The Effects of Depth-Related Environmental Factors on Traits in Acropora cervicornis Raised in Nurseries
Abstract
:1. Introduction
2. Materials and Methods
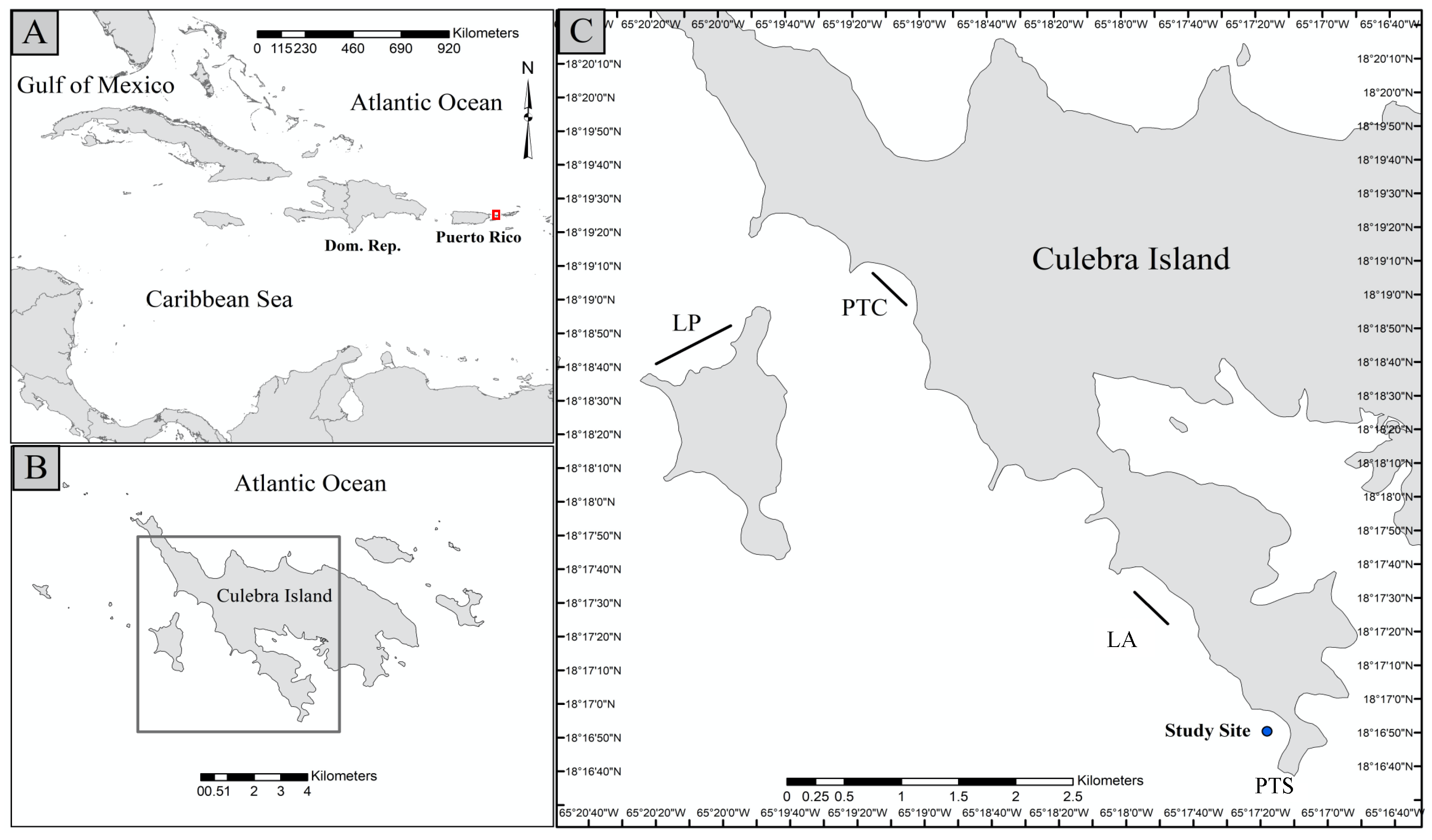
2.1. Study Site
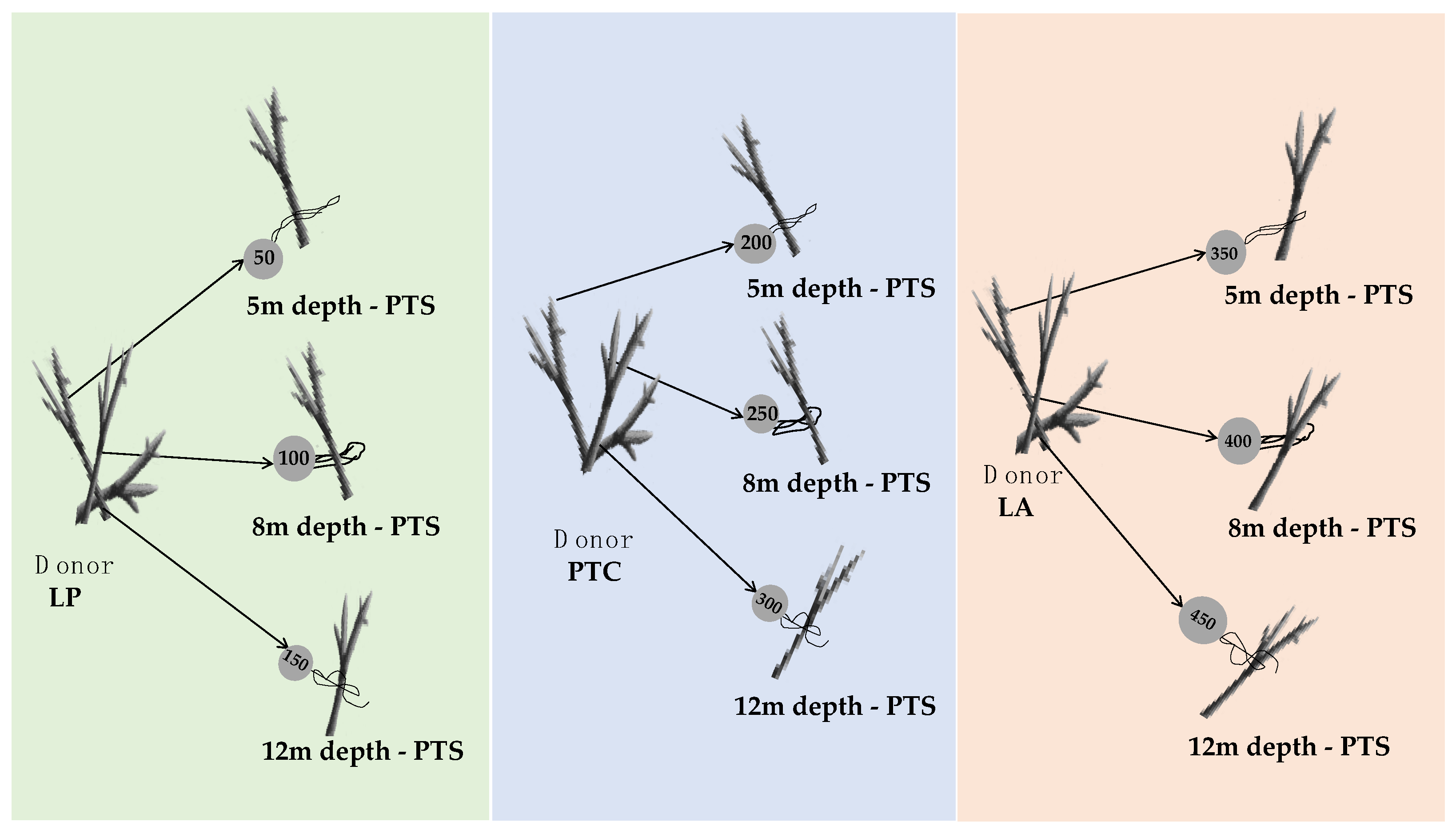
2.2. Coral Collection and Gardening
2.3. Environmental Measurements
2.4. Survival Analysis
2.5. Monthly Growth Rates
2.6. Coral Growth Analysis
3. Results
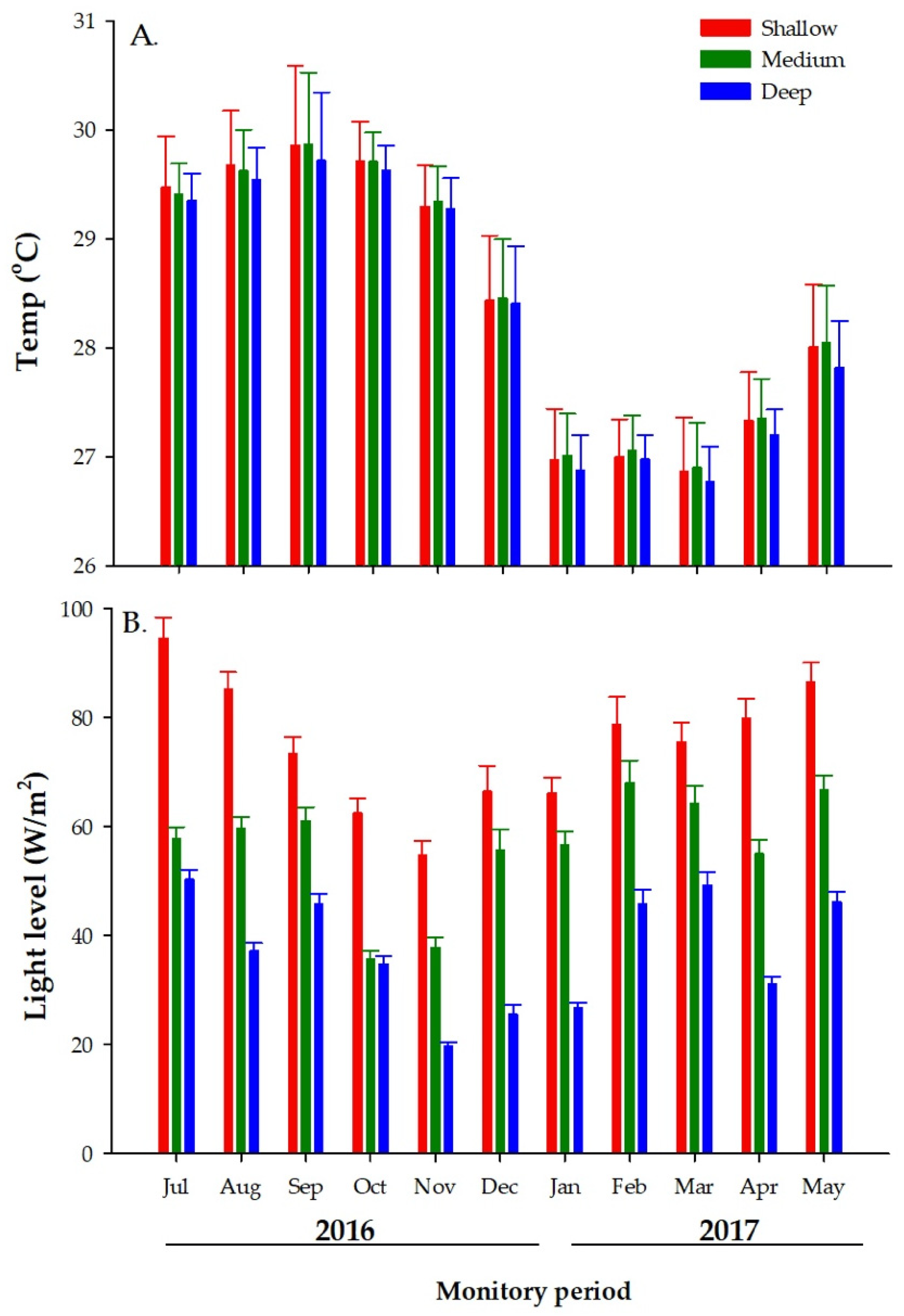
3.1. Environmental Measurements
3.1.1. Temperature
3.1.2. Light Levels
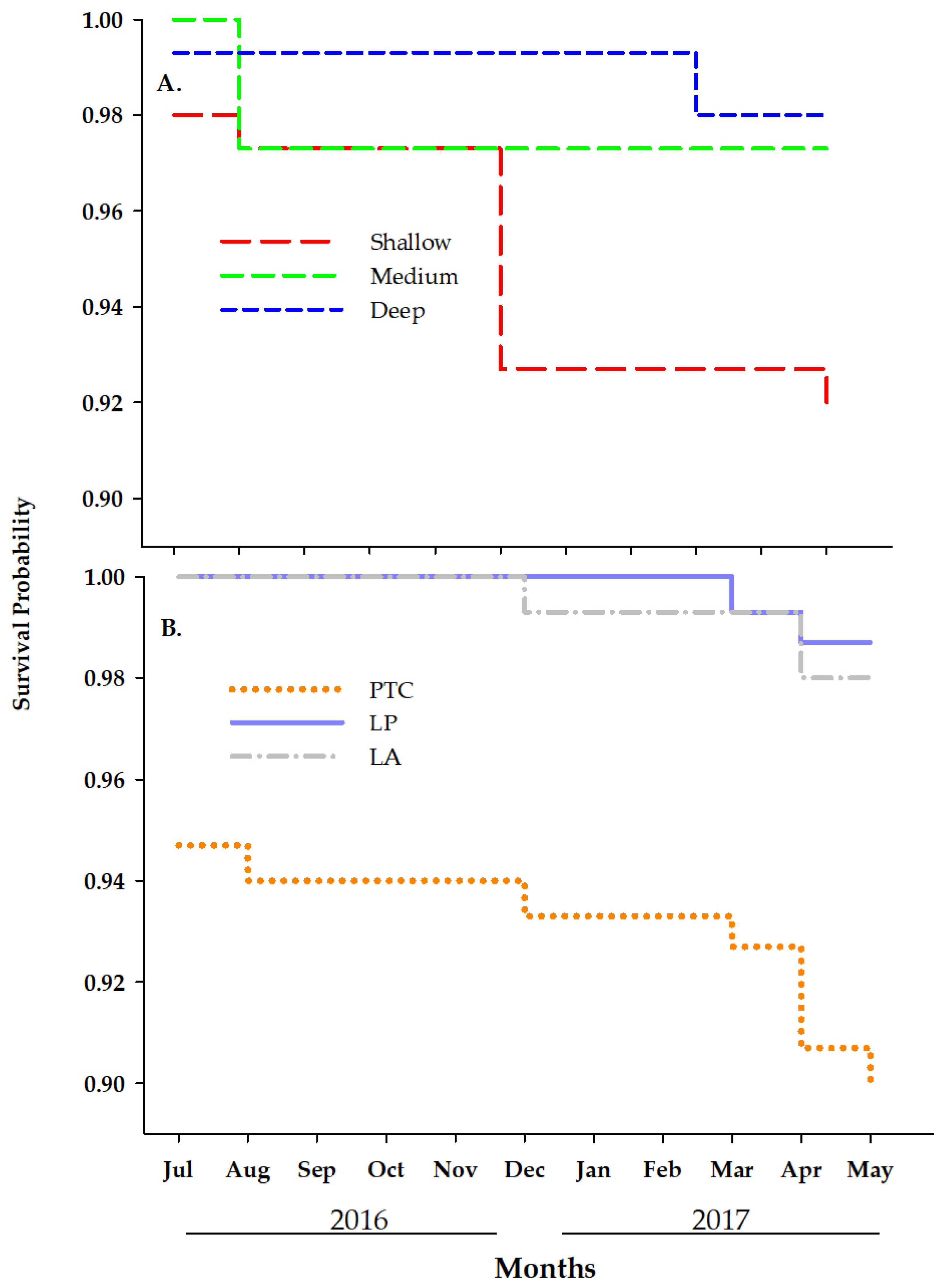
3.1.3. Survival
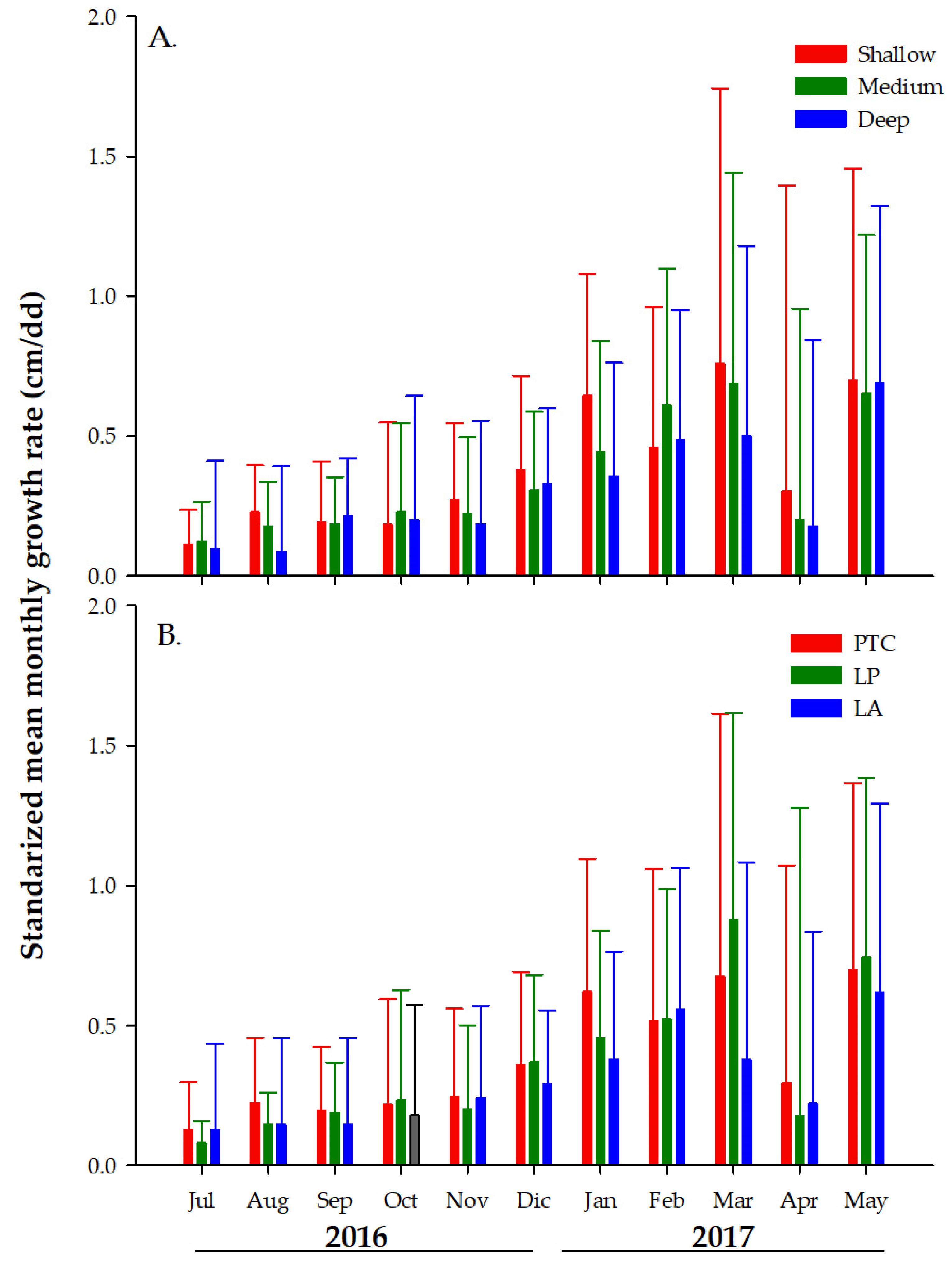
3.2. Monthly Growth Rate (MGR) of Colonies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gardner, T.A.; Côté, I.M.; Gill, J.A.; Grant, A.; Watkinson, A.R. Long-term region-wide declines in Caribbean corals. Science 2003, 301, 958–960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopéz-Peréz, A.; Culpul-Magananes, A.; Ahumada-Sempoal, A.; Medina-Rosas, P.; Reyes-Bonilla, H.; Herrero-Perézrul, M.D.; Reyes-Hernández, C.; Lara-Hernández, J. The coral communities of the Isla Marias archipelago Mexico; structure and biogeographic relevance to the Eastern Pacific. Mar. Ecol. 2016, 37, 679–690. [Google Scholar] [CrossRef]
- Cramer, K.L.; Jackson, J.B.C.; Donovan, M.K.; Greenstein, B.J.; Korpanty, C.A.; Cook, G.M.; Pandolfi, J.M. Widespread loss of Caribbean acroporid corals was underway before coral bleaching and disease outbreaks. Sci. Adv. 2020, 6, eaax9395. [Google Scholar] [CrossRef] [Green Version]
- Pandolfi, J.M.; Bradbury, R.H.; Sala, E.; Hughes, T.P.; Bjorndal, K.A.; Cooke, R.G.; McAdle, D.; McClenachan, L.; Newman, M.J.H.; Paredes, G.; et al. Global trajectories of the long-term decline of coral reef ecosystems. Science 2003, 301, 955–958. [Google Scholar] [CrossRef] [Green Version]
- Bellwood, D.R.; Hughes, T.P.; Folke, C.; Nyström, M. Confronting the coral reef crisis. Nature 2004, 429, 827–833. [Google Scholar] [CrossRef]
- Miller, M.; Bourque, A.; Bohnsack, J. An analysis of the loss of acroporid corals at Looe Key, Florida, USA: 1983–2000. Coral Reefs 2002, 21, 179–182. [Google Scholar] [CrossRef]
- Heron, S.F.; Maynard, J.A.; van Hooidonk, R.; Eakin, C.M. Warming trends and bleaching stress of the World’s coral reefs 1985–2012. Sci. Rep. 2016, 6, 38402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jokiel, P.L.; Coles, S.L. Response of Hawaiian and other Indo-Pacific Reef corals to elevated temperature. Coral Reefs 1990, 8, 155–162. [Google Scholar] [CrossRef]
- Reaser, J.K.; Pomerance, R.; Thomas, P.O. Coral bleaching and global climate change: Scientific findings and policy recommendations. Conserv. Biol. 2000, 14, 1500–1511. [Google Scholar] [CrossRef]
- Frade, P.R.; Bongaerts, P.; Englebert, N.; Rogers, A.; González-Rivero, M.; Hoegh-Guldberg, O. Deep reefs of the Great Barrier Reef offer limited thermal refuge during mass coral bleaching. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz-Diaz, C.P.; Toledo-Hernández, C.; Sabat, A.M.; Marcano, M. Immune response to a pathogen in corals. J. Theor. Biol. 2013, 332, 141–148. [Google Scholar] [CrossRef]
- Nieves-González, A.; Ruiz-Diaz, C.P.; Toledo-Hernández, C.; Ramírez-Lugo, J.S. A mathematical model of the interactions between Acropora cervicornis and its environment. Ecol. Model. 2019, 406, 7–22. [Google Scholar] [CrossRef]
- Keller, B.D.; Gleason, D.F.; McLeod, E.; Woodley, C.M.; Airame, S.; Causey, B.D.; Friedlander, A.M.; Grober-Dunsmore, R.G.; Johnson, J.E.; Miller, S.L.; et al. Climate change, coral reef ecosystems, and management options for marine protected areas. Environ. Manage. 2009, 44, 1069–1088. [Google Scholar] [CrossRef] [Green Version]
- Toledo-Hernández, C.; Yoshioka, P.; Bayman, P.; Sabat, A. Impact of disease and detachment on growth and survivorship of sea fans Gorgonia ventalina. Mar. Ecol. Progr. Ser. 2009, 393, 47–54. [Google Scholar] [CrossRef]
- O’Donnell, K.E.; Lohr, K.E.; Bartels, E.; Patterson, J.T. Evaluation of staghorn coral (Acropora cervicornis, Lamarck 1816) production techniques in an ocean-based nursery with consideration of coral genotype. J. Exp. Mar. Bio. Ecol. 2017, 487, 53–58. [Google Scholar] [CrossRef]
- Goreau, T.F.; Goreau, N.I. Coral Reef Project-Papers in memory of Dr. Thomas F. Goreau. 17. The Ecology of Jamaican Reefs. II. Geomorphology, Zonation, and Sedimentary Phases. Bull. Mar. Sci. 1973, 23, 399–464. [Google Scholar]
- Mercado-Molina, A.E.; Ruiz-Diaz, C.P.; Sabat, A.M. Branching dynamics of transplanted colonies of the threatened coral Acropora cervicornis: Morphogenesis, complexity, and modeling. J. Exp. Mar. Biol. Ecol. 2016, 482, 134–141. [Google Scholar] [CrossRef]
- Bruckner, A.W. Proceeding of the Caribbean Acropora Workshop: Potential Application of the U.S. Endangered Species Act as a Conservation Strategy; NOAA Technical Memorandum NMFS-OPR: Sliver Spring, MD, USA, 2002; 199p. [Google Scholar]
- Graham, N.A.J.; Nash, K.L. The importance of structural complexity in coral reef ecosystems. Coral Reefs 2003, 32, 315–326. [Google Scholar] [CrossRef]
- Yanovski, R.; Nelson, P.A.; Abelson, A. Structural Complexity in Coral Reefs: Examination of a Novel Evaluation Tool on Different Spatial Scales. Front. Ecol. Evol. 2017, 5, 27. [Google Scholar] [CrossRef] [Green Version]
- Jackson, J.; Donovan, M.; Cramer, K.; Lam, V. Status and Trends of Caribbean Coral Reefs 1970–2012; Global Coral Reef Monitoring Network, IUCN: Gland, Switzerland, 2014. [Google Scholar]
- Aronson, R.B.; Precht, W.F. White-band disease and the changing face of Caribbean coral reefs. Hydrobiologia 2001, 460, 25–38. [Google Scholar] [CrossRef]
- Goergen, E.A.; Ostroff, Z.; Gilliam, D.S. Genotype and attachment technique influence the growth and survival of line nursery corals. Restor. Ecol. 2017, 26, 622–628. [Google Scholar] [CrossRef]
- Young, C.N.; Schopmeyer, S.A.; Lirman, D. A review of reef restoration and coral propagation using the threatened genus Acropora in the Caribbean and Western Atlantic. Bull. Mar. Sci. 2012, 88, 1075–1098. [Google Scholar] [CrossRef] [Green Version]
- Lirman, D.; Schopmeyer, S.; Galvan, V.; Drury, C.; Baker, A.C.; Baums, I.B. Growth Dynamics of the Threatened Caribbean Staghorn Coral Acropora cervicornis: Influence of Host Genotype, Symbiont Identity, Colony Size, and Environmental Setting. PLoS ONE 2014, 9, e107253. [Google Scholar] [CrossRef]
- Mercado-Molina, A.; Ruiz-Diaz, C.P.; Sabat, A.M. Demographics and dynamics of two restored populations of the threatened reef-building coral Acropora cervicornis. J. Nat. Conserv. 2015, 24, 17–23. [Google Scholar] [CrossRef]
- Rinkevich, B. Restoration strategies for coral reefs damaged by recreational activities: The use of sexual and asexual recruits. Rest. Ecol. 1995, 3, 241–251. [Google Scholar] [CrossRef]
- Shaish, L.; Levy, G.; Gomez, E.; Rinkevich, B. Fixed and suspended coral nurseries in the Philippines: Establishing the first step in the gardening concept of reef restoration. J. Exp. Mar. Biol. Ecol. 2008, 358, 86–97. [Google Scholar] [CrossRef]
- Levy, G.; Shaish, L.; Haim, A.; Rinkevich, B. Mid-water rope nursery- testing design and performance of a novel reef restoration instrument. Ecol. Eng. 2010, 36, 560–569. [Google Scholar] [CrossRef]
- Baums, I.B.; Baker, A.C.; Davies, W.S.; Grottoli, A.G.; Kenkel, C.D.; Kitchen, S.A.; Kuffner, I.B.; LaJeunesse, T.C.; Matz, M.V.; Miller, M.W.; et al. Considerations for maximizing the adaptive potential of restored coral populations in the western Atlantic. Ecol. Appl. 2019, 29, e01978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shafir, S.; Abady, S.; Rinkevich, B. Improved sustainable maintenance for mid-water coral nursery by the application of an anti- fouling agent. J. Exp. Mar. Biol. Ecol. 2009, 368, 124–128. [Google Scholar] [CrossRef]
- Griffin, S.; Spathias, H.; Moore, T.; Baums, I.; Griffin, B.A. Scaling up Acropora nurseries in the Caribbean and improving techniques. In Proceedings of the 12th International Coral Reef Symposio, Cairns, Australia, 9–13 July 2012. [Google Scholar]
- Putchim, L.; Thongtham, N.; Hewett, A.; Chansang, H. Survival and growth of Acropora spp. in-water nursery and after transplantation at Phi Phi Islands, Andaman Sea Thailand. In Proceedings of the 11th International Coral Reef Symposium, Fort Lauderdale, FL, USA, 7–11 July 2008. [Google Scholar]
- Foo, S.A.; Asner, G.P. Sea surface temperature in coral reef restoration outcomes. Environ. Res. Lett. 2020, 15, 074045. [Google Scholar] [CrossRef]
- Bridge, T.C.L.; Hoey, A.S.; Campbell, S.J.; Muttaqin, E.; Rudi, E.; Fadli, N.; Baird, A.H. Depth-dependent mortality of reef corals following a severe bleaching event: Implications for thermal refuges and population recovery [version 3; peer review: 2 approved, 1 approved with reservations]. F1000Research 2014, 2, 187. [Google Scholar] [CrossRef]
- Enochs, I.C.; Manzello, D.P.; Carlton, R.; Schopmyeyer, S.; van Hooidonk, R.; Lirman, D. Effects of light and elevated pCO2 on the growth and photochemical efficiency of Acropora cervicornis. Coral Reefs 2014, 33, 477–485. [Google Scholar] [CrossRef]
- Therneau, T.M.; Grambsch, P.M. Modeling Survival Data: Extending the Cox Model; Springer: New York, NY, USA, 2000; ISBN 0-387-98784-3. [Google Scholar]
- Kohler, K.E.; Gill, S.M. Coral Point Count with Excel extensions (CPCe): A Visual Basic program for the determination of coral and substrate coverage using random point count methodology. Comput. Geosci. 2006, 32, 1259–1269. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environmental for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Rowan, R.; Knowlton, N.; Baker, A.C.; Baker, A.; Jara, J. Landscape ecology of algal symbionts creates variation in episodes of coral bleaching. Nature 1997, 388, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Marshall, P.A.; Baird, A.H. Bleaching of corals on the Great Barrier Reef: Differential susceptibilities among taxa. Coral Reefs 2000, 19, 155–163. [Google Scholar] [CrossRef]
- Morais, J.; Santos, B.A. Limited potential of deep reefs to serve as refuges for tropical southwestern Atlantic corals. Ecosphere 2018, 9, e02281. [Google Scholar] [CrossRef] [Green Version]
- Schopmeyer, S.A.; Lirman, D.; Bertels, E.; Gilliem, D.S.; Goergen, E.A.; Griffin, S.P.; Johnson, M.E.; Lustic, C.; Maxwell, K.; Walter, C.S. Regional restoration benchmarks for Acropora cervicornis. Coral Reefs 2017, 36, 1047–1057. [Google Scholar] [CrossRef]
- Muir, P.R.; Marshall, P.A.; Abdulla, A.; Aguirre, J.D.; Muir, P.R. Species identity and depth predict bleaching severity in reef-building corals: Shall the deep inherit the reef? Proc. R. Soc. B 2017, 284, 20171551. [Google Scholar] [CrossRef]
- Riegl, B.; Piller, W.E. Possible refugia for reefs in times of environmental stress. Int. J. Earth. Sci. 2003, 92, 520–531. [Google Scholar] [CrossRef]
- Glynn, P.W. Coral reef bleaching: Facts, hypotheses, and implications. Glob. Chang. Biol. 1996, 2, 495–509. [Google Scholar] [CrossRef]
- Palmer, C.V.; Traylor-Knowles, N. Towards an integrated network of coral immune mechanisms. Proc. R. Soc. B Biol. Sci. 2012, 279, 4106–4114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drury, C.; Manzello, D.; Lirman, D. Genotype and local environment dynamically influence growth, disturbance response and survivorship in the threatened coral, Acropora cervicornis. PLoS ONE 2017, 12, e0174000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quinn, N.J.; Kojis, B.L. Evaluationg the potential of natural preproductiona dnarticical techniques to increase Acropora cervicornis population at Discovery Bay, Jamaica. Rev. Biol. Trop. 2010, 54, 105–116. [Google Scholar]
- Bowden-Kerby, A.; Quinn, N.; Stennet, M.; Mejia, A. Acropora cervicornis Restoration to Support Coral Reef Conservation in the Caribbean. NOAA Coast. Zone 2012, 5, 7–10. [Google Scholar]
- Hackerrott, S.; Martell, H.A.; Eirin-López, J.M. Coral environmental memory: Causes, mechanisms, and consequences for future reefs. Trends Ecol. Evol. 2021, 36, 1011–1016. [Google Scholar] [CrossRef]
- Baker, P.A.; Weber, J.N. Coral growth rate: Variation with depth. Earth Planet. Sci. Lett. 1975, 27, 57–61. [Google Scholar] [CrossRef]
- Budd, A.F.; Fukami, H.; Smith, N.D.; Knowlton, N. Taxonomic classification of the reef coral family Mussidae (Cnidaria: Anthozoa: Scleractinia). Zool. J. Linn. Soc. 2012, 166, 465–529. [Google Scholar] [CrossRef] [Green Version]
- Highsmith, R.C. Coral growth rates and environmental control of density banding. J. Exp. Mar. Biol. Ecol. 1979, 37, 105–125. [Google Scholar] [CrossRef]
- Huston, M. Variation in coral growth rates with depth at Discovery Bay, Jamaica. Coral Reefs 1985, 4, 19–25. [Google Scholar] [CrossRef]
- Kinzie, R.A., III. The zonation of West Inidan gorgonians. Bull. Mar. Sci. 1973, 23, 93–155. [Google Scholar]
- Birkeland, C. The importance of rate of biomass accumulation in early successional stage of benthic communities to the survival of coral recruit. In Proceedings of the Third International Coral Reef Symposium, Miami, FL, USA, 1 January 1977; University of Miami: Miami, FL, USA; Volume 1, pp. 5–21. [Google Scholar]
- Kahng, S.E.; Watanabe, T.K.; Hu, H.M.; Watanabe, T.; Shen, C.C. Moderate zooanthellate coral growth rates in the lower photic zone. Coral Reef 2020, 39, 1273–1284. [Google Scholar] [CrossRef]
- Torres, J.L.; Armstrong, R.A.; Corredor, J.E.; Gilbes, F. Physiological Responses of Acropora cervicornis to Increased Solar Irradiance. Photochem. Photobiol. 2007, 83, 839–850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weil, E.; Hammerman, N.M.; Becicka, R.L.; Cruz-Motta, J.J. Growth dynamics in Acropora cervicornis and A. prolifera in southwest Puerto Rico. PeerJ 2020, 8, e8435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernardo, V.-A.; Colley, S.B.; Hoke, S.M.; Thomas, J.D. The reproductive seasonality and gametogenic cycle of Acropora cervicornis off Broward County, Florida, USA. Coral Reefs 2006, 25, 110–122. [Google Scholar]
- Toledo-Hernández, C.; Ruiz-Díaz, C.P.; Hernández-Delgado, E.; Suelimán, S. Devastation of 15-year old community-based coral farming and reef restoration sites in Puerto Rico by major Hurricanes Irma and María. Carb. Nat. 2018, 53, 1–6. [Google Scholar]
- Carrick, J.; Lustic, C.; Lirman, D.; Schopmeyer, S.; Bartels, E.; Burdeno, D.; Dahlgren, C.; Galvan, V.M.; Gilliam, D.; Goergen, L.; et al. Hurricane Impacts on Reef Restoration: The Good, Bad and the Ugly. In Activate Coral Restoration, Techniques for a Changing Planet, 1st ed.; Vaughan, D.E., Ed.; J. Ross Publishing: Fort Lauderdale, FL, USA, 2021; Volume 1, pp. 483–510. [Google Scholar]


| Variable | Df | F Value | p-Value |
|---|---|---|---|
| Temperature °C | |||
| Month | 10 | 2881.47 | 2 × 10−16 *** |
| Depth | 2 | 35.95 | 1 × 10−5 ** |
| Light Levels (W/m2) | |||
| Month | 10 | 7.79 | 5.61 × 10−5 ** |
| Depth | 2 | 103.25 | 2.88 × 10−11 ** |
| Model Variable (Fixed Effects) | Estimate | t Value | p |
|---|---|---|---|
| Intercept | −0.214 | −2.796 | 0.005 ** |
| Month | 0.056 | 4.824 | 1.4 × 10−6 *** |
| Depth Deep | −0.186 | −2.781 | 0.005 ** |
| Depth Medium | −0.275 | −4.917 | 9.15 × 10−7 *** |
| Temperature | 0.303 | −5.375 | 8.06 × 10−8 *** |
| Light Levels | −0.121 | −2.585 | 0.009 ** |
| Locality PTC | 0.185 | 3.324 | 0.001 ** |
| Locality LA | 0.14 | 2.734 | 0.006 ** |
| Temperature: PTC | −0.123 | −3.08 | 0.002 ** |
| Temperature: LA | −0.169 | −4.608 | 4.44 × 10−6 *** |
| Temperature: Light Level | 0.177 | 2.81 | 0.005 ** |
| Deep: Temperature | 0.26 | 2.876 | 0.004 ** |
| Medium: Temperature | 0.279 | 4.315 | 1.64 × 10−5 *** |
| Deep: Light Levels | 0.252 | 4.208 | 2.63 × 10−5 *** |
| Medium: Light Levels | 0.311 | 5.108 | 3.41 × 10−7 *** |
| Deep: Temperature: Light Levels | −0.244 | −3.395 | 0.00067 *** |
| Medium: Temperature: Light Level | −0.368 | −4.765 | 1.96 × 10−6 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Diaz, C.P.; Toledo-Hernández, C.; Sánchez-González, J.L.; Betancourt, B. The Effects of Depth-Related Environmental Factors on Traits in Acropora cervicornis Raised in Nurseries. Water 2022, 14, 212. https://doi.org/10.3390/w14020212
Ruiz-Diaz CP, Toledo-Hernández C, Sánchez-González JL, Betancourt B. The Effects of Depth-Related Environmental Factors on Traits in Acropora cervicornis Raised in Nurseries. Water. 2022; 14(2):212. https://doi.org/10.3390/w14020212
Chicago/Turabian StyleRuiz-Diaz, Claudia Patricia, Carlos Toledo-Hernández, Juan Luis Sánchez-González, and Brenda Betancourt. 2022. "The Effects of Depth-Related Environmental Factors on Traits in Acropora cervicornis Raised in Nurseries" Water 14, no. 2: 212. https://doi.org/10.3390/w14020212
APA StyleRuiz-Diaz, C. P., Toledo-Hernández, C., Sánchez-González, J. L., & Betancourt, B. (2022). The Effects of Depth-Related Environmental Factors on Traits in Acropora cervicornis Raised in Nurseries. Water, 14(2), 212. https://doi.org/10.3390/w14020212







