J. Public Health Dent. 1997, 57, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Bandekar, K. IS 10500: 91 Drinking Water Compared to WHO Guidelines. Indian Waterworks Assoc. 1993, 30, 179–184. [Google Scholar]
- Asian Deveopment Bank. Arsenic and Fluoride in Drinking Water in West Bengal: Characteristics, Implications, and Mitigation; Asian Deveopment Bank: New Delhi, India, 2020. [Google Scholar]
- Podgorski, J.E.; Labhasetwar, P.; Saha, D.; Berg, M. Prediction Modeling and Mapping of Groundwater Fluoride Contamination throughout India. Environ. Sci. Technol. 2018, 52, 9889–9898. [Google Scholar] [CrossRef] [Green Version]
- Samal, A.C.; Bhattacharya, P.; Mallick, A.; Ali, M.M.; Pyne, J.; Santra, S.C. A Study to Investigate Fluoride Contamination and Fluoride Exposure Dose Assessment in Lateritic Zones of West Bengal, India. Environ. Sci. Pollut. Res. 2015, 22, 6220–6229. [Google Scholar] [CrossRef]
- Mondal, N.K.; Pal, K.C.; Kabi, S. Prevalence and Severity of Dental Fluorosis in Relation to Fluoride in Ground Water in the Villages of Birbhum District, West Bengal, India. Environmentalist 2012, 32, 70–84. [Google Scholar] [CrossRef]
- Das, K.; Mondal, N.K. Dental Fluorosis and Urinary Fluoride Concentration as a Reflection of Fluoride Exposure and Its Impact on IQ Level and BMI of Children of Laxmisagar, Simlapal Block of Bankura District, W.B., India. Environ. Monit. Assess. 2016, 188, 1–14. [Google Scholar] [CrossRef]
- Majumdar, K.K. Prevalence of Fluorosis and Pattern of Domestic Filters Use in Two Fluoride Endemic Blocks of West Bengal, India. J. Compr. Heal. 2015, 3, 17–30. [Google Scholar] [CrossRef]
- Bhattacharya, H.N.; Chakrabarti, S.; Chakrabarti, S.; Bhattacharya, H.N. Incidence of Fluoride in the Groundwater of Purulia District, West Bengal: A Geo-Environmental Appraisal. Curr. Sci. 2011, 101, 152–155. [Google Scholar]
- Chakrabarti, S.; Bhattacharya, H.N. Inferring the Hydro-Geochemistry of Fluoride Contamination in Bankura District, West Bengal: A Case Study. J. Geol. Soc. India 2013, 82, 379–391. [Google Scholar] [CrossRef]
- Jacks, G.; Bhattacharya, P.; Chaudhary, V.; Singh, K.P. Controls on the Genesis of Some High-Fluoride Groundwaters in India. Appl. Geochemistry 2005, 20, 221–228. [Google Scholar] [CrossRef]
- Handa, B.K. Geochemistry and Genesis of Fluoride-Containing Ground Waters in India. Ground Water 1975, 13, 275–281. [Google Scholar] [CrossRef]
- Borgnino, L.; Garcia, M.G.; Bia, G.; Stupar, Y.V.; Le Coustumer, P.; Depetris, P.J. Mechanisms of Fluoride Release in Sediments of Argentina’s Central Region. Sci. Total Environ. 2013, 443, 245–255. [Google Scholar] [CrossRef]
- Banerjee, A. Groundwater Fluoride Contamination: A Reappraisal. Geosci. Front. 2015, 6, 277–284. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, S.; Mukherjee, A.; Gupta, S.D.; Bhanja, S.N.; Bhattacharya, A. Predicting Regional-Scale Elevated Groundwater Nitrate Contamination Risk Using Machine Learning on Natural and Human-Induced Factors. ACS ES&T Eng. 2022, 2, 689–702. [Google Scholar] [CrossRef]
- Chakraborty, M.; Sarkar, S.; Mukherjee, A.; Shamsudduha, M.; Ahmed, K.M.; Bhattacharya, A.; Mitra, A. Modeling Regional-Scale Groundwater Arsenic Hazard in the Transboundary Ganges River Delta, India and Bangladesh: Infusing Physically-Based Model with Machine Learning. Sci. Total Environ. 2020, 748, 141107. [Google Scholar] [CrossRef]
- Singha, S.; Pasupuleti, S.; Singha, S.S.; Singh, R.; Kumar, S. Prediction of Groundwater Quality Using Efficient Machine Learning Technique. Chemosphere 2021, 276, 130265. [Google Scholar] [CrossRef]
- Malakar, P.; Mukherjee, A.; Bhanja, S.N.; Sarkar, S.; Saha, D.; Ray, R.K. Deep Learning-Based Forecasting of Groundwater Level Trends in India: Implications for Crop Production and Drinking Water Supply. ACS ES&T Eng. 2021, 1, 965–977. [Google Scholar] [CrossRef]
- Malakar, P.; Mukherjee, A.; Bhanja, S.N.; Ray, R.K.; Sarkar, S.; Zahid, A. Machine-Learning-Based Regional-Scale Groundwater Level Prediction Using GRACE. Hydrogeol. J. 2021, 29, 1027–1042. [Google Scholar] [CrossRef]
- Bhanja, S.N.; Malakar, P.; Mukherjee, A.; Rodell, M.; Mitra, P.; Sarkar, S. Using Satellite-Based Vegetation Cover as Indicator of Groundwater Storage in Natural Vegetation Areas. Geophys. Res. Lett. 2019, 46, 8082–8092. [Google Scholar] [CrossRef]
- Bhanja, S.N.; Mukherjee, A.; Rangarajan, R.; Scanlon, B.R.; Malakar, P.; Verma, S. Long-Term Groundwater Recharge Rates across India by in Situ Measurements. Hydrol. Earth Syst. Sci. 2019, 23, 711–722. [Google Scholar] [CrossRef] [Green Version]
- Malakar, P.; Mukherjee, A.; Bhanja, S.N.; Ganguly, A.R.; Ray, R.K.; Zahid, A.; Sarkar, S.; Saha, D.; Chattopadhyay, S. Three Decades of Depth-Dependent Groundwater Response to Climate Variability and Human Regime in the Transboundary Indus-Ganges-Brahmaputra-Meghna Mega River Basin Aquifers. Adv. Water Resour. 2021, 149, 103856. [Google Scholar] [CrossRef]
- Tylenda, C.A. Toxicological Profile for Fluorides, Hydrogen Fluoride, and Fluorine; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2003. [Google Scholar]
- Barrow, N.J.; Ellis, A.S. Testing a Mechanistic Model. III. The Effects of PH on Fluoride Retention by a Soil. J. Soil Sci. 1986, 37, 287–293. [Google Scholar] [CrossRef]
- Bower, C.A.; Hatcher, J.T. Adsorption Of Fluoride By Soils And Minerals. Soil Sci. 1967, 103, 151–154. [Google Scholar] [CrossRef]
- Omueti, J.A.I.; Jones, R.L. Fluoride Adsorption By Illinois Soils. J. Soil Sci. 1977, 28, 564–572. [Google Scholar] [CrossRef]
- Brewer, R.F. Diagnostic Criteria for Plants and Soils; University of California Press: Berkeley, CA, USA, 1966; pp. 180–196. [Google Scholar]
- Tracy, P.W.; Robbins, C.W.; Lewis, G.C. Fluorite Precipitation in a Calcareous Soil Irrigated with High Fluoride Water. Soil Sci. Soc. Am. J. 1984, 48, 1013–1016. [Google Scholar] [CrossRef] [Green Version]
- Gilpin, L.; Johnson, A.H. Fluorine in Agricultural Soils of Southeastern Pennsylvania. Soil Sci. Soc. Am. J. 1980, 44, 255–258. [Google Scholar] [CrossRef]
- Shacklette, H.T.; Boerngen, J.G.; Keith, J.R. Selenium, Fluorine, and Arsenic in Surficial Materials of the Conterminous United States; US Department of the Interior, Geological Survey: Washington, DC, USA, 1974.
- Zhu, L.; Zhang, H.H.; Xia, B.; Xu, D.R. Total Fluoride in Guangdong Soil Profiles, China: Spatial Distribution and Vertical Variation. Environ. Int. 2007, 33, 302–308. [Google Scholar] [CrossRef]
- Ramteke, L.P.; Sahayam, A.C.; Ghosh, A.; Rambabu, U.; Reddy, M.R.P.; Popat, K.M.; Rebary, B.; Kubavat, D.; Marathe, K.V.; Ghosh, P.K. Study of Fluoride Content in Some Commercial Phosphate Fertilizers. J. Fluor. Chem. 2018, 210, 149–155. [Google Scholar] [CrossRef]
- Scanlon, B.R.; Stonestrom, D.A.; Reedy, R.C.; Leaney, F.W.; Gates, J.; Cresswell, R.G. Inventories and Mobilization of Unsaturated Zone Sulfate, Fluoride, and Chloride Related to Land Use Change in Semiarid Regions, Southwestern United States and Australia. Water Resour. Res. 2009, 45, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Brindha, K.; Rajesh, R.; Murugan, R.; Elango, L. Fluoride Contamination in Groundwater in Parts of Nalgonda District, Andhra Pradesh, India. Environ. Monit. Assess. 2011, 172, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Bi, Y.; Mi, W.; Xie, S.; Ji, L. Land-Use Change Caused by Anthropogenic Activities Increase Fluoride and Arsenic Pollution in Groundwater and Human Health Risk. J. Hazard. Mater. 2021, 406, 124337. [Google Scholar] [CrossRef] [PubMed]
- Saraf, A.K.; Choudhury, P.R.; Roy, B.; Sarma, B.; Vijay, S.; Choudhury, S. GIS Based Surface Hydrological Modelling in Identification of Groundwater Recharge Zones. Int. J. Remote Sens. 2004, 25, 5759–5770. [Google Scholar] [CrossRef]
- Nag, S.K. Morphometric Analysis Using Remote Sensing Techniques in the Chaka Sub-Basin, Purulia District, West Bengal. J. Indian Soc. Remote Sens. 1998, 26, 69–76. [Google Scholar] [CrossRef]
- Chowdhury, A.; Jha, M.K.; Chowdary, V.M.; Mal, B.C. Integrated Remote Sensing and GIS-based Approach for Assessing Groundwater Potential in West Medinipur District, West Bengal, India. Int. J. Remote Sens. 2009, 30, 231–250. [Google Scholar] [CrossRef]
- Chowdhury, A.; Jha, M.K.; Chowdary, V.M. Delineation of Groundwater Recharge Zones and Identification of Artificial Recharge Sites in West Medinipur District, West Bengal, Using RS, GIS and MCDM Techniques. Environ. Earth Sci. 2010, 59, 1209–1222. [Google Scholar] [CrossRef]
- Mukherjee, A.; Fryar, A.E. Deeper Groundwater Chemistry and Geochemical Modeling of the Arsenic Affected Western Bengal Basin, West Bengal, India. Appl. Geochemistry 2008, 23, 863–894. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004; ISBN 9781439833544. [Google Scholar]
- USGS—Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations. Available online: https://pubs.usgs.gov/tm/06/a43/ (accessed on 29 July 2022).
- Central Ground Water Board. Ground Water Year Book of West Bengal & Andaman & Nicobar Islands (2017-18) Technical Report: Series’ “D” No. 285; Central Ground Water Board: Kolkata, India, 2017. [Google Scholar]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef] [Green Version]
- Prasad, A.M.; Iverson, L.R.; Liaw, A. Newer Classification and Regression Tree Techniques: Bagging and Random Forests for Ecological Prediction. Ecosystems 2006, 9, 181–199. [Google Scholar] [CrossRef]
- Yoon, J. Forecasting of Real GDP Growth Using Machine Learning Models: Gradient Boosting and Random Forest Approach. Comput. Econ. 2021, 57, 247–265. [Google Scholar] [CrossRef]
- Tang, Z.; Mei, Z.; Liu, W.; Xia, Y. Identification of the Key Factors Affecting Chinese Carbon Intensity and Their Historical Trends Using Random Forest Algorithm. J. Geogr. Sci. 2020, 30, 743–756. [Google Scholar] [CrossRef]
- Svetnik, V.; Liaw, A.; Tong, C.; Christopher Culberson, J.; Sheridan, R.P.; Feuston, B.P. Random Forest: A Classification and Regression Tool for Compound Classification and QSAR Modeling. J. Chem. Inf. Comput. Sci. 2003, 43, 1947–1958. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, S.; Li, R.; Shahabi, H. Performance Evaluation of the GIS-Based Data Mining Techniques of Best-First Decision Tree, Random Forest, and Naïve Bayes Tree for Landslide Susceptibility Modeling. Sci. Total Environ. 2018, 644, 1006–1018. [Google Scholar] [CrossRef] [PubMed]
- Speiser, J.L.; Miller, M.E.; Tooze, J.; Ip, E. A Comparison of Random Forest Variable Selection Methods for Classification Prediction Modeling. Expert Syst. Appl. 2019, 134, 93–101. [Google Scholar] [CrossRef]
- Bindal, S.; Singh, C.K. Predicting Groundwater Arsenic Contamination: Regions at Risk in Highest Populated State of India. Water Res. 2019, 159, 65–76. [Google Scholar] [CrossRef]
- Fox, E.W.; Hill, R.A.; Leibowitz, S.G.; Olsen, A.R.; Thornbrugh, D.J.; Weber, M.H. Assessing the Accuracy and Stability of Variable Selection Methods for Random Forest Modeling in Ecology. Environ. Monit. Assess. 2017, 189, 1–20. [Google Scholar] [CrossRef]
- Liaw, A.; Wiener, M. Classification and Regression by RandomForest. R News 2002, 2, 18–22. [Google Scholar]
- US Department of Health and Human Services Federal Panel on Community Water Fluoridation. US Public Health Service Recommendation for Fluoride Concentration in Drinking Water for the Prevention of Dental Caries. Public Health Rep. 2015, 130, 318–331. [Google Scholar] [CrossRef] [Green Version]
- Geological Survey of India. Geological and Mineral Map of West Bengal; Government of India: Kolkata, India, 1999.
- Chen, J.; Wang, F.; Xia, X.; Zhang, L. Major Element Chemistry of the Changjiang (Yangtze River). Chem. Geol. 2002, 187, 231–255. [Google Scholar] [CrossRef]
- Wang, M.; Yang, L.; Li, J.; Liang, Q. Hydrochemical Characteristics and Controlling Factors of Surface Water in Upper Nujiang River, Qinghai-Tibet Plateau. Minerals 2022, 12, 490. [Google Scholar] [CrossRef]
- Gan, Y.; Zhao, K.; Deng, Y.; Liang, X.; Ma, T.; Wang, Y. Groundwater Flow and Hydrogeochemical Evolution in the Jianghan Plain, Central China. Hydrogeol. J. 2018, 26, 1609–1623. [Google Scholar] [CrossRef]
- Zhou, P.; Li, M.; Lu, Y. Hydrochemistry and Isotope Hydrology for Groundwater Sustainability of the Coastal Multilayered Aquifer System (Zhanjiang, China). Geofluids 2017, 2017, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Mallick, J.; Singh, C.; AlMesfer, M.; Kumar, A.; Khan, R.; Islam, S.; Rahman, A. Hydro-Geochemical Assessment of Groundwater Quality in Aseer Region, Saudi Arabia. Water 2018, 10, 1847. [Google Scholar] [CrossRef]
- Refat Nasher, N.M.; Humayan Ahmed, M. Groundwater Geochemistry and Hydrogeochemical Processes in the Lower Ganges-Brahmaputra-Meghna River Basin Areas, Bangladesh. J. Asian Earth Sci. X 2021, 6, 100062. [Google Scholar] [CrossRef]
- Ahmed, N.; Bodrud-Doza, M.; Islam, S.M.D.U.; Choudhry, M.A.; Muhib, M.I.; Zahid, A.; Hossain, S.; Moniruzzaman, M.; Deb, N.; Bhuiyan, M.A.Q. Hydrogeochemical Evaluation and Statistical Analysis of Groundwater of Sylhet, North-Eastern Bangladesh. Acta Geochim. 2019, 38, 440–455. [Google Scholar] [CrossRef]
- Gautam, A.; Rai, S.C.; Rai, S.P.; Ram, K. Sanny Impact of Anthropogenic and Geological Factors on Groundwater Hydrochemistry in the Unconfined Aquifers of Indo-Gangetic Plain. Phys. Chem. Earth 2022, 126, 103109. [Google Scholar] [CrossRef]
- Liu, Y.; Jin, M.; Ma, B.; Wang, J. Distribution and Migration Mechanism of Fluoride in Groundwater in the Manas River Basin, Northwest China. Hydrogeol. J. 2018, 26, 1527–1546. [Google Scholar] [CrossRef]
- Ling, Y.; Podgorski, J.; Sadiq, M.; Rasheed, H.; Eqani, S.A.M.A.S.; Berg, M. Monitoring and Prediction of High Fluoride Concentrations in Groundwater in Pakistan. Sci. Total Environ. 2022, 839, 156058. [Google Scholar] [CrossRef]
- Cao, H.; Xie, X.; Wang, Y.; Liu, H. Predicting Geogenic Groundwater Fluoride Contamination throughout China. J. Environ. Sci. 2022, 115, 140–148. [Google Scholar] [CrossRef]
- Kimambo, V.; Bhattacharya, P.; Mtalo, F.; Mtamba, J.; Ahmad, A. Fluoride Occurrence in Groundwater Systems at Global Scale and Status of Defluoridation – State of the Art. Groundw. Sustain. Dev. 2019, 9, 100223. [Google Scholar] [CrossRef]
- Ozsvath, D.L. Fluoride and Environmental Health: A Review. Rev. Environ. Sci. Biotechnol. 2009, 8, 59–79. [Google Scholar] [CrossRef]
- Pickering, W.F. The Mobility of Soluble Fluoride in Soils. Environ. Pollution. Ser. B Chem. Phys. 1985, 9, 281–308. [Google Scholar] [CrossRef]
- Saxena, V.K.; Ahmed, S. Inferring the Chemical Parameters for the Dissolution of Fluoride in Groundwater. Environ. Geol. 2003, 43, 731–736. [Google Scholar] [CrossRef]
- Addison, M.J.; Rivett, M.O.; Phiri, P.; Mleta, P.; Mblame, E.; Wanangwa, G.; Kalin, R.M. Predicting Groundwater Vulnerability to Geogenic Fluoride Risk: A Screening Method for Malawi and an Opportunity for National Policy Redefinition. Water 2020, 12, 3123. [Google Scholar] [CrossRef]
- Malakar, P.; Mukherjee, A.; Bhanja, S.N. Groundwater Recharge under Varied Land Use Regions in the Semi-Arid Parts of Western West Bengal, India. AGUFM 2016, 2016, H31C-1380. [Google Scholar]
- Carpenter, R. Factors Controlling the Marine Geochemistry of Fluorine. Geochim. Cosmochim. Acta 1969, 33, 1153–1167. [Google Scholar] [CrossRef]
- Cronin, S.J.; Manoharan, V.; Hedley, M.J.; Loganathan, P. Fluoride: A Review of Its Fate, Bioavailability, and Risks of Fluorosis in Grazed-Pasture Systems in New Zealand. New Zeal. J. Agric. Res. 2000, 43, 295–321. [Google Scholar] [CrossRef]
- Fan, X.; Parker, D.J.; Smith, M.D. Adsorption Kinetics of Fluoride on Low Cost Materials. Water Res. 2003, 37, 4929–4937. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.D.; Binning, P.; Stipp, S.L.S. Fluoride Removal by Calcite: Evidence for Fluorite Precipitation and Surface Adsorption. Environ. Sci. Technol. 2005, 39, 9561–9568. [Google Scholar] [CrossRef]
- Hamdi, N.; Srasra, E. Removal of Fluoride from Acidic Wastewater by Clay Mineral: Effect of Solid-Liquid Ratios. Desalination 2007, 206, 238–244. [Google Scholar] [CrossRef]
- Tor, A. Removal of Fluoride from an Aqueous Solution by Using Montmorillonite. Desalination 2006, 201, 267–276. [Google Scholar] [CrossRef]
- Diop, S.N.; Dieme, M.M.; Diallo, M.A.; Diawara, C.K. Fluoride Excess Removal from Brackish Drinking Water in Senegal by Using KSF and K10 Montmorillonite Clays. J. Water Resour. Prot. 2022, 14, 21–34. [Google Scholar] [CrossRef]
- Sreedevi, P.D.; Ahmed, S.; Made, B.; Ledoux, E.; Gandolfi, J.M. Association of Hydrogeological Factors in Temporal Variations of Fluoride Concentration in a Crystalline Aquifer in India. Environ. Geol. 2006, 50, 1–11. [Google Scholar] [CrossRef]
- Lin, D.; Jin, M.; Liang, X.; Zhan, H. Estimating Groundwater Recharge beneath Irrigated Farmland Using Environmental Tracers Fluoride, Chloride and Sul-Fate. Hydrogeol. J. 2013, 21, 1469–1480. [Google Scholar] [CrossRef]
- Venkateswarlu, P.; Armstrong, W.D.; Singer, L. Absorption of Fluoride and Chloride by Barley Roots. Plant Physiol. 1965, 40, 255–261. [Google Scholar] [CrossRef] [Green Version]
- Doull, J.; Boekelheide, K.; Farishian, B.G.; Isaacson, R.L.; Klotz, J.B.; Kumar, J.V.; Limeback, H.; Poole, C.; Puzas, J.E.; Reed, N.M.R.; et al. Fluoride in Drinking Water: A Scientific Review of EPA’s Standards; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Kaminsky, L.S.; Mahoney, M.C.; Leach, J.; Melius, J.; Miller, M.J. Fluoride: Benefits and Risks of Exposure. Crit. Rev. Oral Biol. Med. 1990, 1, 261–281. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Das, P.; Shaw, A.; Mukherjee, A.; Civil, W.; Petersen, J.O.; Gooddy, D.C.; Wakefield, O.; Finlayson, A.; Krishan, G.; et al. Deep Urban Groundwater Vulnerability in India Revealed through the Use of Emerging Organic Contaminants and Residence Time Tracers. Environ. Pollut. 2018, 240, 938–949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tripathy, S.S.; Bersillon, J.L.; Gopal, K. Removal of Fluoride from Drinking Water by Adsorption onto Alum-Impregnated Activated Alumina. Sep. Purif. Technol. 2006, 50, 310–317. [Google Scholar] [CrossRef]
- Islam, M.; Patel, R.K. Evaluation of Removal Efficiency of Fluoride from Aqueous Solution Using Quick Lime. J. Hazard. Mater. 2007, 143, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Trabucco, A.; Zomer, R. Global aridity index and potential evapotranspiration (ET0) climate database v2. CGIAR Consort. Spat. Inf. 2018, 10, m9. [Google Scholar] [CrossRef]
- Sivasankar, V.; Darchen, A.; Omine, K.; Sakthivel, R. Fluoride: A World Ubiquitous Compound, Its Chemistry, and Ways of Contamination. In Surface Modified Carbons as Scavengers for Fluoride from Water; Springer International Publishing: Cham, Swizerland, 2016; pp. 5–32. ISBN 9783319406862. [Google Scholar]
- Thapa, R.; Gupta, S.; Gupta, A.; Reddy, D.V.; Kaur, H. Geochemical and Geostatistical Appraisal of Fluoride Contamination: An Insight into the Quaternary Aquifer. Sci. Total Environ. 2018, 640–641, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Hengl, T.; De Jesus, J.M.; Heuvelink, G.B.M.M.; Gonzalez, M.R.; Kilibarda, M.; Blagotić, A.; Shangguan, W.; Wright, M.N.; Geng, X.; Bauer-Marschallinger, B.; et al. SoilGrids250m: Global Gridded Soil Information Based on Machine Learning. PLoS ONE 2017, 12, e0169748. [Google Scholar] [CrossRef] [Green Version]
- Waikar, M.L.; Nilawar, A.P. Identification of Groundwater Potential Zone Using Remote Sensing and GIS Technique. 2007, Volume 3297. Available online: https://www.researchgate.net/publication/271690625_Identification_of_Groundwater_Potential_Zone_using_Remote_Sensing_and_GIS_Technique (accessed on 12 August 2022).
- Lehner, B.; Verdin, K.; Jarvis, A. Technical Documentation Version 1.0; USGS Earth Resources Observation & Science: Sioux Falls, SD, USA, 2006. [Google Scholar]
- Karro, E.; Indermitte, E.; Saava, A.; Haamer, K.; Marandi, A. Fluoride Occurrence in Publicly Supplied Drinking Water in Estonia. Environ. Geol. 2006, 50, 389–396. [Google Scholar] [CrossRef]
- India-WRIS. Available online: https://indiawris.gov.in/wris/#/ (accessed on 12 August 2022).
- Gong, G.; Mattevada, S.; O’Bryant, S.E. Comparison of the Accuracy of Kriging and IDW Interpolations in Estimating Groundwater Arsenic Concentrations in Texas. Environ. Res. 2014, 130, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Salzman, S.A.; Allinson, G.; Stagnitti, F.; Hill, R.J.; Thwaites, L.; Ierodiaconou, D.; Carr, R.; Sherwood, J.; Versace, V. Adsorption and Desorption Characteristics of Fluoride in the Calcareous and Siliceous Sand Sheet Aquifers of South-West Victoria, Australia. WIT Trans. Ecol. Environ. 2008, 111, 159–174. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, A.; Verma, S.; Gupta, S.; Henke, K.R.; Bhattacharya, P. Influence of Tectonics, Sedimentation and Aqueous Flow Cycles on the Origin of Global Groundwater Arsenic: Paradigms from Three Continents. J. Hydrol. 2014, 518, 284–299. [Google Scholar] [CrossRef]
- Mukherjee, A.; Gupta, S.; Coomar, P.; Fryar, A.E.; Guillot, S.; Verma, S.; Bhattacharya, P.; Bundschuh, J.; Charlet, L. Plate Tectonics Influence on Geogenic Arsenic Cycling: From Primary Sources to Global Groundwater Enrichment. Sci. Total Environ. 2019, 683, 793–807. [Google Scholar] [CrossRef]
- del Pilar Alvarez, M.; Carol, E. Geochemical Occurrence of Arsenic, Vanadium and Fluoride in Groundwater of Patagonia, Argentina: Sources and Mobilization Processes. J. S. Am. Earth Sci. 2019, 89, 1–9. [Google Scholar] [CrossRef]
- Parvez, I.A.; Ram, A. Probabilistic Assessment of Earthquake Hazards in the Indian Subcontinent. Pure Appl. Geophys. 1999, 154, 23–40. [Google Scholar] [CrossRef]
- Jain, A.K.; Banerjee, D.M.; Kale, V.S. Tectonics of the Indian Subcontinent; Society of Earth Scientists Series; Springer International Publishing: Cham, Swizerland, 2020; ISBN 978-3-030-42844-0. [Google Scholar]
- Siebert, S.; Henrich, V.; Frenken, K.; Burke, J. Global Map of Irrigation Areas Version 5; Rheinische Friedrich-Wilhelms-University: Bonn, Germany; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013; Volume 2, pp. 1299–1327. [Google Scholar]




| Variables * | Pleistocene Alluvium (n = 12) | Residual Soils (n = 10) | CGC (n = 7) | |||
|---|---|---|---|---|---|---|
| Mean | (±SD) | Mean | (±SD) | Mean | (±SD) | |
| EC (µs/cm) 25 °C | 445 | 134 | 358 | 122 | 948 | 400 |
| Total Alkalinity as CaCO3 | 191 | 61 | 173 | 68 | 218 | 69 |
| Total Hardness as CaCO3 | 104.9 | 46 | 139.6 | 73.8 | 246.8 | 89.1 |
| Salinity | 219 | 66 | 175 | 59 | 476 | 214 |
| pH | 6.9 | 0.3 | 6.5 | 0.2 | 6.7 | 0.4 |
| Ca | 29.7 | 14.0 | 44.7 | 28.4 | 80.2 | 30.0 |
| Mg | 8.2 | 4.4 | 7.8 | 4.3 | 13.2 | 4.5 |
| Na | 13.4 | 4.8 | 16.2 | 10.8 | 76.7 | 73.4 |
| K | 0.9 | 0.9 | 1.2 | 1.3 | 1.5 | 0.9 |
| CO3 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| HCO3 | 232.7 | 75.1 | 211.8 | 82.8 | 266.4 | 84.7 |
| Cl | 28.3 | 36.3 | 14.7 | 9.8 | 127.0 | 71.7 |
| SO4 | 3.8 | 6.1 | 4.9 | 5.5 | 53.7 | 30.5 |
| NO3 | 5.1 | 12.0 | 7.7 | 7.6 | 44.6 | 40.1 |
| F | 0.3 | 0.2 | 0.2 | 0.1 | 0.4 | 0.2 |
| Area | Site ID | Depth (m) | Mean Sediment F (mg/kg) | WC 1 (g/g) | Mean Sediment F (mg/L) | Normalized F Inventory (kg/ha/m) | (Data from Malakar et al. [84]) Recharge Rate (mm/year) | Mean Groundwater F (mg/L) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| CMB 2 | WTF 3 | CGWB (Max) | This Study (Max) | |||||||
| PLEISTOCENE ALLUVIUM | PA1(G) | 7.9 | 1.1 | 0.01 | 137 | 148 | ||||
| PA2(F) | 6.1 | 2.1 | 0.03 | 179 | 212 | |||||
| PA3(A) | 7.9 | 1.1 | 0.03 | 38 | 149 | |||||
| PA1-1(G) | 6.7 | 0.1 | 0.05 | 1.2 | 6 | |||||
| PA1-2(A) | 7.3 | 0.0 | 0.06 | 0.4 | 3 | |||||
| PA1-3(F) | 6.1 | 1.7 | 168 | |||||||
| PA2-1(G) | 7.3 | 0.3 | 35 | |||||||
| PA2-2(F) | 6.1 | 0.4 | 35 | |||||||
| PA2-3(A) | 7.3 | 0.2 | 28 | |||||||
| PA3-1(F) | 7.3 | 0.6 | 68 | |||||||
| PA3-2(A) | 7.3 | 0.4 | 42 | |||||||
| PA3-3(G) | 7.3 | 0.3 | 41 | |||||||
| Agricultural Ø | 7.5 | 0.4 | 0.05 | 19 | 56 | 132 | ||||
| Forest Ø | 6.4 | 1.2 | 0.03 | 179 | 121 | 20 | ||||
| Grasslands Ø | 7.3 | 0.5 | 0.03 | 69 | 58 | 74 | ||||
| Mean | 7.1 | 0.7 | 0.04 | 71 | 78 | 44 | 49 | 0.6(4.3) | 0.3(0.6) | |
| CGC | CGC1 | 7.3 | 2.2 | 261 | 22 | 30 | 0.3(0.5) | 0.4(0.6) | ||
| RESIDUAL SOILS | RS1(A) | 6.1 | 0.4 | 0.02 | 24 | 43 | ||||
| RS2(G) | 7.3 | 0.6 | 0.01 | 64 | 73 | |||||
| RS3(F) | 7.9 | 0.3 | 0.02 | 19 | 38 | |||||
| RS1-1(A) | 7.9 | 2.0 | 0.01 | 189 | 267 | |||||
| RS1-2(G) | 8.5 | 0.6 | 0.01 | 68 | 80 | |||||
| RS1-3(F) | 9.1 | 1.0 | 0.01 | 169 | 144 | |||||
| RS2-1(G) | 9.1 | 1.5 | 0.01 | 294 | 225 | |||||
| RS2-2(F) | 8.5 | 2.3 | 0.01 | 523 | 325 | |||||
| RS2-3(A) | 8.5 | 2.0 | 0.01 | 417 | 277 | |||||
| RS3-1(G) | 7.3 | 4.4 | 0.02 | 198 | 535 | |||||
| RS3-2(A) | 7.9 | 2.2 | 0.03 | 85 | 286 | |||||
| RS3-3(F) | 7.3 | 2.5 | 0.02 | 128 | 300 | |||||
| Agricultural Ø | 7.6 | 1.7 | 0.02 | 179 | 218 | 132 | ||||
| Forest Ø | 8.2 | 1.5 | 0.01 | 210 | 202 | 20 | ||||
| Grasslands Ø | 8.1 | 1.8 | 0.01 | 156 | 228 | 74 | ||||
| Mean | 8.0 | 1.7 | 0.01 | 182 | 216 | 47 | 53 | 0.2(1.6) | 0.2(0.4) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aind, D.A.; Malakar, P.; Sarkar, S.; Mukherjee, A. Controls on Groundwater Fluoride Contamination in Eastern Parts of India: Insights from Unsaturated Zone Fluoride Profiles and AI-Based Modeling. Water 2022, 14, 3220. https://doi.org/10.3390/w14203220
Aind DA, Malakar P, Sarkar S, Mukherjee A. Controls on Groundwater Fluoride Contamination in Eastern Parts of India: Insights from Unsaturated Zone Fluoride Profiles and AI-Based Modeling. Water. 2022; 14(20):3220. https://doi.org/10.3390/w14203220
Chicago/Turabian StyleAind, David Anand, Pragnaditya Malakar, Soumyajit Sarkar, and Abhijit Mukherjee. 2022. "Controls on Groundwater Fluoride Contamination in Eastern Parts of India: Insights from Unsaturated Zone Fluoride Profiles and AI-Based Modeling" Water 14, no. 20: 3220. https://doi.org/10.3390/w14203220
APA StyleAind, D. A., Malakar, P., Sarkar, S., & Mukherjee, A. (2022). Controls on Groundwater Fluoride Contamination in Eastern Parts of India: Insights from Unsaturated Zone Fluoride Profiles and AI-Based Modeling. Water, 14(20), 3220. https://doi.org/10.3390/w14203220











