Investigating the Electrocoagulation Treatment of Landfill Leachate by Iron/Graphite Electrodes: Process Parameters and Efficacy Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Characterization of Yasuj Landfill Leachate
2.2. Experimental Design
2.3. Sample Preparation for Analysis
2.4. Physical and Chemical Analytical Details
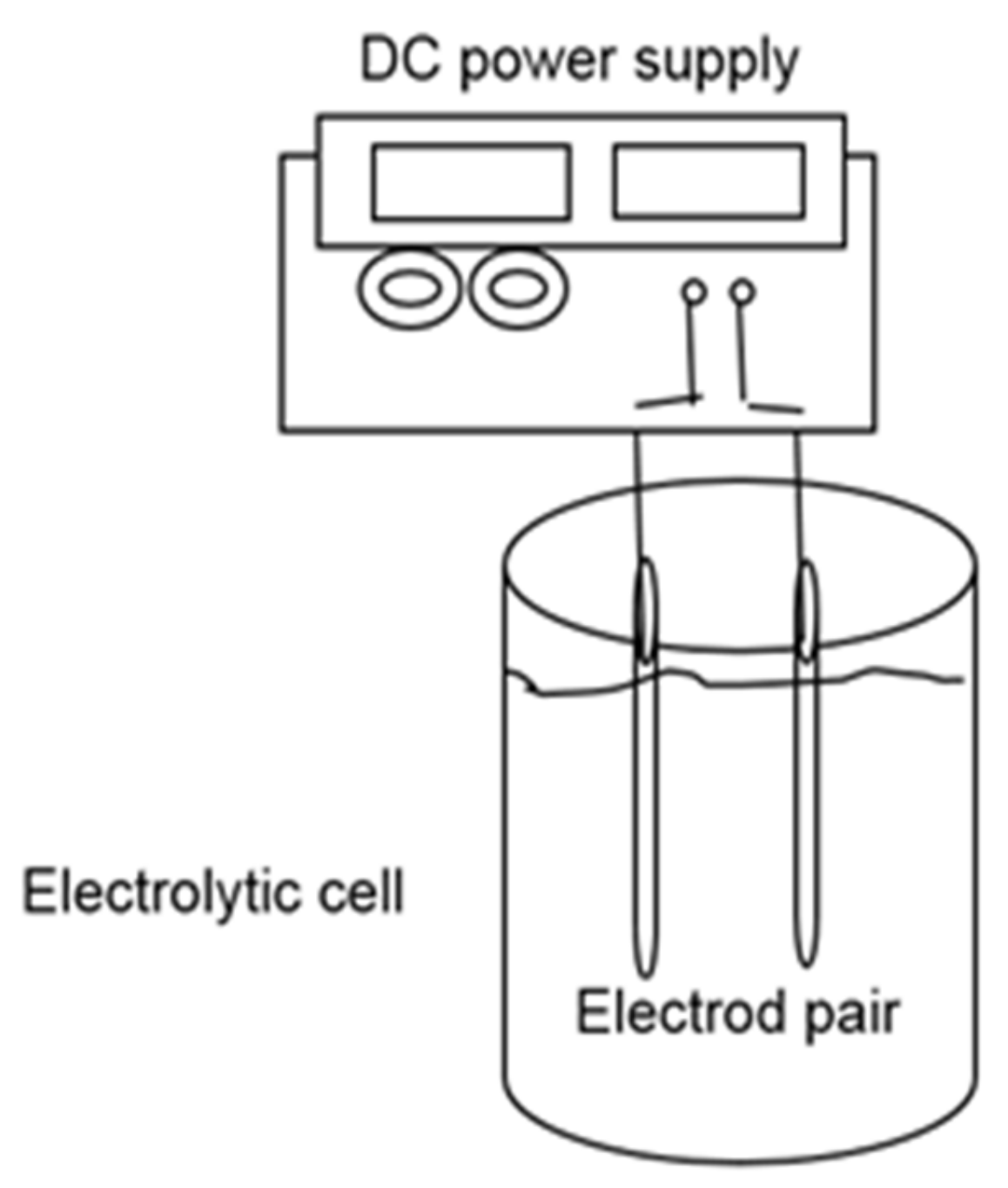
2.5. Electrocoagulation Monitoring
2.6. Statistical Analysis
3. Results and Discussion
3.1. Characteristics of the Yasuj Leachate
3.2. Leachate Treatment
3.2.1. Chemical Flocculation
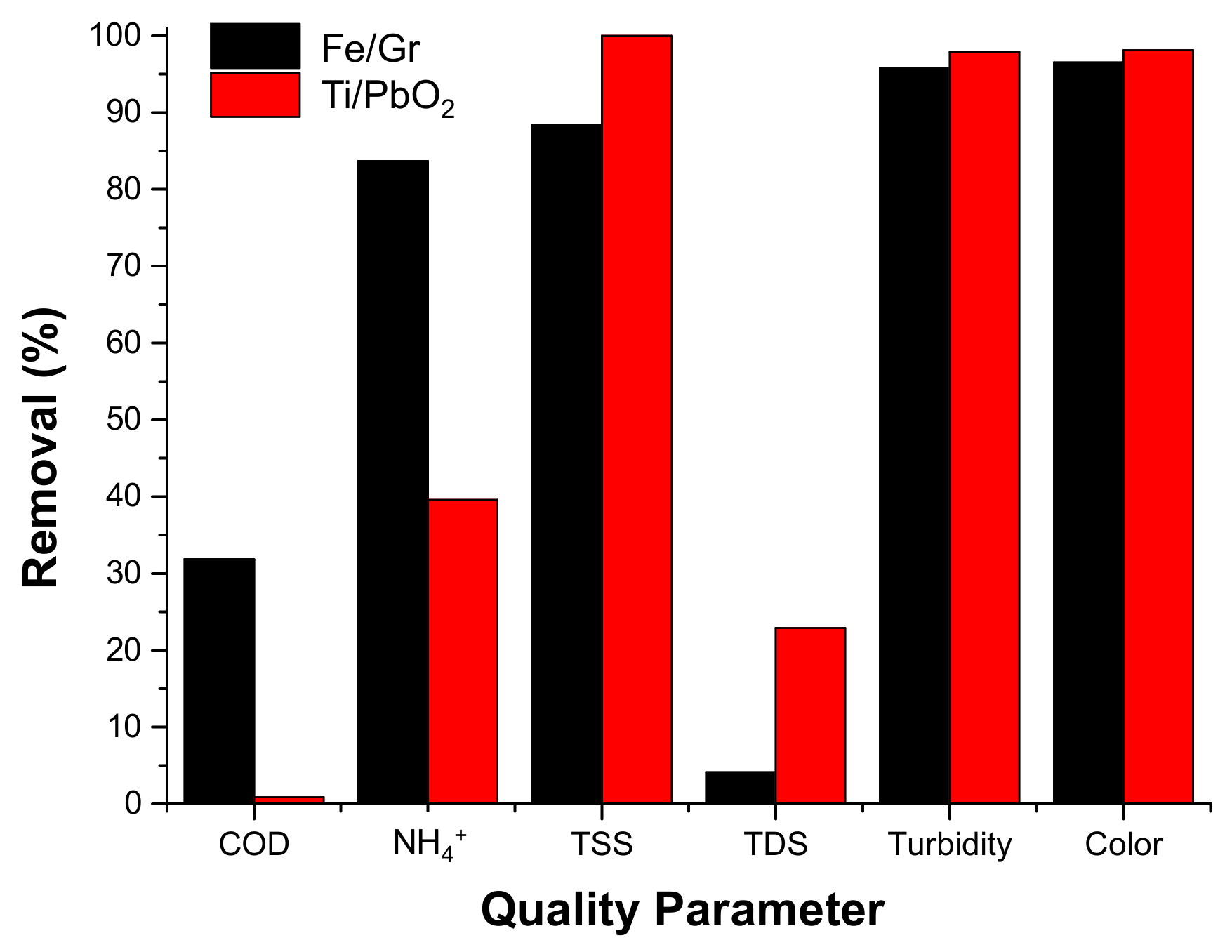
3.2.2. Electrocoagulation Process
3.2.3. Effect of Operational Parameters on the Treatment Efficacy
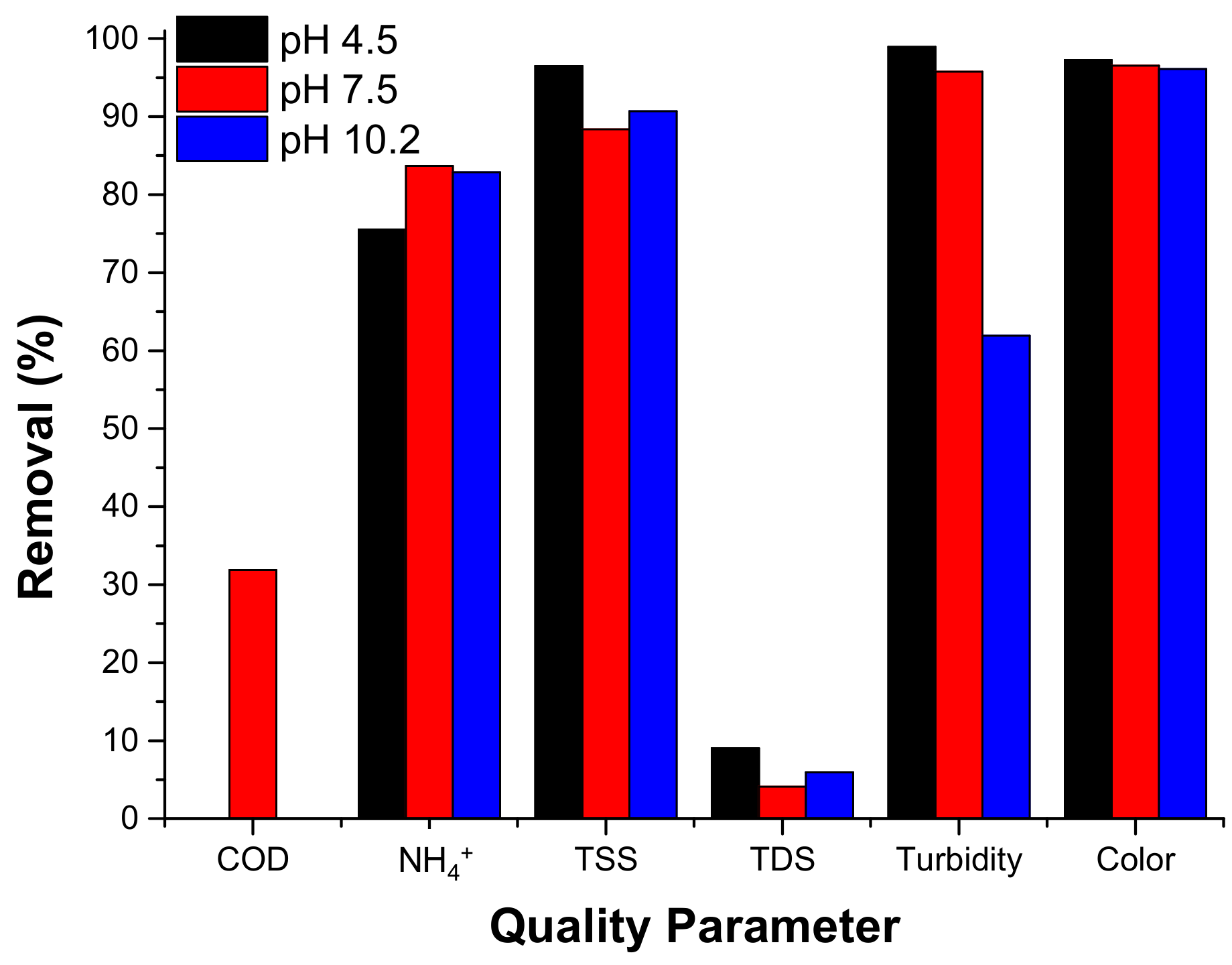
3.2.3.1. pH Variation
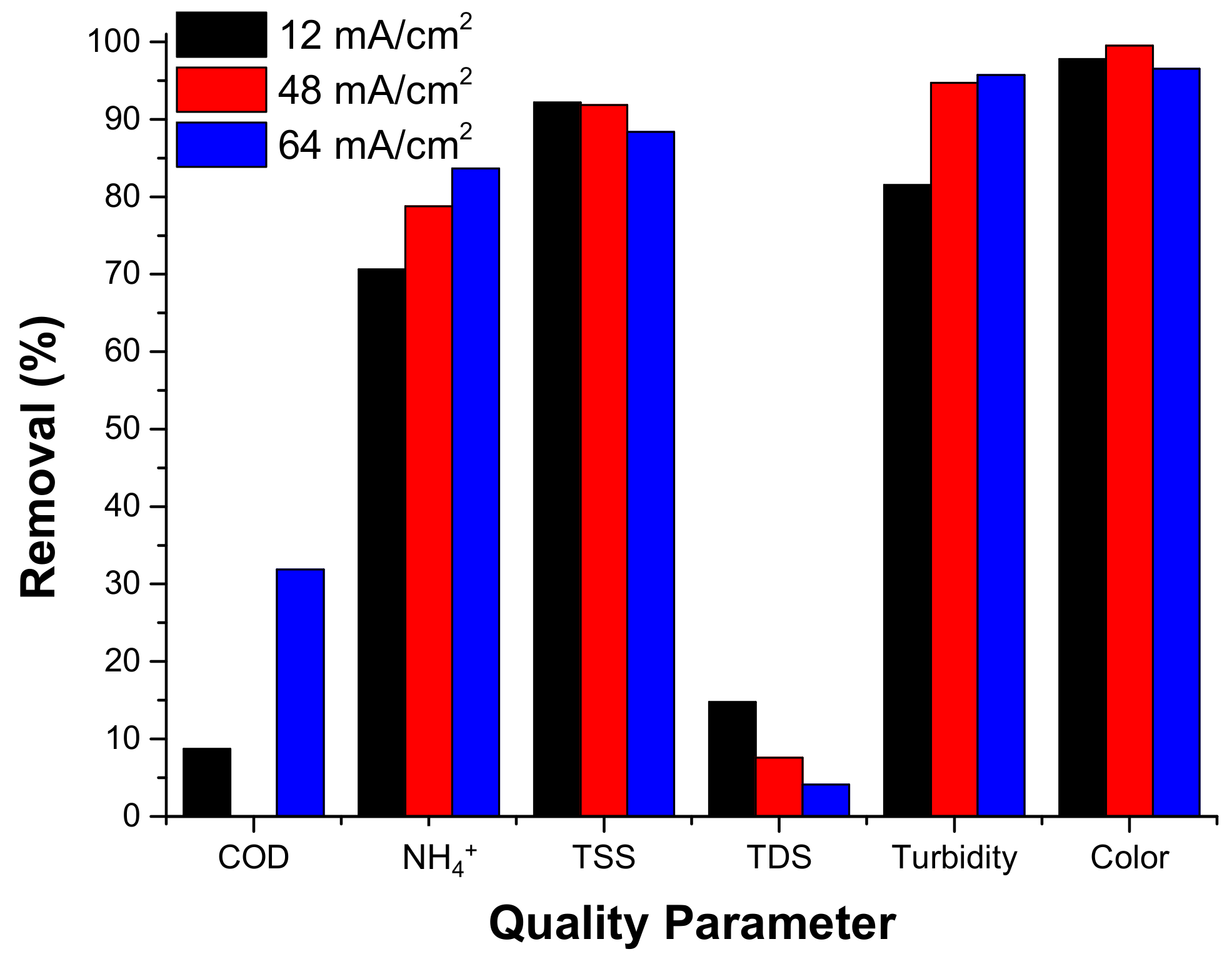
3.2.3.2. Current Density Effect
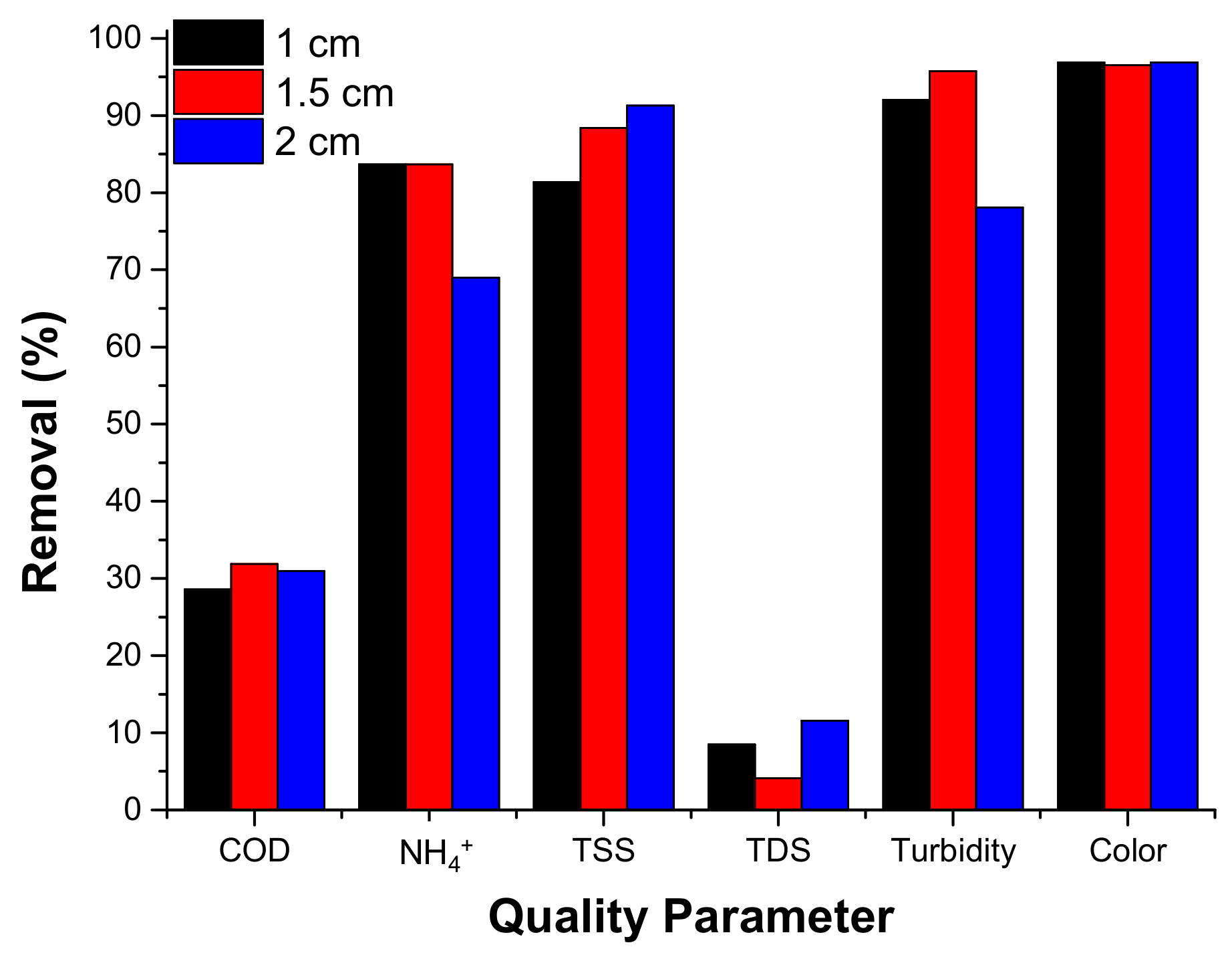
3.2.3.3. Effect of the Inter-Electrode Distance
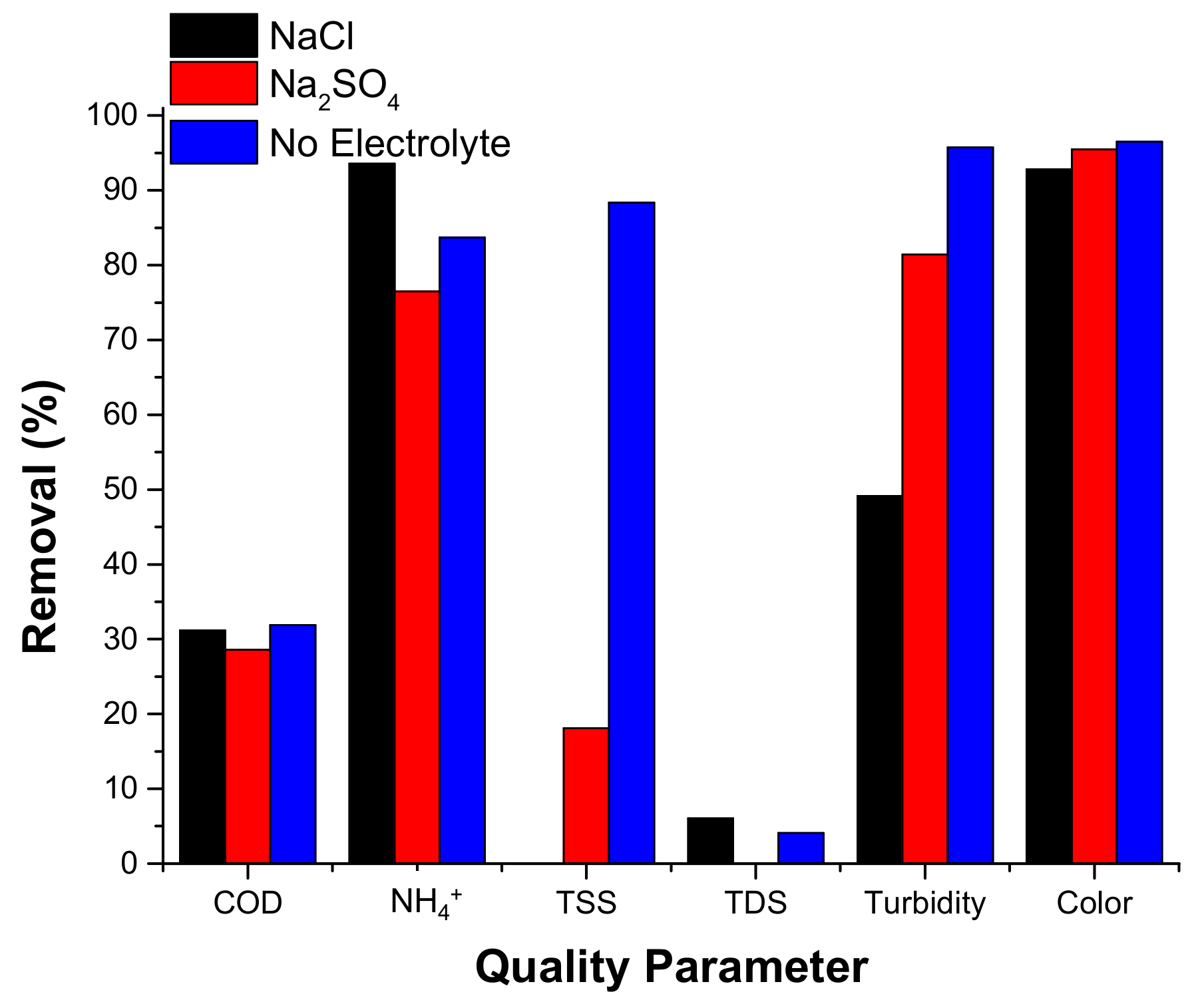
3.2.3.4. Electrolyte Type
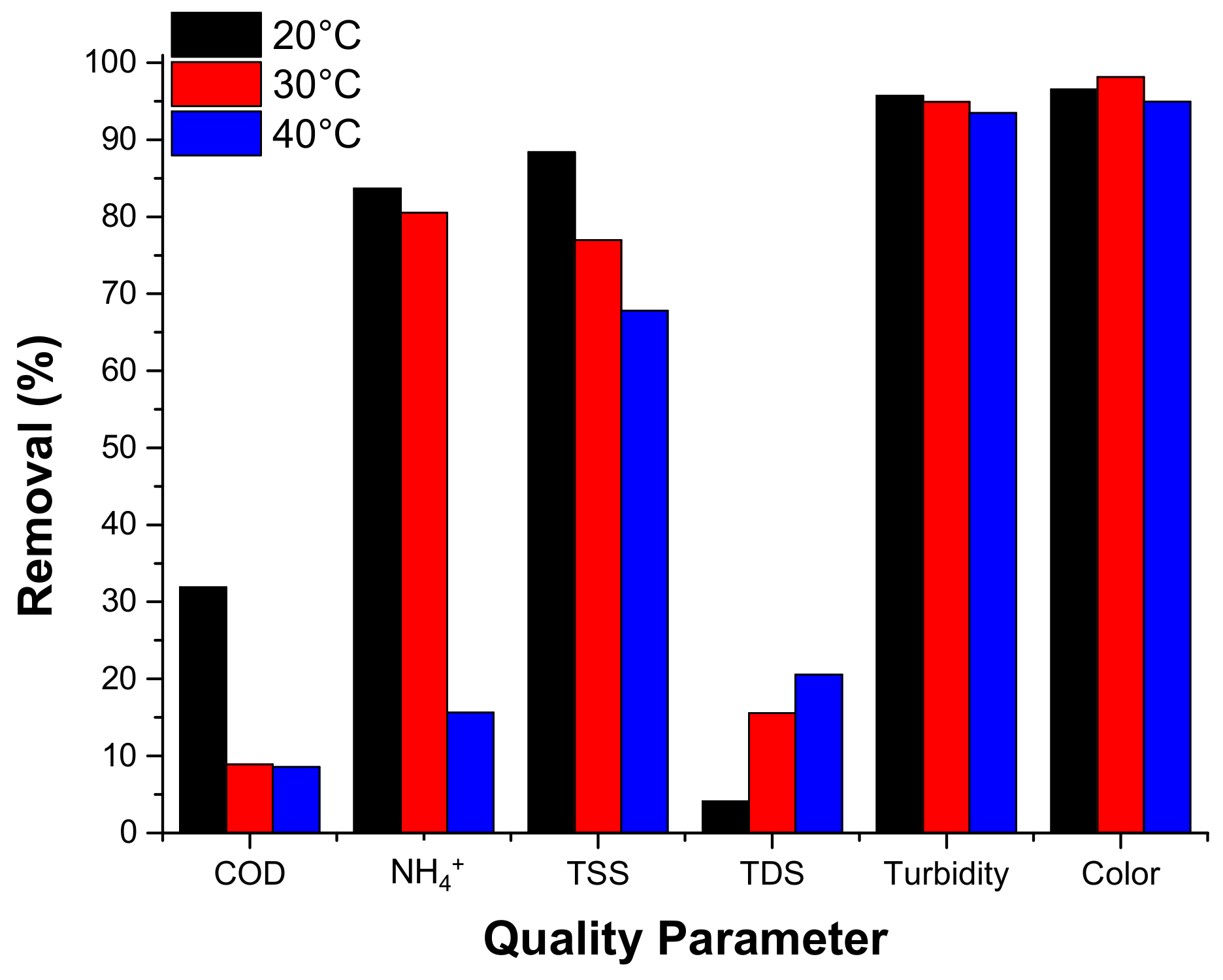
3.2.3.5. Temperature Effect
3.2.3.6. The Effect of Electrolysis Time
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Electrical Coagulation Stage (Stage 2)
References
- Dang, Y.; Lei, Y.; Liu, Z.; Xue, Y.; Sun, D.; Wang, L.-Y.; Holmes, D.E. Impact of fulvic acids on bio-methanogenic treatment of municipal solid waste incineration leachate. Water Res. 2016, 106, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, F.; Samaei, M.R.; Khodadadi, H.; Karimi, A.; Maleknia, H. Effects of materials recovery facility construction on the release of fungal bioaerosols: A case study in southern of Iran. Fresenius Environ. Bull. 2016, 25, 1513–1519. [Google Scholar]
- Moreira, F.C.; Soler, J.; Fonseca, A.; Saraiva, I.; Boaventura, R.A.; Brillas, E.; Vilar, V.J. Incorporation of electrochemical advanced oxidation processes in a multistage treatment system for sanitary landfill leachate. Water Res. 2015, 81, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Moody, C.M.; Townsend, T.G. A comparison of landfill leachates based on waste composition. Waste Manag. 2017, 63, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Nurisepehr, M.; Jorfi, S.; Kalantary, R.R.; Akbari, H.; Soltani, R.D.C.; Samaei, M.R. Sequencing treatment of landfill leachate using ammonia stripping, Fenton oxidation and biological treatment. Waste Manag. Res. 2012, 30, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, B.K.; Kumar, M. Sequential coagulation/flocculation and microwave-persulfate processes for landfill leachate treatment: Assessment of bio-toxicity, effect of pretreatment and cost-analysis. Waste Manag. 2019, 85, 18–29. [Google Scholar] [CrossRef]
- Kjeldsen, P.; Barlaz, M.A.; Rooker, A.P.; Baun, A.; Ledin, A.; Christensen, T.H. Present and Long-Term Composition of MSW Landfill Leachate: A Review. Crit. Rev. Environ. Sci. Technol. 2002, 32, 297–336. [Google Scholar] [CrossRef]
- Costa, A.M.; Alfaia, R.G.D.S.M.; Campos, J.C. Landfill leachate treatment in Brazil—An overview. J. Environ. Manag. 2019, 232, 110–116. [Google Scholar] [CrossRef]
- Renou, S.; Givaudan, J.; Poulain, S.; Dirassouyan, F.; Moulin, P. Landfill leachate treatment: Review and opportunity. J. Hazard. Mater. 2008, 150, 468–493. [Google Scholar] [CrossRef]
- Abbasi, F.; Samaei, M.R.; Azhdarpoor, A.; Jalili, M.; Maleknia, H.; Mehdizadeh, A. Removal, optimization and kinetic modelling of high concentration of methyl tertiary butyl ether from aqueous solutions using copper oxide nanoparticles and hydrogen peroxide. Desalination Water Treat. 2020, 181, 278–288. [Google Scholar] [CrossRef]
- Abbasi, F.; Samaei, M.R.; Manoochehri, Z.; Jalili, M.; Yazdani, E. The effect of incubation temperature and growth media on index microbial fungi of indoor air in a hospital building in Shiraz, Iran. J. Build. Eng. 2020, 31, 101294. [Google Scholar] [CrossRef]
- Rezaie, E.; Sadeghi, M.; Khoramabadi, G.S. A Study on the leachate treatment by using electrochemical process. J. Res. Environ. Health 2016, 1, 297–305. [Google Scholar]
- Eslami, H.; Samaei, M.R.; Shahsavani, E.; Ebrahimi, A.A. Biodegradation and fate of linear alkylbenzene sulfonate in integrated fixed-film activated sludge using synthetic media. Desalination Water Treat. 2017, 92, 128–133. [Google Scholar] [CrossRef] [Green Version]
- Chemlal, R.; Tassist, A.; Drouiche, M.; Lounici, H.; Mameri, N. Microbiological aspects study of bioremediation of diesel-contaminated soils by biopile technique. Int. Biodeterior. Biodegrad. 2012, 75, 201–206. [Google Scholar] [CrossRef]
- Fudala-Ksiazek, S.; Sobaszek, M.; Luczkiewicz, A.; Pieczynska, A.; Ofiarska, A.; Fiszka-Borzyszkowska, A.; Sawczak, M.; Ficek, M.; Bogdanowicz, R.; Siedlecka, E. Influence of the boron doping level on the electrochemical oxidation of raw landfill leachates: Advanced pre-treatment prior to the biological nitrogen removal. Chem. Eng. J. 2018, 334, 1074–1084. [Google Scholar] [CrossRef]
- Miao, L.; Yang, G.; Tao, T.; Peng, Y. Recent advances in nitrogen removal from landfill leachate using biological treatments—A review. J. Environ. Manag. 2019, 235, 178–185. [Google Scholar] [CrossRef]
- Ding, J.; Wang, K.; Wang, S.; Zhao, Q.; Wei, L.; Huang, H.; Yuan, Y.; Dionysiou, D. Electrochemical treatment of bio-treated landfill leachate: Influence of electrode arrangement, potential, and characteristics. Chem. Eng. J. 2018, 344, 34–41. [Google Scholar] [CrossRef]
- Samadi, M.; Saghi, M.; Rahmani, A.; Hasanvand, J.; Rahimi, S.; Syboney, M.S. Hamadan landfill leachate treatment by coagulation-flocculation process. J. Environ. Health Sci. Eng. 2010, 7, 253–258. [Google Scholar]
- Teh, C.Y.; Budiman, P.M.; Shak, K.P.Y.; Wu, T.Y. Recent Advancement of Coagulation–Flocculation and Its Application in Wastewater Treatment. Ind. Eng. Chem. Res. 2016, 55, 4363–4389. [Google Scholar] [CrossRef]
- Deng, Y.; Zhu, X.; Chen, N.; Feng, C.; Wang, H.; Kuang, P.; Hu, W. Review on electrochemical system for landfill leachate treatment: Performance, mechanism, application, shortcoming, and improvement scheme. Sci. Total Environ. 2020, 745, 140768. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, Y.; Jia, H.; Guo, J.; Meng, X.; Wang, J. Electrochemical methods for landfill leachate treatment: A review on electrocoagulation and electrooxidation. Sci. Total Environ. 2022, 806, 150529. [Google Scholar] [CrossRef]
- Liu, X.; Novak, J.T.; He, Z. Removal of landfill leachate ultraviolet quenching substances by electricity induced humic acid precipitation and electrooxidation in a membrane electrochemical reactor. Sci. Total Environ. 2019, 689, 571–579. [Google Scholar] [CrossRef]
- Liu, X.; Novak, J.T.; He, Z. Synergistically coupling membrane electrochemical reactor with Fenton process to enhance landfill leachate treatment. Chemosphere 2020, 247, 125954. [Google Scholar] [CrossRef]
- Maldonado, V.Y.; Landis, G.M.; Ensch, M.; Becker, M.F.; Witt, S.E.; Rusinek, C.A. A flow-through cell for the electrochemical oxidation of perfluoroalkyl substances in landfill leachates. J. Water Process. Eng. 2021, 43, 102210. [Google Scholar] [CrossRef]
- Pierangeli, G.M.F.; Ragio, R.A.; Benassi, R.F.; Gregoracci, G.B.; Subtil, E.L. Pollutant removal, electricity generation and microbial community in an electrochemical membrane bioreactor during co-treatment of sewage and landfill leachate. J. Environ. Chem. Eng. 2021, 9, 106205. [Google Scholar] [CrossRef]
- Tejera, J.; Hermosilla, D.; Gascó, A.; Miranda, R.; Alonso, V.; Negro, C.; Blanco, Á. Treatment of mature landfill leachate by electrocoagulation followed by Fenton or UVA-LED photo-Fenton processes. J. Taiwan Inst. Chem. Eng. 2021, 119, 33–44. [Google Scholar] [CrossRef]
- Azhdarpoor, A.; Abbasi, L.; Samaei, M.R. Investigation of a new double-stage aerobic-anoxic continuous-flow cyclic baffled bioreactor efficiency for wastewater nutrient removal. J. Environ. Manag. 2018, 211, 1–8. [Google Scholar] [CrossRef]
- Feng, H.; Chen, Z.; Wang, X.; Chen, S.; Crittenden, J. Electrochemical advanced oxidation for treating ultrafiltration effluent of a landfill leachate system: Impacts of organics and inorganics and economic evaluation. Chem. Eng. J. 2021, 413, 127492. [Google Scholar] [CrossRef]
- Pierpaoli, M.; Jakobczyk, P.; Sawczak, M.; Łuczkiewicz, A.; Fudala-Książek, S.; Bogdanowicz, R. Carbon nanoarchitectures as high-performance electrodes for the electrochemical oxidation of landfill leachate. J. Hazard. Mater. 2021, 401, 123407. [Google Scholar] [CrossRef]
- Sato, Y.; Zeng, Q.; Meng, L.; Chen, G. Importance of Combined Electrochemical Process Sequence and Electrode Arrangements: A Lab-scale Trial of Real Reverse Osmosis Landfill Leachate Concentrate. Water Res. 2021, 192, 116849. [Google Scholar] [CrossRef]
- Zielińska, M.; Kulikowska, D.; Stańczak, M. Adsorption—Membrane process for treatment of stabilized municipal landfill leachate. Waste Manag. 2020, 114, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Brasil, Y.L.; Moreira, V.R.; Lebron, Y.A.; Moravia, W.G.; Amaral, M.C. Combining yeast MBR, Fenton and nanofiltration for landfill leachate reclamation. Waste Manag. 2021, 132, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.D.M.E.; Alves, V.M.; Dantas, E.R.; Scotti, L.; Lopes, W.S.; Muratov, E.N.; Scotti, M.T. Chemical safety assessment of transformation products of landfill leachate formed during the Fenton process. J. Hazard. Mater. 2021, 419, 126438. [Google Scholar] [CrossRef] [PubMed]
- Soares, L.; Dal-Bó, A.G.; Bernardin, A.M. Use of enameling wastewater in the wet milling process for ‘monoporosa’ tile composition. Clean. Eng. Technol. 2021, 5, 100338. [Google Scholar] [CrossRef]
- Asaithambi, P.; Govindarajan, R.; Yesuf, M.B.; Alemayehu, E. Removal of color, COD and determination of power consumption from landfill leachate wastewater using an electrochemical advanced oxidation processes. Sep. Purif. Technol. 2020, 233, 115935. [Google Scholar] [CrossRef]
- Veli, S.; Arslan, A.; Isgoren, M.; Bingol, D.; Demiral, D. Experimental design approach to COD and color removal of landfill leachate by the electrooxidation process. Environ. Challenges 2021, 5, 100369. [Google Scholar] [CrossRef]
- Shadi, A.M.H.; Kamaruddin, M.A.; Emmanuela, M.I.; Niza, N.M. Performance of electroflotation on the treatment of landfill leachate and combined with other methods: Recent studies. Int. J. Environ. Eng. 2020, 10, 374. [Google Scholar] [CrossRef]
- Deepa, M.; Revathy, P.; Student, P. Validation of Document Clustering based on Purity and Entropy measures. Int. J. Adv. Res. Comput. Commun. Eng. 2012, 1, 147–152. [Google Scholar]
- Fernandes, A.; Santos, D.; Pacheco, M.J.; Ciríaco, L.; Lopes, A. Nitrogen and organic load removal from sanitary landfill leachates by anodic oxidation at Ti/Pt/PbO2, Ti/Pt/SnO2-Sb2O4 and Si/BDD. Appl. Catal. B Environ. 2014, 148, 288–294. [Google Scholar] [CrossRef]
- Ghanbari, F.; Wang, Q.; Hassani, A.; Wacławek, S.; Rodríguez-Chueca, J.; Lin, K.-Y.A. Electrochemical activation of peroxides for treatment of contaminated water with landfill leachate: Efficacy, toxicity and biodegradability evaluation. Chemosphere 2021, 279, 130610. [Google Scholar] [CrossRef]
- Morozesk, M.; Bonomo, M.; Rocha, L.; Duarte, I.; Zanezi, E.; Jesus, H.; Fernandes, M.; Matsumoto, S. Landfill leachate sludge use as soil additive prior and after electrocoagulation treatment: A cytological assessment using CHO-k1 cells. Chemosphere 2016, 158, 66–71. [Google Scholar] [CrossRef]
- Sadeghi, M.; Tadrisi, M.; Bay, A.; Dadban, Y. Treatment of municipal solid wastes leachate using electrocoagulation. J. Health Field 2017, 4, 1–8. [Google Scholar]
- Priya, N.; Esakku, S.M.N.; Palanivelu, K. Electrochemical Treatment of Landfill Leachate. Indian Chem. Engr. B 2005, 47, 272. [Google Scholar]
- Luu, T.L. Post treatment of ICEAS-biologically landfill leachate using electrochemical oxidation with Ti/BDD and Ti/RuO2 anodes. Environ. Technol. Innov. 2020, 20, 101099. [Google Scholar] [CrossRef]
- Panizza, M.; Martinez-Huitle, C.A. Role of electrode materials for the anodic oxidation of a real landfill leachate—Comparison between Ti–Ru–Sn ternary oxide, PbO2 and boron-doped diamond anode. Chemosphere 2013, 90, 1455–1460. [Google Scholar] [CrossRef]
- Boudjema, N.; Drouiche, N.; Abdi, N.; Grib, H.; Lounici, H.; Pauss, A.; Mameri, N. Treatment of Oued El Harrach river water by electrocoagulation noting the effect of the electric field on microorganisms. J. Taiwan Inst. Chem. Eng. 2014, 45, 1564–1570. [Google Scholar] [CrossRef]
- Chemlal, R.; Abdi, N.; Drouiche, N.; Lounici, H.; Pauss, A.; Mameri, N. Rehabilitation of Oued Smar landfill into a recreation park: Treatment of the contaminated waters. Ecol. Eng. 2013, 51, 244–248. [Google Scholar] [CrossRef]
- Panizza, M.; Delucchi, M.; Cerisola, G. Electrochemical degradation of anionic surfactants. J. Appl. Electrochem. 2005, 35, 357–361. [Google Scholar] [CrossRef]
- Labanowski, J.; Pallier, V.; Feuillade-Cathalifaud, G. Study of organic matter during coagulation and electrocoagulation processes: Application to a stabilized landfill leachate. J. Hazard. Mater. 2010, 179, 166–172. [Google Scholar] [CrossRef]
- Zuo, X.; Zhang, Y.; Si, L.; Zhou, B.; Zhao, B.; Zhu, L.; Jiang, X. One-step electrochemical preparation of sulfonated graphene/polypyrrole composite and its application to supercapacitor. J. Alloy. Compd. 2016, 688, 140–148. [Google Scholar] [CrossRef]
- Foo, K.; Hameed, B. An overview of landfill leachate treatment via activated carbon adsorption process. J. Hazard. Mater. 2009, 171, 54–60. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater. 2021. Available online: https://www.wef.org/resources/publications/books/StandardMethods (accessed on 21 November 2021).
- Farzadkia, M.; Vanani, A.F.; Golbaz, S.; Sajadi, H.S. Characterization and evaluation of treatability of wastewater generated in Khuzestan livestock slaughterhouses and assessing of their wastewater treatment systems. Glob. Nest J. 2016, 18, 108–118. [Google Scholar]
- Dia, O.; Drogui, P.; Buelna, G.; Dubé, R.; Ihsen, B.S. Electrocoagulation of bio-filtrated landfill leachate: Fractionation of organic matter and influence of anode materials. Chemosphere 2017, 168, 1136–1141. [Google Scholar] [CrossRef]
- Rice, E.W.; Baird, R.B.; Eaton, A.D. Standard Methods for the Examination of Water and Wastewater. American Public Health Association, American Water Works Association, Water Environment Federation. 2017. Available online: https://www.awwa.org/Store/Product-Details/productId/65266295 (accessed on 3 August 2021).
- Kashitarash, Z.E.; Taghi, S.M.; Kazem, N.; Abbass, A.; Alireza, R. Application of iron nanaoparticles in landfill leachate treatment—Case study: Hamadan landfill leachate. Iran. J. Environ. Health Sci. Eng. 2012, 9, 36. [Google Scholar] [CrossRef] [Green Version]
- Fard, M.P.; Mahvi, A.H.; Asgari, A.; Moradnia, M. Tehran University of Medical Science Heavy Metals Monitoring in Leachate from Landfill Site of Qazvin, Iran. Arch. Hyg. Sci. 2017, 6, 44–48. [Google Scholar] [CrossRef]
- Fudala-Ksiazek, S.; Pierpaoli, M.; Kulbat, E.; Luczkiewicz, A. A modern solid waste management strategy—The generation of new by-products. Waste Manag. 2016, 49, 516–529. [Google Scholar] [CrossRef]
- Ilhan, F.; Kurt, U.; Apaydin, O.; Gönüllü, M.T. Treatment of leachate by electrocoagulation using aluminum and iron electrodes. J. Hazard. Mater. 2008, 154, 381–389. [Google Scholar] [CrossRef]
- Li, X.; Song, J.; Guo, J.; Wang, Z.; Feng, Q. Landfill leachate treatment using electrocoagulation. Procedia Environ. Sci. 2011, 10, 1159–1164. [Google Scholar] [CrossRef] [Green Version]
- Ricordel, C.; Djelal, H. Treatment of landfill leachate with high proportion of refractory materials by electrocoagulation: System performances and sludge settling characteristics. J. Environ. Chem. Eng. 2014, 2, 1551–1557. [Google Scholar] [CrossRef]
- Cossu, R.; Polcaro, A.M.; Lavagnolo, M.C.; Mascia, M.; Palmas, S.; Renoldi, F. Electrochemical Treatment of Landfill Leachate: Oxidation at Ti/PbO2 and Ti/SnO2 Anodes. Environ. Sci. Technol. 1998, 32, 3570–3573. [Google Scholar] [CrossRef]
- Lin, H.; Peng, H.; Feng, X.; Li, X.; Zhao, J.; Yang, K.; Liao, J.; Cheng, D.; Liu, X.; Lv, S.; et al. Energy-efficient for advanced oxidation of bio-treated landfill leachate effluent by reactive electrochemical membranes (REMs): Laboratory and pilot scale studies. Water Res. 2021, 190, 116790. [Google Scholar] [CrossRef] [PubMed]
- Cruz, L.P.S.; Alve, L.P.; Santos, A.V.S.; Esteves, M.B.; Gomes, Í.V.S.; Nunes, L.S.S. Assessment of BTEX Concentrations in Air Ambient of Gas Stations Using Passive Sampling and the Health Risks for Workers. J. Environ. Prot. 2017, 8, 12–25. [Google Scholar] [CrossRef] [Green Version]
- Turro, E.; Su, S.-Y.; Gonçalves, Â.J.M.; Coin, L.; Richardson, S.; Lewin, A. Haplotype and isoform specific expression estimation using multi-mapping RNA-seq reads. Genome Biol. 2011, 12, R13. [Google Scholar] [CrossRef] [PubMed]
- Panizza, M.; Cerisola, G. Applicability of electrochemical methods to carwash wastewaters for reuse. Part 1: Anodic oxidation with diamond and lead dioxide anodes. J. Electroanal. Chem. 2010, 638, 28–32. [Google Scholar] [CrossRef]
- Bashir, M.J.; Isa, M.H.; Kutty, S.R.M.; Awang, Z.B.; Aziz, H.A.; Mohajeri, S.; Farooqi, I.H. Landfill leachate treatment by electrochemical oxidation. Waste Manag. 2009, 29, 2534–2541. [Google Scholar] [CrossRef]
- Bouhezila, F.; Hariti, M.; Lounici, H.; Mameri, N. Treatment of the OUED SMAR town landfill leachate by an electrochemical reactor. Desalination 2011, 280, 347–353. [Google Scholar] [CrossRef]
- Mandal, P.; Gupta, A.K.; Dubey, B.K. Role of inorganic anions on the performance of landfill leachate treatment by electrochemical oxidation using graphite/PbO2 electrode. J. Water Process. Eng. 2020, 33, 101119. [Google Scholar] [CrossRef]
- Ambauen, N.; Muff, J.; Mai, N.L.; Hallé, C.; Trinh, T.T.; Meyn, T. Insights into the Kinetics of Intermediate Formation during Electrochemical Oxidation of the Organic Model Pollutant Salicylic Acid in Chloride Electrolyte. Water 2019, 11, 1322. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.-K.; Lo, S.-L.; Chen, T.-Y. Regeneration and reuse of leachate from a municipal solid waste landfill. J. Environ. Biol. 2014, 35, 1123–1129. [Google Scholar]

| Process | Experimental Condition | Efficiency of Removal | Power Consumption (kWhr/m3) | References | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Initial COD (ppm) | Catalyst | pH | Time | Current Density | COD | Color | NH3 | |||
| Photo-electro-Fenton | 3000–5000 | H2O2 | 1.5–5 | 0.07–0.35 A/dm2 | 97 | 100 | - | 3.1 | Adapted from [35] | |
| Electro-oxidation | Stainless steel | 7 | 35 min | 4.3 mA/cm2 | 42 | 97 | [36] | |||
| Electrochemical | Boron-doped diamond | 64 mA/cm2 | 100 | 127.5 | [28] | |||||
| Reverse osmosis with electrochemical | 8750 | Ti-PbO2 | 3.5 h | 141 A/m2 | 96 | 28.7 | [30] | |||
| Fe2O3 NPs+ Electroflotation | 2200 | 120 min | 96 | 100 | 99 | [37] | ||||
| Parameter | PH | Color (TCU) | Turbidity (NTU) | EC (ms/cm3) | TDS (mg/L) | TSS (mg/L) | NH4+ (mg/L) | COD (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Raw | 6.25 | 19,600 | 3320 | 14.62 | 32,280 | 2326 | 486 | 22,000 |
| Blank | 7 | 0 | 2 | 220 | 500 | 0 | 0 | 0 |
| Sediment leachate (blank for sedimentation process) | 6 | 13,200 | 1740 | 13.76 | 21,480 | 825 | 486 | 22,153 |
| Leachate + Lime | 10 | 8500 | 1355 | 13.8 | 21,820 | 884 | 385 | 22,371 |
| * standards of Iran | <8.5 | <50 | <1000 | <100 | <2.5 | <200 |
| Parameters | Values |
|---|---|
| Electrolysis Time (min) | 30, 120, 240 |
| Electrolyte Type | NaCI, Na2SO4, No electrolyte |
| Ph | 3, 7.5, 10.2 |
| Current density (mA/cm2) | 16, 48, 64 |
| Electrode distance (mm) | 10, 15, 20 |
| Temperature (°C) | Room temperature 20, 30, 40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rookesh, T.; Samaei, M.R.; Yousefinejad, S.; Hashemi, H.; Derakhshan, Z.; Abbasi, F.; Jalili, M.; Giannakis, S.; Bilal, M. Investigating the Electrocoagulation Treatment of Landfill Leachate by Iron/Graphite Electrodes: Process Parameters and Efficacy Assessment. Water 2022, 14, 205. https://doi.org/10.3390/w14020205
Rookesh T, Samaei MR, Yousefinejad S, Hashemi H, Derakhshan Z, Abbasi F, Jalili M, Giannakis S, Bilal M. Investigating the Electrocoagulation Treatment of Landfill Leachate by Iron/Graphite Electrodes: Process Parameters and Efficacy Assessment. Water. 2022; 14(2):205. https://doi.org/10.3390/w14020205
Chicago/Turabian StyleRookesh, Tahereh, Mohammad Reza Samaei, Saeed Yousefinejad, Hassan Hashemi, Zahra Derakhshan, Fariba Abbasi, Mahrokh Jalili, Stefanos Giannakis, and Muhammad Bilal. 2022. "Investigating the Electrocoagulation Treatment of Landfill Leachate by Iron/Graphite Electrodes: Process Parameters and Efficacy Assessment" Water 14, no. 2: 205. https://doi.org/10.3390/w14020205
APA StyleRookesh, T., Samaei, M. R., Yousefinejad, S., Hashemi, H., Derakhshan, Z., Abbasi, F., Jalili, M., Giannakis, S., & Bilal, M. (2022). Investigating the Electrocoagulation Treatment of Landfill Leachate by Iron/Graphite Electrodes: Process Parameters and Efficacy Assessment. Water, 14(2), 205. https://doi.org/10.3390/w14020205







