Applications of Nano-Zeolite in Wastewater Treatment: An Overview
Abstract
1. Introduction
- Producing green zeolites by using agricultural and industrial wastes as sources for the structural elements, e.g., fly and biomass ashes containing silicon, Aluminum ash and slag;
- Improving the performance of the solvo-thermal preparation method by using ionic liquid, where these liquids are used to improve the solvation power, reduce the vapor-pressure and increase the thermal stability of the produced zeolite;
- Producing green zeolites by reducing the consumption of chemicals and water; this trend is dependent on the use of alternative synthesis routes, e.g., vapor phase transition (Dry Gel Conversion), and mechano-chemistry processes;
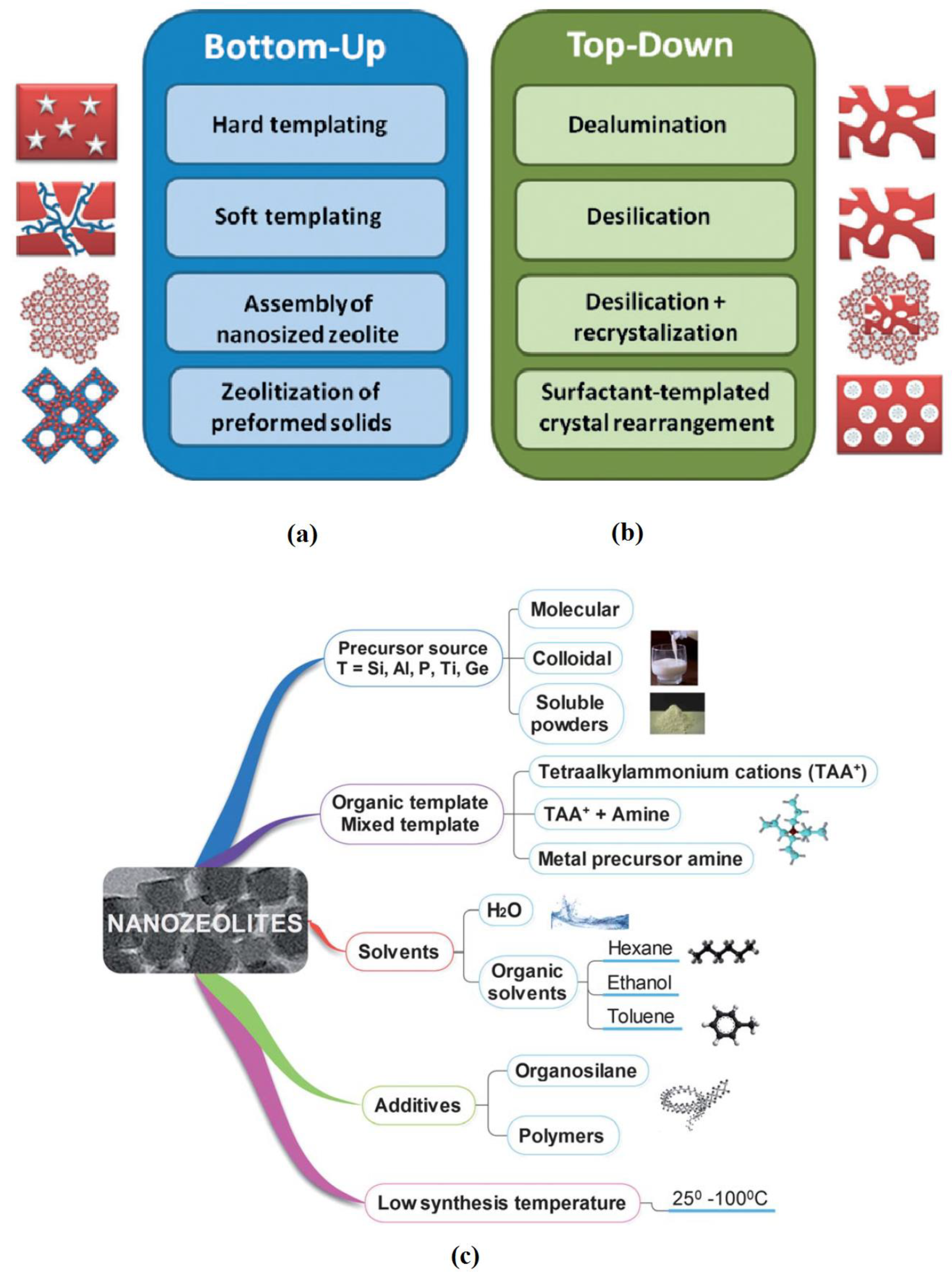
- Preparation of hierarchical zeolite to enhance the accessibility of the pores; this trend relies on either the modification of the preparation scheme (bottom up) or post preparation modification (top down) techniques, Figure 2a,b illustrates these techniques;
- Preparation of zeolite nano-particles to improve the selective separation, to enhance the sorption and de-sorption properties, and subsequently to reduce the size of the wastewater processing units. The key factors that affect the properties of the prepared nano-zeolites are illustrated in Figure 2c;
- Preparation of nano-sheets (2D) to improve the performance of selective separation process by reducing the diffusion path and improving the catalytic activity.
2. Advances in the Investigations of Nano-Zeolites as Ion-Exchanger/Sorbent
2.1. Testing Scheme to Optimize the Ion Exchange/Sorbent Applications
- Particle size, morphology, and surface properties determination: usually, these properties are determined using microscopic techniques. For nano-materials, Scanning Electron Microscope (SEM), Transmission Elector Microscope (TEM), and Scanning Probe Microscope (SPM) are widely used;
- Chemical compositions identification and detection of impurities: Spectroscopic analysis are widely used, e.g., Ultra-Violet Spectroscopy (UV), Fourier Transform Infra-Red Spectroscopy (FTIR), X-ray Photoelectron Spectroscopy (XPS), Energy-Dispersive X-ray spectroscopy (EDX);
- Material crystallinity: Wide Angle X-ray Diffraction (WAXD), Small Angle X-ray scattering (SAXS), and Ultra Small Angle Scattering(UAXS or USAXS) are widely used;
- Pore characteristics, the pore volume, porosity, and specific surface area could be measured via nitrogen absorption and application of Brunauer–Emmett–Teller (BET) model;
- The tendency of the material to agglomerate is usually identified by measuring zeta potential and the hydrodynamic radius;
- The ability of the material to act as cationic or anionic exchanger is usually determined by identifying the zero point charge;
- Cation Exchange Capacity (CEC) procedures are widely used to assess the capacity of the cationic exchanger.
2.2. Batch Investigations
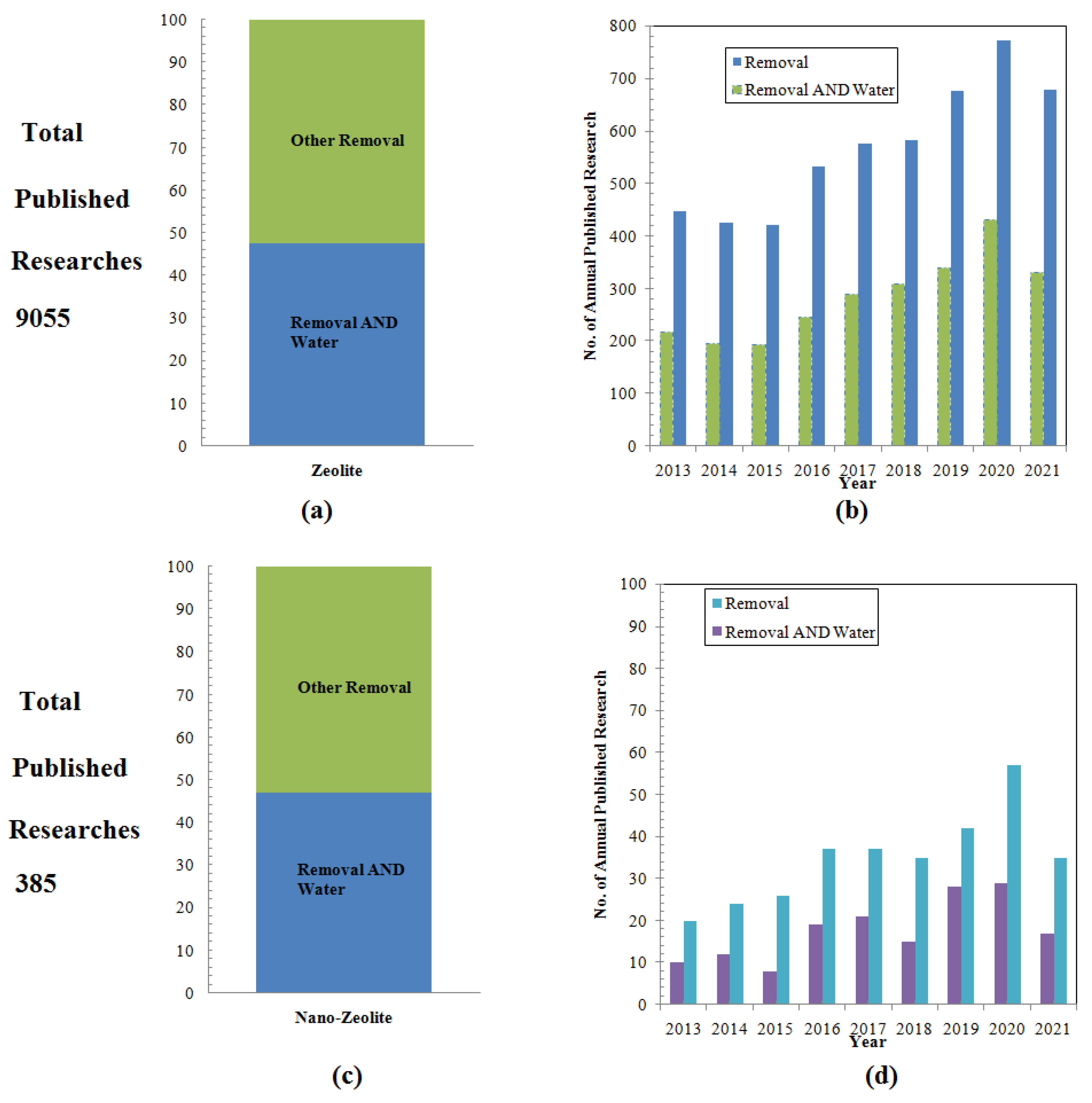
2.3. Application in Removing Radioactive Contaminants
- Different types of synthetic nano-zeolite were investigated that include zeolite A, zeolite Y, zeolite X, CHA, and MOR. The listed research employed the hydrothermal (HT) preparation method and mostly utilized analytical grade chemicals during the preparation.
- Both magnetic and polymeric Nano-zeolites composites were tested for the removal of the radioactive contaminants. In particular, natural zeolite was ground to nano-scale then immobilized in polymeric matrix, i.e.,Poly vinyl alcohol(PVA), Alginate (ALG)/poly-ethyleneoxide (PEO), and tested.
- All of the listed research was conducted by using single contaminant solution, where in-active contaminants were used to reduce the radiological exposure of the researchers.
- The investigated sorbent mass to waste volume fall in the range (1 ≤ m/V ≤ 20), where regeneration studies are limited to one study.
2.4. Application in Industrial Wastewater Treatment
2.4.1. Metal Removal Studies
- All the listed studies investigated only magnetic and polymeric composites, not the nano-zeolite particles. The magnetic composites included the use of magnetite and cobalt ferrite, where polymeric composites include various single and binary polymer, e.g., PVA, chtiosan, …
- Different types of natural and synthetic nano-zeolites were investigated. Natural zeolites were not identified or presented as CLP, where the synthetic nano-zeolites include zeolite Na-X, zeolite Y, Faujasite (FAU), ZSM-5, HZSM5, and MOR. The listed research employed the hydrothermal (HT) preparation method and one research work investigated the use of silica and alumina wastes for the preparation of the nano-zeolite.
- All of the listed research was conducted by using a single contaminant solution, the sorbent mass to volume ratio fall in the range (0.5 ≤ m/V ≤ 20) and regeneration studies were investigated in a comparatively large amount of research.
2.4.2. Organic Contaminants’ Sorption Studies
- Natural and synthesized nano-zeolites of different types, i.e., Nano-zeolites X, MOR, ZSM5, and Sodalite were investigated, where both green and conventional preparation routes were adopted,
- Nano-zeolite particles were mainly investigated for the removal of different types of dyes, i.e., MG, CV, MB, BR (18,41,46), and only one research studied the polymeric composite of natural nano-zeolite. No study addressed the inorganic nano-zeolite composite,
- The sorbent dosage falls in the range (0.3–10), which is relatively narrower than those studied for the radioactive contaminants and for metal removal studies.
- The studies were conducted using single contaminant solution and the regeneratbility studies are very limited.
3. Advances in Nano-Zeolite-Composites Applications in Photo-Catalytic Degradations
3.1. Testing Scheme to Optimize the Photo-Catalytic Degradation Applications
3.2. Batch Investigations
3.3. Applications in Organic Contaminant Degradation
- Most of the conducted research utilized natural CLP grounded to the nano-scale, and limited research utilized synthetic ZSM-5 nano zeolites for their applications in the preparation of photo-catalytic composites.
- The composites included metal oxides e.g., ZnO, CuO, FeO, NiO, and metal sulfide, e.g., ZnS, NiS, CuS, and PbS.
- Most of the conducted experiments employed a single contaminant solution, where solutions of model organic contaminant, e.g., 4-Nitrophenol, Dyes, e.g., Rhodamine B, Methylene blue, personal care products and pharmathetical compounds, e.g., Metronidazole, Tetracycline, Cefuroxime, Benzophenone. Real effluent, i.e., fish pond water, was only used in one research.
- The tested photo-catalytic composite dosage was in the range (0.025–3), where, in some studies, oxidizer, e.g., H2O2, was supplemented to the contaminant solution.
4. Advances in Nano-Zeolite Applications in Membrane Separation
4.1. Testing Scheme to Optimize the Membrane Applications
4.2. Identification of Membrane Performance
4.3. The Membrane Investigations
- Zeolite-Y, Na-X, FAU, Na-A, and beta were used as an active layer in the membrane composite, via depositing a thin film on the substrate or embedding onto the membrane matrix;
- Different modifications for the nano-zeolites were proposed including the addition of metals, e.g., Cu, oxides, e.g., TiO2, and organic modification, e.g., D-tyrosine. These modifications were used to increase the fouling resistance.
- The applications were restricted to MF, UF, and NF, despite there are some research studies that tested the use of nano-zolite in membrane applications for dehydration of different products using RO and forward osmosis, but these membrane types were not tested for wastewater treatment.
5. Conclusions
- Despite it being found that the use of nano-zeolite has enhanced the performance of the treatment process and subsequently can reduce the size and land requirement of the wastewater treatment plant, there is a need to consider the reduction of the materials footprints; this could be achieved by following greener nano-zeolite preparation techniques, i.e., use of bio-materials and wastes as sources for the preparation, use of biosolvent, and low temperature processes.
- Batch experiment for nano-zeolite applications in ion exchange/sorption process is a major research field with the highest number of published papers. This forms a database that can assist with the wide-scale application of several types of nano-zeolites for the removal of different contaminants including radioactive, metal, and organic. Research that assesses the continuous application of nano-zeolites in this field is very limited, where there is a need to assess the hydraulic and sorptive performance of this type of application.
- Despite nano-zeolite being able to be modified to act as anion exchanger/sorbent, these research investigations are very limited. Moreover, the research that includes the application of complicated solutions is missing, i.e., real wastewater. Finally, the application of nano-zeolites for the treatment of corrosive wastewater stream is not sufficiently addressed.
- For photo-degradation applications, there is a need to consider the application on complex/ real wastewater effluent, where the research only focuses on the use of a single contaminant solution.
- For membrane separation, the application of nano-zeolite in RO and forward osmosis is missing in the field of water and wastewater treatment.
- Hierarchical and 2D zeolites were not investigated yet in any application related to water and wastewater treatment.
- For all the presented applications, neither the cost analysis for the preparation and application of nano-zeolite in water and wastewater treatment nor the pilot scale applications were addressed. These types of investigations can help in paving the way toward the wide application of these materials in the industry.
- For each application, the research that addressed the life cycle management of nano-zeolite is missing. In particular, clear assessment of the end of life cycle management options for the exhausted nano-materials should be conducted.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tortajada, C. Contributions of recycled wastewater to clean water and sanitation Sustainable Development Goals. NPJ Clean Water 2020, 3, 22. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Hung, Y.-T. Application of Ionizing Radiation in Wastewater Treatment: An Overview. Water 2020, 12, 19. [Google Scholar] [CrossRef]
- Guth, J.L.; Kessler, H. Synthesis of Aluminosilicate Zeolites and Related Silica-Based Materials. In Catalysis and Zeolites Fundamentals and Applications; Weitkamp, J., Puppe, L., Eds.; Springer: Berlin/Heidelberg, Germany, 1999; pp. 1–52. [Google Scholar]
- Jha, B.; Singh, D.N. Basics of zeolites. In Fly Ash Zeolites; Springer: Singapore, 2016; pp. 5–31. [Google Scholar]
- Millini, R.; Belluss, G. Zeolite Science and Perspectives. In Zeolites in Catalysis: Properties and Applications; Cejka, J., Morris, R.E., Nachtigall, P., Eds.; RSC Catalysis Series No. 28; The Royal Society of Chemistry: Cambridge, UK, 2017. [Google Scholar]
- Tsai, Y.L.; Huang, E.; Li, Y.H.; Hung, H.T.; Jiang, J.H.; Liu, T.C.; Fang, J.N.; Chen, H.F. Raman Spectroscopic Characteristics of Zeolite Group Minerals. Minerals 2021, 11, 167. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Metwally, S.S.; El-Kamash, A.M. Life Cycle of Ion Exchangers in Nuclear Industry: Application and Management of Spent Exchangers. In Handbook of Ecomaterials; Martínez, L., Kharissova, O., Kharisov, B., Eds.; Springer: Cham, Switzerland, 2019; Volume 5, pp. 3709–3732. [Google Scholar] [CrossRef]
- Byrappa, K.; Yoshimura, M. Handbook of Hydrothermal Technology; Noyes Publications: Park Ridge, IL, USA, 2001. [Google Scholar]
- Abdel Rahman, R.O.; Ojovan, M.I. Application of nano-materials in radioactive waste management. In Environmental Science and Engineering; Zhang, T.C., Gurjar, B.R., Govil, J.N., Eds.; Studium Press, LLC: New York, NY, USA, 2017; Volume 10, pp. 361–378. [Google Scholar]
- Abdel Rahman, R.O.; Ibrahim, H.A.; Abdel Monem, N.M. Long-term performance of Zeolite Na A-X blend as backfill material in near surface disposal vault. Chem. Eng. J. 2009, 149, 143–152. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Ibrahim, H.A.; Hanafy, M.; Abdel Monem, N.M. Assessment of synthetic zeolite NaA-X as sorbing barrier for strontium in a radioactive disposal facility. Chem. Eng. J. 2010, 157, 100–112. [Google Scholar] [CrossRef]
- El-Naggar, M.R.; El-Kamash, A.M.; El-Dessouky, M.I.; Ghonaim, A.K. Two-step method for preparation of NaA-X zeolite blend from fly ash for removal of cesium ions. J. Hazard. Mater. 2008, 154, 963–972. [Google Scholar] [CrossRef]
- Ames, L.L., Jr. Synthesis of a clinoptilolite-like zeolite. Am. Miner. 1963, 48, 1374–1381. [Google Scholar]
- Jacobs, P.A.; Flanigen, E.M.; Jansen, J.C.; van Bekkum, H. Introduction to Zeolite Science and Practice; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Abdel Moamen, O.A.; Ismail, I.M.; Abdel Monem, N.M.; Abdel Rahman, R.O. Factorial design analysis for optimizing the removal of cesium and strontium ions on synthetic nano-sized zeolites. J. Taiwan Inst. Chem. Eng. 2015, 55, 133–144. [Google Scholar] [CrossRef]
- Gil, A.; Korili, S.A. Management and valorization of aluminum saline slags: Current status and future trends. Chem. Eng. J. 2016, 289, 74–84. [Google Scholar] [CrossRef]
- Ma, L.J.; Han, L.N.; Chen, S.; Hu, J.L.; Chang, L.P.; Bao, W.R.; Wang, J. Rapid synthesis of magnetic zeolite materials from fly ash and iron-containing wastes using supercritical water for elemental mercury removal from flue gas. Fuel Process. Technol. 2019, 189, 39–48. [Google Scholar] [CrossRef]
- Kuroki, S.; Hashishin, T.; Morikawa, T.; Yamashita, K.; Matsuda, M. Selective synthesis of zeolites A and X from two industrial wastes: Crushed stone powder and aluminum ash. J. Environ. Manag. 2019, 231, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Yoldi, M.; Fuentes-Ordoñez, E.G.; Korili, S.A.; Gil, A. Zeolite synthesis from industrial wastes. Microporous Mesoporous Mater. 2019, 287, 183–191. [Google Scholar] [CrossRef]
- Gao, W.; Amoo, C.C.; Zhang, G.; Javed, M.; Mazonde, B.; Lu, C.; Yang, R.; Xing, C.; Tsubaki, N. Insight into solvent-free synthesis of MOR zeolite and its laboratory scale production. Microporous Mesoporous Mater. 2019, 280, 187–194. [Google Scholar] [CrossRef]
- Mei, J.; Duan, A.; Wang, X. A Brief Review on Solvent-Free Synthesis of Zeolites. Materials 2021, 14, 788. [Google Scholar] [CrossRef]
- Li, K.; Valla, J.; Garcia-Martinez, J. Realizing the Commercial Potential of Hierarchical Zeolites: New Opportunities in Catalytic Cracking. ChemCatChem 2014, 6, 46–66. [Google Scholar] [CrossRef]
- Feliczak-Guzik, A. Hierarchical zeolites: Synthesis and catalytic properties. Microporous Mesoporous Mater. 2018, 259, 33–45. [Google Scholar] [CrossRef]
- Mintova, S.; Gilson, J.P.; Valtchev, V. Advances in nanosized Zeolites. Nanoscale 2013, 5, 6693. [Google Scholar] [CrossRef]
- Pan, T.; Wu, Z.; Yip, A.C.K. Advances in the Green Synthesis of Microporous and Hierarchical Zeolites: A Short Review. Catalysts 2019, 9, 274. [Google Scholar] [CrossRef]
- Cao, Z.; Zeng, S.; Xu, Z.; Arvanitis, A.; Yang, S.; Gu, X.; Dong, J. Ultrathin ZSM-5 zeolite nanosheet laminated membrane for high-flux desalination of concentrated brines. Sci. Adv. 2018, 4, eaau8634. [Google Scholar] [CrossRef]
- Rehman, A.u.; Arepalli, D.; Alam, S.F.; Kim, M.-Z.; Choi, J.; Cho, C.H. Two-Dimensional MFI Zeolite Nanosheets Exfoliated by Surfactant Assisted Solution Process. Nanomaterials 2021, 11, 2327. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Abdel Moamen, O.A.; Hanafy, M.; Abdel Monem, N.M. Preliminary investigation of zinc transport through zeolite-X barrier: Linear isotherm assumption. Chem. Eng. J. 2012, 185–186, 61–70. [Google Scholar] [CrossRef]
- Choi, H.J.; Yu, S.W.; Kim, K.H. Efficient use of Mg-modified zeolite in the treatment of aqueous solution contaminated with heavy metal toxic ions. J. Taiwan Inst. Chem. Eng. 2016, 63, 482–489. [Google Scholar] [CrossRef]
- Tran, H.N.; Viet, P.V.; Chao, H.P. Surfactant modified zeolite as amphiphilic and dual-electronic adsorbent for removal of cationic and oxyanionic metal ions and organic compounds. Ecotoxicol. Environ. Saf. 2018, 147, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Rad, L.R.; Anbia, M. Zeolite-based compositesfor the adsorption of toxic matters from water: A review. J. Environ. Chem. Eng. 2021, 9, 106088. [Google Scholar]
- Rodríguez-Iznaga, I.; Rodríguez-Fuentes, G.; Petranovskii, V. Ammonium modified natural clinoptilolite to remove manganese, cobalt and nickel ions from wastewater: Favorable conditions to the modification and selectivity to the cations. Microporous Mesoporous Mater. 2018, 255, 200–210. [Google Scholar] [CrossRef]
- Zhang, H.; Li, A.; Zhang, W.; Shuang, C. Combination of Na-modified Zeolite and anion exchange resin for advanced treatment of a high ammoniaenitrogen content municipal effluent. J. Colloid Interface Sci. 2016, 468, 128–135. [Google Scholar] [CrossRef]
- Wen, J.; Dong, H.; Zeng, G. Application of zeolite in removing salinity/sodicity from wastewater: A review of mechanisms, challenges and opportunities. J. Clean. Prod. 2018, 197, 1435–1446. [Google Scholar] [CrossRef]
- Jiménez-Reyes, M.; Almazán-Sánchez, P.T.; Solache-Ríos, M. Radioactive waste treatments by using zeolites. A short review. J. Environ. Radioact. 2021, 233, 106610. [Google Scholar] [CrossRef]
- Fatima, H.; Djamel, N.; Samira, A.; Mahfoud, B. Modelling and adsorption studies of removal uranium (VI) ions on synthesised zeolite NaY. Desalin. Water Treat. 2013, 51, 5583–5591. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Abdel Moamen, O.A.; El-Masry, E.H. Life cycle of polymer nanocomposites matrices in hazardous waste management. In Handbook of Polymer and Ceramic Nanotechnology; Hussain, C.M., Thomas, S., Eds.; Springer Nature: Cham, Switzerland, 2021; pp. 1603–1625. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Abdel Moamen, O.A.; Abdelmonem, N.; Ismail, I.M. Optimizing the removal of strontium and cesium ions from binary solutions on magnetic nano-zeolite using response surface methodology (RSM) and artificial neural network (ANN). Environ. Res. 2019, 173, 397–410. [Google Scholar] [CrossRef]
- Abdel Rahman, R.O.; Ojovan, M.I. Toward Sustainable Cementitious Radioactive Waste Forms: Immobilization of Problematic Operational Wastes. Sustainability 2021, 13, 11992. [Google Scholar] [CrossRef]
- Dyer, A.; Hriljac, J.; Evans, N.; Stokes, I.; Rand, P.; Kellet, S.; Harjula, R.; Moller, T.; Maher, Z.; Heatlie-Branson, R.; et al. The use of columns of the zeolite clinoptilolite in the remediation of aqueous nuclear waste streams. J. Radioanal. Nucl. Chem. 2018, 318, 2473–2491. [Google Scholar] [CrossRef] [PubMed]
- Osmanlioglu, E. Treatment of radioactive liquid waste by sorption on natural zeolite in Turkey. J. Hazard. Mater. 2006, 137, 332–335. [Google Scholar] [CrossRef]
- El-Dessouky, M.I.; El-Kamash, A.M.; El-Sourougy, M.R.; Aly, H.F. Simulation of Zeolite Fixed Bed Columns Used for Treatment of Liquid Radioactive Wastes. Radiochem. Acta 2000, 88, 879–884. [Google Scholar] [CrossRef]
- El-Kamash, A.M.; Zaki, A.A.; El Geleel, M.A. Modeling Batch Kinetics and Thermodynamics of Zinc and Cadmium Ions Removal from Waste Solution Using Synthetic Zeolite A. J. Hazard. Mater. 2005, 127, 211–220. [Google Scholar] [CrossRef]
- El-Rahman, K.M.A.; El-Kamash, A.M.; El-Sourougy, M.R.; Abdel-Moniem, N.M. Thermodynamic Modeling for the Removal of Cs+, Sr2+, Ca2+, and Mg2+ Ions from Aqueous Waste Solution Using Zeolite A. J. Radioanal. Nucl. Chem. 2006, 268, 221–230. [Google Scholar] [CrossRef]
- Fang, X.-H.; Fang, F.; Lu, C.-H.; Zheng, L. Removal of Cs+, Sr2+, and Co2+ ions from the mixture of organics and suspended solids aqueous solutions by zeolites. Nucl. Eng. Technol. 2017, 49, 556–561. [Google Scholar] [CrossRef]
- El-Kamash, A.M. Evaluation of zeolite A for the sorptive removal of Cs+ and Sr2+ ions from aqueous solutions using batch and fixed bed column operations. J. Hazard. Mater. 2008, 151, 432–445. [Google Scholar] [CrossRef]
- Abd El-Rahman, K.M.; El-Sourougy, M.R.; Abdel-Monem, N.M.; Ismail, I.M. Modeling the Sorption Kinetics of Cesium and Strontium Ions on Zeolite A. J. Nucl. Radiochem. Sci. 2006, 7, 21–27. [Google Scholar] [CrossRef]
- Baek, W.; Ha, S.; Hong, S.; Kim, S.; Kim, Y. Cation exchange of cesium and cation selectivity of natural zeolites: Chabazite, stilbite, and heulandite. Microporous Mesoporous Mater. 2018, 264, 159–166. [Google Scholar] [CrossRef]
- Lihareva, N.; Petrov, O.; Dimowa, L.; Tzvetanova, Y.; Piroeva, I.; Ublekov, F.; Nikolov, A. Ion exchange of Cs+ and Sr2+ by natural clinoptilolite from bi-cationic solutions and XRD control of their structural positioning. J. Radioanal. Nucl. Chem. 2020, 323, 1093–1102. [Google Scholar] [CrossRef]
- Mosai, A.K.; Chimuka, L.; Cukrowska, E.M.; Kotzé, I.A.; Tutu, H. The recovery of rare earth elements (REEs) from aqueous solutions using natural zeolite and bentonite. Water Air Soil Pollut. 2019, 230, 188. [Google Scholar] [CrossRef]
- Chen, Z.; Lu, S. Investigation of the effect of pH, ionic strength, foreign ions, temperature, soil humic substances on the sorption of 152+154Eu(III) onto NKF-6 zeolite. J. Radioanal. Nucl. Chem. 2016, 309, 717–728. [Google Scholar] [CrossRef]
- Abdi, M.R.; Shakur, H.R.; Saraee, K.R.E.; Sadeghi, M. Effective removal of uranium ions from drinking water using CuO/X zeolite based nanocomposites: Effects of nano concentration and cation exchange. J. Radioanal. Nucl. Chem. 2014, 300, 1217–1225. [Google Scholar] [CrossRef]
- Lazaridis, N.K.; Karapantsios, T.D.; Georgantas, D. Kinetic analysis for the removal of a reactive dye from aqueous solution onto hydrotalcite by adsorption. Water Res. 2003, 37, 3023–3033. [Google Scholar] [CrossRef]
- Krajňák, A.; Viglašová, E.; Galamboš, M.; Krivosudský, L. Kinetics, thermodynamics and isotherm parameters of uranium (VI) adsorption on natural and HDTMA-intercalated bentonite and zeolite. Desalin. Water Treat. 2018, 127, 272–281. [Google Scholar] [CrossRef]
- Al-Shaybe, M.; Khalili, F. Adsorption of thorium (IV) and uranium (VI) by Tulul al-shabba zeolitic tuff. Jordan J. Earth Environ. Sci. 2009, 2, 108–109. [Google Scholar]
- Nyembe, D.W.; Mamba, B.B.; Mulaba-Bafubiandi, A.F. The Effect of Si and Fe Impurities on the Removal of Cu2+ and Co2+ from Co/Cu aqueous colutions using natural clinoptilolite as an ion-exchanger. Desalin. Water Treat. 2010, 21, 335–345. [Google Scholar] [CrossRef]
- Rodríguez, A.; S’aez, P.; Díez, E.; G’omez, J.M.; García, J.; Bernabé, I. Highly efficient low-cost zeolite for cobalt removal from aqueous solutions: Characterization and performance. Am. Inst. Chem. Eng. Environ. Prog. 2018, 38, S352–S365. [Google Scholar] [CrossRef]
- Irannajad, M.; Haghighi, H.K. Removal of Co2+, Ni2+, and Pb2+ by manganese oxide-coated zeolite: Equilibrium, thermodynamics, and kinetics studies. Clay Miner. 2017, 65, 52–62. [Google Scholar] [CrossRef]
- Liang, J.; Li, J.; Li, X.; Liu, K.; Wu, L.; Shan, G. The sorption behavior of CHA-type zeolite for removing radioactive strontium from aqueous solutions. Sep. Purif. Technol. 2020, 230, 115874. [Google Scholar] [CrossRef]
- Hassan, R.S.; Abass, M.R.; Eid, M.A.; Abdel-Galil, E.A. Sorption of some radionuclides from liquid waste solutions using anionic clay hydrotalcite sorbent. Appl. Radiat. Isot. 2021, 178, 109985. [Google Scholar] [CrossRef]
- Abdel Moamen, O.A.; Ibrahim, H.A.; Abdelmonem, N.; Ismail, I.M. Thermodynamic analysis for the sorptive removal of cesium and strontium ions onto synthesized magnetic nano Zeolite. Microporous Mesoporous Mater. 2016, 223, 187–195. [Google Scholar] [CrossRef]
- Faghihian, H.; Moayed, M.; Firooz, A.; Iravan, M. Synthesis of a novel magnetic zeolite nanocomposite for removal of Cs+ and Sr2+ from aqueous solution: Kinetic, equilibrium, and thermodynamic studies. J. Colloid Interface Sci. 2013, 393, 445–451. [Google Scholar] [CrossRef]
- Sharma, P.; Tomar, R. Sorption behaviour of nanocrystalline MOR type zeolite for Th(IV) and Eu(III) removal from aqueous waste by batch treatment. J. Colloid Interface Sci. 2011, 362, 144–156. [Google Scholar] [CrossRef]
- Lee, K.Y.; Kim, K.W.; Park, M.; Kim, J.; Oh, M.; Lee, E.H.; Chung, D.Y.; Moon, J.K. Novel application of nanozeolite for radioactive cesium removal from high-salt wastewater. Water Res. 2016, 95, 134–141. [Google Scholar] [CrossRef]
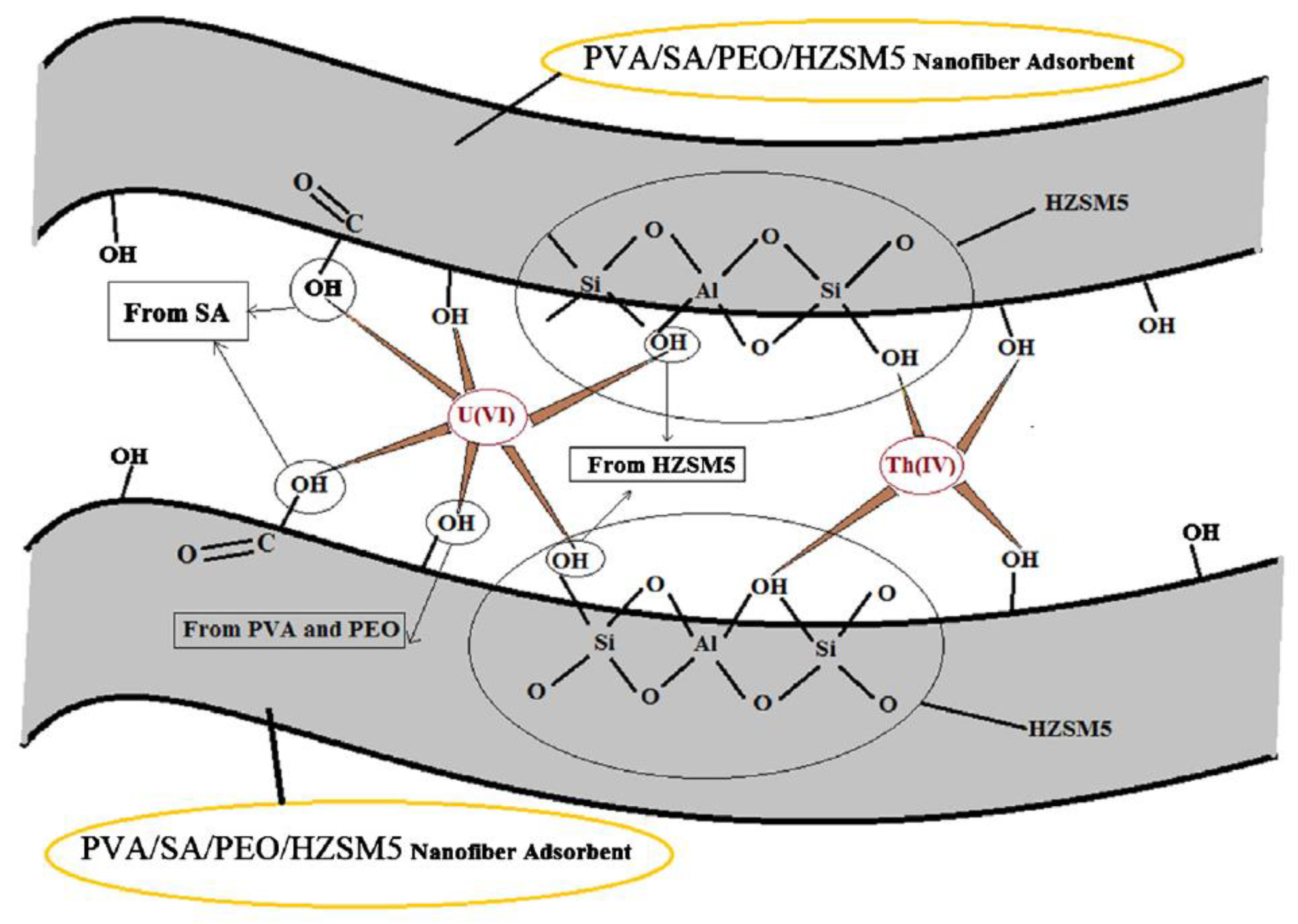
- Talebi, M.; Abbasizadeh, S.; Keshtkar, A.R. Evaluation of Single and Simultaneous Thorium and Uranium Sorption from Water Systems by an Electrospun PVA/SA/PEO/HZSM5 Nanofiber. Process Saf. Environ. Prot. 2017, 109, 340–356. [Google Scholar] [CrossRef]
- Zahakifar, F.; Keshtkar, A.R.; Talebi, M. Performance evaluation of sodium alginate/polyvinyl alcohol/ polyethylene oxide/ZSM5 zeolite hybrid adsorbent for ion uptake from aqueous solutions: A case study of thorium (IV). J. Radioanal. Nucl. Chem. 2021, 327, 65–72. [Google Scholar] [CrossRef]
- Murukutti, M.K.; Jena, H. Synthesis of nano-crystalline zeolite-A and zeolite-X from Indian coal fly ash, its characterization and performance evaluation for the removal of Cs+ and Sr2+ from simulated nuclear waste. J. Hazard. Mater. 2022, 423, 127085. [Google Scholar] [CrossRef] [PubMed]
- Paris, E.C.; Malafatti, J.O.; Musetti, H.C.; Manzoli, A.; Zenatti, A.; Escote, M.T. Faujasite zeolite decorated with cobalt ferrite nanoparticles for improving removal and reuse in Pb2+ ions adsorption. Chin. J. Chem. Eng. 2020, 28, 1884–1890. [Google Scholar] [CrossRef]
- Tabatabaeefar, A.; Keshtkar, A.R.; Talebi, M.; Abolghasemi, H. Polyvinyl alcohol/alginate/zeolite nanohybrid for removal of metals. Chem. Eng. Technol. 2020, 43, 343–354. [Google Scholar] [CrossRef]
- Abdelrahman, E.A.; El-Reash, Y.G.A.; Youssef, H.M.; Kotp, Y.H.; Hegazey, R.M. Utilization of rice husk and waste aluminum cans for the synthesis of some nanosized zeolite, zeolite/zeolite, and geopolymer/zeolite products for the efficient removal of Co (II), Cu (II), and Zn (II) ions from aqueous media. J. Hazard. Mater. 2021, 401, 123813. [Google Scholar] [CrossRef]
- Shariatinia, Z.; Bagherpour, A. Synthesis of zeolite NaY and its nanocomposites with chitosan as adsorbents for lead(II) removal from aqueous solution. Powder Technol. 2018, 338, 744–763. [Google Scholar] [CrossRef]
- Ghanavati, L.; Hekmati, A.H.; Rashidi, A.; Shafiekhani, A. Application of electrospun polyamide-6/modified zeolite nanofibrous composite to remove acid blue 74 dye from textile dyeing wastewater. J. Text. Inst. 2020, 112, 1730–1742. [Google Scholar] [CrossRef]
- Pizarro, C.; Rubio, M.A.; Escudey, M.; Albornoz, M.F.; Muñoz, D.; Denardin, J.; Fabris, J.D. Nanomagnetite-zeolite composites in the removal of arsenate from aqueous systems. J. Braz. Chem. Soc. 2015, 26, 1887–1896. [Google Scholar] [CrossRef]
- Shafiee, M.; Abedi, M.A.; Abbasizadeh, S.; Sheshdeh, R.K.; Mousavi, S.E.; Shohani, S. Effect of zeolite hydroxyl active site distribution on adsorption of Pb (II) and Ni (II) pollutants from water system by polymeric nanofibers. Sep. Sci. Technol. 2020, 55, 1994–2011. [Google Scholar] [CrossRef]
- Rad, L.R.; Momeni, A.; Ghazani, B.F.; Irani, M.; Mahmoudi, M.; Noghreh, B. Removal of Ni2+ and Cd2+ ions from aqueous solutions using electrospun PVA/zeolite nanofibrous adsorbent. Chem. Eng. J. 2014, 256, 119–127. [Google Scholar] [CrossRef]
- Isawi, H. Using zeolite/polyvinyl alcohol/sodium alginate nanocomposite beads for removal of some heavy metals from wastewater. Arab. J. Chem. 2020, 13, 5691–5716. [Google Scholar] [CrossRef]
- Abdelrahman, E.A. Synthesis of zeolite nanostructures from waste aluminum cans for efficient removal of malachite green dye from aqueous media. J. Mol. Liq. 2018, 253, 72–82. [Google Scholar] [CrossRef]
- Abdelrahman, E.A.; Hegazey, R.M.; Alharbi, A. Facile synthesis of mordenite nanoparticles for efficient removal of Pb(II) ions from aqueous media. J. Inorg. Organomet. Polym. Mater. 2020, 30, 1369–1383. [Google Scholar] [CrossRef]
- Borandegi, M.; Nezamzadeh-Ejhieh, A. Enhanced removal efficiency of clinoptilolite nano-particles toward Co(II) from aqueous solution by modification with glutamic acid. Colloids Surf. A Physicochem. Eng. Asp. 2015, 479, 35–45. [Google Scholar] [CrossRef]
- Heidari-Chaleshtori, M.; Nezamzadeh-Ejhieh, A. Clinoptilolitenano-particles modified with aspartic acid for removal of Cu(II) from aqueous solutions: Isotherms and kinetic aspects. New J. Chem. 2015, 39, 9396–9406. [Google Scholar] [CrossRef]
- Fakari, S.; Nezamzadeh-Ejhieh, A. Synergistic effects of ion exchange and complexation processes in cysteine-modified clinoptilolite nanoparticles for removal of Cu(II) from aqueous solutions in batch and continuous flow systems. New J. Chem. 2017, 41, 3811–3820. [Google Scholar] [CrossRef]
- Mehrali-Afjani, M.; Nezamzadeh-Ejhieh, A. Efficient solid amino acid–clinoptilolite nanoparticles adsorbent for Mn(II) removal: A comprehensive study on designing the experiments, thermodynamic and kinetic aspects. Solid State Sci. 2020, 101, 106124. [Google Scholar] [CrossRef]
- Nezamzadeh-Ejhieh, A.; Kabiri-Samani, M. Effective removal of Ni(II) from aqueous solutions by modification of nano particles of clinoptilolite with dimethylglyoxime. J. Hazard. Mater. 2013, 260, 339–349. [Google Scholar] [CrossRef]
- Shafiof, M.S.; Nezamzadeh-Ejhieh, A. A comprehensive study on the removal of Cd(II) from aqueous solution on a novel pentetic acid-clinoptilolite nanoparticles adsorbent: Experimental design, kinetic and thermodynamic aspects. Solid State Sci. 2020, 99, 106071. [Google Scholar] [CrossRef]
- Shakur, H.R.; Saraee, K.R.E.; Abdi, M.R.; Azimi, G. A novel PAN/NaX/ZnO nanocomposite absorbent: Synthesis, characterization, removal of uranium anionic species from contaminated water. J. Mater. Sci. 2016, 51, 9991–10004. [Google Scholar] [CrossRef]
- Chen, J.; Gao, Q.; Zhang, X.; Liu, Y.; Wang, P.; Jiao, Y.; Yang, Y. Nanometer mixed-valence silver oxide enhancing adsorption of ZIF-8 for removal of iodide in solution. Sci. Total Environ. 2019, 646, 634–644. [Google Scholar] [CrossRef]
- Shakur, H.R.; Saraee, K.R.E.; Abdi, M.R.; Azimi, G. Selective removal of uranium ions from contaminated waters using modified-X nanozeolite. Appl. Rad. Isot. 2016, 118, 43–55. [Google Scholar] [CrossRef]
- Gasser, M.S.; Mekhamer, H.S.; Abdel Rahman, R.O. Optimization of the utilization of Mg/Fe hydrotalcite like Compounds in the removal of Sr(II) from aqueous solution. J. Environ. Chem. Eng. 2016, 4, 4619–4630. [Google Scholar] [CrossRef]
- Gasser, M.S.; El Sherif, E.; Abdel Rahman, R.O. Modification of Mg-Fe hydrotalcite using Cyanex 272 for lanthanides separation. Chem. Eng. J. 2017, 316C, 758–769. [Google Scholar] [CrossRef]
- Gasser, M.S.; El Sherif, E.; Mekhamer, H.S.; Abdel Rahman, R.O. Assessment of Cyanex 301 impregnated resin for its potential use to remove cobalt from aqueous solutions. Environ. Res. 2020, 185, 109402. [Google Scholar] [CrossRef] [PubMed]
- Phillip, E.; Khoo, K.S.; Yusof, M.A.W.; Abdel Rahman, R.O. Assessment of POFA-cementitious material as backfill barrier in DSRS borehole disposal: 226Ra confinement. J. Environ. Manag. 2021, 280, 111703. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, H.A.; Abdel Moamen, O.A.; Monem, N.A.; Ismail, I.M. Assessment of kinetic and isotherm models for competitive sorption of Cs+ and Sr2+ from binary metal solution onto nanosized zeolite. Chem. Eng. Commun. 2018, 205, 1274–1287. [Google Scholar] [CrossRef]
- Hassan, H.S.; Abdel Moamen, O.A.; Zaher, W.F. Adaptive Neuro-Fuzzy inference system analysis on sorption studies of strontium and cesium cations onto a novel impregnated nano-zeolite. Adv. Powder Technol. 2020, 31, 1125–1139. [Google Scholar] [CrossRef]
- Abdel Moamen, O.A.; Hassan, H.S.; Zaher, W.F. Taguchi L16 optimization approach for simultaneous removal of Cs+ and Sr2+ ions by a novel scavenger. Ecotoxicol. Environ. Saf. 2020, 189, 110013. [Google Scholar] [CrossRef]
- Barczyk, K.; Mozgawa, W.; Król, M. Studies of anions sorption on natural zeolites. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 133, 876–882. [Google Scholar] [CrossRef]
- Filippidis, A.; Godelitsas, A.; Charistos, D.; Misaelides, P.; Kassoli-Fournaraki, A. The chemical behavior of natural zeolites in aqueous environments: Interactions between low-silica zeolites and 1 M NaC1 solutions of different initial pH-values. Appl. Clay Sci. 1996, 11, 199–209. [Google Scholar] [CrossRef]
- Chutia, P.; Kato, S.; Kojima, T.; Satokawa, S. Adsorption of As(V) on surfactant-modified natural zeolites. J. Hazard. Mater. 2009, 162, 204–211. [Google Scholar] [CrossRef]
- De Gennaro, B.; Catalanotti, L.; Bowman, R.S.; Mercurio, M. Anion exchange selectivity of surfactant modified clinoptilolite-rich tuff for environmental remediation. J. Colloid Interface Sci. 2014, 430, 178–183. [Google Scholar] [CrossRef]
- Li, Z. Influence of Solution pH and Ionic Strength on Chromate Uptake by Surfactant-Modified Zeolite. J. Environ. Eng. 2004, 130, 205–208. [Google Scholar] [CrossRef]
- Leyva-Ramos, R.; Jacobo-Azuara, A.; Diaz-Flores, P.E.; Guerrero-Coronado, R.M.; Mendoza-Barron, J.; BerberMendoza, M.S. Adsorption of chromium(VI) from an aqueous solution on a surfactant-modified zeolite. Colloids Surf. A Physicochem. Eng. Asp. 2008, 330, 35–41. [Google Scholar] [CrossRef]
- Mendoza-Barrón, J.; Jacobo-Azuara, A.; Leyva-Ramos, R.; Berber-Mendoza, M.S.; Guerrero-Coronado, R.M.; Fuentes-Rubio, L.; Martínez-Rosales, J.M. Adsorption of arsenic (V) from a water solution onto a surfactant-modified zeolite. Adsorption 2010, 17, 489–496. [Google Scholar] [CrossRef]
- Haggerty, G.M.; Bowman, R.S. Sorption of chromate and other inorganic anions by organo-zeolite. Environ. Sci. Technol. 1994, 28, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Ghadiri, S.K.; Nabizadeh, R.; Mahvi, A.H.; Nasseri, S.; Kazemian, H.; Mesdaghinia, A.R.; Nazmara, S. Methyl tert-butyl ther adsorption on surfactant modified natural zeolites. Iran. J. Environ. Health Sci. Eng. 2010, 7, 241–252. [Google Scholar]
- Armağanb, B.; Turan, M.; ęlik, M.S. Equilibrium studies on the adsorption of reactive azo dyes into zeolite. Desalination 2004, 170, 33–39. [Google Scholar] [CrossRef]
- Gómez-Hortigüela, L.; Pérez-Pariente, J.; García, R.; Chebude, Y.; Díaz, I. Natural zeolites from Ethiopia for elimination of fluoride from drinking water. Sep. Purif. Technol. 2013, 120, 224–229. [Google Scholar] [CrossRef]
- Gómez-Hortigüela, L.; Pinar, A.B.; Pérez-Pariente, J.; Sani, T.; Chebude, Y.; Díaz, I. Ion-exchange in natural zeolite stilbite and significance in defluoridation ability. Microporous Mesoporous Mater. 2014, 193, 93–102. [Google Scholar] [CrossRef]
- Adem, M.; Sani, T.; Chebude, Y.; Fetter, G.; Bosch, P.; Diaz, I. Comparison of the defluoridation capacity of zeolites from Ethiopia and Mexico. Bull. Chem. Soc. Ethiop. 2015, 29, 53. [Google Scholar] [CrossRef]
- Cai, Q.; Turner, B.D.; Sheng, D.; Sloan, S. The kinetics of fluoride sorption by zeolite: Effects of cadmium, barium and manganese. J. Contam. Hydrol. 2015, 177–178, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Fang, Q.; Dong, J.; Cheng, X.; Xu, J. Removal of fluoride from drinking water by natural stilbite zeolite modified with Fe(III). Desalination 2011, 277, 121–127. [Google Scholar] [CrossRef]
- Velazquez-Peña, G.C.; Solache-Ríos, M.; Martínez-Miranda, V. Competing Effects of Chloride, Nitrate, and Sulfate Ions on the Removal of Fluoride by a Modified Zeolitic Tuff. Water Air Soil Pollut. 2014, 226, 2236. [Google Scholar] [CrossRef]
- Zhang, Z.; Tan, Y.; Zhong, M. Defluorination of wastewater by calcium chloride modified natural zeolite. Desalination 2011, 276, 246–252. [Google Scholar] [CrossRef]
- Peng, S.; Zeng, Q.; Guo, Y.; Niu, B.; Zhang, X.; Hong, S. Defluoridation from aqueous solution by chitosan modified natural zeolite. J. Chem. Technol. Biotechnol. 2013, 88, 1707–1714. [Google Scholar] [CrossRef]
- Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry and Use. Anal. Chim. Acta 1975, 75, 493. [Google Scholar] [CrossRef]
- Kosmulski, M. Surface Charging and Points of Zero Charge; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Rožić, M.; Šipušić, Đ.I.; Sekovanić, L.; Miljanić, S.; Ćurković, L.; Hrenović, J. Sorption phenomena of modification of clinoptilolite tuffs by surfactant cations. J. Colloid Interface Sci. 2009, 331, 295–301. [Google Scholar] [CrossRef]
- Li, Z. Sorption Kinetics of Hexadecyltrimethylammonium on Natural Clinoptilolite. Langmuir 1999, 15, 6438–6445. [Google Scholar] [CrossRef]
- Ghomashi, P.; Charkhi, A.; Kazemeini, M.; Yousefi, T. Removal of Fluoride from Wastewater by Natural and Modified Nano Clinoptilolite Zeolite. J. Water Environ. Nanotechnol. 2020, 5, 270–282. [Google Scholar]
- Shevade, S.; Ford, R.G. Use of synthetic zeolites for arsenate removal from pollutant water. Water Res. 2004, 38, 3197–3204. [Google Scholar] [CrossRef] [PubMed]
- Ragnarsdottir, K.V. Dissolution kinetics of heulandite at pH 2–12 and 25 °C. Geochem. Cosmochim. Acta 1993, 57, 2439–2449. [Google Scholar] [CrossRef]
- Gilani, N.S.; Tilami, S.E.; Azizi, S.N. One-step green synthesis of nano-sodalite zeolite and its performance for the adsorptive removal of crystal violet. J. Chin. Chem. Soc. 2021, 68, 2264–2273. [Google Scholar] [CrossRef]
- Yürekli, Y. Determination of adsorption capacities of NaX Nano-particles against heavy metals and dyestuff. J. Fac. Eng. Archit. Gaz. 2019, 34, 2113–2124. [Google Scholar]
- Nassara, M.Y.; Abdelrahman, E.A.; Aly, A.A.; Mohamed, T.Y. A facile synthesis of mordenite zeolite nanostructures for efficient bleaching of crude soybean oil and removal of methylene blue dye from aqueous media. J. Mol. Liq. 2017, 248, 302–313. [Google Scholar] [CrossRef]
- Goyal, N.; Barman, S.; Bulasara, V.K. Efficient removal of bisphenol S from aqueous solution by synthesized nano-zeolite secony mobil-5. Microporous Mesoporous Mater. 2018, 259, 184–194. [Google Scholar] [CrossRef]
- Ghifari, M.A.; Nuraini, A.; Permatasari, D.; Kamila, N.; Imanullah, T.; Astuti, Y. Nano-Zeolite Modification Using Cetylpiridinium Bromide for the Removal of Remazol Black B and Remazol Yellow G Dyes. Adv. Sci. Lett. 2017, 23, 6502–6505. [Google Scholar] [CrossRef]
- Hassaninejad-Darzi, S.K.; Kavyani, S.; Torkamanzadeh, M.; Tilaki, R.D. Applicability of ZSM-5 nanozeolite to removal of ternary basic dyes: An adsorption study using high-accuracy UV/Vis-chemometric methods. Monatsh, Chem. 2017, 148, 2037–2049. [Google Scholar] [CrossRef]
- Sivalingam, S.; Sen, S. Efficient removal of textile dye using nanosized fly ash derived zeolite-x: Kinetics and process optimization study. J. Taiwan Inst. Chem. Eng. 2019, 96, 305–314. [Google Scholar] [CrossRef]
- Sarabadan, M.; Bashiri, H.; Mousavi, S.M. Adsorption of crystal violet dye by a zeolite-montmorillonite nano-adsorbent: Modelling, kinetic and equilibrium studies. Clay Miner. 2019, 54, 357–368. [Google Scholar] [CrossRef]
- Robles-Mora, G.; Barrera-Cortés, J.; Valdez-Castro, L.; Solorza-Feria, O.; García-Díaz, C. Polycyclic Aromatic Hydrocarbon Sorption by Functionalized Humic Acids Immobilized in Micro- and Nano-Zeolites. Sustainability 2021, 13, 10391. [Google Scholar] [CrossRef]
- Hu, G.; Yang, J.; Duan, X.; Farnood, R.; Yang, C.; Yang, J.; Liu, W.; Liu, Q. Recent developments and challenges in zeolite-based composite photocatalysts for environmental applications. Chem. Eng. J. 2021, 417, 129209. [Google Scholar] [CrossRef]
- Miklos, D.B.; Remy, C.; Jekel, M.; Linden, K.G.; Drewes, J.E.; Hübner, U. Evaluation of advanced oxidation processes for water and wastewater treatment—A critical review. Water Res. 2018, 139, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Marcelo, C.R.; Puiatti, G.A.; Nascimento, M.A.; Oliveira, A.F.; Lopes, R.P. Degradation of the Reactive Blue 4 Dye in Aqueous Solution Using Zero-Valent Copper Nanoparticles. J. Nanomater. 2018, 2018, 4642038. [Google Scholar] [CrossRef]
- Sun, S.; Xiao, W.; You, C.; Zhou, W.; Garba, Z.N.; Wang, L.; Yuan, Z. Methods for preparing and enhancing photocatalytic activity of basic bismuth nitrate. J. Clean. Prod. 2021, 294, 126350. [Google Scholar] [CrossRef]
- Viter, R.; Iatsunskyi, I. Optical Spectroscopy for Characterization of Metal Oxide Nanofibers. In Handbook of Nanofibers; Barhoum, A., Bechelany, M., Makhlouf, A., Eds.; Springer Nature: Cham, Switzerland, 2019. [Google Scholar]
- Irshad, M.A.; Nawaz, R.; ur Rehman, M.Z.; Adrees, M.; Rizwan, M.; Ali, S.; Ahmad, S.; Tasleem, S. Synthesis, characterization and advanced sustainable applications of titanium dioxide nanoparticles: A review. Ecotoxicol. Environm. Saf. 2021, 212, 111978. [Google Scholar] [CrossRef]
- Anandan, S.; Yoon, M. Photocatalytic activities of the nano-sized TiO2-supported Y-zeolites. J. Photochem. Photobiol. C 2003, 4, 5–18. [Google Scholar] [CrossRef]
- Fonseca-Cervantes, O.R.; Pérez-Larios, A.; Arellano, V.H.R.; Sulbaran-Rangel, B.; Guzmán González, C.A. Effects in Band Gap for Photocatalysis in TiO2 Support by Adding Gold and Ruthenium. Processes 2020, 8, 1032. [Google Scholar] [CrossRef]
- Velásquez, J.; Valencia, S.; Rios, L.; Restrepo, G.; Marín, J. Characterization and photocatalytic evaluation of polypropylene and polyethylene pellets coated with P25 TiO2 using the controlled-temperature embedding method. Chem. Eng. J. 2012, 203, 398–405. [Google Scholar] [CrossRef]
- Šuligoj, A.; Pavlović, J.; Arčon, I.; Rajić, N.; Tušar, N.N. SnO2-Containing Clinoptilolite as a Composite Photocatalyst for Dyes Removal from Wastewater under Solar Light. Catalysts 2020, 10, 253. [Google Scholar] [CrossRef]
- Zou, W.; Gao, B.; Ok, Y.S.; Dong, L. Integrated adsorption and photocatalytic degradation of volatile organic compounds (VOCs) using carbon-based nanocomposites: A critical review. Chemosphere 2019, 218, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Valente, J.P.S.; Padilha, P.M.; Florentino, A.O. Studies on the adsorption and kinetics of photodegradation of a model compound for heterogeneous photocatalysis onto TiO2. Chemosphere 2006, 64, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Armenise, S.; Garcia-Bordeje, E.; Valverde, J.L.; Romeo, E.; Monzón, A. A Langmuir–Hinshelwood approach to the kinetic modelling of catalytic ammonia decomposition in an integral reactor. Phys. Chem. Chem. Phys. 2013, 15, 12104–12117. [Google Scholar] [CrossRef]
- Chekema, C.T.; Goetza, V.; Richardsonb, Y.; Plantarda, G.; Blinc, J. Modelling of adsorption/photodegradation phenomena on AC-TiO2 composite catalysts for water treatment detoxification. Catal. Today 2019, 328, 183–188. [Google Scholar] [CrossRef]
- Giovannetti, R.; Rommozzi, E.; D’Amato, C.A.; Zannotti, M. Kinetic Model for Simultaneous Adsorption/Photodegradation Process of Alizarin Red S in Water Solution by Nano-TiO2 under Visible Light. Catalysts 2016, 6, 84. [Google Scholar] [CrossRef]
- Sun, P.; Zhang, J.; Liu, W.; Wang, Q.; Cao, W. Modification to L-H Kinetics Model and Its Application in the Investigation on Photodegradation of Gaseous Benzene by Nitrogen-Doped TiO2. Catalysts 2018, 8, 326. [Google Scholar] [CrossRef]
- Ali, M.H.H.; Goher, M.E.; Al-Afify, A.D.G. Kinetics and Adsorption Isotherm Studies of Methylene Blue Photodegradation Under UV Irradiation Using reduced Graphene Oxide-TiO2 Nanocomposite in Different Wastewaters Effluents. Egypt. J. Aquat. Biol. Fish. 2019, 23, 253–263. [Google Scholar] [CrossRef]
- Kakhki, R.M.; Karimian, A.; Hasan-nejad, H.; Ahsani, F. Zinc Oxide–Nanoclinoptilolite as a Superior Catalyst for Visible Photo-Oxidation of Dyes and Green Synthesis of Pyrazole Derivatives. J. Inorg. Organomet. Polym. Mater 2019, 29, 1358–1367. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Y.; Diao, J.; Qiang, M.; Chen, Z. Synthesis and Photocatalytic Activity of Hierarchical Zn-ZSM-5 Structures. Catalysts 2021, 11, 797. [Google Scholar] [CrossRef]
- Nezamzadeh-Ejhieh, A.; Shirzadi, A. Enhancement of the photocatalytic activity of Ferrous Oxide by doping onto the nano-clinoptilolite particles towards photodegradation of tetracycline. Chemosphere 2014, 107, 136–144. [Google Scholar] [CrossRef]
- Pourtaheri, A.; Nezamzadeh-Ejhieh, A. Enhancement in photocatalytic activity of NiO by supporting onto an Iranian clinoptilolite nano-particles of aqueous solution of cefuroxime pharmaceutical capsule. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 137, 338–344. [Google Scholar] [CrossRef]
- Bahrami, M.; Nezamzadeh-Ejhieh, A. Effect of supporting and hybridizing of FeO and ZnO semiconductors onto an Iranian clinoptilolite nano-particles and the effect of ZnO/FeO ratio in the solar photodegradation of fish ponds waste water. Mater. Sci. Semicond. Process. 2014, 27, 833–840. [Google Scholar] [CrossRef]
- Babaahamdi-Milani, M.; Nezamzadeh-Ejhieh, A. A comprehensive study on photocatalytic activity of supported Ni/Pb sulfide and oxide systems onto natural zeolite nanoparticles. J. Hazard. Mater. 2016, 318, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Nezamzadeh-Ejhieh, A.; Zabihi-Mobarakeh, H. Heterogeneous photodecolorization of mixture of methylene blue and bromophenol blue using CuO-nano-clinoptilolite. J. Ind. Eng. Chem. 2014, 20, 1421–1431. [Google Scholar] [CrossRef]
- Derikvandi, H.; Nezamzadeh-Ejhieh, A. Comprehensive study on enhanced photocatalytic activity of heterojunction ZnS-NiS/zeolite nanoparticles: Experimental design based on response surface methodology (RSM), impedance spectroscopy and GC-MASS studies. J. Colloid Interface Sci. 2017, 490, 652–664. [Google Scholar] [CrossRef]
- Esmaili-Hafshejani, J.; Nezamzadeh-Ejhieh, A. Increased photocatalytic activity of Zn(II)/Cu(II) oxides and sulfides by coupling and supporting them onto clinoptilolite nanoparticles in the degradation of benzophenone aqueous solution. J. Hazard. Mater. 2016, 316, 194–203. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Lyu, Z.; Gu, Q.; Zhang, L.; Wang, J. Ceramic-based membranes for water and wastewater treatment. Colloids Surf. A 2019, 578, 123513. [Google Scholar] [CrossRef]
- Far, R.M.; Van der Bruggen, B.; Verliefdea, A.; Cornelissena, E. A review of zeolite materials used in membranes for water purification: History, applications, challenges and future trends. J. Chem. Technol. Biotechnol. 2021. [Google Scholar] [CrossRef]
- Sarkar, S.; Chakraborty, S. Nanocomposite polymeric membrane a new trend of water and wastewater treatment: A short review. Groundw. Sustain. Dev. 2021, 12, 100533. [Google Scholar] [CrossRef]
- Ismail, A.F.; Padaki, M.; Hilal, N.; Matsuura, T.; Laua, W.J. Thin film composite membrane—Recent development and future potential. Desalination 2015, 356, 140–148. [Google Scholar] [CrossRef]
- Adam, M.R.; Othman, M.H.D.; Samah, R.A.; Puteh, M.H.; Ismail, A.F.; Mustafa, A.; Rahman, M.A.; Jaafara, J. Current trends and future prospects of ammonia removal in wastewater: A comprehensive review on adsorptive membrane development. Sep. Purif. Technol. 2019, 213, 114. [Google Scholar] [CrossRef]
- Kim, S.; Nam, S.N.; Jang, A.; Jang, M.; Park, C.M.; Son, A.; Her, N.; Heo, J.; Yoon, Y. Review of adsorption–membrane hybrid systems for water and wastewater treatment. Chemosphere 2022, 286, 131916. [Google Scholar] [CrossRef]
- Cui, J.; Li, F.; Wang, Y.; Zhang, Q.; Ma, W.; Huang, C. Electrospun nanofiber membranes for wastewater treatment applications. Sep. Purif. Technol. 2020, 250, 117116. [Google Scholar] [CrossRef]
- Habiba, U.; Siddique, T.A.; Lee, J.J.L.; Joo, T.C.; Ang, B.C.; Afifi, A.M. Adsorption study of methyl orange by chitosan/polyvinyl alcohol/zeolite electrospun composite nanofibrous membrane. Carbohydr. Polym. 2018, 191, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Habiba, U.; Afifi, A.M.; Salleh, A.; Ang, B.C. Chitosan/(polyvinyl alcohol)/zeolite electrospun composite nanofibrous membrane for adsorption of Cr6+, Fe3+ and Ni2+. J. Hazard. Mater. 2017, 322, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Saranya, R.; Arthanareeswaran, G.; Ismail, A.F. Enhancement of anti-fouling properties during the treatment of paper mill effluent using functionalized zeolite and activated carbon nanomaterials based ultrafiltration. J. Chem. Technol. Biotechnol. 2019, 94, 2805–2815. [Google Scholar] [CrossRef]
- Anis, S.F.; Lalia, B.S.; Lesimple, A.; Hashaikeh, R.; Hilal, N. Electrically conductive membranes for contemporaneous dye rejection and degradation. Chem. Eng. J. 2022, 428, 131184. [Google Scholar] [CrossRef]
- Sultana, N.; Rahman, R. Electrospun nanofiber composite membranes based on cellulose acetate/nano-zeolite for the removal of oil from oily wastewater. Emerg. Mater. 2021. [Google Scholar] [CrossRef]
- Moghimifar, V.; Livari, A.E.; Raisi, A.; Aroujalian, A. Enhancing the antifouling property of polyethersulfone ultrafiltration membranes using NaX zeolite and titanium oxide nanoparticles. RSC Adv. 2015, 5, 55964–55976. [Google Scholar] [CrossRef]
- Esmaeili, A.; Saremnia, B. Comparison study of adsorption and nanofiltration methods for removal of total petroleum hydrocarbons from oil-field wastewater. J. Pet. Sci. Eng. 2018, 171, 403–413. [Google Scholar] [CrossRef]
- Yu, C.; Wu, J.; Zin, G.; Di Luccio, M.; Wen, D.; Qilin, L. D-Tyrosine loaded nanocomposite membranes for environmental-friendly, long-term biofouling control. Water Res. 2018, 30, 105–114. [Google Scholar] [CrossRef]
- Omer, A.M.; Dey, R.; Eltaweil, A.S.; Abd El-Monaem, E.M.; Ziora, Z.M. Insights into recent advances of chitosan-based adsorbents for sustainable removal of heavy metals and anions. Arab. J. Chem. 2022, 15, 103543. [Google Scholar] [CrossRef]
- Yurekli, Y. Removal of heavy metals in wastewater by using zeolite nano-particles impregnated polysulfone membranes. J. Hazard. Mater. 2016, 309, 53–64. [Google Scholar] [CrossRef] [PubMed]
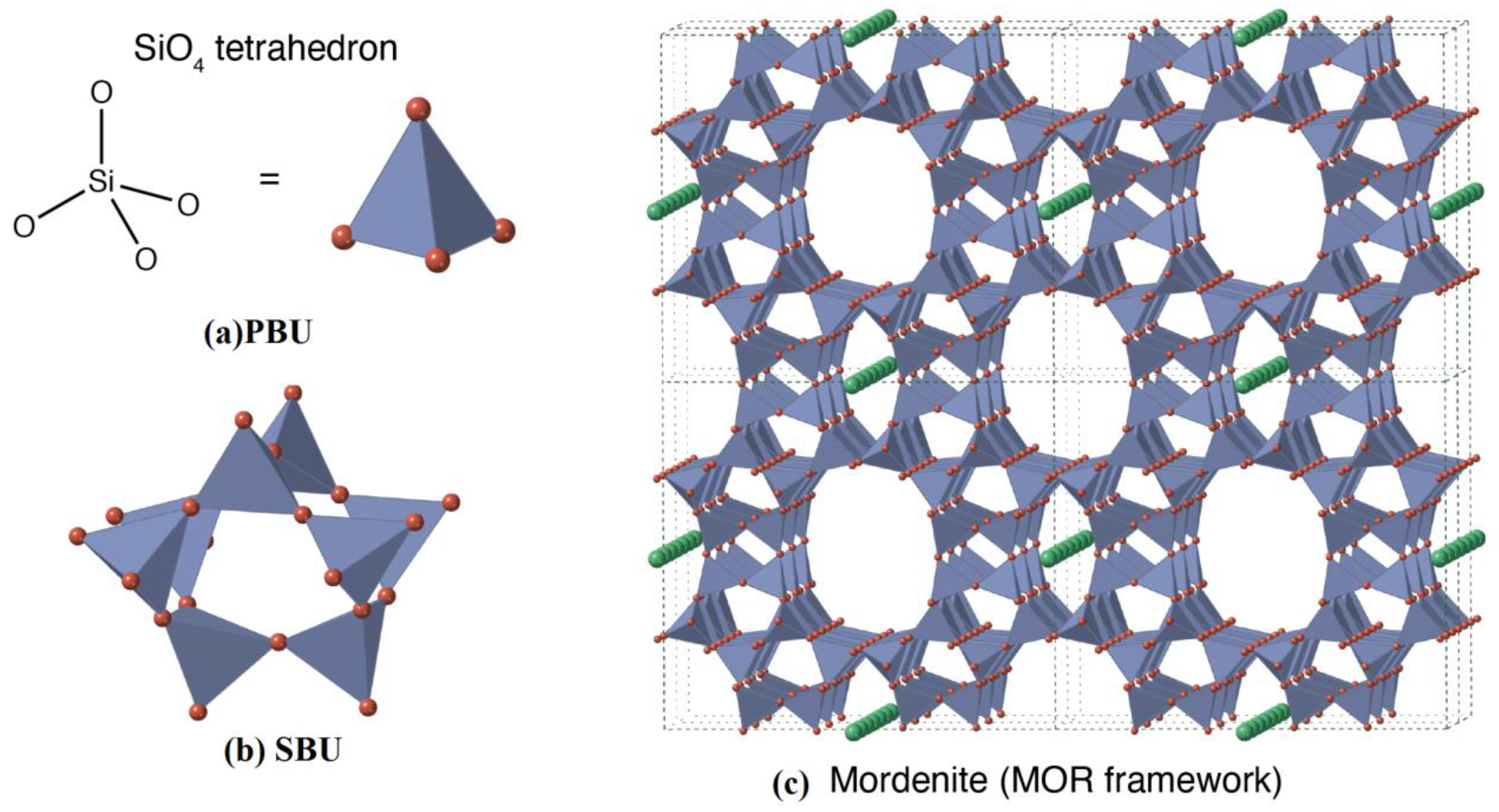
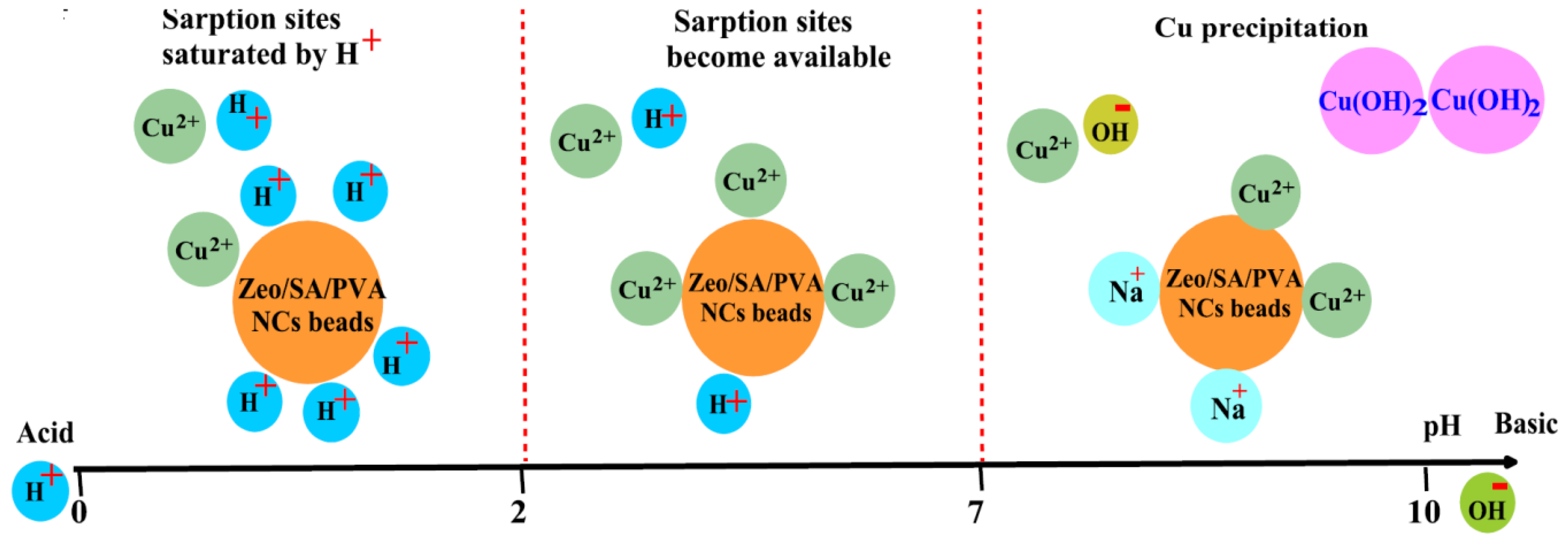
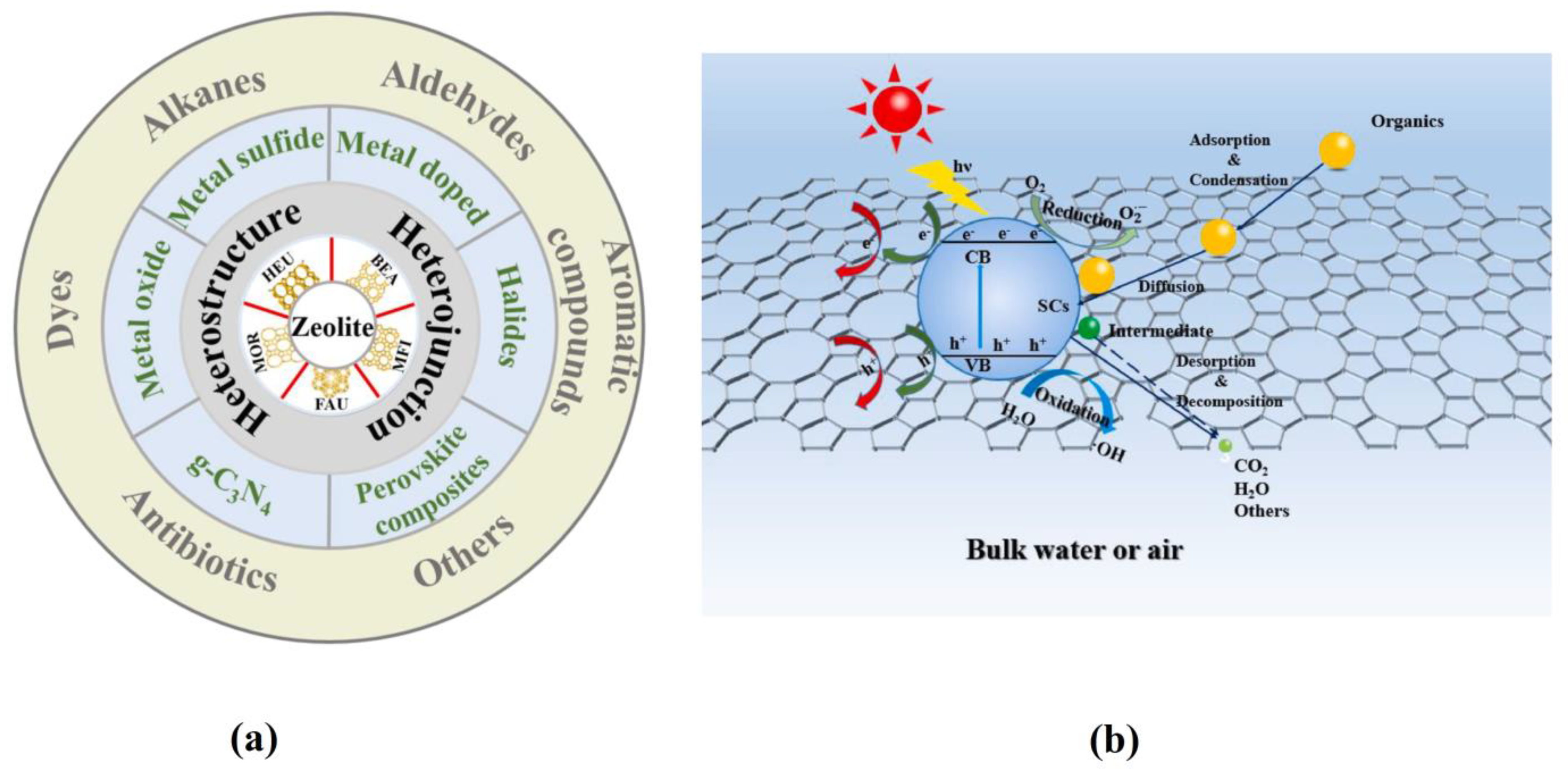
| Exchanger | Preparation Conditions | Ref |
|---|---|---|
| Zeolite Na-A | Si:Al < 3, at 100 °C for 2–3 days | [8] |
| Zeolite Na-X | Si:Al ratios of 2.8:1, at 50 °C (6 h) * and 100 °C (3 h) for 2–3 days | [8] |
| Zeolite Na A-X blend | Si:Al ratios of 2:1, at 80 °C (2 h for gelification), 25 °C (24 h for ageing), and 90 °C (8 h for crystallization). | [10,11,12] |
| Clinoptilolite | Different silica, alumina and alkali moleratios, temperatures (250–300 °C) and pressure (42.5–81.6 atm) for 2–5 days. | [13] |
| Analcite | Si:Al ratios of 2:1, at 275 °C for 2–3 days | [14] |
| Mordenite | Si:Al = 6:1 at 275 °C for 2–3 days | [14] |
| Zeolite y, nano scale | SiO2:Al2O3 = 4.35:1, at 100 °C, 2 days | [15] |
| Technique | Feature | Advantage | Limitation |
|---|---|---|---|
| OFAT | Evaluate isolated effects of the studied factors on a single performance measure Empirical, mechanistic, and black box models are used to analyze the data | Allow the determination of mechanisms, interpolate and extrapolate the process performance | Does not allow the determination of the effect of interaction between the factors that affect the performance |
| MVT | Evaluate the effects of the studied factors variability and their interactions on single and multi performance measures | Identify the main influencing factors, Provide insights into the system reliability | Does not allow the determination of the mechanism |
| Model | Equation | Model Features | |
|---|---|---|---|
| KINETIC | PFO | Rate model used to determine the rate constant(k1, min−1) and sorbed contaminant amount per unit mass of zeolite at equilibrium (qe, mmol.g−1), Entails that the reaction rate is limited by only one process or mechanism on a single class of sorbing sites and that all sites are of the time dependent type, The reaction might be controlled by diffusion through the boundary layer. | |
| PSO | Rate model used to determine the rate constant(k2, g.mmol−1.min−1) and sorbed contaminant amount per unit mass of Zeolite at equilibrium (qe, mmol.g−1), Entails that the rate of sorption is directly proportional to the number of active surface sites and that the rate limiting step may be a chemical sorption involving valence forces through sharing or exchange of electrons between the adsorbent and the adsorbate. | ||
| DKM | A rate model that assumes that the reaction proceed via two subsequent mechanisms. It allows the calculation of the sorbed amount of contaminant in at equilibrium and identification of the rate constant for each mechanism | ||
| El. | Used to determine the initial sorption rate (α, mEq.g−1.min−1) and the desorption constant (β, mmol.g−1). Entails that the reaction increases exponentially with time. | ||
| IPM | Used to quantify the boundary layer effect (Cpi, mmol.g−1), and determine the rate constant of the sorption stage (Kpi, mmol.g−1.min−0.5), Entails that the reaction involves diffusion mechanism and allow the assessment of the contribution of the boundary layer in the reaction | ||
| HPM | Used to determine the rate controlling step and calculate the diffusion coefficient (D, m2/s), Film diffusion model entails that the rate determining step is the contaminants diffusion through the liquid film around the Zeolite particles, Particle diffusion model entails that the rate determining step is the contaminants diffusion into the Zeolite particles. | ||
| ISOTHERM | F | Used to determine Freundlich constant indicative of the relative adsorption capacity (Kf, mmol.g−1), Freundlich intensity constant indicative of the relative sorption capacity (n), Empirical model employed to describe the interaction between contaminants and heterogeneous sorbent, It suggests that sorption energy exponentially decreases on filling of the sorption centers of the sorbent. | |
| L | Used to determine the mono-layer capacity (Qo, mmol.g−1) and Langmuir constant (b) Assumes that the sorption takes place at specific homogenous sites, energetically equivalent, within the sorbent, The sorbent has a finite capacity for the contaminants. | ||
| DR | Used to determine the maximum amount of ion that can be sorbed onto unit weight of zeolite (qm, mmol.g−1) constant related to sorption energy (mol2.K.J−2), Polanyi sorption potential, ε is the work required to remove a molecule to infinity from its location in the sorption space, Employed to describe the interaction between contaminants and heterogeneous sorbent, Used to differentiate between physical and chemical sorption. | ||
| Thermo | & | Used to determining the thermodynamic parameters, i.e., Gibbs free energy change (ΔG, KJ.mol−1) and the change in entropy (ΔS, kJ.mol−1.K−1) and in enthalpy ΔH, kJ.mol−1) from the real thermodynamic equilibrium constant (Kc). | |
| Cont. | Nano-Zeolite | m/Vg/L | Kinetic Investigations | Capacity | Thermodynamic Parameters | Regeneration | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type | Size, nm | Composite | Preparation | Co, mmol/L | pH | teq, min | Temp, K | Model | Qο @RT mmol/g | ΔH kJ.mol−1 | ΔS J.mol−1.K | (-) ΔG @RT kJ.mol−1 | ||||
| Cs1 | Zeolite Y | 20–50 | - | HT, Chemicals | 2 | 5.82 | 6 | 60 | 298–313 | PSO | 6.72 | - | - | - | [15] MVT | |
| Zeolite Y | <100 | Mangetite | 2 | - | - | - | - | - | 1.17 | 2.75 | 35 | 7.93 | - | [61] | ||
| Zeolite A | >50 | Magnetic | 10 | 10 | 8 | 120 | 298 | PSO | 1.724 | 3.08 | 60 | 15.0 | [62] | |||
| CHA | <510 | - | 1 | 0.752 | - | 1 | RT | PSO | 0.3 | -20.02 | −14 | 15.8 | [64] | |||
| Zeolite A | 82 ± 9 | - | HT, fly ash | 10 | 0.752 | 7 | 1440 | RT | - | - | - | - | - | - | * [67] | |
| Zeolite X | 86 ± 12 | - | HT, fly ash | 10 | 0.752 | 7 | 1440 | RT | - | - | - | - | - | - | * [67] | |
| Eu3 | MOR1 | Sphere, D = 50 | - | HT, Chemicals | 4 | 3 | 1 | 1440 | 303 ± 1 | PSO | 2.72 | 8.81 | 74.75 | 13.8 | - | [63] |
| MOR2 | - | 2.87 | 7.05 | 69.75 | 14.1 | - | ||||||||||
| MOR3 | Rod, L = 400, D = 25:50 | - | 2.98 | 9.64 | 79.02 | 14.3 | - | |||||||||
| MOR4 | - | 3.50 | 10.15 | 82.84 | 14.9 | - | ||||||||||
| Sr2 | Zeolite Y | 20–50 | - | HT, Chemicals | 2 | 19.72 | 6 | 60 | 298–313 | PSO | 15.42 | - | - | - | - | [15] MVT |
| Zeolite Y | <100 | Mangetite | 2 | - | - | - | - | - | 1.38 | 8.43 | 41 | 7.16 | [61] | |||
| Zeolite A | >50 | Magnetic | 10 | 20 | 8 | 120 | 298 | PSO | 1.016 | 12.16 | 100 | 18.1 | [62] | |||
| CHA-3 | 100–300 | - | HT, Chemicals | 10 | 1.14 | - | 60 | 298 | PSO | 0.131 | - | - | - | 5 | [54] | |
| Zeolite A | 82 ± 9 | - | HT, fly ash | 10 | 1.14 | 7 | 1440 | RT | - | - | - | - | - | - | * [67] | |
| Zeolite X | 86 ± 12 | - | HT, fly ash | 10 | 11.4 | 7 | 1440 | RT | - | - | - | - | - | - | * [67] | |
| Natural | 109.9 | PVA/ALG | - | 20 | 0.285 | 6 | 120 | 298 | PSO | - | - | - | - | - | [76] | |
| Th4 | MOR1 | Sphere, D = 50 | - | HT, Chemicals | 4 | 4 | 1 | 1440 | 303 ± 1 | PSO | 1.18 | 11.74 | 75.86 | 11.2 | - | [63] |
| MOR2 | - | 1.23 | 11.2 | 73.65 | 11.3 | - | ||||||||||
| MOR3 | Rod, L = 400 D = 25:50 | - | 1.11 | 11.78 | 78.72 | 11.0 | - | |||||||||
| MOR4 | - | 1.55 | 9.74 | 71.20 | 11.8 | - | ||||||||||
| HZSM-5 | Fiber D = 98 | PVA/ALG/PEO | HT, Chemicals | 1 | - | 5.5 | 240 | 298 | DKM | 1.138 | 35.67 | 145.3 | 7.704 | - | [65] MVT | |
| ZSM5 | - | PVA/ALG/PEO | HT, Chemicals | 1 | 0.517 | 5 | 150 | 298 | DKM | 0.569 | 25.962 | 97.2 | 3.039 | - | [66] MVT | |
| U4 | HZSM-5 | Fiber D = 98 | PVA/ALG/PEO | HT, Chemicals | 1 | - | 5.5 | 240 | 298 | DKM | 0.577 | 21.34 | 81.9 | 3.11 | - | [65] MVT |
| Cont. | Nano-Zeolite | m/Vg/L | Kinetic Investigations | Capacity | Thermodynamic Parameters | Regeneration | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type | Size, nm | Composite | Preparation | Co, mmol/L | pH | teq, min | Temp, K | Model | Qο @RT mmol/g | ΔH Kj.mol−1 | ΔS J.mol−1K | (-) ΔG @RT Kj.Mol−1 | ||||
| Al3 | Natural | 109.9 | PVA/ALG | - | 20 | 0.926 | 6 | 120 | 298 | PSO | 0.438 | - | - | - | 10 | [76] |
| As3 | - | 50 | Magnetite | - | 16 | 1.33 | - | 60 | - | - | 0.059 | - | - | - | - | [73] |
| Cd2 | Zeolite NaX | Fiber >170 | PVA | HT, chemicals | 0.5 | 0.446 | 5 | 298–318 | PSO | 7.279 | 11.099 | 82 | 13.24 | 5 | [75] | |
| - | 109.9 | PVA/ALG | Natural | 20 | 0.222 | 6 | 120 | 298 | PSO | 0.411 | - | - | - | 10 | [76] | |
| CLP | - | DTPA | Natural | 0.2 | 8.763 | 5.9 | 205 | - | PSO | 1.235 | −10.3 | 102.89 | −20.4 | - | [84] MVT | |
| Co2 | FAU | <150 | Geopolymer | HT, Si &Al wastes | 2 | 4.237 | 8 | 20 | 298 | PSO | 2.27 | −106.88 | 323 | 203.1 | - | [70] |
| ZSM-5 | - | PVA/ALG | HT, chemicals | 1 | 1.695 | - | 240 | 298 | DKM | 1.255 | 16.47 | 55 | 0.16 | - | [69] | |
| - | 12 | GLU | Natural | 10 | 10 | - | 360 | 298 | PSO | 0.179 | - | - | - | - | ** [79] | |
| Cu2 | CLP | 59 | APS | Natural | 5 | 9.448 | - | 300 | 298 | PSO | 1.36 | - | - | - | - | ** [80] |
| FAU | <150 | Geopolymer | HT, Si &Al wastes | 2 | 3.937 | 8 | 20 | 298 | PSO | 1.987 | −150.76 | 464 | 239 | - | [70] | |
| - | 109.9 | PVA/ALG | Natural | 20 | 0.394 | 6 | 120 | 298 | PSO | 0.764 | - | - | - | 10 | [76] | |
| CLP | <40 | CYS | Natural | 15 | 7.874 | - | 1800 | 298 | PSO | 0.521 | - | - | - | - | ** [81] | |
| Fe3 | - | 109.9 | PVA/ALG | Natural | 20 | 0.448 | 6 | 120 | 298 | PSO | 0.845 | - | - | - | 10 | [76] |
| Li3 | - | 109.9 | PVA/ALG | Natural | 20 | 3.62 | 6 | 120 | 298 | PSO | 5.527 | - | - | - | 10 | [76] |
| Mn2 | Natural | 109.9 | PVA/ALG | Natural | 20 | 0.455 | 6 | 120 | 298 | PSO | 0.781 | - | - | - | 10 | [76] |
| CLP | - | GLU | Natural | 5 | 10.6 | 3.5 | 120 | 298 | PSO | 0.101 | - | - | - | 4 | ** [82] MVT | |
| Ni2 | Zeolite NaX | Fiber >170 | PVA | HT, chemicals | 0.5 | 0.341 | 5 | 298–318 | PSO | 5.738 | 6.018 | 60 | 11.9 | - | [75] | |
| - | 109.9 | PVA/ALG | Natural | 20 | 0.426 | 6 | 120 | 298 | PSO | 0.812 | - | - | - | 10 | [76] | |
| CLP | - | DMG | Natural | 10 | 1.707 | 5.5 | 1400 | 298 | PSO | 0.96@293 | - | - | - | - | ** [83] | |
| Pb2 | FAU | 150–250 | Cobalt ferrite | HT, Chemicals | - | 1.038 | 7 | 60 | 298 | PSO | 2.91 | - | - | - | - | [68] |
| Zeolite Y | 150–300 | - | HT, Chemicals | 0.4 | 0.483 | 6 | 60 | 299 | PSO | 2.19 | - | - | - | - | [71] | |
| 30–50 | chitosan | 0.265 | - | - | - | - | [71] | |||||||||
| - | 109.9 | PVA/ALG | Natural | 20 | 0.121 | 6 | 120 | 298 | PSO | 0.229 | - | - | - | 10 | [76] | |
| HZSM-5 | Fiber | PVP/chitosan | HT, chemicals | 1 | 0.48 | 5.5 | 240 | 298 | DKM | 1.46 | 78.35 | 146.68 | 8.22 | - | [74] | |
| MOR | 35.50, | PEG-EG | HT, chemicals | 3 | 0.241 | 8 | 180 | 298 | PSO | 0.084 | −68.82 | 218 | 133.7 | 5 | [78] | |
| Zn2 | FAU | <150 | Geopolymer | HT, Si &Al wastes | 2 | 3.823 | 8 | 20 | 298 | PSO | 2.017 | −83.1 | 250 | 154.0 | - | [70] |
| - | 109.9 | PVA/ALG | Natural | 20 | 0.382 | 6 | 120 | 298 | PSO | 0.739 | - | - | - | 10 | [76] | |
| Cont. | Nano-Zeolite | Preparation Technique | m/Vg/L | Kinetic Investigations | Capacity | Thermodynamic Parameters | Regeneration | Ref. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type | Size, nm | Composite | Co, mg/L | pH | teq, min | Temp, K | Model | Qο @RT mg/g | ΔH kJ.mol−1 | ΔS J.mol−1K | (-)ΔG @RT kJ.mol−1 | |||||
| AB-74 | Zeolite | 40–500 | PA-6 | Natural | 20 | - | 120 | - | PSO | 166.66 | - | - | - | - | [72] MVT | |
| MG | ZF | 46.56 | - | HT, Al waste and Si chemical | 2 | 700 | - | 120 | RT | PSO | 226.757 | −5.819 | 19 | 11.48 | - | [77] |
| ZM | 26.28 | - | 2 | 700 | - | 180 | RT | PSO | 239.234 | −5.715 | 14 | 9.887 | - | [77] | ||
| ZS | 75.83 | - | 2 | 50 | - | 40 | RT | PSO | 29.744 | −22.62 | 65 | 41.99 | - | [77] | ||
| ZT | 38.73 | - | 2 | 50 | - | 50 | RT | PSO | 25.221 | −22.473 | 69 | 43.03 | - | [77] | ||
| CV | Sodalite | 40–90 | - | Low- temp, chemicals | 0.3 | 20 | - | 40 | RT | PSO | 227.2 | 28.006 | 108.22 | 4.225 | * [120] | |
| Zeolite X | 19–39 | - | HT, coal fly ash | 0.75 | - | - | - | - | PSO | 234.57 | - | - | - | 10 | [126] MVT | |
| MB | Zeolite-X | 170 | - | HT, Chemicals | 2.5 | 100 | - | 5 | RT | PSO | 0.1 | - | - | - | - | [121] |
| MOR | 55.34 | - | 10 | - | - | 120 | RT | PFO | 1.72 | −18.98 | 4.6 | 17.6 | - | [122] | ||
| BR-41 | ZSM5 | 40–100 | - | 1.2 | 17, M | 7 | 60 | RT | PSO | 13.76, μM/g | - | - | - | - | [125] MVT | |
| BR-18 | 1.2 | 33, M | 60 | RT | PSO | 28.49, μM/g | - | - | - | - | ||||||
| BR-46 | 1.2 | 20, M | 60 | RT | PSO | 27.6, μM/g | - | - | - | - | ||||||
| Model | Equation | Model Features |
|---|---|---|
| LH | Linear: | The model assumes that the rate of the photo-degradation reaction proportional to the fraction of the surface by the contaminant, It assumes that the available contaminants on the surface are sorbed following the Langmuir monolayer model, The model does not consider the reactions of the intermediates, Kx the rate constant for sorption (x = ad) and photo-degradation (x = LH) |
| Non-linear: | ||
| FO | Assumes that the overall degradation process is a first order reaction valid for diluted solutions Kf is the apparent first order rate constant | |
| Cont. | Nano-Zeolite | Illumination Source | m/Vg/L | Optimum Conditions | Model | Reuse | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type | Size, nm | Composite | Preparation | Co, ppm | pH | teq, min | ||||||
| MB | CLP | 50 | ZnO | Natural | Fluorescence lamp, 60 W | 0.25 | 10 | - | 50 | FO | 4 | [146] |
| MB | ZSM-5 | - | ZnO | HT-chemical | UV-Mercury lamp, 500 W | 0.2 | 50 | - | 30 | FO | 6 | [147] |
| TC | CLP | 100 | FeO | Natural | Hg lamp, 30 W | 0.2 | - | 4.3 | 200 | FO | 6 | [148] |
| CF | CLP | 10–70 | NiO | Natural | Hg lamp, 35 W | 0.025 | - | 4.3 | 200 | LH | - | [149] |
| FP | CLP | - | FeO-ZnO | Natural | Sunlight | 0.1 | - | 8.3 | 140 | - | 5 | [150] |
| 4-NP | CLP | 52 | NiS/PbS | Natural | Hg lamp, 30 W | 0.5 | - | 7.5 | 200 | FO | - | [151] |
| MB | CLP | 100 | CuO | Natural | Hg lamp, 75 W | 0.2 | 7 | 5.9 | 180 | FO | - | [152] |
| MZ | CLP | 30 | ZnS/NiS | Natural | Hg lamp, 35 W | 3 | 4 | 3 | 150 | - | - | [153] MVT |
| BP | CLP | - | ZnO/CuO | Natural | 2 Hg lamp, each 35 W | 0.12 | 30 | 7.5 | 300 | FO | 5 | [154] |
| ZnS/CuS | 0.1 | 30 | 7.5 | 5 | ||||||||
| Cont. | Membrane | Contact Angle, o | Optimum Conditions | Fouling | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Active Layer | Substrate | Type | Co, ppm | Flux, lm2/h | R, % | Pressure, bar | ||||
| CV | Zeolite-Y | CNS | UF- conductive | 40 | 20 | 210 | 100 | @3 volte | - | [165] |
| Engine Oil | Beta | CA | MF | 67.7 | 2% | - | 97 | Vacuum- | - | [166] |
| Oil | Zeolite NaX-TiO2 | PES | UF | - | - | - | - | - | - | [167] |
| TPH | Zeolite -NaA | Polyaniline | NF | - | 35.1-78.0 | 96.99 | 77.79 | 5 | - | [168] |
| Paper mill effluent | Cu–Zeolite | PES | UF | 73.4–74.8 | COD = 1840, PH = 7.6, BOD = 660, So4 = 205, Cl = 340 | 38.9 | COD = 89 BOD = 90.5 | 4.14 | Fouling | [164] |
| Syenthtic wastewater | FAU- D-tyrosine | NF270 | NF | 20 | - | - | - | - | Bio fouling | [169] |
| As | Zeolite | Chitosan | - | 74.2–59.2 | 1000 | - | 94 | - | - | [170] |
| Ni | Zeolite-Na-X | PSf | UF | 67 | 500 | 21 | 91 | 1 | - | [171] |
| Pb | 21 | 42 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, R.O.A.; El-Kamash, A.M.; Hung, Y.-T. Applications of Nano-Zeolite in Wastewater Treatment: An Overview. Water 2022, 14, 137. https://doi.org/10.3390/w14020137
Rahman ROA, El-Kamash AM, Hung Y-T. Applications of Nano-Zeolite in Wastewater Treatment: An Overview. Water. 2022; 14(2):137. https://doi.org/10.3390/w14020137
Chicago/Turabian StyleRahman, Rehab O. Abdel, Ahmed M. El-Kamash, and Yung-Tse Hung. 2022. "Applications of Nano-Zeolite in Wastewater Treatment: An Overview" Water 14, no. 2: 137. https://doi.org/10.3390/w14020137
APA StyleRahman, R. O. A., El-Kamash, A. M., & Hung, Y.-T. (2022). Applications of Nano-Zeolite in Wastewater Treatment: An Overview. Water, 14(2), 137. https://doi.org/10.3390/w14020137








