The Effect of Trophic Modes on Biomass and Lipid Production of Five Microalgal Strains
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microalgal Strains and Growth Conditions
2.2. Analyses
2.2.1. Biomass Optical Density and Ash-Free Dry Weight (AFDW)
2.2.2. Glucose Concentration
2.2.3. Lipid Extraction and Quantification
2.2.4. Analysis of Fatty Acid Composition
3. Results and Discussion
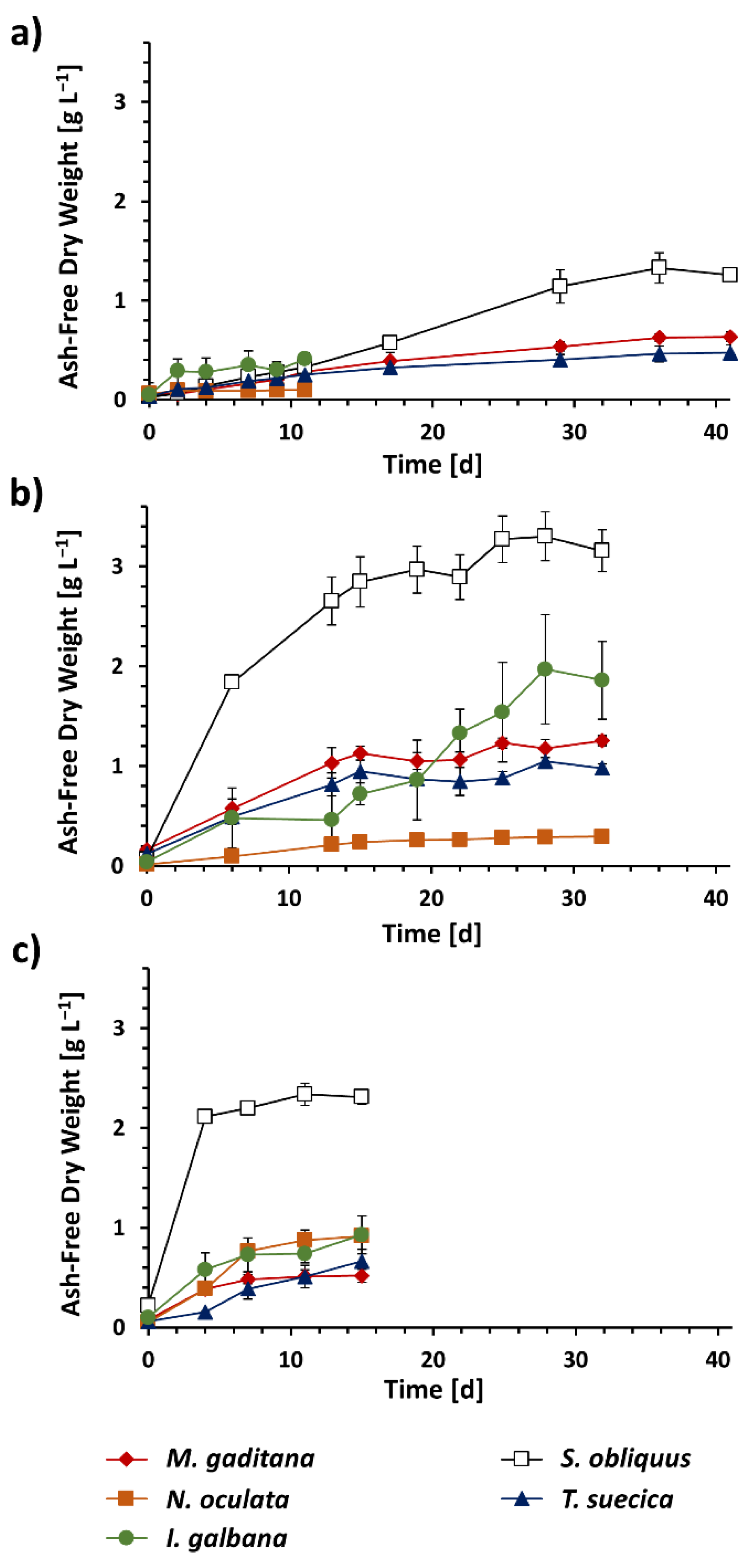
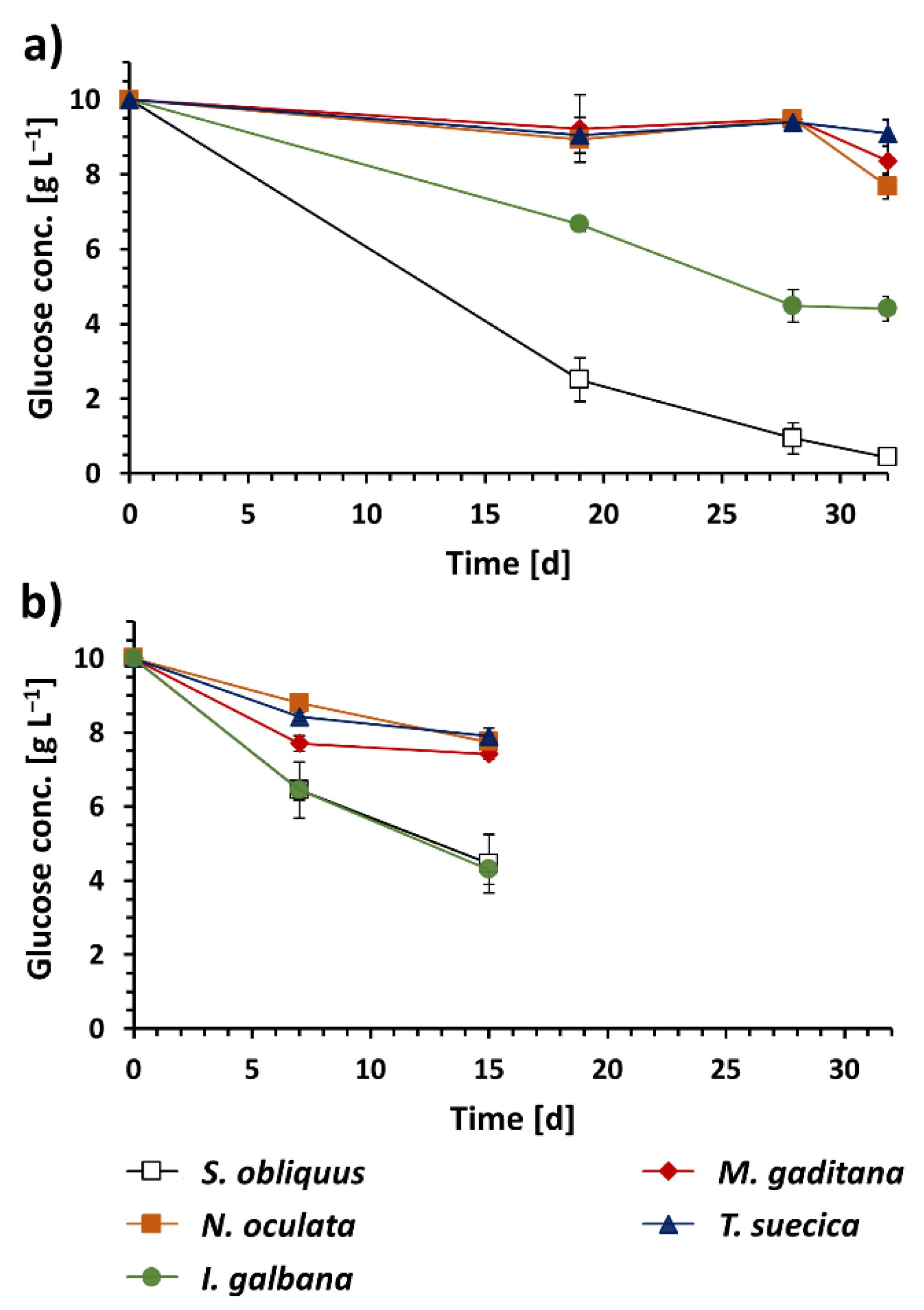
3.1. Algal Biomass Production under Different Trophic Conditions
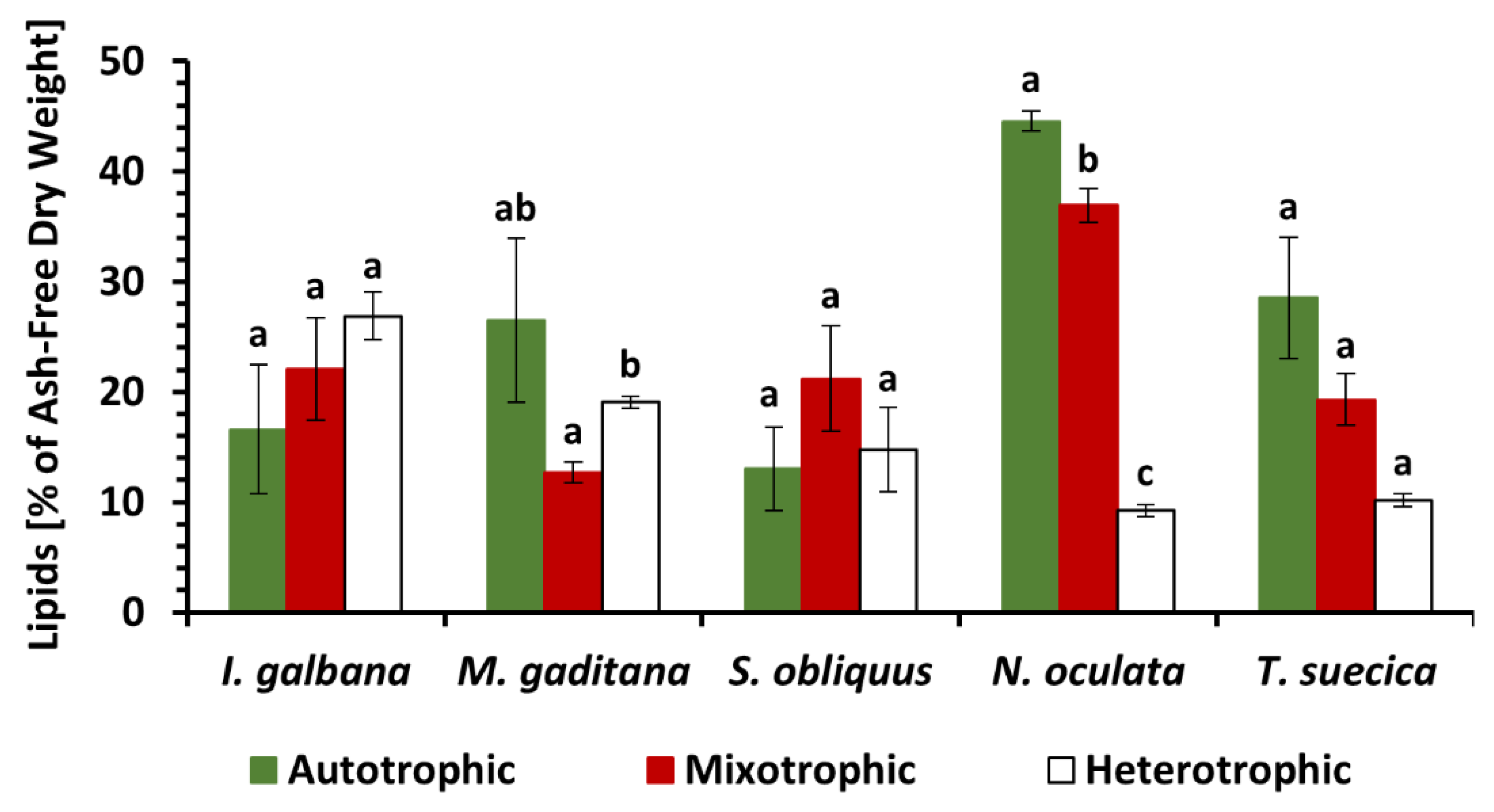
3.2. Lipid Content and Fatty Acid Composition under Autotrophic, Mixotrophic and Heterotrophic Nutrition
3.3. Implications for the Development of Algal Biorefineries
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Punia, S.; Sandhu, K.S.; Siroha, A.K.; Dhull, S.B. Omega 3-metabolism, absorption, bioavailability and health benefits–A review. PharmaNutrition 2019, 10, 100162. [Google Scholar] [CrossRef]
- Lu, Q.; Li, H.; Xiao, Y.; Liu, H. A state-of-the-art review on the synthetic mechanisms, production technologies, and practical application of polyunsaturated fatty acids from microalgae. Algal Res. 2021, 55, 102281. [Google Scholar] [CrossRef]
- Kothri, M.; Mavrommati, M.; Elazzazy, A.M.; Baeshen, M.N.; Moussa, T.A.A.; Aggelis, G. Microbial sources of polyunsaturated fatty acids (PUFAs) and the prospect of organic residues and wastes as growth media for PUFA-producing microorganisms. FEMS Microbiol. Lett. 2020, 367, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cvitković, D.; Dragović-Uzelac, V.; Dobrinčić, A.; Čož-Rakovac, R.; Balbino, S. The effect of solvent and extraction method on the recovery of lipid fraction from Adriatic Sea macroalgae. Algal Res. 2021, 56, 102291. [Google Scholar] [CrossRef]
- Suganya, T.; Varman, M.; Masjuki, H.H.; Renganathan, S. Macroalgae and microalgae as a potential source for commercial applications along with biofuels production: A biorefinery approach. Renew. Sustain. Energy Rev. 2016, 55, 909–941. [Google Scholar] [CrossRef]
- Sosa-Hernández, J.E.; Romero-Castillo, K.D.; Parra-Arroyo, L.; Aguilar-Aguila-Isaías, M.A.; García-Reyes, I.E.; Ahmed, I.; Parra-Saldivar, R.; Bilal, M.; Iqbal, H.M.N. Mexican microalgae biodiversity and state-of-the-art extraction strategies to meet sustainable circular economy challenges: High-value compounds and their applied perspectives. Mar. Drugs 2019, 17, 174. [Google Scholar] [CrossRef] [Green Version]
- Brennan, L.; Owende, P. Biofuels from microalgae-A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Silva, C.; Soliman, E.; Cameron, G.; Fabiano, L.A.; Seider, W.D.; Dunlop, E.H.; Coaldrake, A.K. Commercial-scale biodiesel production from algae. Ind. Eng. Chem. Res. 2014, 53, 5311–5324. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, X.; Liu, J. Screening of Isochrysis strains for simultaneous production of docosahexaenoic acid and fucoxanthin. Algal Res. 2019, 41, 101545. [Google Scholar] [CrossRef]
- Patel, A.K.; Choi, Y.Y.; Sim, S.J. Emerging prospects of mixotrophic microalgae: Way forward to sustainable bioprocess for environmental remediation and cost-effective biofuels. Bioresour. Technol. 2020, 300, 122741. [Google Scholar] [CrossRef]
- Valdovinos-García, E.M.; Barajas-Fernández, J.; de los Ángeles Olán-Acosta, M.; Petriz-Prieto, M.A.; Guzmán-López, A.; Bravo-Sánchez, M.G. Techno-Economic Study of CO2 Capture of a Thermoelectric Plant Using Microalgae (Chlorella vulgaris) for Production of Feedstock for Bioenergy. Energies 2020, 13, 413. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Mi, Y.; Zhao, C.; Wei, Q. A comprehensive review on carbon source effect of microalgae lipid accumulation for biofuel production. Sci. Total Environ. 2021, 806, 151387. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.; Pereira, H.; Campos, J.; Marques, A.; Varela, J.; Silva, J. Heterotrophy as a tool to overcome the long and costly autotrophic scale-up process for large scale production of microalgae. Sci. Rep. 2019, 9, 13935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zili, F.; Bouzidi, N.; Ammar, J.; Zakhama, W.; Ghoul, M.; Sayadi, S.; Ben Ouada, H. Mixotrophic cultivation promotes growth, lipid productivity, and PUFA production of a thermophilic Chlorophyta strain related to the genus Graesiella. J. Appl. Phycol. 2017, 29, 35–43. [Google Scholar] [CrossRef]
- Jareonsin, S.; Pumas, C. Advantages of Heterotrophic microalgae as a host for phytochemicals production. Front. Bioeng. Biotechnol. 2021, 9, 628597. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.J.; Joun, J.; Hong, M.E.; Patel, A.K. Split mixotrophy: A novel cultivation strategy to enhance the mixotrophic biomass and lipid yields of Chlorella protothecoides. Bioresour. Technol. 2019, 291, 121820. [Google Scholar] [CrossRef]
- López, G.; Yate, C.; Ramos, F.A.; Cala, M.P.; Restrepo, S.; Baena, S. Production of polyunsaturated fatty acids and lipids from autotrophic, mixotrophic and heterotrophic cultivation of Galdieria sp. strain USBA-GBX-832. Sci. Rep. 2019, 9, 10791. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yin, J.; Ge, Y.; Han, H.; Liu, M.; Gao, F. Improved lipid productivity of Scenedesmus obliquus with high nutrient removal efficiency by mixotrophic cultivation in actual municipal wastewater. Chemosphere 2021, 285, 131475. [Google Scholar] [CrossRef]
- Penhaul Smith, J.K.; Hughes, A.D.; McEvoy, L.; Thornton, B.; Day, J.G. The carbon partitioning of glucose and DIC in mixotrophic, heterotrophic and photoautotrophic cultures of Tetraselmis suecica. Biotechnol. Lett. 2021, 43, 729–743. [Google Scholar] [CrossRef]
- Choi, H.J.; Lee, S.M. Biomass and oil content of microalgae under mixotrophic conditions. Environ. Eng. Res. 2015, 20, 25–32. [Google Scholar] [CrossRef]
- Mata, T.M.; Melo, A.C.; Meireles, S.; Mendes, A.M.; Martins, A.A.; Caetano, N.S. Potential of microalgae Scenedesmus obliquus grown in brewery wastewater for biodiesel production. Chem. Eng. Trans. 2013, 32, 901–906. [Google Scholar] [CrossRef]
- Girard, J.M.; Roy, M.L.; Hafsa, M.B.; Gagnon, J.; Faucheux, N.; Heitz, M.; Tremblay, R.; Deschênes, J.S. Mixotrophic cultivation of green microalgae Scenedesmus obliquus on cheese whey permeate for biodiesel production. Algal Res. 2014, 5, 241–248. [Google Scholar] [CrossRef]
- Han, W.; Jin, W.; Li, Z.; Wei, Y.; He, Z.; Chen, C.; Qin, C.; Chen, Y.; Tu, R.; Zhou, X. Cultivation of microalgae for lipid production using municipal wastewater. Process Saf. Environ. Prot. 2021, 155, 155–165. [Google Scholar] [CrossRef]
- Culture Collection of Algae & Protozoa. Available online: https://www.ccap.ac.uk/index.php/media-recipes/ (accessed on 1 September 2020).
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Araujo, P.; Nguyen, T.T.; Frøyland, L.; Wang, J.; Kang, J.X. Evaluation of a rapid method for the quantitative analysis of fatty acids in various matrices. J. Chromatogr. A 2008, 1212, 106–113. [Google Scholar] [CrossRef] [Green Version]
- Alkhamis, Y.; Qin, J.G. Comparison of N and P requirements of Isochrysis galbana under phototrophic and mixotrophic conditions. J. Appl. Phycol. 2015, 27, 2231–2238. [Google Scholar] [CrossRef]
- Pagnanelli, F.; Altimari, P.; Trabucco, F.; Toro, L. Mixotrophic growth of Chlorella vulgaris and Nannochloropsis oculata: Interaction between glucose and nitrate. J. Chem. Technol. Biotechnol. 2014, 89, 652–661. [Google Scholar] [CrossRef]
- Song, Y.; Wang, X.; Cui, H.; Ji, C.; Xue, J.; Jia, X.; Ma, R.; Li, R. Enhancing growth and oil accumulation of a palmitoleic acid–rich Scenedesmus obliquus in mixotrophic cultivation with acetate and its potential for ammonium-containing wastewater purification and biodiesel production. J. Environ. Manage. 2021, 297, 113273. [Google Scholar] [CrossRef]
- Menegol, T.; Romero-Villegas, G.I.; López-Rodríguez, M.; Navarro-López, E.; López-Rosales, L.; Chisti, Y.; Cerón-García, M.C.; Molina-Grima, E. Mixotrophic production of polyunsaturated fatty acids and carotenoids by the microalga Nannochloropsis gaditana. J. Appl. Phycol. 2019, 31, 2823–2832. [Google Scholar] [CrossRef]
- Moon, M.; Kim, C.W.; Park, W.K.; Yoo, G.; Choi, Y.E.; Yang, J.W. Mixotrophic growth with acetate or volatile fatty acids maximizes growth and lipid production in Chlamydomonas reinhardtii. Algal Res. 2013, 2, 352–357. [Google Scholar] [CrossRef]
- Chiu, S.Y.; Kao, C.Y.; Tsai, M.T.; Ong, S.C.; Chen, C.H.; Lin, C.S. Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresour. Technol. 2009, 100, 833–838. [Google Scholar] [CrossRef]
- Alkhamis, Y.; Qin, J.G. Cultivation of Isochrysis galbana in phototrophic, heterotrophic, and mixotrophic conditions. Biomed Res. Int. 2013, 2013, 983465. [Google Scholar] [CrossRef] [Green Version]
- Shekarabi, S.P.H.; Mehrgan, M.S. Cultivation of Tetraselmis suecica using a fishmeal factory effluent: Effect on the growth, biochemical composition, and nutrient removal. J. Appl. Phycol. 2021, 33, 1949–1959. [Google Scholar] [CrossRef]
- Msanne, J.; Polle, J.; Starkenburg, S. An assessment of heterotrophy and mixotrophy in Scenedesmus and its utilization in wastewater treatment. Algal Res. 2020, 48, 101911. [Google Scholar] [CrossRef]
- Onay, M. Bioethanol production via different saccharification strategies from H. tetrachotoma ME03 grown at various concentrations of municipal wastewater in a flat-photobioreactor. Fuel 2019, 239, 1315–1323. [Google Scholar] [CrossRef]
- Yang, C.; Hua, Q.; Shimizu, K. Energetics and carbon metabolism during growth of microalgal cells under photoautotrophic, mixotrophic and cyclic light-autotrophic/dark-heterotrophic conditions. Biochem. Eng. J. 2000, 6, 87–102. [Google Scholar] [CrossRef]
- Ohse, S.; Bianchini Derner, R.; Ávila Ozório, R.; Gordo Corrêa, R.; Badiale Furlong, E.; Roberto Cunha, P.C. Lipid content and fatty acid profiles in ten species of microalgae. Idesia (Arica) 2015, 33, 93–101. [Google Scholar] [CrossRef] [Green Version]
- Abomohra, A.E.F.; Eladel, H.; El-Esawi, M.; Wang, S.; Wang, Q.; He, Z.; Feng, Y.; Shang, H.; Hanelt, D. Effect of lipid-free microalgal biomass and waste glycerol on growth and lipid production of Scenedesmus obliquus: Innovative waste recycling for extraordinary lipid production. Bioresour. Technol. 2018, 249, 992–999. [Google Scholar] [CrossRef]
- Camacho-Rodríguez, J.; González-Céspedes, A.M.; Cerón-García, M.C.; Fernández-Sevilla, J.M.; Acién-Fernández, F.G.; Molina-Grima, E. A quantitative study of eicosapentaenoic acid (EPA) production by Nannochloropsis gaditana for aquaculture as a function of dilution rate, temperature and average irradiance. Appl. Microbiol. Biotechnol. 2014, 98, 2429–2440. [Google Scholar] [CrossRef]
- Abirami, S.; Murugesan, S.; Narender, S. Effect of various pretreatment methods prior to extraction of omega 3 fatty acids from Nannochloropsis gaditana. Int. J. Appl. Res. 2016, 2, 81–85. [Google Scholar]
- Park, S.J.; Choi, Y.E.; Kim, E.J.; Park, W.K.; Kim, C.W.; Yang, J.W. Serial optimization of biomass production using microalga Nannochloris oculata and corresponding lipid biosynthesis. Bioprocess Biosyst. Eng. 2012, 35, 3–9. [Google Scholar] [CrossRef]
- Shen, X.F.; Gao, L.J.; Zhou, S.B.; Huang, J.L.; Wu, C.Z.; Qin, Q.W.; Zeng, R.J. High fatty acid productivity from Scenedesmus obliquus in heterotrophic cultivation with glucose and soybean processing wastewater via nitrogen and phosphorus regulation. Sci. Total Environ. 2020, 708, 134596. [Google Scholar] [CrossRef]
- Yang, Z.; Gao, F.; Liu, J.; Yang, J.; Liu, M.; Ge, Y. Improving sedimentation and lipid production of microalgae in the photobioreactor using saline wastewater. Bioresour. Technol. 2021, 126392. [Google Scholar] [CrossRef]
- Dev Sarkar, R.; Singh, H.B.; Chandra Kalita, M. Enhanced lipid accumulation in microalgae through nanoparticle-mediated approach, for biodiesel production: A mini-review. Heliyon 2021, 7, e08057. [Google Scholar] [CrossRef]
- Dierge, E.; Larondelle, Y.; Feron, O. Cancer diets for cancer patients: Lessons from mouse studies and new insights from the study of fatty acid metabolism in tumors. Biochimie 2020, 178, 56–68. [Google Scholar] [CrossRef]
- Toelzer, C.; Gupta, K.; Yadav, S.K.N.; Borucu, U.; Davidson, A.D.; Kavanagh Williamson, M.; Shoemark, D.K.; Garzoni, F.; Staufer, O.; Milligan, R.; et al. Free fatty acid binding pocket in the locked structure of SARS-CoV-2 spike protein. Science 2020, 3255, eabd3255. [Google Scholar] [CrossRef]
- Layé, S.; Nadjar, A.; Joffre, C.; Bazinet, R.P. Anti-inflammatory effects of omega-3 fatty acids in the brain: Physiological mechanisms and relevance to pharmacology. Pharmacol. Rev. 2018, 70, 12–38. [Google Scholar] [CrossRef]
- Guo, X.F.; Li, K.L.; Li, J.M.; Li, D. Effects of EPA and DHA on blood pressure and inflammatory factors: A meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2019, 59, 3380–3393. [Google Scholar] [CrossRef]
- Cengiz Sahin, S. Scenedesmus obliquus: A potential natural source for cosmetic industry. Int. J. Second. Metab. 2019, 6, 129–136. [Google Scholar] [CrossRef] [Green Version]
- da Silva, M.E.T.; Correa, K.d.P.; Martins, M.A.; da Matta, S.L.P.; Martino, H.S.D.; Coimbra, J.S.d.R. Food safety, hypolipidemic and hypoglycemic activities, and in vivo protein quality of microalga Scenedesmus obliquus in Wistar rats. J. Funct. Foods 2020, 65, 103711. [Google Scholar] [CrossRef]
- Dayana Priyadharshini, S.; Suresh Babu, P.; Manikandan, S.; Subbaiya, R.; Govarthanan, M.; Karmegam, N. Phycoremediation of wastewater for pollutant removal: A green approach to environmental protection and long-term remediation. Environ. Pollut. 2021, 290, 117989. [Google Scholar] [CrossRef]
- Patnaik, R.; Mallick, N. Utilization of Scenedesmus obliquus biomass as feedstock for biodiesel and other industrially important co-products: An integrated paradigm for microalgal biorefinery. Algal Res. 2015, 12, 328–336. [Google Scholar] [CrossRef]
- Gilbert-López, B.; Mendiola, J.A.; Fontecha, J.; Van Den Broek, L.A.M.; Sijtsma, L.; Cifuentes, A.; Herrero, M.; Ibáñez, E. Downstream processing of Isochrysis galbana: A step towards microalgal biorefinery. Green Chem. 2015, 17, 4599–4609. [Google Scholar] [CrossRef] [Green Version]
- Ammar, S.H.; Khadim, H.J.; Mohamed, A.I. Cultivation of Nannochloropsis oculata and Isochrysis galbana microalgae in produced water for bioremediation and biomass production. Environ. Technol. Innov. 2018, 10, 132–142. [Google Scholar] [CrossRef]
- Chukhutsina, V.U.; Fristedt, R.; Morosinotto, T.; Croce, R. Photoprotection strategies of the alga Nannochloropsis gaditana. Biochim. Biophys. Acta-Bioenerg. 2017, 1858, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Ajjawi, I.; Verruto, J.; Aqui, M.; Soriaga, L.B.; Coppersmith, J.; Kwok, K.; Peach, L.; Orchard, E.; Kalb, R.; Xu, W.; et al. Lipid production in Nannochloropsis gaditana is doubled by decreasing expression of a single transcriptional regulator. Nat. Biotechnol. 2017, 35, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Cheirsilp, B.; Torpee, S. Enhanced growth and lipid production of microalgae under mixotrophic culture condition: Effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 2012, 110, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Cid, A.; Abalde, J.; Herrero, C. High yield mixotrophic cultures of the marine microalga Tetraselmis suecica (Kylin) Butcher (Prasinophyceae). J. Appl. Phycol. 1992, 4, 31–37. [Google Scholar] [CrossRef] [Green Version]
| Class | Genus/Species | Habitat | Strain |
|---|---|---|---|
| Prymnesiophyceae | I. galbana | Marine | CCAP 927/1 |
| Eustigmatophyceae | M. gaditana | Marine | CCAP 849/5 |
| Chlorophyceae | S. obliquus | Freshwater | CCAP 276/3A |
| Eustigmatophyceae | N. oculata | Marine | CCAP 849/1 |
| Chlorodendrophyceae | T. suecica | Marine | CCAP 66/4 |
| Genus/Species | Autotrophic | Mixotrophic | Heterotrophic |
|---|---|---|---|
| I. galbana | 0.033 ± 0.0049 | 0.057 ± 0.011 | 0.055 ± 0.013 |
| M. gaditana | 0.014 ± 0.0011 | 0.041 ± 0.0031 | 0.029 ± 0.0041 |
| S. obliquus | 0.030 ± 0.0010 | 0.130 ± 0.0094 | 0.140 ± 0.0032 |
| N. oculata | 0.003 ± 0.0015 | 0.011 ± 0.0018 | 0.058 ± 0.0014 |
| T. suecica | 0.011 ± 0.0017 | 0.033 ± 0.0060 | 0.040 ± 0.0076 |
| Nutrition | C12:0 | C16:0 | C16:1n7 | C18:0 | C18:1n9c/ C18:3n3 | C18:2n6c | C20:5n3 | C22:6n3 | |
|---|---|---|---|---|---|---|---|---|---|
| I. galbana | Autotrophic | 0.0 | 18.0 | 0.0 | 12.9 | 14.0 | 11.4 | 0.0 | 0.0 |
| Mixotrophic | 1.3 | 44.6 | 2.0 | 28.5 | 65.0 | 38.1 | 0.5 | 0.0 | |
| Heterotrophic | 0.0 | 74.0 | 2.5 | 61.9 | 77.4 | 87.9 | 1.3 | 0.4 | |
| M. gaditana | Autotrophic | 0.0 | 68.6 | 1.9 | 7.9 | 108.6 | 9.9 | 8.1 | 0.0 |
| Mixotrophic | 7.0 | 22.9 | 1.1 | 8.2 | 9.1 | 20.6 | 0.6 | 0.0 | |
| Heterotrophic | 0.0 | 63.7 | 3.5 | 33.4 | 66.9 | 51.1 | 0.0 | 0.0 | |
| S. obliquus | Autotrophic | 0.0 | 30.8 | 0.0 | 10.6 | 30.3 | 2.8 | 0.0 | 0.0 |
| Mixotrophic | 0.6 | 61.2 | 3.6 | 12.5 | 57.7 | 27.5 | 1.2 | 0.0 | |
| Heterotrophic | 0.4 | 23.1 | 0.6 | 10.9 | 17.1 | 10.1 | 1.1 | 0.0 | |
| N. oculata | Autotrophic | 8.0 | 70.8 | 0.0 | 45.3 | 31.8 | 24.8 | 0.0 | 4.8 |
| Mixotrophic | 0.0 | 61.3 | 3.5 | 23.7 | 48.3 | 28.6 | 2.2 | 2.3 | |
| Heterotrophic | 0.0 | 31.2 | 0.5 | 15.4 | 28.6 | 18.3 | 0.0 | 0.5 | |
| T. suecica | Autotrophic | 0.0 | 66.5 | 1.1 | 10.1 | 99.6 | 11.7 | 9.2 | 0.0 |
| Mixotrophic | 0.6 | 43.2 | 2.3 | 19.9 | 42.4 | 22.9 | 1.0 | 0.0 | |
| Heterotrophic | 0.0 | 22.6 | 1.4 | 11.7 | 22.3 | 15.0 | 1.1 | 0.0 |
| Genus/ Species | Nutrition | Organic Carbon/ Nitrogen Source | Biomass Production | Lipid Production | Reference |
|---|---|---|---|---|---|
| S. obliquus | Mixotrophic | LFAH15-WG10 | 0.248 g·L−1·d−1 | 59.7 mg·L−1·d−1 | [40] |
| S. obliquus | Autotrophic | - | 0.15 g·L−1·d−1 | 36.9 mg·L−1·d−1 | [22] |
| Mixotrophic | 40% Cheese whey | 0.28 g·L−1·d−1 | 37.8 mg·L−1·d−1 | ||
| Heterotrophic | 40% Cheese whey | 0.21 g·L−1·d−1 | - | ||
| Nannochloropsis sp. | Autotrophic | - | 0.016 g·L−1·d−1 | 30 mg·L−1·d−1 | [59] |
| Mixotrophic | glucose | 0.044 g·L−1·d−1 | 1200 mg·L−1·d−1 | ||
| Heterotrophic | glucose | 0.013 g·L−1·d−1 | 40 mg·L−1·d−1 | ||
| N. oculata | Autotrophic | - | 0.387 g·L−1·d−1 | 37.9 mg·L−1·d−1 | [43] |
| Mixotrophic | Yeast | 1.011 g·L−1·d−1 | 109.2 mg·L−1·d−1 | ||
| I. galbana | Autotrophic | - | 0.011 g·L−1·d−1 | [34] | |
| Mixotrophic | Glycerol | 0.022 g·L−1·d−1 | - | ||
| Heterotrophic | Glycerol | Inhibited | |||
| T. suecica | Autotrophic | - | 0.93 × 106 cell mL−1 | 2.3 mg·L−1·d−1 | [60] |
| Mixotrophic | Yeast extract:glucose 4:1 | 2.97 × 106 cell mL−1 | 9.3 mg·L−1·d−1 | ||
| I. galbana | Autotrophic | - | 0.033 g·L−1·d−1 | 6.18 mg·L−1·d−1 | Present study |
| Mixotrophic | Glucose | 0.057 g·L−1·d−1 | 24.85 mg·L−1·d−1 | ||
| Heterotrophic | Glucose | 0.055 g·L−1·d−1 | 16.67 mg·L−1·d−1 | ||
| M. gaditana | Autotrophic | - | 0.014 g·L−1·d−1 | 4.07 mg·L−1·d−1 | Present study |
| Mixotrophic | Glucose | 0.041 g·L−1·d−1 | 7.82 mg·L−1·d−1 | ||
| Heterotrophic | Glucose | 0.029 g·L−1·d−1 | 6.6 mg·L−1·d−1 | ||
| S. obliquus | Autotrophic | - | 0.030 g·L−1·d−1 | 4 mg·L−1·d−1 | Present study |
| Mixotrophic | Glucose | 0.13 g·L−1·d−1 | 21.97 mg·L−1·d−1 | ||
| Heterotrophic | Glucose | 0.14 g·L−1·d−1 | 22.77 mg·L−1·d−1 | ||
| N. oculata | Autotrophic | - | 0.0020 g·L−1·d−1 | 2.04 mg·L−1·d−1 | Present study |
| Mixotrophic | Glucose | 0.011 g·L−1·d−1 | 6 mg·L−1·d−1 | ||
| Heterotrophic | Glucose | 0.058 g·L−1·d−1 | 5.67 mg·L−1·d−1 | ||
| T. suecica | Autotrophic | - | 0.011 g·L−1·d−1 | 3.27 mg·L−1·d−1 | Present study |
| Mixotrophic | Glucose | 0.033 g·L−1·d−1 | 6 mg·L−1·d−1 | ||
| Heterotrophic | Glucose | 0.074 g·L−1·d−1 | 9.33 mg·L−1·d−1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicodemou, A.; Kallis, M.; Agapiou, A.; Markidou, A.; Koutinas, M. The Effect of Trophic Modes on Biomass and Lipid Production of Five Microalgal Strains. Water 2022, 14, 240. https://doi.org/10.3390/w14020240
Nicodemou A, Kallis M, Agapiou A, Markidou A, Koutinas M. The Effect of Trophic Modes on Biomass and Lipid Production of Five Microalgal Strains. Water. 2022; 14(2):240. https://doi.org/10.3390/w14020240
Chicago/Turabian StyleNicodemou, Andonia, Michalis Kallis, Anastasia Agapiou, Androulla Markidou, and Michalis Koutinas. 2022. "The Effect of Trophic Modes on Biomass and Lipid Production of Five Microalgal Strains" Water 14, no. 2: 240. https://doi.org/10.3390/w14020240






