Environmental Hazards of an Unrecultivated Liquid Waste Disposal Site on Soil and Groundwater
Abstract
:1. Introduction
2. Materials and Methods
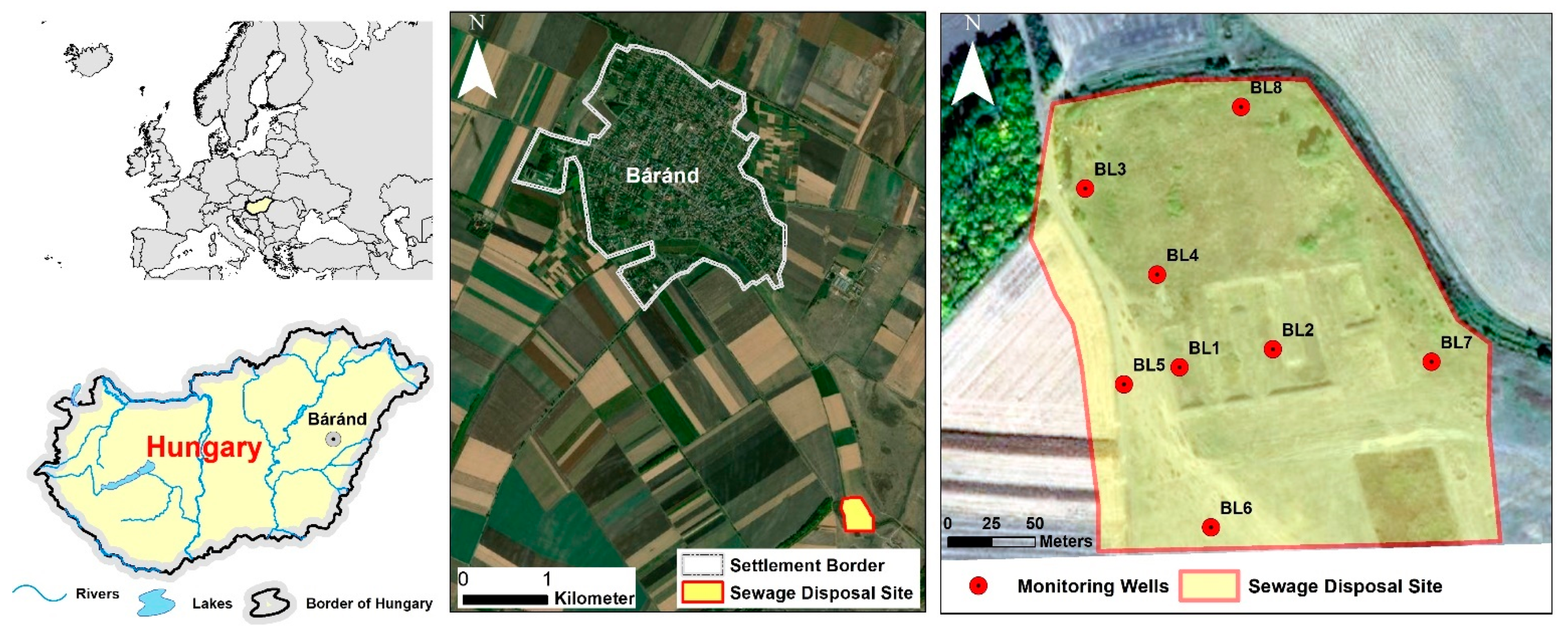
2.1. Site Location and Characteristics
2.2. Field Sampling and Laboratory Analysis
2.3. Statistical Analysis and GIS
3. Results and Discussion
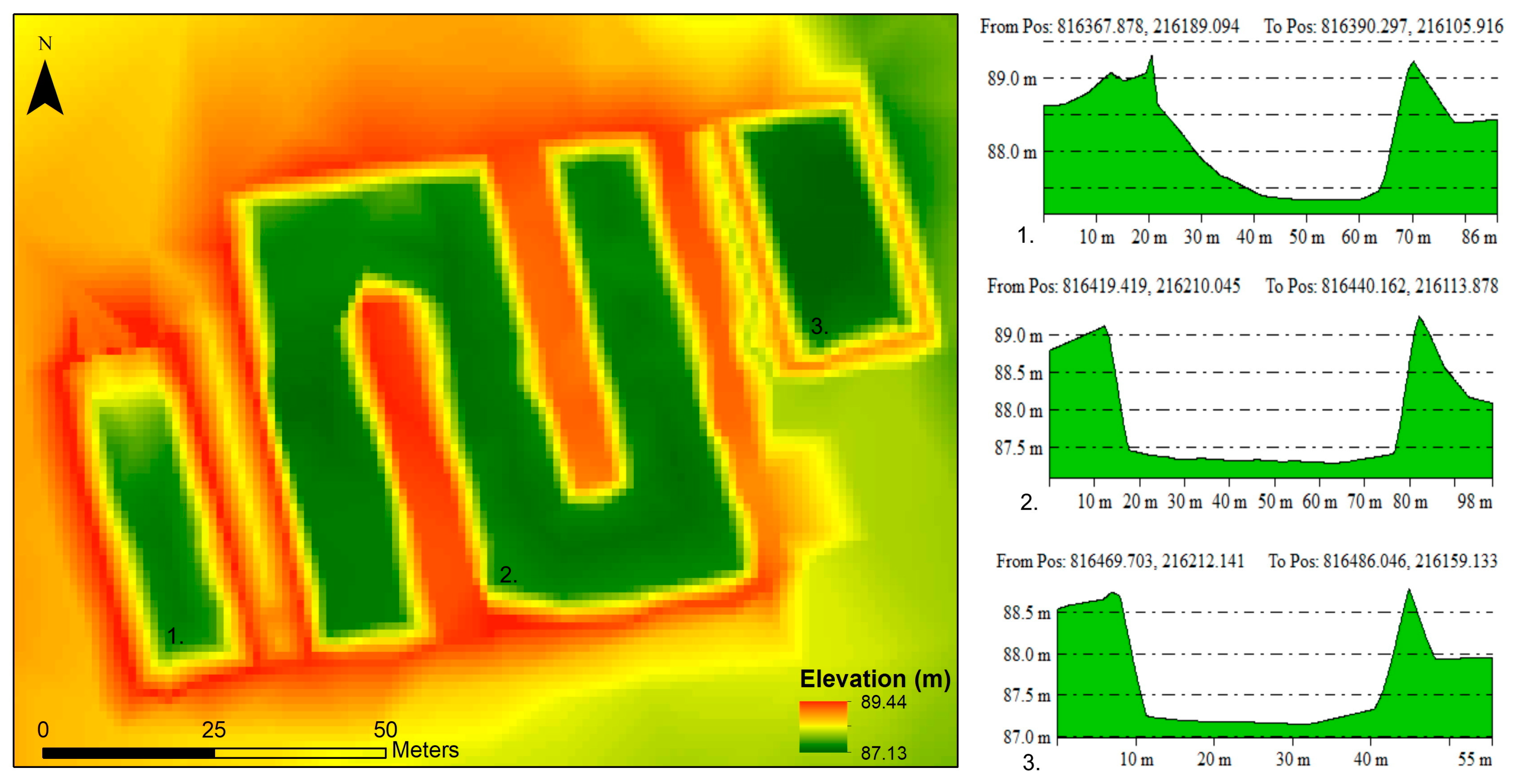
3.1. Digital Elevation Model of the Disposal Site
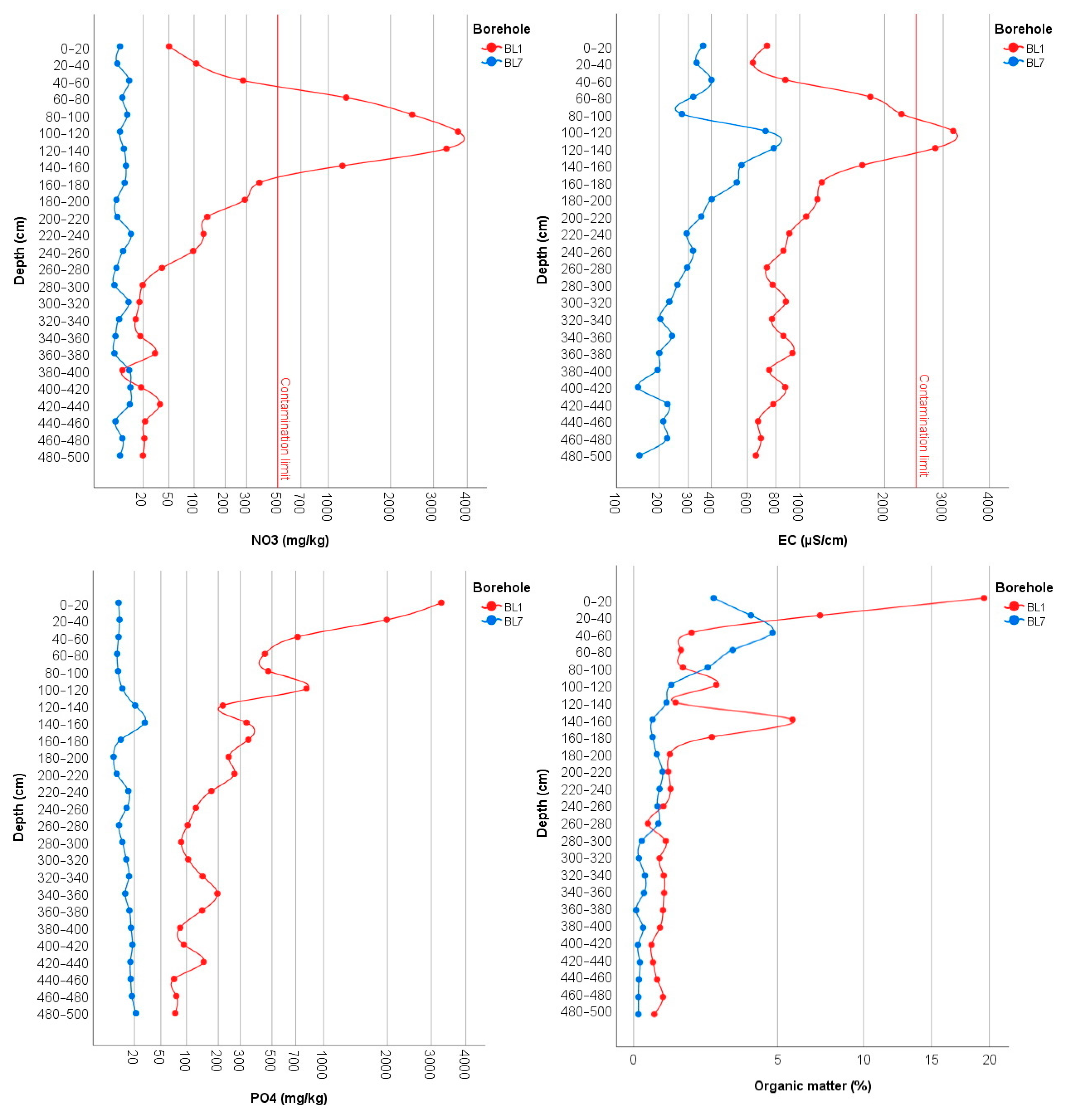
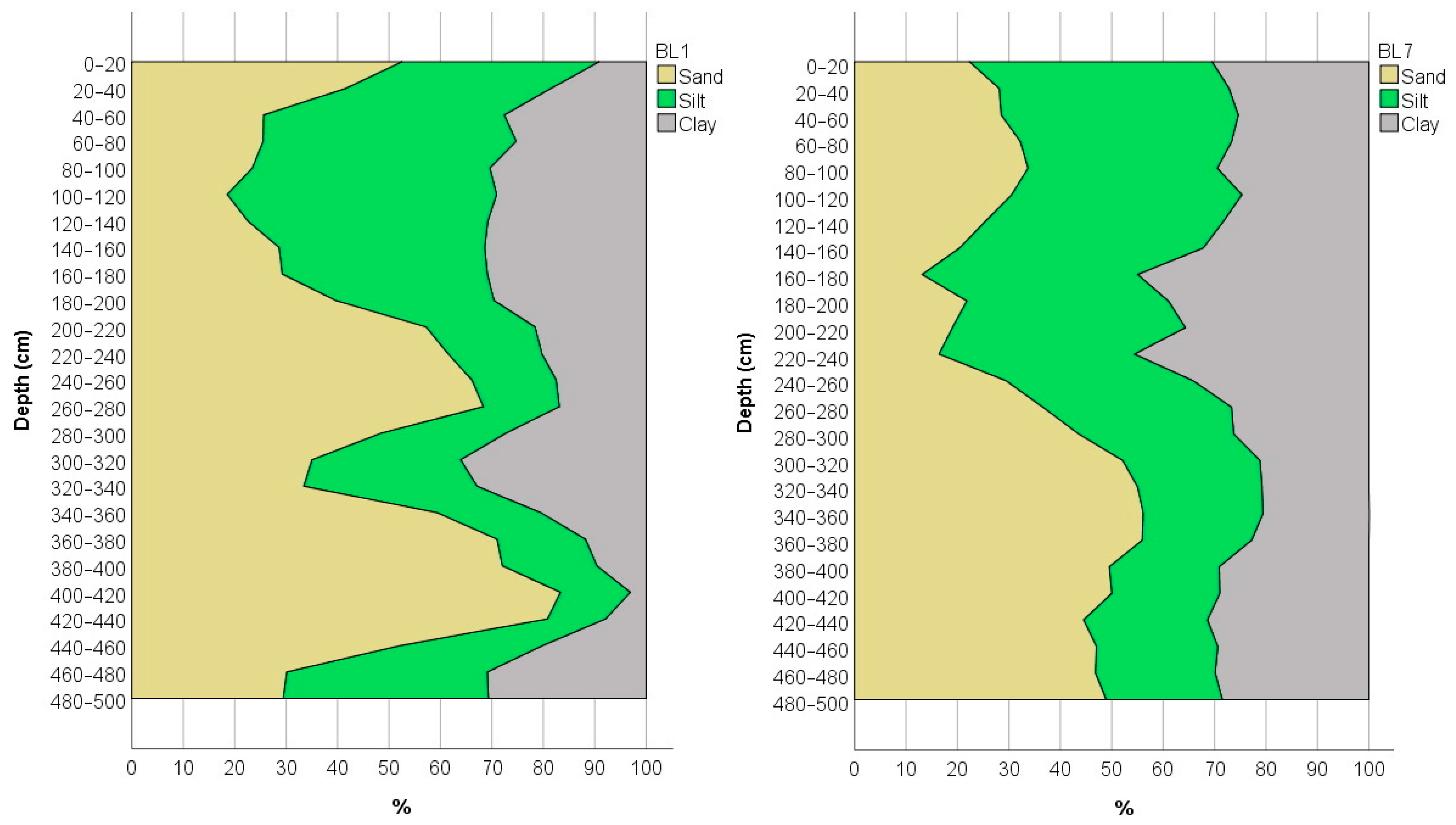
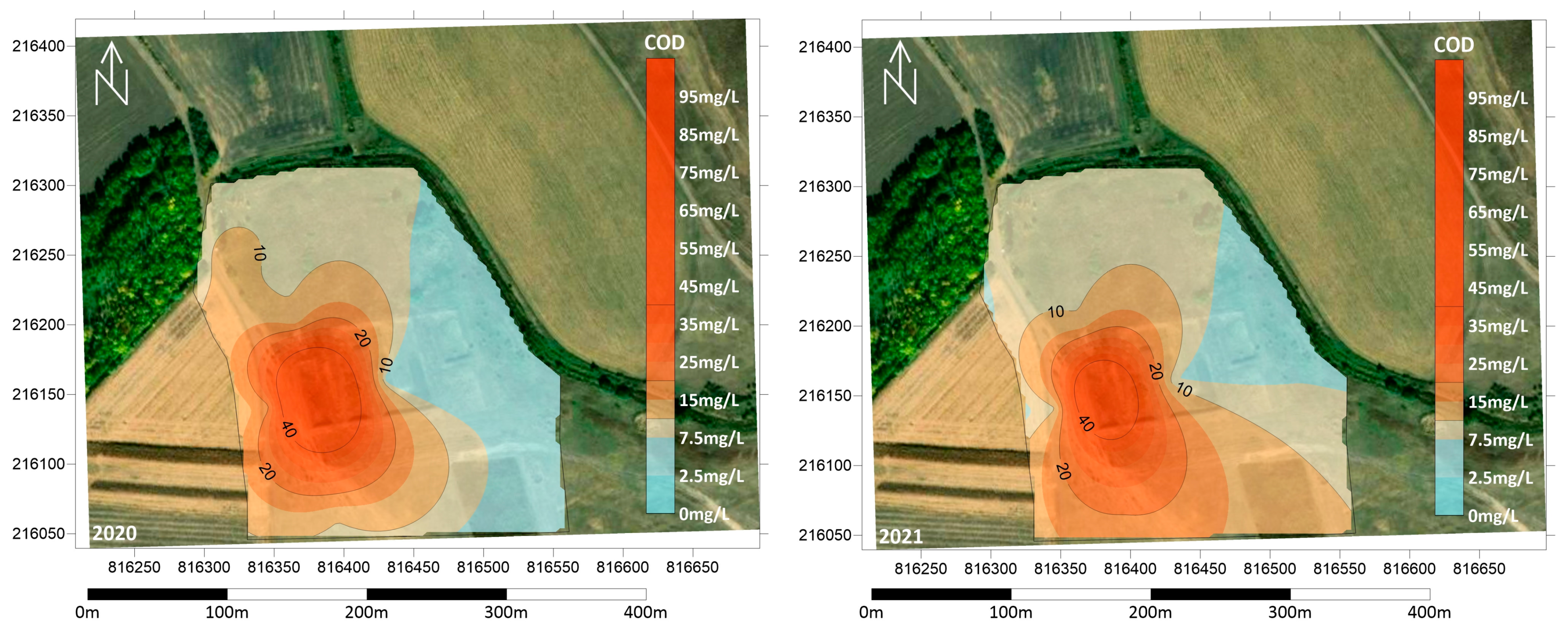
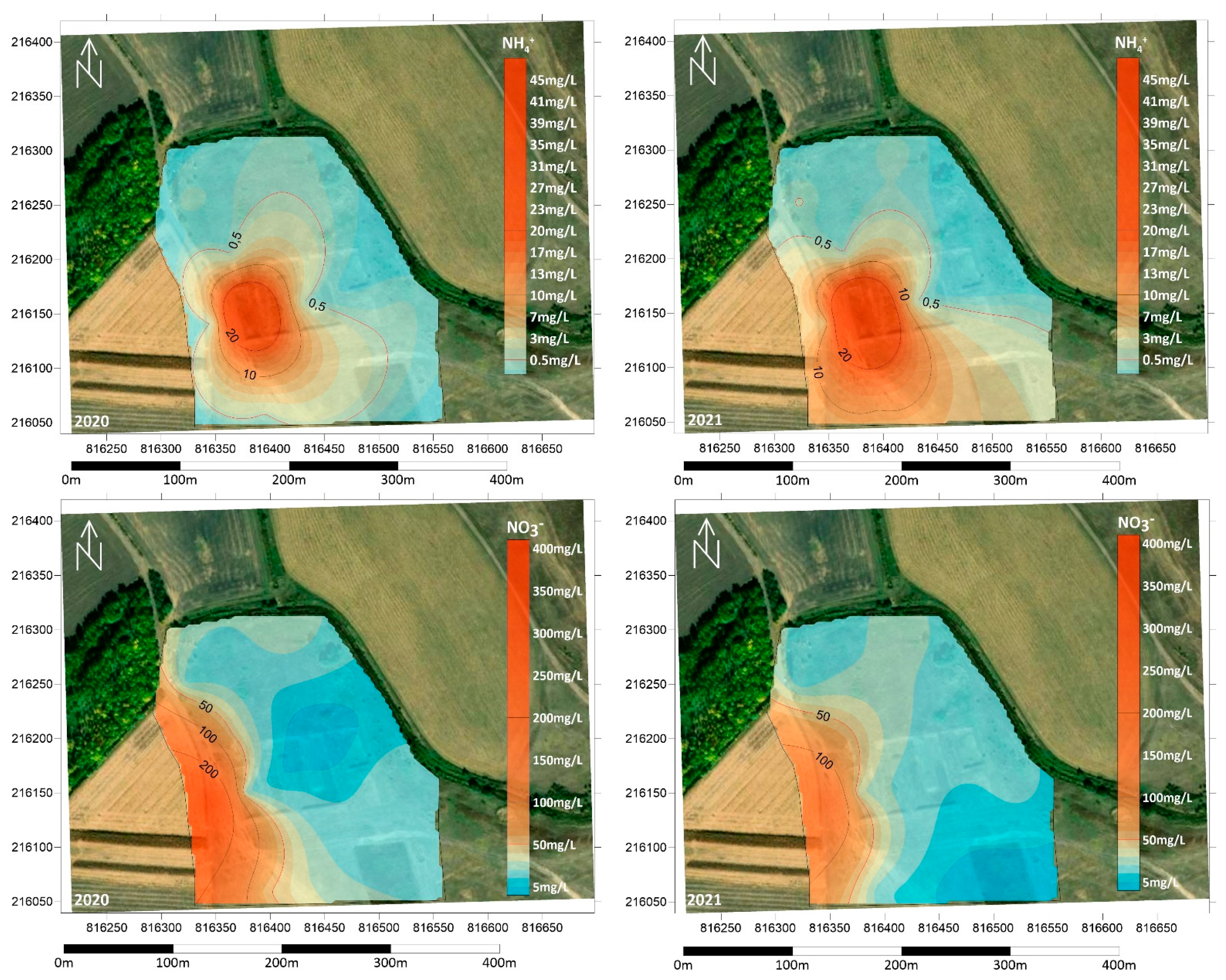
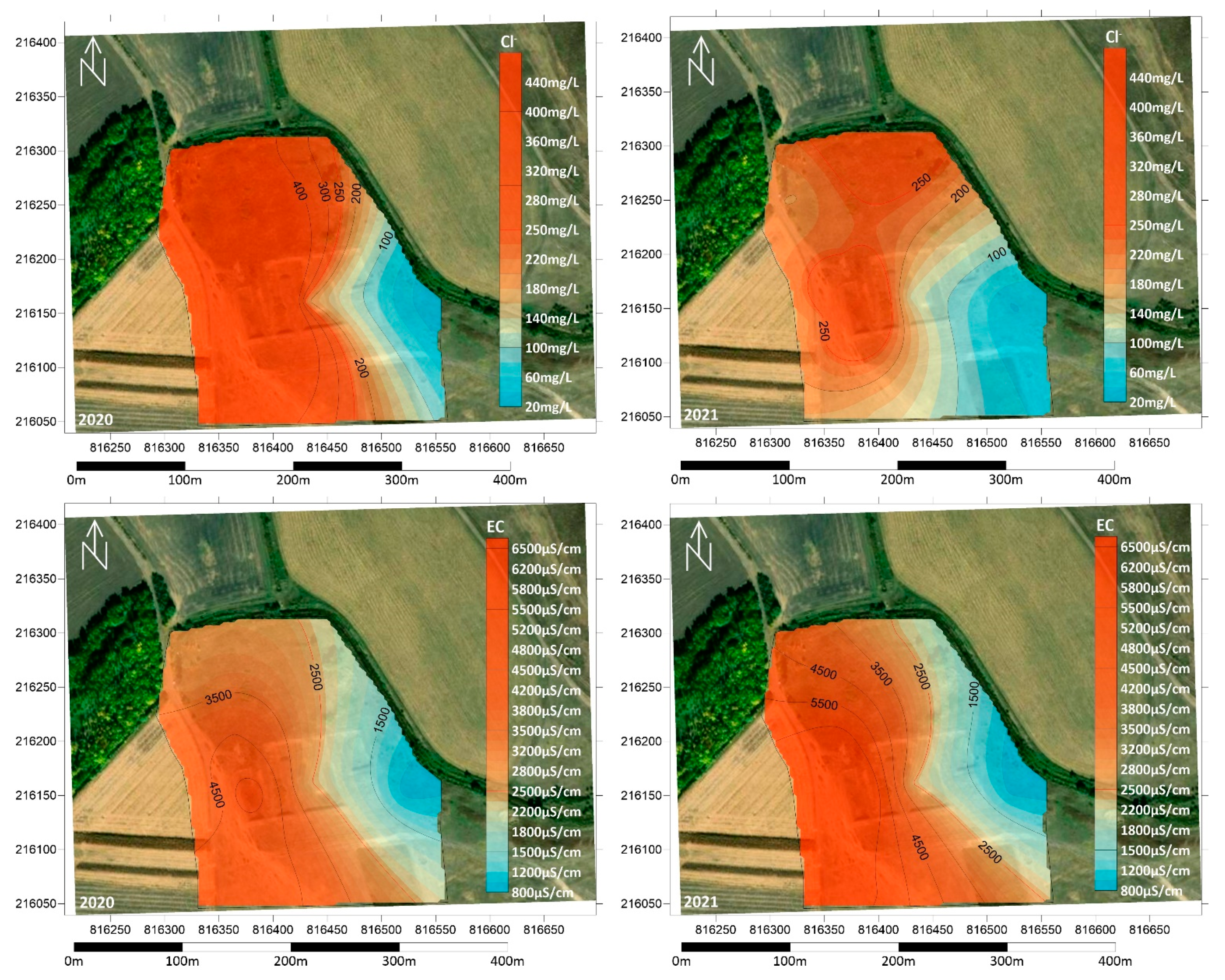
3.2. Physical and Chemical Measurements of the Soil
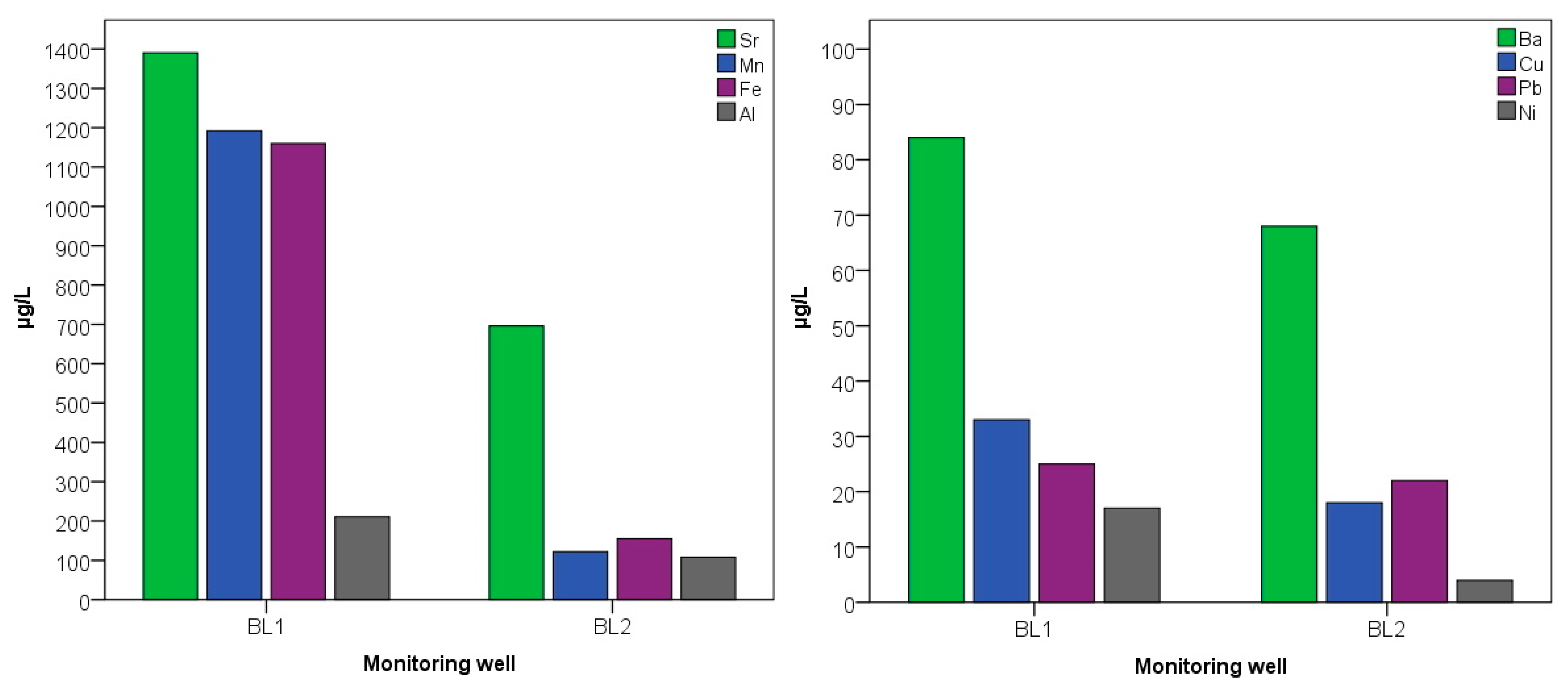
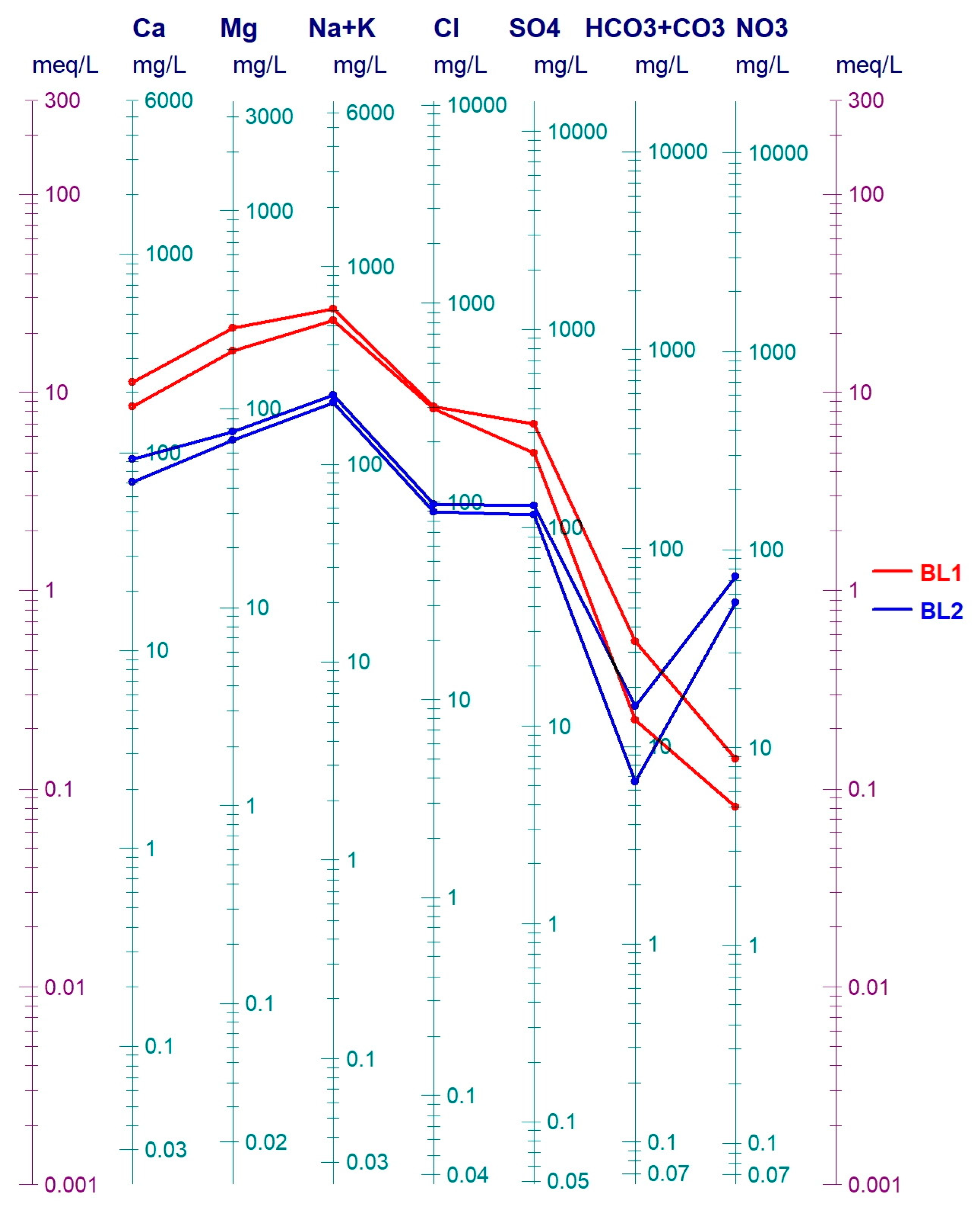
3.3. Groundwater Contamination in the Area
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bugajski, P.M.; Kurek, K.; Młyński, D.; Operacz, A. Designed and real hydraulic load of household wastewater treatment plants. J. Water Land Dev. 2019, 40, 155–160. [Google Scholar] [CrossRef]
- Janža, M.; Prestor, J.; Pestotnik, S.; Jamnik, B. Nitrogen mass balance and pressure impact model applied to an urban aquifer. Water 2020, 12, 1171. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, W.; Ghanem, M. Effect of wastewater on the spring water quality of Sarida Catchment–West Bank. Arab. J. Basic Appl. Sci. 2021, 28, 292–299. [Google Scholar] [CrossRef]
- Ghanem, M.; Ahmad, W.; Keilani, Y.; Sawaftah, F.; Schelter, L.; Schuettrumpf, H. Spring water quality in the central West Bank, Palestine. J. Asian Earth Sci. X 2021, 5, 100052. [Google Scholar] [CrossRef]
- Roba, C.; Bălc, R.; Andreica, D.; Pădurean, A.; Pogăcean, P.; Chertes, T.; Moldovan, F.; Mocan, B.; Roșu, C. Assessment of groundwater quality in nw of romania and its suitability for drinking and agricultural purposes. Environ. Eng. Manag. J. (EEMJ) 2021, 20, 435–437. [Google Scholar] [CrossRef]
- Mester, T.; Szabó, G.; Balla, D. Assessment of shallow groundwater purification processes after the construction of a municipal sewerage network. Water 2021, 13, 1946. [Google Scholar] [CrossRef]
- Hastuti, E.; Riyana, R.; Joy, B.; Supratman, U.; Pamekas, R. Integrated Community Onsite Sanitation System for Close Loop Faecal Management. In E3S Web of Conferences; EDP Sciences: Les Ulis, France, 2021; Volume 249. [Google Scholar]
- Khalil, C.; Al Hageh, C.; Korfali, S.; Khnayzer, R.S. Municipal leachates health risks: Chemical and cytotoxicity assessment from regulated and unregulated municipal dumpsites in Lebanon. Chemosphere 2018, 208, 1–13. [Google Scholar] [CrossRef]
- Amusan, A.A.; Ige, D.V.; Olawale, R. Characteristics of soils and crops’ uptake of metals in municipal waste dump sites in Nigeria. J. Hum. Ecol. 2005, 17, 167–171. [Google Scholar] [CrossRef]
- Haile, D.; Gabbiye, N. Groundwater quality assessment of Chilanchil Abay watershed: The case of Bahir-Dar City waste disposal site. In Advances of Science and Technology; Springer: Cham, Switzerland, 2020; pp. 36–58. [Google Scholar]
- Mukherjee, A.G.; Wanjari, U.R.; Chakraborty, R.; Renu, K.; Vellingiri, B.; George, A.; Gopalakrishnan, A.V. A review on modern and smart technologies for efficient waste disposal and management. J. Environ. Manag. 2021, 297, 113347. [Google Scholar] [CrossRef]
- Han, D.; Tong, X.; Currell, M.J.; Cao, G.; Jin, M.; Tong, C. Evaluation of the impact of an uncontrolled landfill on surrounding groundwater quality, Zhoukou, China. J. Geochem. Explor. 2014, 136, 24–39. [Google Scholar] [CrossRef]
- Moore, E.; Udom, G.; Ngobiri, N. Assessment and impact of current sewage disposal practices in selected niger delta environment. Afr. J. Environ. Nat. Sci. 2019, 2, 77–86. [Google Scholar]
- Oakley, H.R. The disposal of communal waste. J. R. Soc. Arts 1967, 115, 533–546. [Google Scholar]
- Lyubimova, T.P.; Lepikhin, A.P.; Parshakova, Y.N.; Tsiberkin, K.B. Numerical modeling of liquid-waste infiltration from storage facilities into surrounding groundwater and surface-water bodies. J. Appl. Mech. Tech. 2016, 57, 1208–1216. [Google Scholar] [CrossRef]
- Tanna, R.N.; Moncur, M.C.; Birks, S.J.; Gibson, J.J.; Ptacek, C.J.; Mayer, B.; Wieser, M.E.; Wrona, F.J.; Munkittrick, K.R. Utility of a multi-tracer approach as a component of adaptive monitoring for municipal wastewater impacts. Water Qual. Res. J. 2020, 55, 327–341. [Google Scholar] [CrossRef]
- Hughes, G.M. Selection of refuse disposal sites in northeastern Illinois. Environ. Geol. 17 1967, 26. Available online: https://www.ideals.illinois.edu/bitstream/handle/2142/78852/selectionofrefus17hugh.pdf?sequence=1 (accessed on 5 December 2021).
- Kanmani, S.; Gandhimathi, R. Assessment of heavy metal contamination in soil due to leachate migration from an open dumping site. Appl. Water Sci. 2013, 3, 193–205. [Google Scholar] [CrossRef] [Green Version]
- Gorzelak, M.; Dąbrowska, D. Assessment of changes in the quality of ground water in the area of landfill site in Poczesna (South Poland) using the LWPI index. Environ. Socio-Econ. Stud. 2021, 9, 35–43. [Google Scholar] [CrossRef]
- Anomohanran, O.; Utieyin, B.S.; Ofomola, M.O. Delineation of groundwater vulnerability to pollution around dumpsites in Sapele, Nigeria. Int. J. Hydrol. Sci. Technol. 2021, 12, 59–76. [Google Scholar] [CrossRef]
- Wakida, F.T.; Lerner, D.N. Non-agricultural sources of groundwater nitrate: A review and case study. Water Res. 2005, 39, 3–16. [Google Scholar] [CrossRef]
- Fudala-Ksiazek, S.; Kulbat, E.; Luczkiewicz, A. Nitrification, denitrification, and dephosphatation capability of activated sludge during co-treatment of intermediate-age landfill leachates with municipal wastewater. Environ. Technol. 2018, 39, 986–996. [Google Scholar] [CrossRef] [PubMed]
- Buss, S.R.; Herbert, A.W.; Morgan, P.; Thornton, S.F.; Smith, J.W.N. A review of ammonium attenuation in soil and groundwater. Q. J. Eng. Geol. Hydrogeol. 2004, 37, 347–359. [Google Scholar] [CrossRef]
- Stevenson, F.J. Origin and distribution of nitrogen in soil. Soil Nitrogen 1965, 10, 1–42. [Google Scholar]
- Knops, J.M.; Tilman, D. Dynamics of soil nitrogen and carbon accumulation for 61 years after agricultural abandonment. Ecology 2000, 81, 88–98. [Google Scholar] [CrossRef]
- Anikwe, M.A.N.; Nwobodo, K.C.A. Long term effect of municipal waste disposal on soil properties and productivity of sites used for urban agriculture in Abakaliki, Nigeria. Bioresour. Technol. 2002, 83, 241–250. [Google Scholar] [CrossRef]
- Reynolds-Vargas, J.; Fraile-Merino, J.; Hirata, R. Trends in Nitrate Concentrations and Determination of Its Origin Using Stable Isotopes (¹⁸O and ¹⁵N) in Groundwater of the Western Central Valley, Costa Rica. Ambio 2006, 35, 229–236. [Google Scholar] [CrossRef]
- Stollenwerk, K.G. Simulation of phosphate transport in sewage-contaminated groundwater, Cape Cod, Massachusetts. Appl. Geochem. 1996, 11, 317–324. [Google Scholar] [CrossRef]
- LeBlanc, D.R. Sewage Plume in a Sand and Gravel Aquifer, Cape Cod, Massachusetts (Vol. 2218); Department of the Interior, US Geological Survey: Alexandria, VA, USA, 1985.
- Hirschberg, K.J.B. Liquid-waste disposal in Perth, A hydrogeological assessment. West. Aust. Geol. Surv. Rep. 1986, 19, 55–61. [Google Scholar]
- Yoada, R.M.; Chirawurah, D.; Adongo, P.B. Domestic waste disposal practice and perceptions of private sector waste management in urban Accra. BMC Public Health 2014, 14, 697. [Google Scholar] [CrossRef] [Green Version]
- Appleyard, S.J. Impact of liquid waste disposal on potable groundwater resources near Perth, Western Australia. Environ. Geol. 1996, 28, 106–110. [Google Scholar] [CrossRef]
- Szabó, G.; Bessenyei, É.; Hajnal, A.; Csige, I.; Szabó, G.; Tóth, C.; Posta, J.; Mester, T. The Use of Sodium to Calibrate the Transport Modeling of Water Pollution in Sandy Formations Around an Uninsulated Sewage Disposal Site. Water Air Soil Pollut. 2016, 227, 45. [Google Scholar] [CrossRef] [Green Version]
- Ringo, J. Status of Sewage Disposal in Dodoma Municipality, Tanzania. Int. J. Mar. Atmos. Earth Sci. 2016, 4, 24–34. [Google Scholar]
- Marek, D.; Baun, M.; Dąbrowski, M. The challenge of implementing European Union environmental law in the new member states: The Urban Waste Water Treatment Directive in the Czech Republic and Poland. Environ. Plan. C Polit. Space 2017, 35, 1117–1135. [Google Scholar] [CrossRef]
- Somlyódy, L.; Buzás, K.; Clement, A.; Melicz, Z. Strategies for approximating EU legislation in Hungary: The Sajó River case. Water Sci. Technol. 1999, 40, 87–94. [Google Scholar] [CrossRef]
- Somlyódy, L.; Patziger, M. Urban wastewater development in Central and Eastern Europe. Water Sci. Technol. 2012, 66, 1081–1087. [Google Scholar] [CrossRef]
- Mester, T.; Balla, D.; Szabó, G. Assessment of Groundwater Quality Changes in the Rural Environment of the Hungarian Great Plain Based on Selected Water Quality Indicators. Water Air Soil Pollut. 2020, 231, 1–14. [Google Scholar] [CrossRef]
- Hungarian Central Statistical Office. 2021. Available online: https://www.ksh.hu/stadat_files/kor/hu/kor0067.html (accessed on 3 December 2021).
- Michéli, E.; Fuchs, M.; Hegymegi, P.; Stefanovits, P. Classification of the major soils of Hungary and their correlation with the World Reference Base for Soil Resources (WRB). Agrokém. és Talajt. 2006, 55, 19–28. [Google Scholar] [CrossRef]
- Dövényi, Z. Magyarország Kistájainak Katasztere [Inventory of Microregions in Hungary]; MTA Földrajztudományi Kutatóintézet—MTA Geographical Research Institute: Budapest, Hungary, 2010; 876p. [Google Scholar]
- Hungarian Central Statistical Office (HSCO). 2021. Available online: https://nyilvantarto.hu/hu/statisztikak?stat=kozerdeku (accessed on 11 July 2021).
- Mester, T.; Balla, D.; Karancsi, G.; Bessenyei, É.; Szabó, G. Effects of nitrogen loading from domestic wastewater on groundwater quality. Water SA 2019, 45, 349–358. [Google Scholar] [CrossRef] [Green Version]
- Müller, H.W.; Dohrmann, R.; Klosa, D.; Rehder, S.; Eckelmann, W. Comparison of two procedures for particle-size analysis: Köhn pipette and X-ray granulometry. J. Plant Nutr. Soil Sci. 2009, 172, 172–179. [Google Scholar] [CrossRef]
- Ślósarczyk, K.; Witkowski, A.J. Preliminary Evaluation of the Possible Occurrence of Pesticides in Groundwater Contaminated with Nitrates—A Case Study from Southern Poland. Water 2021, 13, 3091. [Google Scholar] [CrossRef]
- El Mountassir, O.; Bahir, M.; Ouazar, D.; Chehbouni, A.; Carreira, P.M. Temporal and spatial assessment of groundwater contamination with nitrate using nitrate pollution index (NPI), groundwater pollution index (GPI), and GIS (case study: Essaouira basin, Morocco). Environ. Sci. Pollut. Res. 2021, 1–18. [Google Scholar] [CrossRef]
- Aravinthasamy, P.; Karunanidhi, D.; Subramani, T.; Roy, P.D. Demarcation of groundwater quality domains using GIS for best agricultural practices in the drought-prone Shanmuganadhi River basin of South India. Environ. Sci. Pollut. Res. 2021, 28, 18423–18435. [Google Scholar] [CrossRef]
- Beauchamp, E.G.; Drury, C.F. Ammonium fixation, release, nitrification, and immobilization in high-and low-fixing soils. Soil Sci. Soc. Am. J. 1991, 55, 125–129. [Google Scholar] [CrossRef]
- Agehara, S.; Warncke, D.D. Soil moisture and temperature effects on nitrogen release from organic nitrogen sources. Soil Sci. Soc. Am. J. 2005, 69, 1844–1855. [Google Scholar] [CrossRef] [Green Version]
- Leonard, M.; Gilpin, B. Potential Impacts of On-Site Sewage Disposal on Groundwater; Institute of Environmental Science and Research Limited: Porirua, New Zealand, 2006. [Google Scholar]
- Al Yaqout, A.F. Assessment and analysis of industrial liquid waste and sludge disposal at unlined landfill sites in arid climate. Waste Manag. 2003, 23, 817–824. [Google Scholar] [CrossRef]
- Okoye, B.S.; Umeora, C.O.; Ifebi, O.C.; Onwuzuligbo, C.C. Effects of sewage disposal systems on the environment in public housing estates in enugu metrpolis. Afr. J. Environ. 2018, 1, 120–130. [Google Scholar]
- Talalaj, I.A.; Biedka, P. Use of the landfill water pollution index (LWPI) for groundwater quality assessment near the landfill sites. Environ. Sci. Pollut. Res. 2016, 23, 24601–24613. [Google Scholar] [CrossRef] [Green Version]
- Brennan, R.B.; Clifford, E.; Devroedt, C.; Morrison, L.; Healy, M.G. Treatment of landfill leachate in municipal wastewater treatment plants and impacts on effluent ammonium concentrations. J. Environ. Manag. 2017, 188, 64–72. [Google Scholar] [CrossRef] [Green Version]
- James, A.; Percy, M.; Ameh, O.S. Heavy metals pollution status of the Katima Mulilo Urban open land wastewater disposal centre and the immediate vicinity. Cogent Environ. Sci. 2020, 6, 1726093. [Google Scholar] [CrossRef]
- Gulyás, G.; Pitás, V.; Fazekas, B.; Kárpáti, Á. Heavy metal balance in a communal wastewater treatment plant. Hung. J. Ind. Chem. 2015, 43, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Szabó, G.; Bessenyei, É. Studying groundwater pollution in the surroundings of a recultivated sewage disposal site in eastern Hungary. J. Selçuk Univ. Nat. Appl. Sci. 2013, 1–12. Available online: http://josunas.selcuk.edu.tr/login/index.php/josunas/article/view/66 (accessed on 29 December 2021).

| Parameter | Limit | Parameter | Limit |
|---|---|---|---|
| pH | <6.5; 9.0< | Aluminium (Al) | 200 µg/L |
| Electrical con. (EC) | 2500 µS/cm | Barium (Ba) | 250 µg/L |
| Ammonium (NH4+) | 0.5 mg/L | Copper (Cu) | 200 µg/L |
| Nitrite (NO2−) | 0.5 mg/L | Cobalt (Co) | 20 µg/L |
| Nitrate (NO3−) | 50 mg/L | Chromium (Cr) | 50 µg/L |
| Chloride (Cl−) | 250 mg/L | Nickel (Ni) | 20 µg/L |
| Phosphate (PO43−) | 0.5 mg/L | Lead (Pb) | 10 µg/L |
| Sulphate (SO42−) | 250 mg/L | Zinc (Zn) | 200 µg/L |
| Sodium (Na) | 200 mg/L |
| 2020 | 2021 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | EC | COD | NH4+ | NO2− | NO3− | PO43− | Cl− | pH | EC | COD | NH4+ | NO2− | NO3− | PO43− | Cl− | |
| Unit | µS/cm | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | µS/cm | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | ||
| Limit | 6.5/9.5 | 2500 | 4.5 | 0.5 | 0.5 | 50 | 0.5 | 250 | 6.5/9.5 | 2500 | 4.5 | 0.5 | 0.5 | 50 | 0.5 | 250 |
| BL1 | 7.19 | 6320 | 98.4 | 48.3 | 0.658 | 6.8 | 4.12 | 790 | 7.25 | 9320 | 74.4 | 56.3 | 0.735 | 8.9 | 3.69 | 463.8 |
| BL2 | 7.26 | 2610 | 5.4 | 0.53 | 0.125 | 8.4 | 0.63 | 207 | 7.29 | 2510 | 7.8 | 0.52 | 0.165 | 14 | 0.78 | 130.2 |
| BL3 | 6.99 | 3060 | 10.9 | 0.34 | 0.019 | 17.3 | 0.47 | 544 | 7.05 | 4870 | 9.3 | 0.63 | 0.015 | 12.5 | 0.53 | 198.3 |
| BL4 | 7.16 | 4590 | 11.7 | 0.71 | 0.058 | 51.8 | 0.38 | 490 | 7.12 | 7010 | 10.9 | 0.75 | 0.076 | 57.6 | 0.41 | 241.4 |
| BL5 | 7.09 | 3920 | 18.1 | 0.67 | 0.063 | 393.0 | 0.61 | 452 | 7.08 | 7060 | 9.3 | 6.14 | 0.059 | 194.8 | 0.65 | 228.5 |
| BL6 | 6.94 | 5220 | 10.1 | 0.57 | 0.018 | 54.5 | 2.56 | 286 | 7.02 | 6610 | 18.8 | 9.67 | 0.021 | 12.8 | 2.84 | 163.8 |
| BL7 | 7.24 | 895 | 6.9 | 0.23 | 0.024 | 19.3 | 0.43 | 48.3 | 7.18 | 937 | 7.8 | 0.28 | 0.014 | 12.1 | 0.32 | 38.8 |
| BL8 | 6.92 | 2680 | 9.5 | 0.35 | 0.029 | 30.4 | 0.55 | 350.4 | 6.86 | 2560 | 9.3 | 0.34 | 0.033 | 24.3 | 0.47 | 319 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mester, T.; Szabó, G.; Sajtos, Z.; Baranyai, E.; Szabó, G.; Balla, D. Environmental Hazards of an Unrecultivated Liquid Waste Disposal Site on Soil and Groundwater. Water 2022, 14, 226. https://doi.org/10.3390/w14020226
Mester T, Szabó G, Sajtos Z, Baranyai E, Szabó G, Balla D. Environmental Hazards of an Unrecultivated Liquid Waste Disposal Site on Soil and Groundwater. Water. 2022; 14(2):226. https://doi.org/10.3390/w14020226
Chicago/Turabian StyleMester, Tamás, György Szabó, Zsófi Sajtos, Edina Baranyai, Gergely Szabó, and Dániel Balla. 2022. "Environmental Hazards of an Unrecultivated Liquid Waste Disposal Site on Soil and Groundwater" Water 14, no. 2: 226. https://doi.org/10.3390/w14020226
APA StyleMester, T., Szabó, G., Sajtos, Z., Baranyai, E., Szabó, G., & Balla, D. (2022). Environmental Hazards of an Unrecultivated Liquid Waste Disposal Site on Soil and Groundwater. Water, 14(2), 226. https://doi.org/10.3390/w14020226






