Characteristics of Bacterial Community in Pelteobagrus fulvidraco Integrated Multi-Trophic Aquaculture System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Construction of Aquaculture System
2.2. Culture Management
2.3. Sample Collection
2.4. Water Analysis
2.5. 16S rRNA Gene Amplicon Sequencing
2.6. Sequence Analysis
2.7. Bioinformatics Analysis
2.8. Statistic Analysis
3. Results
3.1. Changes in Water Quality
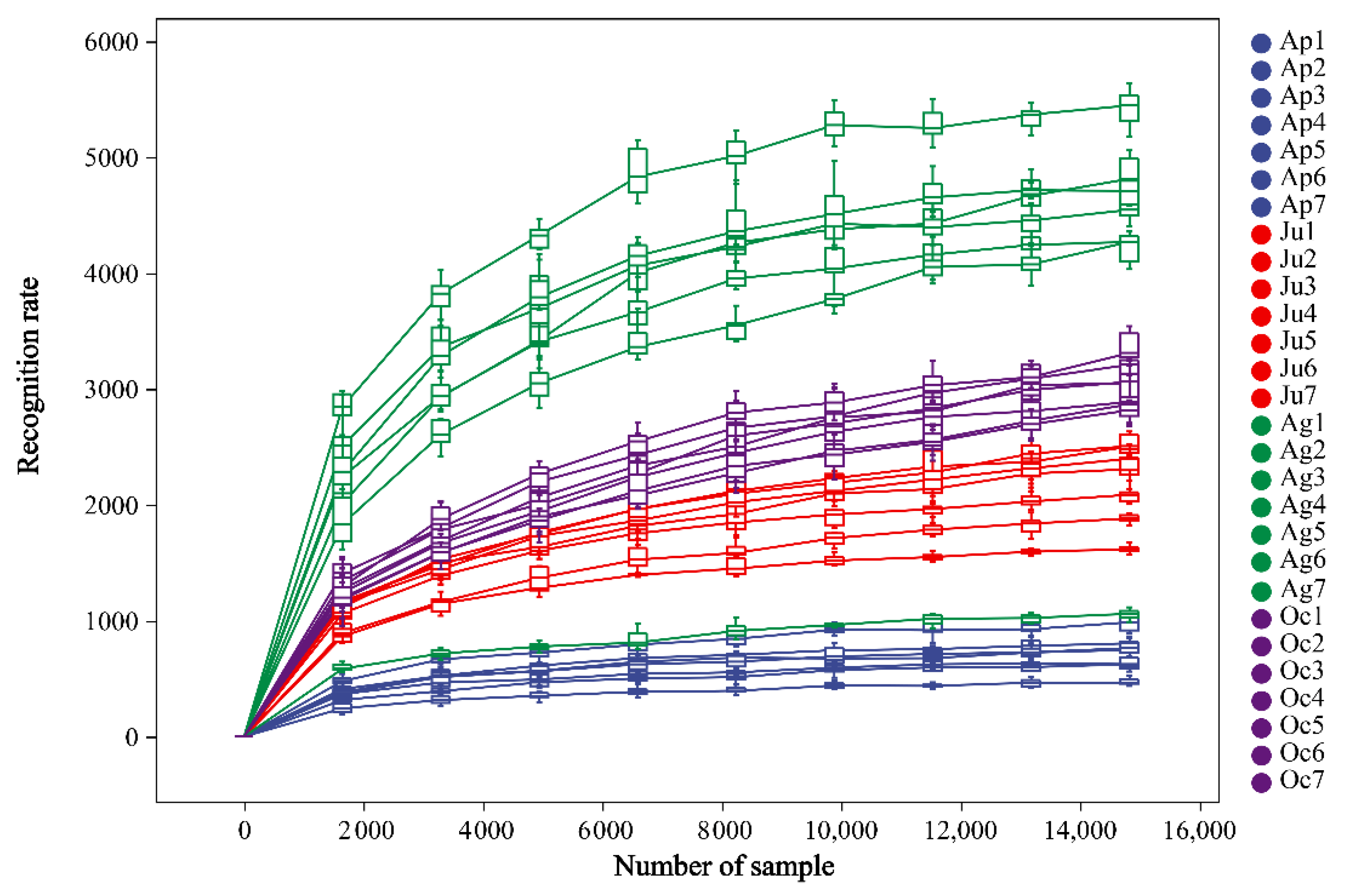
3.2. Seasonal Variation of Bacterial Community Richness and Diversity
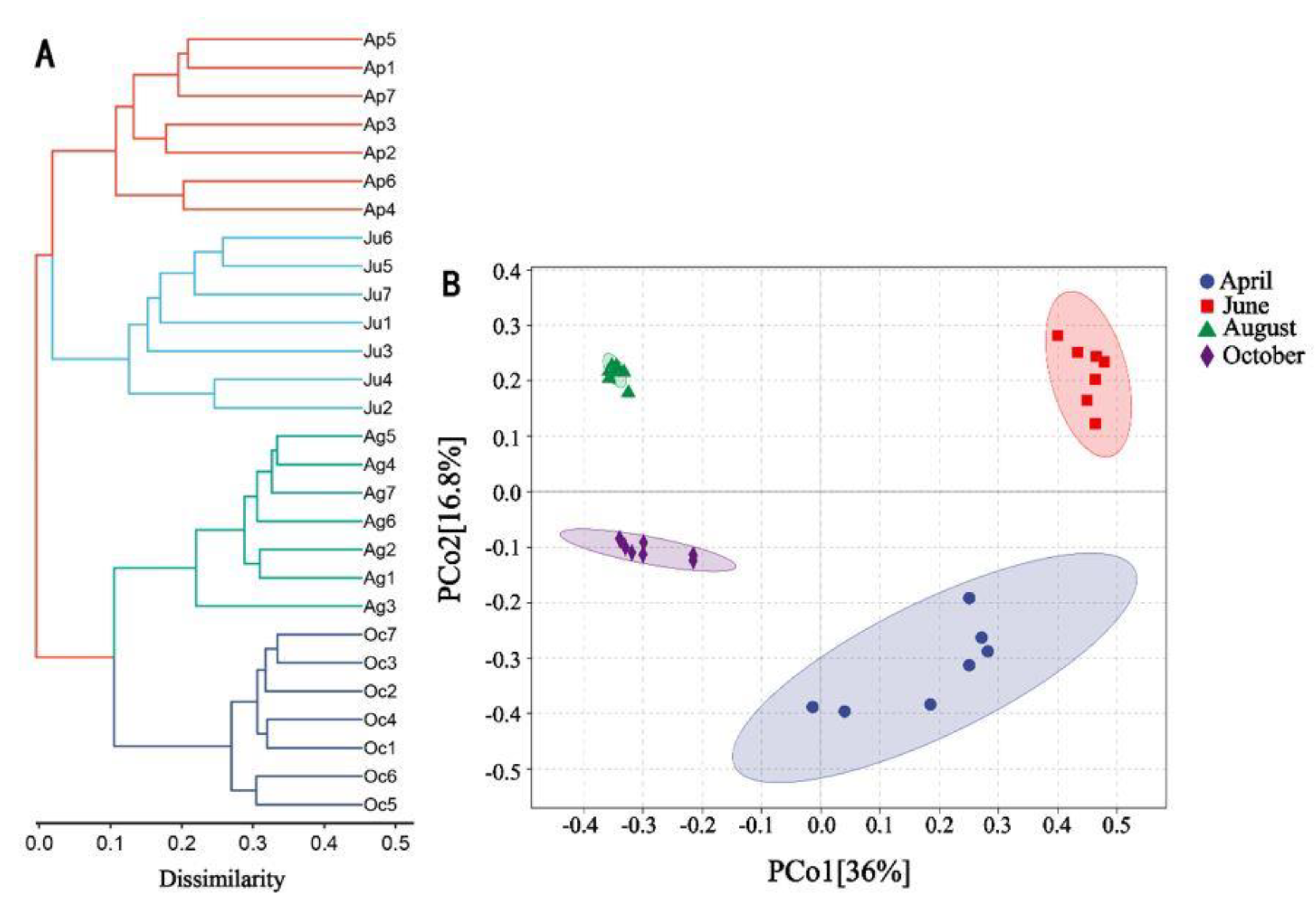
3.3. Differences in Microbial Communities
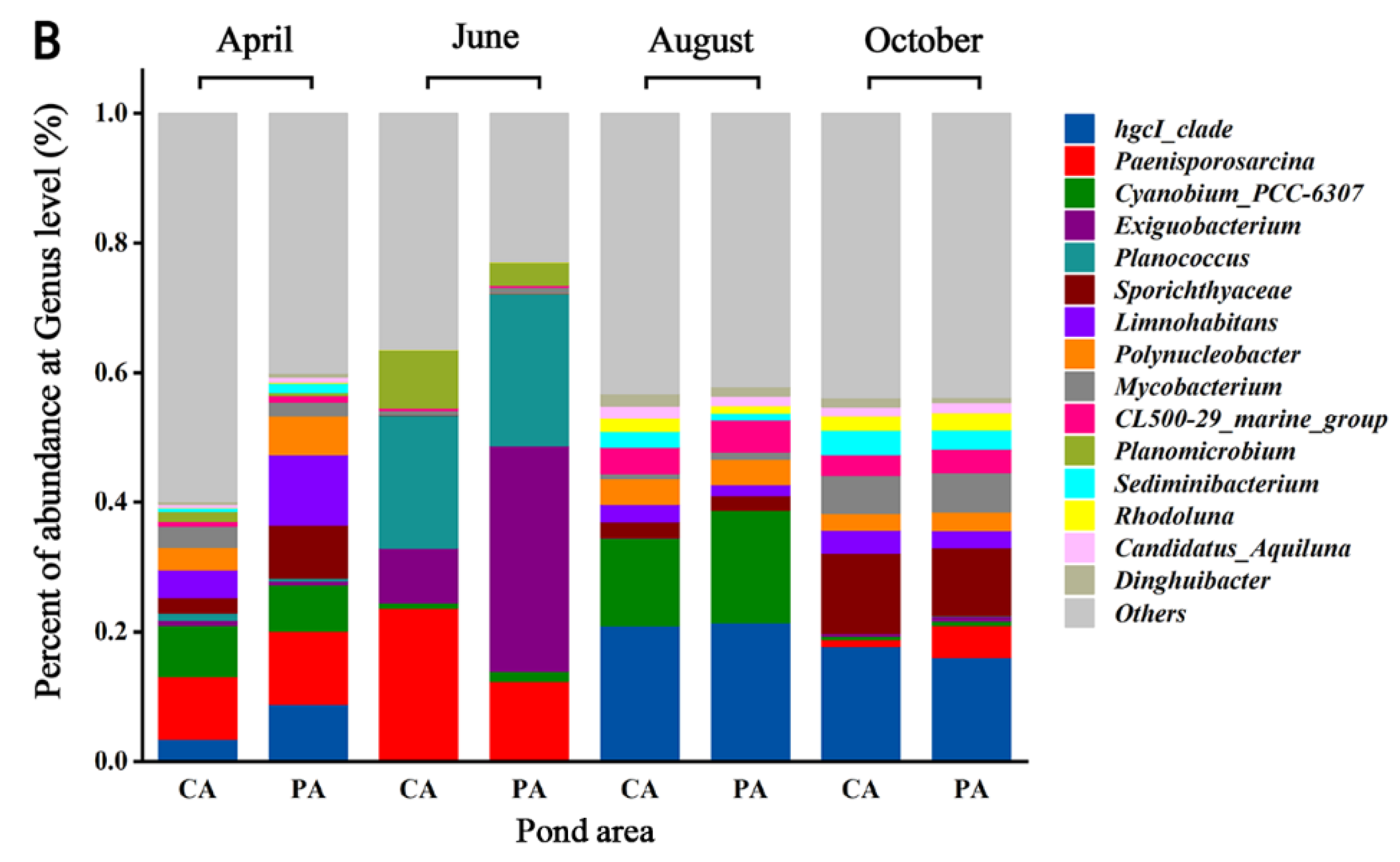
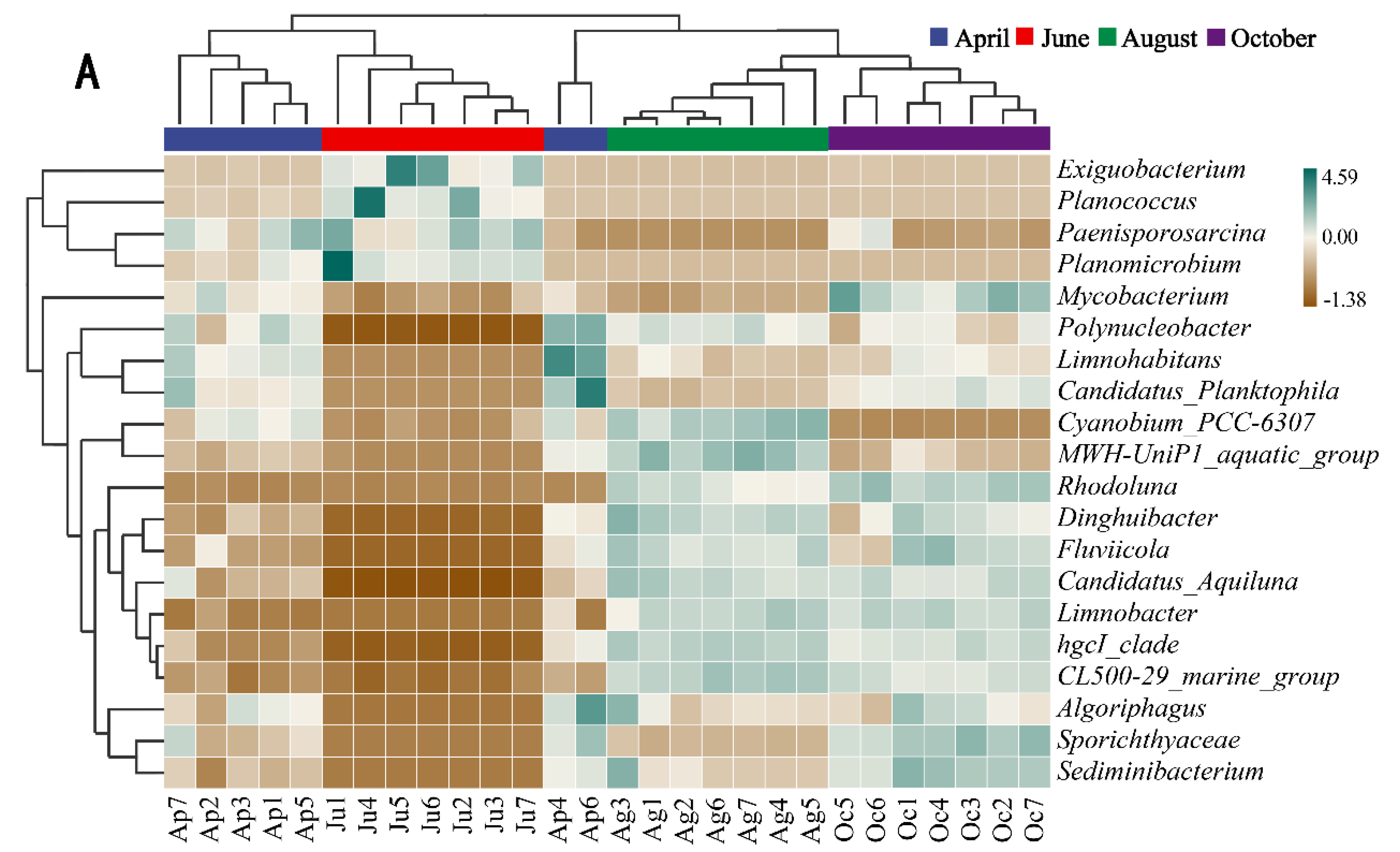
3.4. Seasonal Variation of Bacterial Community Composition
3.5. Relationship between Bacterial Community and Physicochemical Factors
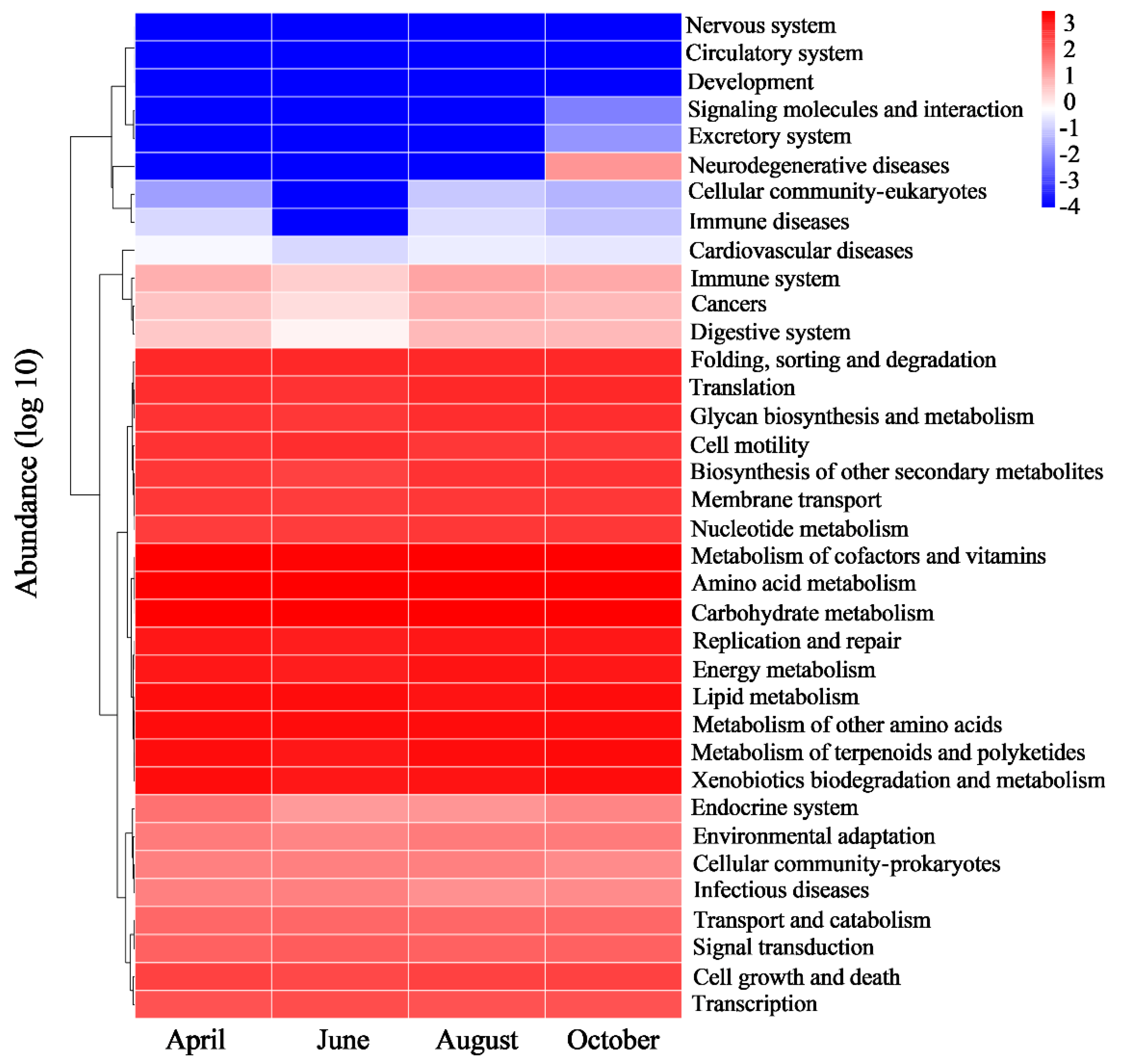
3.6. Bacterial Community Functional Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAMA (Fisheries Administration of the Ministry of agriculture and rural affairs of the people’s Republic of China); NTEC (National fisheries Technology Extension Center); CSF (China society of fisheries). China Fisheries Statistics Yearbook; China Agricultural Press: Beijing, China, 2021. (In Chinese) [Google Scholar]
- Liu, X.G.; Shao, Z.Y.; Cheng, G.F.; Lu, S.M.; Gu, Z.J.; Zhu, H.; Shen, H.Y.; Wang, J.; Chen, X.L. Ecological engineering in pond aquaculture: A review from the whole-process perspective in China. Rev. Aquacult. 2021, 13, 1060–1076. [Google Scholar] [CrossRef]
- Feng, J.; Tian, X.L.; Dong, S.L.; He, R.P.; Zhang, K.; Zhang, D.X.; Li, L.; Zhang, Q.Q.; Zhang, T. Comparative analysis of the energy fluxes and trophic structure of polyculture ecosystems of Portunus trituberculatus based on Ecopath model. Aquaculture 2018, 496, 185–196. [Google Scholar] [CrossRef]
- Tang, Q.S.; Fang, J.G.; Zhang, J.H.; Jiang, Z.J.; Liu, H.M. Impacts of multiple stress on coastal ocean ecosystems and Integrated Multi-trophic Aquaculture. Prog. Fish. Sci. 2013, 34, 11. [Google Scholar]
- Biswas, G.; Kumar, P.; Ghoshal, T.K.; Kailasam, M.; De, D.; Bera, A.; Mandal, B.; Sukumaran, K.; Vijayan, K.K. Integrated multi-trophic aquaculture (IMTA) outperforms conventional polyculture with respect to environmental remediation, productivity and economic return in brackishwater ponds. Aquaculture 2020, 516, 734626. [Google Scholar] [CrossRef]
- Liu, X.G. Pond Aquaculture Ecological Engineering; Agriculture Press: Beijing, China, 2018. (In Chinese) [Google Scholar]
- Zhu, H.; Cheng, S.P.; Wang, J.; Liu, X.G.; Lai, Q.F. Construction and operation evaluation of ecologically engineered cultivation system of saline-alkaline pond. Fish Mod. 2018, 4, 27–33. [Google Scholar]
- Azam, F.; Long, R.A. Oceangraphy: Sea snow microcosms. Nature 2001, 414, 495–498. [Google Scholar] [CrossRef]
- Cotner, J.B.; Biddanda, B.A. Small players, large role: Microbial influence on biogeochemical processes in pelagic aquatic ecosystems. Ecosystems 2002, 5, 105–121. [Google Scholar] [CrossRef]
- Yeo, S.K.; Huggett, M.J.; Eiler, A.; Rappé, M.S. Coastal Bacterioplankton Community Dynamics in Response to a Natural Disturbance. PLoS ONE 2013, 8, e56207. [Google Scholar] [CrossRef]
- Eswaran, R.; Khandeparker, L. Influence of salinity stress on bacterial community composition and β-glucosidase activity in a tropical estuary: Elucidation through microcosm experiments. Mar. Environ. Res. 2020, 159, 104997. [Google Scholar] [CrossRef]
- Kan, J.; Wang, K.; Chen, F. Temporal variation and detection limit of an estuarine bacterioplankton community analyzed by denaturing gradient gel electrophoresis (DGGE). Aquat. Microb. Ecol. 2006, 42, 7–18. [Google Scholar] [CrossRef] [Green Version]
- Cardona, E.; Gueguen, Y.; Magré, K.; Lorgeoux, B.; Piquemal, D.; Pierrat, F.; Noguier, F.; Saulnier, D. BacteriaI community characterization of water and inestine of the shrimp Litopenaeus stylirostris in a biofloc system. BMC. Microbiol. 2016, 16, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anthony, C.Y.; Eric, W.T. Within-and between-lake variability in the composition of bacterioplankton communities: Investigations using multiple spatial Scales. Appl. Environ. Microbiol. 2004, 70, 214–223. [Google Scholar]
- Vieira, R.P.; Gonzalez, A.M.; Cardoso, A.M.; Oliveira, D.N.; Albano, R.M.; Clementino, M.M.; Martins, O.B.; Paranhos, R. Relationships between bacterial diversity and environmental variables in a tropical marine environment, Rio de Janeiro. Environ. Microbiol. 2008, 10, 189–199. [Google Scholar] [CrossRef]
- Wever, A.D.; Muylaert, K.; Gucht, K.V.D.; Pirlot, S.; Cocquyt, C.; Descy, J.P.; Plisnier, P.P.; Vyverman, W. Bacterial community composition in Lake Tanganyika: Vertical and horizontal heterogeneity. Appl. Environ. Microbiol. 2005, 71, 5029–5037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moriarty, D.J.W. The role of microorganisms in aquaculture ponds. Aquaculture 1997, 151, 333–349. [Google Scholar] [CrossRef]
- SEPA (State Environmental Protection Administration). Water and Wastewater Monitoring and Analysis Methods, 4th ed.; China Environmental Science Press: Beijing, China, 2002. (In Chinese) [Google Scholar]
- Liu, C.S.; Zhao, D.F.; Ma, W.J.; Guo, Y.D.; Wang, A.J.; Wang, Q.L.; Lee, D.J. Denitrifying sulfide removal process on high-salinity wastewaters in the presence of Halomonas sp. Appl. Microbiol. Biotechnol. 2016, 100, 1421–1426. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.F.; Zhou, Y.Q.; Chen, Y.R.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef] [Green Version]
- Callahan, B.J.; Mcmurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [Green Version]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J. Introducing mothur: Open-Source, Platform-Independent, Community-Supported Software for Describing and Comparing Microbial Communities. Appl. Environ. Microbiol. 2009, 75, 7537. [Google Scholar] [CrossRef] [Green Version]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome. Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barberan, A.; Bates, S.T.; Casamayor, E.O.; Fierer, N. Using network analysis to explore co-occurrence patterns in soil microbial communities. ISME. J. 2012, 6, 343–351. [Google Scholar] [CrossRef]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef]
- Ministry of Agriculture of the People’s Republic of China. Requirements for Water Discharge from Freshwater Aquaculture Pond; Fisheries industry standard of the people’s Republic of China, SC/T 9101-2007; China Agriculture Press: Beijing, China, 2007. [Google Scholar]
- Zhang, X.L.; Wang, Q.; Zhang, G.Q.; Zhou, L.; Li, Y.F.; Zhang, Y.; Zhao, S.Y. Spatial variation of phytoplankton community structure of in-pond raceway system. South China Fish. Sci. 2021, 17, 36–45. [Google Scholar]
- Zhang, A.F.; Huang, J.F.; Zhang, H.X.; Tao, Z.Y.; Yu, Z.J.; Hu, Z.G. Changes in water quality and density of plankton in a juvenile yellow catfish rearing pond. Chin. J. Fish. 2021, 34, 71–75. [Google Scholar]
- Shpigel, M.; Shauli, L.; Odintsov, V.; Ben-Ezra, D.; Neori, A.; Guttman, L. The sea urchin, Paracentrotus lividus, in an Integrated MultiTrophic Aquaculture (IMTA) system with fish (Sparus aurata) and seaweed (Ulva lactuca): Nitrogen partitioning and proportional configurations. Aquaculture 2018, 490, 260–269. [Google Scholar] [CrossRef]
- Zhang, H.J.; Guo, Z.X.; Zhao, D.Y.; He, F. Variation of water quality in recirculating aquaculture system based on paddy wetland. Environ. Sci. Technol. 2020, 43, 159–164. [Google Scholar]
- Feng, F.; Fang, T.; Liu, J.T. Vertical distribution of species of nitrogen and phosphorus in the sediments of lake Donghu. Environ. Sci. 2006, 27, 1078–1082. [Google Scholar]
- Yi, S.M.; Dai, D.W.; Wu, H.; Chai, S.T.; Liu, S.J.; Meng, Q.X.; Zhou, Z.M. Dietary concentrate-to-forage ratio affects rumen bacterial community composition and metabolome of yaks. Front. Nutr. 2022, 9, 927206. [Google Scholar] [CrossRef]
- Tian, X.L.; Zheng, Y.Y.; Liu, B.J.; Wang, F.; Dong, S.L. Abundance dynamics and community functional diversity of bacteria in Grass carp polyculture systems. Period. Ocean. Univ. China 2012, 42, 19–27. [Google Scholar]
- Hoang, M.N.; Nguyen, P.N.; Bossier, P. Water quality, animal performance, nutrient budgets and microbial community in the biofloc-based polyculture system of white shrimp, Litopenaeus vannamei and gray mullet, Mugil cephalus. Aquaculture 2019, 515, 734610. [Google Scholar] [CrossRef]
- Wang, Y.T.; Li, B.; Wang, H.H.; Wu, F.L.; Zhang, L. Bacterial diversity of pond water and sediment in different culture systems of yellow catfish (Pelteobagrus fulvidraco) by Miseq sequencing. Acta Microbiol. Sin. 2020, 44, 781–789. [Google Scholar]
- Mancuso, C.; Franzmann, P.; Burton, H.; Nichols, P. Microbial community structure and biomass estimates of a methanogenic Antarctic Lake ecosystem as determined by phospholipid analyses. Microb. Ecol. 1990, 19, 73–95. [Google Scholar] [CrossRef] [PubMed]
- Stefano, A.; Si, T. Stability criteria for complex ecosystems. Nature 2012, 483, 205–208. [Google Scholar]
- Cheng, Y.Y. Research on Microbial Community Ecology in Sediment of a Pond Cultured Mainly with Grass Carp (Ctenopharyngodon indellus); Graduate School of Chinese Academy of Sciences: Beijing, China, 2011. [Google Scholar]
- Zhao, X.G. Influence of the Cultured Odontobutis Obscurus to the Microbial Community, Water Quality and Rice Traits in Rice-Fish System; Shanghai Ocean University: Shanghai, China, 2016. [Google Scholar]
- Fan, L.M.; Barry, K.; Hu, G.D.; Meng, S.L.; Chao, S.; Qiu, L.P.; Zheng, Y.; Wu, W.; Qu, J.H.; Chen, J.Z.; et al. Characterizing bacterial communities in tilapia pond surface sediment and their responses to pond differences and temporal variations. World J. Microbiol. Biotechnol. 2017, 33, 1. [Google Scholar] [CrossRef]
- Salcher, M.M. Same same but different: Ecological niche partitioning of planktonic freshwater prokaryotes. J. Limnol. 2014, 73, 74–87. [Google Scholar] [CrossRef] [Green Version]
- Ameryk, A.; Kownacka, J.; Zalewski, M.; Piwosz, K. Typical freshwater and marine bacterial lineages dynamics at salinity between 0 and 4 in the Vistula Lagoon. Estuar. Coast. Shelf Sci. 2021, 250, 107100. [Google Scholar] [CrossRef]
- Du, Y.Y.; Lou, Z.Y.; Zhang, Y.P.; Li, W.; Wang, T. Effect of cage culture on community structure and influencing factors of bacterioplankton in Liujiaxia Reservoir. Fresh. Fish. 2018, 48, 11–16. [Google Scholar]
- Wang, S.D.; Li, X.; Zhang, M.Z.; Jiang, H.B.; Wang, R.X.; Qian, Y.X.; Li, M. Ammonia stress disrupts intestinal microbial community and amino acid metabolism of juvenile yellow catfish (Pelteobagrus fulvidraco). Ecotoxicol. Environ. Saf. 2021, 227, 112932. [Google Scholar] [CrossRef]
- Li, Q.X.; Cao, L.X.; Tan, H.M.; Sun, J.; Cui, Y.; Xu, R.L. Bacterial communities in the waterlogged wooden cultural relics from the “Nanhai No.1” shipwreck. Acta Microbiol. Sin. 2018, 58, 1439–1452. [Google Scholar]
- Hu, Y.X.; Peng, Y.; Li, R.W.; Huang, J.; Zhou, Z.; Hu, S.; Wang, Y.C.; Qiu, G.S. Plankton diversity and community characteristics in Danjiangkou Reservoir based on environmental DNA metabarcoding. J. Lake Sci. 2021, 33, 1650–1659. [Google Scholar]
- Reed, H.E.; Martiny, J.B. Microbial composition affects the functioning of estuarine sediments. ISME J. 2013, 7, 868–879. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.Y.; Gou, H.L.; Gong, Y.H.; Ji, Y.T.; Su, X.L. Seasonal dynamics of the coastal bacterioplankton at intensive fish-farming areas of the Yellow Sea, China revealed by high-throughput sequencing. Mar. Pollut. Bull. 2019, 139, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Dahle, S.W.; Bakke, I.; Birkeland, M.; Nordøy, K.; Dalum, A.S.; Attramadal, K.J.K. Production of lumpfish (Cyclopterus lumpus L.) in RAS with distinct water treatments: Effects on fish survival, growth, gill health and microbial communities in rearing water and biofilm. Aquaculture 2020, 522, 735097. [Google Scholar] [CrossRef]
- Venkat, H.K.; Sahu, N.P.; Jain, K.K. Effect of feeding Lactobacillus-based probiotics on the gut micro-flora, growth and survival of postlarvae of Macrobrachium rosenbergii (de Man). Aquac. Res. 2004, 35, 501–507. [Google Scholar] [CrossRef]
- Bentzon-Tilia, M.; Gram, L. Monitoring and managing microbes in aquaculture-Towards a sustainable industry. Microb. Biotechnol. 2016, 9, 576–584. [Google Scholar] [CrossRef] [Green Version]
- Zelezniak, A.; Andrejev, S.; Ponomarova, O.; Mende, D.R.; Bork, P.; Patil, K.R. Metabolic dependencies drive species co-occurrence in diverse microbial communities. Proc. Natl. Acad Sci. USA 2015, 112, 6449–6454. [Google Scholar] [CrossRef] [Green Version]
- Perez-Garcia, O.; Lear, G.; Singhal, N. Metabolic network modeling of microbial interactions in natural and engineered environmental systems. Front. Microbiol. 2016, 7, 673. [Google Scholar] [CrossRef]




| Area | Site | April | June | August | October |
|---|---|---|---|---|---|
| CA | I | Ap1 | Ju1 | Ag1 | Oc1 |
| Ⅱ | Ap2 | Ju2 | Ag2 | Oc2 | |
| Ⅲ | Ap3 | Ju3 | Ag3 | Oc3 | |
| PA | Ⅳ | Ap4 | Ju4 | Ag4 | Oc4 |
| Ⅴ | Ap5 | Ju5 | Ag5 | Oc5 | |
| Ⅵ | Ap6 | Ju6 | Ag6 | Oc6 | |
| Ⅶ | Ap7 | Ju7 | Ag7 | Oc7 |
| Time | Area | T (°C) | DO (mg/L) | TN (mg/L) | -N (mg/L) | -N (μg/L) | -N (mg/L) | TP (mg/L) | -P (μg/L) | N:P |
|---|---|---|---|---|---|---|---|---|---|---|
| April | CA | 18.63 ± 0.05 A | 12.94 ± 0.80 A | 1.84 ± 0.09 A | 0.34 ± 0.13 A | 3.85 ± 2.70 | 0.95 ± 0.21 αA | 0.14 ± 0.05 | 1.42 ± 1.20 | 14.12 ± 3.84 AB |
| PA | 18.45 ± 0.11 A | 12.39 ± 3.00 | 2.40 ± 1.32 | 0.39 ± 0.14 AB | 3.3 ± 1.37 B | 0.43 ± 0.20 βAB | 0.11 ± 0.01 AB | 0.99 ± 0.74 A | 14.84 ± 1.48 | |
| June | CA | 26.73 ± 0.09 B | 12.59 ± 0.81 A | 1.47 ± 0.01 B | 0.05 ± 0.00 B | 6.68 ± 1.40 α | 0.10 ± 0.05 B | 0.14 ± 0.01 | 3.93 ± 1.69 | 10.27 ± 0.75 B |
| PA | 26.65 ± 0.09 B | 12.08 ± 2.03 | 1.55 ± 0.34 | 0.04 ± 0.01 B | 3.99 ± 0.56 βB | 0.13 ± 0.06 B | 0.12 ± 0.01 A | 3.78 ± 1.70 A | 13.00 ± 2.42 | |
| August | CA | 31.23 ± 0.05 C | 9.21 ± 0.52 B | 1.85 ± 0.10 A | 0.53 ±0.02 A | 6.39 ± 2.48 | 0.94 ± 0.03 αA | 0.12 ± 0.01 α | 2.65 ± 1.00 | 16.06 ± 1.40 AB |
| PA | 31.3 ± 0.00 C | 9.68 ± 0.87 | 1.77 ± 0.09 | 0.58 ± 0.24 A | 7.86 ± 1.86 A | 0.73 ± 0.07 βA | 0.09 ± 0.01 βAB | 1.68 ± 0.38 A | 19.60 ± 1.88 | |
| October | CA | 21.23 ± 0.055 D | 9.98 ± 0.36 B | 1.71 ± 0.04 A | 0.39 ± 0.07 A | 4.21 ± 1.50 | 0.48 ± 0.08 αB | 0.08 ± 0.02 | 24.00± 16.74 | 24.02 ± 6.23 A |
| PA | 21.28 ± 0.08 D | 9.76 ± 1.37 | 1.72 ± 0.16 | 0.38 ± 0.06 AB | 2.59 ±0. 64 B | 0.38 ± 0.17 βAB | 0.08 ± 0.02 B | 27.90 ± 12.81 B | 22.89 ± 5.08 |
| Time | Area | Chao1 | Shannon |
|---|---|---|---|
| April | CA | 3.78 × 103 ± 1.94 × 103 | 8.48 ± 1.32 a |
| PA | 4.43 × 103 ± 2.01 × 102 A | 9.49 ± 0.53 a | |
| June | CA | 6.87 × 102 ± 78.9 | 5.10 ± 0.47 b |
| PA | 7.44 × 102 ± 1.81 × 102 B | 5.36 ± 0.86 b | |
| August | CA | 2.06 × 103 ± 3.55 × 102 | 8.41 ± 0.21 a |
| PA | 2.27 × 103 ± 2.32 × 102 C | 8.59 ± 0.14 a | |
| October | CA | 3.03 × 103 ± 1.53 × 102 | 8.46 ± 0.11 a |
| PA | 3.03 × 103 ± 2.11 × 102 D | 8.34 ± 0.16 a |
| Number | Name |
|---|---|
| ko00100 | Steroid biosynthesis |
| ko00120 | Primary bile acid biosynthesis |
| ko00121 | Secondary bile acid biosynthesis |
| ko00140 | Steroid hormone biosynthesis |
| ko00196 | Photosynthesis—antenna proteins |
| ko00253 | Tetracycline biosynthesis |
| ko00281 | Geraniol degradation |
| ko00364 | Fluorobenzoate degradation |
| ko00510 | N-Glycan biosynthesis |
| ko00523 | Polyketide sugar unit biosynthesis |
| ko00540 | Lipopolysaccharide biosynthesis |
| ko00621 | Dioxin degradation |
| ko00622 | Xylene degradation |
| ko00626 | Naphthalene degradation |
| ko00633 | Nitrotoluene degradation |
| ko00906 | Carotenoid biosynthesis |
| ko00909 | Sesquiterpenoid biosynthesis |
| ko00941 | Flavonoid biosynthesis |
| ko00943 | Isoflavonoid biosynthesis |
| ko00965 | Betalain biosynthesis |
| ko00980 | Metabolism of xenobiotics by cytochrome P450 |
| ko01055 | Biosynthesis of vancomycin group antibiotics |
| ko01057 | Biosynthesis of type II polyketide products |
| ko02060 | Phosphotransferase system (PTS) |
| ko03040 | Spliceosome |
| ko03050 | Proteasome |
| ko03320 | PPAR signaling pathway |
| ko03450 | Non-homologous end-joining |
| ko04142 | Lysosome |
| ko04144 | Endocytosis |
| ko04145 | Phagosome |
| ko04621 | NOD-like receptor signaling pathway |
| ko04974 | Protein digestion and absorption |
| ko05010 | Alzheimer’s disease |
| ko05100 | Bacterial invasion of epithelial cells |
| ko05110 | Vibrio cholerae infection |
| ko05145 | Toxoplasmosis |
| ko05146 | Amoebiasis |
| ko05150 | Staphylococcus aureus infection |
| ko05322 | Systemic lupus erythematosus |
| ko05410 | Hypertrophic cardiomyopathy (HCM) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.-X.; Liu, X.-G.; Lu, M.; Zhou, R.-F.; Sun, Z.-Y.; Xiao, S.-W. Characteristics of Bacterial Community in Pelteobagrus fulvidraco Integrated Multi-Trophic Aquaculture System. Water 2022, 14, 3192. https://doi.org/10.3390/w14203192
Zhao Y-X, Liu X-G, Lu M, Zhou R-F, Sun Z-Y, Xiao S-W. Characteristics of Bacterial Community in Pelteobagrus fulvidraco Integrated Multi-Trophic Aquaculture System. Water. 2022; 14(20):3192. https://doi.org/10.3390/w14203192
Chicago/Turabian StyleZhao, Yu-Xi, Xing-Guo Liu, Ming Lu, Run-Feng Zhou, Zhao-Yun Sun, and Shu-Wen Xiao. 2022. "Characteristics of Bacterial Community in Pelteobagrus fulvidraco Integrated Multi-Trophic Aquaculture System" Water 14, no. 20: 3192. https://doi.org/10.3390/w14203192
APA StyleZhao, Y.-X., Liu, X.-G., Lu, M., Zhou, R.-F., Sun, Z.-Y., & Xiao, S.-W. (2022). Characteristics of Bacterial Community in Pelteobagrus fulvidraco Integrated Multi-Trophic Aquaculture System. Water, 14(20), 3192. https://doi.org/10.3390/w14203192








