Optimized Removal of Azo Dyes from Simulated Wastewater through Advanced Plasma Technique with Novel Reactor
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
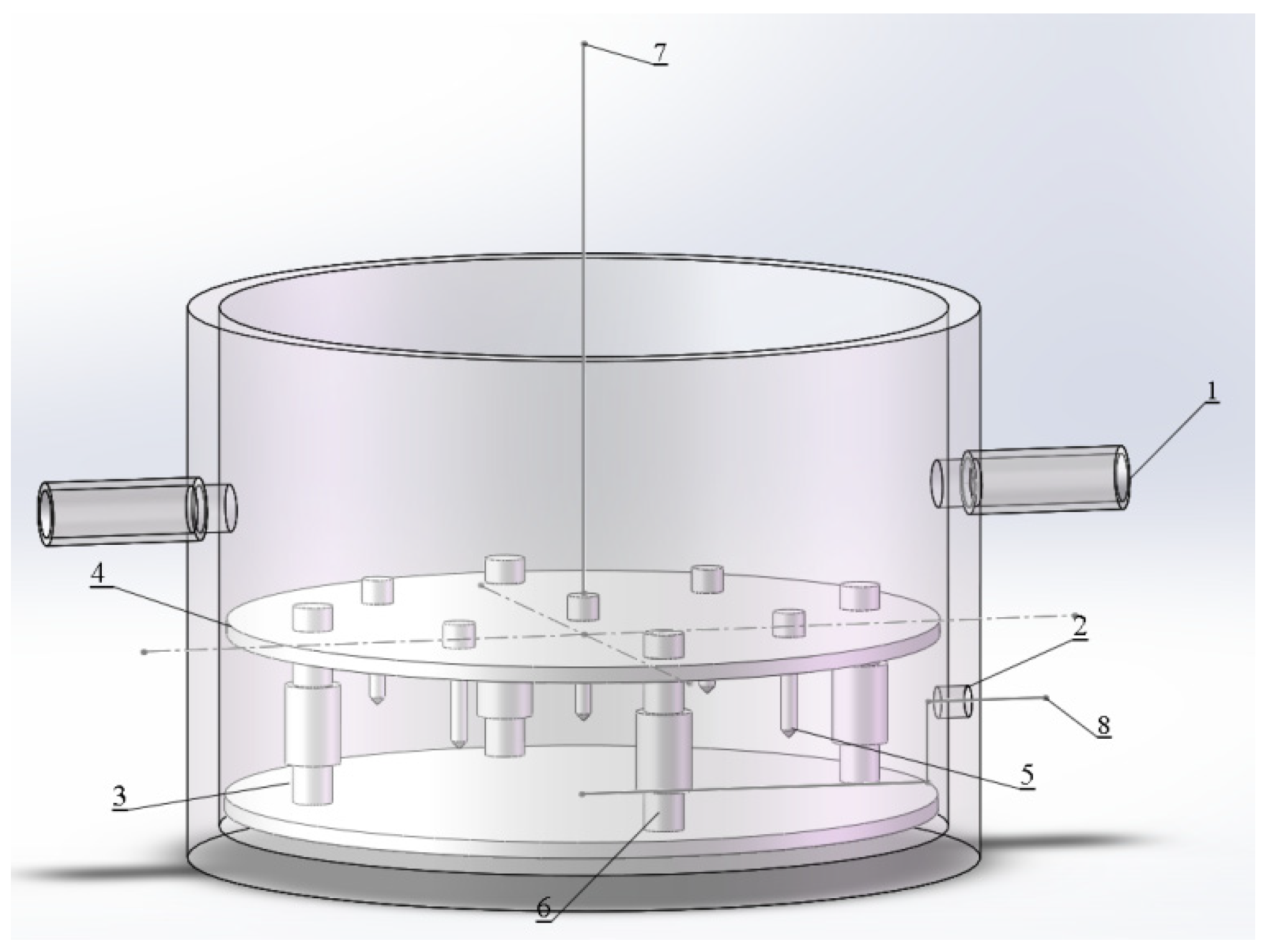
2.2. Experimental Setup and Procedure
2.3. Sample Preparation and Analysis
2.4. Data Analysis
- Removal rate of MO (η) %where η was the removal rate of MO in the solution, C0 and Ct were the concentration of MO solution at times of 0 and t, respectively.
- Energy consumption (W) kWh/gwhere W was the energy consumption of removing organic matter per unit weight, I was the output current, U was the output voltage, t was the discharge time, Δm was the removal weight of organic matter.
- Mineralization rate (XTOC) %where XTOC was the mineralization rate of MO, TOC0 and TOCt were total organic carbon at 0 min and t min, respectively.
2.5. RSM Experimental Design
3. Results and Discussion
3.1. The Influence of Voltage
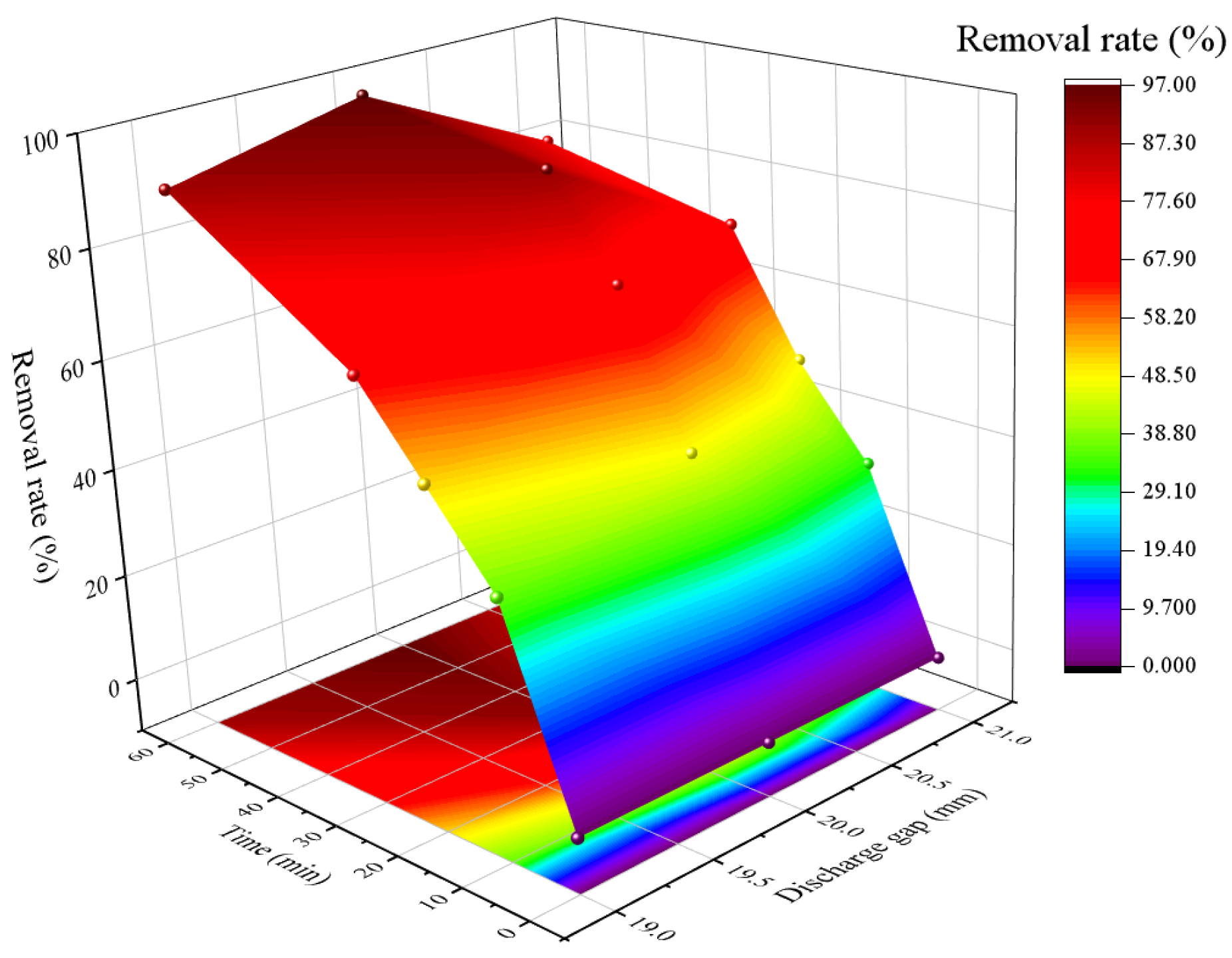
3.2. The Influence of Discharge Gap
3.3. The Influence of Discharge Needle Numbers
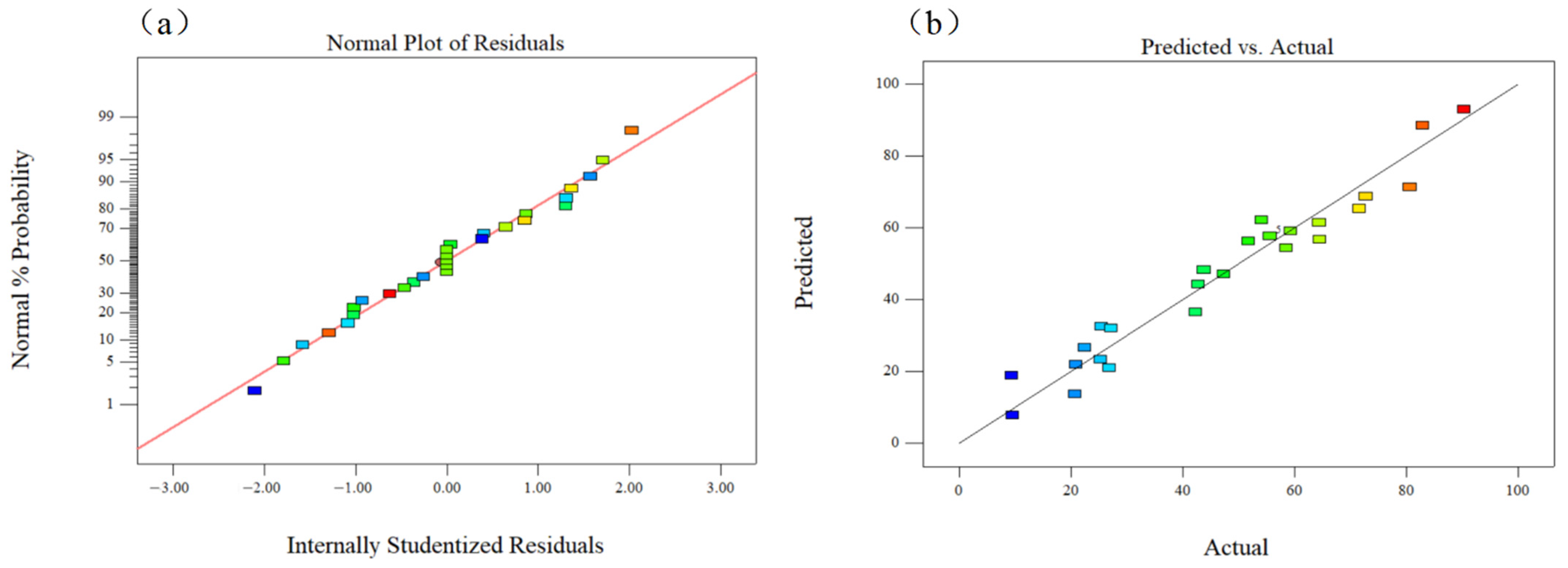
3.4. Analysis of RSM
3.28CD − 8.17A2 − 8.16B2 − 12.12C2 + 2.54D2
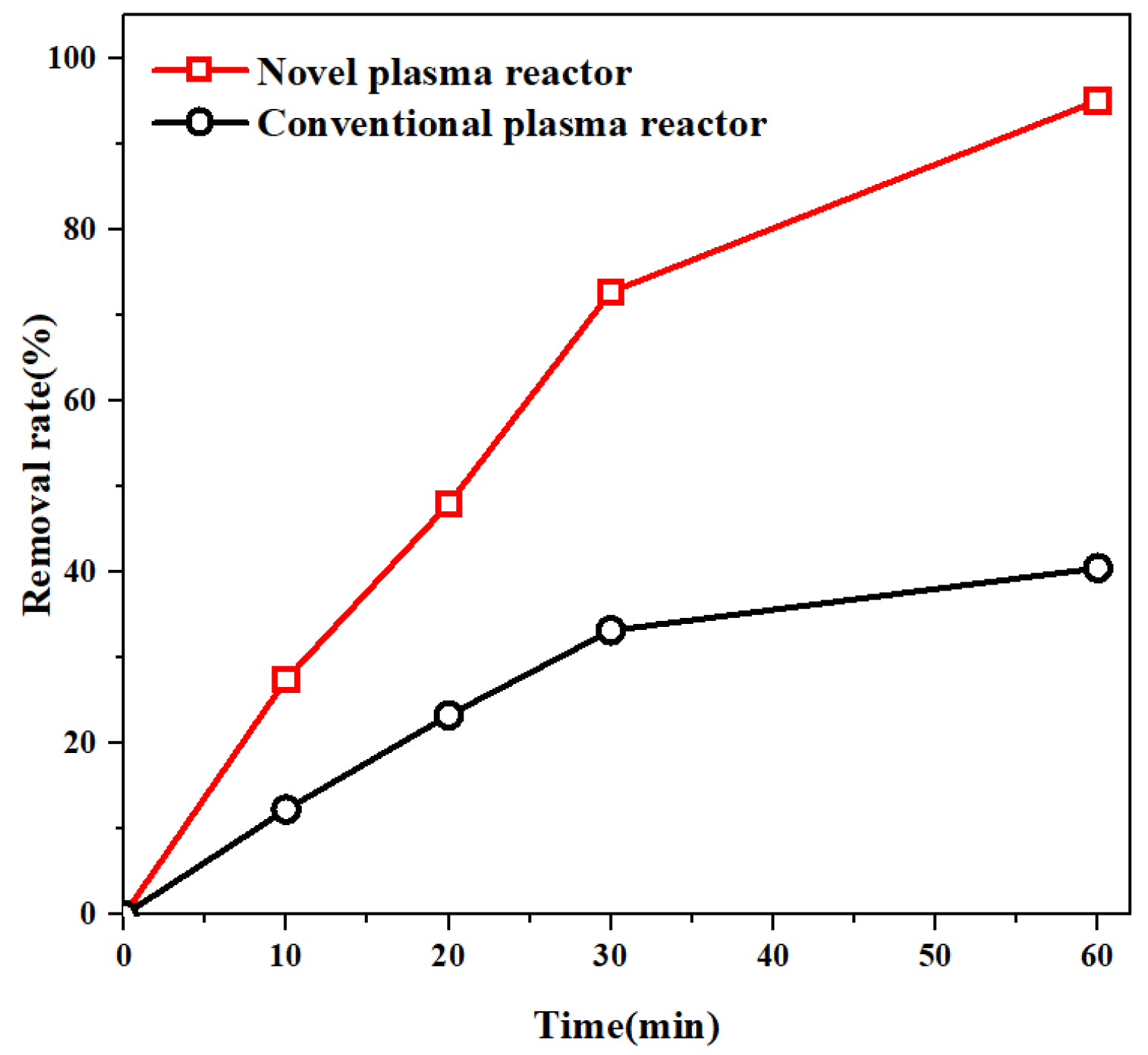
3.5. Advantages of Removal Effect of MO in Novel Plasma Reactor
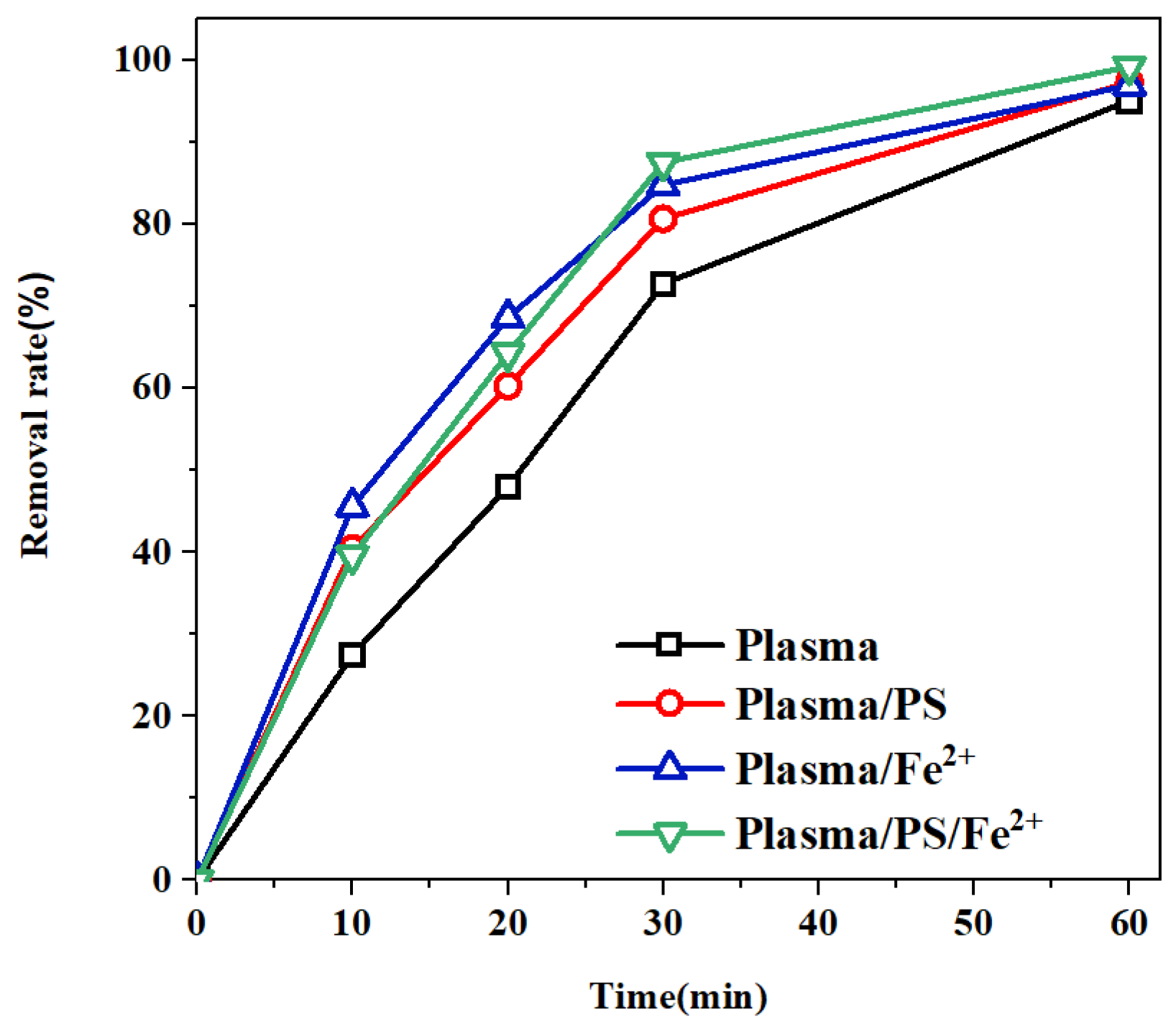
3.6. The Influence of Catalysts on MO
3.7. Removal Mechanism of Plasma for MO Removal
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jamee, R.; Siddique, R. Biodegradation of Synthetic Dyes of Textile Effluent by Microorganisms: An Environmentally and Economically Sustainable Approach. Eur. Microbiol. Immu. 2019, 9, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Ajaz, M.; Shakeel, S.; Rehman, A. Microbial use for azo dye degradation-a strategy for dye bioremediation. Int. Microbiol. 2020, 23, 149–159. [Google Scholar] [CrossRef]
- Selvaraj, V.; Karthika, T.S.; Mansiy, C.; Alagar, M. An over review on recently developed techniques, mechanisms and intermediate involved in the advanced azo dye degradation for industrial applications. J. Mol.Struct. 2021, 1224, 129195. [Google Scholar] [CrossRef]
- Fatima, M.; Farooq, R.; Lindstrom, R.W.; Saeed, M. A review on biocatalytic decomposition of azo dyes and electrons recovery. J. Mol. Liq. 2017, 246, 275–281. [Google Scholar] [CrossRef]
- Chung, K.T. Azo dyes and human health: A review. J. Environ. Sci. Health C 2016, 4, 233–261. [Google Scholar] [CrossRef] [PubMed]
- Pirkarami, A.; Olya, M.E.; Limaee, N.Y. Decolorization of azo dyes by photo electro adsorption process using polyaniline coated electrode. Prog. Org. Coat. 2013, 76, 682–688. [Google Scholar] [CrossRef]
- Yaseen, D.A.; Scholz, M. Textile dye wastewater characteristics and constituents of synthetic effluents: A critical review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef]
- Dahiya, D.; Nigam, P.S. Waste Management by Biological Approach Employing Natural Substrates and Microbial Agents for the Remediation of Dyes’ Wastewater. Appl. Sci. 2020, 10, 2958. [Google Scholar] [CrossRef]
- Donkadokula, N.Y.; Kola, A.K.; Naz, I.; Saroj, D. A review on advanced physico-chemical and biological textile dye wastewater treatment techniques. Rev. Environ. Sci. Biotechnol. 2020, 19, 543–560. [Google Scholar] [CrossRef]
- Li, C.; Zhang, M.H.; Song, C.W.; Tao, P.; Sun, M.H.; Shao, M.H.; Wang, T.H. Enhanced Treatment Ability of Membrane Technology by Integrating an Electric Field for Dye Wastewater Treatment: A Review. J. Aoac. Int. 2018, 101, 1341–1352. [Google Scholar] [CrossRef] [PubMed]
- Katheresan, V.; Kanseto, J.; Lau, S.Y. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Perez, A.; Poznyak, T.; Chairez, I.; Guzman-Zavaleta, Z.J.; Alfaro-Ponce, M. Influence of Sodium Sulfate on the Direct Red 28 Degradation by Ozone in a Wastewater Recycling Process: A Stoichiometric and Novel Image Analysis. Mater. Lett. 2019, 42, 428–438. [Google Scholar] [CrossRef]
- Liu, Y.; Li, C.L.; Bao, J.; Wang, X.; Yu, W.J.; Shao, L.X. Degradation of Azo Dyes with Different Functional Groups in Simulated Wastewater by Electrocoagulation. Water 2022, 14, 123. [Google Scholar] [CrossRef]
- Shanker, U.; Rani, M.; Jassal, V. Degradation of hazardous organic dyes in water by nanomaterials. Environ. Chem. Lett. 2017, 15, 623–642. [Google Scholar] [CrossRef]
- Jiang, B.; Zheng, J.T.; Qiu, S.; Wu, M.B.; Zhang, Q.H.; Yan, Z.F.; Xue, Q.Z. Review on electrical discharge plasma technology for wastewater remediation. J. Colloid Interface Sci. 2014, 236, 348–368. [Google Scholar] [CrossRef]
- Fan, J.W.; Wu, H.X.; Liu, R.Y.; Meng, L.Y.; Sun, Y.J. Review on the treatment of organic wastewater by discharge plasma combined with oxidants and catalysts. Environ. Sci. Pollut. R. 2021, 28, 2522–2548. [Google Scholar] [CrossRef] [PubMed]
- Shang, K.F.; Li, J.; Morent, R. Hybrid electric discharge plasma technologies for water decontamination: A short review. Plasma. Sci. Technol. 2019, 21, 043001. [Google Scholar] [CrossRef]
- Cui, M.H.; Liu, W.Z.; Cui, D. Recent advancements in azo dye decolorization in bio-electrochemical systems (BESs): Insights into decolorization mechanism and practical application. Water Res. 2021, 203, 117512. [Google Scholar] [CrossRef] [PubMed]
- Goncharuk, W.; Klishchenko, R.E.; Kornienko, I.V. Destruction of GT Azo Active Orange Dye in the Flow-Through Plasma-Chemical Reactor. J. Water Chem. Technol. 2018, 40, 185–189. [Google Scholar] [CrossRef]
- Sen, S.K.; Raut, S.; Bandyopadhyay, P.; Raut, S. Fungal decolouration and degradation of azo dyes: A review. Fungal. Biol. Rev. 2016, 30, 112–133. [Google Scholar] [CrossRef]
- Topolovec, B.; Skoro, N.; Puac, N.; Petrovic, M. Pathways of organic micropollutants degradation in atmospheric pressure plasma processing -A review. Chemosphere 2022, 294, 133606. [Google Scholar] [CrossRef]
- Russo, M.; Lervolino, G.; Vaiano, V.; Palma, V. Non-Thermal Plasma Coupled with Catalyst for the Degradation of Water Pollutants: A Review. Catalysts 2020, 10, 1438. [Google Scholar] [CrossRef]
- Magureanu, M.; Bradu, C.; Parvulescu, V.I. Plasma processes for the treatment of water contaminated with harmful organic compounds. J. Phys. D Appl. Phys. 2018, 51, 313002. [Google Scholar] [CrossRef]
- Ma, S.; Lee, S.; Kim, K.; Im, J.; Jeon, H. Purification of organic pollutants in cationic thiazine and azo dye solutions using plasma-based advanced oxidation process via submerged multi-hole dielectric barrier discharge. Sep. Purif. Technol. 2021, 255, 117715. [Google Scholar] [CrossRef]
- Sarangapani, C.; Dixit, Y.; Milosavljevic, V.; Bourke, P.; Sullivan, C.; Cullen, P.J. Optimization of atmospheric air plasma for degradation of organic dyes in wastewater. Water Sci. Technol. 2017, 75, 207–219. [Google Scholar] [CrossRef]
- Wu, L.H.; Xie, Q.L.; Lv, Y.B.; Wu, Z.Y.; Liang, X.J.; Lu, M.Z.; Nie, Y. Degradation of Methylene Blue via Dielectric Barrier Discharge Plasma Treatment. Water 2019, 11, 1818. [Google Scholar] [CrossRef]
- Koppenol, W.H. Names for inorganic radicals (IUPAC recommendations 2000). Pure. Appl. Chem. 2000, 72, 437–446. [Google Scholar] [CrossRef][Green Version]
- Essiptchouk, A.; Petraconi, G.; Miranda, F.; Saraiva, A.C.V.; Charakhovski, L. Glycerine degradation by submerged plasma. J. Phys. D Appl. Phys. 2019, 52, 465201. [Google Scholar] [CrossRef]
- Shang, K.F.; Li, W.F.; Wang, X.J.; Lu, N.; Jiang, N.; Li, J. Degradation of p-nitrophenol by DBD plasma/Fe2+/persulfate oxidation process. Sep. Purif. Technol. 2019, 218, 106–112. [Google Scholar] [CrossRef]
- Chen, W.M.; Gu, Z.P.; Guo, S.P.; Li, Q.B. Microwave-assisted Fe-0-activated persulfate process for treating explosives in production wastewater. Chem. Eng. J. 2020, 391, 123497. [Google Scholar] [CrossRef]
- Peng, J.B.; Wang, Z.X.; Wang, S.Y.; Liu, J.; Zhang, Y.Z.; Wang, B.J.; Gong, Z.M.; Wang, M.J.; Dong, H.; Shi, J.L. Enhanced removal of methylparaben mediated by cobalt/carbon nanotubes (Co/CNTs) activated peroxymonosulfate in chloride-containing water: Reaction kinetics, mechanisms and pathways. Chem. Eng. J. 2021, 409, 128176. [Google Scholar] [CrossRef]
- Qi, C.D.; Wen, Y.N.; Zhao, Y.J.; Dai, Y.H.; Li, Y.P.; Xu, C.M.; Yang, S.G.; He, H. Enhanced degradation of organic contaminants by Fe(III)/peroxymonosulfate process with L-cysteine. Chin. Chem. Lett. 2022, 33, 2125–2128. [Google Scholar] [CrossRef]
- Stratton, G.R.; Bellona, C.L.; Dai, F.; Holsen, T.M.; Thagard, S.M. Plasma-based water treatment: Conception and application of a new general principle for reactor design. Chem. Eng. J. 2015, 273, 543–550. [Google Scholar] [CrossRef]
- Oke, S.R.; Ige, O.O.; Falodun, O.E.; Okoro, A.M.; Mphahlele, M.R.; Olubambi, P.A. Powder metallurgy of stainless steels and composites: A review of mechanical alloying and spark plasma sintering. Int. J. Adv. Manuf. Tech. 2019, 102, 3271–3290. [Google Scholar] [CrossRef]
- Zhang, C.X.; Sun, Y.B.; Yu, Z.Q.; Zhang, G.Y.; Feng, J.W. Simultaneous removal of Cr(VI) and acid orange 7 from water solution by dielectric barrier discharge plasma. Chemosphere 2018, 191, 527–536. [Google Scholar] [CrossRef]
- Zhang, C.; Qiu, J.T.; Kong, F.; Hou, X.M.; Fang, Z.; Yin, Y.; Shao, T. Plasma surface treatment of Cu by nanosecond-pulse diffuse discharges in atmospheric air. Plasma. Sci. Technol. 2018, 20, 014001. [Google Scholar] [CrossRef]
- Sun, B.; Aye, N.N.; Gao, Z.Y.; Lv, D.; Zhu, X.M.; Sato, M. Characteristics of gas-liquid pulsed discharge plasma reactor and dye decoloration efficiency. J. Environ. Sci. 2012, 24, 840–845. [Google Scholar] [CrossRef]
- Kakhki, R.M.; Rahni, S.Y.; Karimian, A. Removal of Methyl Orange from aqueous solutions by a novel, high efficient and low cost copper-modified nanoalum. Inorg. Nano-Met. Chem. 2021, 51, 1291–1296. [Google Scholar]
- Ko, S.J.; Yamaguchi, T.; Salles, F.; Oh, J.M. Systematic utilization of layered double hydroxide nanosheets for effective removal of Methyl Orange from an aqueous system by pi-pi stacking-induced nanoconfinement. J. Environ. Manag. 2021, 277, 111455. [Google Scholar] [CrossRef]
- Bagheri, F.; Chaibakhsh, N. Efficient visible-light photocatalytic ozonation for dye degradation using Fe2O3/MoS2 nanocomposite. Sep. Sci. Technol. 2021, 56, 3022–3032. [Google Scholar] [CrossRef]
- Meiyazhagan, S.; Yugeswaran, S.; Ananthapadmanabhan, P.V.; Suresh, K. Process and kinetics of dye degradation using microplasma and its feasibility in textile effluent detoxification. J. Water Process Eng. 2020, 37, 101519. [Google Scholar] [CrossRef]
- Hafeez, A.; Javed, F.; Fazal, T.; Shezaed, N.; Amjad, U.E.S.; Rehman, M.S.U. Intensification of ozone generation and degradation of azo dye in non-thermal hybrid corona-DBD plasma micro-reactor. Chem. Eng. Processing 2021, 159, 108205. [Google Scholar] [CrossRef]
- Takahashi, K.; Takeda, M.; Konno, R.; Takaki, K.; Satta, N. Influence of Electric Parameters on Hydroxyl Radical Production by Positive Pulsed Discharge Inside of a Bubble in Water. IEEE Trans. Plasma Sci. 2019, 47, 1105–1113. [Google Scholar] [CrossRef]
- Guo, H.; Wang, Y.W.; Liao, L.N.; Li, Z.; Pan, S.J.; Puyang, C.D.; Su, Y.Y.; Zhang, Y.; Wang, T.C.; Ren, J.Y. Review on remediation of organic-contaminated soil by discharge plasma: Plasma types, impact factors, plasma-assisted catalysis, and indexes for remediation. Chem. Eng. J. 2022, 436, 135239. [Google Scholar] [CrossRef]
- Hu, X.Y.; Wang, B.W. Removal of pefloxacin from wastewater by dielectric barrier discharge plasma: Mechanism and degradation pathways. J. Environ. Chem. Eng. 2021, 9, 105720. [Google Scholar] [CrossRef]
- Lee, H.D.; Kim, J.O.; Chung, J.W. Degradation of Methyl Orange by pulsed corona discharge process in water. Desalin. Water Treat. 2015, 53, 2767–2773. [Google Scholar] [CrossRef]
- Xiang, H.J.; Lei, B.; Yuan, X.C.; Lv, Q.A.; Zhang, Q. Design and Simulation of New Type Reactor in the Wastewater Treatment System Based on Discharge Plasma. IEEE Trans. Plasma Sci. 2019, 47, 952–957. [Google Scholar] [CrossRef]
- Jadaa, W.; Prakash, A.; Ray, A.K. Modeling of Degradation of Diazo Dye in Swirl-Flow Photocatalytic Reactor: Response Surface Approach. Catalysts 2020, 10, 1418. [Google Scholar] [CrossRef]
- Wang, J.K.; Yao, Z.P.; Wang, Y.J.; Xia, Q.X.; Chu, H.Y.; Jiang, Z.H. Preparation of immobilized coating Fenton-like catalyst for high efficient degradation of phenol. Environ. Pollut. 2017, 224, 552–558. [Google Scholar] [CrossRef]
- Liu, Y.; Qu, G.Z.; Sun, Q.H.; Jia, H.Z.; Wang, T.C.; Zhu, L.Y. Endogenously activated persulfate by non-thermal plasma for Cu(II)-EDTA decomplexation: Synergistic effect and mechanisms. Chem. Eng. J. 2021, 406, 126774. [Google Scholar] [CrossRef]
- Yang, D.Z.; Zhou, X.F.; Liang, J.P.; Xu, Q.N.; Wang, H.L.; Yang, K.; Wang, B.; Wang, W.C. Degradation of methylene blue in liquid using high-voltage pulsed discharge plasma synergizing iron-based catalyst-activated persulfate. J. Phys. D Appl. Phys. 2021, 54, 244002. [Google Scholar] [CrossRef]
- Li, H.; Song, R.Y.; Wang, Y.Y.; Zhong, R.W.; Zhang, Y.; Zhou, J.; Wang, T.C.; Zhu, L.Y. Simultaneous removal of antibiotic-resistant bacteria and its resistance genes in water by plasma oxidation: Highlights the effects of inorganic ions. Sep. Purif. Technol. 2022, 218, 119672. [Google Scholar] [CrossRef]
- Yuan, R.X.; Ramjaun, S.N.; Wang, Z.H.; Liu, J.S. Effects of chloride ion on degradation of Acid Orange 7 by sulfate radical-based advanced oxidation process: Implications for formation of chlorinated aromatic compounds. J. Hazard. Mater. 2011, 196, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Kamat, P.V.; Padmaja, S.; Au, V.; Madison, S.A. Free radical induced oxidation of the azo dye Acid Yellow 9. J. Chem. Soc. Perkin Trans. 2 1999, 6, 1219–1223. [Google Scholar] [CrossRef]
- Wang, B.W.; Xu, M.; Chi, C.M.; Chao, W.; Dajun, M.J. Degradation of Methyl Orange using dielectric barrier discharge water falling film reactor. J. Adv. Oxid. Technol. 2017, 20, 20170021. [Google Scholar] [CrossRef]




| Parameter | Horizontal Extent | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| Time (min) | 10 | 35 | 60 |
| Voltage (kV) | 11 | 13 | 15 |
| Discharge gap (mm) | 19 | 20 | 21 |
| Discharge needle numbers | 3 | 4 | 5 |
| The Experimental Number | Time (min) | Voltage (kV) | Discharge Gap (mm) | Discharge Needle Numbers | Removal Rate (%) |
|---|---|---|---|---|---|
| 1 | 60.00 | 13.00 | 20.00 | 3.00 | 71.52 |
| 2 | 10.00 | 13.00 | 21.00 | 4.00 | 9.54 |
| 3 | 60.00 | 13.00 | 20.00 | 5.00 | 90.25 |
| 4 | 35.00 | 15.00 | 20.00 | 5.00 | 72.73 |
| 5 | 35.00 | 13.00 | 19.00 | 5.00 | 64.4 |
| 6 | 10.00 | 15.00 | 20.00 | 4.00 | 26.84 |
| 7 | 60.00 | 11.00 | 20.00 | 4.00 | 43.77 |
| 8 | 10.00 | 13.00 | 20.00 | 3.00 | 25.26 |
| 9 | 35.00 | 11.00 | 20.00 | 5.00 | 64.53 |
| 10 | 60.00 | 13.00 | 21.00 | 4.00 | 80.59 |
| 11 | 35.00 | 15.00 | 20.00 | 3.00 | 54.08 |
| 12 | 60.00 | 13.00 | 19.00 | 4.00 | 55.58 |
| 13 | 35.00 | 11.00 | 19.00 | 4.00 | 20.88 |
| 14 | 35.00 | 13.00 | 20.00 | 4.00 | 59.24 |
| 15 | 10.00 | 11.00 | 20.00 | 4.00 | 20.76 |
| 16 | 10.00 | 13.00 | 19.00 | 4.00 | 9.41 |
| 17 | 35.00 | 15.00 | 21.00 | 4.00 | 47.34 |
| 18 | 35.00 | 13.00 | 20.00 | 4.00 | 59.24 |
| 19 | 10.00 | 13.00 | 20.00 | 5.00 | 25.49 |
| 20 | 35.00 | 13.00 | 19.00 | 3.00 | 42.35 |
| 21 | 35.00 | 13.00 | 20.00 | 4.00 | 59.24 |
| 22 | 35.00 | 11.00 | 21.00 | 4.00 | 27.24 |
| 23 | 35.00 | 15.00 | 19.00 | 4.00 | 58.45 |
| 24 | 35.00 | 13.00 | 21.00 | 3.00 | 42.78 |
| 25 | 35.00 | 13.00 | 20.00 | 4.00 | 59.24 |
| 26 | 35.00 | 11.00 | 20.00 | 3.00 | 22.48 |
| 27 | 60.00 | 15.00 | 20.00 | 4.00 | 82.83 |
| 28 | 35.00 | 13.00 | 21.00 | 5.00 | 51.7 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 13,001.19 | 14 | 928.66 | 18.99 | <0.0001 | significant |
| A-Time | 7866.37 | 1 | 7866.37 | 160.89 | <0.0001 | |
| B-Voltage | 1694.80 | 1 | 1694.80 | 34.66 | <0.0001 | |
| C-discharge gap | 5.49 | 1 | 5.49 | 0.11 | 0.7424 | |
| D-discharge needle numbers | 1019.92 | 1 | 1019.92 | 20.86 | 0.0004 | |
| AB | 271.92 | 1 | 271.92 | 5.56 | 0.0334 | |
| AC | 154.75 | 1 | 154.75 | 3.17 | 0.0969 | |
| AD | 85.56 | 1 | 85.56 | 1.75 | 0.2071 | |
| BC | 76.30 | 1 | 76.30 | 1.56 | 0.2321 | |
| BD | 136.89 | 1 | 136.89 | 2.80 | 0.1165 | |
| CD | 43.10 | 1 | 43.10 | 0.88 | 0.3637 | |
| A2 | 443.32 | 1 | 443.32 | 8.86 | 0.0100 | |
| B2 | 432.13 | 1 | 432.13 | 8.84 | 0.0101 | |
| C2 | 952.96 | 1 | 952.96 | 19.49 | 0.0006 | |
| D2 | 41.94 | 1 | 41.94 | 0.86 | 0.3700 | |
| Residual | 684.49 | 14 | 48.89 | |||
| Lack of Fit | 684.49 | 10 | 68.45 | |||
| Pure Error | 0.000 | 4 | 0.000 | |||
| Cor Total | 13,685.68 | 28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Song, J.-W.; Bao, J.; Shen, X.-J.; Li, C.-L.; Wang, X.; Shao, L.-X. Optimized Removal of Azo Dyes from Simulated Wastewater through Advanced Plasma Technique with Novel Reactor. Water 2022, 14, 3152. https://doi.org/10.3390/w14193152
Liu Y, Song J-W, Bao J, Shen X-J, Li C-L, Wang X, Shao L-X. Optimized Removal of Azo Dyes from Simulated Wastewater through Advanced Plasma Technique with Novel Reactor. Water. 2022; 14(19):3152. https://doi.org/10.3390/w14193152
Chicago/Turabian StyleLiu, Yang, Jia-Wei Song, Jia Bao, Xin-Jun Shen, Cheng-Long Li, Xin Wang, and Li-Xin Shao. 2022. "Optimized Removal of Azo Dyes from Simulated Wastewater through Advanced Plasma Technique with Novel Reactor" Water 14, no. 19: 3152. https://doi.org/10.3390/w14193152
APA StyleLiu, Y., Song, J.-W., Bao, J., Shen, X.-J., Li, C.-L., Wang, X., & Shao, L.-X. (2022). Optimized Removal of Azo Dyes from Simulated Wastewater through Advanced Plasma Technique with Novel Reactor. Water, 14(19), 3152. https://doi.org/10.3390/w14193152









